Nanoporous Silica-Based Protocells at Multiple Scales for Designs of Life and Nanomedicine
Abstract
:1. Introduction
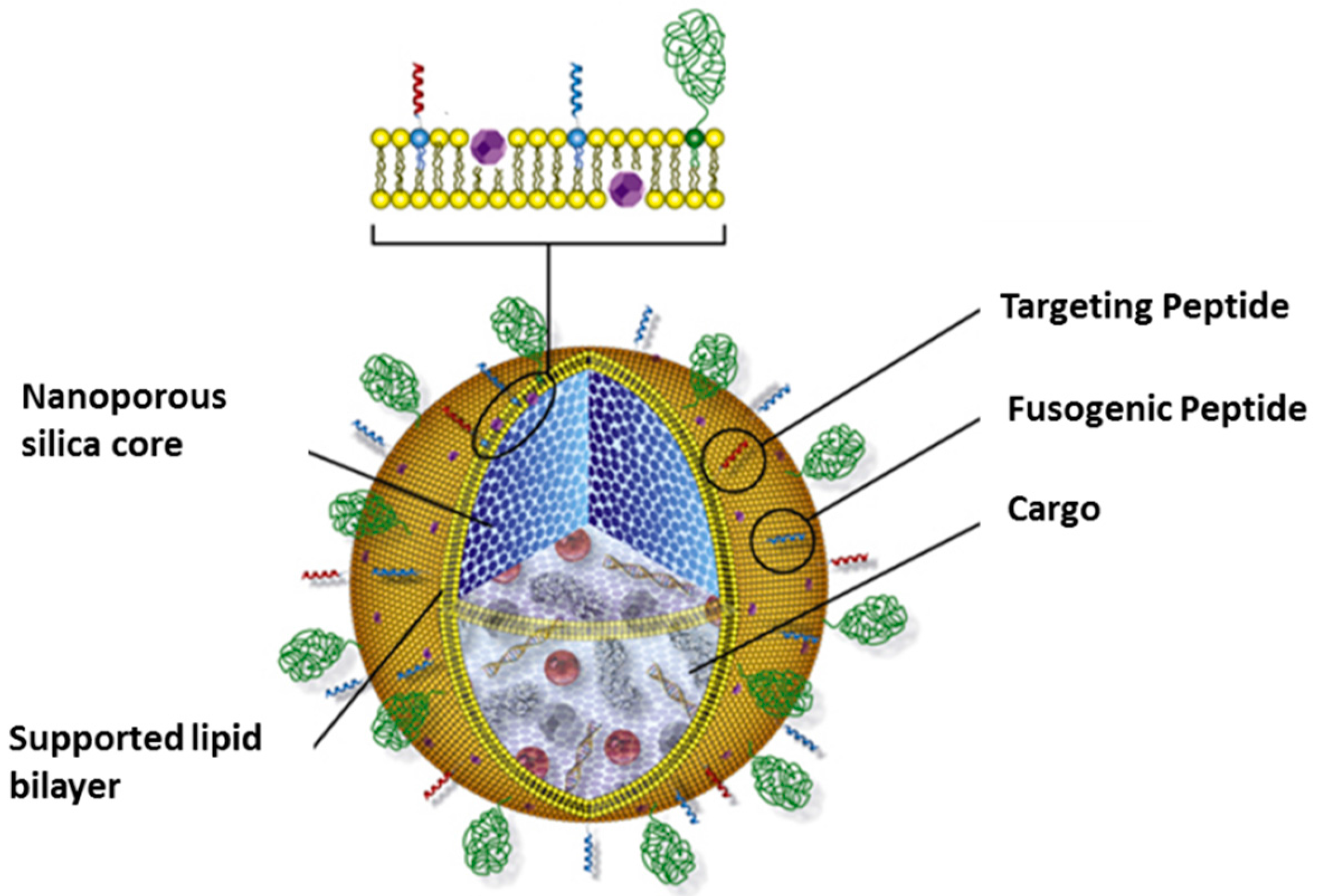
2. Design, Synthesis and Advantages
3. Nanoporous Silica-Based Protocells
3.1. Reconstitution of Membrane Proteins in the Lipid Bilayer of Protocells
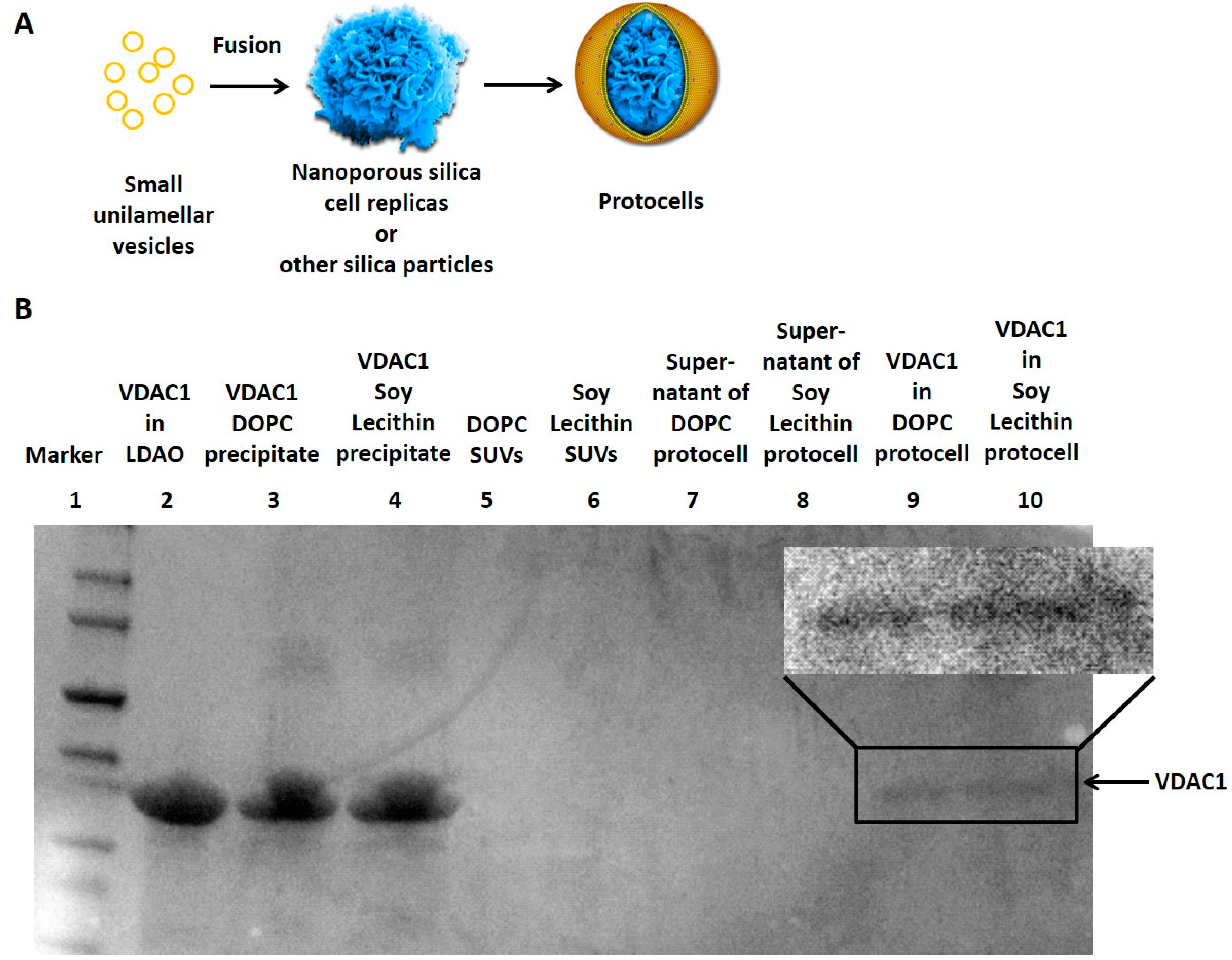
3.2. Reconstitution of Biochemical Reactions inside Porous Silica Core of Protocells
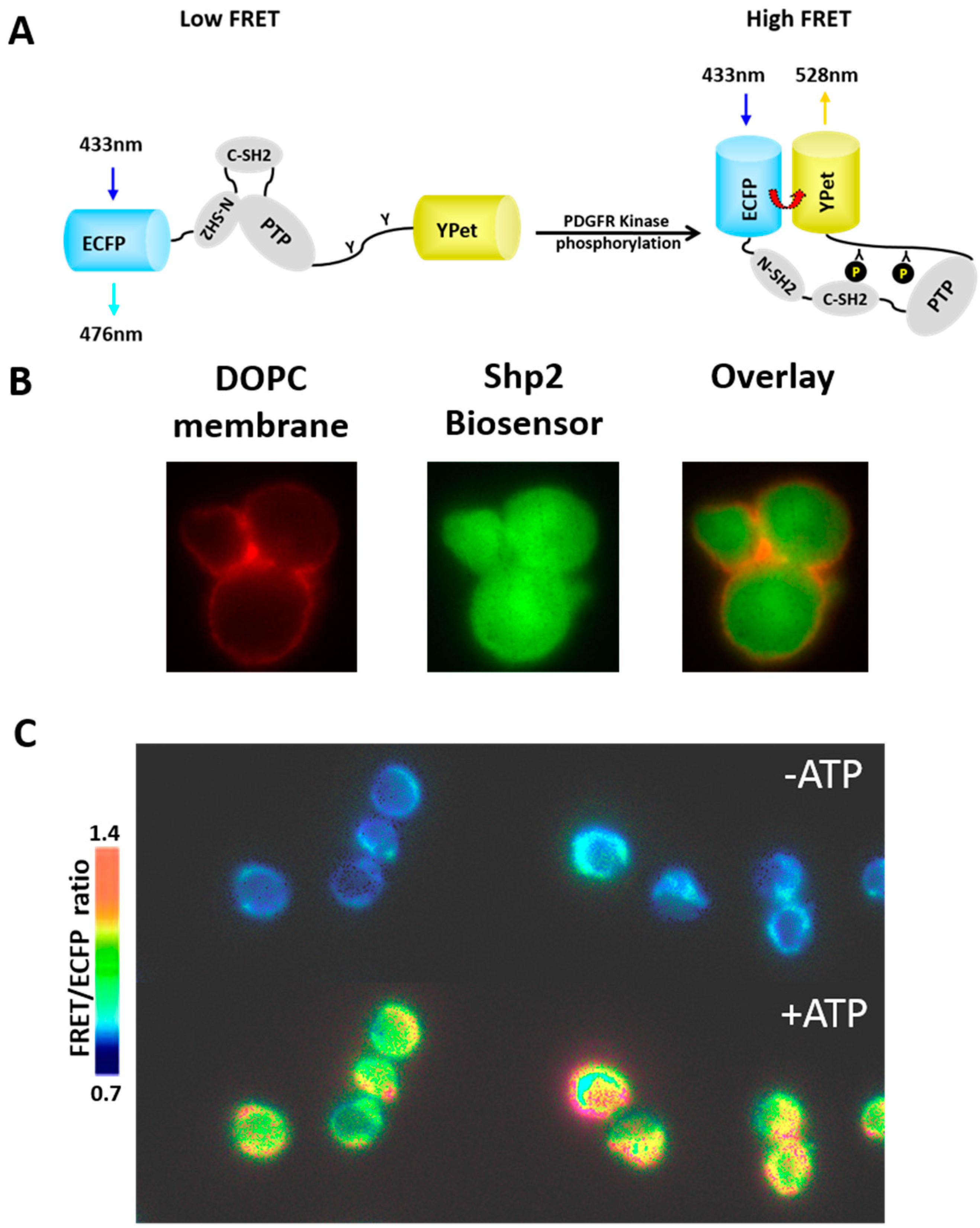
3.3. Protocells as Nanocarriers for Targeted Drug Delivery
4. Conclusions and Future Prospects
Acknowledgments
Conflicts of Interest
References
- Endy, D. Foundations for engineering biology. Nature 2005, 438, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.G.; Glass, J.I.; Lartigue, C.; Noskov, V.N.; Chuang, R.-Y.; Algire, M.A.; Benders, G.A.; Montague, M.G.; Ma, L.; Moodie, M.M.; et al. Creation of a bacterial cell controlled by a chemically synthesized genome. Science 2010, 329, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Stace-Naughton, A.; Jiang, X.; Brinker, C.J. Porous nanoparticle supported lipid bilayers (protocells) as delivery vehicles. J. Am. Chem. Soc. 2009, 131, 1354–1355. [Google Scholar] [CrossRef] [PubMed]
- Ashley, C.E.; Carnes, E.C.; Phillips, G.K.; Padilla, D.; Durfee, P.N.; Brown, P.A.; Hanna, T.N.; Liu, J.; Phillips, B.; Carter, M.B.; et al. The targeted delivery of multicomponent cargos to cancer cells by nanoporous particle-supported lipid bilayers. Nat. Mater. 2011, 10, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Oberholzer, T.; Luisi, P.L. The use of liposomes for constructing cell models. J. Biol. Phys. 2002, 28, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Adamala, K.; Luisi, P.L. Experimental systems to explore life origin: perspectives for understanding primitive mechanisms of cell division. Results Probl. Cell Differ. 2012, 53, 1–9. [Google Scholar]
- Szostak, J.W.; Bartel, D.P.; Luisi, P.L. Synthesizing life. Nature 2001, 409, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Rigaud, J.L.; Levy, D. Reconstitution of membrane proteins into liposomes. Methods Enzymol. 2003, 372, 65–86. [Google Scholar] [PubMed]
- Oberholzer, T.; Albrizio, M.; Luisi, P.L. Polymerase chain reaction in liposomes. Chem. Biol. 1995, 2, 677–682. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.K.; Yu, X.; Patil, A.J.; Li, M.; Mann, S. Cytoskeletal-like Supramolecular Assembly and Nanoparticle-Based Motors in a Model Protocell. Angew. Chem. Int. Ed. 2011, 50, 9343–9347. [Google Scholar] [CrossRef]
- Stano, P.; Carrara, P.; Kuruma, Y.; Pereira de Souza, T.; Luisi, P.L. Compartmentalized reactions as a case of soft-matter biotechnology: Synthesis of proteins and nucleic acids inside lipid vesicles. J. Mater. Chem. 2011, 21, 18887–18902. [Google Scholar] [CrossRef]
- Kita, H.; Matsuura, T.; Sunami, T.; Hosoda, K.; Ichihashi, N.; Tsukada, K.; Urabe, I.; Yomo, T. Replication of Genetic Information with Self-Encoded Replicase in Liposomes. Chembiochem 2008, 9, 2403–2410. [Google Scholar] [CrossRef] [PubMed]
- Hammer, D.A.; Kamat, N.P. Towards an artificial cell. FEBS Lett. 2013, 586, 2882–2890. [Google Scholar] [CrossRef]
- Huang, X.; Li, M.; Green, D.C.; Williams, D.S.; Patil, A.J.; Mann, S. Interfacial assembly of protein-polymer nano-conjugates into stimulus-responsive biomimetic protocells. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef]
- Huang, X.; Patil, A.J.; Li, M.; Mann, S. Design and Construction of Higher-Order Structure and Function in Proteinosome-Based Protocells. J. Am. Chem. Soc. 2014, 136, 9225–9234. [Google Scholar] [CrossRef] [PubMed]
- Martino, C.; Kim, S.H.; Horsfall, L.; Abbaspourrad, A.; Rosser, S.J.; Cooper, J.; Weitz, D.A. Protein expression, aggregation, and triggered release from polymersomes as artificial cell-like structures. Angew. Chem. Int. Ed. 2012, 51, 6416–6420. [Google Scholar] [CrossRef]
- Li, M.; Huang, X.; Tang, T.-Y.D.; Mann, S. Synthetic cellularity based on non-lipid micro-compartments and protocell models. Curr. Opin. Chem. Biol. 2014, 22, 1–11. [Google Scholar] [CrossRef]
- Kumar, R.K.; Li, M.; Olof, S.N.; Patil, A.J.; Mann, S. Artificial Cytoskeletal Structures Within Enzymatically Active Bio-inorganic Protocells. Small 2013, 9, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Harbron, R.L.; Weaver, J.V.M.; Binks, B.P.; Mann, S. Electrostatically gated membrane permeability in inorganic protocells. Nat. Chem. 2013, 5, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Markstrom, M.; Gunnarsson, A.; Orwar, O.; Jesorka, A. Dynamic microcompartmentalization of giant unilamellar vesicles by sol-gel transition and temperature induced shrinking/swelling of poly(N-isopropyl acrylamide). Soft Matter. 2007, 3, 587–595. [Google Scholar] [CrossRef]
- Tang, T.-Y.D.; Hak, C.R.C.; Thompson, A.J.; Kuimova, M.K.; Williams, D.S.; Perriman, A.W.; Mann, S. Fatty acid membrane assembly on coacervate microdroplets as a step towards a hybrid protocell model. Nat. Chem. 2014, 6, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Kaehr, B.; Townson, J.L.; Kalinich, R.M.; Awad, Y.H.; Swartzentruber, B.S.; Dunphy, D.R.; Brinker, C.J. Cellular complexity captured in durable silica biocomposites. Proc. Natl. Acad. Sci. USA 2012, 109, 17336–17341. [Google Scholar] [CrossRef] [PubMed]
- Pontani, L.L.; van der Gucht, J.; Salbreux, G.; Heuvingh, J.; Joanny, J.F.; Sykes, C. Reconstitution of an actin cortex inside a liposome. Biophys. J. 2009, 96, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Takiguchi, K.; Yamada, A.; Negishi, M.; Tanaka-Takiguchi, Y.; Yoshikawa, K. Entrapping desired amounts of actin filaments and molecular motor proteins in giant liposomes. Langmuir 2008, 24, 11323–11326. [Google Scholar] [CrossRef] [PubMed]
- Nomura, S.M.; Tsumoto, K.; Hamada, T.; Akiyoshi, K.; Nakatani, Y.; Yoshikawa, K. Gene expression within cell-sized lipid vesicles. Chembiochem 2003, 4, 1172–1175. [Google Scholar] [CrossRef] [PubMed]
- Stachowiak, J.C.; Richmond, D.L.; Li, T.H.; Li, A.P.; Parekh, S.H.; Fletcher, D.A. Unilamellar vesicle formation and encapsulation by microfluidic jetting. Proc. Natl. Acad. Sci. USA 2008, 105, 4697–4702. [Google Scholar] [CrossRef] [PubMed]
- Ellis, R.J. Macromolecular crowding: Obvious but underappreciated. Trends Biochem. Sci. 2001, 26, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Ellis, R.J.; Minton, A.P. Cell biology: Join the crowd. Nature 2003, 425, 27–28. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.X.; Rivas, G.; Minton, A.P. Macromolecular crowding and confinement: Biochemical, biophysical, and potential physiological consequences. Annu. Rev. Biophys. 2008, 37, 375–397. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, R.B.; Brinker, C.J. Aqueous sol-gel process for protein encapsulation. Chem. Mater. 2000, 12, 2434–2441. [Google Scholar] [CrossRef]
- Brinker, C.J. Hydrolysis and Condensation of Silicates—Effects on Structure. J. Non-Cryst. Solids 1988, 100, 31–50. [Google Scholar] [CrossRef]
- Natarajan, S.K.; Selvaraj, S. Mesoporous silica nanoparticles: Importance of surface modifications and its role in drug delivery. RSC Adv. 2014, 4, 14328–14334. [Google Scholar] [CrossRef]
- Lu, Y.; Fan, H.; Stump, A.; Ward, T.L.; Rieker, T.; Brinker, C.J. Aerosol-assisted self-assembly of mesostructured spherical nanoparticles. Nature 1999, 398, 223–226. [Google Scholar] [CrossRef]
- Brinker, C.J.; Lu, Y.; Sellinger, A.; Fan, H. Evaporation-induced self-assembly: Nanostructures made easy. Adv. Mater. 1999, 11, 579–585. [Google Scholar] [CrossRef]
- Davis, R.W.; Flores, A.; Barrick, T.A.; Cox, J.M.; Brozik, S.M.; Lopez, G.P.; Brozik, J.A. Nanoporous microbead supported bilayers: Stability, physical characterization, and incorporation of functional transmembrane proteins. Langmuir 2007, 23, 3864–3872. [Google Scholar] [CrossRef] [PubMed]
- Sackmann, E. Supported membranes: Scientific and practical applications. Science 1996, 271, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Troutier, A.L.; Ladaviere, C. An overview of lipid membrane supported by colloidal particles. Adv. Colloid Interface Sci. 2007, 133, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Koenig, B.W.; Krueger, S.; Orts, W.J.; Majkrzak, C.F.; Berk, N.F.; Silverton, J.V.; Gawrisch, K. Neutron Reflectivity and Atomic Force Microscopy Studies of a Lipid Bilayer in Water Adsorbed to the Surface of a Silicon Single Crystal. Langmuir 1996, 12, 1343–1350. [Google Scholar] [CrossRef]
- Johnson, S.J.; Bayerl, T.M.; Weihan, W.; Noack, H.; Penfold, J.; Thomas, R.K.; Kanellas, D.; Rennie, A.R.; Sackmann, E. Coupling of spectrin and polylysine to phospholipid monolayers studied by specular reflection of neutrons. Biophys. J. 1991, 60, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Varma, S.; Teng, M.; Scott, H.L. Nonintercalating nanosubstrates create asymmetry between bilayer leaflets. Langmuir 2012, 28, 2842–2848. [Google Scholar] [CrossRef] [PubMed]
- Nordlund, G.; Ng, J.B.S.; Bergström, L.; Brzezinski, P. A membrane-reconstituted multisubunit functional proton pump on mesoporous silica particles. ACS Nano 2009, 3, 2639–2646. [Google Scholar] [CrossRef] [PubMed]
- Oliynyk, V.; Mille, C.; Ng, J.B.S.; von Ballmoos, C.; Corkery, R.W.; Bergström, L. Selective and ATP-driven transport of ions across supported membranes into nanoporous carriers using gramicidin A and ATP synthase. Phys. Chem. Chem. Phys. 2013, 15, 2733–2740. [Google Scholar] [CrossRef] [PubMed]
- Stano, P.; Pereira de Souza, T.; Carrara, P.; Altamura, E.; D’Aguanno, E.; Caputo, M.; Luisi, P.L.; Mavelli, F. Recent Biophysical Issues about the Preparation of Solute-Filled Lipid Vesicles. Mech. Adv. Mater. Struct. 2014. [Google Scholar] [CrossRef]
- Noireaux, V.; Libchaber, A. A vesicle bioreactor as a step toward an artificial cell assembly. Proc. Natl. Acad. Sci. USA 2004, 101, 17669–17674. [Google Scholar] [CrossRef] [PubMed]
- Rostovtseva, T.; Colombini, M. VDAC channels mediate and gate the flow of ATP: Implications for the regulation of mitochondrial function. Biophys. J. 1997, 72, 1954–1962. [Google Scholar] [CrossRef] [PubMed]
- Hiller, S.; Garces, R.G.; Malia, T.J.; Orekhov, V.Y.; Colombini, M.; Wagner, G. Solution structure of the integral human membrane protein VDAC-1 in detergent micelles. Science 2008, 321, 1206–1210. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.Y.; Raschle, T.; Hiller, S.; Wagner, G. Solution NMR spectroscopic characterization of human VDAC-2 in detergent micelles and lipid bilayer nanodiscs. Biochim. Biophys. Acta 2012, 1818, 1562–1569. [Google Scholar] [CrossRef] [PubMed]
- Kuruma, Y.; Stano, P.; Ueda, T.; Luisi, P.L. A synthetic biology approach to the construction of membrane proteins in semi-synthetic minimal cells. Biochim. Biophys. Acta 2009, 1788, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, M. Ordered Mesoporous Materials for Bioadsorption and Biocatalysis. Chem. Mater. 2005, 17, 4577–4593. [Google Scholar] [CrossRef]
- Tallquist, M.; Kazlauskas, A. PDGF signaling in cells and mice. Cytokine Growth Factor Rev. 2004, 15, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Chan, G.; Kalaitzidis, D.; Neel, B.G. The tyrosine phosphatase Shp2 (PTPN11) in cancer. Cancer Metastasis Rev. 2008, 27, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Loh, M.L.; Vattikuti, S.; Schubbert, S.; Reynolds, M.G.; Carlson, E.; Lieuw, K.H.; Cheng, J.W.; Lee, C.M.; Stokoe, D.; Bonifas, J.M.; et al. Mutations in PTPN11 implicate the SHP-2 phosphatase in leukemogenesis. Blood 2004, 103, 2325–2331. [Google Scholar] [CrossRef] [PubMed]
- Tartaglia, M.; Gelb, B.D. Noonan syndrome and related disorders: Genetics and pathogenesis. Annu. Rev. Genomics Hum. Genet. 2005, 6, 45–68. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Lu, S.; Ouyang, M.; Lin, L.J.; Zhuo, Y.; Liu, B.; Chien, S.; Neel, B.G.; Wang, Y. Antagonism between binding site affinity and conformational dynamics tunes alternative cis-interactions within Shp2. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef]
- Kim, B.Y.; Rutka, J.T.; Chan, W.C. Nanomedicine. New Engl. J. Med. 2010, 363, 2434–2443. [Google Scholar] [CrossRef] [PubMed]
- Slingerland, M.; Guchelaar, H.J.; Gelderblom, H. Liposomal drug formulations in cancer therapy: 15 years along the road. Drug Discov. Today 2011, 17, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Song, W.; Wang, A.; Zhu, P.; Fei, J.; Li, J. Lipid coated mesoporous silica nanoparticles as photosensitive drug carriers. Phys. Chem. Chem. Phys. 2010, 12, 4418–4422. [Google Scholar] [CrossRef] [PubMed]
- Kendall, E.L.; Ngassam, V.N.; Gilmore, S.F.; Brinker, C.F.; Parikh, A.N. Lithographically defined macroscale modulation of lateral fluidity and phase separation realized via patterned nanoporous silica-supported phospholipid bilayers. J. Am. Chem. Soc. 2013, 135, 15718–15721. [Google Scholar] [CrossRef] [PubMed]
- Miele, E.; Spinelli, G.P.; Miele, E.; di Fabrizio, E.; Ferretti, E.; Tomao, S.; Gulino, A. Nanoparticle-based delivery of small interfering RNA: Challenges for cancer therapy. Int. J. Nanomed. 2012, 7, 3637–3657. [Google Scholar]
- Ashley, C.E.; Carnes, E.C.; Epler, K.E.; Padilla, D.P.; Phillips, G.K.; Castillo, R.E.; Wilkinson, D.C.; Wilkinson, B.S.; Burgard, C.A.; Kalinich, R.M.; et al. Delivery of small interfering RNA by peptide-targeted mesoporous silica nanoparticle-supported lipid bilayers. ACS Nano 2012, 6, 2174–2188. [Google Scholar] [CrossRef] [PubMed]
- Luisi, P.L. Toward the engineering of minimal living cells. Anat. Rec. 2002, 268, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Luisi, P.L.; Ferri, F.; Stano, P. Approaches to semi-synthetic minimal cells: A review. Naturwissenschaften 2006, 93, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ichihashi, N.; Matsuura, T.; Kita, H.; Sunami, T.; Suzuki, H.; Yomo, T. Constructing Partial Models of Cells. Cold Spring Harb. Persp. Biol. 2010, 2. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Jakobsson, E.; Wang, Y.; Brinker, C.J. Nanoporous Silica-Based Protocells at Multiple Scales for Designs of Life and Nanomedicine. Life 2015, 5, 214-229. https://doi.org/10.3390/life5010214
Sun J, Jakobsson E, Wang Y, Brinker CJ. Nanoporous Silica-Based Protocells at Multiple Scales for Designs of Life and Nanomedicine. Life. 2015; 5(1):214-229. https://doi.org/10.3390/life5010214
Chicago/Turabian StyleSun, Jie, Eric Jakobsson, Yingxiao Wang, and C. Jeffrey Brinker. 2015. "Nanoporous Silica-Based Protocells at Multiple Scales for Designs of Life and Nanomedicine" Life 5, no. 1: 214-229. https://doi.org/10.3390/life5010214
APA StyleSun, J., Jakobsson, E., Wang, Y., & Brinker, C. J. (2015). Nanoporous Silica-Based Protocells at Multiple Scales for Designs of Life and Nanomedicine. Life, 5(1), 214-229. https://doi.org/10.3390/life5010214




