The “Bone Block Technique”: Reconstruction of Bone Defects Caused by Osteomyelitis Using Corticocancellous Bone Blocks from the Iliac Crest and the Induced Membrane Technique
Abstract
1. Introduction
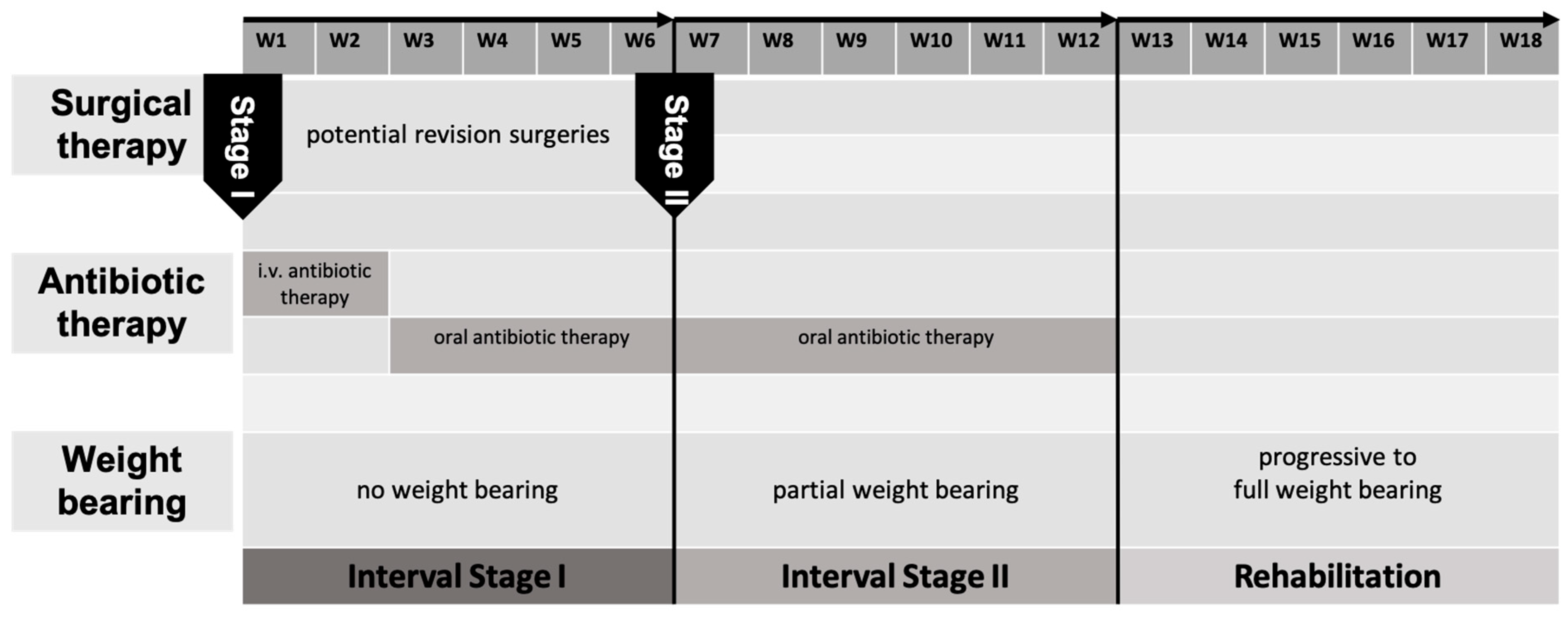
1.1. Surgical Technique
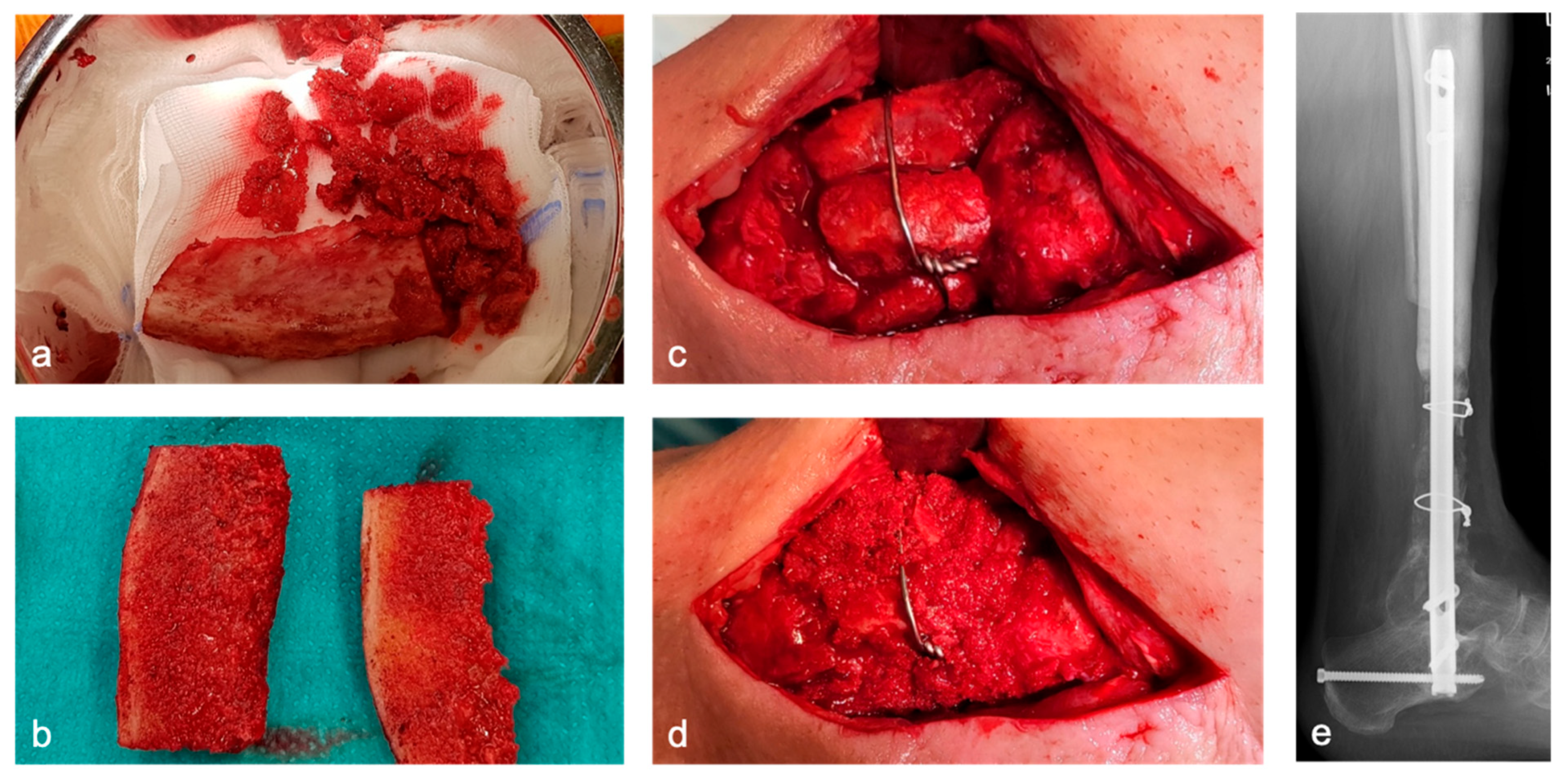
1.1.1. BBT Stage I
1.1.2. BBT Stage II
2. Materials and Methods
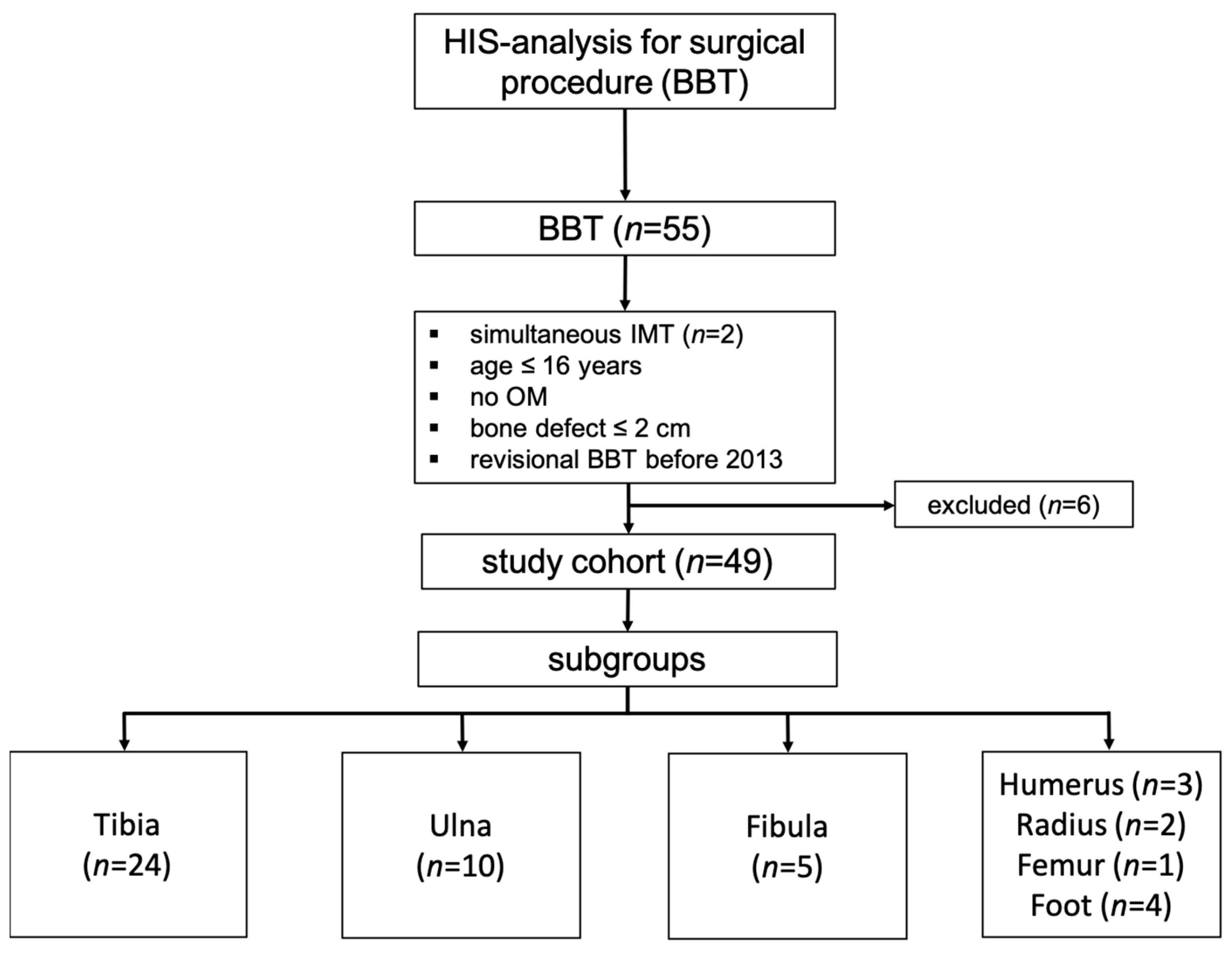
2.1. Study Design
2.2. Data Acquisition
2.3. Investigated Parameters
2.4. Statistical Analysis
3. Results
3.1. Study Cohort
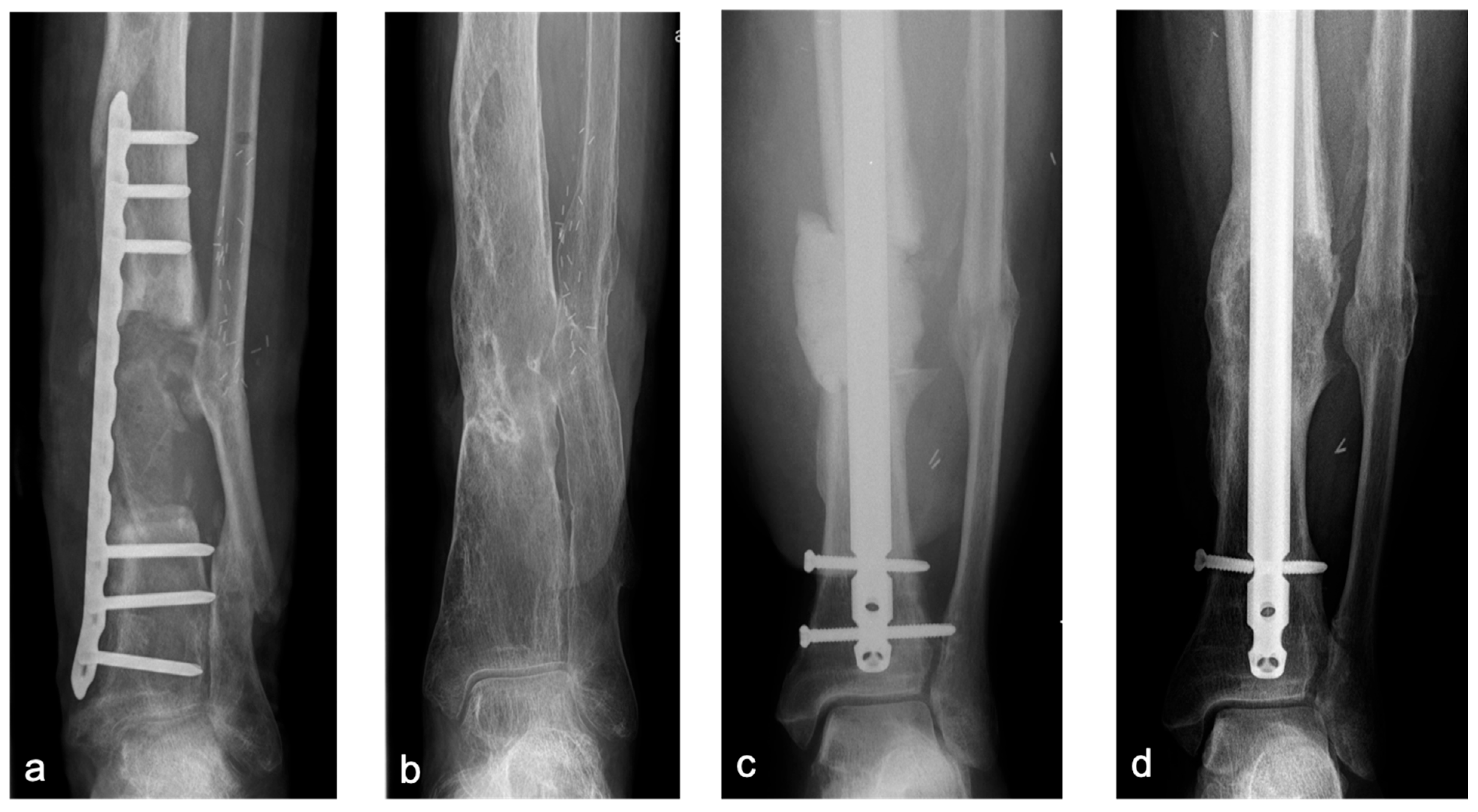
3.2. Surgical Data
3.3. Consolidation
3.4. Infectiology
3.5. Complications
4. Discussion
4.1. Fixation Strategies in Stage I and II
4.2. Grafting Technique and Bone Healing
4.3. Recurrent Infections
4.4. Complications
4.5. Limitations
4.6. Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BBT | Bone block technique |
| IMT | Induced membrane technique |
| MT | Masquelet technique |
| SSC | Surgical site complication |
| DO | Distraction osteogenesis |
| PMMA | Polymethylmethacrylate |
| CCBG | Corticocancellous bone graft |
| CB | Cancellous bone |
| HIS | Hospital information system |
| FU | Follow-up |
| SEM | Standard error of the mean |
| BMI | Body mass index |
| ASA | American Society of Anesthesiologists |
| cm | Centimeters |
References
- Gupta, G.; Majhee, A.; Rani, S.; Shekhar, S.; Prasad, P.; Chauhan, G. A comparative study between bone transport technique using Ilizarov/LRS fixator and induced membrane (Masquelet) technique in management of bone defects in the long bones of lower limb. J. Fam. Med. Prim. Care 2022, 11, 3660. [Google Scholar] [CrossRef] [PubMed]
- Wakefield, S.M.; Papakostidis, C.; Giannoudis, V.P.; Mandía-Martínez, A.; Giannoudis, P.V. Distraction osteogenesis versus induced membrane technique for infected tibial non-unions with segmental bone loss: A systematic review of the literature and meta-analysis of available studies. Eur. J. Trauma Emerg. Surg. 2023, 50, 705–721. [Google Scholar] [CrossRef]
- Xie, L.; Huang, Y.; Zhang, L.; Si, S.; Yu, Y. Ilizarov method and its combined methods in the treatment of long bone defects of the lower extremity: Systematic review and meta-analysis. BMC Musculoskelet. Disord. 2023, 24, 891. [Google Scholar] [CrossRef] [PubMed]
- Rohilla, R.; Sharma, P.K.; Wadhwani, J.; Das, J.; Singh, R.; Beniwal, D. Prospective randomized comparison of bone transport versus Masquelet technique in infected gap nonunion of tibia. Arch. Orthop. Trauma Surg. 2022, 142, 1923–1932. [Google Scholar] [CrossRef]
- Mathieu, L.; Durand, M.; Collombet, J.M.; de Rousiers, A.; de l’Escalopier, N.; Masquelet, A.C. Induced membrane technique: A critical literature analysis and proposal for a failure classification scheme. Eur. J. Trauma Emerg. Surg. 2021, 47, 1373–1380. [Google Scholar] [CrossRef]
- Masquelet, A.C. Induced Membrane Technique: Pearls and Pitfalls. J. Orthop. Trauma 2017, 31, S36–S38. [Google Scholar] [CrossRef] [PubMed]
- Giannoudis, P.V.; Faour, O.; Goff, T.; Kanakaris, N.; Dimitriou, R. Masquelet technique for the treatment of bone defects: Tips-tricks and future directions. Injury 2011, 42, 591–598. [Google Scholar] [CrossRef]
- Kanakaris, N.K.; Harwood, P.J.; Mujica-Mota, R.; Mohrir, G.; Chloros, G.; Giannoudis, P.V. Treatment of tibial bone defects: Pilot analysis of direct medical costs between distraction osteogenesis with an Ilizarov frame and the Masquelet technique. Eur. J. Trauma Emerg. Surg. 2023, 49, 951–964. [Google Scholar] [CrossRef]
- Masquelet, A.C.; Begue, T. The Concept of Induced Membrane for Reconstruction of Long Bone Defects. Orthop. Clin. N. Am. 2010, 41, 27–37. [Google Scholar] [CrossRef]
- Masquelet, A.C.; Fitoussi, F.; Begue, T.; Muller, G.P. Reconstruction of the long bones by the induced membrane and spongy autograft. Ann. Chir. Plast. Esthet. 2000, 45, 346–353. [Google Scholar]
- Morelli, I.; Drago, L.; George, D.A.; Gallazzi, E.; Scarponi, S.; Romanò, C.L. Masquelet technique: Myth or reality? A systematic review and meta-analysis. Injury 2016, 47, S68–S76. [Google Scholar] [CrossRef]
- Karger, C.; Kishi, T.; Schneider, L.; Fitoussi, F.; Masquelet, A.C. Treatment of posttraumatic bone defects by the induced membrane technique. Orthop. Traumatol. Surg. Res. 2012, 98, 97–102. [Google Scholar] [CrossRef]
- Fischer, C.; Schipper, S.; Langwald, S.; Klauke, F.; Kobbe, P.; Mendel, T.; Hückstädt, M. Modified Masquelet technique: Technique of the induced membrane in the course of time. Unfallchirurgie 2024, 127, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, L.; Murison, J.C.; de Rousiers, A.; de L’eScalopier, N.; Lutomski, D.; Collombet, J.-M.; Durand, M. The Masquelet technique: Can disposable polypropylene syringes be an alternative to standard PMMA spacers? A rat bone defect model. Clin. Orthop. Relat. Res. 2021, 479, 2737–2751. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, L.; Durand, M.; Demoures, T.; Steenman, C.; Masquelet, A.C.; Collombet, J.M. Repeated Induced-Membrane Technique Failure without Infection: A Series of Three Consecutive Procedures Performed for a Single Femur Defect. Case Rep. Orthop. 2020, 2020, 8892226. [Google Scholar] [CrossRef]
- Foster, A.L.; Moriarty, T.F.; Zalavras, C.; Morgenstern, M.; Jaiprakash, A.; Crawford, R.; Burch, M.-A.; Boot, W.; Tetsworth, K.; Miclau, T.; et al. The influence of biomechanical stability on bone healing and fracture-related infection: The legacy of Stephan Perren. Injury 2021, 52, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Beeharry, M.W.; Ahmad, B. Principles of Fracture Healing and Fixation: A Literature Review. Cureus 2024, 16, e76250. [Google Scholar] [CrossRef]
- Hast, M.; Glatt, V.; Archdeacon, M.; Ledet, E.; Lewis, G.; Ahn, J.; Haller, J. Biomechanics of fracture healing: How best to optimize your construct in the OR. OTA Int. 2024, 7, e304. [Google Scholar] [CrossRef]
- Elliott, D.S.; Newman, K.J.H.; Forward, D.P.; Hahn, D.M.; Ollivere, B.; Kojima, K.; Handley, R.; Rossiter, N.D.; Wixted, J.J.; Smith, R.M.; et al. A unified theory of bone healing and nonunion: BHN theory. Bone Jt. J. 2016, 98, 884–891. [Google Scholar] [CrossRef]
- Perren, S.M. Evolution of the internal fixation of long bone fractures the scientific basis of biological internal fixation: Choosing a new balance between stability and biology. J. Bone Jt. Surg. Br. 2002, 84, 1093–1110. [Google Scholar] [CrossRef]
- El-Alfy, B.S.; Ali, A.M. Management of segmental skeletal defects by the induced membrane technique. Indian J. Orthop. 2015, 49, 643–648. [Google Scholar] [CrossRef]
- Oestern, H.J.; Tscherne, H.; Sturm, J.; Nerlich, M. Classification of the severity of injury. Unfallchirurg 1985, 88, 465–472. [Google Scholar]
- Mi, M.; Papakostidis, C.; Wu, X.; Giannoudis, P.V. Mixed results with the Masquelet technique: A fact or a myth? Injury 2020, 51, 132–135. [Google Scholar] [CrossRef]
- Kawakami, R.; Konno, S.I.; Ejiri, S.; Hatashita, S. 141 Infected Bone Defects After Limb-Threatening Trauma Fukushima. 2015. Volume 61. Available online: https://www.jstage.jst.go.jp/browse/fms (accessed on 10 February 2025).
- Wang, X.; Luo, F.; Huang, K.; Xie, Z. Induced membrane technique for the treatment of bone defects due to post-traumatic osteomyelitis. Bone Jt. Res. 2016, 5, 101–105. [Google Scholar] [CrossRef]
- Apard, T.; Bigorre, N.; Cronier, P.; Duteille, F.; Bizot, P.; Massin, P. Two-stage reconstruction of post-traumatic segmental tibia bone loss with nailing. Orthop. Traumatol. Surg. Res. 2010, 96, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Zappaterra, T.; Ghislandi, X.; Adam, A.; Huard, S.; Gindraux, F.; Gallinet, D.; Lepage, D.; Garbuio, P.; Tropet, Y.; Obert, L. Reconstruction des pertes de substance osseuse du membre supérieur par la technique de la membrane induite, étude prospective à propos de neuf cas. Chir. Main 2011, 30, 255–263. [Google Scholar] [CrossRef]
- Andrzejowski, P.; Masquelet, A.; Giannoudis, P.V. Induced Membrane Technique (Masquelet) for Bone Defects in the Distal Tibia, Foot, and Ankle: Systematic Review, Case Presentations, Tips, and Techniques. Foot Ankle Clin. 2020, 25, 537–586. [Google Scholar] [CrossRef]
- Bourgeois, M.; Loisel, F.; Bertrand, D.; Nallet, J.; Gindraux, F.; Adam, A.; Lepage, D.; Sergent, P.; Leclerc, G.; Rondot, T.; et al. Management of forearm bone loss with induced membrane technique. Hand Surg. Rehabil. 2020, 39, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Klifto, C.S.; Gandi, S.D.; Sapienza, A. Bone Graft Options in Upper-Extremity Surgery. J. Hand Surg. 2018, 43, 755–761.e2. [Google Scholar] [CrossRef] [PubMed]
- Scholz, A.O.; Gehrmann, S.; Glombitza, M.; Kaufmann, R.A.; Bostelmann, R.; Flohe, S.; Windolf, J. Reconstruction of septic diaphyseal bone defects with the induced membrane technique. Injury 2015, 46, S121–S124. [Google Scholar] [CrossRef]
- Moghaddam, A.; Zietzschmann, S.; Bruckner, T.; Schmidmaier, G. Treatment of atrophic tibia non-unions according to “diamond concept”: Results of one- and two-step treatment. Injury 2015, 46, S39–S50. [Google Scholar] [CrossRef]
- Stafford, P.R.; Norris, B.L. Reamer-irrigator-aspirator bone graft and bi Masquelet technique for segmental bone defect nonunions: A review of 25 cases. Injury 2010, 41, S72–S77. [Google Scholar] [CrossRef]
- Findeisen, S.; Gräfe, N.; Schwilk, M.; Ferbert, T.; Helbig, L.; Haubruck, P.; Schmidmaier, G.; Tanner, M. Use of Autologous Bone Graft with Bioactive Glass as a Bone Substitute in the Treatment of Large-Sized Bone Defects of the Femur and Tibia. J. Pers. Med. 2023, 13, 1644. [Google Scholar] [CrossRef]
- Kaou, M.H.; Furkó, M.; Balázsi, K.; Balázsi, C. Advanced Bioactive Glasses: The Newest Achievements and Breakthroughs in the Area. Nanomaterials 2023, 13, 2287. [Google Scholar] [CrossRef]
- Dell’aquila, A.M.; dos Reis, G.N.B.; Cuba, G.T.; Targa, W.H.d.C.; Bongiovanni, J.C.; Durigon, T.S.; Salles, M.J.; dos Reis, F.B. Outcome and Predictors of Treatment Failure in Chronic Osteomyelitis Using Bioactive Glass Granules and Putty Formulations. Antibiotics 2023, 12, 1720. [Google Scholar] [CrossRef]
- Van Vugt, T.A.G.; Geurts, J.A.P.; Blokhuis, T.J. Treatment of infected tibial non-unions using a BMAC and S53P4 BAG combination for reconstruction of segmental bone defects: A clinical case series. Injury 2021, 52, S67–S71. [Google Scholar] [CrossRef] [PubMed]
- Van Gestel, N.A.P.; Geurts, J.; Hulsen, D.J.W.; Van Rietbergen, B.; Hofmann, S.; Arts, J.J. Clinical Applications of S53P4 Bioactive Glass in Bone Healing and Osteomyelitic Treatment: A Literature Review. Biomed. Res. Int. 2015, 2015, 684826. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Yang, Y.; Liu, B.; Li, X.; Tian, Y. Application of 3D-printed porous prosthesis for the reconstruction of infectious bone defect with concomitant severe soft tissue lesion: A case series of 13 cases. BMC Musculoskelet. Disord. 2024, 25, 1090. [Google Scholar] [CrossRef]
- Li, Z.; Lu, M.; Zhang, Y.; Wang, J.; Wang, Y.; Gong, T.; He, X.; Luo, Y.; Zhou, Y.; Min, L.; et al. 3D-Printed Personalized Lattice Implant as an Innovative Strategy to Reconstruct Geographic Defects in Load-Bearing Bones. Orthop. Surg. 2024, 16, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Xing, Y.; Li, X.; Liu, B.; Liu, N.; Huo, Y.; Tian, Y. 3D-printed titanium porous prosthesis combined with the Masquelet technique for the management of large femoral bone defect caused by osteomyelitis. BMC Musculoskelet. Disord. 2024, 25, 474. [Google Scholar] [CrossRef]
- Liu, B.; Tan, Q.; Wang, Z.; Hou, G.; Wang, C.; Tian, Y. Applying 3D-Printed Porous Ti6Al4V Prostheses to Repair Osteomyelitis-Induced Partial Bone Defects of Lower Limbs: Finite Element Analysis and Clinical Outcomes. Orthop. Surg. 2025, 17, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Chen, C.; Zhang, B.; Yao, C.; Zhang, Y. Advances in 3D-printed scaffold technologies for bone defect repair: Materials, biomechanics, and clinical prospects. Biomed. Eng. Online 2025, 24, 51. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.; Langwald, S.; Klauke, F.; Kobbe, P.; Mendel, T.; Hückstädt, M. Introducing the Pearl-String Technique: A New Concept in the Treatment of Large Bone Defects. Life 2025, 15, 414. [Google Scholar] [CrossRef]
- Hückstädt, M.; Fischer, C.; Mitin, M.; Klauke, F.; Langwald, S.; Mendel, T.; Kobbe, P.; Schipper, S. Modified Masquelet technique for reconstruction of critical bone defects. Unfallchirurgie 2024, 127, 738–742. [Google Scholar] [CrossRef] [PubMed]

| Gender | Age [years] | BMI [KG/m2] | Smoking | Diabetes | ASA Classification | Initial Open Fractures |
|---|---|---|---|---|---|---|
| male: 35 (71.4%) female:14 (28.6%) | 51.45 y ± 2.09 y (17.6–76.85) | 29.23 ± 0.96 (19.45–48.67) | n = 25 (51.0%) | n = 9 (18.4%) | ASA 1: n = 10 (20.4%) ASA 2: n = 35 (71.4%) ASA 3: n = 4 (8.2%) | n = 19 (38.8%) |
| Stage Two Fixation | Osteosynthese und MT Simultan | Bone Defect Length [mm] | Amount of Iliac Crest Grafts Used | Type of Iliac Crest Grafts Used | Origin of Iliac Crest Grafts | Arrangement of Iliac Crest Grafts | Fixation of Iliac Crest Grafts | Flap Coverage |
|---|---|---|---|---|---|---|---|---|
| locking plate n = 32 (65.3%) intramedullary nail n = 15 (30.7%) external fixation (Ilizarov) n = 1 (2%) cast n = 1 (2%) | n = 20 (40.8%) | 42.08 ± 2.19 mm (21.3–83.9) | one graft n = 27 (55.1%) two grafts n = 21 (42.9%) three grafts n = 1 (2%) | bicortical n = 10 (20.4%) tricortical n = 39 (79.6%) | anterior n = 47 posterior n = 1 anterior + posterior n = 1 | single n = 23 (46.9%) parallel n = 22 (44.9%) In line n = 4 (8.2%) | Press-fit n = 35 (71.4%) cerclage wire n = 2 (4.1%) screws n = 12 (24.5%) | n = 17 (34.7%) |
| Pathogens Detected at Stage One BBT | Recurrent Infection (n = 8) | Revision Surgeries Before Stage Two BBT | Interval Between Stage One and Two BBT [Days] | Temporary Fixation After Stage One BBT | Pathogen Detection at Stage Two BBT | Patients Without Antibiotic Therapy Before Stage Two BBT | Patients with Continuation of Antibiotic Therapy After Stage Two BBT |
|---|---|---|---|---|---|---|---|
| coagulase-negative staphylococcus n = 28 staphylococcus aureus n = 16 mixed infection n = 18 enterococcus n = 4 streptococcus n = 3 cutibacterium acnes n = 10 Gram-negative bacteria n = 6 others n = 8 | coagulase-negative staphylococcus n = 4 staphylococcus aureus n = 3 enterococcus n = 1 | n= 3.3 ± 2.1 (1–8) | 57.1 ± 4d (2–136) | locking plate n = 19 (38.8%) intramedullary nail n = 6 (12.2%) carbon rod n = 1 (2%) external fixateur n = 4 (8.2%) K-wire and cast n = 4 (8.2%) cast n = 15 (30.6%) | n = 12 (24.5%) | n = 19 (38.8%) | n = 32 (65.3%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hückstädt, M.; Fischer, C.; Weissmann, A.; Langwald, S.; Schröter, P.; Klauke, F.; Mendel, T.; Hofmann, G.O.; Kobbe, P.; Schipper, S. The “Bone Block Technique”: Reconstruction of Bone Defects Caused by Osteomyelitis Using Corticocancellous Bone Blocks from the Iliac Crest and the Induced Membrane Technique. Life 2025, 15, 1340. https://doi.org/10.3390/life15091340
Hückstädt M, Fischer C, Weissmann A, Langwald S, Schröter P, Klauke F, Mendel T, Hofmann GO, Kobbe P, Schipper S. The “Bone Block Technique”: Reconstruction of Bone Defects Caused by Osteomyelitis Using Corticocancellous Bone Blocks from the Iliac Crest and the Induced Membrane Technique. Life. 2025; 15(9):1340. https://doi.org/10.3390/life15091340
Chicago/Turabian StyleHückstädt, Marc, Christian Fischer, Alexander Weissmann, Steffen Langwald, Patrick Schröter, Friederike Klauke, Thomas Mendel, Gunther O. Hofmann, Philipp Kobbe, and Sandra Schipper. 2025. "The “Bone Block Technique”: Reconstruction of Bone Defects Caused by Osteomyelitis Using Corticocancellous Bone Blocks from the Iliac Crest and the Induced Membrane Technique" Life 15, no. 9: 1340. https://doi.org/10.3390/life15091340
APA StyleHückstädt, M., Fischer, C., Weissmann, A., Langwald, S., Schröter, P., Klauke, F., Mendel, T., Hofmann, G. O., Kobbe, P., & Schipper, S. (2025). The “Bone Block Technique”: Reconstruction of Bone Defects Caused by Osteomyelitis Using Corticocancellous Bone Blocks from the Iliac Crest and the Induced Membrane Technique. Life, 15(9), 1340. https://doi.org/10.3390/life15091340





