Impact of Intermittent Hypoxia Related to Obstructive Sleep Apnoea Syndrome on Low-Grade Inflammation in Hypertensive Patients: Potential Implications for Cardiovascular Risk
Abstract
1. Introduction
2. Materials and Methods
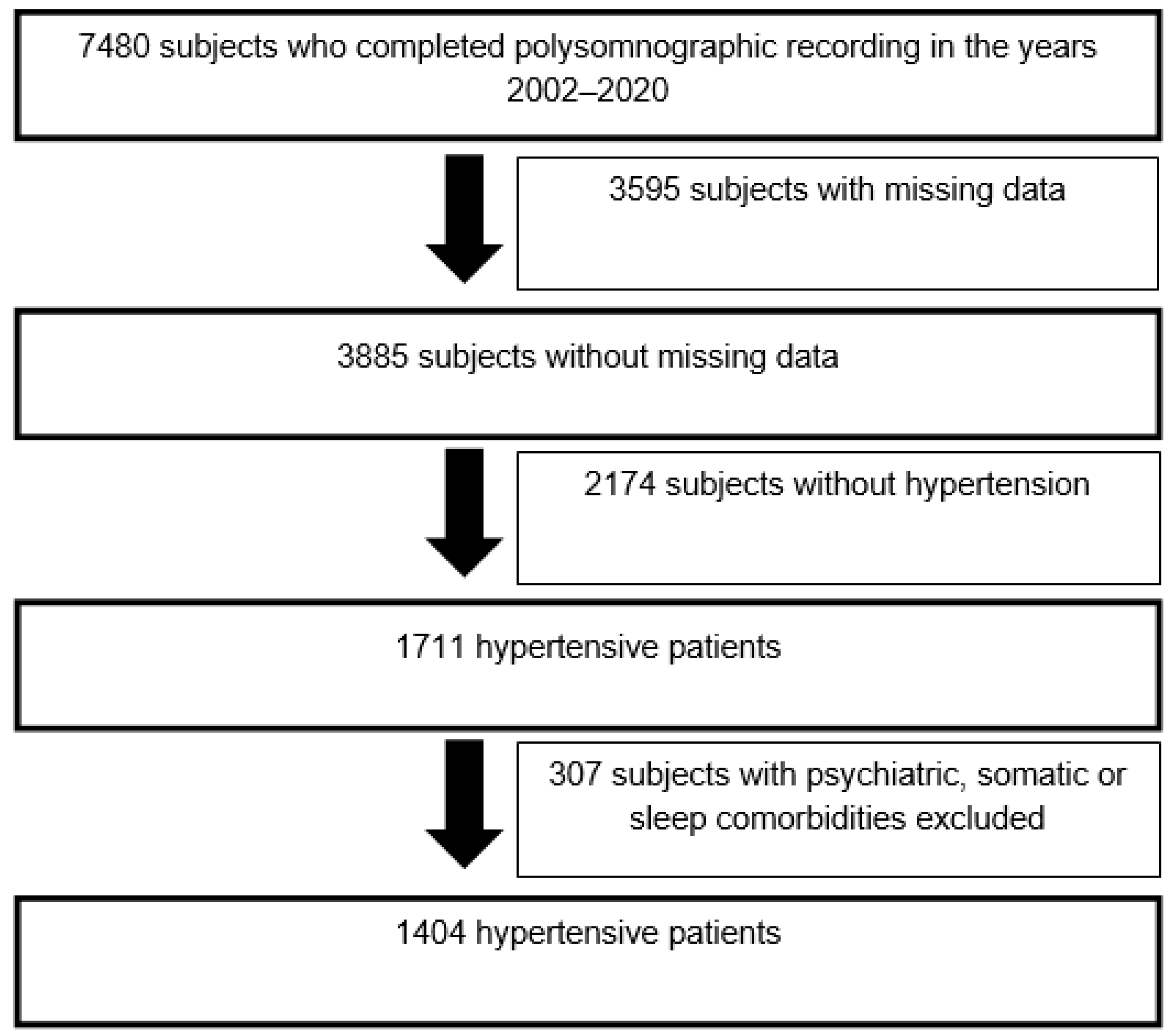
2.1. Population
2.2. Methods
2.2.1. Medical and Psychiatric Assessment
2.2.2. Sleep Evaluation and Study
2.3. Statistical Analyses
- -
- For gender, the presence of obesity and smoking status in model 1.
- -
- For gender, the presence of obesity, smoking status, presence of type 2 diabetes, presence of dyslipidaemia and aspirin therapy in model 2.
- -
- For gender, the presence of obesity, smoking status, presence of type 2 diabetes, presence of dyslipidaemia, aspirin therapy and Epworth Sleepiness Scale scores in model 3.
2.4. Data Collected
3. Results
3.1. Polysomnographic Data (Table 3)
| Whole Sample (n = 1404) | Subjects without LGI (n = 929) | Subjects with LGI (n = 475) | p-Value | |
|---|---|---|---|---|
| Sleep latency (min) | 26.3 (14.4–50.5) | 25.0 (14.3–49.0) | 28.0 (14.7–51.0) | 0.256 |
| Sleep efficiency (%) | 76.6 (66.2–83.9) | 76.8 (66.7–83.9) | 76.3 (65.2–84.0) | 0.680 |
| Sleep period time (min) | 449.5 (408.5–482.8) | 451.0 (410.5–485.5) | 445.7 (406.0–478.5) | 0.173 |
| Total sleep time (min) | 376.0 (326.4–416.5) | 379.0 (327.5–418.0) | 373.0 (322.0–414.5) | 0.231 |
| % stage 1 | 8.1 (5.4–11.5) | 8.2 (5.6–11.2) | 7.9 (5.2–11.8) | 0.470 |
| % stage 2 | 54.9 (47.2–61.4) | 55.0 (47.4–61.3) | 54.8 (46.2–61.7) | 0.755 |
| % Stage 3 | 2.3 (0.1–7.9) | 2.3 (0.1–7.6) | 2.6 (0.2–8.2) | 0.325 |
| % REM | 15.5 (10.9–19.3) | 15.6 (11.3–19.4) | 15.2 (10.2–19.3) | 0.115 |
| REM latency (min) | 85.0 (60.0–137.5) | 84.2 (60.0–132.0) | 87.5 (60.9–153.8) | 0.036 |
| % wake after sleep onset | 14.0 (8.4–22.6) | 13.9 (8.4–22.4) | 14.2 (8.1–23.2) | 0.987 |
| Number of awakenings | 32 (22–47) | 32 (22–47) | 32 (21–48) | 0.487 |
| Micro-arousal index | 13 (8–22) | 13 (8–21) | 14 (8–25) | 0.119 |
| Apnoea–hypopnoea index | 7 (2–22) | 7 (2–21) | 8 (2–28) | 0.151 |
| Oxygen desaturation index | 3 (1–11) | 3 (1–10) | 4 (1–15) | 0.001 |
| Total time under 90% of SaO2 (min) | 6.3 (0.3–49.8) | 4.3 (0.0–36.5) | 13.0 (0.5–78.0) | <0.001 |
| PLMS index | 2 (0–11) | 2 (0–12) | 1 (0–10) | 0.033 |
| Median (P25–P75) | Median (P25–P75) | Median (P25–P75) | Wilcoxon test |
3.2. Univariate Analyses (Table 4)
| Variables | Categories | % | Subjects without LGI | Subjects with LGI | p-Value Chi² | OR (CI 95%) | p-Value |
|---|---|---|---|---|---|---|---|
| Gender | Female (n = 435) male (n = 969) | 31.0% 69.0% | 25.2% 74.8% | 42.3% 57.7% | <0.001 | 1 0.46 (0.36 to 0.58) | <0.001 |
| Age (years) | <50 (n = 574) ≥50 (n = 830) | 40.9% 59.1% | 40.8% 59.2% | 41.1% 58.9% | 0.926 | 1 0.99 (0.79 to 1.24) | 0.926 |
| BMI (kg/m²) | <30 (n = 738) ≥30 (n = 666) | 52.5% 47.5% | 62.1% 37.9% | 33.9% 66.1% | <0.001 | 1 3.20 (2.54 to 4.03) | <0.001 |
| Smoking | No (n = 1131) Yes (n = 273) | 80.6% 19.4% | 82.4% 17.6% | 77.1% 22.9% | 0.018 | 1 1.39 (1.06 to 1.82) | 0.018 |
| Alcohol | No (n = 887) Yes (n = 517) | 63.2% 36.8% | 62.3% 37.7% | 64.8% 35.2% | 0.355 | 1 0.90 (0.71 to 1.13) | 0.355 |
| Caffeine | No (n = 279) Yes (n = 1125) | 19.9% 80.1% | 21.1% 78.9% | 17.5% 82.5% | 0.107 | 1 1.26 (0.95 to 1.68) | 0.108 |
| Type 2 diabetes | No (n = 1106) Yes (n = 298) | 78.8% 21.2% | 81.6% 18.4% | 73.3% 26.7% | <0.001 | 1 1.62 (1.24 to 2.10) | <0.001 |
| Dyslipidaemia status | No (n = 526) Untreated (n = 488) Treated (n = 390) | 37.5% 34.8% 27.7% | 37.6% 32.0% 30.4% | 37.3% 40.2% 22.5% | 0.001 | 1 1.27 (0.98 to 1.64) 0.75 (0.56 to 0.99) | 0.001 |
| Hypertension status | Untreated (n = 533) Controlled (n = 527) Uncontrolled (n = 344) | 38.0% 37.5% 24.5% | 39.3% 37.9% 22.8% | 35.4% 36.8% 27.8% | 0.104 | 1 1.08 (0.84 to 1.40) 1.35 (1.02 to 1.80) | 0.104 |
| Number of antihypertensive treatments | 0 (n = 533) 1 (n = 510) 2 (n = 236) ≥3 (n = 125) | 38.0% 36.3% 16.8% 8.9% | 39.3% 36.1% 16.6% 8.0% | 35.4% 36.8% 17.3% 10.5% | 0.316 | 1 1.13 (0.88 to 1.47) 1.16 (0.84 to 1.60) 1.45 (0.97 to 2.16) | 0.318 |
| Cardiovascular comorbidities | No (n = 1119) Yes (n = 285) | 79.7% 20.3% | 79.9% 20.1% | 79.4% 20.6% | 0.825 | 1 1.03 (0.78 to 1.36) | 0.825 |
| Aspirin therapy | No (n = 1084) Yes (n = 320) | 77.2% 22.8% | 75.4% 34.6% | 80.8% 19.2% | 0.020 | 1 0.72 (0.55 to 0.95) | 0.021 |
| OSAS status | No (n = 566) Mild (n = 362) Moderate to severe with ODI < 15/h (n = 217) Moderate to severe without ODI ≥ 15/h (n = 259) | 40.3% 25.8% 15.5% 18.4% | 40.6% 27.3% 16.3% 15.8% | 39.8% 22.7% 13.9% 23.6% | 0.003 | 1 0.85 (0.64 to 1.13) 0.87 (0.62 to 1.22) 1.52 (1.12 to 2.05) | 0.003 |
| Insomnia disorder | No (n = 469) Sleep deprivation alone (n = 314) Insomnia without short sleep duration (n = 367) Insomnia with short sleep duration (n = 254) | 33.4% 22.4% 26.1% 18.1% | 34.8% 21.9% 25.7% 17.6% | 30.7% 23.4% 27.0% 18.9% | 0.508 | 1 1.21 (0.89 to 1.64) 1.18 (0.89 to 1.58) 1.21 (0.88 to 1.68) | 0.509 |
| Sleep movement disorders | No (n = 1112) Moderate to severe PLMs alone (n = 110) RLS alone or combined with PLMs (n = 182) | 79.2% 7.8% 13.0% | 78.2% 8.0% 13.8% | 81.3% 7.6% 11.1% | 0.325 | 1 0.91 (0.60 to 1.39) 0.77 (0.55 to 1.09) | 0.326 |
| ESS | <11 (n = 843) ≥11 and <14 (n = 245) ≥14 (n = 316) | 60.0% 17.5% 22.5% | 61.8% 17.8% 20.4% | 56.6% 16.8% 26.6% | 0.035 | 1 1.03 (0.76 to 1.40) 1.42 (1.08 to 1.85) | 0.036 |
| Major depression | No (n = 783) Remitted (n = 295) Current (n = 326) | 55.8% 21.0% 23.2% | 56.8% 20.8% 22.4% | 53.7% 21.5% 24.8% | 0.485 | 1 1.09 (0.82 to 1.45) 1.17 (0.90 to 1.54) | 0.485 |
| LGI | No (n = 929) Yes (n = 475) | 66.2% 33.8% | |||||
| Median (P25–P75) | Wilcoxon test | ||||||
| Age (years) | 52 (45–60) | 53 (45–60) | 52 (45–59) | 0.380 | |||
| BMI (kg/m²) | 29.6 (26.2–33.8) | 28.4 (25.3–32.1) | 32.2 (28.4–37.1) | <0.001 | |||
| CRP (mg/L) | 1.9 (1.0–3.8) | 1.2 (0.1–1.9) | 4.9 (3.8–7.0) | <0.001 | |||
| ESS | 9 (6–13) | 9 (5–13) | 9 (6–14) | 0.005 | |||
| ISI | 14 (9–18) | 14 (9–17) | 14 (10–18) | 0.164 | |||
| BDI | 4 (2–8) | 4 (2–8) | 4 (2–8) | 0.056 |
3.3. Multivariate Analyses (Table 5)
| Variables | Model 1 OR Adjusted (CI 95%) | p-Value | Model 2 OR Adjusted (CI 95%) | p-Value | Model 3 OR Adjusted (CI 95%) | p-Value |
|---|---|---|---|---|---|---|
| OSAS No Mild Moderate to severe with ODI < 15/h Moderate to severe without ODI ≥ 15/h | 1 0.92 (0.68 to 1.25) 0.88 (0.61 to 1.27) 1.50 (1.06 to 2.13) | 0.023 | 1 0.95 (0.70 to 1.30) 0.93 (0.64 to 1.34) 1.51 (1.06 to 2.14) | 0.041 | 1 0.95 (0.70 to 1.30) 0.93 (0.64 to 1.35) 1.51 (1.06 to 2.14) | 0.045 |
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AHI | apnoea–hypopnoea index |
| CPAP | continuous positive airway pressure |
| CRP | C-reactive protein |
| DSM | Diagnostic and Statistical Manual of Mental Disorders |
| ODI | oxygen desaturation index |
| OSAS | obstructive sleep apnoea syndrome |
References
- Xiao, L.; Harrison, D.G. Inflammation in Hypertension. Can. J. Cardiol. 2020, 36, 635–647. [Google Scholar] [CrossRef] [PubMed]
- Kanclerska, J.; Wieckiewicz, M.; Nowacki, D.; Szymanska-Chabowska, A.; Poreba, R.; Mazur, G.; Martynowicz, H. Sleep architecture and vitamin D in hypertensives with obstructive sleep apnea: A polysomnographic study. Dent. Med. Probl. 2024, 61, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Jayedi, A.; Rahimi, K.; Bautista, L.E.; Nazarzadeh, M.; Zargar, M.S.; Shab-Bidar, S. Inflammation markers and risk of developing hypertension: A meta-analysis of cohort studies. Heart 2019, 105, 686–692. [Google Scholar] [CrossRef]
- Hage, F.G. C-reactive protein and hypertension. J. Hum. Hypertens. 2014, 28, 410–415. [Google Scholar] [CrossRef]
- Adukauskienė, D.; Čiginskienė, A.; Adukauskaitė, A.; Pentiokinienė, D.; Šlapikas, R.; Čeponienė, I. Clinical relevance of high sensitivity C-reactive protein in cardiology. Medicina 2016, 52, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Cortez, A.F.; Muxfeldt, E.S.; Cardoso, C.R.; Salles, G.F. Prognostic Value of C-Reactive Protein in Resistant Hypertension. Am. J. Hypertens. 2016, 29, 992–1000. [Google Scholar] [CrossRef]
- Liu, H.H.; Cao, Y.X.; Sun, D.; Jin, J.L.; Zhang, H.W.; Guo, Y.L.; Zhu, C.G.; Wu, N.Q.; Gao, Y.; Dong, Q.T.; et al. High-sensitivity C-reactive protein and hypertension: Combined effects on coronary severity and cardiovascular outcomes. Hypertens. Res. 2019, 42, 1783–1793. [Google Scholar] [CrossRef]
- Maio, R.; Perticone, M.; Suraci, E.; Sciacqua, A.; Sesti, G.; Perticone, F. Endothelial dysfunction and C-reactive protein predict the incidence of heart failure in hypertensive patients. ESC Heart Fail. 2021, 8, 399–407. [Google Scholar] [CrossRef]
- Silvestre-Roig, C.; Braster, Q.; Ortega-Gomez, A.; Soehnlein, O. Neutrophils as regulators of cardiovascular inflammation. Nat. Rev. Cardiol. 2020, 17, 327–340. [Google Scholar] [CrossRef]
- Kim-Mitsuyama, S.; Soejima, H.; Yasuda, O.; Node, K.; Jinnouchi, H.; Yamamoto, E.; Sekigami, T.; Ogawa, H.; Matsui, K. Reduction in hsCRP levels is associated with decreased incidence of cardiovascular events in Japanese hypertensive women but not in men. Sci. Rep. 2020, 10, 17040. [Google Scholar] [CrossRef]
- Fang, M.; Qian, Q.; Zhao, Z.; Zhu, L.; Su, J.; Li, X. High-Sensitivity C-Reactive Protein Combined with Low-Density Lipoprotein Cholesterol as the Targets of Statin Therapy in Patients with Acute Coronary Syndrome. Int. Heart J. 2018, 59, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Amezcua-Castillo, E.; González-Pacheco, H.; Sáenz-San Martín, A.; Méndez-Ocampo, P.; Gutierrez-Moctezuma, I.; Massó, F.; Sierra-Lara, D.; Springall, R.; Rodríguez, E.; Arias-Mendoza, A.; et al. C-Reactive Protein: The Quintessential Marker of Systemic Inflammation in Coronary Artery Disease-Advancing toward Precision Medicine. Biomedicines 2023, 11, 2444. [Google Scholar] [CrossRef] [PubMed]
- Imani, M.M.; Sadeghi, M.; Farokhzadeh, F.; Khazaie, H.; Brand, S.; Dürsteler, K.M.; Brühl, A.; Sadeghi-Bahmani, D. Evaluation of Blood Levels of C-Reactive Protein Marker in Obstructive Sleep Apnea: A Systematic Review, Meta-Analysis and Meta-Regression. Life 2021, 11, 362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, W.; Liu, K.; Chen, Z.; Fei, Q.; Ahmad, N.; Yi, M. The causal associations of altered inflammatory proteins with sleep duration, insomnia and daytime sleepiness. Sleep 2023, 46, zsad207. [Google Scholar] [CrossRef]
- Jiménez-Jiménez, F.J.; Alonso-Navarro, H.; García-Martín, E.; Agúndez, J.A.G. Inflammatory factors and restless legs syndrome: A systematic review and meta-analysis. Sleep Med. Rev. 2023, 68, 101744. [Google Scholar] [CrossRef] [PubMed]
- Lv, R.; Liu, X.; Zhang, Y.; Dong, N.; Wang, X.; He, Y.; Yue, H.; Yin, Q. Pathophysiological mechanisms and therapeutic approaches in obstructive sleep apnea syndrome. Signal Transduct. Target. Ther. 2023, 8, 218. [Google Scholar] [PubMed]
- Alterki, A.; Abu-Farha, M.; Al Shawaf, E.; Al-Mulla, F.; Abubaker, J. Investigating the Relationship between Obstructive Sleep Apnoea, Inflammation and Cardio-Metabolic Diseases. Int. J. Mol. Sci. 2023, 24, 6807. [Google Scholar] [CrossRef] [PubMed]
- Van der Touw, T.; Andronicos, N.M.; Smart, N. Is C-reactive protein elevated in obstructive sleep apnea? a systematic review and meta-analysis. Biomarkers 2019, 24, 429–435. [Google Scholar] [CrossRef]
- Willame, H.; Wacquier, B.; Point, C.; Dosogne, M.; Al Faker, M.; Loas, G.; Hein, M. The association between type 2 diabetes and anhedonic subtype of major depression in hypertensive individuals. J. Clin. Hypertens. 2022, 24, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Hein, M.; Wacquier, B.; Lanquart, J.P.; Loas, G. Risk of Cardiovascular Disease in Apnoeic Individuals: Role of Comorbid Insomnia Disorder. Life 2022, 12, 944. [Google Scholar] [CrossRef]
- Li, F.; Huang, H.; Song, L.; Hao, H.; Ying, M. Effects of Obstructive Sleep Apnea Hypopnea Syndrome on Blood Pressure and C-Reactive Protein in Male Hypertension Patients. J. Clin. Med. Res. 2016, 8, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Thomopoulos, C.; Tsioufis, C.; Dimitriadis, K.; Tsiachris, D.; Tousoulis, D.; Manolis, A.; Alchanatis, M.; Kallikazaros, I.; Stefanadis, C. Obstructive sleep apnoea syndrome is associated with enhanced sub-clinical inflammation and asymmetric dimethyl-arginine levels in hypertensives. J. Hum. Hypertens. 2009, 23, 65–67. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Qian, X.; Yin, T.; Li, T.; Kang, C.; Guo, R.; Sun, B.; Liu, C. High levels of inflammation and insulin resistance in obstructive sleep apnea patients with hypertension. Inflammation 2012, 35, 1507–1511. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, H.; Guo, C.; Duan, X.; Hu, F.; Yang, W.; Cui, J.; Song, L.; Chun, Y.; Yuan, J.; et al. Association between severity of obstructive sleep apnea and high-sensitivity C-reactive protein in patients with hypertrophic obstructive cardiomyopathy. Clin. Cardiol. 2020, 43, 803–811. [Google Scholar] [CrossRef]
- Fukuoka, R.; Kohno, T.; Kohsaka, S.; Yanagisawa, R.; Kawakami, T.; Hayashida, K.; Kanazawa, H.; Yuasa, S.; Maekawa, Y.; Sano, M.; et al. Nocturnal intermittent hypoxia and short sleep duration are independently associated with elevated C-reactive protein levels in patients with coronary artery disease. Sleep Med. 2017, 29, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Więckiewicz, M.; Lavigne, G.; Martynowicz, H. Decrypting the putative interrelation between sleep bruxism, masticatory muscle pain and sleep breathing disorders: Nosology and the role of hypoxia. Dent. Med. Probl. 2024, 61, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.T.; Bundy, J.D.; Kelly, T.N.; Reed, J.E.; Kearney, P.M.; Reynolds, K.; Chen, J.; He, J. Global disparities of hypertension prevalence and control: A systematic analysis of population-based studies from 90 countries. Circulation 2016, 134, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Draelants, L.; Point, C.; Wacquier, B.; Lanquart, J.P.; Loas, G.; Hein, M. 10-Year Risk for Cardiovascular Disease Associated with COMISA (Co-Morbid Insomnia and Sleep Apnea) in Hypertensive Subjects. Life 2023, 13, 1379. [Google Scholar] [CrossRef] [PubMed]
- Bernier, V.; Debarge, M.H.; Hein, M.; Ammendola, S.; Mungo, A.; Loas, G. Major Depressive Disorder, Inflammation, and Nutrition: A Tricky Pattern? Nutrients 2023, 15, 3438. [Google Scholar] [CrossRef] [PubMed]
- Pearson, T.A.; Mensah, G.A.; Alexander, R.W.; Anderson, J.L.; Cannon, R.O.; Criqui, M.; Fadl, Y.Y.; Fortmann, S.P.; Hong, Y.; Myers, G.L.; et al. Markers of inflammation and cardiovascular disease: Application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 2003, 107, 499–511. [Google Scholar] [CrossRef]
- Castro, A.R.; Silva, S.O.; Soares, S.C. The Use of High Sensitivity C-Reactive Protein in Cardiovascular Disease Detection. J. Pharm. Pharm. Sci. 2018, 21, 496–503. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; Text Rev; American Psychiatric Publishing: Arlington, VA, USA, 2000. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing: Washington, DC, USA, 2013. [Google Scholar]
- Beck, A.T.; Steer, R.A.; Ball, R.; Ranieri, W. Comparison of Beck Depression Inventories -IA and -II in psychiatric outpatients. J. Pers. Assess. 1996, 67, 588–597. [Google Scholar] [CrossRef]
- Johns, M.W. A new method for measuring daytime sleepiness: The Epworth sleepiness scale. Sleep 1991, 14, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Morin, C.M. Insomnia: Psychological Assessment and Management; Guilford Press: New York, NY, USA, 1993. [Google Scholar]
- Kushida, C.A.; Littner, M.R.; Morgenthaler, T.; Alessi, C.A.; Bailey, D.; Coleman, J.; Friedman, L.; Hirshkowitz, M.; Kapen, S.; Kramer, M.; et al. Practice Parameters for the Indications for Polysomnography and Related Procedures: An Update for 2005. Sleep 2005, 28, 499–521. [Google Scholar] [CrossRef] [PubMed]
- Iber, C.; Ancoli-Israel, S.; Chesson, A.; Quan, S.F.; for the American Academy of Sleep Medicine. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications, 1st ed.; American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- Ferri, R.; Koo, B.B.; Picchietti, D.L.; Fulda, S. Periodic leg movements during sleep: Phenotype, neurophysiology, and clinical significance. Sleep Med. 2017, 31, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J.; Gozal, D.; Iber, C.; Kapur, V.K.; Marcus, C.L.; Mehra, R.; Parthasarathy, S.; Quan, S.F.; et al. Rules for scoring respiratory events in sleep: Update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J. Clin. Sleep Med. 2012, 8, 597–619. [Google Scholar] [CrossRef] [PubMed]
- Edinger, J.D.; Bonnet, M.H.; Bootzin, R.R.; Doghramji, K.; Dorsey, C.M.; Espie, C.A.; Jamieson, A.O.; McCall, W.V.; Morin, C.M.; Stepanski, E.J.; et al. Derivation of research diagnostic criteria for insomnia: Report of an American Academy of Sleep Medicine Work Group. Sleep 2004, 27, 1567–1596. [Google Scholar] [CrossRef] [PubMed]
- Fleetham, J.; Ayas, N.; Bradley, D.; Ferguson, K.; Fitzpatrick, M.; George, C.; Hanly, P.; Hill, F.; Kimoff, J.; Kryger, M.; et al. Canadian Thoracic Society guidelines: Diagnosis and treatment of sleep disordered breathing in adults. Can. Respir. J. 2006, 13, 387–392. [Google Scholar] [CrossRef]
- Muraki, I.; Tanigawa, T.; Yamagishi, K.; Sakurai, S.; Ohira, T.; Imano, H.; Kitamura, A.; Kiyama, M.; Sato, S.; Shimamoto, T.; et al. Nocturnal intermittent hypoxia and C reactive protein among middle-aged community residents: A cross-sectional survey. Thorax 2010, 65, 523–527. [Google Scholar] [CrossRef]
- Haba-Rubio, J.; Marti-Soler, H.; Tobback, N.; Andries, D.; Marques-Vidal, P.; Vollenweider, P.; Preisig, M.; Heinzer, R. Clinical significance of periodic limb movements during sleep: The HypnoLaus study. Sleep Med. 2018, 41, 45–50. [Google Scholar] [CrossRef]
- Allen, R.P.; Picchietti, D.L.; Garcia-Borreguero, D.; Ondo, W.G.; Walters, A.S.; Winkelman, J.W.; Zucconi, M.; Ferri, R.; Trenkwalder, C.; Lee, H.B.; et al. Restless legs syndrome/Willis-Ekbom disease diagnostic criteria: Updated International Restless Legs Syndrome Study Group (IRLSSG) consensus criteria--history, rationale, description, and significance. Sleep Med. 2014, 15, 860–873. [Google Scholar] [CrossRef] [PubMed]
- Hein, M.; Lanquart, J.P.; Loas, G.; Hubain, P.; Linkowski, P. Insomnia with short sleep duration as risk factor for type 2 diabetes: A systematic review of the literature. Rev. Med. Brux. 2020, 41, 98–104. [Google Scholar] [CrossRef]
- Peduzzi, P.; Concato, J.; Feinstein, A.R.; Holford, T.R. Importance of events per independent variable in proportional hazards regression analysis. II. Accuracy and precision of regression estimates. J. Clin. Epidemiol. 1995, 48, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Peduzzi, P.; Concato, J.; Kemper, E.; Holford, T.R.; Feinstein, A.R. A simulation study of the number of events per variable in logistic regression analysis. J. Clin. Epidemiol. 1996, 49, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Delongui, F.; Kallaur, A.P.; Oliveira, S.R.; Bonametti, A.M.; Grion, C.M.; Morimoto, H.K.; Simão, A.N.; Magalhães, G.G.; Reiche, E.M. Serum levels of high sensitive C reactive protein in healthy adults from southern Brazil. J. Clin. Lab. Anal. 2013, 27, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Vu, T.H.; Liu, K.; Lloyd-Jones, D.M.; Stamler, J.; Pirzada, A.; Shah, S.J.; Garside, D.B.; Daviglus, M.L. Favorable Levels of All Major Cardiovascular Risk Factors at Younger Ages and High-Sensitivity C-Reactive Protein 39 Years Later—The Chicago Healthy Aging Study. Prev. Med. Rep. 2015, 2, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Aboukhater, D.; Morad, B.; Nasrallah, N.; Nasser, S.A.; Sahebkar, A.; Kobeissy, F.; Boudaka, A.; Eid, A.H. Inflammation and hypertension: Underlying mechanisms and emerging understandings. J. Cell. Physiol. 2023, 238, 1148–1159. [Google Scholar] [CrossRef] [PubMed]
- Iwashima, Y.; Horio, T.; Kamide, K.; Rakugi, H.; Ogihara, T.; Kawano, Y. C-reactive protein, left ventricular mass index, and risk of cardiovascular disease in essential hypertension. Hypertens. Res. 2007, 30, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Rossi, G.P.; Bisogni, V.; Rossitto, G.; Maiolino, G.; Cesari, M.; Zhu, R.; Seccia, T.M. Practice Recommendations for Diagnosis and Treatment of the Most Common Forms of Secondary Hypertension. High Blood Press. Cardiovasc. Prev. 2020, 27, 547–560. [Google Scholar]
- Barbaro, N.R.; Fontana, V.; Modolo, R.; De Faria, A.P.; Sabbatini, A.R.; Fonseca, F.H.; Anhê, G.F.; Moreno, H. Increased arterial stiffness in resistant hypertension is associated with inflammatory biomarkers. Blood Press. 2015, 24, 7–13. [Google Scholar] [CrossRef]
- Hwang, A.Y.; Dietrich, E.; Pepine, C.J.; Smith, S.M. Resistant Hypertension: Mechanisms and Treatment. Curr. Hypertens. Rep. 2017, 19, 56. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Corrado, E.; Coppola, G.; Muratori, I.; Mezzani, A.; Novo, G.; Novo, S. The predictive role of C-reactive protein in patients with hypertension and subclinical atherosclerosis. Intern. Med. J. 2009, 39, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Zdrojewski, T.; Chwojnicki, K.; Bandosz, P.; Konarski, R.; Wyrzykowski, B. Distribution of C-reactive protein and its relation to arterial hypertension in a country representing a high-risk region for cardiovascular diseases. Blood Press. 2006, 15, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Virdis, A.; Dell’Agnello, U.; Taddei, S. Impact of inflammation on vascular disease in hypertension. Maturitas 2014, 78, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Hein, M.; Lanquart, J.P.; Hubain, P.; Loas, G. Risk of resistant hypertension associated with restless legs syndrome and periodic limb movements during sleep: A study on 673 treated hypertensive individuals. Sleep Med. 2019, 63, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Svensson, M.; Venge, P.; Janson, C.; Lindberg, E. Relationship between sleep-disordered breathing and markers of systemic inflammation in women from the general population. J. Sleep Res. 2012, 21, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Arnardottir, E.S.; Maislin, G.; Schwab, R.J.; Staley, B.; Benediktsdottir, B.; Olafsson, I.; Juliusson, S.; Romer, M.; Gislason, T.; Pack, A.I. The interaction of obstructive sleep apnea and obesity on the inflammatory markers C-reactive protein and interleukin-6: The Icelandic Sleep Apnea Cohort. Sleep 2012, 35, 921–932. [Google Scholar] [CrossRef] [PubMed]
- Gouveris, H.; Bahr, K.; Jahn, C.; Matthias, C.; Simon, P. The Apnea-Hypopnea Index Underestimates Systemic Inflammation in Women with Sleep-Disordered Breathing. J. Women’s Health 2018, 27, 920–926. [Google Scholar]
- Punjabi, N.M.; Newman, A.B.; Young, T.B.; Resnick, H.E.; Sanders, M.H. Sleep-disordered breathing and cardiovascular disease: An outcome-based definition of hypopneas. Am. J. Respir. Crit. Care Med. 2008, 177, 1150–1155. [Google Scholar] [CrossRef] [PubMed]
- Lavalle, S.; Masiello, E.; Iannella, G.; Magliulo, G.; Pace, A.; Lechien, J.R.; Calvo-Henriquez, C.; Cocuzza, S.; Parisi, F.M.; Favier, V.; et al. Unraveling the Complexities of Oxidative Stress and Inflammation Biomarkers in Obstructive Sleep Apnea Syndrome: A Comprehensive Review. Life 2024, 14, 425. [Google Scholar] [CrossRef]
- Archontogeorgis, K.; Nena, E.; Papanas, N.; Steiropoulos, P. Biomarkers to improve diagnosis and monitoring of obstructive sleep apnea syndrome: Current status and future perspectives. Pulm. Med. 2014, 2014, 930535. [Google Scholar] [CrossRef]
- Hein, M.; Mungo, A.; Hubain, P.; Loas, G. Excessive daytime sleepiness in adolescents: Current treatment strategies. Sleep Sci. 2020, 13, 157–171. [Google Scholar]
- Jin, F.; Liu, J.; Zhang, X.; Cai, W.; Zhang, Y.; Zhang, W.; Yang, J.; Lu, G.; Zhang, X. Effect of continuous positive airway pressure therapy on inflammatory cytokines and atherosclerosis in patients with obstructive sleep apnea syndrome. Mol. Med. Rep. 2017, 16, 6334–6339. [Google Scholar] [CrossRef] [PubMed]
- Panoutsopoulos, A.; Kallianos, A.; Kostopoulos, K.; Seretis, C.; Koufogiorga, E.; Protogerou, A.; Trakada, G.; Kostopoulos, C.; Zakopoulos, N.; Nikolopoulos, I. Effect of CPAP treatment on endothelial function and plasma CRP levels in patients with sleep apnea. Med. Sci. Monit. 2012, 18, CR747–CR751. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Pan, L.; Ren, D.; Xie, X. Impact of continuous positive airway pressure on C-reactive protein in patients with obstructive sleep apnea: A meta-analysis. Sleep Breath. 2013, 17, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Pan, L.; Ren, D.; Du, C.; Guo, Y. Effects of continuous positive airway pressure therapy on systemic inflammation in obstructive sleep apnea: A meta-analysis. Sleep Med. 2013, 14, 1139–1150. [Google Scholar] [CrossRef] [PubMed]
- Cai, A.; Wang, L.; Zhou, Y. Hypertension and obstructive sleep apnea. Hypertens. Res. 2016, 39, 391–395. [Google Scholar] [CrossRef]
- Parati, G.; Lombardi, C.; Hedner, J.; Bonsignore, M.R.; Grote, L.; Tkacova, R.; Lévy, P.; Riha, R.; Bassetti, C.; Narkiewicz, K.; et al. Recommendations for the management of patients with obstructive sleep apnoea and hypertension. Eur. Respir. J. 2013, 41, 523–538. [Google Scholar] [CrossRef] [PubMed]
- Hopps, E.; Caimi, G. Obstructive Sleep Apnea Syndrome: Links Between Pathophysiology and Cardiovascular Complications. Clin. Investig. Med. 2015, 38, E362–E370. [Google Scholar] [CrossRef]
- Gupta, L.; Thomas, J.; Ravichandran, R.; Singh, M.; Nag, A.; Panjiyar, B.K. Inflammation in Cardiovascular Disease: A Comprehensive Review of Biomarkers and Therapeutic Targets. Cureus 2023, 15, e45483. [Google Scholar] [CrossRef]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Hypertension according to the diagnostic criteria of the World Health Organization: mean systolic blood pressure ≥140 mmHg or mean diastolic blood pressure ≥90 mmHg or self-reported diagnosis of clinically demonstrated hypertension or taking antihypertensive medication | Acute and/or uncontrolled hepatic, pancreatic, pulmonary, cardiovascular, renal or infectious diseases |
| Age ≥ 18 years old | Past or current severe psychiatric diseases (psychotic disorder, bipolar disorder and substance use disorder) |
| CRP levels < 10 mg/L | Past or current cancer, autoimmune or inflammatory diseases |
| Absence of pregnancy | Past or current malformation or lesion (head trauma, lesions of cerebral respiratory centres, craniofacial malformations and abnormal chest deformities) |
| Central disorders of hypersomnolence, OSAS already known or course of treatment before Sleep Laboratory and sleep apnoea syndrome with predominantly central component |
| Analyses | Results |
|---|---|
| Comparison of polysomnographic parameters | Hypertensive patients with low-grade inflammation have higher parameters related to intermittent hypoxia (oxygen desaturation index and total time under 90% of oxygen saturation) than those without low-grade inflammation. |
| Prevalence of low-grade inflammation in hypertensive individuals | 33.8% |
| Univariate analyses | Confounding factors identified: gender, body mass index, smoking, type 2 diabetes, dyslipidaemia status, aspirin therapy and Epworth Sleepiness Scale scores. |
| Multivariate analyses | Unlike mild OSAS and moderate to severe OSAS with low intermittent hypoxia, only moderate to severe OSAS with high intermittent hypoxia was significantly associated with higher risk of low-grade inflammation in hypertensive patients after adjustment for confounding factors. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hein, M.; Wacquier, B.; Conenna, M.; Lanquart, J.-P.; Point, C. Impact of Intermittent Hypoxia Related to Obstructive Sleep Apnoea Syndrome on Low-Grade Inflammation in Hypertensive Patients: Potential Implications for Cardiovascular Risk. Life 2024, 14, 592. https://doi.org/10.3390/life14050592
Hein M, Wacquier B, Conenna M, Lanquart J-P, Point C. Impact of Intermittent Hypoxia Related to Obstructive Sleep Apnoea Syndrome on Low-Grade Inflammation in Hypertensive Patients: Potential Implications for Cardiovascular Risk. Life. 2024; 14(5):592. https://doi.org/10.3390/life14050592
Chicago/Turabian StyleHein, Matthieu, Benjamin Wacquier, Matteo Conenna, Jean-Pol Lanquart, and Camille Point. 2024. "Impact of Intermittent Hypoxia Related to Obstructive Sleep Apnoea Syndrome on Low-Grade Inflammation in Hypertensive Patients: Potential Implications for Cardiovascular Risk" Life 14, no. 5: 592. https://doi.org/10.3390/life14050592
APA StyleHein, M., Wacquier, B., Conenna, M., Lanquart, J.-P., & Point, C. (2024). Impact of Intermittent Hypoxia Related to Obstructive Sleep Apnoea Syndrome on Low-Grade Inflammation in Hypertensive Patients: Potential Implications for Cardiovascular Risk. Life, 14(5), 592. https://doi.org/10.3390/life14050592






