Mineral-Mediated Oligoribonucleotide Condensation: Broadening the Scope of Prebiotic Possibilities on the Early Earth
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
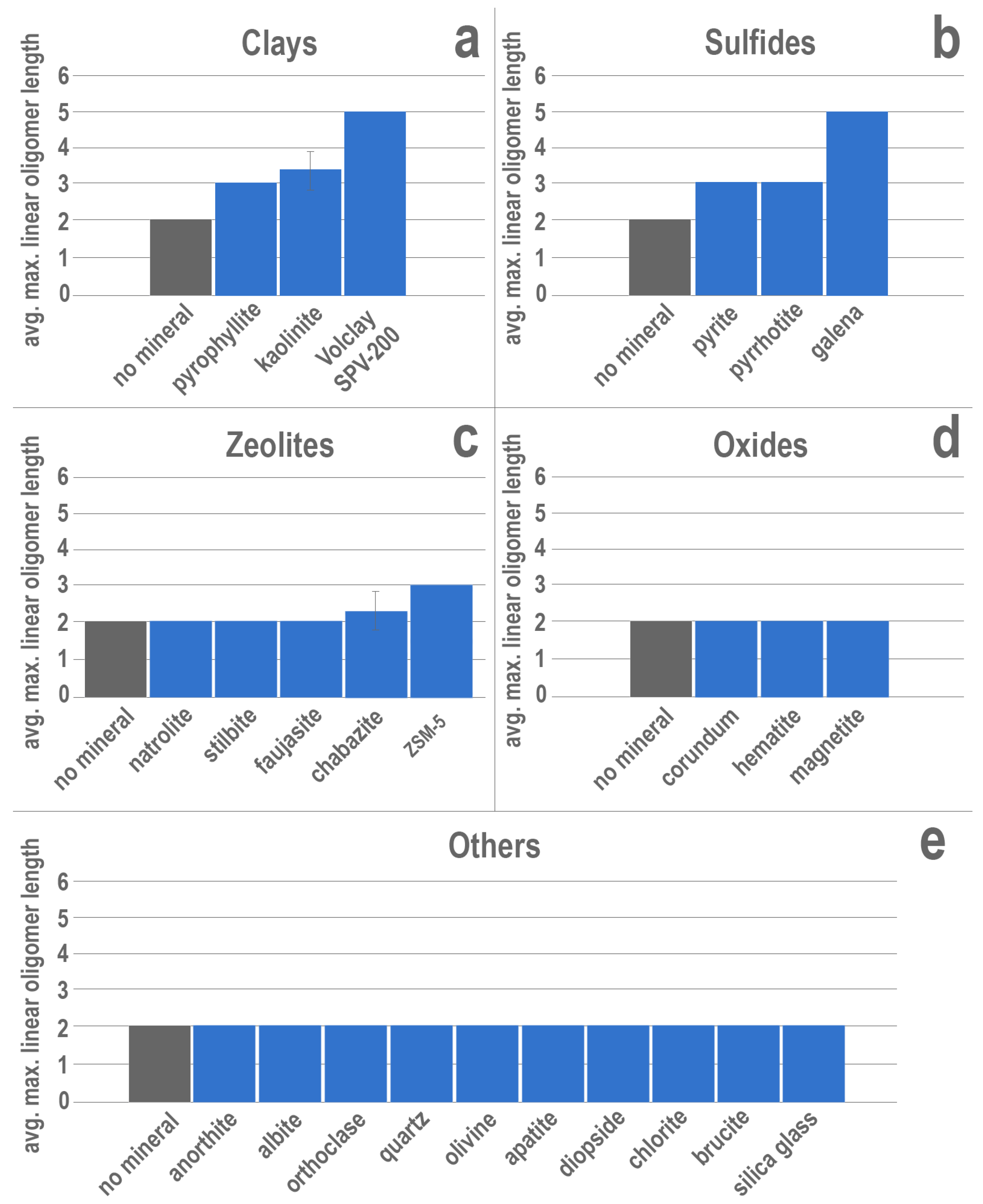
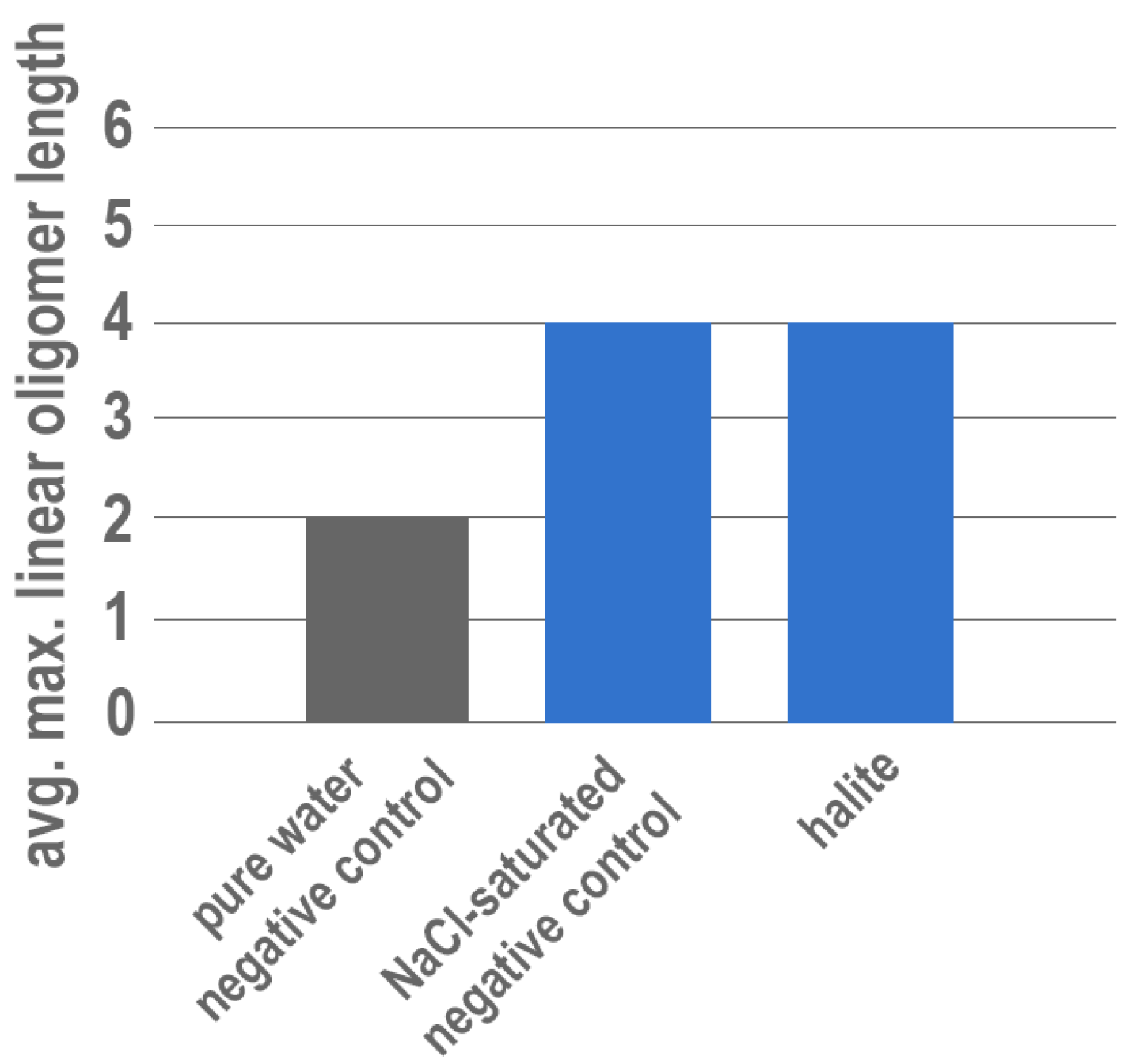
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
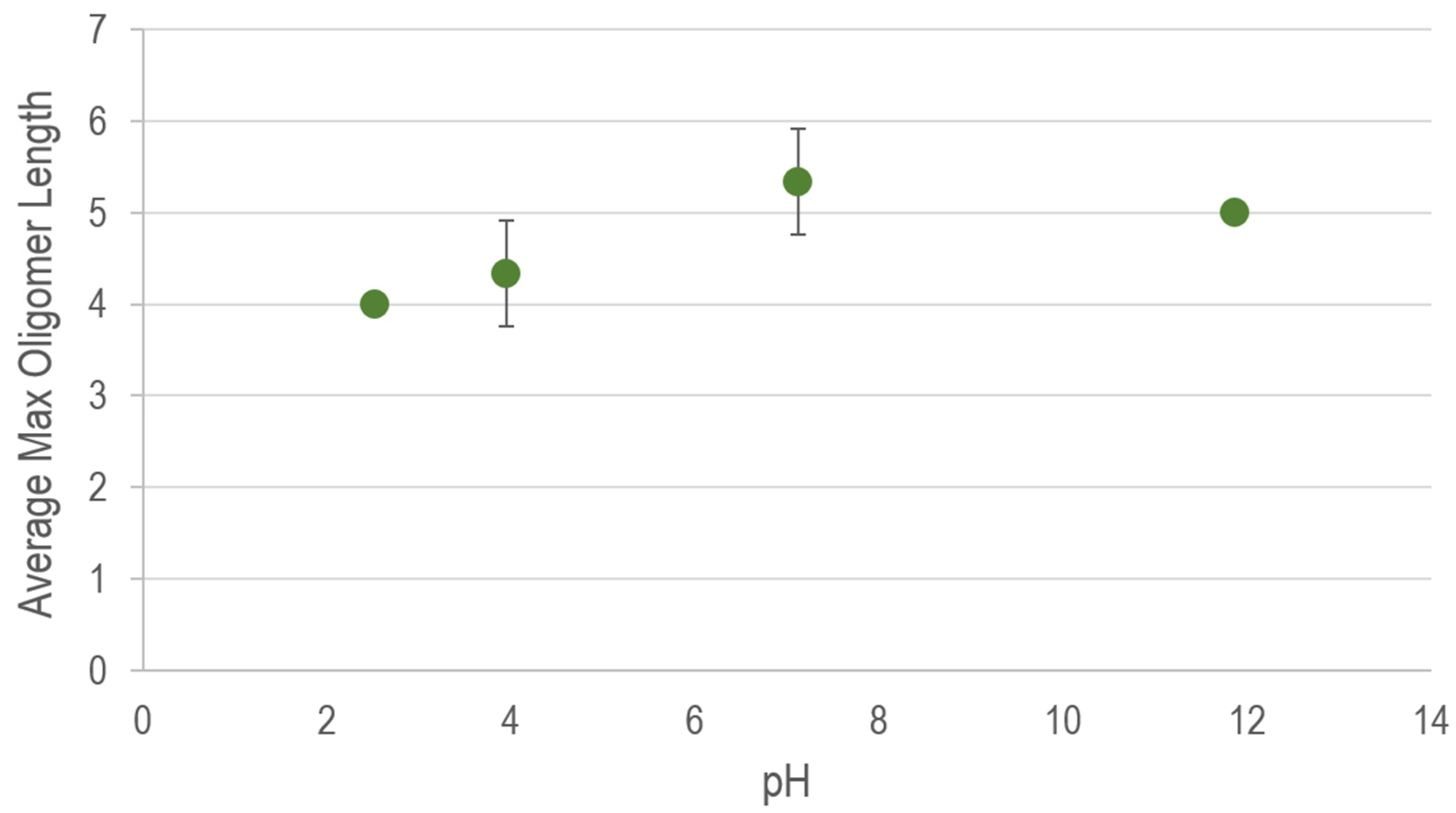
Appendix A
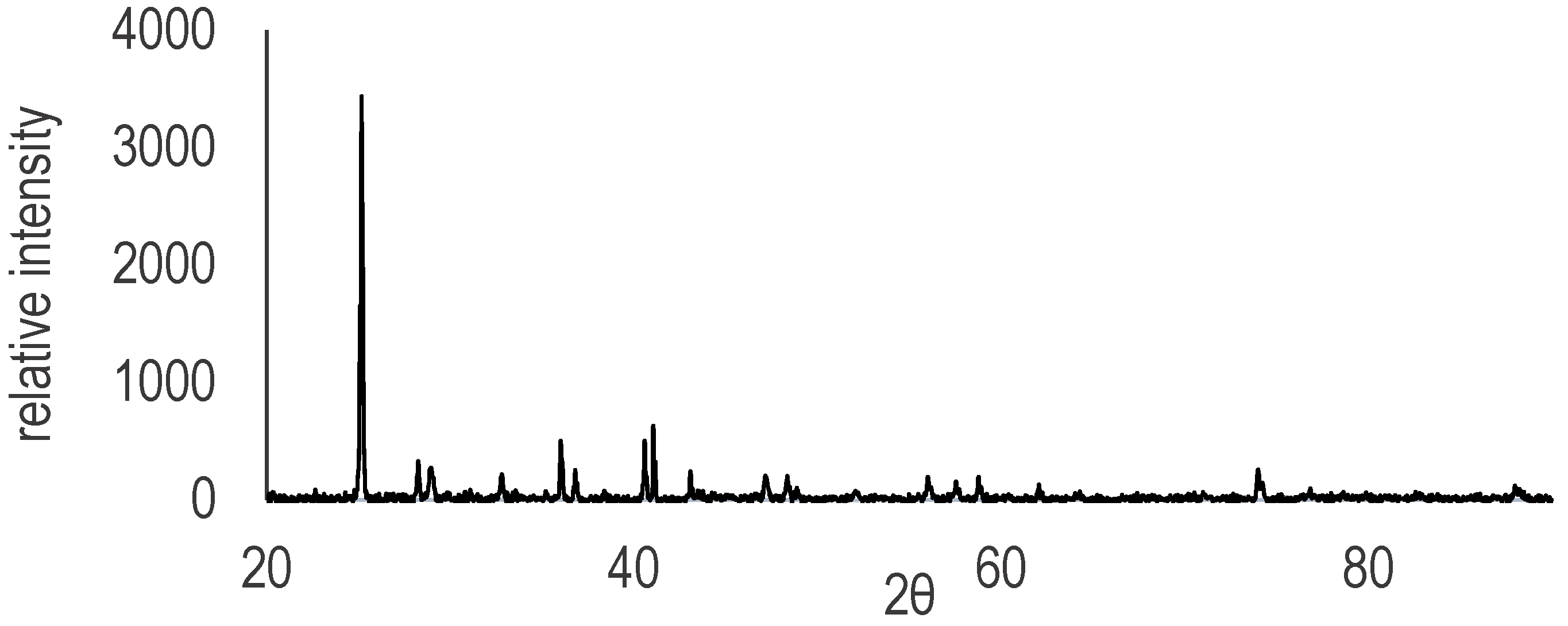
Appendix B
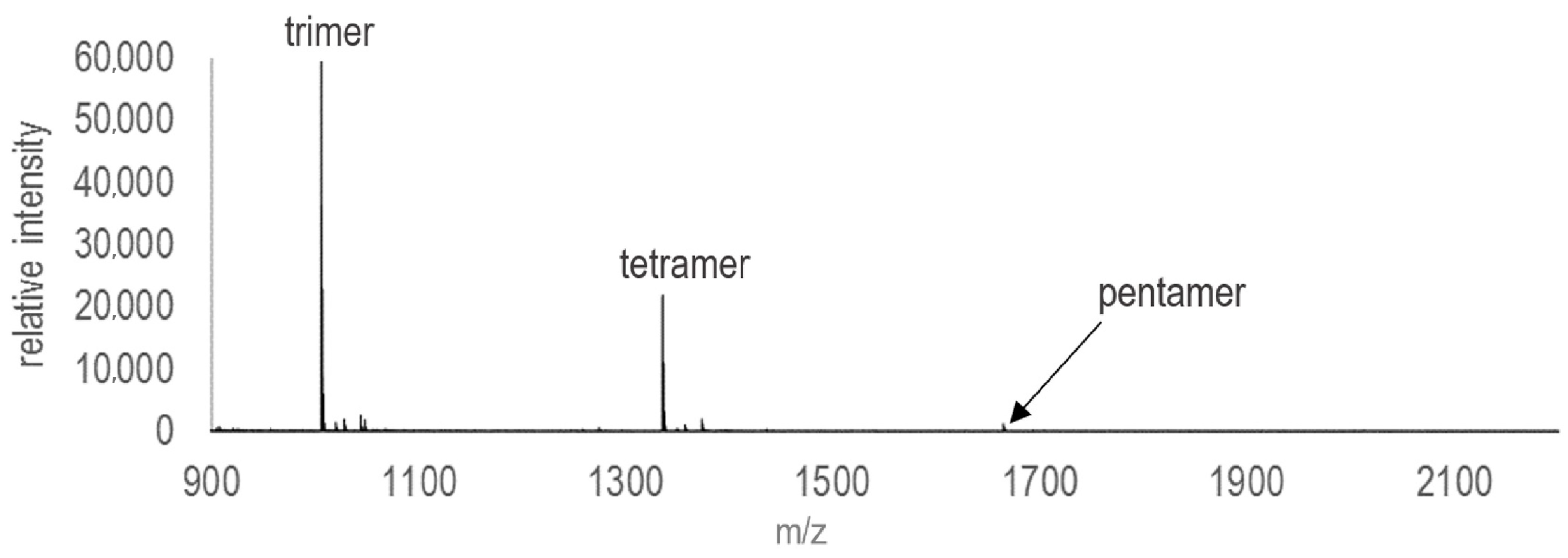
References
- Cafferty, B.J.; Hud, N.V. Abiotic Synthesis of RNA in Water: A Common Goal of Prebiotic Chemistry and Bottom-up Synthetic Biology. Curr. Opin. Chem. Biol. 2014, 22, 146–157. [Google Scholar] [CrossRef]
- Ying, J.; Ding, R.; Liu, Y.; Zhao, Y. Prebiotic Chemistry in Aqueous Environment: A Review of Peptide Synthesis and Its Relationship with Genetic Code. Chin. J. Chem. 2021, 39, 2264–2272. [Google Scholar] [CrossRef]
- Tolstoguzov, V. Why Were Polysaccharides Necessary? Orig. Life Evol. Biosph. 2004, 34, 571–597. [Google Scholar] [CrossRef]
- Shock, E.L. Stability of Peptides in High-Temperature Aqueous Solutions. Geochim. Cosmochim. Acta 1992, 56, 3481–3491. [Google Scholar] [CrossRef]
- Hulshof, J.; Ponnamperuma, C. Prebiotic Condensation Reactions in an Aqueous Medium: A Review of Condensing Agents. Orig. Life 1976, 7, 197–224. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.-F. Adsorption and Polymerization of Amino Acids on Mineral Surfaces: A Review. Orig. Life Evol. Biosph. 2008, 38, 211–242. [Google Scholar] [CrossRef]
- Nelson, D.L.; Cox, M.M. Lehninger Principles of Biochemistry, 4th ed.; W.H. Freeman: New York, NY, USA, 2005; ISBN 978-0-7167-4339-2. [Google Scholar]
- Ross, D.; Deamer, D. Dry/Wet Cycling and the Thermodynamics and Kinetics of Prebiotic Polymer Synthesis. Life 2016, 6, 28. [Google Scholar] [CrossRef]
- Ferris, J.P.; Joshi, P.C.; Wang, K.-J.; Miyakawa, S.; Huang, W. Catalysis in Prebiotic Chemistry: Application to the Synthesis of RNA Oligomers. Adv. Space Res. 2004, 33, 100–105. [Google Scholar] [CrossRef]
- Weimann, B.J.; Lohrmann, R.; Orgel, L.E.; Schneider-Bernloehr, H.; Sulston, J.E. Template-Directed Synthesis with Adenosine-5′-Phosphorimidazolide. Science 1968, 161, 387. [Google Scholar] [CrossRef]
- Shabarova, Z.A. Synthetic Nucleotide-Peptides. In Progress in Nucleic Acid Research and Molecular Biology; Elsevier: Amsterdam, The Netherlands, 1970; Volume 10, pp. 145–182. ISBN 978-0-12-540010-7. [Google Scholar]
- Costanzo, G.; Pino, S.; Ciciriello, F.; Di Mauro, E. Generation of Long RNA Chains in Water. J. Biol. Chem. 2009, 284, 33206–33216. [Google Scholar] [CrossRef]
- Prabahar, K.J.; Ferris, J.P. Adenine Derivatives as Phosphate-Activating Groups for the Regioselective Formation of 3′,5′-Linked Oligoadenylates on Montmorillonite: Possible Phosphate-Activating Groups for the Prebiotic Synthesis of RNA. J. Am. Chem. Soc. 1997, 119, 4330–4337. [Google Scholar] [CrossRef]
- Sawai, H. Catalysis of Internucleotide Bond Formation by Divalent Metal Ions. J. Am. Chem. Soc. 1976, 98, 7037–7039. [Google Scholar] [CrossRef]
- Ferris, J.P.; Ertem, G. Montmorillonite Catalysis of RNA Oligomer Formation in Aqueous Solution. A Model for the Prebiotic Formation of RNA. J. Am. Chem. Soc. 1993, 115, 12270–12275. [Google Scholar] [CrossRef]
- de Castro Silva, F.; Lima, L.C.B.; Silva-Filho, E.C.; Fonseca, M.G.; Lambert, J.-F.; Jaber, M. A Comparative Study of Alanine Adsorption and Condensation to Peptides in Two Clay Minerals. Appl. Clay Sci. 2020, 192, 105617. [Google Scholar] [CrossRef]
- Ferris, J.P. Montmorillonite-Catalysed Formation of RNA Oligomers: The Possible Role of Catalysis in the Origins of Life. Philos. Trans. R. Soc. B Biol. Sci. 2006, 361, 1777–1786. [Google Scholar] [CrossRef]
- Joshi, P.C.; Aldersley, M.F.; Delano, J.W.; Ferris, J.P. Mechanism of Montmorillonite Catalysis in the Formation of RNA Oligomers. J. Am. Chem. Soc. 2009, 131, 13369–13374. [Google Scholar] [CrossRef] [PubMed]
- Burcar, B.T.; Jawed, M.; Shah, H.; McGown, L.B. In Situ Imidazole Activation of Ribonucleotides for Abiotic RNA Oligomerization Reactions. Orig. Life Evol. Biosph. 2015, 45, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Coari, K.M.; Martin, R.C.; Jain, K.; McGown, L.B. Nucleotide Selectivity in Abiotic RNA Polymerization Reactions. Orig. Life Evol. Biosph. 2017, 47, 305–321. [Google Scholar] [CrossRef] [PubMed]
- Banin, A. Quantitative Ion Exchange Process for Clay. Patent No. 3725528, 3 April 1973. [Google Scholar]
- Banin, A.; Lawless, J.G.; Mazzurco, J.; Church, F.M.; Margulies, L.; Orenberg, J.B. PH Profile of the Adsorption of Nucleotides onto Montmorillonite: II. Adsorption and Desorption of 5′-AMP in Iron-Calcium Montmorillonite Systems. Orig. Life Evol. Biosph. 1985, 15, 89–101. [Google Scholar] [CrossRef]
- Hazen, R.M.; Papineau, D.; Bleeker, W.; Downs, R.T.; Ferry, J.M.; McCoy, T.J.; Sverjensky, D.A.; Yang, H. Mineral Evolution. Am. Mineral. 2008, 93, 1693–1720. [Google Scholar] [CrossRef]
- Moberly, R. Amorphous Marine Muds from Tropically Weathered Basalt. Am. J. Sci. 1963, 261, 767–772. [Google Scholar] [CrossRef]
- Bischoff, J.L.; Dickson, F.W. Seawater-Basalt Interaction at 200 °C and 500 Bars: Implications for Origin of Sea-Floor Heavy-Metal Deposits and Regulation of Seawater Chemistry. Earth Planet. Sci. Lett. 1975, 25, 385–397. [Google Scholar] [CrossRef]
- Bain, D.C.; Russell, J.D. Swelling Minerals in a Basalt and Its Weathering Products from Morvern, Scotland: I. Interstratified Montmorillonite-Vermiculite-Illite. Clay Miner. 1980, 15, 445–451. [Google Scholar] [CrossRef]
- Zolotov, M.Y.; Mironenko, M.V. Formation and Fate of Phyllosilicates on the Surface of Mars: Geochemical Modeling of Aqueous Weathering. In Proceedings of the Seventh International Conference on Mars, Pasadena, CA, USA, 9–13 July 2007; Volume 3365. [Google Scholar]
- Schirrmeister, L.; Sto¨rr, M. The Weathering of Basaltic Rocks in Burundi and Vietnam. CATENA 1994, 21, 243–256. [Google Scholar] [CrossRef]
- Cavosie, A.J.; Valley, J.W.; Wilde, S.A. The Oldest Terrestrial Mineral Record. In Earth’s Oldest Rocks; Elsevier: Amsterdam, The Netherlands, 2019; pp. 255–278. ISBN 978-0-444-63901-1. [Google Scholar]
- Korenaga, J. Hadean Geodynamics and the Nature of Early Continental Crust. Precambrian Res. 2021, 359, 106178. [Google Scholar] [CrossRef]
- Watson, E.B.; Harrison, T.M. Zircon Thermometer Reveals Minimum Melting Conditions on Earliest Earth. Science 2005, 308, 841–844. [Google Scholar] [CrossRef]
- Trail, D.; Tailby, N.; Wang, Y.; Mark Harrison, T.; Boehnke, P. Aluminum in Zircon as Evidence for Peraluminous and Metaluminous Melts from the H Adean to Present. Geochem. Geophys. Geosyst. 2017, 18, 1580–1593. [Google Scholar] [CrossRef]
- Hazen, R.M.; Sverjensky, D.A.; Azzolini, D.; Bish, D.L.; Elmore, S.C.; Hinnov, L.; Milliken, R.E. Clay Mineral Evolution. Am. Mineral. 2013, 98, 2007–2029. [Google Scholar] [CrossRef]
- Bernal, J.D. The Physical Basis of Life. Proc. Phys. Soc. 1949, 62, 537–558. [Google Scholar] [CrossRef]
- Pearce, B.K.D.; Tupper, A.S.; Pudritz, R.E.; Higgs, P.G. Constraining the Time Interval for the Origin of Life on Earth. Astrobiology 2018, 18, 343–364. [Google Scholar] [CrossRef] [PubMed]
- Demolon, A.; Barbier, G. Conditions de formation et constitution du complexe argilo-humique des sols. C. R. Hebd. Sances Acadmie Sci. 1929, 188, 654–656. [Google Scholar]
- Ensminger, L.E.; Gieseking, J.E. The Adsorption of Proteins by Montmorillonite Clays. Soil Sci. 1939, 48, 467–474. [Google Scholar] [CrossRef]
- Ensminger, L.E.; Gieseking, J.E. Resistance of Clay Adsorbed Proteins to Proteolytic Hydrolysis. Soil Sci. 1942, 53, 205–210. [Google Scholar] [CrossRef]
- Ensminger, L.E.; Gieseking, J.E. The Adsorption of Proteins by Montmorillonite Clays and Its Effect on Base-Exchange Capacity. Soil Sci. 1941, 51, 125–132. [Google Scholar] [CrossRef]
- Cairns-Smith, A.G. Genetic Takeover and the Mineral Origins of Life; Cambridge University Press: Cambridge, New York, NY, USA, 1982; ISBN 978-0-521-23312-5. [Google Scholar]
- Cairns-Smith, A.G. The Origin of Life and the Nature of the Primitive Gene. J. Theor. Biol. 1966, 10, 53–88. [Google Scholar] [CrossRef] [PubMed]
- Lailach, G.E.; Thomas, T.D.; Brindley, G.W. Absorption of Pyrimidines, Purines, and Nucleosides by Li, Na, Mg, and Ca Montmorillonite (Clay Organic Studies XII). Clays Clay Miner. 1968, 16, 258–293. [Google Scholar] [CrossRef]
- Lailach, G.E. Absorption of Pyrimidines, Purines, and Nucleosides by Co-, Ni-, Cu-, and Fe(III)-Montmorillonite (Clay-Organic Studies XIII). Clays Clay Miner. 1968, 16, 295–301. [Google Scholar] [CrossRef]
- Ferris, J.P.; Huang, C.-H.; Hagan, W.J. Clays as Prototypical Enzymes for the Prebiological Formation of Phosphate Esters. Orig. Life Evol. Biosph. 1986, 16, 473–474. [Google Scholar] [CrossRef]
- Ferris, J.P.; Ertem, G.; Agarwal, V. Mineral Catalysis of the Formation of Dimers of 5′-AMP in Aqueous Solution: The Possible Role of Montmorillonite Clays in the Prebiotic Synthesis of RNA. Orig. Life Evol. Biosph. 1989, 19, 165–178. [Google Scholar] [CrossRef] [PubMed]
- McCollom, T.M.; Ritter, G.; Simoneit, B.R.T. Lipid Synthesis under Hydrothermal Conditions by Fischer-Tropsch-Type Reactions. Orig. Life Evol. Biosph. 1999, 29, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Hanczyc, M.M. Experimental Models of Primitive Cellular Compartments: Encapsulation, Growth, and Division. Science 2003, 302, 618–622. [Google Scholar] [CrossRef] [PubMed]
- Ferris, J.P.; Hill, A.R.; Liu, R.; Orgel, L.E. Synthesis of Long Prebiotic Oligomers on Mineral Surfaces. Nature 1996, 381, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Nisbet, E.G. Origin of Life: RNA and Hot-Water Springs. Nature 1986, 322, 206. [Google Scholar] [CrossRef]
- Smith, J.V. Biochemical Evolution. I. Polymerization on Internal, Organophilic Silica Surfaces of Dealuminated Zeolites and Feldspars. Proc. Natl. Acad. Sci. USA 1998, 95, 3370–3375. [Google Scholar] [CrossRef]
- Burcar, B.T. Astrobiological Exploration of RNA Oligomerization and Reaction Product Characterization for Prebiotically Based Chemical Reactions. Rensselaer Polytechnic Institute: Troy, NY, USA, 2015. [Google Scholar]
- Pedreira-Segade, U.; Feuillie, C.; Pelletier, M.; Michot, L.J.; Daniel, I. Adsorption of Nucleotides onto Ferromagnesian Phyllosilicates: Significance for the Origin of Life. Geochim. Cosmochim. Acta 2016, 176, 81–95. [Google Scholar] [CrossRef]
- Biondi, E.; Furukawa, Y.; Kawai, J.; Benner, S.A. Adsorption of RNA on Mineral Surfaces and Mineral Precipitates. Beilstein J. Org. Chem. 2017, 13, 393–404. [Google Scholar] [CrossRef]
- Jerome, C.A.; Kim, H.-J.; Mojzsis, S.J.; Benner, S.A.; Biondi, E. Catalytic Synthesis of Polyribonucleic Acid on Prebiotic Rock Glasses. Astrobiology 2022, 22, 629–636. [Google Scholar] [CrossRef]
- Miyakawa, S.; Joshi, P.C.; Gaffey, M.J.; Gonzalez-Toril, E.; Hyland, C.; Ross, T.; Rybij, K.; Ferris, J.P. Studies in the Mineral and Salt-Catalyzed Formation of RNA Oligomers. Orig. Life Evol. Biosph. 2006, 36, 343–361. [Google Scholar] [CrossRef]
- Burcar, B.T.; Barge, L.M.; Trail, D.; Watson, E.B.; Russell, M.J.; McGown, L.B. RNA Oligomerization in Laboratory Analogues of Alkaline Hydrothermal Vent Systems. Astrobiology 2015, 15, 509–522. [Google Scholar] [CrossRef]
- Pfeffer, S.; Lagos-Quintana, M.; Tuschl, T. Clonin of Small RNA Molecules. Curr. Protoc. Mol. Biol. 2005, 72, 26.4.1–26.4.18. [Google Scholar] [CrossRef]
- Evangelou, V.P. Pyrite Chemistry: The Key for Abatement of Acid Mine Drainage. In Acidic Mining Lakes; Geller, W., Klapper, H., Salomons, W., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; pp. 197–222. ISBN 978-3-642-71956-1. [Google Scholar]
- Castleberry, C.M.; Chou, C.-W.; Limbach, P.A. Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry of Oligonucleotides. In Current Protocols in Nucleic Acid Chemistry; Beaucage, S.L., Bergstrom, D.E., Herdewijn, P., Matsuda, A., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; pp. 10.1.1–10.1.21. ISBN 978-0-471-14270-6. [Google Scholar]
- Zagorevskii, D.V.; Aldersley, M.F.; Ferris, J.P. MALDI Analysis of Oligonucleotides Directly from Montmorillonite. J. Am. Soc. Mass Spectrom. 2006, 17, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, L.M.; Dong, Y.; Joshi, P.C.; Aldersley, M.F.; Ferris, J.P.; McGown, L.B. Signal Enhancement of Abiotically-Synthesized RNA Oligonucleotides and Other Biopolymers Using Unmodified Fused Silica in MALDI-MS. J. Am. Soc. Mass Spectrom. 2011, 22, 1100–1104. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, G.; Saladino, R.; Botta, G.; Giorgi, A.; Scipioni, A.; Pino, S.; Di Mauro, E. Generation of RNA Molecules by a Base-Catalysed Click-Like Reaction. ChemBioChem 2012, 13, 999–1008. [Google Scholar] [CrossRef]
- Burcar, B.T.; Cassidy, L.M.; Moriarty, E.M.; Joshi, P.C.; Coari, K.M.; McGown, L.B. Potential Pitfalls in MALDI-TOF MS Analysis of Abiotically Synthesized RNA Oligonucleotides. Orig. Life Evol. Biosph. 2013, 43, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Sawai, H.; Orgel, L.E. Oligonucleotide Synthesis Catalyzed by the Zn2+ Ion. J. Am. Chem. Soc. 1975, 97, 2532–3533. [Google Scholar] [CrossRef]
- Sleeper, H.L.; Lohrmann, R.; Orgel, L.E. Template-Directed Synthesis of Oligoadenylates Catalyzed by Pb2+ Ions. J. Mol. Evol. 1979, 13, 203–214. [Google Scholar] [CrossRef]
- Keller, W.D. Environmental Aspects of Clay Minerals. SEPM J. Sediment. Res. 1970, 40, 788–813. [Google Scholar] [CrossRef]
- Lockhart, N.C. Electrical Properties and the Surface Characteristics and Structure of Clays. II. Kaolinite—A Nonswelling Clay. J. Colloid Interface Sci. 1980, 74, 520–529. [Google Scholar] [CrossRef]
- Ulian, G.; Moro, D.; Valdrè, G. Water Adsorption Behaviour on (001) Pyrophyllite Surface from Ab Initio Density Functional Theory Simulations. Appl. Clay Sci. 2021, 212, 106221. [Google Scholar] [CrossRef]
- Marantos, I.; Christidis, G.E.; Ulmanu, M. Zeolite Formation and Deposits. In Handbook of Natural Zeolites; Bentham Science: Oak Park, IL, USA, 2012; pp. 28–51. ISBN 978-1-60805-446-6. [Google Scholar]
- Smith, J.V.; Arnold, F.P.; Parsons, I.; Lee, M.R. Biochemical Evolution III: Polymerization on Organophilic Silica-Rich Surfaces, Crystal–Chemical Modeling, Formation of First Cells, and Geological Clues. Proc. Natl. Acad. Sci. USA 1999, 96, 3479–3485. [Google Scholar] [CrossRef] [PubMed]
- Wachtershauser, G. Before Enzymes and Templates: Theory of Surface Metabolism. Microbiol. Rev. 1988, 52, 33. [Google Scholar] [CrossRef] [PubMed]
- Wächtershäuser, G. Pyrite Formation, the First Energy Source for Life: A Hypothesis. Syst. Appl. Microbiol. 1988, 10, 207–210. [Google Scholar] [CrossRef]
- Bebié, J.; Schoonen, M.A.A. Pyrite and Phosphate in Anoxia and an Origin-of-Life Hypothesis. Earth Planet. Sci. Lett. 1999, 171, 1–5. [Google Scholar] [CrossRef]
- Mizuuchi, R.; Blokhuis, A.; Vincent, L.; Nghe, P.; Lehman, N.; Baum, D. Mineral Surfaces Select for Longer RNA Molecules. Chem. Commun. 2019, 55, 2090–2093. [Google Scholar] [CrossRef]
- Sawai, H. Oligonucleotide Formation Catalyzed by Divalent Metal Ions. The Uniqueness of the Ribosyl System. J. Mol. Evol. 1988, 27, 181–186. [Google Scholar] [CrossRef]
- Kanavarioti, A.; Monnard, P.-A.; Deamer, D.W. Eutectic Phases in Ice Facilitate Nonenzymatic Nucleic Acid Synthesis. Astrobiology 2001, 1, 271–281. [Google Scholar] [CrossRef]
| Mineral Name | Mineral Type | General Formula |
|---|---|---|
| Montmorillonite (Volclay SPV-200) | Clay (Phyllosilicate) | (Na, Ca)0.33(Al, Mg)2(Si4O10)(OH)2·nH2O |
| Pyrophyllite | Clay (Phyllosilicate) | Al2Si4O10(OH)2 |
| Kaolinite | Clay (Phyllosilicate) | Al2Si2O5(OH)4 |
| Nontronite | Clay (Phyllosilicate) | (CaO0.5, Na)0.3Fe3+2(Si, Al)4O10(OH)2·nH2O |
| Pyrite | Sulfide | FeS2 |
| Pyrrhotite | Sulfide | Fe1−xS (x = 0 to 0.2) |
| Galena | Sulfide | PbS |
| Natrolite | Zeolite | Na2Al2Si3O10·2H2O |
| Stilbite | Zeolite | NaCa4(Si27Al9)O72·28(H2O) |
| Faujasite (synthetic) | Zeolite | (Na2, Ca, Mg)3.5[Al7Si17O48]·32(H2O) |
| Chabazite | Zeolite | (Ca, K2, Na2)2[Al2Si4O12]2·12H2O |
| ZSM-5 (synthetic) | Zeolite | NanAlnSi96–nO192·16H2O (0 < n < 27) |
| Corundum | Oxide | Al2O3 |
| Hematite | Oxide | Fe2O3 |
| Magnetite | Oxide | Fe2+Fe3+2O4 |
| Anorthite | Feldspar (Tectosilicate) | CaAl2Si2O8 |
| Albite | Feldspar (Tectosilicate) | NaAlSi3O8 |
| Orthoclase | Feldspar (Tectosilicate) | KAlSi3O8 |
| Quartz | Tectosilicate | SiO2 |
| Olivine | Nesosilicate | (Mg, Fe)2SiO4 |
| Black Smoker Chimney | (Polymineralic) | N/A |
| Apatite | Phosphate | Ca5(PO4)3(F, Cl, OH) |
| Diopside | Pyroxene (Inosilicate) | MgCaSi2O6 |
| Chlorite | Phyllosilicate | (Fe, Mg, Al)6(Si, Al)4O10(OH)8 |
| Brucite | Hydroxide | Mg(OH)2 |
| Silica (synthetic) | (Glass) | SiO2 |
| Instrument Parameter | Value |
|---|---|
| Detection Mode | Reflectron |
| Ionization Mode | Negative |
| Ion Source 1 | 19 kV |
| Ion Source 2 | 16.7 kV |
| Lens | 8.4 kV |
| Pulsed Ion Extraction | 140 ns |
| Relative Laser Intensity | 75–85% |
| Sets of Shots Per Sample | 5 |
| Shots Per Set | 1000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riggi, V.S.; Watson, E.B.; Steele, A.; Rogers, K.L. Mineral-Mediated Oligoribonucleotide Condensation: Broadening the Scope of Prebiotic Possibilities on the Early Earth. Life 2023, 13, 1899. https://doi.org/10.3390/life13091899
Riggi VS, Watson EB, Steele A, Rogers KL. Mineral-Mediated Oligoribonucleotide Condensation: Broadening the Scope of Prebiotic Possibilities on the Early Earth. Life. 2023; 13(9):1899. https://doi.org/10.3390/life13091899
Chicago/Turabian StyleRiggi, Vincent S., E. Bruce Watson, Andrew Steele, and Karyn L. Rogers. 2023. "Mineral-Mediated Oligoribonucleotide Condensation: Broadening the Scope of Prebiotic Possibilities on the Early Earth" Life 13, no. 9: 1899. https://doi.org/10.3390/life13091899
APA StyleRiggi, V. S., Watson, E. B., Steele, A., & Rogers, K. L. (2023). Mineral-Mediated Oligoribonucleotide Condensation: Broadening the Scope of Prebiotic Possibilities on the Early Earth. Life, 13(9), 1899. https://doi.org/10.3390/life13091899






