Development of In Situ Product Recovery (ISPR) System Using Amberlite IRA67 for Enhanced Biosynthesis of Hyaluronic Acid by Streptococcus zooepidemicus
Abstract
1. Introduction
2. Materials and Methods
2.1. Glycerol Stock of Streptococcus zooepidemicus
2.2. Ion-Exchange Resins and HA Adsorption Capacity
2.3. Cultivation of S. zooepidemicus and Experimental Design
2.3.1. Inoculum
2.3.2. Batch Fermentation of S. zooepidemicus
2.4. Analytical Methods
2.4.1. Cell Concentration
2.4.2. HA Concentration
2.4.3. Lactic Acid Assay
- V = final volume [mL];
- MW = molecular weight of L-lactic acid [g/mol];
- ε = extinction coefficient of NADH at 340 nm = 6300 [l × mol−1 × cm−1];
- d = light path [cm];
- v = sample volume [mL].
2.4.4. Glucose Assay
2.4.5. Scanning Electron Microscope
2.4.6. Viscosity Analysis
2.5. Statistical Analysis
3. Results and Discussion
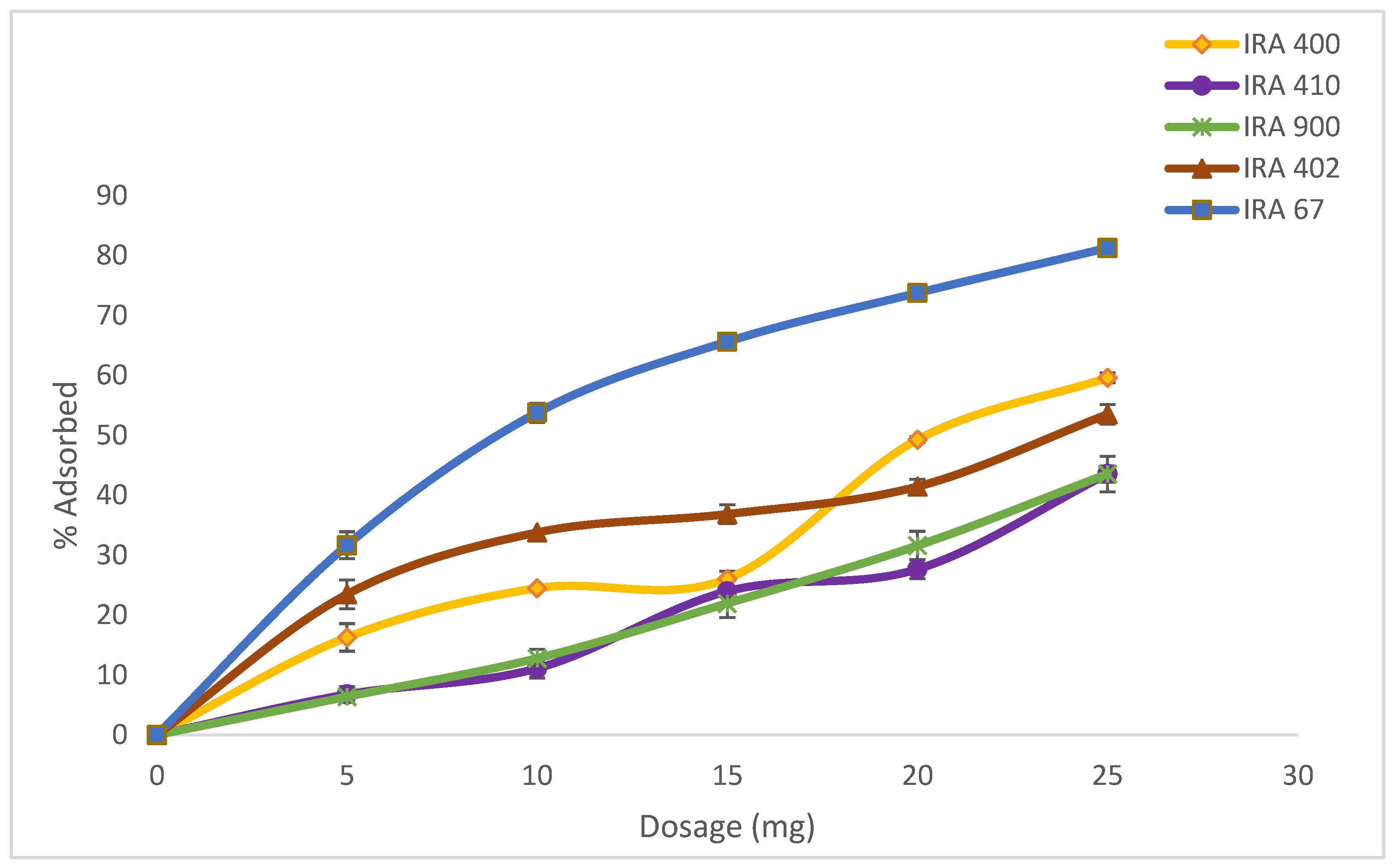
3.1. Ion-Exchange Resins Adsorption Capacity of HA
3.2. Effect of Different Dosages of Amberlite IRA67 on HA Production by S. zooepidemicus
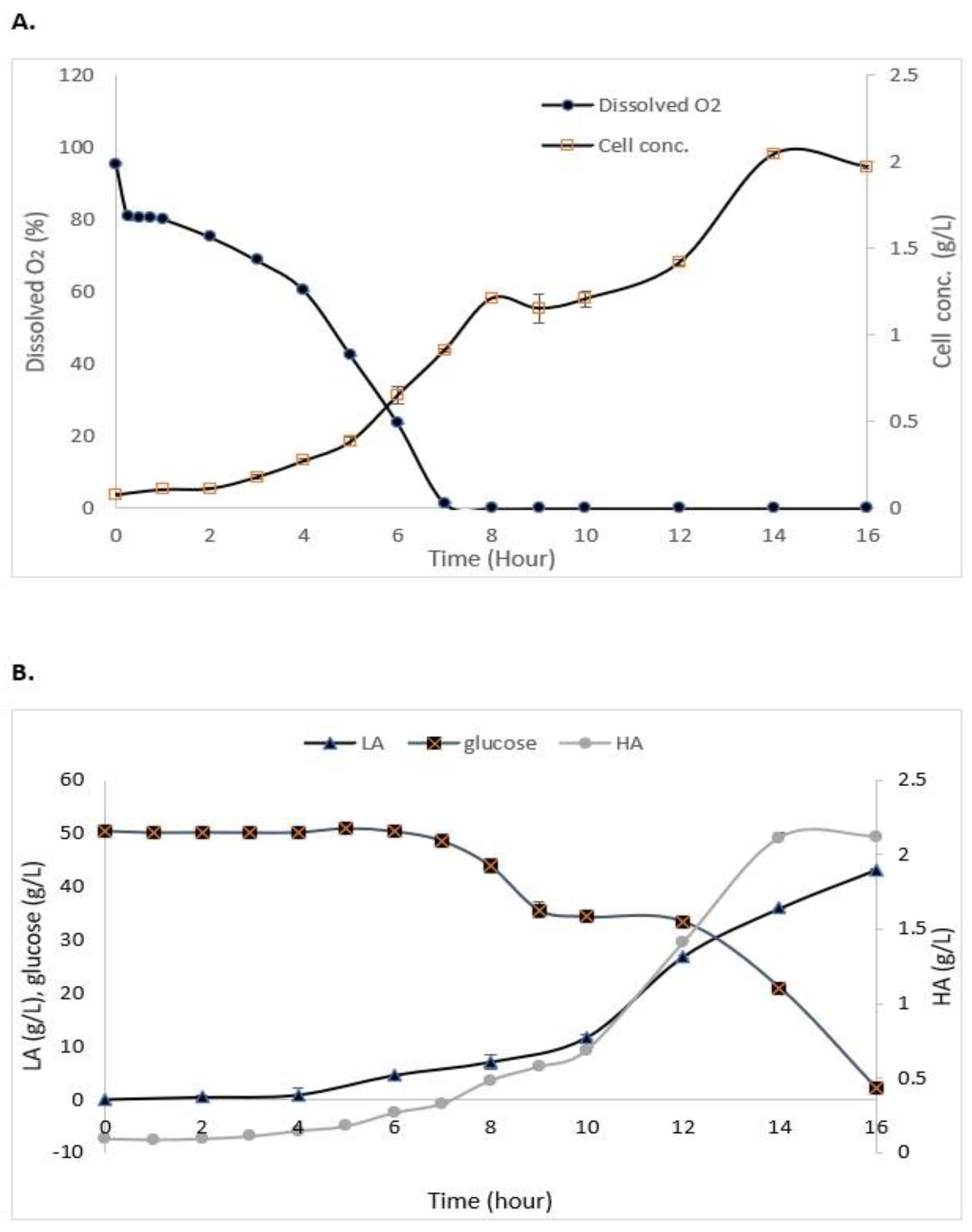
3.3. Batch Fermentation of S. zooepidemicus
3.4. Scanning Electron Microscope of Fermentation Broth and the IRA67 Resin
3.5. Viscosity Analysis on Fermentation Media
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Juncan, A.M.; Moisă, D.G.; Santini, A.; Morgovan, C.; Rus, L.L.; Vonica-țincu, A.L.; Loghin, F. Advantages of Hyaluronic Acid and Its Combination with Other Bioactive Ingredients in Cosmeceuticals. Molecules 2021, 26, 4429. [Google Scholar] [CrossRef] [PubMed]
- Harrer, D.; Sanchez Armengol, E.; Friedl, J.D.; Jalil, A.; Jelkmann, M.; Leichner, C.; Laffleur, F. Is Hyaluronic Acid the Perfect Excipient for the Pharmaceutical Need? Int. J. Pharm. 2021, 601, 120589. [Google Scholar] [CrossRef]
- Lierova, A.; Kasparova, J.; Filipova, A.; Cizkova, J.; Pekarova, L.; Korecka, L.; Mannova, N.; Bilkova, Z.; Sinkorova, Z. Hyaluronic Acid: Known for Almost a Century, but Still in Vogue. Pharmaceutics 2022, 14, 838. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Ma, Y.; Huang, Y.; Li, S.; Xu, H.; Su, E. Current Advances in the Biosynthesis of Hyaluronic Acid with Variable Molecular Weights. Carbohydr. Polym. 2021, 269, 118320. [Google Scholar] [CrossRef] [PubMed]
- Marinho, A.; Nunes, C.; Reis, S. Hyaluronic Acid: A Key Ingredient in the Therapy of Inflammation. Biomolecules 2021, 11, 1518. [Google Scholar] [CrossRef]
- Yasin, A.; Ren, Y.; Li, J.; Sheng, Y.; Cao, C.; Zhang, K. Advances in Hyaluronic Acid for Biomedical Applications. Front. Bioeng. Biotechnol. 2022, 10, 910290. [Google Scholar] [CrossRef]
- Hashimoto, M.; Maeda, K. New Functions of Low-Molecular-Weight Hyaluronic Acid on Epidermis Filaggrin Production and Degradation. Cosmetics 2021, 8, 118. [Google Scholar] [CrossRef]
- Rodriguez-Marquez, C.D.; Arteaga-Marin, S.; Rivas-Sánchez, A.; Autrique-Hernández, R.; Castro-Muñoz, R. A Review on Current Strategies for Extraction and Purification of Hyaluronic Acid. Int. J. Mol. Sci. 2022, 23, 6038. [Google Scholar] [CrossRef]
- Yao, Z.Y.; Qin, J.; Gong, J.S.; Ye, Y.H.; Qian, J.Y.; Li, H.; Xu, Z.H.; Shi, J.S. Versatile Strategies for Bioproduction of Hyaluronic Acid Driven by Synthetic Biology. Carbohydr. Polym. 2021, 264, 118015. [Google Scholar] [CrossRef]
- Liu, L.; Yang, H.; Zhang, D.; Du, G.; Chen, J.; Wang, M.; Sun, J. Enhancement of Hyaluronic Acid Production by Batch Culture of Streptococcus zooepidemicus with N-Dodecane as an Oxygen Vector. J. Microbiol. Biotechnol. 2009, 19, 596–603. [Google Scholar] [CrossRef]
- Sze, J.H.; Brownlie, J.C.; Love, C.A. Biotechnological Production of Hyaluronic Acid: A Mini Review. 3 Biotech 2016, 6, 67. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, Y.; Li, Z.; Ren, Y.; Zhao, Y.; Zhao, G. Efficient Production of High-Molecular-Weight Hyaluronic Acid with a Two-Stage Fermentation. RSC Adv. 2018, 8, 36167–36171. [Google Scholar] [CrossRef] [PubMed]
- Pan, N.C.; Vignoli, J.A.; Baldo, C.; Pereira, H.C.B.; da Silva, R.S.d.S.F.; Celligoi, M.A.P.C. Effect of Fermentation Conditions on the Production of Hyaluronic Acid by Streptococcus zooepidemicus ATCC 39920. Acta Sci.-Biol. Sci. 2015, 37, 411–417. [Google Scholar] [CrossRef]
- Mohan, N.; Tadi, S.R.R.; Pavan, S.S.; Sivaprakasam, S. Deciphering the Role of Dissolved Oxygen and N-Acetyl Glucosamine in Governing Higher Molecular Weight Hyaluronic Acid Synthesis in Streptococcus zooepidemicus Cell Factory. Appl. Microbiol. Biotechnol. 2020, 104, 3349–3365. [Google Scholar] [CrossRef]
- Hasegawa, S.; Nagatsuru, M.; Shibutani, M.; Yamamoto, S.; Hasebe, S. Productivity of Concentrated Hyaluronic Acid Using a Maxblend Fermentor. J. Biosci. Bioeng. 1999, 88, 68–71. [Google Scholar] [CrossRef]
- Lai, Z.-W.; Rahim, R.A.; Ariff, A.B.; Mohamad, R. Biosynthesis of High Molecular Weight Hyaluronic Acid by Streptococcus zooepidemicus Using Oxygen Vector and Optimum Impeller Tip Speed. J. Biosci. Bioeng. 2012, 114, 286–291. [Google Scholar] [CrossRef]
- Ren, X. Application of Hydrocarbon and Perfluorocarbon Oxygen Vectors to Enhance Heterologous Production of Hyaluronic Acid in Engineered Bacillus Subtilis. Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2017. [Google Scholar]
- Gao, M.-T.; Shimamura, T.; Ishida, N.; Takahashi, H. PH-Uncontrolled Lactic Acid Fermentation with Activated Carbon as an Adsorbent. Enzym. Microb. Technol. 2011, 48, 526–530. [Google Scholar] [CrossRef]
- Othman, M.; Ariff, A.B.; Rios-Solis, L.; Halim, M. Extractive Fermentation of Lactic Acid in Lactic Acid Bacteria Cultivation: A Review. Front. Microbiol. 2017, 8, 2285. [Google Scholar] [CrossRef]
- Othman, M.; Ariff, A.B.; Kapri, M.R.; Rios-Solis, L.; Halim, M. Growth Enhancement of Probiotic Pediococcus Acidilactici by Extractive Fermentation of Lactic Acid Exploiting Anion-Exchange Resin. Front. Microbiol. 2018, 9, 2554. [Google Scholar] [CrossRef]
- Pradhan, N.; Rene, E.R.; Lens, P.N.L.; Dipasquale, L.; D’Ippolito, G.; Fontana, A.; Panico, A.; Esposito, G. Adsorption Behaviour of Lactic Acid on Granular Activated Carbon and Anionic Resins: Thermodynamics, Isotherms and Kinetic Studies. Energies 2017, 10, 665. [Google Scholar] [CrossRef]
- Gamaethiralalage, J.G.; Singh, K.; Sahin, S.; Yoon, J.; Elimelech, M.; Suss, M.E.; Liang, P.; Biesheuvel, P.M.; Zornitta, R.L.; De Smet, L. Recent Advances in Ion Selectivity with Capacitive Deionization. Energy Environ. Sci. 2021, 14, 1095–1120. [Google Scholar] [CrossRef]
- Gluszcz, P.; Jamroz, T.; Sencio, B.; Ledakowicz, S. Equilibrium and Dynamic Investigations of Organic Acids Adsorption onto Ion-Exchange Resins. Bioprocess Biosyst. Eng. 2004, 26, 185–190. [Google Scholar] [CrossRef]
- Choi, S.; Choi, W.; Kim, S.; Lee, S.-Y.; Noh, I.; Kim, C.-W. Purification and Biocompatibility of Fermented Hyaluronic Acid for Its Applications to Biomaterials. Biomater. Res. 2014, 18, 6. [Google Scholar] [CrossRef] [PubMed]
- Han, H.Y.; Jang, S.H.; Kim, E.C.; Park, J.K.; Han, Y.J.; Lee, C.; Park, H.S.; Kim, Y.C.; Park, H.J. Microorganism Producing Hyaluronic Acid and Purification Method of Hyaluronic Acid. WIPO (PCT). WO2004016771A1, 26 February 2004. [Google Scholar]
- Luongo, V.; Palma, A.; Rene, E.R.; Fontana, A.; Pirozzi, F.; Esposito, G.; Lens, P.N.L. Lactic Acid Recovery from a Model of Thermotoga Neapolitana Fermentation Broth Using Ion Exchange Resins in Batch and Fixed-Bed Reactors. Sep. Sci. Technol. 2019, 54, 1008–1025. [Google Scholar] [CrossRef]
- Bitter, T.; Muir, H.M. A Modified Uronic Acid Carbazole Reaction. Anal. Biochem. 1962, 4, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.S.; Ramanan, R.N.; Ling, T.; Shuhaimi, M.; Ariff, A. Enhanced Production of Periplasmic Interferon Alpha-2b by Escherichia coli Using Ion-Exchange Resin for In Situ Removal of Acetate in the Culture. Biochem. Eng. J. 2011, 58–59, 124–132. [Google Scholar] [CrossRef]
- Bishai, M.; De, S.; Adhikari, B.; Banerjee, R. A Platform Technology of Recovery of Lactic Acid from a Fermentation Broth of Novel Substrate Zizyphus oenophlia. 3 Biotech 2015, 5, 455–463. [Google Scholar] [CrossRef]
- Kołodyńska, D. Polyacrylate Anion Exchangers in Sorption of Heavy Metal Ions with the Biodegradable Complexing Agent. Chem. Eng. J. 2009, 150, 280–288. [Google Scholar] [CrossRef]
- Hashim, H.; Ahmad, W.Y.W.; Zubairi, S.I.; Maskat, M.Y. Effect of PH on Adsorption of Organic Acids and Phenolic Compounds by Amberlite Ira 67 Resin. J. Teknol. 2019, 81, 69–81. [Google Scholar] [CrossRef]
- Samah, R.; Zainol, N.; Yee, P.; Pawing, C.; Abd-Aziz, S. Adsorption of Vanillin Using Macroporous Resin H103. Adsorpt. Sci. Technol. 2013, 31, 599–610. [Google Scholar] [CrossRef]
- Bautista, L.; Plata, M.; José, A.; Martínez, M. Application of an Effective Diffusion Model to the Adsorption of Aspartame on Functionalised Divinylbenzene–Styrene Macroporous Resins. J. Food Eng. 2003, 59, 319–325. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, K.; Li, J.; Wang, Q.; Guo, J. Separation of Vanillin and Syringaldehyde from Oxygen Delignification Spent Liquor by Macroporous Resin Adsorption. CLEAN–Soil Air Water 2010, 38, 1074–1079. [Google Scholar] [CrossRef]
- Santos, A.G.; de Albuquerque, T.L.; Ribeiro, B.D.; Coelho, M.A.Z. In Situ Product Recovery Techniques Aiming to Obtain Biotechnological Products: A Glance to Current Knowledge. Biotechnol. Appl. Biochem. 2021, 68, 1044–1057. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.-J.; Liu, Y.-S.; Zhou, L.-G.; Wu, J.-Y. Enhanced Beauvericin Production with in Situ Adsorption in Mycelial Liquid Culture of Fusarium Redolens Dzf2. Process Biochem. 2009, 44, 1063–1067. [Google Scholar] [CrossRef]
- Jahn, A.; Nielsen, P.H. Extraction of Extracellular Polymeric Substances (EPS) from Biofilms Using a Cation Exchange Resin. Water Sci. Technol. 1995, 32, 157–164. [Google Scholar] [CrossRef]
- Zhao, J.; Li, Y.; Shan, T.; Mou, Y.; Zhou, L. Enhancement of Diepoxin ζ Production with in Situ Resin Adsorption in Mycelial Liquid Culture of the Endophytic Fungus Berkleasmium Sp. Dzf12 from Dioscorea Zingiberensis. World J. Microbiol. Biotechnol. 2011, 27, 2753–2758. [Google Scholar] [CrossRef]
- Wang, W.; Maimaiti, A.; Shi, H.; Wu, R.; Wang, R.; Li, Z.; Qi, D.; Yu, G.; Deng, S. Adsorption Behavior and Mechanism of Emerging Perfluoro-2-Propoxypropanoic Acid (GenX) on Activated Carbons and Resins. Chem. Eng. J. 2019, 364, 132–138. [Google Scholar] [CrossRef]
- Halim, M.; Rios-Solis, L.; Micheletti, M.; Ward, J.M.; Lye, G.J. Microscale Methods to Rapidly Evaluate Bioprocess Options for Increasing Bioconversion Yields: Application to the ω-Transaminase Synthesis of Chiral Amines. Bioprocess Biosyst. Eng. 2014, 37, 931–941. [Google Scholar] [CrossRef]
- Oslan, S.N.H.; Halim, M.; Ramle, N.A.; Saad, M.Z.; Tan, J.S.; Kapri, M.R.; Ariff, A.B. Improved Stability of Live Attenuated Vaccine GdhA Derivative Pasteurella Multocida B:2 by Freeze Drying Method for Use as Animal Vaccine. Cryobiology 2017, 79, 1–8. [Google Scholar] [CrossRef]
- Rohit, S.G.; Jyoti, P.K.; Subbi, R.R.T.; Naresh, M.; Senthilkumar, S. Kinetic Modeling of Hyaluronic Acid Production in Palmyra Palm (Borassus flabellifer) Based Medium by Streptococcus zooepidemicus MTCC 3523. Biochem. Eng. J. 2018, 137, 284–293. [Google Scholar] [CrossRef]
- Gao, H.-J.; Du, G.-C.; Chen, J. Analysis of Metabolic Fluxes for Hyaluronic Acid (HA) Production by Streptococcus Zooepidemicus. World J. Microbiol. Biotechnol. 2006, 22, 399–408. [Google Scholar] [CrossRef]
- Oslan, S.N.H.; Tan, J.S.; Abbasiliasi, S.; Ziad Sulaiman, A.; Saad, M.Z.; Halim, M.; Ariff, A.B. Integrated Stirred-Tank Bioreactor with Internal Adsorption for the Removal of Ammonium to Enhance the Cultivation Performance of GdhA Derivative Pasteurella multocida B:2. Microorganisms 2020, 8, 1654. [Google Scholar] [CrossRef]
- Cui, S.; Zhao, J.; Zhang, H.; Chen, W. High-Density Culture of Lactobacillus Plantarum Coupled with a Lactic Acid Removal System with Anion-Exchange Resins. Biochem. Eng. J. 2016, 115, 80–84. [Google Scholar] [CrossRef]
- Tan, J.S.; Ling, T.C.; Mustafa, S.; Tam, Y.J.; Ramanan, R.N.; Ariff, A.B. An Integrated Bioreactor-Expanded Bed Adsorption System for the Removal of Acetate to Enhance the Production of Alpha-Interferon-2b by Escherichia coli. Process Biochem. 2013, 48, 551–558. [Google Scholar] [CrossRef]
- Quintero, J.; Acosta, A.; Mejía, C.; Ríos, R.; Torres, A.M. Purification of Lactic Acid Obtained from a Fermentative Process of Cassava Syrup Using Ion Exchange Resins. Rev. Fac. Ing. 2012, 65, 139–151. [Google Scholar]
- Chahuki, F.F.; Aminzadeh, S.; Jafarian, V.; Tabandeh, F.; Khodabandeh, M. Hyaluronic Acid Production Enhancement via Genetically Modification and Culture Medium Optimization in Lactobacillus acidophilus. Int. J. Biol. Macromol. 2019, 121, 870–881. [Google Scholar] [CrossRef] [PubMed]
- Saharkhiz, S.; Babaeipour, V. The Dilution Effect of Media Culture on Mixing Time, Kla O2, and Hyaluronic Acid Production in S. zooepidemicus Fed-Batch Culture. Biotechnol. Lett. 2021, 43, 2217–2222. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, M.; Li, T.; Fu, L.; Cao, W.; Liu, H. Construction of Efficient Streptococcus Zooepidemicus Strains for Hyaluoronic Acid Production Based on Identification of Key Genes Involved in Sucrose Metabolism. AMB Express 2016, 6, 121. [Google Scholar] [CrossRef]
- Shah, M.; Badle, S.; Ramachandran, K.B. Hyaluronic Acid Production and Molecular Weight Improvement by Redirection of Carbon Flux towards Its Biosynthesis Pathway. Biochem. Eng. J. 2013, 80, 53–60. [Google Scholar] [CrossRef]
- Cheng, F.; Luozhong, S.; Guo, Z.; Yu, H.; Stephanopoulos, G. Enhanced Biosynthesis of Hyaluronic Acid Using Engineered Corynebacterium glutamicum via Metabolic Pathway Regulation. Biotechnol. J. 2017, 12. [Google Scholar] [CrossRef]
- Kaur, M.; Jayaraman, G. Hyaluronan Production and Molecular Weight Is Enhanced in Pathway-Engineered Strains of Lactate Dehydrogenase-Deficient Lactococcus lactis. Metab. Eng. Commun. 2016, 3, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Woo, J.E.; Seong, H.J.; Lee, S.Y.; Jang, Y.-S. Metabolic Engineering of Escherichia coli for the Production of Hyaluronic Acid from Glucose and Galactose. Front. Bioeng. Biotechnol. 2019, 7, 351. [Google Scholar] [CrossRef] [PubMed]
- Duffeck, H.C.B.P.; Pan, N.C.; Saikawa, G.I.A.; da Rocha, S.P.D.; Baldo, C.; Celligoi, M.A.P.C. Biomedical Potential of Hyaluronic Acid from Streptococcus zooepidemicus Produced in Sugarcane Molasses. Braz. J. Dev. 2020, 6, 49963–49980. [Google Scholar] [CrossRef]
- Garrigues, C.; Mercade, M.; Cocaign-Bousquet, M.; Lindley, N.D.; Loubiere, P. Regulation of Pyruvate Metabolism in Lactococcus lactis Depends on the Imbalance between Catabolism and Anabolism. Biotechnol. Bioeng. 2001, 74, 108–115. [Google Scholar] [CrossRef]
- Jagannath, S.; Ramachandran, K.B. Influence of Competing Metabolic Processes on the Molecular Weight of Hyaluronic Acid Synthesized by Streptococcus zooepidemicus. Biochem. Eng. J. 2010, 48, 148–158. [Google Scholar] [CrossRef]
- Poolman, B.; Bosman, B.; Kiers, J.A.N.; Konings, W.N. Control of Glycolysis by Glyceraldehyde-3-Phosphate Dehydrogenase in Streptococcus cremoris and Streptococcus lactis. J. Bacteriol. 1987, 169, 5887–5890. [Google Scholar] [CrossRef]
- Yalçın, O.; Baylan, N.; Çehreli, S. Competitive Adsorption of Anti-Parkinson Drugs on Different Amberlite Resins from Water: Quantitative Analysis by Ultra Performance Liquid Chromatography (UPLC). Ind. Eng. Chem. Res. 2021, 60, 11789–11801. [Google Scholar] [CrossRef]
- Tolner, B.; Smith, L.; Begent, R.H.J.; Chester, K.A. Expanded-Bed Adsorption Immobilized-Metal Affinity Chromatography. Nat. Protoc. 2006, 1, 1213–1222. [Google Scholar] [CrossRef]
- Ferrer-Polonio, E.; Mendoza-Roca, J.A.; Iborra-Clar, A.; Pastor-Alcañiz, L. Adsorption of Raw and Treated by Membranes Fermentation Brines from Table Olives Processing for Phenolic Compounds Separation and Recovery. J. Chem. Technol. Biotechnol. 2016, 91, 2094–2102. [Google Scholar] [CrossRef]
- Iyyappan, J.; Baskar, G.; Bharathiraja, B.; Saravanathamizhan, R. Malic Acid Production from Biodiesel Derived Crude Glycerol Using Morphologically Controlled Aspergillus Niger in Batch Fermentation. Bioresour. Technol. 2018, 269, 393–399. [Google Scholar] [CrossRef]
- Ferreira, R.G.; Azzoni, A.R.; Santana, M.H.A.; Petrides, D. Techno-Economic Analysis of a Hyaluronic Acid Production Process Utilizing Streptococcal Fermentation. Processes 2021, 9, 241. [Google Scholar] [CrossRef]
- Yu, J.; Quan, H.; Huang, Z.; Li, P.; Chang, S. Synthesis of a Heavy-Oil Viscosity Reducer Containing a Benzene Ring and Its Viscosity Reduction Mechanism. ChemistrySelect 2022, 7, e202102694. [Google Scholar] [CrossRef]
- Liu, Y.; Qian, Y.; Yong, X.; Jia, H.; Wei, P.; Zhou, J. Effects of Granular Activated Carbon and Temperature on the Viscosity and Methane Yield of Anaerobically Digested of Corn Straw with Different Dry Matter Concentrations. Bioresour. Technol. 2021, 332, 125109. [Google Scholar] [CrossRef] [PubMed]
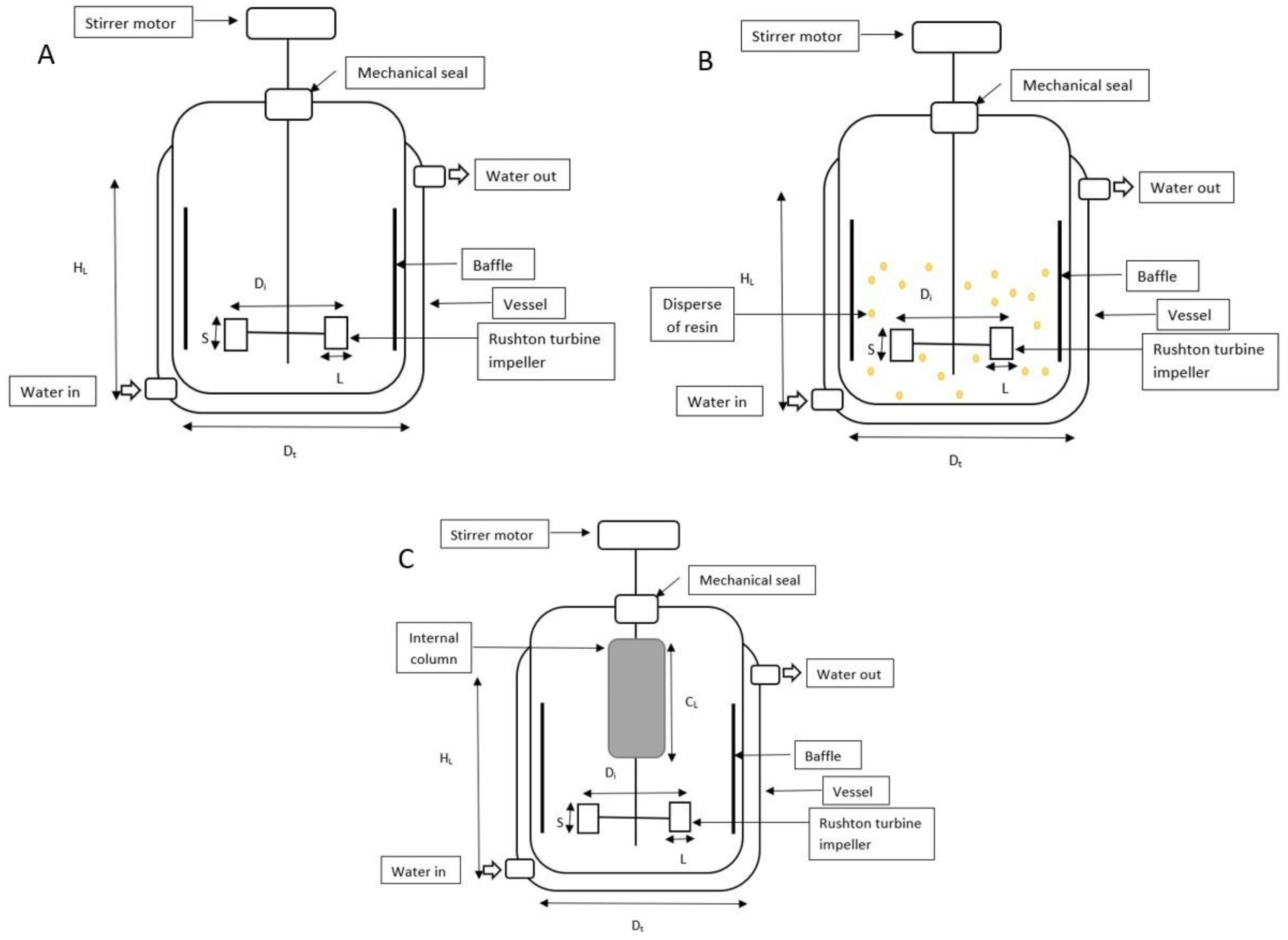




| Dimension and Variables | Value |
|---|---|
| Di (m) | 0.083 |
| Dt (m) | 0.130 |
| HL (m) | 0.120 |
| S (m) | 0.010 |
| Di/Dt (m) | 0.638 |
| L (M) | 0.015 |
| CL (m) | 0.120 |
| Speed (rev/min) | 200 |
| Airflow rate (vvm) | 1.00 |
| Type of Resin (Amberlite) | Amount of HA Adsorbed (%) | Characteristic | Particle Size (mm) | Ionic Form |
|---|---|---|---|---|
| IRA400 Cl | 58.73 ± 0.84 | Strong base | 0.6–0.75 | Cl- |
| IRA900 Cl | 42.19 ± 1.35 | Strong base | 0.6–0.75 | Cl- |
| IRA410 Cl | 40.58 ± 2.96 | Strong base | 0.64–0.8 | Cl- |
| IRA402 Cl | 51.85 ± 1.63 | Strong base | 0.6–0.75 | Cl- |
| IRA67 | 81.80 ± 0.56 | Weak base | 0.5–0.75 | Free base |
| Growth Phase | HA Concentration (g/L) |
|---|---|
| Lag | 0.148 ± 0.80 |
| Late exponential | 0.537 ± 1.23 |
| Stationary | 1.174 ± 0.003 |
| IRA67 Dosage (g/L) | HA in Media (g/L) | HA Eluted (g/L) | Total HA (g/L) |
|---|---|---|---|
| 0 | 0.312 ± 0.002 | - | 0.312 ± 0.002 |
| 15 | 0.236 ± 0.001 | 0.232 ± 0.003 | 0.468 ± 0.002 |
| 25 | 0.285 ± 0.001 | 0.296 ± 0.002 | 0.581 ± 0.001 |
| 35 | 0.323 ± 0.001 | 0.298 ± 0.002 | 0.621 ± 0.002 |
| 45 | 0.564 ± 0.018 | 0.312 ± 0.003 | 0.876 ± 0.007 |
| 50 | 0.356 ± 0.001 | 0.818 ± 0.004 | 1.174 ± 0.003 |
| 55 | 0.287 ± 0.010 | 0.857 ± 0.005 | 1.144 ± 0.006 |
| 60 | 0.404 ± 0.002 | 0.704 ± 0.005 | 1.107 ± 0.004 |
| Time * | Cell Concentration (g/L) | Dissolved Oxygen (%) | Glucose (g/L) | HA (g/L) | LA (g/L) |
|---|---|---|---|---|---|
| 0 | 0.00 | 98.0 | 50.00 ± 0.03 | 0.00 | 0.032 ± 0.24 |
| 14 | 2.547 ± 0.24 | 0.00 | 33.51 ± 0.23 | 2.763 ± 0.04 | 55.629 ± 0.15 |
| 15 | 2.325 ± 0.40 | 0.00 | 20.62 ± 0.10 | 2.872 ± 0.06 | 58.672 ± 0.09 |
| 24 | 2.478 ± 0.06 | 0.00 | 1.27 ± 0.78 | 2.713 ± 0.18 | 42.789 ± 0.10 |
| Cultivation Variable | Before Addition of IRA67 (mPa·s) * | After Addition of IRA67 (mPa·s) * | After 24 h (mPa·s) * |
|---|---|---|---|
| Control fermentation (no resin) | - | - | 60.5 ± 0.05 |
| Disperse of IRA67 | 60.7 ± 0.05 | 41.4 ± 0.1 | 40.0 ± 0.1 |
| Internal column of IRA67 | 58.8 ± 0.1 | 47.0 ± 0.1 | 23.7 ± 0.05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdullah Thaidi, N.I.; Mohamad, R.; Wasoh, H.; Kapri, M.R.; Ghazali, A.B.; Tan, J.S.; Rios-Solis, L.; Halim, M. Development of In Situ Product Recovery (ISPR) System Using Amberlite IRA67 for Enhanced Biosynthesis of Hyaluronic Acid by Streptococcus zooepidemicus. Life 2023, 13, 558. https://doi.org/10.3390/life13020558
Abdullah Thaidi NI, Mohamad R, Wasoh H, Kapri MR, Ghazali AB, Tan JS, Rios-Solis L, Halim M. Development of In Situ Product Recovery (ISPR) System Using Amberlite IRA67 for Enhanced Biosynthesis of Hyaluronic Acid by Streptococcus zooepidemicus. Life. 2023; 13(2):558. https://doi.org/10.3390/life13020558
Chicago/Turabian StyleAbdullah Thaidi, Nur Imanina, Rosfarizan Mohamad, Helmi Wasoh, Mohammad Rizal Kapri, Ahmad Badruddin Ghazali, Joo Shun Tan, Leonardo Rios-Solis, and Murni Halim. 2023. "Development of In Situ Product Recovery (ISPR) System Using Amberlite IRA67 for Enhanced Biosynthesis of Hyaluronic Acid by Streptococcus zooepidemicus" Life 13, no. 2: 558. https://doi.org/10.3390/life13020558
APA StyleAbdullah Thaidi, N. I., Mohamad, R., Wasoh, H., Kapri, M. R., Ghazali, A. B., Tan, J. S., Rios-Solis, L., & Halim, M. (2023). Development of In Situ Product Recovery (ISPR) System Using Amberlite IRA67 for Enhanced Biosynthesis of Hyaluronic Acid by Streptococcus zooepidemicus. Life, 13(2), 558. https://doi.org/10.3390/life13020558






