Evaluation of the Immunomodulatory Effects of a Probiotics and Natural Extract-Based Formulation in Bacterial-Induced Prostatitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Formulation Testing
2.3. Cell Cultures
2.3.1. Caco-2 Cell Culture
2.3.2. LNCaP Cell Culture
2.3.3. THP-1 Cell Culture
2.3.4. Bacterial Strain
2.4. In Vitro Intestinal Epithelium Model
2.5. Medium Conditioning by Probiotic Adhesion to Intestinal Mucosa
2.6. In Vitro Model of Innate Immune System
2.7. Determination of Cytotoxicity
2.8. THP-1-Conditioned Medium
2.9. Evaluation of Anti-Inflammatory Activity
2.10. Statistical Analysis
3. Results
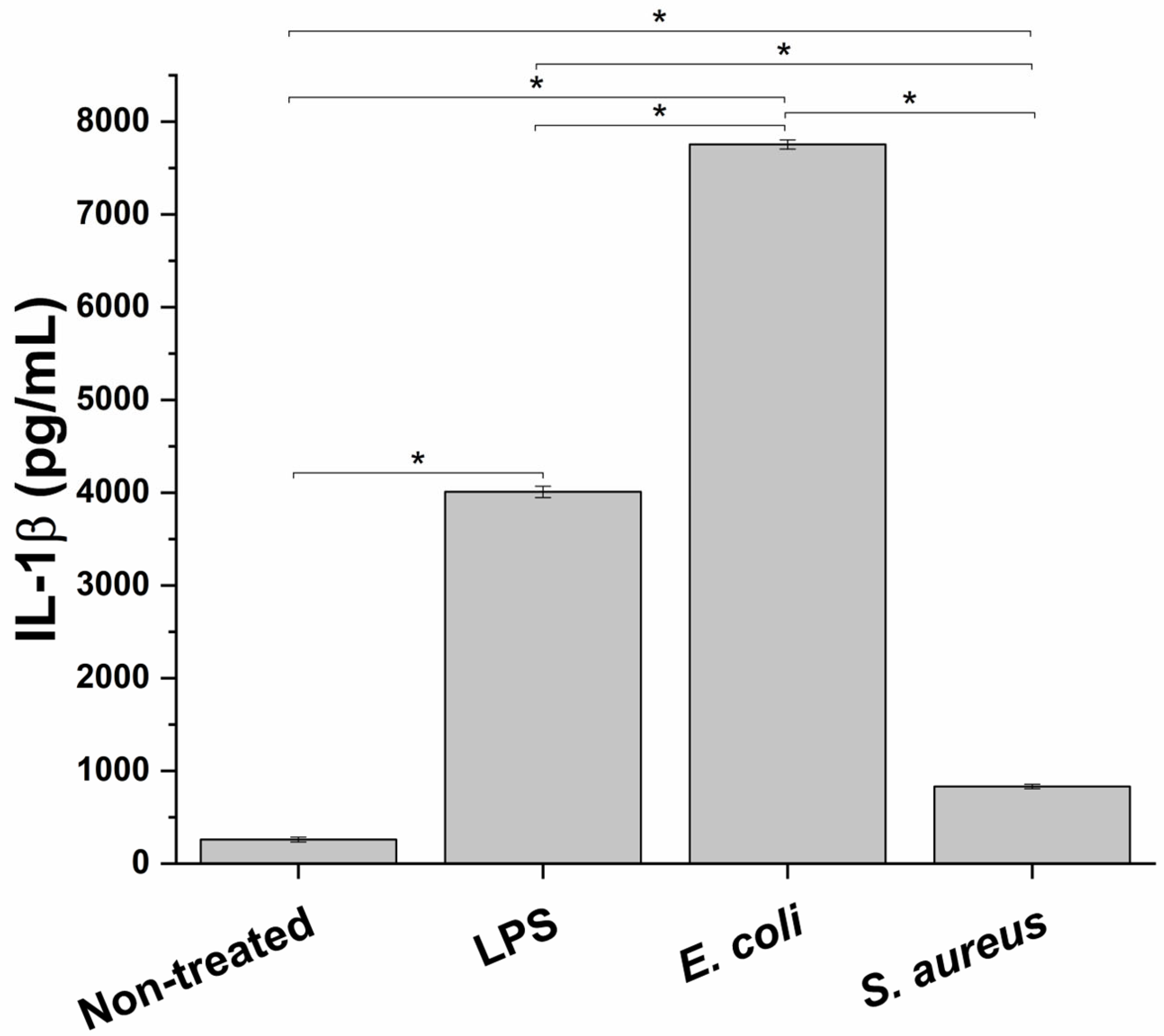
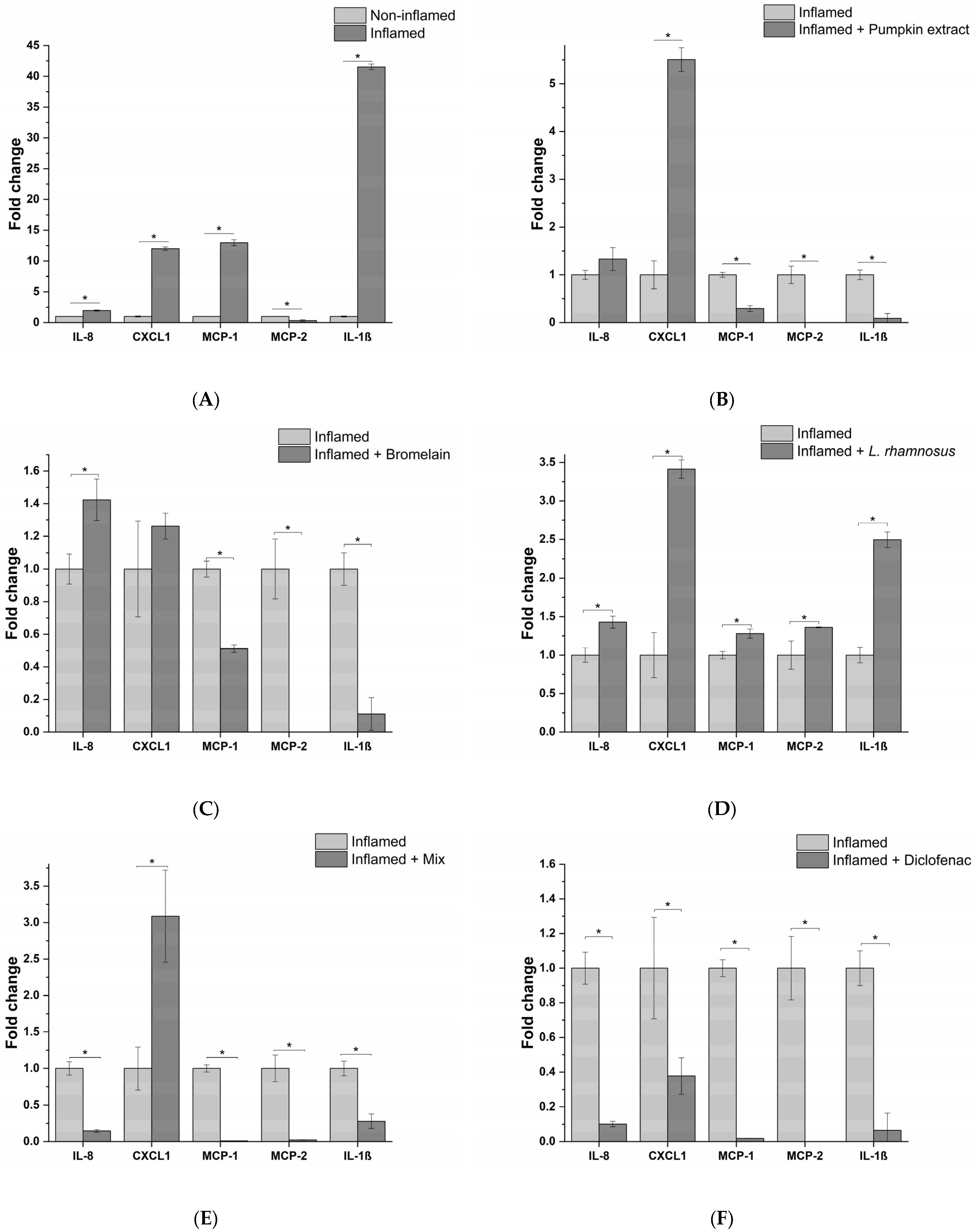
3.1. Impact of Different Bacterial Strains and Bacterial Endotoxin on Innate Immune System In Vitro Model Inflammation
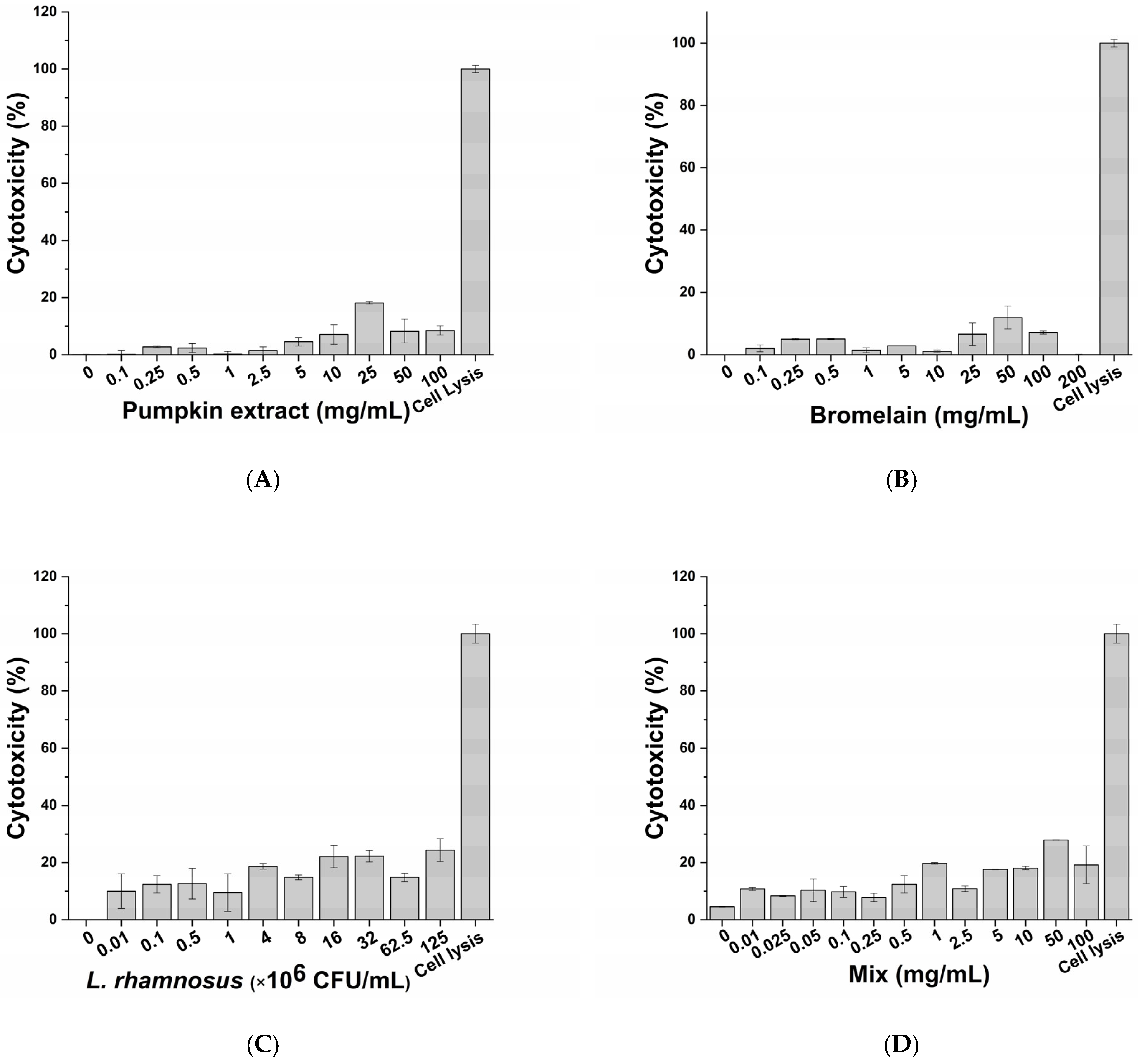
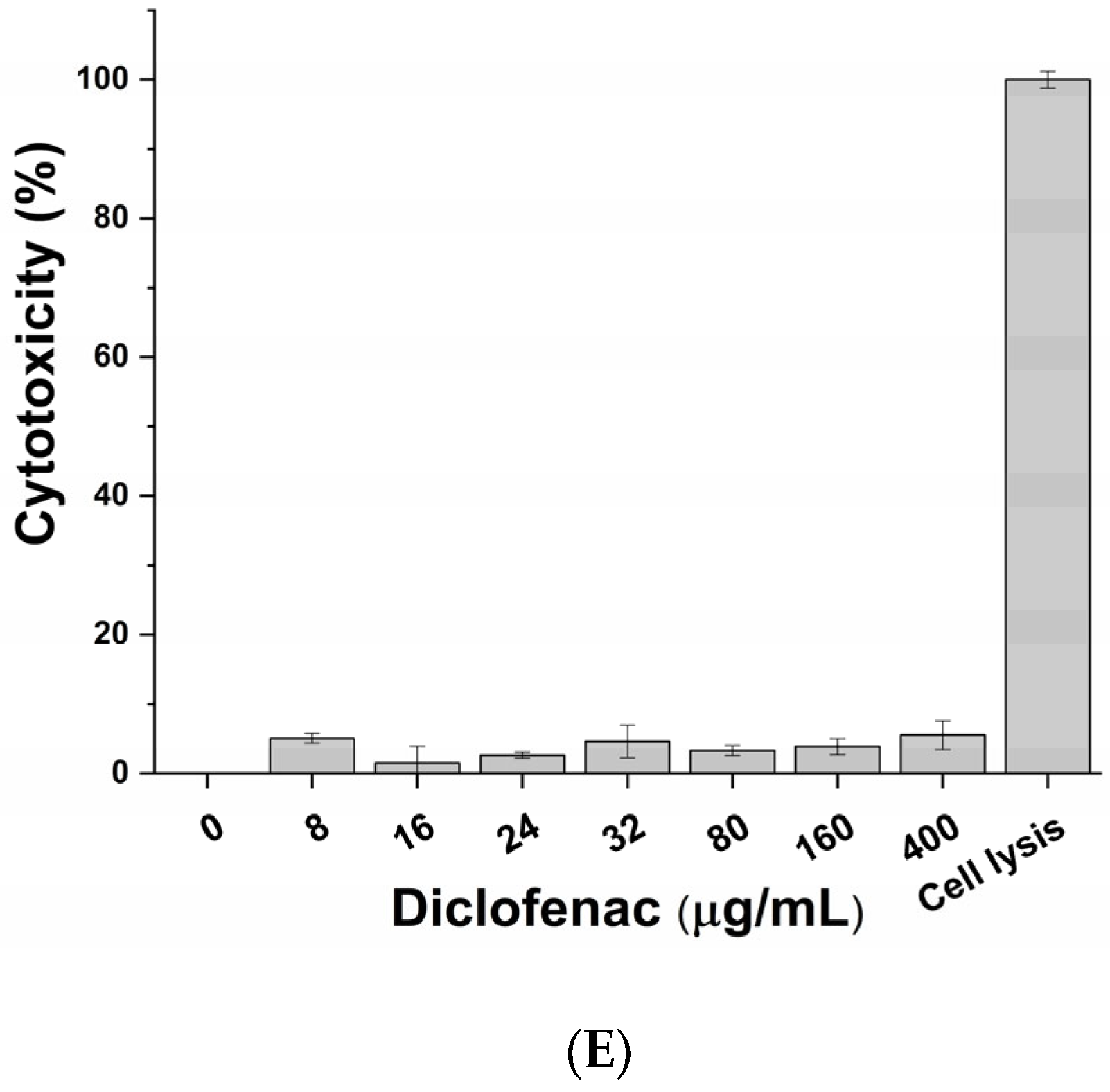
3.2. Cytotoxic Effect of Active Principles and Their Mix on In Vitro Cell Model
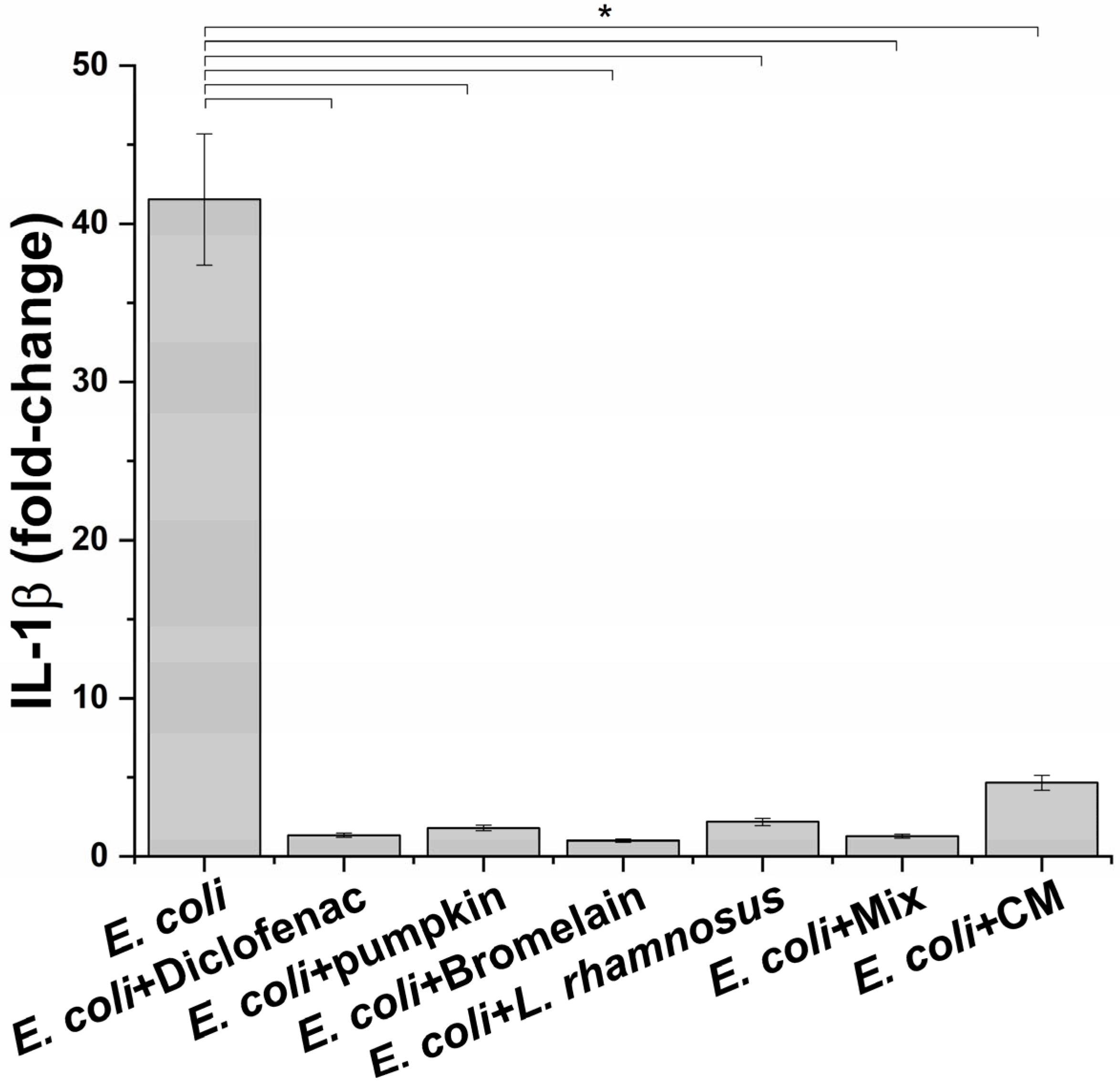
3.3. Anti-Inflammatory Activity of Treatments on Immune System In Vitro Model
3.4. Impact of THP-1-Conditioned Milieus on Prostate Epithelium In Vitro Model Inflammation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pontari, M.A. Chronic Prostatitis/Chronic Pelvic Pain Syndrome. Urol. Clin. N. Am. 2008, 35, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Wenninger, R.E.B.K.; Heiman, J.R.; Rothman, I.; Berghuis, J.P. Sickness impact of chronic nonbacterial prostatitis and its correlates. J. Urol. 1996, 155, 965–968. [Google Scholar] [CrossRef] [PubMed]
- Pontari, M.A.; Ruggieri, M.R. Mechanisms in Prostatitis/Chronic Pelvic Pain Syndrome. J. Urol. 2008, 179, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Mazzoli, S. Conventional bacteriology in prostatitis patients: Microbiological bias, problems and epidemiology on 1686 microbial isolates. Arch. Ital. Urol. Androl. 2007, 79, 71–75. [Google Scholar] [PubMed]
- Nickel, J.C.; Roehrborn, C.G.; O’Leary, M.P.; Bostwick, D.G.; Somerville, M.C.; Rittmaster, R.S. The Relationship between Prostate Inflammation and Lower Urinary Tract Symptoms: Examination of Baseline Data from the REDUCE Trial. Eur. Urol. 2008, 54, 1379–1384. [Google Scholar] [CrossRef] [PubMed]
- Conte, M.P.; Aleandri, M.; Marazzato, M.; Conte, A.L.; Ambrosi, C.; Nicoletti, M.; Zagaglia, C.; Gambara, G.; Palombi, F.; De Cesaris, P.; et al. The Adherent/Invasive Escherichia coli Strain LF82 Invades and Persists in Human Prostate Cell Line RWPE-1, Activating a Strong Inflammatory Response. Infect. Immun. 2016, 84, 3105–3113. [Google Scholar] [CrossRef] [PubMed]
- Sfanos, K.S.; De Marzo, A.M. Prostate cancer and inflammation: The evidence. Histopathology 2012, 60, 199–215. [Google Scholar] [CrossRef]
- Haverkamp, J.; Charbonneau, B.; Ratliff, T.L. Prostate inflammation and its potential impact on prostate cancer: A current review. J. Cell. Biochem. 2008, 103, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Kanda, Y.; Osaki, M.; Okada, F. Chemopreventive Strategies for Inflammation-Related Carcinogenesis: Current Status and Future Direction. Int. J. Mol. Sci. 2017, 18, 867. [Google Scholar] [CrossRef]
- Al-Okbi, S.Y. Nutraceuticals of anti-inflammatory activity as complementary therapy for rheumatoid arthritis. Toxicol. Ind. Health 2014, 30, 738–749. [Google Scholar] [CrossRef]
- Barve, K.H.; Kulkarni, Y.A.; Gaikwad, A.B. Nutraceuticals as Therapeutic Agents for Inflammation; Elsevier Inc.: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Lee, J.; Koo, N.; Min, D.B. Reactive Oxygen Species, Aging, and Antioxidative Nutraceuticals. Compr. Rev. Food Sci. Food Saf. 2004, 3, 21–33. [Google Scholar] [CrossRef]
- Balgoon, M.J.; Al-Zahrani, M.H.; Al Jaouni, S.; Ayuob, N. Combined Oral and Topical Application of Pumpkin (Cucurbita pepo L.) Alleviates Contact Dermatitis Associated With Depression Through Downregulation Pro-Inflammatory Cytokines. Front. Pharmacol. 2021, 12, 663417. [Google Scholar] [CrossRef] [PubMed]
- Rathnavelu, V.; Alitheen, N.B.; Sohila, S.; Kanagesan, S.; Ramesh, R. Potential role of bromelain in clinical and therapeutic applications (Review). Biomed. Rep. 2016, 5, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yang, J.; Lu, F. Urethral dysbacteriosis as an underlying, primary cause of chronic prostatitis: Potential implications for probiotic therapy. Med. Hypotheses 2009, 73, 741–743. [Google Scholar] [CrossRef] [PubMed]
- Amdekar, S.; Singh, V.; Singh, D.D. Probiotic Therapy: Immunomodulating Approach Toward Urinary Tract Infection. Curr. Microbiol. 2011, 63, 484–490. [Google Scholar] [CrossRef]
- Candela, M.; Perna, F.; Carnevali, P.; Vitali, B.; Ciati, R.; Gionchetti, P.; Rizzello, F.; Campieri, M.; Brigidi, P. Interaction of probiotic Lactobacillus and Bifidobacterium strains with human intestinal epithelial cells: Adhesion properties, competition against enteropathogens and modulation of IL-8 production. Int. J. Food Microbiol. 2008, 125, 286–292. [Google Scholar] [CrossRef]
- De Vrese, M.; Schrezenmeir, J. Probiotics and non-intestinal infectious conditions. Br. J. Nutr. 2002, 88, s59–s66. [Google Scholar] [CrossRef]
- Collins, M.M.; Stafford, R.; O’Leary, M.P.; Barry, M.J. How common is prostatitis? A national survey of physician visits. J. Urol. 1998, 159, 1224–1228. [Google Scholar] [CrossRef]
- Britton, J.J.C.C. Prostatitis. AUA Updat. Ser. 1998, 17, 154–159. [Google Scholar]
- Takeuchi, O.; Akira, S. Pattern Recognition Receptors and Inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef]
- Gatti, G.; Rivero, V.; Motrich, R.D.; Maccioni, M. Prostate epithelial cells can act as early sensors of infection by up-regulating TLR4 expression and proinflammatory mediators upon LPS stimulation. J. Leukoc. Biol. 2006, 79, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Mogensen, T.H. Pathogen Recognition and Inflammatory Signaling in Innate Immune Defenses. Clin. Microbiol. Rev. 2009, 22, 240–273. [Google Scholar] [CrossRef] [PubMed]
- Hochreiter, W.W.; Nadler, R.B.; Koch, A.E.; Campbell, P.L.; Ludwig, M.; Weidner, W.; Schaeffer, A.J. Cme Article Evaluation of the Cytokines Interleukin 8 and Prostatic Secretions. Urology 2000, 4295, 1025–1029. [Google Scholar] [CrossRef] [PubMed]
- Nadler, R.B.; Koch, A.E.; Calhoun, E.A.; Campbell, P.L.; Pruden, D.L.; Bennett, C.L.; Yarnold, P.R.; Schaeffer, A.J. IL-1β and TNF-α in prostatic secretions are indicators in the evaluation of men with chronic prostatitis. J. Urol. 2000, 164, 214–218. [Google Scholar] [CrossRef]
- Koch, A.E.; Kunkel, S.L.; Pearce, W.H.; Shah, M.R.; Parikh, D.; Evanoff, H.L.; Haines, G.K.; Burdick, M.D.; Strieter, R.M. Enhanced production of the chemotactic cytokines interleukin-8 and monocyte chemoattractant protein-1 in human abdominal aortic aneurysms. Am. J. Pathol. 1993, 142, 1423–1431. [Google Scholar]
- Koch, A.E.; Kunkel, S.L.; Shah, M.R.; Hosaka, S.; Halloran, M.M.; Haines, G.K.; Burdick, M.D.; Pope, R.M. Growth-related gene product alpha. A chemotactic cytokine for neutrophils in rheumatoid arthritis. J. Immunol. 1995, 155, 3660–3666. [Google Scholar] [CrossRef]
- Voronov, E.; Shouval, D.S.; Krelin, Y.; Cagnano, E.; Benharroch, D.; Iwakura, Y.; Dinarello, C.A.; Apte, R.N. IL-1 is required for tumor invasiveness and angiogenesis. Proc. Natl. Acad. Sci. USA 2003, 100, 2645–2650. [Google Scholar] [CrossRef]
- Gong, X.; Gong, W.; Kuhns, D.B.; Ben-Baruch, A.; Howard, O.M.Z.; Wang, J.M. Monocyte Chemotactic Protein-2 (MCP-2) Uses CCR1 AND CCR2B as Its Functional Receptors. J. Biol. Chem. 1997, 272, 11682–11685. [Google Scholar] [CrossRef]
- Yamagami, S.; Tanaka, H.; Endo, N. Monocyte chemoattractant protein-2 can exert its effects through the MCP-1 receptor (CC CKR2B). FEBS Lett. 1997, 400, 329–332. [Google Scholar] [CrossRef]
- Moore, U.M.; Kaplow, J.M.; Pleass, R.D.; Castro, S.W.; Naik, K.; Lynch, C.N.; Daly, S.; Roach, A.G.; Jaye, M.; Williams, R.J. Monocyte chemoattractant protein-2 is a potent agonist of CCR2B. J. Leukoc. Biol. 1997, 62, 911–915. [Google Scholar] [CrossRef]
- Murphy, P.M.; Baggiolini, M.; Charo, I.F.; Hébert, C.A.; Horuk, R.; Matsushima, K.; Miller, L.H.; Oppenheim, J.J.; Power, C.A. International union of pharmacology. XXII. Nomenclature for chemokine receptors. Pharmacol. Rev. 2000, 52, 145–176. [Google Scholar] [PubMed]
- Alam, J.V.D.R.; Forsythe, P.; Stafford, S.; Heinrich, J.; Bravo, R.; Proost, P. Monocyte chemotactic protein-2, monocyte chemotactic protein-3, and fibroblast-induced cytokine. Three new chemokines induce chemotaxis and activation of basophils. J. Immunol. 1994, 153, 3155–3159. [Google Scholar] [CrossRef]
- Uguccioni, M.; D’Apuzzo, M.; Loetscher, M.; Dewald, B.; Baggiolini, M. Actions of the chemotactic cytokines MCP-1, MCP-2, MCP-3, RANTES, MIP-1α and MIP-1β on human monocytes. Eur. J. Immunol. 1995, 25, 64–68. [Google Scholar] [CrossRef]
- Taub, D.D.; Proost, P.; Murphy, W.J.; Anver, M.; Longo, D.L.; Van Damme, J.; Oppenheim, J.J. Monocyte chemotactic protein-1 (MCP-1), -2, and -3 are chemotactic for human T lymphocytes. J. Clin. Investig. 1995, 95, 1370–1376. [Google Scholar] [CrossRef] [PubMed]
- Noso, N.; Proost, P.; Vandamme, J.; Schroder, J. Human Monocyte Chemotactic Proteins-2 and -3 (MCP-2 and MCP-3) Attract Human Eosinophils and Desensitize the Chemotactic Responses Towards RANTES. Biochem. Biophys. Res. Commun. 1994, 200, 1470–1476. [Google Scholar] [CrossRef]
- Duque, G.A.; Descoteaux, A. Macrophage Cytokines: Involvement in Immunity and Infectious Diseases. Front. Immunol. 2014, 5, 491. [Google Scholar] [CrossRef]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte Chemoattractant Protein-1 (MCP-1): An Overview. J. Interf. Cytokine Res. 2009, 29, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Waugh, D.J.; Wilson, C. The Interleukin-8 Pathway in Cancer. Clin. Cancer Res. 2008, 14, 6735–6741. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.; Gray, K.P.; Harshman, L.C.; Evan, C.; Nakabayashi, M.; Fichorova, R.; Rider, J.; Mucci, L.; Kantoff, P.; Sweeney, C.J. Elevated IL-8, TNF-α, and MCP-1 in men with metastatic prostate cancer starting androgen-deprivation therapy (ADT) are associated with shorter time to castration-resistance and overall survival. Prostate 2014, 74, 820–828. [Google Scholar] [CrossRef]
- Veltri, R.W.; Miller, M.C.; Zhao, G.; Ng, A.; Marley, G.M.; Wright, G.L., Jr.; Vessella, R.L.; Ralph, D. Interleukin-8 Serum Levels in Patients With Prostate Cancer. Urology 1999, 4295, 139–147. [Google Scholar] [CrossRef]
- Lee, Y.-C.; Gajdošik, M.Š.; Josic, D.; Clifton, J.G.; Logothetis, C.; Yu-Lee, L.-Y.; Gallick, G.E.; Maity, S.N.; Lin, S.-H. Secretome Analysis of an Osteogenic Prostate Tumor Identifies Complex Signaling Networks Mediating Cross-talk of Cancer and Stromal Cells Within the Tumor Microenvironment. Mol. Cell. Proteom. 2015, 14, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Tseng, C.; Zhang, Y.; Sirin, O.; Corn, P.G.; Li-Ning-Tapia, E.M.; Troncoso, E.M.L.-N.P.; Davis, J.; Pettaway, C.; Ward, J.; et al. CXCL1 mediates obesity-associated adipose stromal cell trafficking and function in the tumour microenvironment. Nat. Commun. 2016, 7, 11674. [Google Scholar] [CrossRef] [PubMed]
| Active Principles | Form |
|---|---|
| Pumpkin d.e. (tit. 40% in fatty acids) | Powder |
| Bromelain (2400 GDU/g) | Powder |
| L. rhamnosus (LRH020 300 B/g) | Powder |
| Treatment | Highest Non-Cytotoxic Concentration |
|---|---|
| Pumpkin extract | 100 mg/mL |
| Bromelain | 200 mg/mL |
| L. rhamnosus | 125 × 106 CFU/mL |
| Mix | 100 mg/mL |
| Diclofenac | 400 μg/mL |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Murzilli, S.; Mirone, V.; Micheletto, M.; Tedesco, E.; Maira, G.D.; Benetti, F.; Vanelli, A. Evaluation of the Immunomodulatory Effects of a Probiotics and Natural Extract-Based Formulation in Bacterial-Induced Prostatitis. Life 2023, 13, 389. https://doi.org/10.3390/life13020389
Murzilli S, Mirone V, Micheletto M, Tedesco E, Maira GD, Benetti F, Vanelli A. Evaluation of the Immunomodulatory Effects of a Probiotics and Natural Extract-Based Formulation in Bacterial-Induced Prostatitis. Life. 2023; 13(2):389. https://doi.org/10.3390/life13020389
Chicago/Turabian StyleMurzilli, Stefania, Vincenzo Mirone, Marta Micheletto, Erik Tedesco, Giovanni Di Maira, Federico Benetti, and Arianna Vanelli. 2023. "Evaluation of the Immunomodulatory Effects of a Probiotics and Natural Extract-Based Formulation in Bacterial-Induced Prostatitis" Life 13, no. 2: 389. https://doi.org/10.3390/life13020389
APA StyleMurzilli, S., Mirone, V., Micheletto, M., Tedesco, E., Maira, G. D., Benetti, F., & Vanelli, A. (2023). Evaluation of the Immunomodulatory Effects of a Probiotics and Natural Extract-Based Formulation in Bacterial-Induced Prostatitis. Life, 13(2), 389. https://doi.org/10.3390/life13020389








