Gender Specificities of Cardiac Troponin Serum Levels: From Formation Mechanisms to the Diagnostic Role in Case of Acute Coronary Syndrome
Abstract
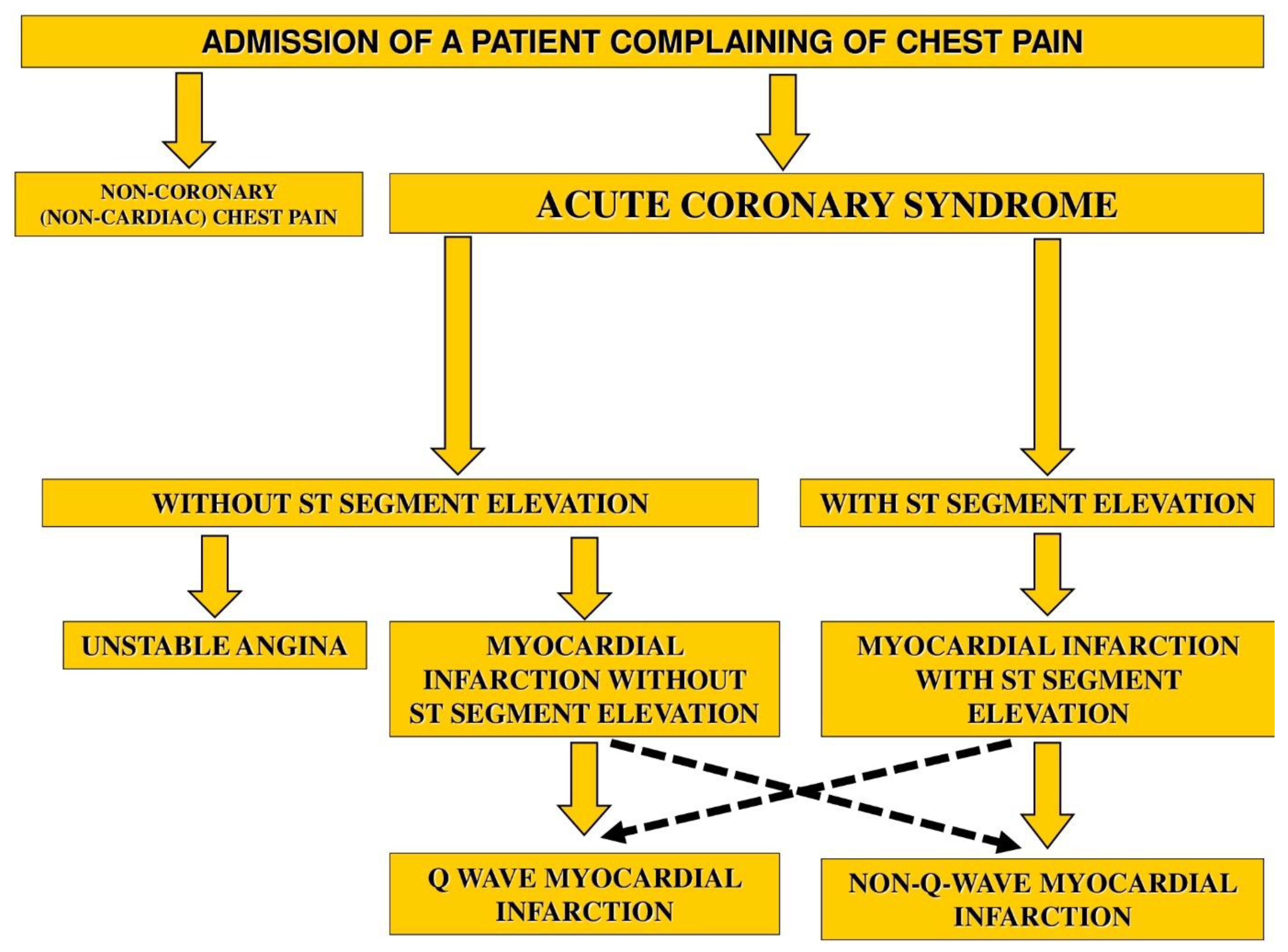
1. Introduction
2. Characteristics and Benefits of Using New Highly Sensitive Laboratory Tests
3. Gender Specificities of Cardiac Troponin Levels
4. Possible Mechanisms for the Formation of Gender Specificities in Cardiac Troponins
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jinnouchi, H.; Sato, Y.; Sakamoto, A.; Cornelissen, A.; Mori, M.; Kawakami, R.; Gadhoke, N.V.; Kolodgie, F.D.; Virmani, R.; Finn, A.V. Calcium deposition within coronary atherosclerotic lesion: Implications for plaque stability. Atherosclerosis 2020, 306, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Narula, N.; Olin, J.W.; Narula, N. Pathologic Disparities between Peripheral Artery Disease and Coronary Artery Disease. Arter. Thromb. Vasc. Biol. 2020, 40, 1982–1989. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M.; Grigorieva, Y.V.; Pavlova, T.V.; Duplyakov, D.V. Diagnostic significance of complete blood count in cardiovascular patients; Samara State Medical University. Russ. J. Cardiol. 2020, 25, 3923. [Google Scholar] [CrossRef]
- Staroverov, I.I.; Shakhnovich, R.M.; Gilyarov, M.Y.; Komarov, A.L.; Konstantinova, E.V.; Panchenko, E.P.; Yavelov, I.S. Eurasian Clinical Guidelines on Diagnosis and Treatment of Acute Coronary Syndrome with St Segment Elevation (Stemi). Eurasian Heart J. 2020, 1, 4–77. (In Russian) [Google Scholar] [CrossRef]
- Collet, J.-P.; Thiele, H.; Barbato, E.; Barthélémy, O.; Bauersachs, J.; Bhatt, D.L.; Dendale, P.; Dorobantu, M.; Edvardsen, T.; Folliguet, T.; et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur. Heart J. 2021, 42, 1289–1367. [Google Scholar] [CrossRef]
- Naghavi, M.; Wang, H.; Lozano, R.; Davis, A.; Liang, X.; Zhou, M.; Vollset, S.E.; Abbasoglu Ozgoren, A.; Abdalla, S.; Abd-Allah, F.; et al. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 385, 117–171. [Google Scholar]
- Erlikh, A.D.; Gratsiansky, N.A. Registry of Acute Coronary Syndromes RECORD-3. Characteristics of Patients and Treatment During Initial Hospitalization. Kardiologiia 2016, 56, 16–24. (In Russian) [Google Scholar] [CrossRef]
- Russian Society of Cardiology. Clinical practice guidelines for Acute ST-segment elevation myocardial infarction. Russ. J. Cardiol. 2020, 25, 4103. (In Russian) [Google Scholar] [CrossRef]
- Barbarash, O.L.; Duplyakov, D.V.; Zateischikov, D.A.; Panchenko, E.P.; Shakhnovich, R.M.; Yavelov, I.S.; Yakovlev, A.N.; Abugov, S.A.; Alekyan, B.G.; Arkhipov, M.V.; et al. 2020 Clinical practice guidelines for Acute coronary syndrome without ST segment elevation. Russ. J. Cardiol. 2021, 26, 4449. (In Russian) [Google Scholar] [CrossRef]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth Universal Definition of Myocardial Infarction (2018). Circulation 2018, 138, e618–e651. [Google Scholar] [CrossRef]
- Chaulin, A.M. Cardiac Troponins Metabolism: From Biochemical Mechanisms to Clinical Practice (Literature Review). Int. J. Mol. Sci. 2021, 22, 10928. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A. Cardiac Troponins: Contemporary Biological Data and New Methods of Determination. Vasc. Health Risk Manag. 2021, 17, 299–316. [Google Scholar] [CrossRef] [PubMed]
- De Lemos, J.A.; Drazner, M.H.; Omland, T.; Ayers, C.R.; Khera, A.; Rohatgi, A.; Hashim, I.; Berry, J.D.; Das, S.R.; Morrow, D.A.; et al. Association of Troponin T Detected with a Highly Sensitive Assay and Cardiac Structure and Mortality Risk in the General Population. JAMA 2010, 304, 2503–2512. [Google Scholar] [CrossRef] [PubMed]
- Zeller, T.; Tunstall-Pedoe, H.; Saarela, O.; Ojeda, F.; Schnabel, R.B.; Tuovinen, T.; Woodward, M.; Struthers, A.; Hughes, M.; Kee, F.; et al. High population prevalence of cardiac troponin I measured by a high-sensitivity assay and cardiovascular risk estimation: The MORGAM Biomarker Project Scottish Cohort. Eur. Heart J. 2013, 35, 271–281. [Google Scholar] [CrossRef]
- Altaf, A.; Shah, H.; Salahuddin, M. Gender based differences in clinical and Angiographic characteristics and outcomes of Acute Coronary Syndrome (ACS) in Asian population. Pak. J. Med. Sci. 2019, 35, 1349–1354. [Google Scholar] [CrossRef]
- Lobo, R.; De Michieli, L.; Jaffe, A.S. Sex-Specific 99th Percentile URLs for Cardiac Troponin Assays—Their Time Has Come. Clin. Chem. 2020, 67, 197–200. [Google Scholar] [CrossRef]
- Peacock, W.F.; Baumann, B.M.; Rivers, E.J.; Davis, T.E.; Handy, B.; Jones, C.W.; Hollander, J.E.; Limkakeng, A.T.; Mehrotra, A.; Than, M.; et al. Using Sex-specific Cutoffs for High-sensitivity Cardiac Troponin T to Diagnose Acute Myocardial Infarction. Acad. Emerg. Med. 2020, 28, 463–466. [Google Scholar] [CrossRef]
- Chaulin, A.M.; Duplyakov, D.V. Analytical Review of Modern Information on the Physiological and Pathochemical Mechanisms of the Release of Cardiospecific Proteins from Muscle Tissue, Methodology and Technologies of Their Research, Interpretation of the Results. Lab. Diagn. East. Eur. 2022, 11, 78–97. (In Russian) [Google Scholar] [CrossRef]
- Conrad, M.J.; Jarolim, P. Cardiac Troponins and High-sensitivity Cardiac Troponin Assays. Clin. Lab. Med. 2014, 34, 59–73. [Google Scholar] [CrossRef]
- Xu, R.-Y.; Zhu, X.-F.; Yang, Y.; Ye, P. High-sensitive cardiac troponin T. J. Geriatr. Cardiol. 2013, 10, 102–109. [Google Scholar]
- Chaulin, A.M. Cardiac troponins: Current information on the main analytical characteristics of determination methods and new diagnostic possibilities. Medwave 2021, 21, e002132. [Google Scholar] [CrossRef] [PubMed]
- Katus, H.A.; Remppis, A.; Neumann, F.J.; Scheffold, T.; Diederich, K.W.; Vinar, G.; Noe, A.; Matern, G.; Kuebler, W. Diagnostic efficiency of troponin T measurements in acute myocardial infarction. Circulation 1991, 83, 902–912. [Google Scholar] [CrossRef]
- Chaulin, A.M.; Abashina, O.E.; Duplyakov, D.V. High-sensitivity cardiac troponins: Detection and central analytical characteristics. Cardiovasc. Ther. Prev. 2021, 20, 2590. (In Russian) [Google Scholar] [CrossRef]
- Galvani, M.; Bachetti, C. High-sensitivity troponin in emergency room practice: Pros and cons. J. Cardiovasc. Med. 2018, 19 (Suppl. 1), e68–e71. [Google Scholar] [CrossRef] [PubMed]
- Jaffe, A.S. The 10 commandments of troponin, with special reference to high sensitivity assays. Heart 2011, 97, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M.; Karslyan, L.S.; Bazyuk, E.V.; Nurbaltaeva, D.A.; Duplyakov, D.V. Clinical and Diagnostic Value of Cardiac Markers in Human Biological Fluids. Kardiologiia 2019, 59, 66–75. (In Russian) [Google Scholar] [CrossRef]
- Apple, F.S.; Collinson, P.O.; IFCC Task Force on Clinical Applications of Cardiac Biomarkers. Analytical Characteristics of High-Sensitivity Cardiac Troponin Assays. Clin. Chem. 2012, 58, 54–61. [Google Scholar] [CrossRef]
- Apple, F.S.; Jaffe, A.S.; Collinson, P.; Mockel, M.; Ordonez-Llanos, J.; Lindahl, B.; Hollander, J.; Plebani, M.; Than, M.; Chan, M. IFCC educational materials on selected analytical and clinical applications of high sensitivity cardiac troponin assays. Clin. Biochem. 2014, 48, 201–203. [Google Scholar] [CrossRef]
- Reichlin, T.; Hochholzer, W.; Bassetti, S.; Steuer, S.; Stelzig, C.; Hartwiger, S.; Biedert, S.; Schaub, N.; Buerge, C.; Potocki, M.; et al. Early Diagnosis of Myocardial Infarction with Sensitive Cardiac Troponin Assays. N. Engl. J. Med. 2009, 361, 858–867. [Google Scholar] [CrossRef]
- Chaulin, A.M. Elevation Mechanisms and Diagnostic Consideration of Cardiac Troponins under Conditions Not Associated with Myocardial Infarction. Part 1. Life 2021, 11, 914. [Google Scholar] [CrossRef]
- Chaulin, A.M. Elevation Mechanisms and Diagnostic Consideration of Cardiac Troponins under Conditions Not Associated with Myocardial Infarction. Part 2. Life 2021, 11, 1175. [Google Scholar] [CrossRef]
- Available online: https://www.ifcc.org/media/478962/high-sensitivity-cardiac-troponin-i-and-t-assay-analytical-characteristics-designated-by-manufacturer-v042021.pdf (accessed on 8 December 2022).
- Chaulin, A.M.; Duplyakov, D.V. Cardiac troponins: Current data on the diagnostic value and analytical characteristics of new determination methods. Cor et Vasa 2021, 63, 486–493. [Google Scholar] [CrossRef]
- Mohebi, R.; Jackson, L.; McCarthy, C.P.; Murtagh, G.; Murphy, S.P.; Abboud, A.; Miksenas, H.; Gaggin, H.K.; Januzzi, J.L. Relation of High-Sensitivity Cardiac Troponin I and Obstructive Coronary Artery Disease in Patients without Acute Myocardial Infarction. Am. J. Cardiol. 2022, 173, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M.; Duplyakov, D.V. Cardioprotective Strategies for Doxorubicin-induced Cardiotoxicity: Present and Future. Ration. Pharmacother. Cardiol. 2022, 18, 103–112. [Google Scholar] [CrossRef]
- Chaulin, A.M.; Abashina, O.E.; Duplyakov, D.V. Pathophysiological mechanisms of cardiotoxicity in chemotherapeutic agents. Russ. Open Med. J. 2020, 9, e0305. [Google Scholar] [CrossRef]
- Chaulin, A.M.; Duplyakov, D.V. Comorbidity in chronic obstructive pulmonary disease and cardiovascular disease. Cardiovasc. Ther. Prev. 2021, 20, 2539. (In Russian) [Google Scholar] [CrossRef]
- Seng, N.S.H.L.; Zeratsion, G.; Pena, O.; Tufail, M.U.; Jim, B. Utility of Cardiac Troponins in Patients with Chronic Kidney Disease. Cardiol. Rev. 2022. online ahead of print. [Google Scholar] [CrossRef]
- Bellia, C.; Lombardo, M.; Della-Morte, D. Use of Troponin as a predictor for cardiovascular diseases in patients with type 2 Diabetes Mellitus. Clin. Chim. Acta 2020, 507, 54–61. [Google Scholar] [CrossRef]
- Chaulin, A.M.; Dupliakov, D.V. Modern Ideas about the Diagnostic Significance and Mechanisms of Increasing the Levels of Cardiac Troponins in Arterial Hypertension in Human Biological Fluids. Cardiol. Belarus 2022, 14, 80–94. [Google Scholar] [CrossRef]
- Foley, J.D., III; Sneed, J.D.; Steinhubl, S.R.; Kolasa, J.; Ebersole, J.L.; Lin, Y.; Kryscio, R.J.; McDevitt, J.T.; Campbell, C.L.; Miller, C.S. Oral fluids that detect cardiovascular disease biomarkers. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 114, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M.; Duplyakov, D.V. High-sensitivity cardiac troponins: Circadian rhythms. Cardiovasc. Ther. Prev. 2021, 20, 2639. (In Russian) [Google Scholar] [CrossRef]
- Mirzaii-Dizgah, I.; Riahi, E. Salivary troponin I as an indicator of myocardial infarction. Indian J. Med. Res. 2013, 138, 861–865. [Google Scholar] [PubMed]
- Chaulin, A.M.; Duplyakova, P.D.; Bikbaeva, G.R.; Tukhbatova, A.A.; Grigorieva, E.V.; Duplyakov, D.V. Concentration of high-sensitivity cardiac troponin I in the oral fluid in patients with acute myocardial infarction: A pilot study. Russ. J. Cardiol. 2020, 25, 3814. [Google Scholar] [CrossRef]
- Chen, J.-Y.; Lee, S.-Y.; Li, Y.-H.; Lin, C.-Y.; Shieh, M.-D.; Ciou, D.-S. Urine High-Sensitivity Troponin I Predict Incident Cardiovascular Events in Patients with Diabetes Mellitus. J. Clin. Med. 2020, 9, 3917. [Google Scholar] [CrossRef] [PubMed]
- Saviñon-Flores, A.I.; Saviñon-Flores, F.; Trejo, G.; Méndez, E.; Ţălu, S.; González-Fuentes, M.A.; Méndez-Albores, A. A review of cardiac troponin I detection by surface enhanced Raman spectroscopy: Under the spotlight of point-of-care testing. Front. Chem. 2022, 10, 1309. [Google Scholar] [CrossRef]
- Chaulin, A.M. Diagnostic value of highly sensitive cardiac troponins and mechanisms of their increase in serum and urine in arterial hypertension. Riv. Ital. Med. Lab. 2021, 17, 99–107. [Google Scholar]
- Chaulin, A.M. Metabolic Pathway of Cardiospecific Troponins: From Fundamental Aspects to Diagnostic Role (Comprehensive Review). Front. Mol. Biosci. 2022, 9, 841277. [Google Scholar] [CrossRef]
- Apple, F.S.; Jaffe, A.S. Men are different than women: It’s true for cardiac troponin too. Clin. Biochem. 2014, 47, 867–868. [Google Scholar] [CrossRef]
- Apple, F.S.; Panteghini, M.; Ravkilde, J.; Mair, J.; Wu, A.H.; Tate, J.; Pagani, F.; Christenson, R.H.; Jaffe, A.S. Quality Specifications for B-Type Natriuretic Peptide Assays. Clin. Chem. 2005, 51, 486–493. [Google Scholar] [CrossRef]
- Punukollu, G.; Gowda, R.M.; Khan, I.A.; Mehta, N.J.; Navarro, V.; Vasavada, B.C.; Sacchi, T.J. Elevated serum cardiac troponin I in rhabdomyolysis. Int. J. Cardiol. 2004, 96, 35–40. [Google Scholar] [CrossRef]
- Li, S.F.; Zapata, J.; Tillem, E. The prevalence of false-positive cardiac troponin I in ED patients with rhabdomyolysis. Am. J. Emerg. Med. 2005, 23, 860–863. [Google Scholar] [CrossRef] [PubMed]
- Apple, F.S.; Quist, H.E.; Doyle, P.J.; Otto, A.P.; Murakami, M.M. Plasma 99th Percentile Reference Limits for Cardiac Troponin and Creatine Kinase MB Mass for Use with European Society of Cardiology/American College of Cardiology Consensus Recommendations. Clin. Chem. 2003, 49, 1331–1336. [Google Scholar] [CrossRef] [PubMed]
- Apple, F.S.; Ler, R.; Murakami, M.M. Determination of 19 Cardiac Troponin I and T Assay 99th Percentile Values from a Common Presumably Healthy Population. Clin. Chem. 2012, 58, 1574–1581. [Google Scholar] [CrossRef] [PubMed]
- Saenger, A.; Beyrau, R.; Braun, S.; Cooray, R.; Dolci, A.; Freidank, H.; Giannitsis, E.; Gustafson, S.; Handy, B.; Katus, H.; et al. Multicenter analytical evaluation of a high-sensitivity troponin T assay. Clin. Chim. Acta 2011, 412, 748–754. [Google Scholar] [CrossRef]
- Gore, M.O.; Seliger, S.L.; Defilippi, C.R.; Nambi, V.; Christenson, R.H.; Hashim, I.A.; Hoogeveen, R.C.; Ayers, C.R.; Sun, W.; McGuire, D.K.; et al. Age- and Sex-Dependent Upper Reference Limits for the High-Sensitivity Cardiac Troponin T Assay. J. Am. Coll. Cardiol. 2014, 63, 1441–1448. [Google Scholar] [CrossRef]
- Roffi, M.; Patrono, C.; Collet, J.-P.; Mueller, C.; Valgimigli, M.; Andreotti, F.; Bax, J.J.; Borger, M.A.; Brotons, C.; Chew, D.P.; et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur. Heart J. 2016, 37, 267–315. [Google Scholar] [CrossRef]
- Mueller-Hennessen, M.; Lindahl, B.; Giannitsis, E.; Biener, M.; Vafaie, M.; Defilippi, C.R.; Christ, M.; Santalo-Bel, M.; Panteghini, M.; Plebani, M.; et al. Diagnostic and prognostic implications using age- and gender-specific cut-offs for high-sensitivity cardiac troponin T—Sub-analysis from the TRAPID-AMI study. Int. J. Cardiol. 2016, 209, 26–33. [Google Scholar] [CrossRef]
- Giménez, M.R.; Twerenbold, R.; Boeddinghaus, J.; Nestelberger, T.; Puelacher, C.; Hillinger, P.; Wildi, K.; Jaeger, C.; Grimm, K.; Heitzelmann, K.-F.; et al. Clinical Effect of Sex-Specific Cutoff Values of High-Sensitivity Cardiac Troponin T in Suspected Myocardial Infarction. JAMA Cardiol. 2016, 1, 912–920. [Google Scholar] [CrossRef]
- McRae, A.; Graham, M.; Abedin, T.; Ji, Y.; Yang, H.; Wang, D.; Southern, D.; Andruchow, J.; Lang, E.; Innes, G.; et al. Sex-specific, high-sensitivity cardiac troponin T cut-off concentrations for ruling out acute myocardial infarction with a single measurement. Can. J. Emerg. Med. 2018, 21, 26–33. [Google Scholar] [CrossRef]
- Shah, A.; Griffiths, M.; Lee, K.K.; McAllister, D.; Hunter, A.L.; Ferry, A.V.; Cruikshank, A.; Reid, A.; Stoddart, M.; Strachan, F.; et al. High sensitivity cardiac troponin and the under-diagnosis of myocardial infarction in women: Prospective cohort study. BMJ 2015, 350, g7873–h626. [Google Scholar] [CrossRef]
- Novack, V.; Cutlip, D.E.; Jotkowitz, A.; Lieberman, N.; Porath, A. Reduction in Sex-Based Mortality Difference with Implementation of New Cardiology Guidelines. Am. J. Med. 2008, 121, 597.e1–597.e8. [Google Scholar] [CrossRef]
- Trambas, C.; Pickering, J.W.; Than, M.; Bain, C.; Nie, L.; Paul, E.; Dart, A.; Broughton, A.; Schneider, H.G. Impact of High-Sensitivity Troponin I Testing with Sex-Specific Cutoffs on the Diagnosis of Acute Myocardial Infarction. Clin. Chem. 2016, 62, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Everett, B.M.; Brooks, M.M.; Vlachos, H.E.; Chaitman, B.R.; Frye, R.L.; Bhatt, D.L. Sex Differences in Cardiac Troponin and the Risk of Death or Major Cardiovascular Events. J. Am. Coll. Cardiol. 2016, 68, 978–980. [Google Scholar] [CrossRef] [PubMed]
- Chauin, A. The Main Causes and Mechanisms of Increase in Cardiac Troponin Concentrations Other Than Acute Myocardial Infarction (Part 1): Physical Exertion, Inflammatory Heart Disease, Pulmonary Embolism, Renal Failure, Sepsis. Vasc. Health Risk Manag. 2021, 17, 601–617. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M.; Dupliakov, D.V. Physical Activity and Cardiac Markers: Part 1. Human. Sport Med. 2022, 22, 15–28. (In Russian) [Google Scholar] [CrossRef]
- Lazzarino, A.I.; Hamer, M.; Gaze, D.; Collinson, P.; Steptoe, A. The Association between Cortisol Response to Mental Stress and High-Sensitivity Cardiac Troponin T Plasma Concentration in Healthy Adults. J. Am. Coll. Cardiol. 2013, 62, 1694–1701. [Google Scholar] [CrossRef]
- Aengevaeren, V.L.; Baggish, A.L.; Chung, E.H.; George, K.; Kleiven, Ø.; Mingels, A.M.; Ørn, S.; Shave, R.E.; Thompson, P.D.; Eijsvogels, T.M. Exercise-Induced Cardiac Troponin Elevations: From Underlying Mechanisms to Clinical Relevance. Circulation 2021, 144, 1955–1972. [Google Scholar] [CrossRef]
- Chaulin, A.M.; Duplyakova, P.D.; Duplyakov, D.V. Circadian rhythms of cardiac troponins: Mechanisms and clinical significance. Russ. J. Cardiol. 2020, 25, 4061. [Google Scholar] [CrossRef]
- Jan, M.I.; Khan, R.A.; Fozia, U.; Ahmad, I.; Khan, N.; Urooj, K.; Shah, A.U.H.A.; Khan, A.U.; Ali, T.; Ishtiaq, A.; et al. C-Reactive Protein and High-Sensitive Cardiac Troponins Correlate with Oxidative Stress in Valvular Heart Disease Patients. Oxid. Med. Cell. Longev. 2022, 2022, 5029853. [Google Scholar] [CrossRef]
- Chaulin, A.M. Phosphorylation and Fragmentation of the Cardiac Troponin T: Mechanisms, Role in Pathophysiology and Laboratory Diagnosis. Int. J. Biomed. 2021, 11, 250–259. [Google Scholar] [CrossRef]
- Chaulin, A.M.; Duplyakov, D.V. Arrhythmogenic effects of doxorubicin. Complex Issues Cardiovasc. Dis. 2020, 9, 69–80. (In Russian) [Google Scholar] [CrossRef]
- Kong, Z.; Nie, J.; Lin, H.; George, K.; Zhao, G.; Zhang, H.; Tong, T.K.; Shi, Q. Sex differences in release of cardiac troponin T after endurance exercise. Biomarkers 2016, 22, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Tiller, N.B.; Wheatley-Guy, C.M.; Fermoyle, C.C.; Robach, P.; Ziegler, B.; Gavet, A.; Schwartz, J.C.; Taylor, B.J.; Constantini, K.; Murdock, R.; et al. Sex-specific Physiological Responses to Ultramarathon. Med. Sci. Sports Exerc. 2022, 54, 1647–1656. [Google Scholar] [CrossRef]
- Schwarzenberger, J.C.; Sun, L.S.; Pesce, M.A.; Heyer, E.J.; Delphin, E.; Almeida, G.M.; Wood, M. Sex-based differences in serum cardiac troponin I, a specific marker for myocardial injury, after cardiac surgery. Crit. Care Med. 2003, 31, 689–693. [Google Scholar] [CrossRef]
- Mendelsohn, M.E. Protective effects of estrogen on the cardiovascular system. Am. J. Cardiol. 2002, 89, 12E–17E; discussion 17E–18E. [Google Scholar] [CrossRef]
- Stearns, J.D.; Dávila-Román, V.G.; Barzilai, B.; Thompson, R.E.; Grogan, K.L.; Thomas, B.; Hogue, C.W. Prognostic Value of Troponin I Levels for Predicting Adverse Cardiovascular Outcomes in Postmenopausal Women Undergoing Cardiac Surgery. Anesth. Analg. 2009, 108, 719–726. [Google Scholar] [CrossRef]
- Bergmann, O.; Bhardwaj, R.D.; Bernard, S.; Zdunek, S.; Barnabé-Heider, F.; Walsh, S.; Zupicich, J.; Alkass, K.; Buchholz, B.A.; Druid, H.; et al. Evidence for Cardiomyocyte Renewal in Humans. Science 2009, 324, 98–102. [Google Scholar] [CrossRef]
- Graham, E.; Bergmann, O. Dating the Heart: Exploring Cardiomyocyte Renewal in Humans. Physiology 2017, 32, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Derks, W.; Bergmann, O. Polyploidy in Cardiomyocytes: Roadblock to Heart Regeneration? Circ. Res. 2020, 126, 552–565. [Google Scholar] [CrossRef]
- Chaulin, A.M. Biology of Cardiac Troponins: Emphasis on Metabolism. Biology 2022, 11, 429. [Google Scholar] [CrossRef]
- White, H.D. Pathobiology of Troponin Elevations: Do Elevations Occur with Myocardial Ischemia as Well as Necrosis? J. Am. Coll. Cardiol. 2011, 57, 2406–2408. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M. Main analytical characteristics of laboratory methods for the determination of cardiac troponins: A review from the historical and modern points of view. Orv. Hetil. 2022, 163, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Uçar, H.; Gür, M.; Kivrak, A.; Koyunsever, N.Y.; Seker, T.; Akilli, R.E.; Türkoğlu, C.; Kaypakli, O.; Sahin, D.Y.; Elbasan, Z.; et al. High-sensitivity cardiac troponin T levels in newly diagnosed hypertensive patients with different left ventricle geometry. Blood Press. 2013, 23, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M. Updated information about methods of identification and diagnostic opportunities of cardiac troponins. Riv. Ital. Della Med. Lab. 2021, 17, 154–164. [Google Scholar] [CrossRef]
- Chaulin, A.M.; Duplyakov, D.V. Environmental factors and cardiovascular diseases. Hyg. Sanit. 2021, 100, 223–228. (In Russian) [Google Scholar] [CrossRef]
- Neal, R.C.; Ferdinand, K.C.; Ycas, J.; Miller, E. Relationship of Ethnic Origin, Gender, and Age to Blood Creatine Kinase Levels. Am. J. Med. 2009, 122, 73–78. [Google Scholar] [CrossRef]
- Chaulin, A.M. False-Positive Causes in Serum Cardiac Troponin Levels. J. Clin. Med. Res. 2022, 14, 80–87. [Google Scholar] [CrossRef]
- Clifford, S.M.; Bunker, A.M.; Jacobsen, J.R.; Roberts, W.L. Age and gender specific pediatric reference intervals for aldolase, amylase, ceruloplasmin, creatine kinase, pancreatic amylase, prealbumin, and uric acid. Clin. Chim. Acta 2011, 412, 788–790. [Google Scholar] [CrossRef]
- Zaninotto, M.; Mion, M.; Novello, E.; Delprete, E.; Moretti, M.; Plebani, M. Creatine-kinase MB mass: Age and sex-associated reference limits in two different platforms that use the same method. Clin. Chim. Acta 2009, 401, 162–164. [Google Scholar] [CrossRef]
- Emokpae, M.A.; Nwagbara, G.O.N.A. Serum Creatine Kinase-MB Isoenzyme Activity among Subjects with Uncomplicated Essential Hypertension: Any Sex Differences. Med.Sci. 2017, 5, 8. [Google Scholar] [CrossRef]
- Mirzaii-Dizgah, I.; Hejazi, S.F.; Riahi, E.; Salehi, M.M. Saliva-based creatine kinase MB measurement as a potential point-of-care testing for detection of myocardial infarction. Clin. Oral. Investig. 2012, 16, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M. On the Effect of Heterophilic Antibodies on Serum Levels of Cardiac Troponins: A Brief Descriptive Review. Life 2022, 12, 1114. [Google Scholar] [CrossRef] [PubMed]
- Chaulin, A.M.; Sergeev, A.K. The Role of Fine Particles (PM 2.5) in the Genesis of Atherosclerosis and Myocardial Damage: Emphasis on Clinical and Epidemiological Data, and Pathophysiological Mechanisms. Cardiol. Res. 2022, 13, 268–282. [Google Scholar] [CrossRef] [PubMed]
- Strunz, C.; Araki, L.; Nogueira, A.; Mansur, A. Gender differences in serum CK-MB mass levels in healthy Braziliansubjects. Braz. J. Med. Biol. Res. 2011, 44, 236–239. [Google Scholar] [CrossRef]
| The Nature of Myocardial Damage | Nosologies/Reasons |
|---|---|
| Myocardial damage caused by ischemia | Myocardial infarction |
| Myocardial damage not caused by ischemia in cardiac pathologies |
|
| Myocardial damage not caused by ischemia in extracardiac pathologies |
|
| Physiological conditions |
|
| Without myocardial damage (false positive factors) |
|
| Manufacturer of the Immunotest | LoD, ng/L | CV, % | Characteristics of the Examined Healthy Population, Number, Gender, Age | 99 Percentile, ng/L | Type of Test Sample | Percentage of Measurable Values (Above LoD, But Less Than 99 Percentile), % |
|---|---|---|---|---|---|---|
| Abbott/Alinity i systems/Alinity i STAT High Sensitive Troponin-I; commercial-OUS | 1.6 | 4.0 | n (21–75 years old) = 1530, including M (21–75 years) = 764, W (21–75 years) = 766 | Total = 26.2. For M = 34.2. For W = 15.6 | Plasma with anticoagulants (lithium heparin or K2EDTA, or K3EDTA) or Serum | Total = 85, for M = 92. for W = 78 |
| Beckman Coulter/Access 2, DxI/Access hsTnI; commercial—OUS | 1.0–2.3 | 3.7 | n (21–99 years old) = 1089, including M = 595, W = 494 | Total = 17.5. For M = 19.8. For W = 11.6. | Plasma containing lithium heparin | Total > 50, no data on gender characteristics |
| bioMérieux VIDAS High Sensitive Troponin I; commercial | 3.2 | 7.0 | n (41–80 years old) = 815, including M (41–80 years old) = 447, W (41–80 years old) = 368. | Total = 19.0. For M = 25.0. For W = 11.0. | Serum or Plasma with anticoagulant (lithium heparin) | No data provided |
| ET Healthcare Pylon hsTnI assay; China FDA approved | 1.2–1.4 | 10.0 | n (15–91 years old) = 863, including M = 425, W = 438. | Total = 27.0. For M = 27.0. For W = 21.0. | Plasma with anticoagulant (EDTA) or serum or whole blood | Total = 91, for M = 94, for W = 89 |
| Fujirebio Lumipulse G G1200 and G600II hsTnI | 2.1 | ≤4.6 | n (18–90 years old) = 1018, including M = 590, W = 428. | Total = 29.6%, For M = 32.8. For W = 27.8. | Plasma with anticoagulant (lithium heparin) | Total = 65, no data on gender characteristics |
| LSI Medience (formerly Mitsubishi) PATHFAST cTnI; commercial | 1.0 | <6.0 | n (18–86 years old) = 474, including M = 238, W = 236 | Total = 15.48. For M = 16.91. For W = 11.46 | Plasma with anticoagulant (sodium heparin or lithium heparin or EDTA) | Total = 76.3, no data on gender characteristics |
| Ortho/VITROS/hsTroponin I; commercial | 0.39–0.86 | <10.0 | n (22–91 years) = 952, including M = 466, W = 486 | Total = 11.0. For M = 12.0. For W = 9.0. | Blood serum | Total > 50, no data on gender characteristics |
| Quidel/Alere Triage True hs-cTnI | 0.7–1.6 | 5.0–5.9 | n = 789, including M = 398, W = 391 | Total = 20.5. For M = 25.7. For W = 14.4 | Blood plasma with anticoagulant EDTA | Total ≥ 50, no data on gender characteristics |
| Roche/cobas e601, e602, E170/cTnT-hs 18-min; commercial | 2.05 | <10 | n (20–71 years old) = 533, including W = 49.7% | Total = 14.0. For M = 16.0. For W = 9 | Serum or plasma with anticoagulants (EDTA, heparin) | Total > 71.5, no data on gender characteristics |
| Siemens ATELLICA High-Sensitivity TnI (TnIH), US and OUS; commercial | 1.6 | <4.0 | n (22–91 years old) = 2007, including M = 1000, W = 1007. | Total = 45.2. For M = 53.5. For W = 34.1. | Serum or plasma with anticoagulant (heparin) | Total = 71, for M = 84, for W = 58. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaulin, A.M. Gender Specificities of Cardiac Troponin Serum Levels: From Formation Mechanisms to the Diagnostic Role in Case of Acute Coronary Syndrome. Life 2023, 13, 267. https://doi.org/10.3390/life13020267
Chaulin AM. Gender Specificities of Cardiac Troponin Serum Levels: From Formation Mechanisms to the Diagnostic Role in Case of Acute Coronary Syndrome. Life. 2023; 13(2):267. https://doi.org/10.3390/life13020267
Chicago/Turabian StyleChaulin, Aleksey Michailovich. 2023. "Gender Specificities of Cardiac Troponin Serum Levels: From Formation Mechanisms to the Diagnostic Role in Case of Acute Coronary Syndrome" Life 13, no. 2: 267. https://doi.org/10.3390/life13020267
APA StyleChaulin, A. M. (2023). Gender Specificities of Cardiac Troponin Serum Levels: From Formation Mechanisms to the Diagnostic Role in Case of Acute Coronary Syndrome. Life, 13(2), 267. https://doi.org/10.3390/life13020267






