pH-Dependence Cytotoxicity Evaluation of Artepillin C against Tumor Cells
Abstract
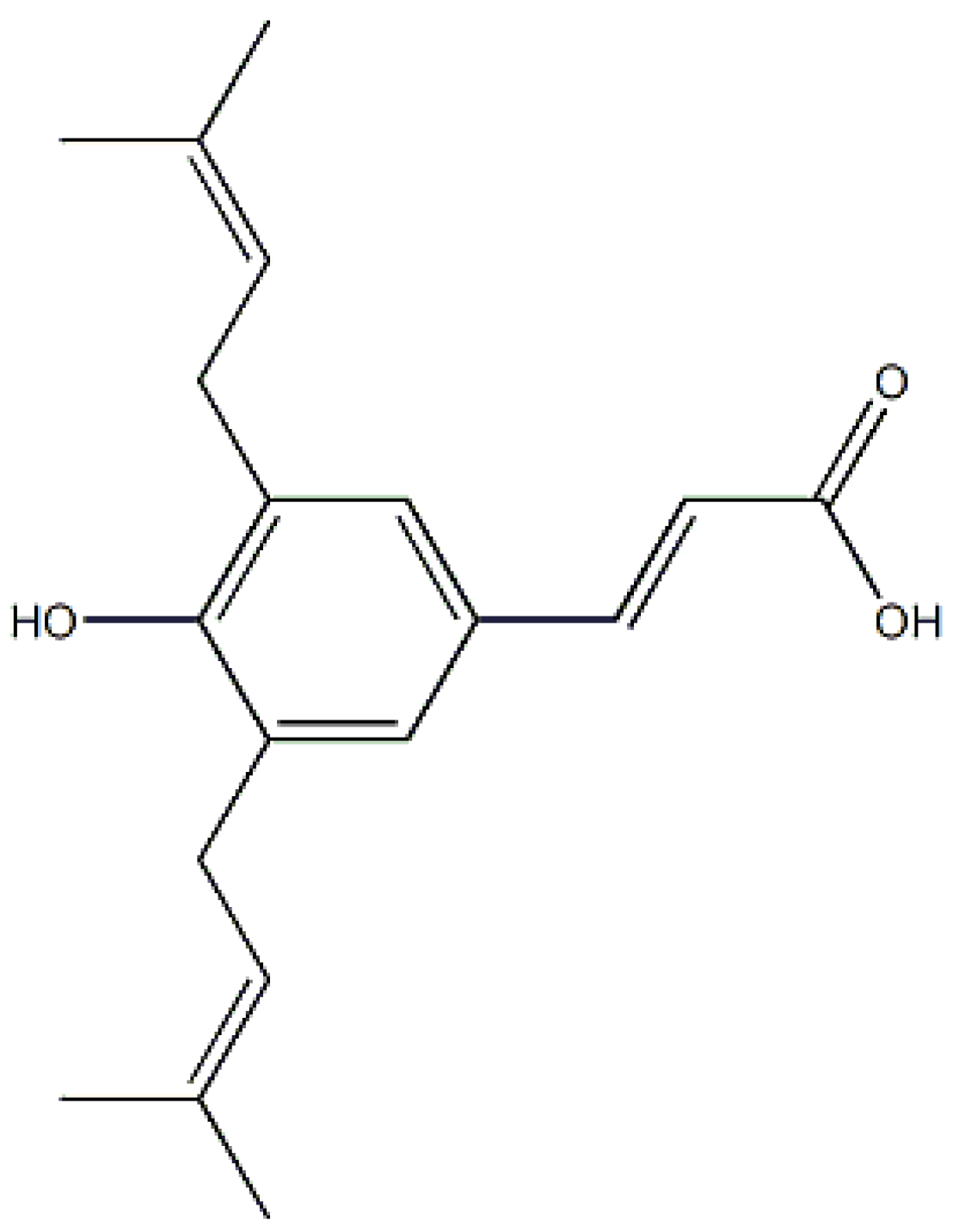
:1. Introduction
2. Materials and Methods
2.1. Artepillin C Solubilization
2.2. In Vitro Culture of Fibroblasts and Glioblastoma Cells
2.3. MTT and LDH Assays
2.4. Microscopy Analyses
3. Results and Discussion
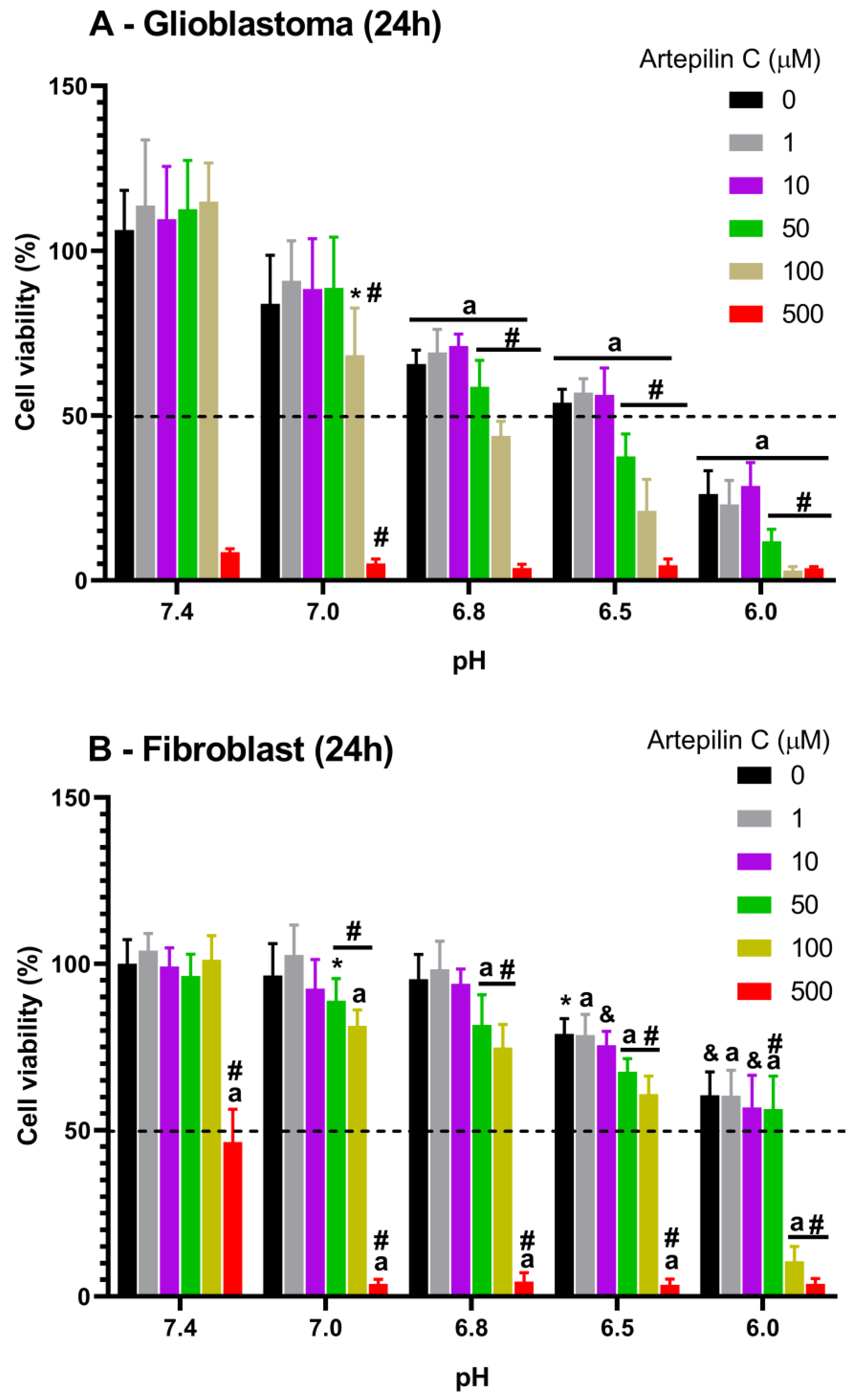
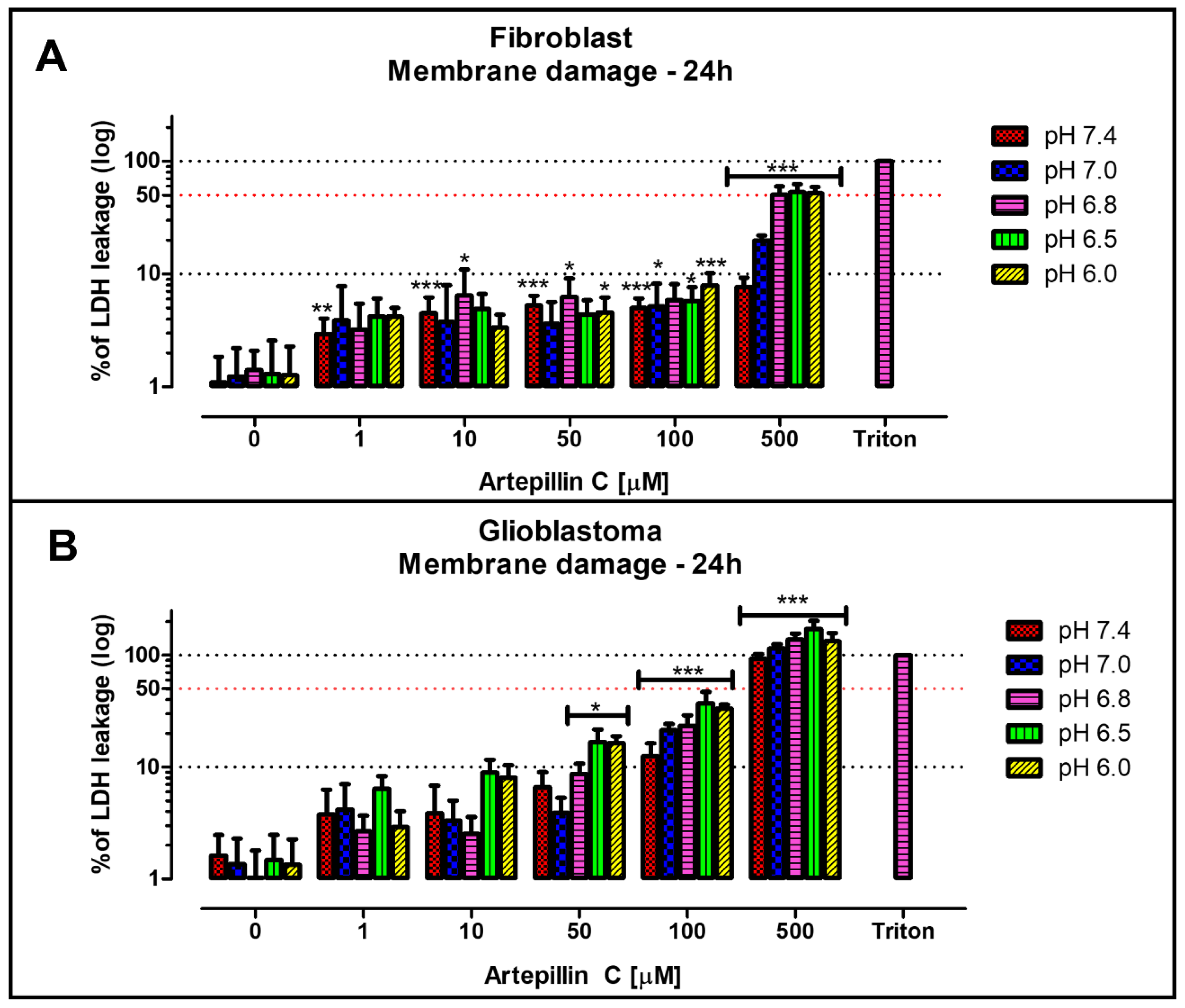
3.1. Cell Viability Studies
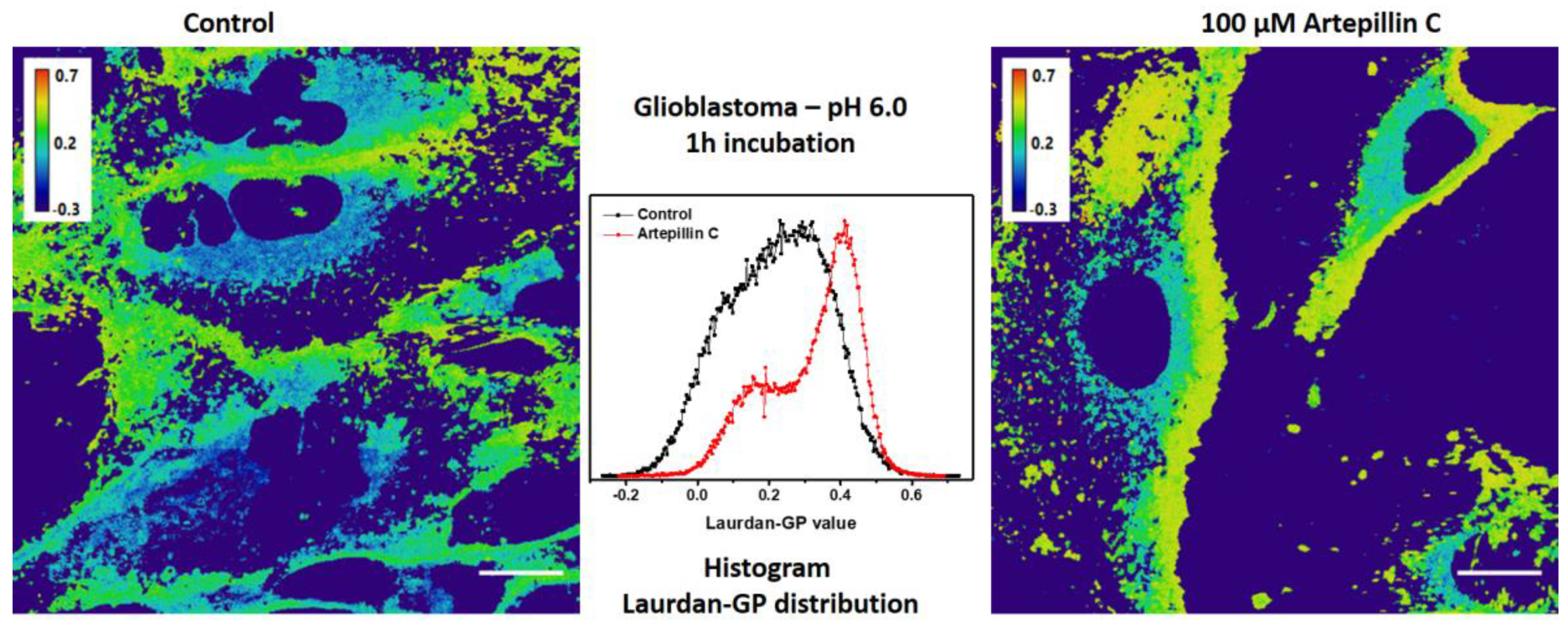
3.2. Live Cell Imaging
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mao, J.J.; Pillai, G.G.; Andrade, C.J.; Ligibel, J.A.; Basu, P.; Cohen, L.; Khan, I.A.; Mustian, K.M.; Puthiyedath, R.; Dhiman, K.S.; et al. Integrative oncology: Addressing the global challenges of cancer prevention and treatment. CA Cancer J. Clin. 2022, 72, 144–164. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Cancer. 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 12 February 2023).
- Huang, M.; Lu, J.J.; Ding, J. Natural Products in Cancer Therapy: Past, Present and Future. Nat. Prod. Bioprospect. 2021, 11, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef] [PubMed]
- Sulaeman, A.; Fikri, A.M.; Mulyati, A.H. The bee propolis for preventing and healing non-communicable diseases. In Functional Foods and Nutraceuticals in Metabolic and Non-Communicable Diseases; Elsevier: Amsterdam, The Netherlands, 2022; pp. 465–479. [Google Scholar] [CrossRef]
- Salatino, A.; Salatino, M.L.F.; Negri, G. How diverse is the chemistry and plant origin of Brazilian propolis? Apidologie 2021, 52, 1075–1097. [Google Scholar] [CrossRef]
- Bhargava, P.; Mahanta, D.; Kaul, A.; Ishida, Y.; Terao, K.; Wadhwa, R.; Kaul, S.C. Experimental Evidence for Therapeutic Potentials of Propolis. Nutrients 2021, 13, 2528. [Google Scholar] [CrossRef]
- Kuropatnicki, A.K.; Szliszka, E.; Krol, W. Historical Aspects of Propolis Research in Modern Times. Evid. Based Complement. Altern. Med. 2013, 2013, 964149. [Google Scholar] [CrossRef]
- Dantas Silva, R.P.; Machado, B.A.S.; de Abreu Barreto, G.; Costa, S.S.; Andrade, L.N.; Amaral, R.G.; Carvalho, A.A.; Padilha, F.F.; Barbosa, J.D.V.; Umsza-Guez, M.A. Antioxidant, antimicrobial, antiparasitic, and cytotoxic properties of various Brazilian propolis extracts. PLoS ONE 2017, 12, e0172585. [Google Scholar] [CrossRef]
- Shahinozzaman, M.; Basak, B.; Emran, R.; Rozario, P.; Obanda, D.N. Artepillin C: A comprehensive review of its chemistry, bioavailability, and pharmacological properties. Fitoterapia 2020, 147, 104775. [Google Scholar] [CrossRef]
- Huang, S.; Zhang, C.-P.; Wang, K.; Li, G.; Hu, F.-L. Recent Advances in the Chemical Composition of Propolis. Molecules 2014, 19, 19610–19632. [Google Scholar] [CrossRef]
- Damodaran, T. Propolis. In Nutraceuticals; Elsevier: Amsterdam, The Netherlands, 2021; pp. 795–812. [Google Scholar] [CrossRef]
- Kumazawa, S.; Hamasaka, T.; Nakayama, T. Antioxidant activity of propolis of various geographic origins. Food Chem. 2004, 84, 329–339. [Google Scholar] [CrossRef]
- Hamasaka, T.; Kumazawa, S.; Fujimoto, T.; Nakayama, T. Antioxidant activity and constituents of propolis collected in various areas of Japan. Food Sci. Technol. Res. Res. 2004, 10, 86–92. [Google Scholar] [CrossRef]
- Veiga, R.; De Mendonça, S.; Mendes, P.; Paulino, N.; Mimica, M.; Netto, A.L.; Lira, I.; López, B.-C.; Negrão, V.; Marcucci, M. Artepillin C and phenolic compounds responsible for antimicrobial and antioxidant activity of green propolis and Baccharis dracunculifolia DC. J. Appl. Microbiol. 2017, 122, 911–920. [Google Scholar] [CrossRef]
- de Oliveira, P.F.; Lima, I.M.D.S.; Munari, C.C.; Bastos, J.K.; Filho, A.A.D.S.; Tavares, D.C. Comparative evaluation of antiproliferative effects of brazilian green propolis, its main source baccharis dracunculifolia, and their major constituents artepillin C and baccharin. Planta Med. 2014, 80, 490–492. [Google Scholar] [CrossRef] [PubMed]
- Camuri, I.J.; Costa, A.B.; Ito, A.S.; Pazin, W.M. Optical absorption and fluorescence spectroscopy studies of Artepillin C, the major component of green propolis. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 198, 71–77. [Google Scholar] [CrossRef]
- Camuri, I.J.; da Costa, A.B.; Ito, A.S.; Pazin, W.M. pH and Charge Effects Behind the Interaction of Artepillin C, the Major Component of Green Propolis, With Amphiphilic Aggregates: Optical Absorption and Fluorescence Spectroscopy Studies. Photochem. Photobiol. 2019, 95, 1345–1351. [Google Scholar] [CrossRef] [PubMed]
- Heiden, M.G.V.; Cantley, L.C.; Thompson, C.B. Understanding the warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Liberti, M.V.; Locasale, J.W.; Biology, C.; Biology, C. The Warburg Effect: How Does it Benefit Cancer Cells? Trends Biochem. Sci. 2017, 41, 211–218. [Google Scholar] [CrossRef]
- Romero-Garcia, S.; Moreno-Altamirano, M.M.B.; Prado-Garcia, H.; Sánchez-García, F.J. Lactate contribution to the tumor microenvironment: Mechanisms, effects on immune cells and therapeutic relevance. Front. Immunol. 2016, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Pazin, W.M.; Ruiz, G.C.M.; de Oliveira, O.N.; Constantino, C.J.L. Interaction of Artepillin C with model membranes: Effects of pH and ionic strength. Biochim. Biophys. Acta Biomembr. 2019, 1861, 410–417. [Google Scholar] [CrossRef]
- Pazin, W.M.; Ruiz, G.C.M.; dos Santos, M.J.; Aoki, P.H.B.; Ito, A.S.; Constantino, C.J.L. The protective effect of Artepillin C against lipid oxidation on model membranes. J. Mol. Liq. 2021, 324, 115089. [Google Scholar] [CrossRef]
- Evans, C.L.; Xie, X.S. Coherent Anti-Stokes Raman Scattering Microscopy: Chemical Imaging for Biology and Medicine. Annu. Rev. Anal. Chem. 2008, 1, 883–909. [Google Scholar] [CrossRef] [PubMed]
- Bagatolli, L.A. To see or not to see: Lateral organization of biological membranes and fluorescence microscopy. Biochim. Biophys. Acta Biomembr. 2006, 1758, 1541–1556. [Google Scholar] [CrossRef] [PubMed]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A summary. Neuro. Oncol. 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Alves, A.L.V.; Gomes, I.N.F.; Carloni, A.C.; Rosa, M.N.; da Silva, L.S.; Evangelista, A.F.; Reis, R.M.; Silva, V.A.O. Role of Glioblastoma Stem Cells in Cancer Therapeutic Resistance: A Perspective on Antineoplastic Agents from Natural Sources and Chemical Derivatives. Stem Cell Res. Ther. 2021, 12, 206. [Google Scholar] [CrossRef]
- Rahimifard, M.; Haghi-Aminjan, H.; Hadjighassem, M.; Jaktaji, R.P.; Bagheri, Z.; Movahed, M.A.; Zarghi, A.; Pourahmad, J. Assessment of cytotoxic effects of new derivatives of pyrazino[1,2-a] benzimidazole on isolated human glioblastoma cells and mitochondria. Life Sci. 2021, 286, 120022. [Google Scholar] [CrossRef]
- Schaefer, S.; Svenstrup, T.H.; Fischer, M.; Guerra, B. D11-mediated inhibition of protein kinase CK2 impairs HIF-1α-mediated signaling in human glioblastoma cells. Pharmaceuticals 2017, 10, 5. [Google Scholar] [CrossRef]
- Hjelmeland, A.B.; Wu, Q.; Heddleston, J.M.; Choudhary, G.S.; MacSwords, J.; Lathia, J.D.; McLendon, R.; Lindner, D.; Sloan, A.; Rich, J.N. Acidic stress promotes a glioma stem cell phenotype. Cell Death Differ. 2011, 18, 829–840. [Google Scholar] [CrossRef]
- van Meerloo, J.; Kaspers, G.J.L.; Cloos, J. Cell Sensitivity Assays: The MTT Assay. Methods Mol. Biol. 2011, 731, 237–245. [Google Scholar]
- Kumar, P.; Nagarajan, A.; Uchil, P.D. Analysis of Cell Viability by the Lactate Dehydrogenase Assay. Cold Spring Harb. Protoc. 2018, 2018, 465–468. [Google Scholar] [CrossRef]
- Brewer, J.; de la Serna, J.B.; Wagner, K.; Bagatolli, L.A. Multiphoton excitation fluorescence microscopy in planar membrane systems. Biochim. Biophys. Acta 2010, 1798, 1301–1308. [Google Scholar] [CrossRef]
- Kobal, M.B.; Pazin, W.M.; Bistaffa, M.J.; Constantino, C.J.; Toledo, K.A.; Aoki, P.H. Correlating Artepillin C cytotoxic activity on HEp-2 cells with bioinspired systems of plasma membranes. Mater. Sci. Eng. C 2020, 112, 110943. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, T.; Zhang, J.; Xu, S.; Liu, H. Disruption of Tumor Cells Using a PH-Activated and Thermosensitive Antitumor Lipopeptide Containing a Leucine Zipper Structure. Langmuir 2018, 34, 8818–8827. [Google Scholar] [CrossRef] [PubMed]
- Navrátilová, J.; Hankeová, T.; Beneš, P.; Šmarda, J. Acidic PH of Tumor Microenvironment Enhances Cytotoxicity of the Disulfiram/Cu2+ Complex to Breast and Colon Cancer Cells. Chemotherapy 2013, 59, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Calcinotto, A.; Filipazzi, P.; Grioni, M.; Iero, M.; De Milito, A.; Ricupito, A.; Cova, A.; Canese, R.; Jachetti, E.; Rossetti, M.; et al. Modulation of Microenvironment Acidity Reverses Anergy in Human and Murine Tumor-Infiltrating T Lymphocytes. Cancer Res. 2012, 72, 2746–2756. [Google Scholar] [CrossRef] [PubMed]
- Valiahdi, S.M.; Egger, A.E.; Miklos, W.; Jungwirth, U.; Meelich, K.; Nock, P.; Berger, W.; Hartinger, C.G.; Galanski, M.S.; Jakupec, M.A.; et al. Influence of Extracellular PH on the Cytotoxicity, Cellular Accumulation, and DNA Interaction of Novel PH-Sensitive 2-Aminoalcoholatoplatinum (II) Complexes. JBIC J. Biol. Inorg. Chem. 2013, 18, 249–260. [Google Scholar] [CrossRef]
- Swietach, P.; Vaughan-Jones, R.D.; Harris, A.L.; Hulikova, A. The Chemistry, Physiology and Pathology of PH in Cancer. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130099. [Google Scholar] [CrossRef]
- Wehr, J.; Sikorski, E.L.; Bloch, E.; Feigman, M.S.; Ferraro, N.J.; Baybutt, T.R.; Snook, A.E.; Pires, M.M.; Thévenin, D. PH-Dependent Grafting of Cancer Cells with Antigenic Epitopes Promotes Selective Antibody-Mediated Cytotoxicity. J. Med. Chem. 2020, 63, 3713–3722. [Google Scholar] [CrossRef]
- Pizzirusso, A.; De Nicola, A.; Sevink, G.J.A.; Correa, A.; Cascella, M.; Kawakatsu, T.; Rocco, M.; Zhao, Y.; Celino, M.; Milano, G. Biomembrane solubilization mechanism by Triton X-100: A computational study of the three stage model. Phys. Chem. Chem. Phys. 2017, 19, 29780–29794. [Google Scholar] [CrossRef]
- De Gelder, J.; De Gussem, K.; Vandenabeele, P.; Moens, L. Reference database of Raman spectra of biological molecules. J. Raman Spectrosc. 2007, 38, 1133–1147. [Google Scholar] [CrossRef]
- Wang, S.X.; Nakamura, N.; Mure, M.; Klinman, J.P.; Sanders-Loehr, J. Characterization of the Native Lysine Tyrosylquinone Cofactor in Lysyl Oxidase by Raman Spectroscopy. J. Biol. Chem. 1997, 272, 28841–28844. [Google Scholar] [CrossRef]
- Owen, D.M.; Rentero, C.; Magenau, A.; Abu-Siniyeh, A.; Gaus, K. Quantitative Imaging of Membrane Lipid Order in Cells and Organisms. Nat. Protoc. 2012, 7, 24–35. [Google Scholar]
- Ventura, A.E.; Pokorna, S.; Huhn, N.; Santos, T.C.B.; Prieto, M.; Futerman, A.H.; Silva, L.C. Cell Lipid Droplet Heterogeneity and Altered Biophysical Properties Induced by Cell Stress and Metabolic Imbalance. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2023, 1868, 159347. [Google Scholar] [CrossRef] [PubMed]
- Endo, S.; Hoshi, M.; Matsunaga, T.; Inoue, T.; Ichihara, K.; Ikari, A. Autophagy inhibition enhances anticancer efficacy of artepillin C, a cinnamic acid derivative in Brazilian green propolis. Biochem. Biophys. Res. Commun. 2018, 497, 437–443. [Google Scholar] [CrossRef] [PubMed]


| pH | Incubation Time | Laurdan GP Values | |||
|---|---|---|---|---|---|
| Fibroblasts | Glioblastomas | ||||
| Control | 100 µM Artepillin C | Control | 100 µM Artepillin C | ||
| 7.4 | 24 h | 0.14 ± 0.02 | 0.21 ± 0.03 | 0.23 ± 0.01 | 0.26 ± 0.01 |
| 0.35 ± 0.04 | 0.37 ± 0.05 | 0.37 ± 0.03 | 0.35 ± 0.03 | ||
| 6.0 | 1 h | 0.16 ± 0.01 | 0.26 ± 0.05 | 0.16 ± 0.01 | 0.31 ± 0.01 |
| 0.34 ± 0.02 | 0.37 ± 0.03 | 0.37 ± 0.01 | 0.40 ± 0.01 | ||
| 4 h | 0.18 ± 0.01 | 0.30 ± 0.01 | 0.19 ± 0.07 | 0.30 ± 0.01 | |
| 0.34 ± 0.01 | 0.38 ± 0.02 | 0.37 ± 0.02 | 0.40 ± 0.01 | ||
| 8 h | 0.21 ± 0.03 | 0.37 ± 0.02 | 0.19 ± 0.02 | 0.35 ± 0.01 | |
| 0.33 ± 0.03 | 0.40 ± 0.01 | 0.34 ± 0.04 | 0.40 ± 0.02 | ||
| 12 h | 0.27 ± 0.04 | 0.27 ± 0.02 | 0.22 ± 0.04 | 0.27 ± 0.02 | |
| 0.34 ± 0.02 | 0.31 ± 0.01 | 0.38 ± 0.01 | 0.39 ± 0.01 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pazin, W.M.; Miranda, R.R.; Toledo, K.A.; Kjeldsen, F.; Constantino, C.J.L.; Brewer, J.R. pH-Dependence Cytotoxicity Evaluation of Artepillin C against Tumor Cells. Life 2023, 13, 2186. https://doi.org/10.3390/life13112186
Pazin WM, Miranda RR, Toledo KA, Kjeldsen F, Constantino CJL, Brewer JR. pH-Dependence Cytotoxicity Evaluation of Artepillin C against Tumor Cells. Life. 2023; 13(11):2186. https://doi.org/10.3390/life13112186
Chicago/Turabian StylePazin, Wallance M., Renata R. Miranda, Karina A. Toledo, Frank Kjeldsen, Carlos J. L. Constantino, and Jonathan R. Brewer. 2023. "pH-Dependence Cytotoxicity Evaluation of Artepillin C against Tumor Cells" Life 13, no. 11: 2186. https://doi.org/10.3390/life13112186
APA StylePazin, W. M., Miranda, R. R., Toledo, K. A., Kjeldsen, F., Constantino, C. J. L., & Brewer, J. R. (2023). pH-Dependence Cytotoxicity Evaluation of Artepillin C against Tumor Cells. Life, 13(11), 2186. https://doi.org/10.3390/life13112186







