The Insulin-like Growth Factor System and Colorectal Cancer
Abstract
1. Introduction
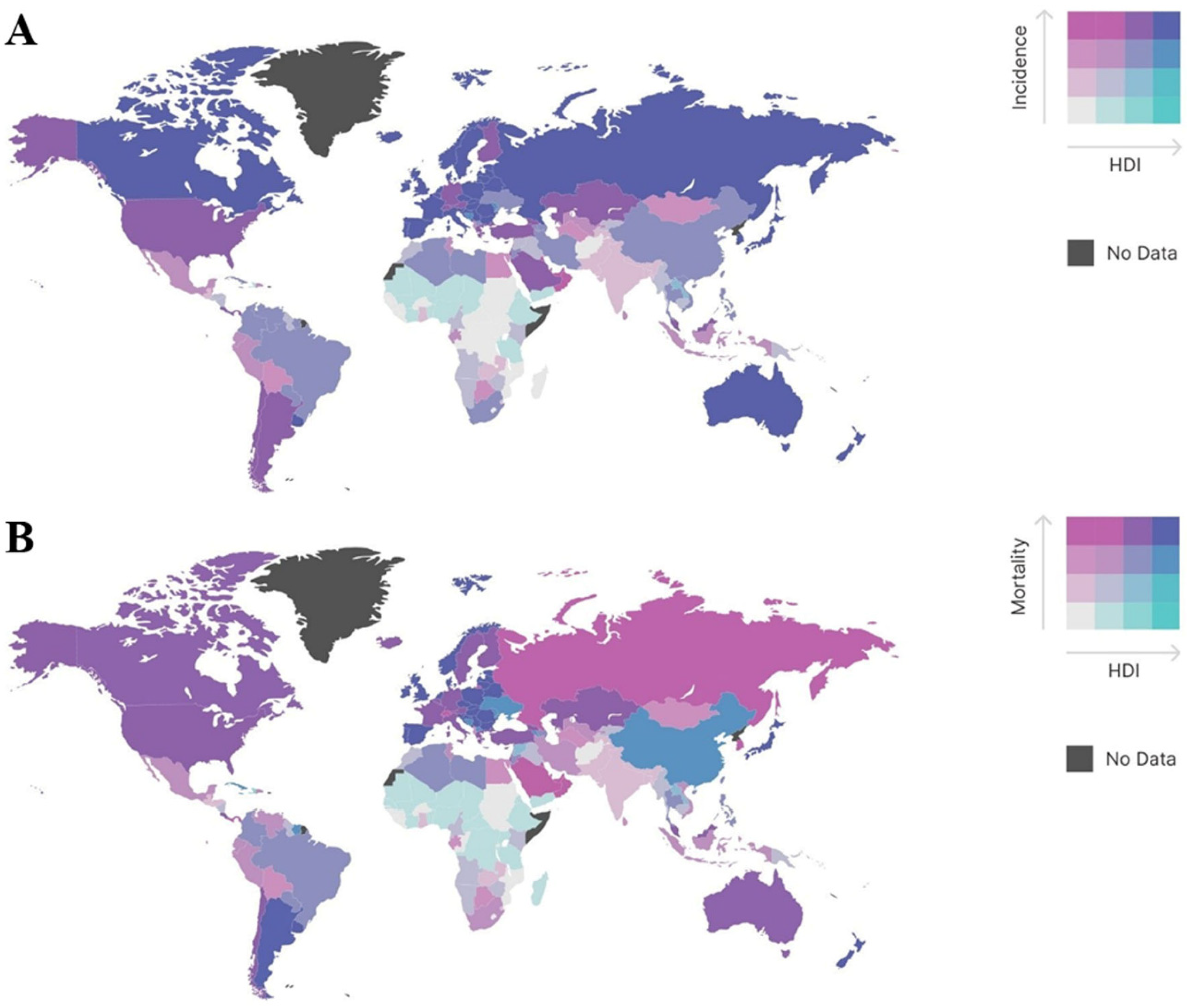
2. Epidemiology of Colorectal Cancer
3. Pathophysiology of the Colorectal Cancer
4. Insulin-like Growth Factors and Colorectal Cancer
4.1. Insulin-like Growth Factors
4.2. Insulin-like Growth Factor Receptors
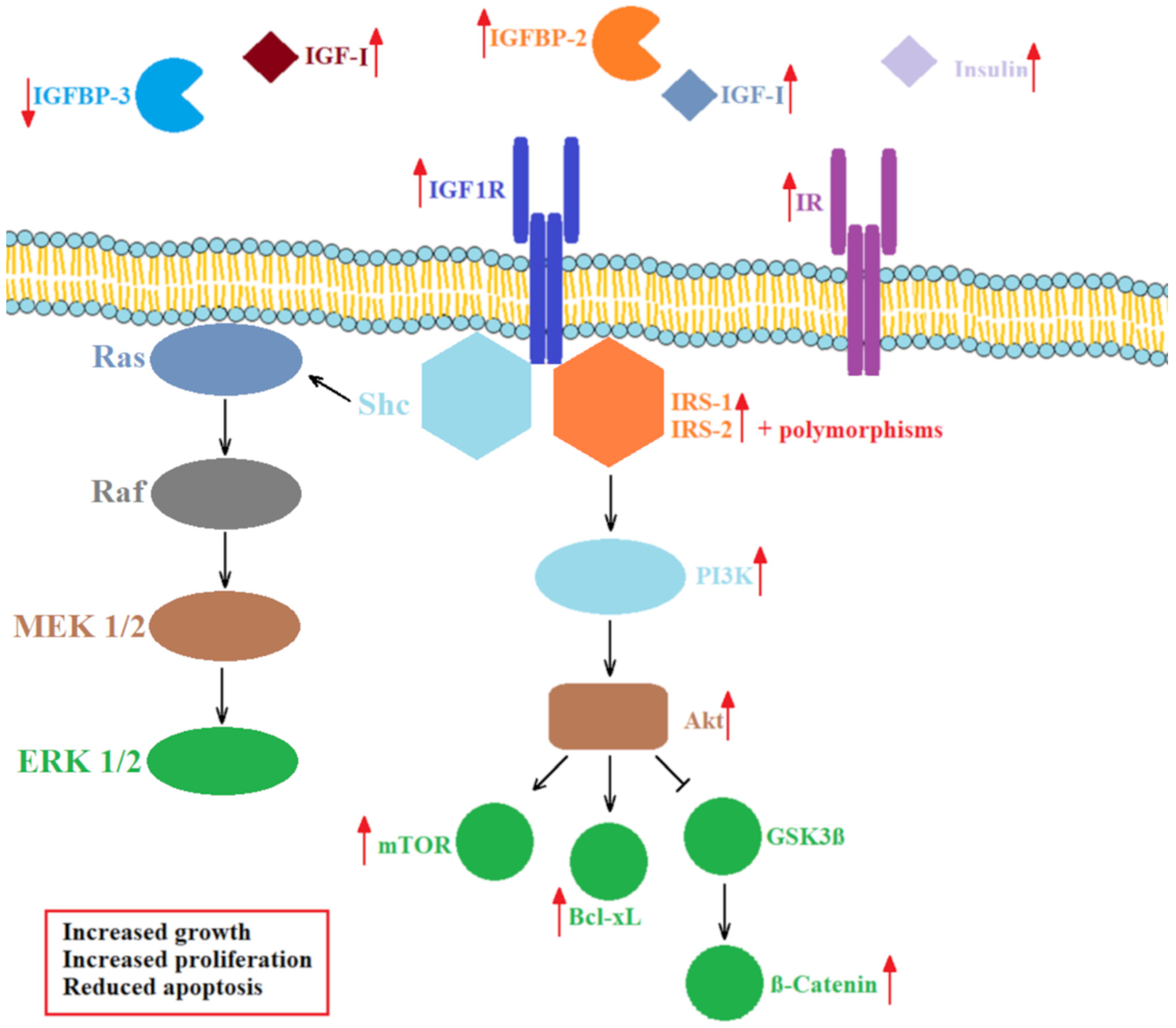
4.3. Cell Signaling via Insulin-like Growth Factor Receptors
4.4. Insulin-like Growth Factor Binding Proteins
4.5. Insulin-like Growth Factor Binding Protein Proteinases
4.6. Genetic, Epigenetic and Post-Transcriptional Regulators of the Insulin-like Growth Factor System
4.6.1. Genetic Variants
4.6.2. DNA Methylation and Imprinting
4.6.3. Regulatory RNAs
4.6.4. RNA-Binding Proteins
4.7. Insulin-like Growth Factor Signaling and the Associated Signaling Pathways
5. Therapeutic Potential of the Insulin-like Growth Factor Signaling Pathways in Treating Colorectal Cancer
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Weiderpass, E.; Soerjomataram, I. The Ever-increasing Importance of Cancer as a Leading Cause of Premature Death Worldwide. Cancer 2021, 127, 3029–3030. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Global Health Estimates 2020: Deaths by Cause, Age, Sex, by Country and by Region. 2000–2019. Available online: https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates/ghe-leading-causes-of-death (accessed on 15 April 2022).
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Patterns and Trends in Colorectal Cancer Incidence and Mortality. Gut 2017, 66, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Li, Y.; Wang, W.; Feng, W.; Shi, O.; Wang, Q. International Incidence Trends in Early- and Late-Onset Colorectal Cancer: A Population-Based Study. Int. J. Colorectal Dis. 2020, 35, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- United Nations Development Programme. Human Development Reports. 2020. Available online: https://hdr.undp.org/content/human-development-report-2020 (accessed on 20 April 2022).
- Chung, R.Y.-N.; Tsoi, K.K.F.; Kyaw, M.H.; Lui, A.R.; Lai, F.T.T.; Sung, J.J.-Y. A Population-Based Age-Period-Cohort Study of Colorectal Cancer Incidence Comparing Asia against the West. Cancer Epidemiol. 2019, 59, 29–36. [Google Scholar] [CrossRef]
- Sierra, M.S.; Forman, D. Burden of Colorectal Cancer in Central and South America. Cancer Epidemiol. 2016, 44, S74–S81. [Google Scholar] [CrossRef]
- Araghi, M.; Soerjomataram, I.; Jenkins, M.; Brierley, J.; Morris, E.; Bray, F.; Arnold, M. Global Trends in Colorectal Cancer Mortality: Projections to the Year 2035. Int. J. Cancer 2019, 144, 2992–3000. [Google Scholar] [CrossRef]
- Wong, M.C.S.; Huang, J.; Lok, V.; Wang, J.; Fung, F.; Ding, H.; Zheng, Z.-J. Differences in Incidence and Mortality Trends of Colorectal Cancer Worldwide Based on Sex, Age, and Anatomic Location. Clin. Gastroenterol. Hepatol. 2021, 19, 955–966.e61. [Google Scholar] [CrossRef]
- Arhin, N.; Ssentongo, P.; Taylor, M.; Olecki, E.J.; Pameijer, C.; Shen, C.; Oh, J.; Eng, C. Age-Standardised Incidence Rate and Epidemiology of Colorectal Cancer in Africa: A Systematic Review and Meta-Analysis. BMJ Open 2022, 12, e052376. [Google Scholar] [CrossRef]
- Cardoso, R.; Guo, F.; Heisser, T.; Hackl, M.; Ihle, P.; de Schutter, H.; van Damme, N.; Valerianova, Z.; Atanasov, T.; Májek, O.; et al. Colorectal Cancer Incidence, Mortality, and Stage Distribution in European Countries in the Colorectal Cancer Screening Era: An International Population-Based Study. Lancet Oncol. 2021, 22, 1002–1013. [Google Scholar] [CrossRef]
- Makhlouf, N.A.; Abdel-Gawad, M.; Mahros, A.M.; Lashen, S.A.; Zaghloul, M.; Eliwa, A.; Elshemy, E.E.; Ali-Eldin, Z.; Abdeltawab, D.; El-Raey, F.; et al. Colorectal Cancer in Arab World: A Systematic Review. World J. Gastrointest. Oncol. 2021, 13, 1791–1798. [Google Scholar] [CrossRef] [PubMed]
- Vekic, B.; Dragojevic-Simic, V.; Jakovljevic, M.; Kalezic, M.; Zagorac, Z.; Dragovic, S.; Zivic, R.; Pilipovic, F.; Simic, R.; Jovanovic, D.; et al. A Correlation Study of the Colorectal Cancer Statistics and Economic Indicators in Selected Balkan Countries. Front. Public Health 2020, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Kokki, I.; Papana, A.; Campbell, H.; Theodoratou, E. Estimating the Incidence of Colorectal Cancer in South East Asia. Croat. Med. J. 2013, 54, 532–540. [Google Scholar] [CrossRef][Green Version]
- Graham, A.; Adeloye, D.; Grant, L.; Theodoratou, E.; Campbell, H. Estimating the Incidence of Colorectal Cancer in Sub–Saharan Africa: A Systematic Analysis. J. Glob. Health 2012, 2, 020404. [Google Scholar] [CrossRef]
- Ali Hussein Alhurry, A.M.; Rezaianzadeh, A.; Rahimikazerooni, S.; Abdzaid Akool, M.; Bahrami, F.; Shahidinia, S.S.; Pourahmad, M. A Review of the Incidence of Colorectal Cancer in the Middle East. Ann. Colorectal Res. 2017, 5, e46292. [Google Scholar] [CrossRef]
- Soriano, L.C.; Soriano-Gabarró, M.; García Rodríguez, L.A. Trends in the Contemporary Incidence of Colorectal Cancer and Patient Characteristics in the United Kingdom: A Population-Based Cohort Study Using the Health Improvement Network. BMC Cancer 2018, 18, 402. [Google Scholar] [CrossRef]
- Cayuela, L.; Rodriguez, S.; Giráldez Gallego, Á.; Cayuela, A. Regional Differences in Colorectal Cancer Mortality Trends, Spain (1980–2018). Rev. Española Enferm. Dig. 2020, 113, 570–575. [Google Scholar] [CrossRef]
- Austin, H.; Jane Henley, S.; King, J.; Richardson, L.C.; Eheman, C. Changes in Colorectal Cancer Incidence Rates in Young and Older Adults in the United States: What Does It Tell Us about Screening. Cancer Causes Control 2014, 25, 191–201. [Google Scholar] [CrossRef]
- Jiang, D.; Zhang, L.; Liu, W.; Ding, Y.; Yin, J.; Ren, R.; Li, Q.; Chen, Y.; Shen, J.; Tan, X.; et al. Trends in Cancer Mortality in China from 2004 to 2018: A Nationwide Longitudinal Study. Cancer Commun. 2021, 41, 1024–1036. [Google Scholar] [CrossRef]
- Colussi, D.; Brandi, G.; Bazzoli, F.; Ricciardiello, L. Molecular Pathways Involved in Colorectal Cancer: Implications for Disease Behavior and Prevention. Int. J. Mol. Sci. 2013, 14, 16365–16385. [Google Scholar] [CrossRef]
- Fearon, E.R.; Vogelstein, B. A Genetic Model for Colorectal Tumorigenesis. Cell 1990, 61, 759–767. [Google Scholar] [CrossRef]
- Bardi, G.; Sukhikh, T.; Pandis, N.; Fenger, C.; Kronborg, O.; Heim, S. Karyotypic Characterization of Colorectal Adenocarcinomas. Genes Chromosomes Cancer 1995, 12, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Powell, S.M.; Zilz, N.; Beazer-Barclay, Y.; Bryan, T.M.; Hamilton, S.R.; Thibodeau, S.N.; Vogelstein, B.; Kinzler, K.W. APC Mutations Occur Early during Colorectal Tumorigenesis. Nature 1992, 359, 235–237. [Google Scholar] [CrossRef] [PubMed]
- Cottrell, S.; Bodmer, W.F.; Bicknell, D.; Kaklamanis, L. Molecular Analysis of APC Mutations in Familial Adenomatous Polyposis and Sporadic Colon Carcinomas. Lancet 1992, 340, 626–630. [Google Scholar] [CrossRef]
- Malumbres, M.; Barbacid, M. RAS Oncogenes: The First 30 Years. Nat. Rev. Cancer 2003, 3, 459–465. [Google Scholar] [CrossRef]
- Guerrero, S.; Casanova, I.; Farré, L.; Mazo, A.; Capellà, G.; Mangues, R. K-Ras Codon 12 Mutation Induces Higher Level of Resistance to Apoptosis and Predisposition to Anchorage-Independent Growth than Codon 13 Mutation or Proto-Oncogene Overexpression. Cancer Res. 2000, 60, 6750–6756. [Google Scholar]
- Imamura, Y.; Morikawa, T.; Liao, X.; Lochhead, P.; Kuchiba, A.; Yamauchi, M.; Qian, Z.R.; Nishihara, R.; Meyerhardt, J.A.; Haigis, K.M.; et al. Specific Mutations in KRAS Codons 12 and 13, and Patient Prognosis in 1075 BRAF Wild-Type Colorectal Cancers. Clin. Cancer Res. 2012, 18, 4753–4763. [Google Scholar] [CrossRef]
- Baker, S.J.; Preisinger, A.C.; Jessup, J.M.; Paraskeva, C.; Markowitz, S.; Willson, J.K.; Hamilton, S.; Vogelstein, B. P53 Gene Mutations Occur in Combination with 17p Allelic Deletions as Late Events in Colorectal Tumorigenesis. Cancer Res. 1990, 50, 7717–7722. [Google Scholar]
- Lanza, G.; Matteuzzi, M.; Gafá, R.; Orvieto, E.; Maestri, I.; Santini, A.; del Senno, L. Chromosome 18q Allelic Loss and Prognosis in Stage II and III Colon Cancer. Int. J. Cancer 1998, 79, 390–395. [Google Scholar] [CrossRef]
- Fishel, R. Mismatch Repair, Molecular Switches, and Signal Transduction. Genes Dev. 1998, 12, 2096–2101. [Google Scholar] [CrossRef]
- Sinicrope, F.A.; Sargent, D.J. Molecular Pathways: Microsatellite Instability in Colorectal Cancer: Prognostic, Predictive, and Therapeutic Implications. Clin. Cancer Res. 2012, 18, 1506–1512. [Google Scholar] [CrossRef] [PubMed]
- Samowitz, W.S.; Albertsen, H.; Herrick, J.; Levin, T.R.; Sweeney, C.; Murtaugh, M.A.; Wolff, R.K.; Slattery, M.L. Evaluation of a Large, Population-Based Sample Supports a CpG Island Methylator Phenotype in Colon Cancer. Gastroenterology 2005, 129, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Teodoridis, J.M.; Hardie, C.; Brown, R. CpG Island Methylator Phenotype (CIMP) in Cancer: Causes and Implications. Cancer Lett. 2008, 268, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Bettington, M.; Walker, N.; Clouston, A.; Brown, I.; Leggett, B.; Whitehall, V. The Serrated Pathway to Colorectal Carcinoma: Current Concepts and Challenges. Histopathology 2013, 62, 367–386. [Google Scholar] [CrossRef]
- Terzić, J.; Grivennikov, S.; Karin, E.; Karin, M. Inflammation and Colon Cancer. Gastroenterology 2010, 138, 2101–2114.e5. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.-T.; Wang, S.; Ye, Y.-J.; Du, R.-Y.; Cui, Z.-R.; Somsouk, M. Constitutive Activation of Stat3 Signaling Pathway in Human Colorectal Carcinoma. World J. Gastroenterol. 2004, 10, 1569. [Google Scholar] [CrossRef]
- Corvinus, F.M.; Orth, C.; Moriggl, R.; Tsareva, S.A.; Wagner, S.; Pfitzner, E.B.; Baus, D.; Kaufman, R.; Huber, L.A.; Zatloukal, K.; et al. Persistent STAT3 Activation in Colon Cancer Is Associated with Enhanced Cell Proliferation and Tumor Growth. Neoplasia 2005, 7, 545–555. [Google Scholar] [CrossRef]
- Sinicrope, F.A.; Gill, S. Role of Cyclooxygenase-2 in Colorectal Cancer. Cancer Metastasis Rev. 2004, 23, 63–75. [Google Scholar] [CrossRef]
- LeRoith, D.; Holly, J.M.P.; Forbes, B.E. Insulin-like Growth Factors: Ligands, Binding Proteins, and Receptors. Mol. Metab. 2021, 52, 101245. [Google Scholar] [CrossRef]
- Salmon, W.D.; Daughaday, W.H. A Hormonally Controlled Serum Factor Which Stimulates Sulfate Incorporation by Cartilage in Vitro. J. Lab. Clin. Med. 1957, 49, 825–836. [Google Scholar]
- Kawamoto, K.; Onodera, H.; Kondo, S.; Kan, S.; Ikeuchi, D.; Maetani, S.; Imamura, M. Expression of Insulin-Like Growth Factor-2 Can Predict the Prognosis of Human Colorectal Cancer Patients: Correlation with Tumor Progression, Proliferative Activity and Survival. Oncology 1998, 55, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Teramae, S.; Miyamoto, H.; Muguruma, N.; Okada, Y.; Goji, T.; Kitamura, S.; Kimura, T.; Kimura, M.; Bando, Y.; Takayama, T. Insulin-like Growth Factor II-Producing Metastatic Colon Cancer with Recurrent Hypoglycemia. Clin. J. Gastroenterol. 2015, 8, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Vrieling, A.; Voskuil, D.W.; Bosma, A.; Majoor, D.M.; van Doorn, J.; Cats, A.; Depla, A.C.T.M.; Timmer, R.; Witteman, B.J.M.; Wesseling, J.; et al. Expression of Insulin-like Growth Factor System Components in Colorectal Tissue and Its Relation with Serum IGF Levels. Growth Horm. IGF Res. 2009, 19, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Kasprzak, A.; Adamek, A. Insulin-Like Growth Factor 2 (IGF2) Signaling in Colorectal Cancer—From Basic Research to Potential Clinical Applications. Int. J. Mol. Sci. 2019, 20, 4915. [Google Scholar] [CrossRef]
- Unger, C.; Kramer, N.; Unterleuthner, D.; Scherzer, M.; Burian, A.; Rudisch, A.; Stadler, M.; Schlederer, M.; Lenhardt, D.; Riedl, A.; et al. Stromal-Derived IGF2 Promotes Colon Cancer Progression via Paracrine and Autocrine Mechanisms. Oncogene 2017, 36, 5341–5355. [Google Scholar] [CrossRef]
- Mancarella, C.; Morrione, A.; Scotlandi, K. Novel Regulators of the IGF System in Cancer. Biomolecules 2021, 11, 273. [Google Scholar] [CrossRef]
- Samani, A.A.; Yakar, S.; LeRoith, D.; Brodt, P. The Role of the IGF System in Cancer Growth and Metastasis: Overview and Recent Insights. Endocr. Rev. 2007, 28, 20–47. [Google Scholar] [CrossRef]
- Davies, M.; Gupta, S.; Goldspink, G.; Winslet, M. The Insulin-like Growth Factor System and Colorectal Cancer: Clinical and Experimental Evidence. Int. J. Colorectal Dis. 2006, 21, 201–208. [Google Scholar] [CrossRef]
- Pandini, G.; Frasca, F.; Mineo, R.; Sciacca, L.; Vigneri, R.; Belfiore, A. Insulin/Insulin-like Growth Factor I Hybrid Receptors Have Different Biological Characteristics Depending on the Insulin Receptor Isoform Involved. J. Biol. Chem. 2002, 277, 39684–39695. [Google Scholar] [CrossRef] [PubMed]
- Belfiore, A.; Malaguarnera, R. Insulin Receptor and Cancer. Endocr. Relat. Cancer 2011, 18, R125–R147. [Google Scholar] [CrossRef]
- Scalia, P.; Giordano, A.; Williams, S.J. The IGF-II–Insulin Receptor Isoform-A Autocrine Signal in Cancer: Actionable Perspectives. Cancers 2020, 12, 366. [Google Scholar] [CrossRef] [PubMed]
- Frasca, F.; Pandini, G.; Scalia, P.; Sciacca, L.; Mineo, R.; Costantino, A.; Goldfine, I.D.; Belfiore, A.; Vigneri, R. Insulin Receptor Isoform A, a Newly Recognized, High-Affinity Insulin-Like Growth Factor II Receptor in Fetal and Cancer Cells. Mol. Cell. Biol. 1999, 19, 3278–3288. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, T.; Kyohara, M.; Terauchi, Y.; Shirakawa, J. The Roles of the IGF Axis in the Regulation of the Metabolism: Interaction and Difference between Insulin Receptor Signaling and IGF-I Receptor Signaling. Int. J. Mol. Sci. 2021, 22, 6817. [Google Scholar] [CrossRef]
- Rieger, L.; O’Connor, R. Controlled Signaling—Insulin-Like Growth Factor Receptor Endocytosis and Presence at Intracellular Compartments. Front. Endocrinol. 2021, 11, 620013. [Google Scholar] [CrossRef]
- El-Shewy, H.M.; Luttrell, L.M. Insulin-Like Growth Factor-2/Mannose-6 Phosphate Receptors. Vitam. Horm. 2009, 80, 667–697. [Google Scholar]
- Duplomb, L.; Chaigne-Delalande, B.; Vusio, P.; Raher, S.; Jacques, Y.; Godard, A.; Blanchard, F. Soluble Mannose 6-Phosphate/Insulin-Like Growth Factor II (IGF-II) Receptor Inhibits Interleukin-6-Type Cytokine-Dependent Proliferation by Neutralization of IGF-II. Endocrinology 2003, 144, 5381–5389. [Google Scholar] [CrossRef][Green Version]
- di Popolo, A.; Memoli, A.; Apicella, A.; Tuccillo, C.; di Palma, A.; Ricchi, P.; Acquaviva, A.M.; Zarrilli, R. IGF-II/IGF-I Receptor Pathway up-Regulates COX-2 MRNA Expression and PGE2 Synthesis in Caco-2 Human Colon Carcinoma Cells. Oncogene 2000, 19, 5517–5524. [Google Scholar] [CrossRef]
- Mishra, L.; Bass, B.; Ooi, B.S.; Sidawy, A.; Korman, L.; Mönig, H. Role of Insulin-like Growth Factor-I (IGF-I) Receptor, IGF-I, and IGF Binding Protein-2 in Human Colorectal Cancers. Growth Horm. IGF Res. 1998, 8, 473–479. [Google Scholar] [CrossRef]
- Weber, M.M.; Fottner, C.; Liu, S.B.; Jung, M.C.; Engelhardt, D.; Baretton, G.B. Overexpression of the Insulin-like Growth Factor I Receptor in Human Colon Carcinomas. Cancer 2002, 95, 2086–2095. [Google Scholar] [CrossRef]
- Nakamura, M.; Miyamoto, S.; Maeda, H.; Zhang, S.; Sangai, T.; Ishii, G.; Hasebe, T.; Endoh, Y.; Saito, N.; Asaka, M.; et al. Low Levels of Insulin-Like Growth Factor Type 1 Receptor Expression at Cancer Cell Membrane Predict Liver Metastasis in Dukes’ C Human Colorectal Cancers. Clin. Cancer Res. 2004, 10, 8434–8441. [Google Scholar] [CrossRef]
- Heckl, S.M.; Pellinghaus, M.; Behrens, H.-M.; Krüger, S.; Schreiber, S.; Röcken, C. Questioning the IGF1 Receptor’s Assigned Role in CRC-a Case for Rehabilitation? BMC Cancer 2020, 20, 704. [Google Scholar] [CrossRef] [PubMed]
- Robajac, D.; Križáková, M.; Masnikosa, R.; Miljuš, G.; Šunderić, M.; Nedić, O.; Katrlík, J. Sensitive Glycoprofiling of Insulin-like Growth Factor Receptors Isolated from Colon Tissue of Patients with Colorectal Carcinoma Using Lectin-Based Protein Microarray. Int. J. Biol. Macromol. 2020, 144, 932–937. [Google Scholar] [CrossRef] [PubMed]
- Nedić, O.; Robajac, D.; Šunderić, M.; Miljuš, G.; Đukanović, B.; Malenković, V. Detection and Identification of Oxidized Insulin-like Growth Factor-Binding Proteins and Receptors in Patients with Colorectal Carcinoma. Free Radic. Biol. Med. 2013, 65, 1195–1200. [Google Scholar] [CrossRef]
- Esposito, D.L.; Aru, F.; Lattanzio, R.; Morgano, A.; Abbondanza, M.; Malekzadeh, R.; Bishehsari, F.; Valanzano, R.; Russo, A.; Piantelli, M.; et al. The Insulin Receptor Substrate 1 (Irs1) in Intestinal Epithelial Differentiation and in Colorectal Cancer. PLoS ONE 2012, 7, e36190. [Google Scholar] [CrossRef] [PubMed]
- Day, E.; Poulogiannis, G.; McCaughan, F.; Mulholland, S.; Arends, M.J.; Ibrahim, A.E.K.; Dear, P.H. IRS2 is a Candidate Driver Oncogene on 13q34 in Colorectal Cancer. Int. J. Exp. Pathol. 2013, 94, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Slattery, M.L.; Samowitz, W.; Curtin, K.; Ma, K.N.; Hoffman, M.; Caan, B.; Neuhausen, S. Associations among IRS1, IRS2, IGF1, and IGFBP3 Genetic Polymorphisms and Colorectal Cancer. Cancer Epidemiol. Biomark. Prev. 2004, 13, 1206–1214. [Google Scholar] [CrossRef]
- Abbruzzese, C.; Diodoro, M.G.; Sperduti, I.; Mileo, A.M.; Pattaro, G.; de Salvo, L.; Cosimelli, M.; Perrotti, N.; Paggi, M.G. Detection of Phosphorylated Insulin Receptor in Colorectal Adenoma and Adenocarcinoma: Implications for Prognosis and Clinical Outcome. J. Cell. Physiol. 2015, 230, 562–567. [Google Scholar] [CrossRef]
- Sekharam, M.; Zhao, H.; Sun, M.; Fang, Q.; Zhang, Q.; Yuan, Z.; Dan, H.C.; Boulware, D.; Cheng, J.Q.; Coppola, D. Insulin-like Growth Factor 1 Receptor Enhances Invasion and Induces Resistance to Apoptosis of Colon Cancer Cells through the Akt/Bcl-x(L) Pathway. Cancer Res. 2003, 63, 7708–7716. [Google Scholar]
- Zhang, Q.; Wang, L.; Song, Z.; Qu, X. Knockdown of Type I Insulin-like Growth Factor Receptor Inhibits Human Colorectal Cancer Cell Growth and Downstream PI3K/Akt, WNT/β-Catenin Signal Pathways. Biomed. Pharmacother. 2015, 73, 12–18. [Google Scholar] [CrossRef]
- Kasprzak, A. Insulin-Like Growth Factor 1 (IGF-1) Signaling in Glucose Metabolism in Colorectal Cancer. Int. J. Mol. Sci. 2021, 22, 6434. [Google Scholar] [CrossRef]
- Fukuda, R.; Hirota, K.; Fan, F.; Jung, Y.D.; Ellis, L.M.; Semenza, G.L. Insulin-like Growth Factor 1 Induces Hypoxia-Inducible Factor 1-Mediated Vascular Endothelial Growth Factor Expression, Which Is Dependent on MAP Kinase and Phosphatidylinositol 3-Kinase Signaling in Colon Cancer Cells. J. Biol. Chem. 2002, 277, 38205–38211. [Google Scholar] [CrossRef] [PubMed]
- Ellis, B.C.; Graham, L.D.; Molloy, P.L. CRNDE, a Long Non-Coding RNA Responsive to Insulin/IGF Signaling, Regulates Genes Involved in Central Metabolism. Biochim. Biophys. Acta Mol. Cell Res. 2014, 1843, 372–386. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Wang, Y.; Tao, C.; Li, Y.; Cao, S.; Yang, X. CRNDE silencing promotes apoptosis and enhances cisplatin sensitivity of colorectal carcinoma cells by inhibiting the Akt/mTORC1-mediated Warburg effect. Oncol. Lett. 2022, 23, 70. [Google Scholar] [CrossRef] [PubMed]
- Bach, L.A. 40 YEARS OF IGF1: IGF-Binding Proteins. J. Mol. Endocrinol. 2018, 61, T11–T28. [Google Scholar] [CrossRef]
- Palmqvist, R.; Stattin, P.; Rinaldi, S.; Biessy, C.; Stenling, R.; Riboli, E.; Hallmans, G.; Kaaks, R. Plasma Insulin, IGF-Binding Proteins-1 and -2 and Risk of Colorectal Cancer: A Prospective Study in Northern Sweden. Int. J. Cancer 2003, 107, 89–93. [Google Scholar] [CrossRef]
- Jiang, B.; Zhang, X.; Du, L.-L.; Wang, Y.; Liu, D.-B.; Han, C.-Z.; Jing, J.-X.; Zhao, X.-W.; Xu, X.-Q. Possible Roles of Insulin, IGF-1 and IGFBPs in Initiation and Progression of Colorectal Cancer. World J. Gastroenterol. 2014, 20, 1608–1613. [Google Scholar] [CrossRef]
- Ollberding, N.J.; Cheng, I.; Wilkens, L.R.; Henderson, B.E.; Pollak, M.N.; Kolonel, L.N.; le Marchand, L. Genetic Variants, Prediagnostic Circulating Levels of Insulin-like Growth Factors, Insulin, and Glucose and the Risk of Colorectal Cancer: The Multiethnic Cohort Study. Cancer Epidemiol. Biomark. Prev. 2012, 21, 810–820. [Google Scholar] [CrossRef]
- Alami, N.; Page, V.; Yu, Q.; Jerome, L.; Paterson, J.; Shiry, L.; Leyland-Jones, B. Recombinant Human Insulin-like Growth Factor-Binding Protein 3 Inhibits Tumor Growth and Targets the Akt Pathway in Lung and Colon Cancer Models. Growth Horm. IGF Res. 2008, 18, 487–496. [Google Scholar] [CrossRef]
- Ali, O.; Cohen, P.; Lee, K.-W. Epidemiology and Biology of Insulin-like Growth Factor Binding Protein-3 (IGFBP-3) as an Anti-Cancer Molecule. Horm. Metab. Res. 2003, 35, 726–733. [Google Scholar] [CrossRef]
- Jenkins, P.J.; Khalaf, S.; Ogunkolade, W.; McCarthy, K.; David, T.; Hands, R.E.; Davies, D.; Bustin, S.A. Differential Expression of IGF-Binding Protein-3 in Normal and Malignant Colon and Its Influence on Apoptosis. Endocr. Relat. Cancer 2005, 12, 891–901. [Google Scholar] [CrossRef]
- Yamamoto, N.; Oshima, T.; Yoshihara, K.; Aoyama, T.; Hayashi, T.; Yamada, T.; Sato, T.; Shiozawa, M.; Yoshikawa, T.; Morinaga, S.; et al. Clinicopathological Significance and Impact on Outcomes of the Gene Expression Levels of IGF-1, IGF-2 and IGF-1R, IGFBP-3 in Patients with Colorectal Cancer: Overexpression of the IGFBP-3 Gene Is an Effective Predictor of Outcomes in Patients with Colorectal Cancer. Oncol. Lett. 2017, 13, 3958–3966. [Google Scholar] [CrossRef] [PubMed]
- Zámorová, M.; Holazová, A.; Miljuš, G.; Robajac, D.; Šunderić, M.; Malenković, V.; Đukanović, B.; Gemeiner, P.; Katrlík, J.; Nedić, O. Analysis of Changes in the Glycan Composition of Serum, Cytosol and Membrane Glycoprotein Biomarkers of Colorectal Cancer Using a Lectin-Based Protein Microarray. Anal. Methods 2017, 9, 2660–2666. [Google Scholar] [CrossRef]
- Vocka, M.; Langer, D.; Fryba, V.; Petrtyl, J.; Hanus, T.; Kalousova, M.; Zima, T.; Petruzelka, L. Novel Serum Markers HSP60, CHI3L1, and IGFBP-2 in Metastatic Colorectal Cancer. Oncol. Lett. 2019, 18, 6284–6292. [Google Scholar] [CrossRef] [PubMed]
- Šunderić, M.; Đukanović, B.; Malenković, V.; Nedić, O. Molecular Forms of the Insulin-like Growth Factor-Binding Protein-2 in Patients with Colorectal Cancer. Exp. Mol. Pathol. 2014, 96, 48–53. [Google Scholar] [CrossRef]
- Jenab, M.; Riboli, E.; Cleveland, R.J.; Norat, T.; Rinaldi, S.; Nieters, A.; Biessy, C.; Tjønneland, A.; Olsen, A.; Overvad, K.; et al. Serum C-Peptide, IGFBP-1 and IGFBP-2 and Risk of Colon and Rectal Cancers in the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 2007, 121, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Otani, T.; Iwasaki, M.; Sasazuki, S.; Inoue, M.; Tsugane, S. Plasma C-Peptide, Insulin-like Growth Factor-I, Insulin-like Growth Factor Binding Proteins and Risk of Colorectal Cancer in a Nested Case-Control Study: The Japan Public Health Center-Based Prospective Study. Int. J. Cancer 2007, 120, 2007–2012. [Google Scholar] [CrossRef]
- Durai, R.; Davies, M.; Yang, W.; Yang, S.; Seifalian, A.; Goldspink, G.; Winslet, M. Biology of Insulin-like Growth Factor Binding Protein-4 and Its Role in Cancer (Review). Int. J. Oncol. 2006, 28, 1317–1325. [Google Scholar] [CrossRef]
- Street, M.; Miraki-Moud, F.; Sanderson, I.; Savage, M.; Giovannelli, G.; Bernasconi, S.; Camacho-Hubner, C. Interleukin-1beta (IL-1beta) and IL-6 Modulate Insulin-like Growth Factor-Binding Protein (IGFBP) Secretion in Colon Cancer Epithelial (Caco-2) Cells. J. Endocrinol. 2003, 179, 405–415. [Google Scholar] [CrossRef]
- Bach, L.A. Recent Insights into the Actions of IGFBP-6. J. Cell Commun. Signal. 2015, 9, 189–200. [Google Scholar] [CrossRef]
- Zhao, C.; Zhu, X.; Wang, G.; Wang, W.; Ju, S.; Wang, X. Decreased Expression of IGFBP6 Correlates with Poor Survival in Colorectal Cancer Patients. Pathol. Res. Pract. 2020, 216, 152909. [Google Scholar] [CrossRef]
- Koyama, N.; Zhang, J.; Huqun; Miyazawa, H.; Tanaka, T.; Su, X.; Hagiwara, K. Identification of IGFBP-6 as an Effector of the Tumor Suppressor Activity of SEMA3B. Oncogene 2008, 27, 6581–6589. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Dozmorov, M.; Oh, Y. IGFBP-3/IGFBP-3 Receptor System as an Anti-Tumor and Anti-Metastatic Signaling in Cancer. Cells 2020, 9, 1261. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.C.; Smartt, H.; Zadeh, H.A.M.; MacFarlane, M.; Paraskeva, C.; Collard, T.J. Insulin-like Growth Factor Binding Protein 3 (IGFBP-3) Potentiates TRAIL-Induced Apoptosis of Human Colorectal Carcinoma Cells through Inhibition of NF-ΚB. Cell Death Differ. 2007, 14, 137–145. [Google Scholar] [CrossRef]
- Lee, K.-W.; Ma, L.; Yan, X.; Liu, B.; Zhang, X.; Cohen, P. Rapid Apoptosis Induction by IGFBP-3 Involves an Insulin-like Growth Factor-Independent Nucleomitochondrial Translocation of RXRα/Nur77. J. Biol. Chem. 2005, 280, 16942–16948. [Google Scholar] [CrossRef]
- Williams, A.C.; Collard, T.J.; Perks, C.M.; Newcomb, P.; Moorghen, M.; Holly, J.M.; Paraskeva, C. Increased P53-Dependent Apoptosis by the Insulin-like Growth Factor Binding Protein IGFBP-3 in Human Colonic Adenoma-Derived Cells. Cancer Res. 2000, 60, 22–27. [Google Scholar]
- Lin, Y.-W.; Weng, X.-F.; Huang, B.-L.; Guo, H.-P.; Xu, Y.-W.; Peng, Y.-H. IGFBP-1 in Cancer: Expression, Molecular Mechanisms, and Potential Clinical Implications. Am. J. Transl. Res. 2021, 13, 813–832. [Google Scholar]
- Kaaks, R.; Toniolo, P.; Akhmedkhanov, A.; Lukanova, A.; Biessy, C.; Dechaud, H.; Rinaldi, S.; Zeleniuch-Jacquotte, A.; Shore, R.E.; Riboli, E. Serum C-Peptide, Insulin-Like Growth Factor (IGF)-I, IGF-Binding Proteins, and Colorectal Cancer Risk in Women. J. Natl. Cancer Inst. 2000, 92, 1592–1600. [Google Scholar] [CrossRef] [PubMed]
- Pickard, A.; McCance, D.J. IGF-Binding Protein 2—Oncogene or Tumor Suppressor? Front. Endocrinol. 2015, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Bach, L.A.; Fu, P.; Yang, Z. Insulin-like Growth Factor-Binding Protein-6 and Cancer. Clin. Sci. 2013, 124, 215–229. [Google Scholar] [CrossRef]
- Fu, P.; Thompson, J.A.; Bach, L.A. Promotion of Cancer Cell Migration. J. Biol. Chem. 2007, 282, 22298–22306. [Google Scholar] [CrossRef]
- Devin, J.L.; Bolam, K.A.; Jenkins, D.G.; Skinner, T.L. The Influence of Exercise on the Insulin-like Growth Factor Axis in Oncology: Physiological Basis, Current, and Future Perspectives. Cancer Epidemiol. Biomark. Prev. 2016, 25, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Durai, R.; Yang, W.; Gupta, S.; Seifalian, A.M.; Winslet, M.C. The Role of the Insulin-like Growth Factor System in Colorectal Cancer: Review of Current Knowledge. Int. J. Colorectal Dis. 2005, 20, 203–220. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E. Insulin, Insulin-Like Growth Factors and Colon Cancer: A Review of the Evidence. J. Nutr. 2001, 131, 3109S–3120S. [Google Scholar] [CrossRef]
- Berk, Ş.; Janssen, J.A.M.J.L.; van Koetsveld, P.M.; Dogan, F.; Değerli, N.; Özcan, S.; Kelestimur, F.; Hofland, L.J. Modifying Effects of Glucose and Insulin/Insulin-Like Growth Factors on Colon Cancer Cells. Front. Oncol. 2021, 11, 645732. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Shen, F.; Weinfeld, M.; Sergi, C. Insulin Growth Factor Binding Protein 7 (IGFBP7)-Related Cancer and IGFBP3 and IGFBP7 Crosstalk. Front. Oncol. 2020, 10, 727. [Google Scholar] [CrossRef]
- Georges, R.B.; Adwan, H.; Hamdi, H.; Hielscher, T.; Linnemann, U.; Berger, M.R. The Insulin-like Growth Factor Binding Proteins 3 and 7 Are Associated with Colorectal Cancer and Liver Metastasis. Cancer Biol. Ther. 2011, 12, 69–79. [Google Scholar] [CrossRef]
- Chan, Y.X.; Alfonso, H.; Chubb, S.A.P.; Ho, K.K.Y.; Fegan, P.G.; Hankey, G.J.; Golledge, J.; Flicker, L.; Yeap, B.B. Higher IGFBP3 is associated with increased incidence of colorectal cancer in older men independently of IGF1. Clin. Endocrinol. 2018, 88, 333–340. [Google Scholar] [CrossRef]
- Diehl, D.; Hessel, E.; Oesterle, D.; Renner-Müller, I.; Elmlinger, M.; Langhammer, M.; Göttlicher, M.; Wolf, E.; Lahm, H.; Hoeflich, A. IGFBP-2 overexpression reduces the appearance of dysplastic aberrant crypt foci and inhibits growth of adenomas in chemically induced colorectal carcinogenesis. Int. J. Cancer 2009, 124, 2220–2225. [Google Scholar] [CrossRef]
- Durai, R.; Yang, S.Y.; Seifalian, A.M.; Goldspink, G.; Winslet, M.C. Role of insulin-like growth factor binding protein-4 in prevention of colon cancer. World J. Surg. Oncol. 2007, 5, 128. [Google Scholar] [CrossRef]
- Nakamura, M.; Miyamoto, S.; Maeda, H.; Ishii, G.; Hasebe, T.; Chiba, T.; Asaka, M.; Ochiai, A. Matrix Metalloproteinase-7 Degrades All Insulin-like Growth Factor Binding Proteins and Facilitates Insulin-like Growth Factor Bioavailability. Biochem. Biophys. Res. Commun. 2005, 333, 1011–1016. [Google Scholar] [CrossRef]
- Ohashi, S.; Natsuizaka, M.; Nakagawa, H. MMP7 and Activation of IGF-1R: A New Insight into Anti-EGFR Therapeutic Resistance in Metastatic Colorectal Cancer. Cancer Biol. Ther. 2011, 11, 184–187. [Google Scholar] [CrossRef]
- Sadowski, T.; Dietrich, S.; Koschinsky, F.; Sedlacek, R. Matrix Metalloproteinase 19 Regulates Insulin-like Growth Factor-Mediated Proliferation, Migration, and Adhesion in Human Keratinocytes through Proteolysis of Insulin-like Growth Factor Binding Protein-3. Mol. Biol. Cell 2003, 14, 4569–4580. [Google Scholar] [CrossRef] [PubMed]
- Walkiewicz, K.; Nowakowska-Zajdel, E.; Kozieł, P.; Muc-Wierzgoń, M. The Role of Some ADAM-Proteins and Activation of the Insulin Growth Factor-Related Pathway in Colorectal Cancer. Cent. Eur. J. Immunol. 2018, 43, 109–113. [Google Scholar] [CrossRef]
- Firth, S.M.; Baxter, R.C. Cellular Actions of the Insulin-Like Growth Factor Binding Proteins. Endocr. Rev. 2002, 23, 824–854. [Google Scholar] [CrossRef] [PubMed]
- Forbes, B.E.; McCarthy, P.; Norton, R.S. Insulin-Like Growth Factor Binding Proteins: A Structural Perspective. Front. Endocrinol. 2012, 3, 38. [Google Scholar] [CrossRef]
- Philippou, A.; Christopoulos, P.F.; Koutsilieris, M. Clinical Studies in Humans Targeting the Various Components of the IGF System Show Lack of Efficacy in the Treatment of Cancer. Mutat. Res. Rev. Mutat. Res. 2017, 772, 105–122. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Chang, H.; Greer, A.; Hillerman, S.; Reeves, K.A.; Hurlburt, W.; Cogswell, J.; Patel, D.; Qi, Z.; Fairchild, C.; et al. IRS2 Copy Number Gain, KRAS and BRAF Mutation Status as Predictive Biomarkers for Response to the IGF-1R/IR Inhibitor BMS-754807 in Colorectal Cancer Cell Lines. Mol. Cancer Ther. 2015, 14, 620–630. [Google Scholar] [CrossRef]
- Wang, Q.; Wei, F.; Lv, G.; Li, C.; Liu, T.; Hadjipanayis, C.G.; Zhang, G.; Hao, C.; Bellail, A.C. The Association of TP53 Mutations with the Resistance of Colorectal Carcinoma to the Insulin-like Growth Factor-1 Receptor Inhibitor Picropodophyllin. BMC Cancer 2013, 13, 521. [Google Scholar] [CrossRef]
- Voutsadakis, I.A. The Landscape of PIK3CA Mutations in Colorectal Cancer. Clin. Colorectal Cancer 2021, 20, 201–215. [Google Scholar] [CrossRef]
- Lin, P.-C.; Lin, J.-K.; Lin, H.-H.; Lan, Y.-T.; Lin, C.-C.; Yang, S.-H.; Chen, W.-S.; Liang, W.-Y.; Jiang, J.-K.; Chang, S.-C. A Comprehensive Analysis of Phosphatase and Tensin Homolog Deleted on Chromosome 10 (PTEN) Loss in Colorectal Cancer. World J. Surg. Oncol. 2015, 13, 186. [Google Scholar] [CrossRef]
- Wong, H.-L.; Delellis, K.; Probst-Hensch, N.; Koh, W.-P.; van den Berg, D.; Lee, H.-P.; Yu, M.C.; Ingles, S.A. A New Single Nucleotide Polymorphism in the Insulin-like Growth Factor I Regulatory Region Associates with Colorectal Cancer Risk in Singapore Chinese. Cancer Epidemiol. Biomark. Prev. 2005, 14, 144–151. [Google Scholar] [CrossRef]
- Samowitz, W.S.; Wolff, R.K.; Ma, K.N.; Andersen, K.; Caan, B.; Slattery, M.L. Polymorphisms in Insulin-Related Genes Predispose to Specific KRAS2 and TP53 Mutations in Colon Cancer. Mutat. Res. Fundam. Mol. Mech. Mutagenesis 2006, 595, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Zecevic, M.; Amos, C.I.; Gu, X.; Campos, I.M.; Jones, J.S.; Lynch, P.M.; Rodriguez-Bigas, M.A.; Frazier, M.L. IGF1 Gene Polymorphism and Risk for Hereditary Nonpolyposis Colorectal Cancer. JNCI J. Natl. Cancer Inst. 2006, 98, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Pechlivanis, S.; Wagner, K.; Chang-Claude, J.; Hoffmeister, M.; Brenner, H.; Försti, A. Polymorphisms in the Insulin like Growth Factor 1 and IGF Binding Protein 3 Genes and Risk of Colorectal Cancer. Cancer Detect. Prev. 2007, 31, 408–416. [Google Scholar] [CrossRef]
- Xiang, H.; Liu, L.; Chu, G.; Wei, S.; Liu, J.; Xu, Y.; Chen, J.; Wang, C.; Wei, Q.-Y.; Nie, S. Association Between Two Functional Polymorphisms of Insulin-Like Growth Factor Binding Protein 3 and Colorectal Cancer Risk in a Chinese Population. J. Toxicol. Environ. Health Part A 2009, 72, 706–711. [Google Scholar] [CrossRef]
- Feik, E.; Baierl, A.; Hieger, B.; Führlinger, G.; Pentz, A.; Stättner, S.; Weiss, W.; Pulgram, T.; Leeb, G.; Mach, K.; et al. Association of IGF1 and IGFBP3 Polymorphisms with Colorectal Polyps and Colorectal Cancer Risk. Cancer Causes Control 2010, 21, 91–97. [Google Scholar] [CrossRef]
- LeRoy, E.C.; Moore, J.H.; Hu, C.; Martínez, M.E.; Lance, P.; Duggan, D.; Thompson, P.A. Genes in the Insulin and Insulin-like Growth Factor Pathway and Odds of Metachronous Colorectal Neoplasia. Hum. Genet. 2011, 129, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Keku, T.O.; Vidal, A.; Oliver, S.; Hoyo, C.; Hall, I.J.; Omofoye, O.; McDoom, M.; Worley, K.; Galanko, J.; Sandler, R.S.; et al. Genetic Variants in IGF-I, IGF-II, IGFBP-3, and Adiponectin Genes and Colon Cancer Risk in African Americans and Whites. Cancer Causes Control 2012, 23, 1127–1138. [Google Scholar] [CrossRef]
- Karimi, K.; Mahmoudi, T.; Karimi, N.; Dolatmoradi, H.; Arkani, M.; Farahani, H.; Vahedi, M.; Parsimehr, E.; Dabiri, R.; Nobakht, H.; et al. Is There an Association between Variants in Candidate Insulin Pathway Genes IGF-I, IGFBP-3, INSR, and IRS2 and Risk of Colorectal Cancer in the Iranian Population? Asian Pac. J. Cancer Prev. 2013, 14, 5011–5016. [Google Scholar] [CrossRef]
- Stanilov, N.S.; Karakolev, I.A.; Deliysky, T.S.; Jovchev, J.P.; Stanilova, S.A. Association of Insulin-like Growth Factor-I Receptor Polymorphism with Colorectal Cancer Development. Mol. Biol. Rep. 2014, 41, 8099–8106. [Google Scholar] [CrossRef]
- Simons, C.C.J.M.; Schouten, L.J.; Godschalk, R.W.L.; van Engeland, M.; van den Brandt, P.A.; van Schooten, F.J.; Weijenberg, M.P. Genetic Variants in the Insulin-like Growth Factor Pathway and Colorectal Cancer Risk in the Netherlands Cohort Study. Sci. Rep. 2015, 5, 14126. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, T.; Majidzadeh-A, K.; Karimi, K.; Farahani, H.; Dabiri, R.; Nobakht, H.; Asadi, A.; Karimi, N.; Arkani, M.; Zali, M.R. Gly972Arg Variant of Insulin Receptor Substrate 1 Gene and Colorectal Cancer Risk in Overweight/Obese Subjects. Int. J. Biol. Markers 2016, 31, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhou, X.; Liu, R.; Cao, Y.; Wang, L.; Chao, X.; Zhang, S. Relationship of SNP Rs35767 in IGF-1 Promoter Region with Susceptibility to Colorectal Cancer. Int. J. Clin. Exp. Pathol. 2018, 11, 5110–5116. [Google Scholar] [PubMed]
- Chao, X.L.; Wang, L.L.; Liu, R.; Li, Y.; Zhou, X.J. Association between CA Repeat Polymorphism in IGF1 Gene Promoter and Colorectal Cancer Risk in a Native Chinese Population. Neoplasma 2019, 66, 1002–1008. [Google Scholar] [CrossRef]
- Dhifallah, H.; Aissi, S.; Njima, M.; Zakhama, A.; Kenani, A. IGF1 Polymorphisms and Colon Cancer Risk in Tunisian Population. La Tunis. Med. 2020, 98, 60–69. [Google Scholar]
- Kattan, S.W.; Allah, A.M.K.A.; Mohamed, K.I.; Alruwetei, A.M.; Hegazy, A.H.; el Gayed, E.M.A. Linking Insulin like Growth Factor-1 (IGF-1) Rs6214 Gene Polymorphism and Its Serum Level with Risk of Colorectal Cancer. Beni-Suef Univ. J. Basic Appl. Sci. 2022, 11, 74. [Google Scholar] [CrossRef]
- Li, X.; Yang, J.; Wang, X.; Gao, X. The Association of IGF1 Rs35767 Polymorphism with Colorectal Cancer Risk in the Chinese Han Population. Nucleosides Nucleotides Nucleic Acids 2022, 1–9. [Google Scholar] [CrossRef]
- Kawasaki, T.; Nosho, K.; Ohnishi, M.; Suemoto, Y.; Kirkner, G.J.; Fuchs, C.S.; Ogino, S. IGFBP3 Promoter Methylation in Colorectal Cancer: Relationship with Microsatellite Instability, CpG Island Methylator Phenotype, P53. Neoplasia 2007, 9, 1091–1098. [Google Scholar] [CrossRef][Green Version]
- Perez-Carbonell, L.; Balaguer, F.; Toiyama, Y.; Egoavil, C.; Rojas, E.; Guarinos, C.; Andreu, M.; Llor, X.; Castells, A.; Jover, R.; et al. IGFBP3 Methylation Is a Novel Diagnostic and Predictive Biomarker in Colorectal Cancer. PLoS ONE 2014, 9, e104285. [Google Scholar] [CrossRef]
- Goel, A.; Arnold, C.N.; Niedzwiecki, D.; Carethers, J.M.; Dowell, J.M.; Wasserman, L.; Compton, C.; Mayer, R.J.; Bertagnolli, M.M.; Boland, C.R. Frequent Inactivation of PTEN by Promoter Hypermethylation in Microsatellite Instability-High Sporadic Colorectal Cancers. Cancer Res. 2004, 64, 3014–3021. [Google Scholar] [CrossRef]
- Baharudin, R.; Ishak, M.; Muhamad Yusof, A.; Saidin, S.; Syafruddin, S.E.; Wan Mohamad Nazarie, W.F.; Lee, L.-H.; Ab Mutalib, N.-S. Epigenome-Wide DNA Methylation Profiling in Colorectal Cancer and Normal Adjacent Colon Using Infinium Human Methylation 450K. Diagnostics 2022, 12, 198. [Google Scholar] [CrossRef] [PubMed]
- Barlow, D.P.; Stöger, R.; Herrmann, B.G.; Saito, K.; Schweifer, N. The Mouse Insulin-like Growth Factor Type-2 Receptor Is Imprinted and Closely Linked to the Tme Locus. Nature 1991, 349, 84–87. [Google Scholar] [CrossRef] [PubMed]
- Giannoukakis, N.; Deal, C.; Paquette, J.; Goodyer, C.G.; Polychronakos, C. Parental Genomic Imprinting of the Human IGF2 Gene. Nat. Genet. 1993, 4, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Takano, Y.; Shiota, G.; Kawasaki, H. Analysis of Genomic Imprinting of Insulin-Like Growth Factor 2 in Colorectal Cancer. Oncology 2000, 59, 210–216. [Google Scholar] [CrossRef]
- Cui, H.; Cruz-Correa, M.; Giardiello, F.M.; Hutcheon, D.F.; Kafonek, D.R.; Brandenburg, S.; Wu, Y.; He, X.; Powe, N.R.; Feinberg, A.P. Loss of IGF2 Imprinting: A Potential Marker of Colorectal Cancer Risk. Science 2003, 299, 1753–1755. [Google Scholar] [CrossRef]
- Tian, F.; Tang, Z.; Song, G.; Pan, Y.; He, B.; Bao, Q.; Wang, S. Loss of Imprinting of IGF2 Correlates with Hypomethylation of the H19 Differentially Methylated Region in the Tumor Tissue of Colorectal Cancer Patients. Mol. Med. Rep. 2012, 5, 1536–1540. [Google Scholar] [CrossRef]
- Belharazem, D.; Magdeburg, J.; Berton, A.-K.; Beissbarth, L.; Sauer, C.; Sticht, C.; Marx, A.; Hofheinz, R.; Post, S.; Kienle, P.; et al. Carcinoma of the Colon and Rectum with Deregulation of Insulin-like Growth Factor 2 Signaling: Clinical and Molecular Implications. J. Gastroenterol. 2016, 51, 971–984. [Google Scholar] [CrossRef]
- Schwarzenbach, H. Biological and Clinical Relevance of H19 in Colorectal Cancer Patients. EBioMedicine 2016, 13, 9–10. [Google Scholar] [CrossRef]
- Yang, J.; Qi, M.; Fei, X.; Wang, X.; Wang, K. LncRNA H19: A Novel Oncogene in Multiple Cancers. Int. J. Biol. Sci. 2021, 17, 3188–3208. [Google Scholar] [CrossRef]
- Yin, Y.; Yan, Z.-P.; Lu, N.-N.; Xu, Q.; He, J.; Qian, X.; Yu, J.; Guan, X.; Jiang, B.-H.; Liu, L.-Z. Downregulation of MiR-145 Associated with Cancer Progression and VEGF Transcriptional Activation by Targeting N-RAS and IRS1. Biochim. Biophys. Acta Gene Regul. Mech. 2013, 1829, 239–247. [Google Scholar] [CrossRef]
- Pekow, J.; Meckel, K.; Dougherty, U.; Butun, F.; Mustafi, R.; Lim, J.; Crofton, C.; Chen, X.; Joseph, L.; Bissonnette, M. Tumor Suppressors MiR-143 and MiR-145 and Predicted Target Proteins API5, ERK5, K-RAS, and IRS-1 Are Differentially Expressed in Proximal and Distal Colon. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 308, G179–G187. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Chen, J.; Gao, C.; Zhu, D.; Xu, X.; Wu, C.; Jiang, J. MicroRNA 497 Inhibits Tumor Growth through Targeting Insulin Receptor Substrate 1 in Colorectal Cancer. Oncol. Lett. 2017, 14, 6379–6386. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ye, S.; Yu, C.; Zhang, G.; Shi, F.; Chen, Y.; Yang, J.; Wu, W.; Zhou, Y. Downregulation of MicroRNA 126 Is Inversely Correlated with Insulin Receptor Substrate 1 Protein Expression in Colorectal Cancer and Is Associated with Advanced Stages of Disease. Oncol. Lett. 2020, 20, 2411–2419. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.T.; Jiang, C.C.; Wang, G.P.; Li, Y.P.; Wang, C.Y.; Guo, X.Y.; Yang, R.H.; Feng, Y.; Wang, F.H.; Tseng, H.-Y.; et al. MicroRNA-497 Targets Insulin-like Growth Factor 1 Receptor and Has a Tumour Suppressive Role in Human Colorectal Cancer. Oncogene 2013, 32, 1910–1920. [Google Scholar] [CrossRef]
- Su, J.; Liang, H.; Yao, W.; Wang, N.; Zhang, S.; Yan, X.; Feng, H.; Pang, W.; Wang, Y.; Wang, X.; et al. MiR-143 and MiR-145 Regulate IGF1R to Suppress Cell Proliferation in Colorectal Cancer. PLoS ONE 2014, 9, e114420. [Google Scholar] [CrossRef]
- Wu, G.; Liu, J.; Wu, Z.; Wu, X.; Yao, X. MicroRNA-184 Inhibits Cell Proliferation and Metastasis in Human Colorectal Cancer by Directly Targeting IGF-1R. Oncol. Lett. 2017, 14, 3215–3222. [Google Scholar] [CrossRef]
- Li, Z.; Pan, W.; Shen, Y.; Chen, Z.; Zhang, L.; Zhang, Y.; Luo, Q.; Ying, X. IGF1/IGF1R and MicroRNA Let-7e down-Regulate Each Other and Modulate Proliferation and Migration of Colorectal Cancer Cells. Cell Cycle 2018, 17, 1212–1219. [Google Scholar] [CrossRef]
- Liu, S.; Zhou, Y.; Zhou, Y.; Wang, J.; Ji, R. Mechanism of MiR 98 Inhibiting Tumor Proliferation and Invasion by Targeting IGF1R in Diabetic Patients Combined with Colon Cancer. Oncol. Lett. 2020, 20, 1719–1726. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, Q.; Li, M.; Jiang, S.; Wang, X. MicroRNA-375 Inhibits Colorectal Cancer Growth by Targeting PIK3CA. Biochem. Biophys. Res. Commun. 2014, 444, 199–204. [Google Scholar] [CrossRef]
- Ke, T.-W.; Wei, P.-L.; Yeh, K.-T.; Chen, W.T.-L.; Cheng, Y.-W. MiR-92a Promotes Cell Metastasis of Colorectal Cancer Through PTEN-Mediated PI3K/AKT Pathway. Ann. Surg. Oncol. 2015, 22, 2649–2655. [Google Scholar] [CrossRef]
- Wu, Y.; Song, Y.; Xiong, Y.; Wang, X.; Xu, K.; Han, B.; Bai, Y.; Li, L.; Zhang, Y.; Zhou, L. MicroRNA-21 (Mir-21) Promotes Cell Growth and Invasion by Repressing Tumor Suppressor PTEN in Colorectal Cancer. Cell. Physiol. Biochem. 2017, 43, 945–958. [Google Scholar] [CrossRef] [PubMed]
- Coronel-Hernández, J.; López-Urrutia, E.; Contreras-Romero, C.; Delgado-Waldo, I.; Figueroa-González, G.; Campos-Parra, A.D.; Salgado-García, R.; Martínez-Gutierrez, A.; Rodríguez-Morales, M.; Jacobo-Herrera, N.; et al. Cell Migration and Proliferation Are Regulated by MiR-26a in Colorectal Cancer via the PTEN–AKT Axis. Cancer Cell Int. 2019, 19, 80. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Guo, X.; Zhang, H.; Xiang, Y.; Chen, J.; Yin, Y.; Cai, X.; Wang, K.; Wang, G.; Ba, Y.; et al. Role of MiR-143 Targeting KRAS in Colorectal Tumorigenesis. Oncogene 2009, 28, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Liu, J.; Huo, J.; Li, K.; Li, K.; Guo, H.; Yang, Y. Downregulation of MiR 143 Modulates KRAS Expression in Colorectal Carcinoma Cells. Oncol. Rep. 2019, 42, 2759–2767. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Pan, X.; Li, Z.; Bai, P.; Jin, H.; Wang, T.; Song, C.; Zhang, L.; Gao, L. Association between Polymorphisms in the Promoter Region of MiR-143/145 and Risk of Colorectal Cancer. Hum. Immunol. 2013, 74, 993–997. [Google Scholar] [CrossRef]
- Liang, Y.; Sun, R.; Li, L.; Yuan, F.; Liang, W.; Wang, L.; Nie, X.; Chen, P.; Zhang, L.; Gao, L. A Functional Polymorphism in the Promoter of MiR-143/145 Is Associated with the Risk of Cervical Squamous Cell Carcinoma in Chinese Women. Medicine 2015, 94, e1289. [Google Scholar] [CrossRef]
- Chu, H.; Zhong, D.; Tang, J.; Li, J.; Xue, Y.; Tong, N.; Qin, C.; Yin, C.; Zhang, Z.; Wang, M. A Functional Variant in MiR-143 Promoter Contributes to Prostate Cancer Risk. Arch. Toxicol. 2016, 90, 403–414. [Google Scholar] [CrossRef]
- Sun, R.; Chen, P.; Li, L.; Sun, H.; Nie, X.; Liang, Y.; Yuan, F.; Pu, Y.; Bai, P.; Zhang, L.; et al. A Polymorphism Rs4705341 in the Flanking Region of MiR-143/145 Predicts Risk and Prognosis of Colorectal Cancer. Oncotarget 2016, 7, 62084–62090. [Google Scholar] [CrossRef]
- Yuan, F.; Sun, R.; Li, L.; Jin, B.; Wang, Y.; Liang, Y.; Che, G.; Gao, L.; Zhang, L. A Functional Variant Rs353292 in the Flanking Region of MiR-143/145 Contributes to the Risk of Colorectal Cancer. Sci. Rep. 2016, 6, 30195. [Google Scholar] [CrossRef]
- Wu, J.; Huang, Q.; Meng, D.; Huang, M.; Li, C.; Qin, T. A Functional Rs353293 Polymorphism in the Promoter of MiR-143/145 Is Associated with a Reduced Risk of Bladder Cancer. PLoS ONE 2016, 11, e0159115. [Google Scholar] [CrossRef]
- Li, C.; Zhang, Y.; Li, Y.; Ma, Q.; Liu, S.; Yao, Y.; Tan, F.; Shi, L.; Yao, Y. The Association of Polymorphisms in MiRNAs with Nonsmall Cell Lung Cancer in a Han Chinese Population. Cancer Manag. Res. 2018, 10, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, G.; Haupt, L.M.; Aquino, E.M.; Lea, R.A.; Sutherland, H.G.; Griffiths, L.R. Single Nucleotide Polymorphisms in MIR143 Contribute to Protection Against Non-Hodgkin Lymphoma (NHL) in Caucasian Populations. Genes 2019, 10, 185. [Google Scholar] [CrossRef] [PubMed]
- Kotarac, N.; Dobrijevic, Z.; Matijasevic, S.; Savic-Pavicevic, D.; Brajuskovic, G. Analysis of Association of Potentially Functional Genetic Variants within Genes Encoding MiR-34b/c, MiR-378 and MiR-143/145 with Prostate Cancer in Serbian Population. EXCLI J. 2019, 18, 515–529. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Li, X.; Quan, X.; Li, H.; Hao, X.; Jiang, M.; Zhou, B. Association between Two Polymorphisms in the Promoter Region of MiR-143/MiR-145 and the Susceptibility of Lung Cancer in Northeast Chinese Nonsmoking Females. DNA Cell Biol. 2019, 38, 814–823. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Pan, J.; Zhang, L.; Wei, Y.; Wang, C. Long Non-Coding RNA CRNDE Sponges MiR-384 to Promote Proliferation and Metastasis of Pancreatic Cancer Cells through Upregulating IRS1. Cell Prolif. 2017, 50, e12389. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Sha, H.; Sun, X.; Zhang, Y.; Wu, Y.; Zhang, J.; Zhang, H.; Wu, J.; Feng, J. CRNDE: An Oncogenic Long Non-Coding RNA in Cancers. Cancer Cell Int. 2020, 20, 162. [Google Scholar] [CrossRef]
- Poliseno, L.; Salmena, L.; Zhang, J.; Carver, B.; Haveman, W.J.; Pandolfi, P.P. A Coding-Independent Function of Gene and Pseudogene MRNAs Regulates Tumour Biology. Nature 2010, 465, 1033–1038. [Google Scholar] [CrossRef]
- Shang, A.-Q.; Wang, W.-W.; Yang, Y.-B.; Gu, C.-Z.; Ji, P.; Chen, C.; Zeng, B.-J.; Wu, J.-L.; Lu, W.-Y.; Sun, Z.-J.; et al. Knockdown of Long Noncoding RNA PVT1 Suppresses Cell Proliferation and Invasion of Colorectal Cancer via Upregulation of MicroRNA-214-3p. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 317, G222–G232. [Google Scholar] [CrossRef]
- Mancarella, C.; Scotlandi, K. IGF2BP3 from Physiology to Cancer: Novel Discoveries, Unsolved Issues, and Future Perspectives. Front. Cell Dev. Biol. 2020, 7, 363. [Google Scholar] [CrossRef]
- Cao, J.; Mu, Q.; Huang, H. The Roles of Insulin-Like Growth Factor 2 MRNA-Binding Protein 2 in Cancer and Cancer Stem Cells. Stem Cells Int. 2018, 2018, 4217259. [Google Scholar] [CrossRef]
- Chen, H.-M.; Lin, C.-C.; Chen, W.-S.; Jiang, J.-K.; Yang, S.-H.; Chang, S.-C.; Ho, C.-L.; Yang, C.-C.; Huang, S.-C.; Chao, Y.; et al. Insulin-Like Growth Factor 2 MRNA-Binding Protein 1 (IGF2BP1) Is a Prognostic Biomarker and Associated with Chemotherapy Responsiveness in Colorectal Cancer. Int. J. Mol. Sci. 2021, 22, 6940. [Google Scholar] [CrossRef] [PubMed]
- Abrantes, A.M.; Serra, M.E.S.; Gonçalves, A.C.; Rio, J.; Oliveiros, B.; Laranjo, M.; Rocha-Gonsalves, A.M.; Sarmento-Ribeiro, A.B.; Botelho, M.F. Hypoxia-Induced Redox Alterations and Their Correlation with 99mTc-MIBI and 99mTc-HL-91 Uptake in Colon Cancer Cells. Nucl. Med. Biol. 2010, 37, 125–132. [Google Scholar] [CrossRef]
- Cao, D.; Hou, M.; Guan, Y.; Jiang, M.; Yang, Y.; Gou, H. Expression of HIF-1alpha and VEGF in Colorectal Cancer: Association with Clinical Outcomes and Prognostic Implications. BMC Cancer 2009, 9, 432. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Xu, Z.; Mao, S.; Chen, W.; Zeng, R.; Zhou, S.; Liu, J. Effect of Hypoxia on Hypoxia Inducible Factor-1α, Insulin-like Growth Factor I and Vascular Endothelial Growth Factor Expression in Hepatocellular Carcinoma HepG2 Cells. Oncol. Lett. 2015, 9, 1142–1148. [Google Scholar] [CrossRef]
- Joung, Y.-H.; Lee, M.-Y.; Lim, E.-J.; Kim, M.-S.; Hwang, T.S.; Kim, S.-Y.; Ye, S.-K.; Lee, J.-D.; Park, T.; Woo, Y.-S.; et al. Hypoxia Activates the IGF-1 Expression through STAT5b in Human HepG2 Cells. Biochem. Biophys. Res. Commun. 2007, 358, 733–738. [Google Scholar] [CrossRef] [PubMed]
- Loh, C.-Y.; Chai, J.; Tang, T.; Wong, W.; Sethi, G.; Shanmugam, M.; Chong, P.; Looi, C. The E-Cadherin and N-Cadherin Switch in Epithelial-to-Mesenchymal Transition: Signaling, Therapeutic Implications, and Challenges. Cells 2019, 8, 1118. [Google Scholar] [CrossRef]
- Canonici, A.; Steelant, W.; Rigot, V.; Khomitch-Baud, A.; Boutaghou-Cherid, H.; Bruyneel, E.; van Roy, F.; Garrouste, F.; Pommier, G.; André, F. Insulin-like Growth Factor-I Receptor, E-Cadherin and Av Integrin Form a Dynamic Complex under the Control of α-Catenin. Int. J. Cancer 2008, 122, 572–582. [Google Scholar] [CrossRef]
- André, F.; Janssens, B.; Bruyneel, E.; van Roy, F.; Gespach, C.; Mareel, M.; Bracke, M. Alpha-Catenin Is Required for IGF-I-Induced Cellular Migration but Not Invasion in Human Colonic Cancer Cells. Oncogene 2004, 23, 1177–1186. [Google Scholar] [CrossRef]
- Vella, V.; Malaguarnera, R.; Nicolosi, M.L.; Morrione, A.; Belfiore, A. Insulin/IGF Signaling and Discoidin Domain Receptors: An Emerging Functional Connection. Biochim. Biophys. Acta Mol. Cell Res. 2019, 1866, 118522. [Google Scholar] [CrossRef]
- Malaguarnera, R.; Nicolosi, M.L.; Sacco, A.; Morcavallo, A.; Vella, V.; Voci, C.; Spatuzza, M.; Xu, S.-Q.; Iozzo, R.V.; Vigneri, R.; et al. Novel Cross-Talk between IGF-IR and DDR1 Regulates IGF-IR Trafficking, Signaling and Biological Responses. Oncotarget 2015, 6, 16084–16105. [Google Scholar] [CrossRef]
- ben Arfi, K.; Schneider, C.; Bennasroune, A.; Bouland, N.; Wolak-Thierry, A.; Collin, G.; Le, C.C.; Toussaint, K.; Hachet, C.; Lehrter, V.; et al. Discoidin Domain Receptor 1 Expression in Colon Cancer: Roles and Prognosis Impact. Cancers 2022, 14, 928. [Google Scholar] [CrossRef] [PubMed]
- Reszegi, A.; Horváth, Z.; Karászi, K.; Regős, E.; Postniková, V.; Tátrai, P.; Kiss, A.; Schaff, Z.; Kovalszky, I.; Baghy, K. The Protective Role of Decorin in Hepatic Metastasis of Colorectal Carcinoma. Biomolecules 2020, 10, 1199. [Google Scholar] [CrossRef]
- Iozzo, R.V.; Buraschi, S.; Genua, M.; Xu, S.-Q.; Solomides, C.C.; Peiper, S.C.; Gomella, L.G.; Owens, R.C.; Morrione, A. Decorin Antagonizes IGF Receptor I (IGF-IR) Function by Interfering with IGF-IR Activity and Attenuating Downstream Signaling. J. Biol. Chem. 2011, 286, 34712–34721. [Google Scholar] [CrossRef] [PubMed]
- Morrione, A.; Neill, T.; Iozzo, R.V. Dichotomy of Decorin Activity on the Insulin-like Growth Factor-I System. FEBS J. 2013, 280, 2138–2149. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Yang, J.; Yue, J.; Chen, Y.; Zhou, H.; Fan, D.; Zhang, Q.; Buraschi, S.; Iozzo, R.V.; Bi, X. Decorin Deficiency Promotes Epithelial-Mesenchymal Transition and Colon Cancer Metastasis. Matrix Biol. 2021, 95, 1–14. [Google Scholar] [CrossRef] [PubMed]
- de Jong, G.; van Dijk, J.P.; van Eijk, H.G. The Biology of Transferrin. Clin. Chim. Acta 1990, 190, 1–46. [Google Scholar] [CrossRef]
- Miljuš, G.; Malenković, V.; Đukanović, B.; Kolundžić, N.; Nedić, O. IGFBP-3/Transferrin/Transferrin Receptor 1 Complexes as Principal Mediators of IGFBP-3 Delivery to Colon Cells in Non-Cancer and Cancer Tissues. Exp. Mol. Pathol. 2015, 98, 431–438. [Google Scholar] [CrossRef]
- Westwood, M.; Aplin, J.D.; Collinge, I.A.; Gill, A.; White, A.; Gibson, J.M. A2-Macroglobulin: A New Component in the Insulin-like Growth Factor/Insulin-like Growth Factor Binding Protein-1 Axis. J. Biol. Chem. 2001, 276, 41668–41674. [Google Scholar] [CrossRef]
- Šunderić, M.; Malenković, V.; Nedić, O. Complexes between Insulin-like Growth Factor Binding Proteins and Alpha-2-Macroglobulin in Patients with Tumor. Exp. Mol. Pathol. 2015, 98, 173–177. [Google Scholar] [CrossRef]
- Gligorijević, N.; Penezić, A.; Nedić, O. Influence of Glyco-Oxidation on Complexes between Fibrin (Ogen) and Insulin-like Growth Factor-Binding Protein-1 in Patients with Diabetes Mellitus Type 2. Free Radic. Res. 2017, 51, 64–72. [Google Scholar] [CrossRef]
- Orrù, S.; Nigro, E.; Mandola, A.; Alfieri, A.; Buono, P.; Daniele, A.; Mancini, A.; Imperlini, E. A Functional Interplay between IGF-1 and Adiponectin. Int. J. Mol. Sci. 2017, 18, 2145. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, M.J.; Cooke, A.; Rosenfeld-Franklin, M.; Buck, E.; Foreman, K.; Landfair, D.; O’Connor, M.; Pirritt, C.; Sun, Y.; Yao, Y.; et al. Discovery of OSI-906: A Selective and Orally Efficacious Dual Inhibitor of the IGF-1 Receptor and Insulin Receptor. Future Med. Chem. 2009, 1, 1153–1171. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Mehrabi Nasab, E.; Hassanpour, F.; Athari, S.S. Linsitinib and Aspirin as the IGF1-R Antagonists, Inhibit Regorafenib-Resistant Chemotherapy in Colon Cancer. Saudi J. Biol. Sci. 2022, 29, 872–877. [Google Scholar] [CrossRef] [PubMed]
- Leiphrakpam, P.D.; Agarwal, E.; Mathiesen, M.; Haferbier, K.L.; Brattain, M.G.; Chowdhury, S. In Vivo Analysis of Insulin-like Growth Factor Type 1 Receptor Humanized Monoclonal Antibody MK-0646 and Small Molecule Kinase Inhibitor OSI-906 in Colorectal Cancer. Oncol. Rep. 2014, 31, 87–94. [Google Scholar] [CrossRef]
- Reidy, D.L.; Vakiani, E.; Fakih, M.G.; Saif, M.W.; Hecht, J.R.; Goodman-Davis, N.; Hollywood, E.; Shia, J.; Schwartz, J.; Chandrawansa, K.; et al. Randomized, Phase II Study of the Insulin-Like Growth Factor-1 Receptor Inhibitor IMC-A12, With or Without Cetuximab, in Patients with Cetuximab- or Panitumumab-Refractory Metastatic Colorectal Cancer. J. Clin. Oncol. 2010, 28, 4240–4246. [Google Scholar] [CrossRef]
- Sclafani, F.; Kim, T.Y.; Cunningham, D.; Kim, T.W.; Tabernero, J.; Schmoll, H.J.; Roh, J.K.; Kim, S.Y.; Park, Y.S.; Guren, T.K.; et al. A Randomized Phase II/III Study of Dalotuzumab in Combination with Cetuximab and Irinotecan in Chemorefractory, KRAS Wild-Type, Metastatic Colorectal Cancer. J. Natl. Cancer Inst. 2015, 107, djv258. [Google Scholar] [CrossRef]
- Wilky, B.A.; Rudek, M.A.; Ahmed, S.; Laheru, D.A.; Cosgrove, D.; Donehower, R.C.; Nelkin, B.; Ball, D.; Doyle, L.A.; Chen, H.; et al. A Phase I Trial of Vertical Inhibition of IGF Signalling Using Cixutumumab, an Anti-IGF-1R Antibody, and Selumetinib, an MEK 1/2 Inhibitor, in Advanced Solid Tumours. Br. J. Cancer 2015, 112, 24–31. [Google Scholar] [CrossRef]
- Tabernero, J.; Chawla, S.P.; Kindler, H.; Reckamp, K.; Chiorean, E.G.; Azad, N.S.; Lockhart, A.C.; Hsu, C.-P.; Baker, N.F.; Galimi, F.; et al. Anticancer Activity of the Type I Insulin-like Growth Factor Receptor Antagonist, Ganitumab, in Combination with the Death Receptor 5 Agonist, Conatumumab. Target. Oncol. 2015, 10, 65–76. [Google Scholar] [CrossRef]
- Zhong, H.; Fazenbaker, C.; Chen, C.; Breen, S.; Huang, J.; Yao, X.; Ren, P.; Yao, Y.; Herbst, R.; Hollingsworth, R.E. Overproduction of IGF-2 Drives a Subset of Colorectal Cancer Cells, Which Specifically Respond to an Anti-IGF Therapeutic Antibody and Combination Therapies. Oncogene 2017, 36, 797–806. [Google Scholar] [CrossRef]
- Oh, Y.J.; Dent, M.W.; Freels, A.R.; Zhou, Q.; Lebrilla, C.B.; Merchant, M.L.; Matoba, N. Antitumor Activity of a Lectibody Targeting Cancer-Associated High-Mannose Glycans. Mol. Ther. 2022, 30, 1523–1535. [Google Scholar] [CrossRef]
- Zong, R.; Chen, X.; Feng, J.; Xu, S. IGF-1R Depletion Sensitizes Colon Cancer Cell Lines to Radiotherapy. Cancer Biomark. 2021, 32, 199–206. [Google Scholar] [CrossRef]
- Li, Y.; Lu, K.; Zhao, B.; Zeng, X.; Xu, S.; Ma, X.; Zhi, Y. Depletion of Insulin-like Growth Factor 1 Receptor Increases Radiosensitivity in Colorectal Cancer. J. Gastrointest. Oncol. 2020, 11, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Baile, M.; Ventero, M.P.; Encinar, J.A.; García-Morales, P.; Poveda-Deltell, M.; Pérez-Valenciano, E.; Barberá, V.M.; Gallego-Plazas, J.; Rodríguez-Lescure, Á.; Martín-Nieto, J.; et al. Differential Effects of IGF-1R Small Molecule Tyrosine Kinase Inhibitors BMS-754807 and OSI-906 on Human Cancer Cell Lines. Cancers 2020, 12, 3717. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, Y.; Zhu, J.; Zheng, H.; Chen, S.; Chen, L.; Yang, H.-S. IGF-1R Inhibition Induces MEK Phosphorylation to Promote Survival in Colon Carcinomas. Signal Transduct. Target. Ther. 2020, 5, 153. [Google Scholar] [CrossRef]
- Sanderson, M.P.; Apgar, J.; Garin-Chesa, P.; Hofmann, M.H.; Kessler, D.; Quant, J.; Savchenko, A.; Schaaf, O.; Treu, M.; Tye, H.; et al. BI 885578, a Novel IGF1R/INSR Tyrosine Kinase Inhibitor with Pharmacokinetic Properties That Dissociate Antitumor Efficacy and Perturbation of Glucose Homeostasis. Mol. Cancer Ther. 2015, 14, 2762–2772. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Zhao, N.; Jiang, M. Isovitexin Attenuates Tumor Growth in Human Colon Cancer Cells through the Modulation of Apoptosis and Epithelial-Mesenchymal Transition via PI3K/Akt/MTOR Signaling Pathway. Biochem. Cell Biol. 2021, 99, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.J.; Hong, E.M.; Park, S.W.; Byun, H.W.; Koh, D.H.; Choi, M.H.; Kae, S.H.; Lee, J. Statin Induces Apoptosis of Human Colon Cancer Cells and Downregulation of Insulin-like Growth Factor 1 Receptor via Proapoptotic ERK Activation. Oncol. Lett. 2016, 12, 250–256. [Google Scholar] [CrossRef]
- Kang, M.-H.; Kim, I.-H.; Nam, T.-J. Phloroglucinol Induces Apoptosis through the Regulation of Insulin-like Growth Factor 1 Receptor Signaling Pathways in Human Colon Cancer HT-29 Cells. Int. J. Oncol. 2014, 45, 1036–1042. [Google Scholar] [CrossRef]
- Wang, J.; Huang, F.; Bai, Z.; Chi, B.; Wu, J.; Chen, X. Curcumol Inhibits Growth and Induces Apoptosis of Colorectal Cancer LoVo Cell Line via IGF-1R and P38 MAPK Pathway. Int. J. Mol. Sci. 2015, 16, 19851–19867. [Google Scholar] [CrossRef]
- Cianciosi, D.; Forbes-Hernández, T.Y.; Regolo, L.; Alvarez-Suarez, J.M.; Quinzi, D.; Sargenti, A.; Bai, W.; Tian, L.; Giampieri, F.; Battino, M. Manuka Honey in Combination with 5-Fluorouracil Decreases Physical Parameters of Colonspheres Enriched with Cancer Stem-like Cells and Reduces Their Resistance to Apoptosis. Food Chem. 2022, 374, 131753. [Google Scholar] [CrossRef]
- Valadez-Bustos, N.; Escamilla-Silva, E.M.; García-Vázquez, F.J.; Gallegos-Corona, M.A.; Amaya-Llano, S.L.; Ramos-Gómez, M. Oral Administration of Microencapsulated B. longum BAA-999 and Lycopene Modulates IGF-1/IGF-1R/IGFBP3 Protein Expressions in a Colorectal Murine Model. Int. J. Mol. Sci. 2019, 20, 4275. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Zheng, W.; Luo, Y.; Ou, X.; Song, L.; Zhang, S.; He, T.; Guo, Z.; Zhu, J.; Shi, H.; et al. Arca Subcrenata Polypeptides Inhibit Human Colorectal Cancer HT-29 Cells Growth via Suppression of IGF-1R/Akt/MTOR Signaling and ATP Production. Nutr. Cancer 2020, 72, 260–272. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Kim, J.-S.; Seo, Y.R.; Park, J.H.Y.; Choi, M.-S.; Sung, M.-K. Carnosic Acid Suppresses Colon Tumor Formation in Association with Antiadipogenic Activity. Mol. Nutr. Food Res. 2014, 58, 2274–2285. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.-H.; Nam, T.-J. Fucoidan Downregulates Insulin-like Growth Factor-I Receptor Levels in HT-29 Human Colon Cancer Cells. Oncol. Rep. 2018, 39, 1516–1522. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-K.; Kim, I.-H.; Kim, J.; Nam, T.-J. Induction of Apoptosis by Laminarin, Regulating the Insulin-like Growth Factor-IR Signaling Pathways in HT-29 Human Colon Cells. Int. J. Mol. Med. 2012, 30, 734–738. [Google Scholar] [CrossRef]
- Hosseini, S.A.; Zand, H.; Cheraghpour, M. The Influence of Curcumin on the Downregulation of MYC, Insulin and IGF-1 Receptors: A Possible Mechanism Underlying the Anti-Growth and Anti-Migration in Chemoresistant Colorectal Cancer Cells. Medicina 2019, 55, 90. [Google Scholar] [CrossRef]
- Lim, D.Y.; Cho, H.J.; Kim, J.; Nho, C.W.; Lee, K.W.; Park, J.H.Y. Luteolin Decreases IGF-II Production and Downregulates Insulin-like Growth Factor-I Receptor Signaling in HT-29 Human Colon Cancer Cells. BMC Gastroenterol. 2012, 12, 9. [Google Scholar] [CrossRef]
- Li, J.; Teng, Y.; Liu, S.; Wang, Z.; Chen, Y.; Zhang, Y.; Xi, S.; Xu, S.; Wang, R.; Zou, X. Cinnamaldehyde Affects the Biological Behavior of Human Colorectal Cancer Cells and Induces Apoptosis via Inhibition of the PI3K/Akt Signaling Pathway. Oncol. Rep. 2016, 35, 1501–1510. [Google Scholar] [CrossRef]

| IGFBP | Role | Possible Modes of Action | Reference |
|---|---|---|---|
| IGFBP-1 | Protective | Decreased concentration leads to an increased concentration of free IGF-I | [99] |
| IGFBP-2 | Tumor promoting | Local increase in free IGF-I due to proteolysis and binding to α5β1 integrin | [60,85,86] |
| IGFBP-3 | Protective | Binding of free IGF-I, pro-apoptotic role through NF-κB inhibition, interaction with retinoid X receptor (RXR)/Nur77 or p53-dependent signaling cascade | [79,80,95,96,97] |
| Tumor promoting | Undefined mechanisms dependent on other members of the IGF system, mainly the concentrations of IGF-I and IGF-II | [81,82] | |
| IGFBP-4 | Protective | Inhibits growth of colon cancer cells | [89,90] |
| IGFBP-5 | Not known | - | - |
| IGFBP-6 | Protective | Inhibition of IGF-II induced proliferation and migration, protection via IGF-independent pathways | [91,92,93,101] |
| Tumor promoting | Stimulation of LIM 1215 colon cancer cell migration | [102] | |
| IGFBP-7 | Possibly protective | Possibly associated with the activation of apoptosis, but further evidence is required | [108] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gligorijević, N.; Dobrijević, Z.; Šunderić, M.; Robajac, D.; Četić, D.; Penezić, A.; Miljuš, G.; Nedić, O. The Insulin-like Growth Factor System and Colorectal Cancer. Life 2022, 12, 1274. https://doi.org/10.3390/life12081274
Gligorijević N, Dobrijević Z, Šunderić M, Robajac D, Četić D, Penezić A, Miljuš G, Nedić O. The Insulin-like Growth Factor System and Colorectal Cancer. Life. 2022; 12(8):1274. https://doi.org/10.3390/life12081274
Chicago/Turabian StyleGligorijević, Nikola, Zorana Dobrijević, Miloš Šunderić, Dragana Robajac, Danilo Četić, Ana Penezić, Goran Miljuš, and Olgica Nedić. 2022. "The Insulin-like Growth Factor System and Colorectal Cancer" Life 12, no. 8: 1274. https://doi.org/10.3390/life12081274
APA StyleGligorijević, N., Dobrijević, Z., Šunderić, M., Robajac, D., Četić, D., Penezić, A., Miljuš, G., & Nedić, O. (2022). The Insulin-like Growth Factor System and Colorectal Cancer. Life, 12(8), 1274. https://doi.org/10.3390/life12081274








