Prevention and Management Strategies for Diabetic Neuropathy
Abstract
:1. Introduction
2. Epidemiology
3. Clinical Manifestations
4. Prevention Strategies
4.1. Glycemic Control
4.2. Lifestyle Modifications
4.3. Footcare
5. Diagnostic Evaluation
6. Management Strategies
6.1. Anticonvulsants
6.2. Serotonin and Norepinephrine Reuptake Inhibitors (SNRIs)
6.3. Tricyclic Antidepressants (TCAs)
6.4. Opioids
6.5. Topical Analgesics
6.6. Intravenous (IV) Medications
7. Novel Therapies
7.1. Neuromodulation Devices
7.1.1. Spinal Cord Stimulation (SCS)
7.1.2. Frequency-Modulated Electromagnetic Neural Stimulation (FREMS)
7.1.3. Transcutaneous Electrical Nerve Stimulation (TENS)
7.1.4. Neuromuscular Electrical Stimulation (NMES)
7.2. Nutraceuticals
7.2.1. α-Lipoic Acid (ALA)
7.2.2. Vitamin B12
7.2.3. Acetyl-L-Carnitine (ALC)
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and Regional Diabetes Prevalence Estimates for 2019 and Projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th Edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pop-Busui, R.; Ang, L.; Boulton, A.J.M.; Feldman, E.L.; Marcus, R.L.; Mizokami-Stout, K.; Singleton, J.R.; Ziegler, D. Diag-nosis and Treatment of Painful Diabetic Peripheral Neuropathy. ADA Clin. Compend. 2022, 2022, 1–32. [Google Scholar] [CrossRef]
- Vileikyte, L.; Gonzalez, J.S. Chapter 14—Recognition and Management of Psychosocial Issues in Diabetic Neuropathy. In Handbook of Clinical Neurology; Zochodne, D.W., Malik, R.A., Eds.; Diabetes and the Nervous System; Elsevier: Amsterdam, The Netherlands, 2014; Volume 126, pp. 195–209. [Google Scholar]
- Pop-Busui, R.; Boulton, A.J.M.; Feldman, E.L.; Bril, V.; Freeman, R.; Malik, R.A.; Sosenko, J.M.; Ziegler, D. Diabetic Neuropathy: A Position Statement by the American Diabetes Association. Diabetes Care 2017, 40, 136–154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hex, N.; Bartlett, C.; Wright, D.; Taylor, M.; Varley, D. Estimating the Current and Future Costs of Type 1 and Type 2 Diabetes in the UK, Including Direct Health Costs and Indirect Societal and Productivity Costs. Diabet. Med. 2012, 29, 855–862. [Google Scholar] [CrossRef]
- Kerr, M.; Barron, E.; Chadwick, P.; Evans, T.; Kong, W.M.; Rayman, G.; Sutton-Smith, M.; Todd, G.; Young, B.; Jeffcoate, W.J. The Cost of Diabetic Foot Ulcers and Amputations to the National Health Service in England. Diabet. Med. 2019, 36, 995–1002. [Google Scholar] [CrossRef]
- Feldman, E.L.; Callaghan, B.C.; Pop-Busui, R.; Zochodne, D.W.; Wright, D.E.; Bennett, D.L.; Bril, V.; Russell, J.W.; Viswanathan, V. Diabetic Neuropathy. Nat. Rev. Dis. Primer 2019, 5, 41. [Google Scholar] [CrossRef]
- Sloan, G.; Selvarajah, D.; Tesfaye, S. Pathogenesis, Diagnosis and Clinical Management of Diabetic Sensorimotor Peripheral Neuropathy. Nat. Rev. Endocrinol. 2021, 17, 400–420. [Google Scholar] [CrossRef]
- Smith, S.; Normahani, P.; Lane, T.; Hohenschurz-Schmidt, D.; Oliver, N.; Davies, A.H. Pathogenesis of Distal Symmetrical Polyneuropathy in Diabetes. Life 2022, 12, 1074. [Google Scholar] [CrossRef]
- Malik, R.A.; Tesfaye, S.; Newrick, P.G.; Walker, D.; Rajbhandari, S.M.; Siddique, I.; Sharma, A.K.; Boulton, A.J.M.; King, R.H.M.; Thomas, P.K.; et al. Sural Nerve Pathology in Diabetic Patients with Minimal but Progressive Neuropathy. Diabetologia 2005, 48, 578–585. [Google Scholar] [CrossRef]
- Dey, I.; Midha, N.; Singh, G.; Forsyth, A.; Walsh, S.K.; Singh, B.; Kumar, R.; Toth, C.; Midha, R. Diabetic Schwann Cells Suffer from Nerve Growth Factor and Neurotrophin-3 Underproduction and Poor Associability with Axons. Glia 2013, 61, 1990–1999. [Google Scholar] [CrossRef]
- Liu, Y.-P.; Shao, S.-J.; Guo, H.-D. Schwann Cells Apoptosis Is Induced by High Glucose in Diabetic Peripheral Neuropathy. Life Sci. 2020, 248, 117459. [Google Scholar] [CrossRef]
- Padilla, A.; Descorbeth, M.; Almeyda, A.L.; Payne, K.; De Leon, M. Hyperglycemia Magnifies Schwann Cell Dysfunction and Cell Death Triggered by PA-Induced Lipotoxicity. Brain Res. 2011, 1370, 64–79. [Google Scholar] [CrossRef] [Green Version]
- Baptista, F.I.; Pinheiro, H.; Gomes, C.A.; Ambrósio, A.F. Impairment of Axonal Transport in Diabetes: Focus on the Putative Mechanisms Underlying Peripheral and Central Neuropathies. Mol. Neurobiol. 2019, 56, 2202–2210. [Google Scholar] [CrossRef]
- Ryle, C.; Donaghy, M. Non-Enzymatic Glycation of Peripheral Nerve Proteins in Human Diabetics. J. Neurol. Sci. 1995, 129, 62–68. [Google Scholar] [CrossRef]
- Fernyhough, P.; Diemel, L.T.; Tomlinson, D.R. Target Tissue Production and Axonal Transport of Neurotrophin-3 Are Reduced in Streptozotocin-Diabetic Rats. Diabetologia 1998, 41, 300–306. [Google Scholar] [CrossRef] [Green Version]
- Sima, A.A.; Lattimer, S.A.; Yagihashi, S.; Greene, D.A. Axo-glial dysjunction. A novel structural lesion that accounts for poorly reversible slowing of nerve conduction in the spontaneously diabetic bio-breeding rat. J. Clin. Investig. 1986, 77, 474–484. [Google Scholar] [CrossRef] [Green Version]
- Said, G.; Slama, G.; Selva, J. Progressive Centripetal Degeneration of Axons in Small Fibre Diabetic Polyneuropathy: A Clinical and Pathological Study. Brain 1983, 106, 791–807. [Google Scholar] [CrossRef]
- Zochodne, D.W. Chapter 26—Mechanisms of Diabetic Neuron Damage: Molecular Pathways. In Diabetes and the Nervous System; Elsevier: Amsterdam, The Netherlands, 2014; Volume 126, pp. 379–399. [Google Scholar]
- Selvarajah, D.; Wilkinson, I.D.; Emery, C.J.; Harris, N.D.; Shaw, P.J.; Witte, D.R.; Griffiths, P.D.; Tesfaye, S. Early Involvement of the Spinal Cord in Diabetic Peripheral Neuropathy. Diabetes Care 2006, 29, 2664–2669. [Google Scholar] [CrossRef] [Green Version]
- Malik, R.A.; Newrick, P.G.; Sharma, A.K.; Jennings, A.; Ah-See, A.K.; Mayhew, T.M.; Jakubowski, J.; Boulton, A.J.; Ward, J.D. Microangiopathy in Human Diabetic Neuropathy: Relationship between Capillary Abnormalities and the Severity of Neuropathy. Diabetologia 1989, 32, 92–102. [Google Scholar] [CrossRef] [Green Version]
- Yasuda, H.; Dyck, P.J. Abnormalities of Endoneurial Microvessels and Sural Nerve Pathology in Diabetic Neuropathy. Neurology 1987, 37, 20–28. [Google Scholar] [CrossRef]
- Tesfaye, S.; Harris, N.; Jakubowski, J.J.; Mody, C.; Wilson, R.M.; Rennie, I.G.; Ward, J.D. Impaired Blood Flow and Arterio-Venous Shunting in Human Diabetic Neuropathy: A Novel Technique of Nerve Photography and Fluorescein Angiography. Diabetologia 1993, 36, 1266–1274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dyck, P.J.; Lais, A.; Karnes, J.L.; O’Brien, P.; Rizza, R. Fiber Loss Is Primary and Multifocal in Sural Nerves in Diabetic Polyneuropathy. Ann. Neurol. 1986, 19, 425–439. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Vanhoutte, P.M. Macro- and Microvascular Endothelial Dysfunction in Diabetes. J. Diabetes 2017, 9, 434–449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Madonna, R.; Balistreri, C.R.; Geng, Y.-J.; De Caterina, R. Diabetic Microangiopathy: Pathogenetic Insights and Novel Therapeutic Approaches. Vascul. Pharmacol. 2017, 90, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Quattrini, C.; Jeziorska, M.; Boulton, A.J.M.; Malik, R.A. Reduced Vascular Endothelial Growth Factor Expression and Intra-Epidermal Nerve Fiber Loss in Human Diabetic Neuropathy. Diabetes Care 2008, 31, 140–145. [Google Scholar] [CrossRef] [Green Version]
- Britland, S.T.; Young, R.J.; Ashutosh, S.K.; Clarke, B.F. Relationship of Endoneurial Capillary Abnormalities to Type and Severity of Diabetic Polyneuropathy. Diabetes 1990, 39, 909–913. [Google Scholar] [CrossRef]
- Malik, R.A.; Veves, A.; Masson, E.A.; Sharma, A.K.; Ah-See, A.K.; Schady, W.; Lye, R.H.; Boulton, A.J. Endoneurial Capillary Abnormalities in Mild Human Diabetic Neuropathy. J. Neurol. Neurosurg. Psychiatry 1992, 55, 557–561. [Google Scholar] [CrossRef] [Green Version]
- Cameron, N.E.; Eaton, S.E.M.; Cotter, M.A.; Tesfaye, S. Vascular Factors and Metabolic Interactions in the Pathogenesis of Diabetic Neuropathy. Diabetologia 2001, 44, 1973–1988. [Google Scholar] [CrossRef] [Green Version]
- Finnerup, N.B.; Attal, N.; Haroutounian, S.; McNicol, E.; Baron, R.; Dworkin, R.H.; Gilron, I.; Haanpää, M.; Hansson, P.; Jensen, T.S.; et al. Pharmacotherapy for Neuropathic Pain in Adults: A Systematic Review and Meta-Analysis. Lancet Neurol. 2015, 14, 162–173. [Google Scholar] [CrossRef] [Green Version]
- Braffett, B.H.; Gubitosi-Klug, R.A.; Albers, J.W.; Feldman, E.L.; Martin, C.L.; White, N.H.; Orchard, T.J.; Lopes-Virella, M.; Lachin, J.M.; Pop-Busui, R. Risk Factors for Diabetic Peripheral Neuropathy and Cardiovascular Autonomic Neuropathy in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study. Diabetes 2020, 69, 1000–1010. [Google Scholar] [CrossRef]
- Hicks, C.W.; Selvin, E. Epidemiology of Peripheral Neuropathy and Lower Extremity Disease in Diabetes. Curr. Diab. Rep. 2019, 19, 86. [Google Scholar] [CrossRef]
- Jaiswal, M.; Lauer, A.; Martin, C.L.; Bell, R.A.; Divers, J.; Dabelea, D.; Pettitt, D.J.; Saydah, S.; Pihoker, C.; Standiford, D.A.; et al. Peripheral Neuropathy in Adolescents and Young Adults with Type 1 and Type 2 Diabetes from the SEARCH for Diabetes in Youth Follow-up Cohort: A Pilot Study. Diabetes Care 2013, 36, 3903–3908. [Google Scholar] [CrossRef] [Green Version]
- Ismail-Beigi, F.; Craven, T.; Banerji, M.A.; Basile, J.; Calles, J.; Cohen, R.M.; Cuddihy, R.; Cushman, W.C.; Genuth, S.; Grimm, R.H.; et al. Effect of Intensive Treatment of Hyperglycaemia on Microvascular Outcomes in Type 2 Diabetes: An Analysis of the ACCORD Randomised Trial. Lancet 2010, 376, 419–430. [Google Scholar] [CrossRef] [Green Version]
- Diabetes Control and Complications Trial Research Group; Nathan, D.M.; Genuth, S.; Lachin, J.; Cleary, P.; Crofford, O.; Davis, M.; Rand, L.; Siebert, C. The Effect of Intensive Treatment of Diabetes on the Development and Progression of Long-Term Complications in Insulin-Dependent Diabetes Mellitus. N. Engl. J. Med. 1993, 329, 977–986. [Google Scholar] [CrossRef]
- Martin, C.L.; Albers, J.W.; Pop-Busui, R.; DCCT/EDIC Research Group. Neuropathy and Related Findings in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Study. Diabetes Care 2014, 37, 31–38. [Google Scholar] [CrossRef] [Green Version]
- Boulton, A.J.M.; Malik, R.A. Chapter 53-Diabetes Mellitus: Neuropathy. In Endocrinology: Adult and Pediatric, 7th ed.; Jameson, J.L., de Groot, L.J., de Kretser, D.M., Giudice, L.C., Grossman, A.B., Melmed, S., Potts, J.T., Jr., Weir, G.C., Eds.; Saunders: Philadelphia, PA, USA, 2016; pp. 920–933. [Google Scholar]
- Maser, R.E.; Steenkiste, A.R.; Dorman, J.S.; Nielsen, V.K.; Bass, E.B.; Manjoo, Q.; Drash, A.L.; Becker, D.J.; Kuller, L.H.; Greene, D.A.; et al. Epidemiological Correlates of Diabetic Neuropathy: Report from Pittsburgh Epidemiology of Diabetes Complications Study. Diabetes 1989, 38, 1456–1461. [Google Scholar] [CrossRef]
- Jarso, G.; Ahmed, A.; Feleke, Y. The Prevalence, Clinical Features and Management of Periphral Neuropathy among Diabetic Patients in Tikur Anbessa and St. Paul’s Specialized University Hospitals, Addis Ababa, Ethiopia. Ethiop. Med. J. 2011, 49, 299–311. [Google Scholar]
- Barbosa, M.; Saavedra, A.; Oliveira, S.; Reis, L.; Rodrigues, F.; Severo, M.; Sittl, R.; Maier, C.; Carvalho, D.M. Prevalence and Determinants of Painful and Painless Neuropathy in Type 1 Diabetes Mellitus. Front. Endocrinol. 2019, 10, 402. [Google Scholar] [CrossRef]
- Callaghan, B.C.; Xia, R.; Banerjee, M.; de Rekeneire, N.; Harris, T.B.; Newman, A.B.; Satterfield, S.; Schwartz, A.V.; Vinik, A.I.; Feldman, E.L.; et al. Metabolic Syndrome Components Are Associated with Symptomatic Polyneuropathy Independent of Glycemic Status. Diabetes Care 2016, 39, 801–807. [Google Scholar] [CrossRef] [Green Version]
- Callaghan, B.C.; Gao, L.; Li, Y.; Zhou, X.; Reynolds, E.; Banerjee, M.; Pop-Busui, R.; Feldman, E.L.; Ji, L. Diabetes and Obesity Are the Main Metabolic Drivers of Peripheral Neuropathy. Ann. Clin. Transl. Neurol. 2018, 5, 397–405. [Google Scholar] [CrossRef]
- World Health Organization. Classification of Diabetes Mellitus; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Sawacha, Z.; Gabriella, G.; Cristoferi, G.; Guiotto, A.; Avogaro, A.; Cobelli, C. Diabetic Gait and Posture Abnormalities: A Biomechanical Investigation through Three Dimensional Gait Analysis. Clin. Biomech. 2009, 24, 722–728. [Google Scholar] [CrossRef]
- Reeves, N.D.; Orlando, G.; Brown, S.J. Sensory-Motor Mechanisms Increasing Falls Risk in Diabetic Peripheral Neuropathy. Medicina 2021, 57, 457. [Google Scholar] [CrossRef] [PubMed]
- Fisher, L.; Hessler, D.M.; Polonsky, W.H.; Mullan, J. When Is Diabetes Distress Clinically Meaningful?: Establishing Cut Points for the Diabetes Distress Scale. Diabetes Care 2012, 35, 259–264. [Google Scholar] [CrossRef] [Green Version]
- Li, C.; Barker, L.; Ford, E.S.; Zhang, X.; Strine, T.W.; Mokdad, A.H. Diabetes and Anxiety in US Adults: Findings from the 2006 Behavioral Risk Factor Surveillance System. Diabet. Med. 2008, 25, 878–881. [Google Scholar] [CrossRef] [PubMed]
- Roy, T.; Lloyd, C.E. Epidemiology of Depression and Diabetes: A Systematic Review. J. Affect. Disord. 2012, 142, S8–S21. [Google Scholar] [CrossRef]
- Naranjo, C.; Del Reguero, L.; Moratalla, G.; Hercberg, M.; Valenzuela, M.; Failde, I. Anxiety, Depression and Sleep Disorders in Patients with Diabetic Neuropathic Pain: A Systematic Review. Expert Rev. Neurother. 2019, 19, 1201–1209. [Google Scholar] [CrossRef] [PubMed]
- de Groot, M.; Anderson, R.; Freedland, K.E.; Clouse, R.E.; Lustman, P.J. Association of Depression and Diabetes Complications: A Meta-Analysis. Psychosom. Med. 2001, 63, 619–630. [Google Scholar] [CrossRef] [PubMed]
- Ahola, A.J.; Thorn, L.M.; Saraheimo, M.; Forsblom, C.; Groop, P.-H. Depression Is Associated with the Metabolic Syndrome among Patients with Type 1 Diabetes. Ann. Med. 2010, 42, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Rubin, R.R.; Gaussoin, S.A.; Peyrot, M.; DiLillo, V.; Miller, K.; Wadden, T.A.; West, D.S.; Wing, R.R.; Knowler, W.C.; Look AHEAD Research Group. Cardiovascular Disease Risk Factors, Depression Symptoms and Antidepressant Medicine Use in the Look AHEAD (Action for Health in Diabetes) Clinical Trial of Weight Loss in Diabetes. Diabetologia 2010, 53, 1581–1589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herzer, M.; Hood, K.K. Anxiety Symptoms in Adolescents with Type 1 Diabetes: Association with Blood Glucose Monitoring and Glycemic Control. J. Pediatr. Psychol. 2010, 35, 415–425. [Google Scholar] [CrossRef]
- Gonzalez, J.S.; Vileikyte, L.; Ulbrecht, J.S.; Rubin, R.R.; Garrow, A.P.; Delgado, C.; Cavanagh, P.R.; Boulton, A.J.M.; Peyrot, M. Depression Predicts First but Not Recurrent Diabetic Foot Ulcers. Diabetologia 2010, 53, 2241–2248. [Google Scholar] [CrossRef] [Green Version]
- Singh, N.; Armstrong, D.G.; Lipsky, B.A. Preventing Foot Ulcers in Patients with Diabetes. JAMA 2005, 293, 217–228. [Google Scholar] [CrossRef]
- Margolis, D.J.; Allen-Taylor, L.; Hoffstad, O.; Berlin, J.A. Diabetic Neuropathic Foot Ulcers: The Association of Wound Size, Wound Duration, and Wound Grade on Healing. Diabetes Care 2002, 25, 1835–1839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prompers, L.; Huijberts, M.; Apelqvist, J.; Jude, E.; Piaggesi, A.; Bakker, K.; Edmonds, M.; Holstein, P.; Jirkovska, A.; Mauricio, D.; et al. High Prevalence of Ischaemia, Infection and Serious Comorbidity in Patients with Diabetic Foot Disease in Europe. Baseline Results from the Eurodiale Study. Diabetologia 2007, 50, 18–25. [Google Scholar] [CrossRef] [Green Version]
- Normahani, P.; Poushpas, S.; Alaa, M.; Bravis, V.; Sounderajah, V.; Aslam, M.; Jaffer, U. Diagnostic Accuracy of Point-of-Care Tests Used to Detect Arterial Disease in Diabetes: TEsting for Arterial Disease in Diabetes (TrEAD) Study. Ann. Surg. 2022. [Google Scholar] [CrossRef]
- Boulton, A.J.M. The Diabetic Foot: From Art to Science. The 18th Camillo Golgi Lecture. Diabetologia 2004, 47, 1343–1353. [Google Scholar] [CrossRef]
- Ragnarson Tennvall, G.; Apelqvist, J. Health-Related Quality of Life in Patients with Diabetes Mellitus and Foot Ulcers. J. Diabetes Complicat. 2000, 14, 235–241. [Google Scholar] [CrossRef]
- Jupiter, D.C.; Thorud, J.C.; Buckley, C.J.; Shibuya, N. The Impact of Foot Ulceration and Amputation on Mortality in Diabetic Patients. I: From Ulceration to Death, a Systematic Review. Int. Wound J. 2016, 13, 892–903. [Google Scholar] [CrossRef] [PubMed]
- Brownrigg, J.R.W.; Apelqvist, J.; Bakker, K.; Schaper, N.C.; Hinchliffe, R.J. Evidence-Based Management of PAD & the Diabetic Foot. Eur. J. Vasc. Endovasc. Surg. 2013, 45, 673–681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, B.M.; Holmes, C.M. Updates on Diabetic Foot and Charcot Osteopathic Arthropathy. Curr. Diabetes Rep. 2018, 18, 74. [Google Scholar] [CrossRef]
- Boulton, A.J.M.; Whitehouse, R.W. The Diabetic Foot. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dhatariya, K., Dungan, K., Grossman, A., Hershman, J.M., Hofland, J., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Boulton, A.J.M. Chapter 8—Diabetic Neuropathy and Foot Complications. In Handbook of Clinical Neurology; Zochodne, D.W., Malik, R.A., Eds.; Diabetes and the Nervous System; Elsevier: Amsterdam, The Netherlands, 2014; Volume 126, pp. 97–107. [Google Scholar]
- van Baal, J.; Hubbard, R.; Game, F.; Jeffcoate, W. Mortality Associated with Acute Charcot Foot and Neuropathic Foot Ulceration. Diabetes Care 2010, 33, 1086–1089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gil, J.; Schiff, A.P.; Pinzur, M.S. Cost Comparison: Limb Salvage Versus Amputation in Diabetic Patients with Charcot Foot. Foot Ankle Int. 2013, 34, 1097–1099. [Google Scholar] [CrossRef]
- Labovitz, J.M.; Shofler, D.W.; Ragothaman, K.K. The Impact of Comorbidities on Inpatient Charcot Neuroarthropathy Cost and Utilization. J. Diabetes Its Complicat. 2016, 30, 710–715. [Google Scholar] [CrossRef]
- Callaghan, B.C.; Little, A.A.; Feldman, E.L.; Hughes, R.A. Enhanced Glucose Control for Preventing and Treating Diabetic Neuropathy. Cochrane Database Syst. Rev. 2012, 6, CD007543. [Google Scholar] [CrossRef]
- American Diabetes Association. 13. Children and Adolescents: Standards of Medical Care in Diabetes—2021. Diabetes Care 2020, 44, S180–S199. [Google Scholar] [CrossRef]
- American Diabetes Association. 6. Glycemic Targets: Standards of Medical Care in Diabetes—2021. Diabetes Care 2020, 44, S73–S84. [Google Scholar] [CrossRef]
- American Diabetes Association. 14. Management of Diabetes in Pregnancy: Standards of Medical Care in Diabetes—2021. Diabetes Care 2020, 44, S200–S210. [Google Scholar] [CrossRef]
- American Diabetes Association. 12. Older Adults: Standards of Medical Care in Diabetes—2021. Diabetes Care 2020, 44, S168–S179. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Guideline. Type 1 Diabetes in Adults: Diagnosis and Management. 2021. Available online: https://www.nice.org.uk/guidance/ng17/resources/type-1-diabetes-in-adults-diagnosis-and-management-pdf-1837276469701 (accessed on 13 November 2021).
- National Institute for Health and Care Excellence (NICE). Guideline. Type 2 Diabetes in Adults Management. 2020. Available online: https://www.nice.org.uk/guidance/ng28/resources/type-2-diabetes-in-adults-management-pdf-1837338615493 (accessed on 13 November 2021).
- National Institute for Health and Care Excellence (NICE). Patient Decision Aid. Type 2 Diabetes: Agreeing My Blood Glucose (HbA1c) Target; Patient Decision Aid. 2022. Available online: https://www.nice.org.uk/guidance/ng28/resources/patient-decision-aid-pdf-2187281198 (accessed on 8 April 2022).
- Gibbons, C.H.; Freeman, R. Treatment-Induced Neuropathy of Diabetes: An Acute, Iatrogenic Complication of Diabetes. Brain J. Neurol. 2015, 138, 43–52. [Google Scholar] [CrossRef]
- Siddique, N.; Durcan, R.; Smyth, S.; Tun, T.K.; Sreenan, S.; McDermott, J.H. Acute Diabetic Neuropathy Following Improved Glycaemic Control: A Case Series and Review. Endocrinol. Diabetes Metab. Case Rep. 2020, 2020. [Google Scholar] [CrossRef] [Green Version]
- Streckmann, F.; Balke, M.; Cavaletti, G.; Toscanelli, A.; Bloch, W.; Décard, B.F.; Lehmann, H.C.; Faude, O. Exercise and Neuropathy: Systematic Review with Meta-Analysis. Sports Med. 2021, 52, 1043–1065. [Google Scholar] [CrossRef] [PubMed]
- Matos, M.; Mendes, R.; Silva, A.B.; Sousa, N. Physical Activity and Exercise on Diabetic Foot Related Outcomes: A Systematic Review. Diabetes Res. Clin. Pract. 2018, 139, 81–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, A.G.; Russell, J.; Feldman, E.L.; Goldstein, J.; Peltier, A.; Smith, S.; Hamwi, J.; Pollari, D.; Bixby, B.; Howard, J.; et al. Lifestyle Intervention for Pre-Diabetic Neuropathy. Diabetes Care 2006, 29, 1294–1299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balducci, S.; Iacobellis, G.; Parisi, L.; Di Biase, N.; Calandriello, E.; Leonetti, F.; Fallucca, F. Exercise Training Can Modify the Natural History of Diabetic Peripheral Neuropathy. J. Diabetes Complicat. 2006, 20, 216–223. [Google Scholar] [CrossRef]
- Ismail, K.; Winkley, K.; Rabe-Hesketh, S. Systematic Review and Meta-Analysis of Randomised Controlled Trials of Psychological Interventions to Improve Glycaemic Control in Patients with Type 2 Diabetes. Lancet 2004, 363, 1589–1597. [Google Scholar] [CrossRef]
- Cradock, K.A.; ÓLaighin, G.; Finucane, F.M.; Gainforth, H.L.; Quinlan, L.R.; Ginis, K.A.M. Behaviour Change Techniques Targeting Both Diet and Physical Activity in Type 2 Diabetes: A Systematic Review and Meta-Analysis. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 18. [Google Scholar] [CrossRef] [Green Version]
- Buggy, A.; Moore, Z. The Impact of the Multidisciplinary Team in the Management of Individuals with Diabetic Foot Ulcers: A Systematic Review. J. Wound Care 2017, 26, 324–339. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Attinger, C.; Smeets, L.; Koller, A.; Bal, A.; Kavarthapu, V. Surgical Strategies for Prevention of Amputation of the Diabetic Foot. J. Clin. Orthop. Trauma 2021, 17, 99–105. [Google Scholar] [CrossRef]
- NHS Digital. Presentation. National Diabetes Foot Care Audit Fourth Annual Report. 2019. Available online: https://files.digital.nhs.uk/FC/18ED7F/NDFA%204AR%20-%20Main%20Report%20v1.1.pdf (accessed on 2 February 2022).
- Dorresteijn, J.A.; Kriegsman, D.M.; Assendelft, W.J.; Valk, G.D. Patient Education for Preventing Diabetic Foot Ulceration. Cochrane Database Syst. Rev. 2012, 10, CD001488. [Google Scholar] [CrossRef] [Green Version]
- Handelsman, Y.; Bloomgarden, Z.T.; Grunberger, G.; Umpierrez, G.; Zimmerman, R.S.; Bailey, T.S.; Blonde, L.; Bray, G.A.; Cohen, A.J.; Dagogo-Jack, S.; et al. American Association of Clinical Endocrinologists and American College of Endocrinology—Clinical Practice Guidelines for Developing A Diabetes Mellitus Comprehensive Care Plan—2015—Executive Summary. Endocr. Pract. 2015, 21, 413–437. [Google Scholar] [CrossRef] [Green Version]
- Tesfaye, S.; Vileikyte, L.; Rayman, G.; Sindrup, S.H.; Perkins, B.A.; Baconja, M.; Vinik, A.I.; Boulton, A.J.M.; Toronto Expert Panel on Diabetic Neuropathy. Painful Diabetic Peripheral Neuropathy: Consensus Recommendations on Diagnosis, Assessment and Management. Diabetes Metab. Res. Rev. 2011, 27, 629–638. [Google Scholar] [CrossRef]
- Turner, R.; Holman, R.R.; Cull, C.A.; Stratton, I.M.; Matthews; Frighi, V.; Manley, S.E.; Neil, A.; McElroy, K.; Wright, D.H.; et al. Intensive Blood-Glucose Control with Sulphonylureas or Insulin Compared with Conventional Treatment and Risk of Complications in Patients with Type 2 Diabetes (UKPDS 33). Lancet 1998, 352, 837–853. [Google Scholar] [CrossRef]
- Leow, M.K.S.; Wyckoff, J. Under-Recognised Paradox of Neuropathy from Rapid Glycaemic Control. Postgrad. Med. J. 2005, 81, 103–107. [Google Scholar] [CrossRef]
- American Diabetes Association. Implications of the United Kingdom Prospective Diabetes Study. Diabetes Care 2002, 25, s28–s32. [Google Scholar] [CrossRef] [Green Version]
- Ang, L.; Jaiswal, M.; Martin, C.; Pop-Busui, R. Glucose Control and Diabetic Neuropathy: Lessons from Recent Large Clinical Trials. Curr. Diab. Rep. 2014, 14, 528. [Google Scholar] [CrossRef]
- Gæde, P.; Vedel, P.; Larsen, N.; Jensen, G.V.H.; Parving, H.-H.; Pedersen, O. Multifactorial Intervention and Cardiovascular Disease in Patients with Type 2 Diabetes. N. Engl. J. Med. 2003, 348, 383–393. [Google Scholar] [CrossRef] [Green Version]
- Maiorino, M.I.; Signoriello, S.; Maio, A.; Chiodini, P.; Bellastella, G.; Scappaticcio, L.; Longo, M.; Giugliano, D.; Esposito, K. Effects of Continuous Glucose Monitoring on Metrics of Glycemic Control in Diabetes: A Systematic Review with Meta-Analysis of Randomized Controlled Trials. Diabetes Care 2020, 43, 1146–1156. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Guideline. Neuropathic Pain in Adults: Pharmacological Management in Non-Specialist Settings. 2020. Available online: https://www.nice.org.uk/guidance/cg173/resources/neuropathic-pain-in-adults-pharmacological-management-in-nonspecialist-settings-pdf-35109750554053 (accessed on 8 November 2021).
- Attal, N.; Cruccu, G.; Baron, R.; Haanpää, M.; Hansson, P.; Jensen, T.S.; Nurmikko, T. EFNS Guidelines on the Pharmacological Treatment of Neuropathic Pain: 2010 Revision. Eur. J. Neurol. 2010, 17, 1113-e88. [Google Scholar] [CrossRef]
- Bril, V.; England, J.; Franklin, G.M.; Backonja, M.; Cohen, J.; Toro, D.D.; Feldman, E.; Iverson, D.J.; Perkins, B.; Russell, J.W.; et al. Evidence-Based Guideline: Treatment of Painful Diabetic Neuropathy: Report of the American Academy of Neurology, the American Association of Neuromuscular and Electrodiagnostic Medicine, and the American Academy of Physical Medicine and Rehabilitation. Neurology 2011, 76, 1758–1765. [Google Scholar] [CrossRef] [Green Version]
- Gabriel, R.; Abdelkader, N.B.; Acosta, T.; Gilis-Januszewska, A.; Gómez-Huelgas, R.; Makrilakis, K.; Kamenov, Z.; Paulweber, B.; Satman, I.; Djordjevic, P.; et al. Early Prevention of Diabetes Microvascular Complications in People with Hyperglycaemia in Europe. EPREDICE Randomized Trial. Study Protocol, Recruitment and Selected Baseline Data. PLoS ONE 2020, 15, e0231196. [Google Scholar] [CrossRef] [Green Version]
- National Institute for Health and Care Excellence (NICE). Guideline. Type 2 Diabetes: Prevention in People at High Risk. 2017. Available online: https://www.nice.org.uk/guidance/ph38/resources/type-2-diabetes-prevention-in-people-at-high-risk-pdf-1996304192197 (accessed on 13 July 2022).
- American Diabetes Association. Standards of Medical Care in Diabetes—2022 Abridged for Primary Care Providers. Clin. Diabetes 2022, 40, 10–38. [Google Scholar] [CrossRef] [PubMed]
- Rudnicki, S.A. Prevention and Treatment of Peripheral Neuropathy after Bariatric Surgery. Curr. Treat. Options Neurol. 2010, 12, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Adam, S.; Azmi, S.; Ho, J.H.; Liu, Y.; Ferdousi, M.; Siahmansur, T.; Kalteniece, A.; Marshall, A.; Dhage, S.S.; Iqbal, Z.; et al. Improvements in Diabetic Neuropathy and Nephropathy After Bariatric Surgery: A Prospective Cohort Study. Obes. Surg. 2021, 31, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Aghili, R.; Malek, M.; Tanha, K.; Mottaghi, A. The Effect of Bariatric Surgery on Peripheral Polyneuropathy: A Systematic Review and Meta-Analysis. Obes. Surg. 2019, 29, 3010–3020. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Diabetic Foot Problems: Prevention and Management. 2019. Available online: https://www.nice.org.uk/guidance/ng19/resources/diabetic-foot-problems-prevention-and-management-pdf-1837279828933 (accessed on 13 July 2022).
- Diabetes UK. Putting Feet First: National Minimum Skills Framework. 2011. Available online: https://diabetes-resources-production.s3-eu-west-1.amazonaws.com/diabetes-storage/migration/pdf/NMSF_16Feb2011.pdf (accessed on 25 November 2021).
- Burgess, J.; Frank, B.; Marshall, A.; Khalil, R.S.; Ponirakis, G.; Petropoulos, I.N.; Cuthbertson, D.J.; Malik, R.A.; Alam, U. Early Detection of Diabetic Peripheral Neuropathy: A Focus on Small Nerve Fibres. Diagnostics 2021, 11, 165. [Google Scholar] [CrossRef]
- Selvarajah, D.; Kar, D.; Khunti, K.; Davies, M.J.; Scott, A.R.; Walker, J.; Tesfaye, S. Diabetic Peripheral Neuropathy: Advances in Diagnosis and Strategies for Screening and Early Intervention. Lancet Diabetes Endocrinol. 2019, 7, 938–948. [Google Scholar] [CrossRef] [Green Version]
- Carmichael, J.; Fadavi, H.; Ishibashi, F.; Shore, A.C.; Tavakoli, M. Advances in Screening, Early Diagnosis and Accurate Staging of Diabetic Neuropathy. Front. Endocrinol. 2021, 12, 671257. [Google Scholar] [CrossRef]
- Feldman, E.L.; Stevens, M.J.; Thomas, P.K.; Brown, M.B.; Canal, N.; Greene, D.A. A Practical Two-Step Quantitative Clinical and Electrophysiological Assessment for the Diagnosis and Staging of Dianetic Neuropathy. Diabetes Care 1994, 17, 9. [Google Scholar] [CrossRef]
- Herman, W.H.; Pop-Busui, R.; Braffett, B.H.; Martin, C.L.; Cleary, P.A.; Albers, J.W.; Feldman, E.L. Use of the Michigan Neuropathy Screening Instrument as a Measure of Distal Symmetrical Peripheral Neuropathy in Type 1 Diabetes: Results from the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications. Diabet. Med. 2012, 29, 937–944. [Google Scholar] [CrossRef] [Green Version]
- Mayeda, L.; Katz, R.; Ahmad, I.; Bansal, N.; Batacchi, Z.; Hirsch, I.B.; Robinson, N.; Trence, D.L.; Zelnick, L.; de Boer, I.H. Glucose Time in Range and Peripheral Neuropathy in Type 2 Diabetes Mellitus and Chronic Kidney Disease. BMJ Open Diabetes Res. Care 2020, 8, e000991. [Google Scholar] [CrossRef] [Green Version]
- Bril, V.; Perkins, B.A. Validation of the Toronto Clinical Scoring System for Diabetic Polyneuropathy. Diabetes Care 2002, 25, 2048–2052. [Google Scholar] [CrossRef] [Green Version]
- Dyck, P.J.; Davies, J.L.; Litchy, W.J.; O’Brien, P.C. Longitudinal Assessment of Diabetic Polyneuropathy Using a Composite Score in the Rochester Diabetic Neuropathy Study Cohort. Neurology 1997, 49, 229–239. [Google Scholar] [CrossRef]
- Bril, V. NIS-LL: The Primary Measurement Scale for Clinical Trial Endpoints in Diabetic Peripheral Neuropathy. Eur. Neurol. 1999, 41, 8–13. [Google Scholar] [CrossRef]
- Dyck, P.J.B.; González-Duarte, A.; Obici, L.; Polydefkis, M.; Wiesman, J.F.; Antonino, I.; Litchy, W.J.; Dyck, P.J. Development of Measures of Polyneuropathy Impairment in HATTR Amyloidosis: From NIS to MNIS + 7. J. Neurol. Sci. 2019, 405, 116424. [Google Scholar] [CrossRef] [Green Version]
- Zilliox, L.A.; Ruby, S.K.; Singh, S.; Zhan, M.; Russell, J.W. Clinical Neuropathy Scales in Neuropathy Associated with Impaired Glucose Tolerance. J. Diabetes Complicat. 2015, 29, 372–377. [Google Scholar] [CrossRef] [Green Version]
- Dyck, P.J.; Karnes, J.L.; Daube, J.; O’brien, P.; Service, F.J. Clinical and neuropathological criteria for the diagnosis and staging of diabetic polyneuropathy. Brain 1985, 108, 861–880. [Google Scholar] [CrossRef]
- Chaudhry, V.; Cornblath, D.R.; Mellits, E.D.; Avila, O.; Freimer, M.L.; Glass, J.D.; Reim, J.; Ronnett, G.V.; Quaskey, S.A.; Kuncl, R.W. Inter- and Intra-Examiner Reliability of Nerve Conduction Measurements in Normal Subjects. Ann. Neurol. 1991, 30, 841–843. [Google Scholar] [CrossRef]
- Himeno, T.; Kamiya, H.; Nakamura, J. Lumos for the Long Trail: Strategies for Clinical Diagnosis and Severity Staging for Diabetic Polyneuropathy and Future Directions. J. Diabetes Investig. 2020, 11, 5–16. [Google Scholar] [CrossRef] [Green Version]
- Rolke, R.; Magerl, W.; Campbell, K.A.; Schalber, C.; Caspari, S.; Birklein, F.; Treede, R.-D. Quantitative Sensory Testing: A Comprehensive Protocol for Clinical Trials. Eur. J. Pain Lond. Engl. 2006, 10, 77–88. [Google Scholar] [CrossRef]
- Petropoulos, I.N.; Ponirakis, G.; Ferdousi, M.; Azmi, S.; Kalteniece, A.; Khan, A.; Gad, H.; Bashir, B.; Marshall, A.; Boulton, A.J.M.; et al. Corneal Confocal Microscopy: A Biomarker for Diabetic Peripheral Neuropathy. Clin. Ther. 2021, 43, 1457–1475. [Google Scholar] [CrossRef]
- Jiang, M.-S.; Yuan, Y.; Gu, Z.-X.; Zhuang, S.-L. Corneal Confocal Microscopy for Assessment of Diabetic Peripheral Neuropathy: A Meta-Analysis. Br. J. Ophthalmol. 2016, 100, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Kalteniece, A.; Ferdousi, M.; Azmi, S.; Khan, S.U.; Worthington, A.; Marshall, A.; Faber, C.G.; Lauria, G.; Boulton, A.J.M.; Soran, H.; et al. Corneal Nerve Loss Is Related to the Severity of Painful Diabetic Neuropathy. Eur. J. Neurol. 2022, 29, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Derry, S.; Bell, R.; Straube, S.; Wiffen, P.J.; Aldington, D.; Moore, R. Pregabalin for Chronic Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2019, 1, CD007076. [Google Scholar] [CrossRef] [PubMed]
- Wiffen, P.J.; Derry, S.; Bell, R.F.; Rice, A.S.; Tölle, T.R.; Phillips, T.; Moore, R.A. Gabapentin for Chronic Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2017, 6, CD007938. [Google Scholar] [CrossRef] [Green Version]
- Vinik, A.; Casellini, C.; Nevoret, M.-L. Table 7, Drugs Approved by the FDA for Treatment of Neuropathic Pain Syndromes. Available online: https://www.ncbi.nlm.nih.gov/books/NBK279175/table/diab-neuropathies.medication/ (accessed on 29 November 2021).
- National Institute for Health and Care Excellence (NICE). British National Formulary. Pregabalin. Available online: https://bnf.nice.org.uk/drug/pregabalin.html (accessed on 11 April 2022).
- Medicines and Healthcare products Regulatory Agency (MHRA). Pregabalin (Lyrica), Gabapentin (Neurontin) and Risk of Abuse and Dependence: New Scheduling Requirements from 1 April. 2019. Available online: https://www.gov.uk/drug-safety-update/pregabalin-lyrica-gabapentin-neurontin-and-risk-of-abuse-and-dependence-new-scheduling-requirements-from-1-april (accessed on 11 April 2022).
- National Institute for Health and Care Excellence (NICE). British National Formulary. Gabapentin. Available online: https://bnf.nice.org.uk/drug/gabapentin.html#indicationsAndDoses (accessed on 11 April 2022).
- Medicines and Healthcare products Regulatory Agency (MHRA). Pregabalin (Lyrica): Reports of Severe Respiratory Depression. 2021. Available online: https://www.gov.uk/drug-safety-update/pregabalin-lyrica-reports-of-severe-respiratory-depression (accessed on 11 April 2022).
- Lunn, M.P.; Hughes, R.A.; Wiffen, P.J. Duloxetine for Treating Painful Neuropathy, Chronic Pain or Fibromyalgia. Cochrane Database Syst. Rev. 2014, 1, CD007115. [Google Scholar] [CrossRef]
- Gao, Y.; Ning, G.; Jia, W.; Zhou, Z.; Xu, Z.; Liu, Z.; Liu, C.; Ma, J.; Li, Q.; Cheng, L.; et al. Duloxetine versus Placebo in the Treatment of Patients with Diabetic Neuropathic Pain in China. Chin. Med. J. 2010, 123, 3184–3192. [Google Scholar] [CrossRef]
- Boyle, J.; Eriksson, M.E.V.; Gribble, L.; Gouni, R.; Johnsen, S.; Coppini, D.V.; Kerr, D. Randomized, Placebo-Controlled Comparison of Amitriptyline, Duloxetine, and Pregabalin in Patients with Chronic Diabetic Peripheral Neuropathic Pain: Impact on Pain, Polysomnographic Sleep, Daytime Functioning, and Quality of Life. Diabetes Care 2012, 35, 2451–2458. [Google Scholar] [CrossRef] [Green Version]
- Kaur, H.; Hota, D.; Bhansali, A.; Dutta, P.; Bansal, D.; Chakrabarti, A. A Comparative Evaluation of Amitriptyline and Duloxetine in Painful Diabetic Neuropathy: A Randomized, Double-Blind, Cross-over Clinical Trial. Diabetes Care 2011, 34, 818–822. [Google Scholar] [CrossRef] [Green Version]
- Khasbage, S.; Shukla, R.; Sharma, P.; Singh, S. A Randomized Control Trial of Duloxetine and Gabapentin in Painful Diabetic Neuropathy. J. Diabetes 2021, 13, 532–541. [Google Scholar] [CrossRef]
- Majdinasab, N.; Kaveyani, H.; Azizi, M. A Comparative Double-Blind Randomized Study on the Effectiveness of Duloxetine and Gabapentin on Painful Diabetic Peripheral Polyneuropathy. Drug Des. Devel. Ther. 2019, 13, 1985–1992. [Google Scholar] [CrossRef] [Green Version]
- Tesfaye, S.; Wilhelm, S.; Lledo, A.; Schacht, A.; Tölle, T.; Bouhassira, D.; Cruccu, G.; Skljarevski, V.; Freynhagen, R. Duloxetine and Pregabalin: High-Dose Monotherapy or Their Combination? The “COMBO-DN Study”—A Multinational, Randomized, Double-Blind, Parallel-Group Study in Patients with Diabetic Peripheral Neuropathic Pain. PAIN 2013, 154, 2616–2625. [Google Scholar] [CrossRef] [Green Version]
- Tanenberg, R.J.; Clemow, D.B.; Giaconia, J.M.; Risser, R.C. Duloxetine Compared with Pregabalin for Diabetic Peripheral Neuropathic Pain Management in Patients with Suboptimal Pain Response to Gabapentin and Treated with or without Antidepressants: A Post Hoc Analysis. Pain Pract. Off. J. World Inst. Pain 2014, 14, 640–648. [Google Scholar] [CrossRef]
- Gallagher, H.C.; Gallagher, R.M.; Butler, M.; Buggy, D.J.; Henman, M.C. Venlafaxine for Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2015, 8, CD011091. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). British National Formulary. Duloxetine. Available online: https://bnf.nice.org.uk/drug/duloxetine.html (accessed on 11 April 2022).
- Moore, R.A.; Derry, S.; Aldington, D.; Cole, P.; Wiffen, P.J. Amitriptyline for Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2015, 7, CD008242. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). British National Formulary. Amitriptyline. Available online: https://bnf.nice.org.uk/drug/amitriptyline-hydrochloride.html (accessed on 11 April 2022).
- Duehmke, R.M.; Derry, S.; Wiffen, P.J.; Bell, R.F.; Aldington, D.; Moore, R.A. Tramadol for Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2017, 6, CD003726. [Google Scholar] [CrossRef]
- Freo, U.; Romualdi, P.; Kress, H.G. Tapentadol for Neuropathic Pain: A Review of Clinical Studies. J. Pain Res. 2019, 12, 1537–1551. [Google Scholar] [CrossRef] [Green Version]
- Harati, Y.; Gooch, C.; Swenson, M.; Edelman, S.; Greene, D.; Raskin, P.; Donofrio, P.; Cornblath, D.; Sachdeo, R.; Siu, C.O.; et al. Double-blind Randomized Trial of Tramadol for the Treatment of the Pain of Diabetic Neuropathy. Neurology 1998, 50, 1842–1846. [Google Scholar] [CrossRef]
- Freeman, R.; Raskin, P.; Hewitt, D.J.; Vorsanger, G.J.; Jordan, D.M.; Xiang, J.; Rosenthal, N.R.; CAPSS-237 Study Group. Randomized Study of Tramadol/Acetaminophen versus Placebo in Painful Diabetic Peripheral Neuropathy. Curr. Med. Res. Opin. 2007, 23, 147–161. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). British National Formulary. Tramadol Hydrochloride. Available online: https://bnf.nice.org.uk/drug/tramadol-hydrochloride.html (accessed on 12 April 2022).
- Dunn, K.E.; Bergeria, C.L.; Huhn, A.S.; Strain, E.C. A Systematic Review of Laboratory Evidence for the Abuse Potential of Tramadol in Humans. Front. Psychiatry 2019, 10, 704. [Google Scholar] [CrossRef]
- Staudt, M.D.; Prabhala, T.; Sheldon, B.L.; Quaranta, N.; Zakher, M.; Bhullar, R.; Pilitsis, J.G.; Argoff, C.E. Current Strategies for the Management of Painful Diabetic Neuropathy. J. Diabetes Sci. Technol. 2020, 16, 341–352. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). British National Formulary. Tapentadol. Available online: https://bnf.nice.org.uk/drug/tapentadol.html#indicationsAndDoses (accessed on 12 April 2022).
- Van Nooten, F.; Treur, M.; Pantiri, K.; Stoker, M.; Charokopou, M. Capsaicin 8% Patch Versus Oral Neuropathic Pain Medications for the Treatment of Painful Diabetic Peripheral Neuropathy: A Systematic Literature Review and Network Meta-Analysis. Clin. Ther. 2017, 39, 787–803.e18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freynhagen, R.; Argoff, C.; Eerdekens, M.; Engelen, S.; Perrot, S. Progressive Response to Repeat Application of Capsaicin 179 Mg (8% w/w) Cutaneous Patch in Peripheral Neuropathic Pain: Comprehensive New Analysis and Clinical Implications. Pain Med. Malden Mass 2021, 22, 2324–2336. [Google Scholar] [CrossRef] [PubMed]
- Mason, L.; Moore, R.A.; Derry, S.; Edwards, J.E.; McQuay, H.J. Systematic Review of Topical Capsaicin for the Treatment of Chronic Pain. BMJ 2004, 328, 991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kulkantrakorn, K.; Chomjit, A.; Sithinamsuwan, P.; Tharavanij, T.; Suwankanoknark, J.; Napunnaphat, P. 0.075% Capsaicin Lotion for the Treatment of Painful Diabetic Neuropathy: A Randomized, Double-Blind, Crossover, Placebo-Controlled Trial. J. Clin. Neurosci. 2019, 62, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Polydefkis, M.; Hauer, P.; Sheth, S.; Sirdofsky, M.; Griffin, J.W.; McArthur, J.C. The Time Course of Epidermal Nerve Fibre Regeneration: Studies in Normal Controls and in People with Diabetes, with and without Neuropathy. Brain 2004, 127, 1606–1615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, B.; Zhou, X.; Zhou, Q.; Wang, H.; Wang, S.; Luo, K. Intra-Venous Lidocaine to Relieve Neuropathic Pain: A Systematic Review and Meta-Analysis. Front. Neurol. 2019, 10, 954. [Google Scholar] [CrossRef] [PubMed]
- Moulin, D.E.; Morley-Forster, P.K.; Pirani, Z.; Rohfritsch, C.; Stitt, L. Intravenous Lidocaine in the Management of Chronic Peripheral Neuropathic Pain: A Randomized-Controlled Trial. Can. J. Anesth. Can. Anesth. 2019, 66, 820–827. [Google Scholar] [CrossRef]
- Aiyer, R.; Mehta, N.; Gungor, S.; Gulati, A. A Systematic Review of NMDA Receptor Antagonists for Treatment of Neuropathic Pain in Clinical Practice. Clin. J. Pain 2018, 34, 450–467. [Google Scholar] [CrossRef]
- Wiffen, P.J.; Derry, S.; Moore, R.A.; Kalso, E.A. Carbamazepine for Chronic Neuropathic Pain and Fibromyalgia in Adults. Cochrane Database Syst. Rev. 2014, 4, CD005451. [Google Scholar] [CrossRef]
- Gill, D.; Derry, S.; Wiffen, P.J.; Moore, R.A. Valproic Acid and Sodium Valproate for Neuropathic Pain and Fibromyalgia in Adults. Cochrane Database Syst. Rev. 2011, 10, CD009183. [Google Scholar] [CrossRef]
- Wiffen, P.J.; Derry, S.; Moore, R.A. Lamotrigine for Chronic Neuropathic Pain and Fibromyalgia in Adults. Cochrane Database Syst. Rev. 2013, 2013, CD006044. [Google Scholar] [CrossRef]
- Birse, F.; Derry, S.; Moore, R.A. Phenytoin for Neuropathic Pain and Fibromyalgia in Adults. Cochrane Database Syst. Rev. 2012, 5, CD009485. [Google Scholar] [CrossRef]
- Gao, Y.; Guo, X.; Han, P.; Li, Q.; Yang, G.; Qu, S.; Yue, L.; Wang, C.-N.; Skljarevski, V.; Dueñas, H.; et al. Treatment of Patients with Diabetic Peripheral Neuropathic Pain in China: A Double-Blind Randomised Trial of Duloxetine vs. Placebo. Int. J. Clin. Pract. 2015, 69, 957–966. [Google Scholar] [CrossRef] [Green Version]
- Alam, U.; Sloan, G.; Tesfaye, S. Treating Pain in Diabetic Neuropathy: Current and Developmental Drugs. Drugs 2020, 80, 363–384. [Google Scholar] [CrossRef]
- Farshchian, N.; Alavi, A.; Heydarheydari, S.; Moradian, N. Comparative Study of the Effects of Venlafaxine and Duloxetine on Chemotherapy-Induced Peripheral Neuropathy. Cancer Chemother. Pharmacol. 2018, 82, 787–793. [Google Scholar] [CrossRef]
- Vrethem, M.; Boivie, J.; Arnqvist, H.; Holmgren, H.; Lindström, T.; Thorell, L.-H. A Comparison of Amitriptyline and Maprotiline in the Treatment of Painful Polyneuropathy in Diabetics and Nondiabetics. Clin. J. Pain 1997, 13, 313–323. [Google Scholar] [CrossRef]
- Morello, C.M.; Leckband, S.G.; Stoner, C.P.; Moorhouse, D.F.; Sahagian, G.A. Randomized Double-Blind Study Comparing the Efficacy of Gabapentin with Amitriptyline on Diabetic Peripheral Neuropathy Pain. Arch. Intern. Med. 1999, 159, 1931–1937. [Google Scholar] [CrossRef]
- Biesbroeck, R.; Bril, V.; Hollander, P.; Kabadi, U.; Schwartz, S.; Singh, S.P.; Ward, W.K.; Bernstein, J.E. A Double-Blind Comparison of Topical Capsaicin and Oral Amitriptyline in Painful Diabetic Neuropathy. Adv. Ther. 1995, 12, 111–120. [Google Scholar]
- Max, M.B.; Culnane, M.; Schafer, S.C.; Gracely, R.H.; Walther, D.J.; Smoller, B.; Dubner, R. Amitriptyline Relieves Diabetic Neuropathy Pain in Patients with Normal or Depressed Mood. Neurology 1987, 37, 589–596. [Google Scholar] [CrossRef]
- Max, M.B.; Lynch, S.A.; Muir, J.; Shoaf, S.E.; Smoller, B.; Dubner, R. Effects of Desipramine, Amitriptyline, and Fluoxetine on Pain in Diabetic Neuropathy. N. Engl. J. Med. 1992, 326, 1250–1256. [Google Scholar] [CrossRef]
- Electronic Medicines Compendium. Amitriptyline 10 Mg Film-Coated Tablets—Summary of Product Characteristics (SmPC)-(Emc). Available online: https://www.medicines.org.uk/emc/product/10849/smpc (accessed on 26 November 2021).
- Sindrup, S.H.; Gram, L.F.; Skjold, T.; Grodum, E.; Brøsen, K.; Beck-Nielsen, H. Clomipramine vs. Desipramine vs. Placebo in the Treatment of Diabetic Neuropathy Symptoms. A Double-Blind Cross-over Study. Br. J. Clin. Pharmacol. 1990, 30, 683–691. [Google Scholar] [CrossRef] [Green Version]
- Max, M.B.; Kishore-Kumar, R.; Schafer, S.C.; Meister, B.; Gracely, R.H.; Smoller, B.; Dubner, R. Efficacy of Desipramine in Painful Diabetic Neuropathy: A Placebo-Controlled Trial. Pain 1991, 45, 3–9. [Google Scholar] [CrossRef]
- Gómez-Pérez, F.J.; Choza, R.; Ríos, J.M.; Reza, A.; Huerta, E.; Aguilar, C.A.; Rull, J.A. Nortriptyline-Fluphenazine vs. Carbamazepine in the Symptomatic Treatment of Diabetic Neuropathy. Arch. Med. Res. 1996, 27, 525–529. [Google Scholar]
- Derry, S.; Wiffen, P.J.; Aldington, D.; Moore, R.A. Nortriptyline for Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2015, 1, CD011209. [Google Scholar] [CrossRef]
- Hearn, L.; Moore, R.A.; Derry, S.; Wiffen, P.J.; Phillips, T. Desipramine for Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2014, 9, CD011003. [Google Scholar] [CrossRef]
- Barohn, R.J.; Gajewski, B.; Pasnoor, M.; Brown, A.; Herbelin, L.L.; Kimminau, K.S.; Mudaranthakam, D.P.; Jawdat, O.; Dimachkie, M.M. Patient Assisted Intervention for Neuropathy: Comparison of Treatment in Real Life Situations (PAIN-CONTRoLS) Study Team Patient Assisted Intervention for Neuropathy: Comparison of Treatment in Real Life Situations (PAIN-CONTRoLS): Bayesian Adaptive Comparative Effectiveness Randomized Trial. JAMA Neurol. 2021, 78, 68–76. [Google Scholar] [CrossRef]
- Electronic Medicines Compendium. Zydol 50 Mg Capsules—Summary of Product Characteristics (SmPC)-(Emc). Available online: https://www.medicines.org.uk/emc/product/81/smpc (accessed on 29 November 2021).
- Sindrup, S.; Konder, R.; Lehmann, R.; Meier, T.; Winkel, M.; Ashworth, J.; Baron, R.; Jensen, T. Randomized Controlled Trial of the Combined Monoaminergic and Opioid Investigational Compound GRT9906 in Painful Polyneuropathy. Eur. J. Pain 2012, 16, 849–859. [Google Scholar] [CrossRef]
- Vinik, A.I.; Shapiro, D.Y.; Rauschkolb, C.; Lange, B.; Karcher, K.; Pennett, D.; Etropolski, M.S. A Randomized Withdrawal, Placebo-Controlled Study Evaluating the Efficacy and Tolerability of Tapentadol Extended Release in Patients with Chronic Painful Diabetic Peripheral Neuropathy. Diabetes Care 2014, 37, 2302–2309. [Google Scholar] [CrossRef] [Green Version]
- Niesters, M.; Proto, P.L.; Aarts, L.; Sarton, E.Y.; Drewes, A.M.; Dahan, A. Tapentadol Potentiates Descending Pain Inhibition in Chronic Pain Patients with Diabetic Polyneuropathy. BJA Br. J. Anaesth. 2014, 113, 148–156. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, S.; Etropolski, M.S.; Shapiro, D.Y.; Rauschkolb, C.; Vinik, A.I.; Lange, B.; Cooper, K.; Van Hove, I.; Haeussler, J. A Pooled Analysis Evaluating the Efficacy and Tolerability of Tapentadol Extended Release for Chronic, Painful Diabetic Peripheral Neuropathy. Clin. Drug Investig. 2015, 35, 95–108. [Google Scholar] [CrossRef] [Green Version]
- Gaskell, H.; Derry, S.; Stannard, C.; Moore, R.A. Oxycodone for Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2016, 7, CD010692. [Google Scholar] [CrossRef] [PubMed]
- McNicol, E.D.; Ferguson, M.C.; Schumann, R. Methadone for Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2017, 5, CD012499. [Google Scholar] [CrossRef] [PubMed]
- McNicol, E.D.; Midbari, A.; Eisenberg, E. Opioids for Neuropathic Pain. Cochrane Database Syst. Rev. 2013, 8, CD006146. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Bley, K. Topical Capsaicin for Pain Management: Therapeutic Potential and Mechanisms of Action of the New High-Concentration Capsaicin 8% Patch. Br. J. Anaesth. 2011, 107, 490–502. [Google Scholar] [CrossRef] [Green Version]
- Kulkantrakorn, K.; Lorsuwansiri, C.; Meesawatsom, P. 0.025% Capsaicin Gel for the Treatment of Painful Diabetic Neuropathy: A Randomized, Double-Blind, Crossover, Placebo-Controlled Trial. Pain Pract. Off. J. World Inst. Pain 2013, 13, 497–503. [Google Scholar] [CrossRef]
- Campbell, C.M.; Kipnes, M.S.; Stouch, B.C.; Brady, K.L.; Kelly, M.; Schmidt, W.K.; Petersen, K.L.; Rowbotham, M.C.; Campbell, J.N. Randomized Control Trial of Topical Clonidine for Treatment of Painful Diabetic Neuropathy. Pain 2012, 153, 1815–1823. [Google Scholar] [CrossRef] [Green Version]
- Derry, S.; Wiffen, P.J.; Moore, R.A.; Quinlan, J. Topical Lidocaine for Neuropathic Pain in Adults. Cochrane Database Syst. Rev. 2014, 2, CD010958. [Google Scholar] [CrossRef]
- Mahoney, J.M.; Vardaxis, V.; Moore, J.L.; Hall, A.M.; Haffner, K.E.; Peterson, M.C. Topical Ketamine Cream in the Treatment of Painful Diabetic Neuropathy: A Randomized, Placebo-Controlled, Double-Blind Initial Study. J. Am. Podiatr. Med. Assoc. 2012, 102, 178–183. [Google Scholar] [CrossRef]
- Lynch, M.E.; Clark, A.J.; Sawynok, J.; Sullivan, M.J.L. Topical 2% Amitriptyline and 1% Ketamine in Neuropathic Pain Syndromes: A Randomized, Double-Blind, Placebo-Controlled Trial. Anesthesiology 2005, 103, 140–146. [Google Scholar] [CrossRef]
- Yilmaz, Z.; Piracha, F.; Anderson, L.; Mazzola, N. Supplements for Diabetes Mellitus: A Review of the Literature. J. Pharm. Pract. 2017, 30, 631–638. [Google Scholar] [CrossRef]
- Le, Y.; Wang, B.; Xue, M. Nutraceuticals Use and Type 2 Diabetes Mellitus. Curr. Opin. Pharmacol. 2022, 62, 168–176. [Google Scholar] [CrossRef]
- Han, T.; Bai, J.; Liu, W.; Hu, Y. THERAPY OF ENDOCRINE DISEASE: A Systematic Review and Meta-Analysis of α-Lipoic Acid in the Treatment of Diabetic Peripheral Neuropathy. Eur. J. Endocrinol. 2012, 167, 465–471. [Google Scholar] [CrossRef] [Green Version]
- Rolim, L.C.; da Silva, E.M.; Flumignan, R.L.; Abreu, M.M.; Dib, S.A. Acetyl-L-Carnitine for the Treatment of Diabetic Peripheral Neuropathy. Cochrane Database Syst. Rev. 2019, 6, CD011265. [Google Scholar] [CrossRef]
- O’Connell, N.E.; Ferraro, M.C.; Gibson, W.; Rice, A.S.; Vase, L.; Coyle, D.; Eccleston, C. Implanted Spinal Neuromodulation Interventions for Chronic Pain in Adults. Cochrane Database Syst. Rev. 2021, 12, CD013756. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Spinal Cord Stimulation for Chronic Pain of Neuropathic or Ischaemic Origin. 2008. Available online: https://www.nice.org.uk/guidance/ta159/resources/spinal-cord-stimulation-for-chronic-pain-of-neuropathic-or-ischaemic-origin-pdf-82598323141573 (accessed on 18 May 2022).
- Food and Drug Administration (FDA). Senza Spinal Cord Stimulation (SCS) System FDA Approval. 2021. Available online: https://www.accessdata.fda.gov/cdrh_docs/pdf13/P130022S039A.pdf (accessed on 14 April 2022).
- Henson, J.V.; Varhabhatla, N.C.; Bebic, Z.; Kaye, A.D.; Yong, R.J.; Urman, R.D.; Merkow, J.S. Spinal Cord Stimulation for Painful Diabetic Peripheral Neuropathy: A Systematic Review. Pain Ther. 2021, 10, 895–908. [Google Scholar] [CrossRef]
- de Vos, C.C.; Meier, K.; Zaalberg, P.B.; Nijhuis, H.J.A.; Duyvendak, W.; Vesper, J.; Enggaard, T.P.; Lenders, M.W.P.M. Spinal Cord Stimulation in Patients with Painful Diabetic Neuropathy: A Multicentre Randomized Clinical Trial. Pain 2014, 155, 2426–2431. [Google Scholar] [CrossRef]
- Slangen, R.; Schaper, N.C.; Faber, C.G.; Joosten, E.A.; Dirksen, C.D.; van Dongen, R.T.; Kessels, A.G.; van Kleef, M. Spinal Cord Stimulation and Pain Relief in Painful Diabetic Peripheral Neuropathy: A Prospective Two-Center Randomized Controlled Trial. Diabetes Care 2014, 37, 3016–3024. [Google Scholar] [CrossRef] [Green Version]
- Chapman, K.B.; Van Roosendaal, B.-K.W.; Van Helmond, N.; Yousef, T.A. Unilateral Dorsal Root Ganglion Stimulation Lead Placement with Resolution of Bilateral Lower Extremity Symptoms in Diabetic Peripheral Neuropathy. Cureus 2020, 12, e10735. [Google Scholar] [CrossRef]
- Eldabe, S.; Espinet, A.; Wahlstedt, A.; Kang, P.; Liem, L.; Patel, N.K.; Vesper, J.; Kimber, A.; Cusack, W.; Kramer, J. Retrospective Case Series on the Treatment of Painful Diabetic Peripheral Neuropathy with Dorsal Root Ganglion Stimulation. Neuromodul. J. Int. Neuromodul. Soc. 2018, 21, 787–792. [Google Scholar] [CrossRef]
- Bosi, E.; Conti, M.; Vermigli, C.; Cazzetta, G.; Peretti, E.; Cordoni, M.C.; Galimberti, G.; Scionti, L. Effectiveness of Frequency-Modulated Electromagnetic Neural Stimulation in the Treatment of Painful Diabetic Neuropathy. Diabetologia 2005, 48, 817–823. [Google Scholar] [CrossRef] [Green Version]
- Bosi, E.; Bax, G.; Scionti, L.; Spallone, V.; Tesfaye, S.; Valensi, P.; Ziegler, D.; on behalf of the FREMS European Trial Study Group. Frequency-Modulated Electromagnetic Neural Stimulation (FREMS) as a Treatment for Symptomatic Diabetic Neuropathy: Results from a Double-Blind, Randomised, Multicentre, Long-Term, Placebo-Controlled Clinical Trial. Diabetologia 2013, 56, 467–475. [Google Scholar] [CrossRef] [Green Version]
- Crasto, W.; Altaf, Q.-A.; Selvaraj, D.R.; Jack, B.; Patel, V.; Nawaz, S.; Murthy, N.; Sukumar, N.; Saravanan, P.; Tahrani, A.A. Frequency Rhythmic Electrical Modulation System (FREMS) to Alleviate Painful Diabetic Peripheral Neuropathy: A Pilot, Randomised Controlled Trial (The FREMSTOP Study). Diabet. Med. 2021, 39, e14710. [Google Scholar] [CrossRef]
- Stein, C.; Eibel, B.; Sbruzzi, G.; Lago, P.D.; Plentz, R.D.M. Electrical Stimulation and Electromagnetic Field Use in Patients with Diabetic Neuropathy: Systematic Review and Meta-Analysis. Braz. J. Phys. Ther. 2013, 17, 93–104. [Google Scholar] [CrossRef] [Green Version]
- Najafi, B.; Crews, R.T.; Wrobel, J.S. A Novel Plantar Stimulation Technology for Improving Protective Sensation and Postural Control in Patients with Diabetic Peripheral Neuropathy: A Double-Blinded, Randomized Study. Gerontology 2013, 59, 473–480. [Google Scholar] [CrossRef]
- Najafi, B.; Talal, T.K.; Grewal, G.S.; Menzies, R.; Armstrong, D.G.; Lavery, L.A. Using Plantar Electrical Stimulation to Improve Postural Balance and Plantar Sensation Among Patients with Diabetic Peripheral Neuropathy: A Randomized Double Blinded Study. J. Diabetes Sci. Technol. 2017, 11, 693–701. [Google Scholar] [CrossRef] [Green Version]
- Hamza, M.A.; White, P.F.; Craig, W.F.; Ghoname, E.S.; Ahmed, H.E.; Proctor, T.J.; Noe, C.E.; Vakharia, A.S.; Gajraj, N. Percutaneous Electrical Nerve Stimulation: A Novel Analgesic Therapy for Diabetic Neuropathic Pain. Diabetes Care 2000, 23, 365–370. [Google Scholar] [CrossRef] [Green Version]
- Ziegler, D.; Ametov, A.; Barinov, A.; Dyck, P.J.; Gurieva, I.; Low, P.A.; Munzel, U.; Yakhno, N.; Raz, I.; Novosadova, M.; et al. Oral Treatment with α-Lipoic Acid Improves Symptomatic Diabetic Polyneuropathy: The SYDNEY 2 Trial. Diabetes Care 2006, 29, 2365–2370. [Google Scholar] [CrossRef] [Green Version]
- Ziegler, D.; Low, P.A.; Litchy, W.J.; Boulton, A.J.M.; Vinik, A.I.; Freeman, R.; Samigullin, R.; Tritschler, H.; Munzel, U.; Maus, J.; et al. Efficacy and Safety of Antioxidant Treatment with α-Lipoic Acid over 4 Years in Diabetic Polyneuropathy: The NATHAN 1 Trial. Diabetes Care 2011, 34, 2054–2060. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.; Cai, X.; Wu, H.; Ji, L. Associations between Metformin Use and Vitamin B12 Levels, Anemia, and Neuropathy in Patients with Diabetes: A Meta-Analysis. J. Diabetes 2019, 11, 729–743. [Google Scholar] [CrossRef]
- American Diabetes Association 3. Prevention or Delay of Type 2 Diabetes: Standards of Medical Care in Diabetes—2021. Diabetes Care 2020, 44, S34–S39. [Google Scholar] [CrossRef]
- Medicines and Healthcare products Regulatory Agency (MHRA). Metformin and Reduced Vitamin B12 Levels: New Advice for Monitoring Patients at Risk. 2022. Available online: https://www.gov.uk/drug-safety-update/metformin-and-reduced-vitamin-b12-levels-new-advice-for-monitoring-patients-at-risk (accessed on 12 July 2022).
- Okada, K.; Tanaka, H.; Temporin, K.; Okamoto, M.; Kuroda, Y.; Moritomo, H.; Murase, T.; Yoshikawa, H. Methylcobalamin Increases Erk1/2 and Akt Activities through the Methylation Cycle and Promotes Nerve Regeneration in a Rat Sciatic Nerve Injury Model. Exp. Neurol. 2010, 222, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Han, W.; Hu, S.; Xu, H. Methylcobalamin: A Potential Vitamin of Pain Killer. Neural Plast. 2013, 2013, e424651. [Google Scholar] [CrossRef] [PubMed]
- Jayabalan, B.; Low, L.L. Vitamin B Supplementation for Diabetic Peripheral Neuropathy. Singap. Med. J. 2016, 57, 55–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Didangelos, T.; Karlafti, E.; Kotzakioulafi, E.; Margariti, E.; Giannoulaki, P.; Batanis, G.; Tesfaye, S.; Kantartzis, K. Vitamin B12 Supplementation in Diabetic Neuropathy: A 1-Year, Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2021, 13, 395. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Health and Care Excellence (NICE). Clinical Knowledge Summaries. Cyanocobalamin. Prescribing Information. Anaemia-B12 and Folate Deficiency. Available online: https://cks.nice.org.uk/topics/anaemia-b12-folate-deficiency/prescribing-information/cyanocobalamin/ (accessed on 14 July 2022).
- Li, S.; Chen, X.; Li, Q.; Du, J.; Liu, Z.; Peng, Y.; Xu, M.; Li, Q.; Lei, M.; Wang, C.; et al. Effects of Acetyl-L-Carnitine and Methylcobalamin for Diabetic Peripheral Neuropathy: A Multicenter, Randomized, Double-Blind, Controlled Trial. J. Diabetes Investig. 2016, 7, 777–785. [Google Scholar] [CrossRef]
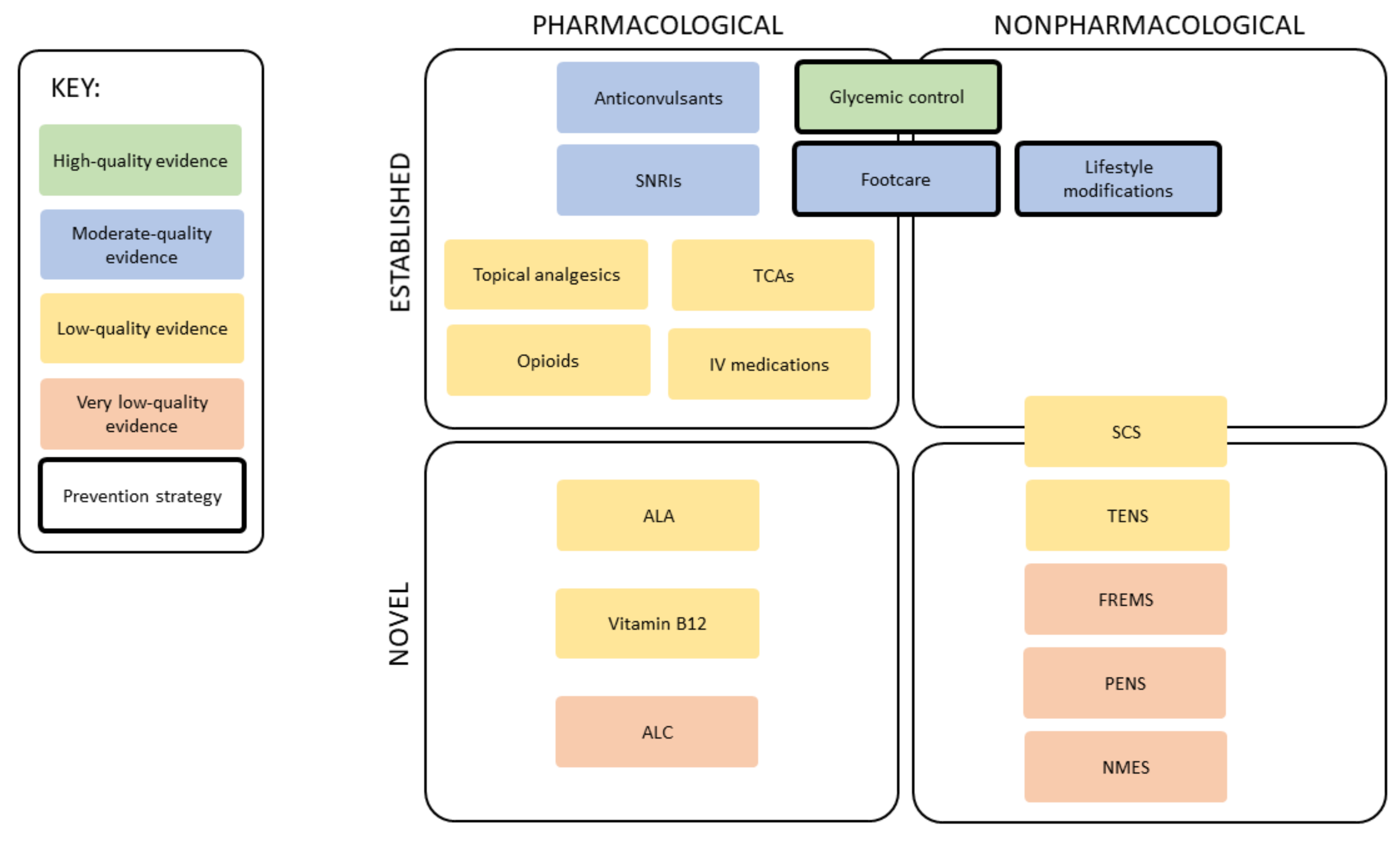
| Prevention Strategy | Indication(s) | Intervention Types | Level of Evidence | Advantages | Disadvantages |
|---|---|---|---|---|---|
| Glycemic control | To reduce the risk of DN | Pharmacological:
Nonpharmacological:
| High-quality—all intervention types that enhance glycemic control for at least 12 months [70] |
|
|
| Lifestyle modifications | To reduce the risk of DN, to prevent progression of DN, to reduce cardiometabolic factors | Nonpharmacological:
| Moderate-quality—supervised exercise programs for DN and DFUs in people with diabetes [80,81] Low-quality—supervised exercise programs for DN in people with prediabetes [82], diet and counselling for DN in people with diabetes |
|
|
| Footcare | To reduce the risk of further foot complications | Pharmacological:
| Low-quality—referral to multidisciplinary footcare services, patient education on footcare |
|
| Pain Management Strategy | Level of Evidence | Advantages | Disadvantages |
|---|---|---|---|
| Anticonvulsants—pregabalin and gabapentin | Moderate-quality [127,128] |
| |
| Serotonin and norepinephrine reuptake inhibitors (SNRIs)—duloxetine and venlafaxine | Moderate-quality (duloxetine) [134,135,136,137,138,139,140,141]; Low-quality (venlafaxine) [142] |
|
|
| Tricyclic antidepressants (TCAs) –amitriptyline | Low-quality [144] |
| |
| Opioids—tramadol and tapentadol | Low-quality [146,147] | ||
| Topical analgesics—topical capsaicin | Moderate-quality (8% capsaicin) [154,155]; Low-quality (0.075% capsaicin) [156,157] | ||
| Intravenous (IV) medications—IV lidocaine and IV ketamine | Low-quality [159,160,161] |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, S.; Normahani, P.; Lane, T.; Hohenschurz-Schmidt, D.; Oliver, N.; Davies, A.H. Prevention and Management Strategies for Diabetic Neuropathy. Life 2022, 12, 1185. https://doi.org/10.3390/life12081185
Smith S, Normahani P, Lane T, Hohenschurz-Schmidt D, Oliver N, Davies AH. Prevention and Management Strategies for Diabetic Neuropathy. Life. 2022; 12(8):1185. https://doi.org/10.3390/life12081185
Chicago/Turabian StyleSmith, Sasha, Pasha Normahani, Tristan Lane, David Hohenschurz-Schmidt, Nick Oliver, and Alun Huw Davies. 2022. "Prevention and Management Strategies for Diabetic Neuropathy" Life 12, no. 8: 1185. https://doi.org/10.3390/life12081185
APA StyleSmith, S., Normahani, P., Lane, T., Hohenschurz-Schmidt, D., Oliver, N., & Davies, A. H. (2022). Prevention and Management Strategies for Diabetic Neuropathy. Life, 12(8), 1185. https://doi.org/10.3390/life12081185






