Combination of Two Manipulative Techniques for the Treatment of Cervicogenic Dizziness: A Randomized Controlled Trial
Abstract
:1. Introduction
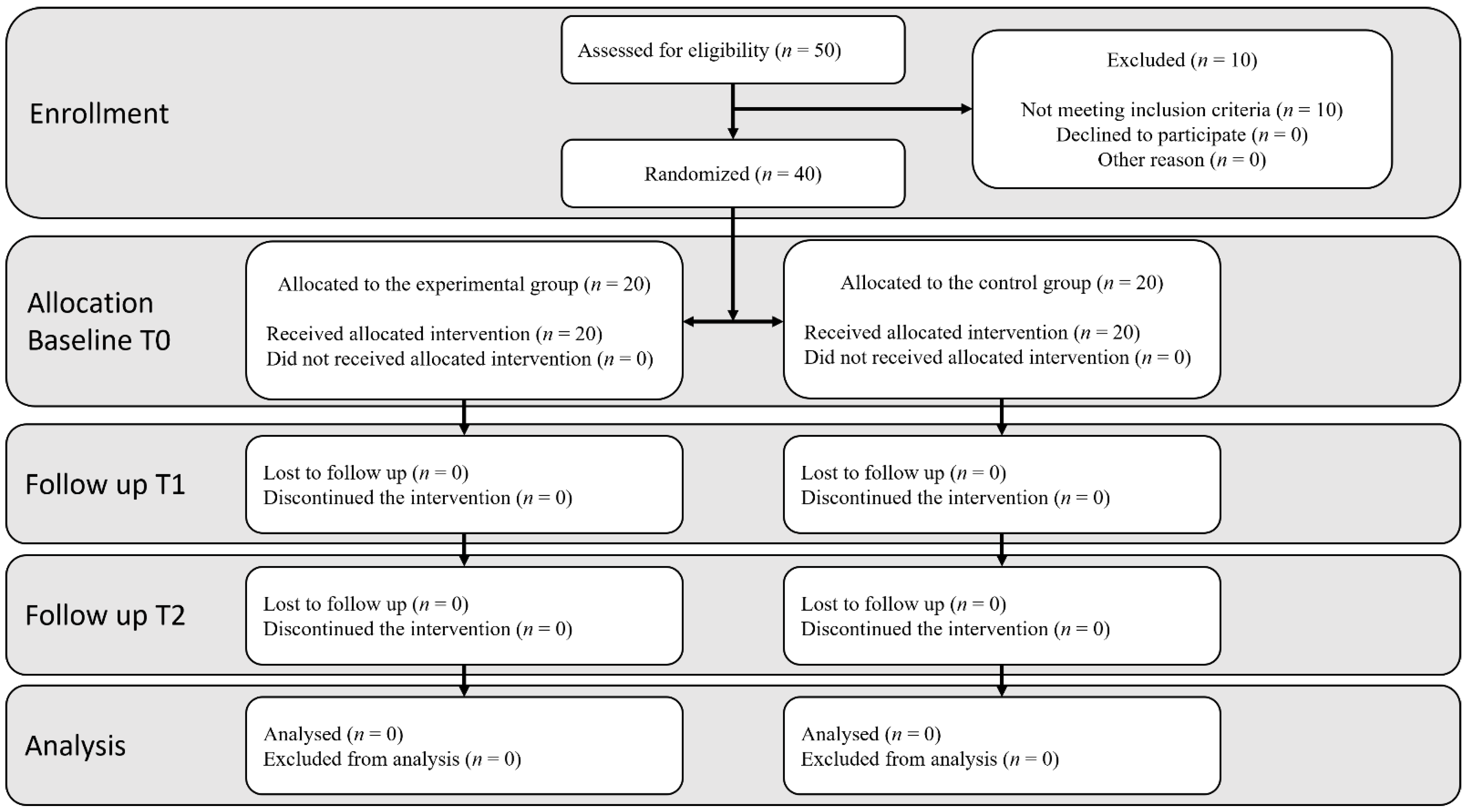
2. Materials and Methods
3. Results
3.1. wVAS
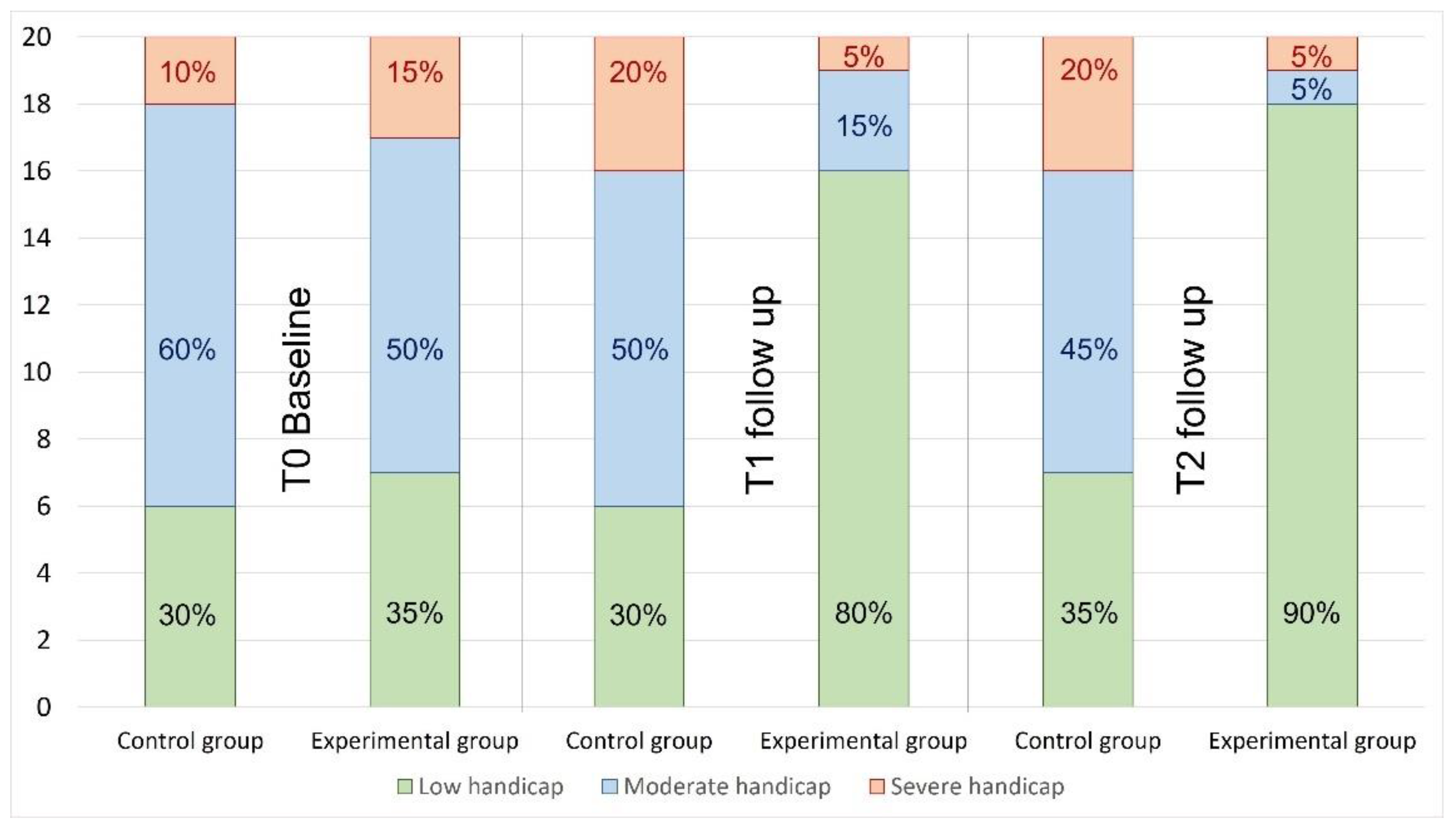
3.2. DHI
3.3. NDI
3.4. UCS Movement (FRT)
3.5. Pressure Pain Threshold
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Casado, V. Atención al paciente neurológico en los Servicios de Urgencias. Revisión de la situación actual en España. Neurologia 2011, 26, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Guerra-Jiménez, G.; Arenas Rodríguez, A.; Falcón González, J.C.; Pérez Plasencia, D.; Ramos Macías, Á. Epidemiología de los trastornos vestibulares en la consulta de otoneurología. Acta Otorrinolaringológica Española 2017, 68, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Olusesi, A.D.; Abubakar, J. 10 Years of vertigo clinic at National Hospital Abuja, Nigeria: What have we learned? Eur. Arch. Oto-Rhino-Laryngol. 2016, 273, 3567–3572. [Google Scholar] [CrossRef]
- Huijbregts, P.; Vidal, P. Dizziness in orthopaedic physical therapy practice: Classification and pathophysiology. J. Man. Manip. Ther. 2004, 12, 199–214. [Google Scholar] [CrossRef]
- Wrisley, D.M.; Sparto, P.J.; Whitney, S.L.; Furman, J.M. Cervicogenic dizziness: A review of diagnosis and treatment. J. Orthop. Sports Phys. Ther. 2000, 30, 755–766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sung, Y.-H. Upper cervical spine dysfunction and dizziness. J. Exerc. Rehabil. 2020, 16, 385–391. [Google Scholar] [CrossRef]
- Boyd-Clark, L.C.; Briggs, C.A.; Galea, M.P. Muscle spindle distribution, morphology, and density in longus colli and multifidus muscles of the cervical spine. Spine 2002, 27, 694–701. [Google Scholar] [CrossRef] [Green Version]
- Dutia, M.B. The muscles and joints of the neck: Their specialisation and role in head movement. Prog. Neurobiol. 1991, 37, 165–178. [Google Scholar] [CrossRef]
- Kristjansson, E.; Treleaven, J. Sensorimotor function and dizziness in neck pain: Implications for assessment and management. J. Orthop. Sports Phys. Ther. 2009, 39, 364–377. [Google Scholar] [CrossRef] [Green Version]
- Devaraja, K. Approach to cervicogenic dizziness: A comprehensive review of its aetiopathology and management. Eur. Arch. Oto-Rhino-Laryngol. 2018, 275, 2421–2433. [Google Scholar] [CrossRef]
- Knapstad, M.; Goplen, F.; Ask, T.; Skouen, J.; Nordahl, S. Associations between pressure pain threshold in the neck and postural control in patients with dizziness or neck pain—A cross-sectional study. BMC Musculoskelet. Disord. 2019, 20, 528. [Google Scholar] [CrossRef]
- Jull, G.; Falla, D.; Treleaven, J.; O’Leary, S. Management of Neck Pain Disorders, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Treleaven, J. Sensorimotor disturbances in neck disorders affecting postural stability, head and eye movement control. Man. Ther. 2008, 13, 2–11. [Google Scholar] [CrossRef]
- Malmström, E.; Westergren, H.; Fransson, P.; Karlberg, M.; Magnusson, M. Experimentally induced deep cervical muscle pain distorts head on trunk orientation. Eur. J. Appl. Physiol. 2013, 113, 2487–2499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Jong, P.T.; de Jong, J.M.; Cohen, B.; Jongkees, L.B. Ataxia and nystagmus induced by injection of local anesthetics in the Neck. Ann. Neurol. 1977, 1, 240–246. [Google Scholar] [CrossRef] [PubMed]
- La, C. Role of eye and neck proprioceptive mechanisms in body orientation and motor coordination. J. Neurophysiol. 1961, 24, 1–11. [Google Scholar] [CrossRef]
- Micarelli, A.; Viziano, A.; Augimeri, I.; Micarelli, B.; Capoccia, D.; Alessandrini, M. Diagnostic route of cervicogenic dizziness: Usefullness of posturography, objective and subjective testing implementation and their correlation. Disabil. Rehabil. 2021, 43, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Knapstad, M.K.; Nordahl, S.H.G.; Goplen, F.K. Clinical characteristics in patients with cervicogenic dizziness: A systematic review. Health Sci. Rep. 2019, 2, e134. [Google Scholar] [CrossRef]
- L’Heureux-Lebeau, B.; Godbout, A.; Berbiche, D.; Saliba, I. Evaluation of paraclinical tests in the diagnosis of cervicogenic dizziness. Otol. Neurotol. 2014, 35, 1858–1865. [Google Scholar] [CrossRef]
- Yahia, A.; Ghroubi, S.; Jribi, S.; Mâlla, J.; Baklouti, S.; Ghorbel, A.; Elleuch, M.H. Chronic neck pain and vertigo: Is a true balance disorder present? Ann. Phys. Rehabil. Med. 2009, 52, 556–567. [Google Scholar] [CrossRef] [Green Version]
- Yahia, A.; Jribi, S.; Ghroubi, S.; Elleuch, M.; Baklouti, S.; Habib Elleuch, M. Evaluation of the posture and muscular strength of the trunk and inferior members of patients with chronic lumbar pain. Jt. Bone Spine 2011, 78, 291–297. [Google Scholar] [CrossRef]
- Reid, S.A.; Callister, R.; Katekar, M.G.; Treleaven, J.M. Utility of a brief assessment tool developed from the dizziness handicap inventory to screen for cervicogenic dizziness: A case control study. Musculoskelet. Sci. Pract. 2017, 30, 42–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, F.C.; Mathew, S.; Littmann, A.E.; MacDonald, C.W. Clinical decision making in the management of patients with cervicogenic dizziness: A case series. J. Orthop. Sports Phys Ther. 2017, 47, 874–884. [Google Scholar] [CrossRef] [PubMed]
- Reneker, J.C.; Clay Moughiman, M.; Cook, C.E. The diagnostic utility of clinical tests for differentiating between cervicogenic and other causes of dizziness after a sports-related concussion: An international Delphi study. J. Sci. Med. Sport 2015, 18, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Yaseen, K.; Hendrick, P.; Ismail, A.; Felemban, M.; Alshehri, M.A. The effectiveness of manual therapy in treating cervicogenic dizziness: A systematic review. J. Phys. Ther. Sci. 2018, 30, 96–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reid, S.; Callister, R.; Katekar, M.; Rivett, D. Effects of cervical spine manual therapy on range of motion, head repositioning and balance in participants with cervicogenic dizziness: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2014. [Google Scholar] [CrossRef]
- Reid, S.; Rivett, D.; Katekar, M.; Callister, R. Sustained natural apophyseal glides (SNAGs) are an effective treatment for cervicogenic dizziness. Man. Ther. 2008, 13, 357–366. [Google Scholar] [CrossRef]
- Reid, S.A.; Callister, R.; Snodgrass, S.J.; Katekar, M.G.; Rivett, D.A. Manual therapy for cervicogenic dizziness: Long-term outcomes of a randomised trial. Man. Ther. 2015, 20, 148–156. [Google Scholar] [CrossRef]
- Malmström, E.-M.; Karlberg, M.; Melander, A.; Magnusson, M.; Moritz, U. Cervicogenic dizziness—Musculoskeletal findings before and after treatment and long-term outcome. Disabil. Rehabil. 2007, 29, 1193–1205. [Google Scholar] [CrossRef]
- Reid, S.A.; Rivett, D.A.; Katekar, M.G.; Callister, R. Efficacy of manual therapy treatments for people with cervicogenic dizziness and pain: Protocol of a randomised controlled trial. BMC Musculoskelet. Disord. 2012, 13, 201. [Google Scholar] [CrossRef] [Green Version]
- Knapstad, M.K.; Nordahl, S.H.G.; Naterstad, I.F.; Ask, T.; Skouen, J.S.; Goplen, F.K. Measuring pressure pain threshold in the cervical region of dizzy patients—The reliability of a pressure algometer. Physiother. Res. Int. 2018, 23, e1736. [Google Scholar] [CrossRef] [Green Version]
- Rushton, A.; Rivett, D.; Carlesso, L.; Flynn, T.; Hing, W.; Kerry, R. International framework for examination of the cervical region for potential of Cervical Arterial Dysfunction prior to Orthopaedic Manual Therapy intervention. Man. Ther. 2014, 19, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Kaltenborn, F. Fisioterapia Manual. Volumen III: Manipulación-Tracción de Las Extremidades y La Columna; Primera: Zaragoza, Spain, 2009. [Google Scholar]
- Carrasco-Uribarren, A.; Rodríguez-Sanz, J.; Hidalgo-García, C.; Tricás-Moreno, J.M.; Balboa-López, D.; Cabanillas-Barea, S. Short-term effects of an upper cervical spine traction-manipulation program in patients with cervicogenic dizziness: A case series study. J. Back Musculoskelet. Rehabil. 2020, 33, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Uribarren, A.; Rodriguez-Sanz, J.; López-de-Celis, C.; Pérez-Guillen, S.; Tricás-Moreno, J.M.; Cabanillas-Barea, S. Short-term effects of the traction-manipulation protocol in dizziness intensity and disability in cervicogenic dizziness: A randomized controlled trial. Disabil. Rehabil. 2021, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Malo-Urriés, M.; Tricás-Moreno, J.M.; Estébanez-de-Miguel, E.; Hidalgo-García, C.; Carrasco-Uribarren, A.; Cabanillas-Barea, S. Immediate effects of upper cervical translatoric mobilization on cervical mobility and pressure pain threshold in patients with cervicogenic headache: A randomized controlled trial. J. Manip. Physiol. Ther. 2017, 40, 649–658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reid, S.A.; Rivett, D.A.; Katekar, M.G.; Callister, R. Comparison of mulligan sustained natural apophyseal glides and maitland mobilizations for treatment of cervicogenic dizziness: A randomized controlled trial. Phys. Ther. 2014, 94, 466–476. [Google Scholar] [CrossRef] [Green Version]
- Rushton, A.; Carlesso, L.C.; Flynn, T.; Hing, W.A.; Kerry, R.; Rubinstein, S.M.; Vogel, S. International Framework for Examination of the Cervical Region for Potential of Vascular Pathologies of the Neck Prior to Orthopaedic Manual Therapy (OMT) Intervention: International IFOMPT Cervical Framework. 2020. Available online: https://www.ifompt.org/site/ifompt/IFOMPT%20Cervical%20Framework%20final%20September%202020.pdf (accessed on 11 June 2022).
- Tamber, A.-L.; Bruusgaard, D. Self-reported faintness or dizziness—Comorbidity and use of medicines. An epidemiological study. Scand. J. Public Health 2009, 37, 613–620. [Google Scholar] [CrossRef]
- Dannenbaum, E.; Chilingaryan, G.; Fung, J. Visual vertigo analogue scale: An assessment questionnaire for visual vertigo. J. Vestib. Res. 2011, 21, 153–159. [Google Scholar] [CrossRef]
- Fong, E.; Li, C.; Aslakson, R.; Agrawal, Y. Systematic review of patient-reported outcome measures in clinical vestibular research. Arch. Phys. Med. Rehabil. 2015, 96, 357–365. [Google Scholar] [CrossRef] [Green Version]
- Perez, N.; Garmendia, I.; García-Granero, M.; Martin, E.; García-Tapia, R. Factor analysis and correlation between dizziness handicap inventory and dizziness characteristics and impact on quality-of-life scales. Acta Otolaryngol. 2001, 545, 145–154. [Google Scholar] [CrossRef]
- Caldara, B.; Asenzo, A.I.; Paglia, G.B.; Ferreri, E.; Gomez, R.S.; Laiz, M.M.; Luques, M.L.; Mangoni, A.P.; Marazzi, C.; Matesa, M.A.; et al. Adaptación cultural y validación del Dizziness Handicap Inventory: Versión Argentina. Acta Otorrinolaringológica Española 2012, 63, 106–114. [Google Scholar] [CrossRef]
- Jacobson, G.P.; Newman, C.W. The development of the dizziness handicap inventory. Arch. Otolaryngol. Head Neck Surg. 1990, 116, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Andrade Ortega, J.A.; Delgado Martínez, A.D.; Almécija Ruiz, R. Validation of a Spanish version of the Neck Disability Index. Medicina Clínica 2008, 130, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Takasaki, H.; Hall, T.; Oshiro, S.; Kaneko, S.; Ikemoto, Y.; Jull, G. Normal kinematics of the upper cervical spine during the Flexion–Rotation Test—In vivo measurements using magnetic resonance imaging. Man. Ther. 2011, 16, 167–171. [Google Scholar] [CrossRef] [Green Version]
- Hall, T.M.; Robinson, K.W.; Fujinawa, O.; Akasaka, K.; Pyne, E.A. Intertester reliability and diagnostic validity of the cervical flexion-rotation test. J. Manip. Physiol. Ther. 2008, 31, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo García, C.; Fanlo Mazas, P.; Malo Urriés, M.; Tricas-Moreno, J.M.; Lucha-Lopez, O.; Estebanezde-Miguel, E.; Bueno-Gracia, E.; Perez-Guillen, S.; Ruiz-de-Escudero, A.; Krauss, J. Short term efficacy of C0-C1 mobilization in the cervical neutral position in upper cervical hypomobility: A randomized controlled trial. J. Int. Acad. Phys. Ther. Res. 2016, 7, 908–914. [Google Scholar] [CrossRef]
- Cohen, J. The concepts of power analysis. In Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 1988; pp. 1–17. [Google Scholar] [CrossRef]
- Heikkilä, H.; Johansson, M.; Wenngren, B.I. Effects of acupuncture, cervical manipulation and NSAID therapy on dizziness and impaired head repositioning of suspected cervical origin: A pilot study. Man. Ther. 2000, 5, 151–157. [Google Scholar] [CrossRef]
- Mutlu, B.; Serbetcioglu, B. Discussion of the dizziness handicap inventory. J. Vestib. Res. 2013, 23, 271–277. [Google Scholar] [CrossRef]
- Whitney, S.L.; Wrisley, D.M.; Brown, K.E.; Furman, J.M. Is perception of handicap related to functional performance in persons with vestibular dysfunction? Otol. Neurotol. 2004, 25, 139–143. [Google Scholar] [CrossRef]
- MacDermid, J.C.; Walton, D.M.; Avery, S.; Blanchard, A.; Etruw, E.; Mcalpine, C.; Goldsmith, C.H. Measurement properties of the neck disability index: A systematic review. J. Orthop. Sport Phys. Ther. 2009, 39, 400–417. [Google Scholar] [CrossRef] [Green Version]
- Strunk, R.G.; Hawk, C. Effects of chiropractic care on dizziness, neck pain, and balance: A single-group, preexperimental, feasibility study. J. Chiropr. Med. 2009, 8, 156–164. [Google Scholar] [CrossRef] [Green Version]
- Minguez-Zuazo, A.; Grande-Alonso, M.; Saiz, B.M.; La Touche, R.; Lara, S.L. Therapeutic patient education and exercise therapy in patients with cervicogenic dizziness: A prospective case series clinical study. J. Exerc. Rehabil. 2016, 12, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Aasa, B.; Berglund, L.; Michaelson, P.; Aasa, U. Individualized low-load motor control exercises and education versus a high-load lifting exercise and education to improve activity, pain intensity, and physical performance in patients with low back pain: A randomized controlled trial. J. Orthop. Sport Phys. Ther. 2015, 45, 77–85. [Google Scholar] [CrossRef] [Green Version]
- Sterling, M.; Pedler, A.; Chan, C.; Puglisi, M.; Vuvan, V.; Vicenzino, B. Cervical lateral glide increases nociceptive flexion reflex threshold but not pressure or thermal pain thresholds in chronic whiplash associated disorders: A pilot randomised controlled trial. Man. Ther. 2010, 15, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Magnusson, M.; Malmström, E.-M. The conundrum of cervicogenic dizziness. In Neuro-Otology; Furman, J.M., Lempert, T., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 365–369. [Google Scholar] [CrossRef]
- Hall, T.; Robinson, K. The flexion-rotation test and active cervical mobility—A comparative measurement study in cervicogenic headache. Man. Ther. 2004, 9, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Ogince, M.; Hall, T.; Robinson, K.; Blackmore, A.M. The diagnostic validity of the cervical flexion-rotation test in C1/2-related cervicogenic headache. Man. Ther. 2007, 12, 256–262. [Google Scholar] [CrossRef]
- Jull, G.; Sterling, M.; Falla, D.; Treleaven, J.; O’Leary, S. Whiplash, Headache, and Neck Pain: Research-Based Directions for Physical Therapies; Churchill Livingstone: London, UK, 2008. [Google Scholar] [CrossRef]
- Curatolo, M.; Petersen-Felix, S.; Arendt-Nielsen, L.; Giani, C.; Zbinden, A.M.; Radanov, B.P. Central hypersensitivity in chronic pain after whiplash injury. Clin. J. Pain 2001, 17, 306–315. [Google Scholar] [CrossRef]
- Seaman, D.R.; Winterstein, J.F. Dysafferentation: A novel term to describe the neuropathophysiological effects of joint complex dysfunction. A look at likely mechanisms of symptom generation. J. Manip. Physiol. Ther. 1998, 21, 267–280. [Google Scholar]
- Sterling, M.; Treleaven, J.; Edwards, S.; Jull, G. Pressure pain thresholds in chronic whiplash associated disorder: Further evidence of altered central pain processing. J. Musculoskelet. Pain 2002, 10, 69–81. [Google Scholar] [CrossRef]
- Sterling, M.; Jull, G.; Vicenzino, B.; Kenardy, J.; Darnell, R. Development of motor system dysfunction following whiplash injury. Pain 2003, 103, 65–73. [Google Scholar] [CrossRef]
- Schmid, A.; Brunner, F.; Wright, A.; Bachmann, L.M. Paradigm shift in manual therapy? Evidence for a central nervous system component in the response to passive cervical joint mobilisation. Man. Ther. 2008, 13, 387–396. [Google Scholar] [CrossRef]
- Nijs, J.; Van Houdenhove, B.; Oostendorp, R.A.B. Recognition of central sensitization in patients with musculoskeletal pain: Application of pain neurophysiology in manual therapy practice. Man. Ther. 2010, 15, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Voogt, L.; de Vries, J.; Meeus, M.; Struyf, F.; Meuffels, D.; Nijs, J. Analgesic effects of manual therapy in patients with musculoskeletal pain: A systematic review. Man. Ther. 2014, 20, 250–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moustafa, I.M.; Diab, A.A.; Harrison, D.E. The effect of normalizing the sagittal cervical configuration on dizziness, neck pain, and cervicocephalic kinesthetic sensibility: A 1-year randomized controlled study. Eur. J. Phys. Rehabil. Med. 2017, 53, 57–71. [Google Scholar] [CrossRef] [PubMed]
- Hawk, C.; Cambron, J.; Pfefer, M. Pilot study of the effect of a limited and extended course of chiropractic care on balance, chronic pain, and dizziness in older adults. J. Manip. Physiol. Ther. 2009, 32, 438–447. [Google Scholar] [CrossRef]
- Hawk, C.; Cambron, J. Chiropractic care for older adults: Effects on balance, dizziness, and chronic pain. J. Manip. Physiol. Ther. 2009, 32, 431–437. [Google Scholar] [CrossRef]
- Bracher, E.S.; Almeida, C.I.; Almeida, R.R.; Duprat, A.C.; Bracher, C.B. A combined approach for the treatment of cervical vertigo. J. Manip. Physiol. Ther. 2000, 23, 96–100. [Google Scholar] [CrossRef]
- Cuenca-Martínez, F.; Bartrina-Rodríguez, I.; Suso-Martí, L.; La Touche, R.; Ferrer-Peña, R. Association between somatosensory, motor and psychological variables by levels of disability in patients with cervicogenic dizziness. Somatosens. Mot. Res. 2018, 35, 247–252. [Google Scholar] [CrossRef]

| Experimental Group (n = 20) | Control Group (n = 20) | p Value | |
|---|---|---|---|
| Gender (M; F) | 4M;16F | 4M;16F | 0.999 |
| Age (years) | 55.90 (11.96) | 52.10 (16.03) | 0.401 |
| Weight (kg) | 65.90 (11.96) | 66.90 (11.56) | 0.730 |
| Height (cm) | 163.30 (8.53) | 162.40 (7.79) | 0.790 |
| wVAS | 74.45 (17.34) | 70.94 (23.48) | 0.585 |
| NDI | 12.45 (5.96) | 14.60 (5.44) | 0.231 |
| TSK-11 | 24.20 (5.72) | 24.45 (6.82) | 0.901 |
| DHI | |||
| Severe | 3 (15%) | 2 (10%) | 0.795 |
| Moderate | 10 (50%) | 12 (60%) | |
| Low | 7 (35%) | 6 (30%) |
| Group | Baseline | Follow-Up | Within Group | Between Group | Follow-Up | Within Group | Between Group |
|---|---|---|---|---|---|---|---|
| T0 | T1 | T0-T1 | T0-T1 | T2 | T0-T2 | T0-T2 | |
| wVAS | |||||||
| Control group | 70.85 ± 23.48 | 61.20 ± 25.89 | p < 0.334 | p < 0.001 d = 1.46 | 67.75 ± 20.38 | p < 0.999 | p < 0.001 d = 1.28 |
| d = 0.39 | d = 0.14 | ||||||
| Experimental group | 74.45 ± 17.34 | 25.65 ± 22.50 | p < 0.001 | 37.05 ± 27.08 | p < 0.001 | ||
| d = 2.39 | d = 1.64 | ||||||
| NDI | |||||||
| Control group | 14.60 ± 5.44 | 13.40 ± 6.84 | p < 0.285 | p < 0.064 d = 0.64 | 13.60 ± 5.56 | p < 0.625 | p < 0.012 d = 0.94 |
| d = 0.19 | d = 0.18 | ||||||
| Experimental group | 12.25 ± 5.96 | 9.35 ± 5.77 | p < 0.001 | 7.95 ± 6.39 | p < 0.002 | ||
| d = 0.53 | d = 0.73 | ||||||
| FRT Most restricted side | |||||||
| Control group | 18.65 ± 6.96 | 19.63 ± 8.04 | p < 0.999 | p < 0.001 d = 1.36 | 20.83 ± 8.26 | p < 0.686 | p < 0.001 d = 1.23 |
| d = 0.13 | d = 0.29 | ||||||
| Experimental group | 17.27 ± 4.61 | 29.40 ± 6.23 | p < 0.001 | 30.03 ± 6.66 | p < 0.001 | ||
| d = 2.21 | d = 2.23 | ||||||
| FRT Less restricted side | |||||||
| Control group | 23.40 ± 7.72 | 20.98 ± 7.70 | p < 0.558 | p < 0.001 d = 1.43 | 21.66 ± 8.48 | p < 0.944 | p < 0.001 d = 1.03 |
| d = 0.31 | d =0.22 | ||||||
| Experimental group | 22.13 ± 6.75 | 31.01 ± 6.41 | p < 0.001 | 29.78 ± 7.27 | p < 0.002 | ||
| d = 1.35 | d = 1.09 | ||||||
| Group | Baseline | Follow-Up | Within Group | Between Group | Follow-Up | Within Group | Between Group |
|---|---|---|---|---|---|---|---|
| T0 | T1 | T0-T1 | T0-T1 | T2 | T0-T2 | T0-T2 | |
| Upper Trapezius (R) (kPa) | |||||||
| Control group | 188.90 ± 66.86 | 178.45 ± 100.14 | p < 0.999 | p < 0.006 d = 0.35 | 191.75 ± 84.20 | p < 0.999 | p < 0.022 d = 0.22 |
| d = 0.12 | d = 0.04 | ||||||
| Experimental group | 166.55 ± 98.53 | 217.10 ± 122.25 | p < 0.022 | 214.90 ± 126.60 | p < 0.016 | ||
| d = 0.46 | d = 0.43 | ||||||
| Upper Trapezius (L) (kPa) | |||||||
| Control group | 174.75 ± 78.46 | 173.10 ± 93.47 | p < 0.999 | p < 0.086 d = 0.34 | 187.20 ± 101.84 | p < 0.999 | p < 0.134 d = 0.32 |
| d = 0.02 | d = 0.14 | ||||||
| Experimental group | 177.65 ± 125.16 | 208.55 ± 112.65 | p < 0.035 | 225.85 ± 137.14 | p < 0.009 | ||
| d = 0.26 | d = 0.37 | ||||||
| C2-3 (R) (kPa) | |||||||
| Control group | 139.15 ± 52.42 | 146.20 ± 60.93 | p < 0.568 | p < 0.273 d = 0.43 | 150.50 ± 53.59 | p < 0.579 | p < 0.639 d = 0.38 |
| d = 0.12 | d = 0.21 | ||||||
| Experimental group | 152.30 ± 97.02 | 179.85 ± 91.67 | p < 0.408 | 172.60 ± 62.56 | p < 0.738 | ||
| d = 0.26 | d = 0.25 | ||||||
| C2-3 (L) (kPa) | |||||||
| Control group | 129.25 ± 54.98 | 150.25 ± 70.93 | p < 0.410 | p < 0.838 d = 0.25 | 139.85 ± 59.71 | p < 0.999 | p < 0.110 d = 0.74 |
| d = 0.33 | d = 0.19 | ||||||
| Experimental group | 152.35 ± 86.11 | 169.75 ± 84.19 | p < 0.399 | 201.55 ± 102.43 | p < 0.071 | ||
| d = 0.20 | d = 0.52 | ||||||
| Suboccipital (R) (kPa) | |||||||
| Control group | 156.70 ± 59.68 | 160.25 ± 66.18 | p < 0.999 | p < 0.070 d = 0.35 | 157.90 ± 59.67 | p < 0.999 | p < 0.043 d = 0.56 |
| d = 0.07 | d = 0.02 | ||||||
| Experimental group | 157.70 ± 97.81 | 193.90 ± 120.61 | p < 0.075 | 201.05 ± 91.94 | p < 0.018 | ||
| d = 0.33 | d = 0.46 | ||||||
| Suboccipital (L) (kPa) | |||||||
| Control group | 132.80 ± 48.77 | 142.55 ± 188.05 | p < 0.064 | p < 0.143 d = 0.30 | 159.45 ± 59.12 | p < 0.999 | p < 0.773 d = 0.42 |
| d = 0.71 | d = 0.14 | ||||||
| Experimental group | 153.20 ± 125.53 | 188.05 ± 102.29 | p < 0.999 | 187.45 ± 73.25 | p < 0.001 | ||
| d = 0.30 | d = 1.64 | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carrasco-Uribarren, A.; Pardos-Aguilella, P.; Pérez-Guillén, S.; López-de-Celis, C.; Rodríguez-Sanz, J.; Cabanillas-Barea, S. Combination of Two Manipulative Techniques for the Treatment of Cervicogenic Dizziness: A Randomized Controlled Trial. Life 2022, 12, 1023. https://doi.org/10.3390/life12071023
Carrasco-Uribarren A, Pardos-Aguilella P, Pérez-Guillén S, López-de-Celis C, Rodríguez-Sanz J, Cabanillas-Barea S. Combination of Two Manipulative Techniques for the Treatment of Cervicogenic Dizziness: A Randomized Controlled Trial. Life. 2022; 12(7):1023. https://doi.org/10.3390/life12071023
Chicago/Turabian StyleCarrasco-Uribarren, Andoni, Pilar Pardos-Aguilella, Silvia Pérez-Guillén, Carlos López-de-Celis, Jacobo Rodríguez-Sanz, and Sara Cabanillas-Barea. 2022. "Combination of Two Manipulative Techniques for the Treatment of Cervicogenic Dizziness: A Randomized Controlled Trial" Life 12, no. 7: 1023. https://doi.org/10.3390/life12071023
APA StyleCarrasco-Uribarren, A., Pardos-Aguilella, P., Pérez-Guillén, S., López-de-Celis, C., Rodríguez-Sanz, J., & Cabanillas-Barea, S. (2022). Combination of Two Manipulative Techniques for the Treatment of Cervicogenic Dizziness: A Randomized Controlled Trial. Life, 12(7), 1023. https://doi.org/10.3390/life12071023










