Antimicrobial Properties of Compounds Isolated from Syzygium malaccense (L.) Merr. and L.M. Perry and Medicinal Plants Used in French Polynesia
Abstract
:1. Introduction
2. Results and Discussion
2.1. Ethnopharmacological Survey
2.2. Antimicrobial Activity and Cytotoxicity of Plant Methanolic Extracts
2.3. Antimicrobial Tests of Methanolic Extract from Syzygium malaccense and Curcuma longa
2.4. Structural Identification
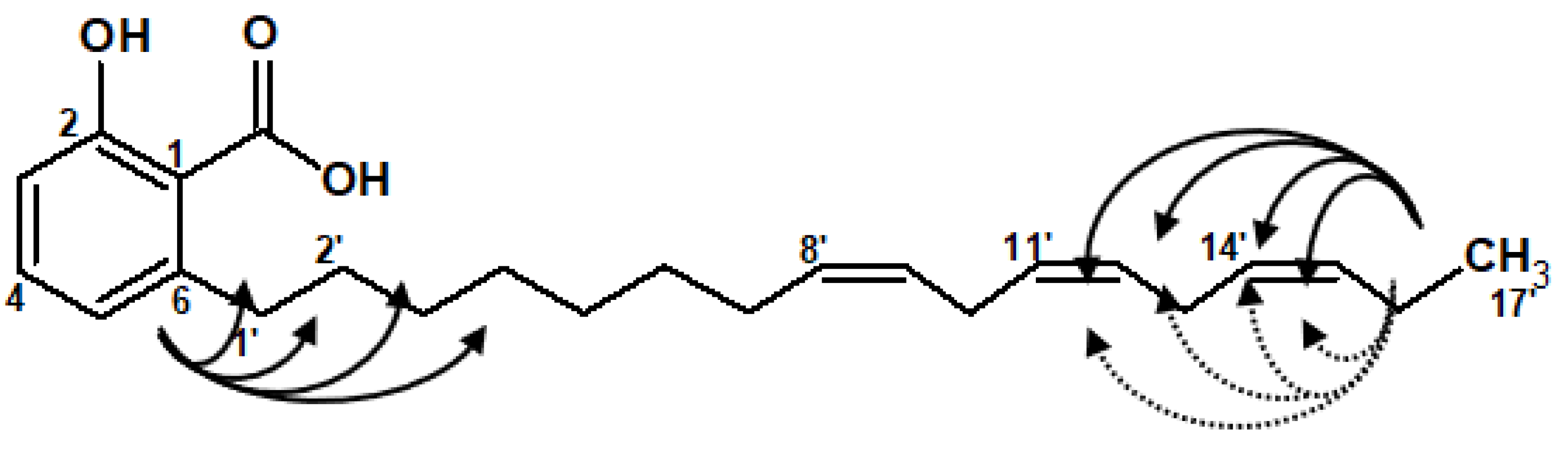
2.4.1. Compound 1: Ginkgolic Acid (C15:0)
2.4.2. Compound 2: Ginkgolic Acid (C15:1, ω5)
2.4.3. Compound 3: Ginkgolic acid (C17:0)
2.4.4. Compound 4: Ginkgolic Acid (C17:1, ω5)
2.4.5. Compound 5: Ginkgolic Acid (C17:3; ω3, ω6, ω9)
2.4.6. Compound 6: Ginkgolic Acid (C17:2; ω6, ω9)
2.4.7. Compound 7: Ginkgolic Acid (C19:1; ω5)
2.5. Antimicrobical Activity of Compounds 1 to 7
2.6. Synergistic Effects of Plant Extracts
2.7. Structure–Activity Relationships
3. Conclusions
4. Materials and Methods
4.1. Ethnopharmacological Survey
4.2. Antimicrobial Activity
4.2.1. Plant Material and Preparation of Extract
4.2.2. MIC and FIC Index Measures
4.2.3. MIC Determination of Isolated Compounds and Standard Molecules Using a Broth Microdilution Method
4.3. Cytotoxic Activity of Crude Plant Extracts
4.4. Bioautography and Isolation of Aliphatic-Salicylic Acids from Syzygium malaccense
4.4.1. Bioautography
4.4.2. Purification of Salicylic Acids Derivatives
4.5. Identification of Salicylic Acids Derivatives
4.5.1. Spectrometric Analysis
4.5.2. Thiomethylation
4.5.3. Compound Spectroscopic Data
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Abreu, A.C.; McBain, A.J.; Simoes, M. Plants as sources of new antimicrobials and resistance-modifying agents. Nat. Prod. Rep. 2012, 29, 1007–1021. [Google Scholar] [CrossRef] [PubMed]
- Jaikumar, K.; Mohamed, S.N.M.; Anand, D.; Saravanan, P. Anticancer activity of Calophyllum inophyllum L., Ethanolic Leaf Extract. Int. J. Pharm. Sci. Res. 2016, 7, 3330–3335. [Google Scholar] [CrossRef]
- Salama, S.M.; Abdulla, M.A.; Alrashdi, A.S.; Ismail, S.; Alkiyumi, S.S.; Golbabapour, S. Hepatoprotective effect of ethanolic extract of Curcuma longa on thioacetamide induced liver cirrhosis in rats. BMC Complement. Altern. Med. 2013, 13, 56–73. [Google Scholar] [CrossRef] [Green Version]
- Dunstan, A.; Noreen, Y.; Serrano, G.; Cox, P.A.; Perera, P.; Bohlin, L. Evaluation of some Samoan and Peruvian medicinal plants by prostaglandin biosynthesis and rat ear oedema assays. J. Ethnopharmacol. 1997, 57, 35–56. [Google Scholar] [CrossRef]
- Savitha, R.C.; Padmavathy, S.; Sundhararajan, A. In vitro antioxidant activities on leaf extracts of Syzygium malaccense (L.) merr and perry. Anc. Sci. Life 2011, 30, 110–113. [Google Scholar] [PubMed]
- Batista, A.G.; da Silva, J.K.; Cazarin, C.B.B.; Biasoto, A.C.T.; Sawaya, A.C.H.F.; Prado, M.A.; Marostica, M.R. Red-jambo (Syzygium malaccense): Bioactive compounds in fruits and leaves. LWT J. Food Sci. Technol. 2017, 76, 284–291. [Google Scholar] [CrossRef]
- Nunes, P.C.; Aquino, J.S.; Rockenbach, I.I.; Stamford, T.L.M. Physico-chemical characterization, bioactive compounds and antioxidant activity of Malay apple (Syzygium malaccense (L.) Merr. & L.M. Perry). PLoS ONE 2016, 11, e0158134. [Google Scholar] [CrossRef] [Green Version]
- Locher, C.P.; Burch, M.T.; Mower, H.F.; Berestecky, J.; Davis, H.; Van Poel, B.; Lasure, A.; VandenBerghe, D.A.; Vlietinck, A.J. Anti-microbial activity and anti-complement activity of extracts obtained from selected Hawaiian medicinal plants. J. ethnopharmacol. 1995, 49, 23–32. [Google Scholar] [CrossRef]
- Hua, Z.; Wu, C.W.; Fan, G.F.; Tang, Z.; Cao, F. The antibacterial activity and mechanism of ginkgolic acid C15:1. BMC Biotechnol. 2017, 17, 5. [Google Scholar] [CrossRef] [Green Version]
- Kubo, I.; Muroi, H.; Himejima, M.; Yamagiwa, Y.; Mera, H.; Tokushima, K.; Ohta, S.; Kamikawa, T. Structure-antibacterial activity relationships of anacardic acids. J. Agric. Food Chem. 1993, 41, 1016–1019. [Google Scholar] [CrossRef]
- Rivero-Cruz, B.E.; Esturau, N.; Sánchez-Nieto, S.; Romero, I.; Castillo-Juárez, I.; Rivero-Cruz, J.F. Isolation of the new anacardic acid 6-[16′Z-nonadecenyl]-salicylic acid and evaluation of its antimicrobial activity against Streptococcus mutans and Porphyromonas gingivalis. Nat. Prod. Res. 2011, 25, 1282–1287. [Google Scholar] [CrossRef] [PubMed]
- Kubo, J.; Ran Lee, J.; Kubo, I. Anti-Helicobacter pylori agents from the Cashew Apple. J. Agric. Food Chem. 1999, 47, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Das, K.; Tiwari, R.K.; Shrivastava, D.K. Techniques for evaluation of medicinal plant products as antimicrobial agent: Current methods and future trends. J. Med. Plants Res. 2010, 4, 104–111. [Google Scholar] [CrossRef]
- Coates, N.J.; Gilpin, M.L.; Gwynn, M.N.; Lewis, D.E.; Milner, P.H.; Spear, S.R.; Tyler, J.W. SB-202742, a novel β-lactamase inhibitor isolated from Spondias mombin. J. Nat. Prod. 1994, 57, 654–657. [Google Scholar] [CrossRef]
- Lie Ken Jie, M.S.; Pasha, M.K.; Alam, M.S. Synthesis and nuclear magnetic resonance properties of all geometrical isomers of conjugated linoleic acids. Lipids 1997, 32, 1041–1044. [Google Scholar] [CrossRef]
- Silverstein, R. Spectrometric Identification of Organic Compounds, 7th ed.; John Wiley & Sons: Chichester, UK; Hoboken, NJ, USA, 2005. [Google Scholar]
- Mansour, M.P.; Holdsworth, D.G.; Forbes, S.E.; Macleod, C.K.; Volkman, J.K. High contents of 24:6(n 3) and 20:1(n 13) fatty acids in the brittle star Amphiura elandiformis from Tasmanian coastal sediments. Biochem. Syst. Ecol. 2005, 33, 659–674. [Google Scholar] [CrossRef]
- Choi, Y.H.; Choi, H.K.; Peltenburg Looman, A.M.G.; Lefeber, A.W.; Verpoorte, R. Quantitative analysis of ginkgolic acids from Ginkgo leaves and products using 1H-NMR. Phytochem. Anal. 2004, 15, 325–330. [Google Scholar] [CrossRef]
- Spencer, G.F.; Tjarks, L.W.; Kleiman, R. Alkyl and phenylalkyl anacardic acids from Knema elegans seed oil. J. Nat. Prod. 1980, 43, 724–730. [Google Scholar] [CrossRef]
- Gellerman, J.L.; Schlenk, H. Methods for isolation and determination of anacardic acids. Anal. Chem. 1968, 40, 739–743. [Google Scholar] [CrossRef]
- Walters, D.S.; Minard, R.; Craig, R.; Mumma, R.O. Geranium defensive agents. III. Structural determination and biosynthetic considerations of anacardic acids of geranium. J. Chem. Ecol. 1987, 14, 743–751. [Google Scholar] [CrossRef]
- Corthout, J.; Pieters, L.; Claeys, M.; Berghe, D.V.; Vlietinck, A. Antibacterial and molluscicidal phenolic acids from Spondias mombin. Planta Med. 1994, 60, 460–463. [Google Scholar] [CrossRef] [PubMed]
- Tahrioui, A.; Ortiz, S.; Azuama, O.C.; Bouffartigues, E.; Benalia, N.; Tortuel, D.; Maillot, O.; Chemat, S.; Kritsanida, M.; Feuilloley, M.; et al. Membrane-interactive compounds from Pistacia lentiscus L. thwart Pseudomonas aeruginosa virulence. Front. Microbiol. 2020, 11, 1068. [Google Scholar] [CrossRef] [PubMed]
- Grand, S. Tahu’a, Tohunga, Kahuna: Le Monde Polynésien des Soins Traditionnels; Au vent des îles Pirae: Papeete, French Polynesia, 2007. [Google Scholar]
- Hooper, A. Tahitian Folk Medicine. In Rank and Status in Polynesia and Melanesia; Douglas, O., Ed.; Société des Océanistes: Paris, France, 1978; pp. 61–80. [Google Scholar]
- Pétard, P. Raau Tahiti: Plantes Médicinales Polynésiennes et Remèdes Tahitiens; Commission du Pacifique Sud: Noumea, France, 1972. [Google Scholar]
- Hemshekhar, M.; Sebastin Santhosh, M.; Kemparaju, K.; Girish, K.S. Emerging roles of anacardic acid and its derivatives: A pharmacological overview. Basic Clin. Pharmacol. Toxicol. 2012, 110, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Marks, J.G.; DeMelfi, T.; McCarthy, M.A.; Witte, E.J.; Castagnoli, N.; Epstein, W.L.; Aber, R.C. Dermatitis from cashew nuts. J. Am. Acad. Dermatol. 1984, 10, 627–631. [Google Scholar] [CrossRef]
- Qian, Y.; Yunru, P.; Erxin, S.; Ming, Z.; Liang, Y.; Zhenhua, Z.; Jinhua, T.; Shulan, S.; Sheng, G.; Jin-Ao, D. Metabolic profiling of the hepatotoxicity and nephrotoxicity of ginkgolic acids in rats using ultra-performance liquid chromatography-high-definition mass spectrometry. Chem. Biol. Interact. 2017, 273, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Florence, J.; Moretti, C. Les Ressources Végétales Polynésiennes, Substances Naturelles en Polynésie Française: Stratégies de Valorisation, Expertise Collégiale; IRD Éditions: Marseille, France, 2013; pp. 3–36. [Google Scholar]
- Florence, J.; Chevillotte, H.; Ollier, C.; Meyer, J.Y. Base de Données Botaniques Nadeaud de l’Herbier de la Polynésie Française (PAP). Institut Louis Malardé; Délégation à la Recherche: Papeete, French Polynesia, 2019; Available online: http://nadeaud.ilm.pf (accessed on 3 March 2022).
- Girardi, C.; Butaud, J.F.; Ollier, C.; Ingert, N.; Weniger, B.; Raharivelomanana, P.; Moretti, C. Herbal medicine in the Marquesas Islands. J. Ethnopharmacol. 2015, 161, 200–213. [Google Scholar] [CrossRef]
- Cos, P.; Vlietinck, A.J.; Berghe, D.V.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro “proof-of-concept. ” J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef]
- Ríos, J.L.; Recio, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol. 2005, 100, 80–84. [Google Scholar] [CrossRef]
- Abedini, A.; Roumy, V.; Mahieux, S.; Biabiany, M.; Standaert-Vitse, A.; Rivière, C.; Sahpaz, S.; Bailleul, F.; Neut, C.; Hennebelle, T. Rosmarinic acid and its methyl ester as antimicrobial components of the hydromethanolic extract of Hyptis atrorubens Poit. (Lamiaceae). Evid.-Based Complement. Altern. Med. 2013, 2013, 604536. [Google Scholar] [CrossRef] [Green Version]
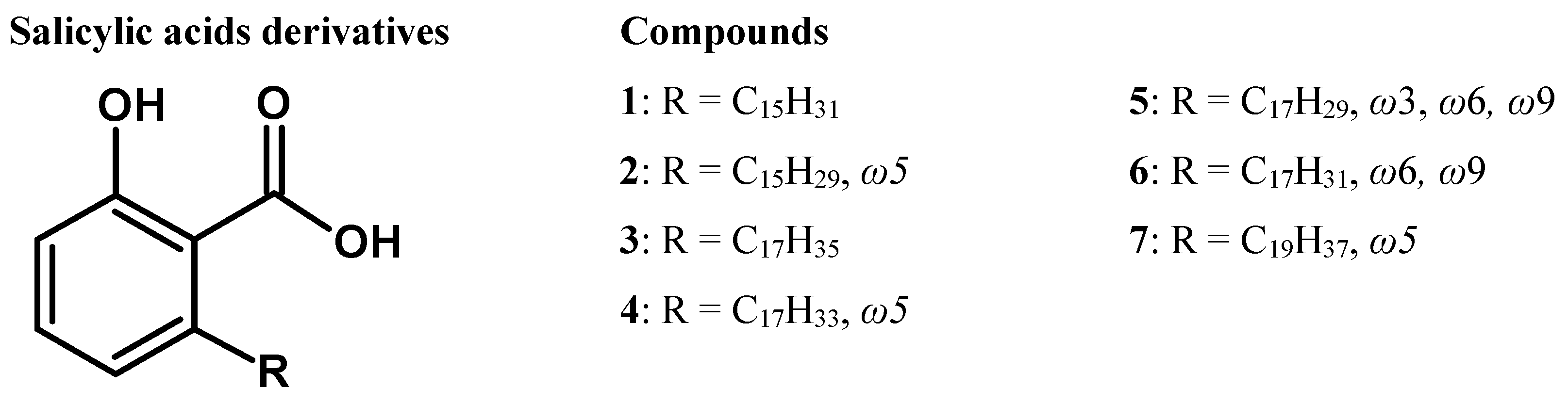
| Latin Name “Vernacular Name” (Botanical Family) (Number of Citation) | Health Problem Category (Part Used; Mode of Administration) |
|---|---|
| Adenostemma viscosum J.R.Forst. & G. Forst. “vaianu” (Asteraceae) (5) | Pneumo. (l.; Po), Uterus (l.; Po), Cystitis (l.; Po), V. Cutaneous symptoms (w.p.; Po), V. diseases (l.; Po and Cat), Wound, Abscess (l.; Po) |
| Aleurites moluccanus (L.) Willd. “ti’a’iri” (Euphorbiaceae) (9) | Aphta (bk.; Po), Cystitis (bk.; Po), Furunculous, Pustule (alm.; Bath), STD (bk.; Po), Oral mycosis (bk.; Garg and Po), Tonsillitis (bk. Garg or Po and Cat.), Wound, Abscess (bk.; Po) |
| Calophyllum inophyllum L. “tamanu” (Calophyllaceae) (8) | Cicatrising (alm. (oil); Cat), Cystitis (bk.; Po), Mycosis (l.; Bath), V. Cutaneous symptoms (l.; Bath), Wound, Abscess (alm. (oil); Cat) (bk.; Po) |
| Cenchrus echinatus L. “piripiri” (Poaceae) (7) | Furunculous, Pustule (a.p.; Po), Leucorrhoea (a.p.; Po), Sinusitis, Rhinitis (a.p.; Fumigation), Wound, Abscess (a.p.; Cat) |
| Centotheca lappacea (L.) Desv. “’ofe’ofe” (Poaceae) (7) | Furunculous, Pustule (a.p.; Po), V. diseases (a.p.; Po and Cat), Wound, Abscess (a.p.; Cat) |
| Citrus× aurantiifolia (Christm.) Swingle “taporo” (Rutaceae) (13) | Pneumo. (f. juice or l.; Po), Uterus, (f. juice; Po), Cystitis (f. juice or l.; Po), Diarrhoea (f. juice; Po or Po and Cat.), STD (f. juice; Po), Heartburn (f. juice; Po), Leucorrhoea (f. juice or l.; Po), Oral mycosis (f. juice; Po), Tonsillitis (f. juice; Po), V. Cutaneous symptoms (f. juice; Cat), Vulvitis (f. juice; Po), Wound, Abscess (f. juice; Cat) |
| Commelina diffusa Burm.f. “ma’a pape” (Commelinaceae) (6) | Wound, Abscess (l. and st.; Cat) |
| Cordia subcordata Lam. “tou” (Boraginaceae) (12) | Acne (l.; ND), Pneumo. (bk. or l.; Po), Conjunctivitis (l.; In eye), Cystitis (bk. or l.; Po), Furunculous, Pustule (bk.; Po), STD (l.; Po), Leucorrhoea (bk.; Po), Oral mycosis (bk. orl.; Po), Sinusitis, Rhinitis (l.; Po), V. Cutaneous symptoms (l.; Cat), Wound, Abscess (bk.; Po) |
| Cordyline fruticosa (L.) A.Chev. “‘auti” (Asparagaceae) (5) | Diarrhoea (b.; Po) (l.; Po and Cat), Heartburn (l.; Po), Otitis (b.; Cat), V. Cutaneous symptoms (b.; Cat) |
| Curcuma longa L. “re’a ma’a” (Zingiberaceae) (15) | Pneumo. (rh.; Po), Uterus (rh.; Po), Cystitis (rh.; Po, genital bath), Furunculous, Pustule (rh.; Po), STD (rh.; Po genital bath) Oral mycosis (rh.; Po), Vaginal Mycosis (rh.; Po and Bath), V. Cutaneous symptoms (rh.; Po), Vulvitis (rh.; Po) |
| Davallia solida (G. Forst.) Sw. var. solida “titi” (Davalliaceae) (9) | “Crumpled lung” (rh.; Po), Pneumo. (Frond or rh.; Po), Cystitis (rh.; Po), Heartburn (rh.; Po), Leucorrhoea (rh.;Po), Tonsillitis (rh.; Po), V. Cutaneous symptoms (rh.; Cat) |
| Fagraea berteroana A.Gray ex Benth. “pua” (Gentianaceae) (6) | Itching, Allergy (fl.; Bath) |
| Ficus prolixa G.Forst. “ ‘ora” (Moraceae) Ind. (6) | Pneumo. (a.r.; Po), STD (a.r.; Po), Ringworm (l.; Bath), Tonsillitis (a.r. Garg), V. disease (a.r.; Po and Cat) |
| Ficus tinctoria G.Forst. subsp. tinctoria “mati” (Moraceae) (5) | Acne (l.f.; ND), Uterus (l.f.; Po), Cystitis (l.f.; Po), Furunculous, Pustule (l.f.; Po), Oral mycosis (l.f.; Po) |
| Hibiscus tiliaceus L. subsp. tiliaceus “purau” (Malvaceae) (5) | Eye irritation (r.; In eye), V. Cutaneous symptoms (bk. Bath), Wound; Abscess (fl.; Bath) |
| Mangifera indica L. “vi popa’a” (Anacardiaceae) (5) | Pneumo. (f.; Po), Furunculous, Pustule (f.; Po and Cat), Tonsillitis (f.; Garg and Po), Wound, Abscess (f.; Po and Cat) |
| Microsorum grossum (Langsd. & Fisch.) S.B.Andrews “metuapua’a” (Polypodiaceae) (10) | “Crumpled lung” (rh.; Po), Pneumo. (rh.; Po), Cystitis (rh.; Po), Diarrhoea (Frond: Po), Furunculous, Pustule (rh.; Po), Heartburn (rh.; Po), Leucorrhoea (rh.; Po), Tonsillitis (rh.; Po), V. Cutaneous symptoms (rh.; Cat) |
| Musa troglodytarum L. “fe’i” (Musaceae) (5) | V. Cutaneous symptoms (f.;Cat), Wound; Abscess (b.; Cat) |
| Musa× paradisiaca L. “mei’a hamoa” or “mei’a rio” (Musaceae) (5) | Wound, Abscess (b. or f.; Cat) |
| Pandanus tectorius Parkinson var. tectorius “fara” (Pandanaceae) (5) | Oral mycosis (a.r.; Po) |
| Paspalum vaginatum Sw. “matie tatahi” (Poaceae) Ind. (8) | Aqueous pustule (a.p.; Cat), Pneumo. (a.p.; Po), Furunculous, Pustule (a.p.; Po), V. Cutaneous symptoms (a.p.; Bath), V. Diseases (a.p.; Po and Cat) |
| Phyllanthus amarus Schumach. & Thonn. “moemoe” (Phyllanthaceae) (10) | Furunculous, Pustule (w.p.; Bath), Otitis (l.; In ear) (w.p. Cat), V. Cutaneous symptoms (r.; Cat) |
| Psidium guajava L. “tuvava” (Myrtaceae) (6) | Pneumo. (l.; Po), Cystitis (l.; Po) STD (l.; Po), Otitis (f.; In ear) |
| Rorippa sarmentosa (Sol. ex G.Forst. ex DC.) J.F.Macbr. “moahau’a’ino” (Brassicaceae) (11) | Pneumo. (w.p.; Po), Uterus (l.; Po), Cicatrasing (w.p.; Cat), Constipation (w.p.; Po), Cystitis (r. or w.p.; Po), Furunculous, Pustule (w.p.; Cat), Heartburn (l.; Po), Oral mycosis (l. Garg and Po) (r.; Po), Purge (w.p.; Po), Tonsillitis (l. or w.p.; Garg and Po or Po) (r.; Garg) |
| Syzygium cumini (L.) Skeels “pistas” (Myrtaceae) (5) | Mycosis (bk.; Cat), V. Cutaneous symptoms (bk.; Cat) |
| Syzygium malaccense (L.) Merr. & L.M.Perry “ ‘ahi’a” (Myrtaceae) (12) | Pneumo. (l.; Po), Uterus (l.; Po), Cystitis (bk. or l.; Po), Furunculous, Pustule (bk. or l.; Po), STD (l.; Po), Oral mycosis (l.; Garg or Po), Tonsillitis (l.; Garg and Po), Vaginal Mycosis (l.; Po and Bath), Vulvitis (l.; Po) |
| Thespesia populnea (L.) Sol. ex Corrêa “miro” (Malvaceae) (9) | Pneumo. (Latex of f.; Po), Conjunctivitis (alm.; Po), Cystitis (bk.; Po), Furunculous, Pustule (alm.; Po) (Latex of f.; Bath), Leucorrhoea (bk. or l.; Po), Mycosis (bk.; Bath), Varicella (Latex of f.; Bath), Wound, Abscess (bk.; Po) |
| Torenia crustacea (L.) Cham. & Schltdl “piri’ate” (Linderniaceae) (6) | Pneumo. (a.p.; Po), Uterus (a.p. or l. Po), Constipation (a.p.; Po), Cystitis (l.; Po), Heartburn (l.; Po), Sinusitis, Rhinitis (l.; Po), V. diseases (a.p.; Po and Cat) |
| Usnea spp. “remu ha’ari” (Parmeliaceae, lichen) (5) | “Crumpled lung” (w.p.; Po), Pneumo. (w.p.; Po), Constipation (w.p.; Po), Cystitis (Lichen; Po) |
| Vigna marina (Burm.) Merr. “pipi” (Fabaceae) (5) | Burn (l.; Bath), Diarrhoea (l.; Po) |
| Malvaceae | Myrtaceae | Calophyllaceae | Boraginaceae | Anacardiaceae | Parmeliaceae | Euphorbiaceae | Pandanaceae | Polypodiaceae | Linderniaceae | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hibiscus tiliaceus | Thespesia populnea | Syzygium cumini | Psidium guajava | Calophyllum inophyllum | Cordia subcordata | Mangifera indica | Usnea spp. | Aleurites moluccanus | Pandanus tectorius var. tectorius | Microsorum grossum | Torenia crustacea | ||||||
| Microorganisms Tested | Bark | Bark | Leaf | Bark | Leaf | Bark | Leaf | Bark | Leaf | Lichen | Bark | Aerial Roots | Rhizome | Aerial Part | |||
| Yeast | Candida albicans | 10286 | 1.2 | 0.6 | 1.2 | 0.15 | 0.6 | 0.6 | 1.2 | 0.6 | 1.2 | 1.2 | - | 1.2 | - | 0.15 | |
| Gram + | Enterococcus faecalis | C159-6 | - | 0.3 | - | - | 0.6 | 0.6 | - | 1.2 | - | 0.6 | - | - | - | - | |
| Enterococcus sp. | 8153 | - | 0.3 | - | - | - | 1.2 | - | - | - | 0.6 | - | - | - | - | ||
| Staphylococcus aureus | 8146 | - | 0.3 | 1.2 | 0.6 | 0.6 | 0.15 | 0.6 | 0.6 | 1.2 | 0.6 | - | - | - | - | ||
| Staphylococcus aureus | 8241 | - | 0.3 | 1.2 | 0.6 | 1.2 | 0.07 | 0.6 | 0.6 | 1.2 | 0.6 | - | - | 1.2 | - | ||
| Staphylococcus aureus | ATCC 6538 | 0.6 | 0.15 | 0.6 | 0.3 | 0.6 | 0.07 | 0.6 | 0.6 | 1.2 | 0.6 | - | 1.2 | 1.2 | - | ||
| Staphylococcus aureus | T28-1 | - | 0.3 | 0.6 | 0.3 | 0.3 | 0.07 | 0.3 | 0.6 | 1.2 | 0.6 | - | - | 1.2 | - | ||
| Staphylococcus aureus | T17-4 | - | 0.3 | 1.2 | 0.3 | 0.6 | 0.07 | 0.6 | 0.6 | 1.2 | 0.6 | - | 1.2 | 1.2 | - | ||
| Staphylococcus epidermidis | T46A1 | 0.6 | 0.15 | 0.6 | 0.3 | 0.3 | 0.15 | 0.6 | 0.3 | 1.2 | 0.6 | 0.6 | - | 1.2 | 0.6 | ||
| Staphylococcus epidermidis | T19A1 | 0.6 | 0.15 | 0.6 | 0.3 | 0.3 | 0.15 | 0.6 | 0.15 | 0.6 | 0.6 | 0.6 | - | 1.2 | 0.6 | ||
| Staphylococcus epidermidis | T21A5 | 1.2 | 0.3 | 0.6 | 0.3 | 0.3 | 0.15 | 0.6 | 0.15 | 0.6 | 0.6 | 1.2 | - | 1.2 | 0.6 | ||
| Staphylococcus warneri | T12A12 | 1.2 | 0.6 | - | 0.3 | 0.6 | 0.3 | - | 0.3 | 1.2 | 1.2 | - | - | 1.2 | - | ||
| Staphylococcus warneri | T26A1 | 1.2 | 1.2 | - | 0.3 | 0.6 | 0.3 | - | 0.3 | 0.6 | - | 1.2 | - | 1.2 | - | ||
| Staphylococcus pettenkoferi | T47.A6 | 1.2 | 0.3 | - | 0.3 | 0.6 | 0.6 | - | 0.6 | 0.6 | 1.2 | 1.2 | - | 1.2 | - | ||
| Streptococcus agalactiae | T38.2 | - | 0.15 | 1.2 | - | 0.6 | 0.15 | 0.6 | - | 1.2 | 0.6 | - | 1.2 | - | 0.3 | ||
| Streptococcus agalactiae | T53C9 | - | 0.15 | 1.2 | - | 0.6 | 0.15 | 0.6 | 0.6 | - | 0.6 | - | 1.2 | - | 0.6 | ||
| Corynebacterium striatum | T40A3 | 1.2 | 0.6 | 0.6 | 0.15 | 0.3 | 0.15 | 0.3 | 0.6 | 1.2 | 0.6 | 1.2 | - | 0.6 | - | ||
| Gram − | Citrobacter freundii | 11041 | - | - | - | - | - | - | - | 1.2 | - | - | - | - | - | - | |
| Citrobacter freundii | 10268 | 1.2 | 1.2 | - | 0.6 | - | - | - | 1.2 | - | - | - | - | - | - | ||
| Proteus mirabilis | 11060 | - | - | - | 0.6 | - | - | - | 1.2 | - | - | - | - | - | - | ||
| Proteus mirabilis | T28-3 | - | - | - | 0.6 | - | - | - | 1.2 | - | - | - | - | - | - | ||
| Pseudomonas aeruginosa | 8131 | - | - | - | 0.6 | 0.6 | - | - | 1.2 | - | 1.2 | 1.2 | - | - | - | ||
| Pseudomonas aeruginosa | ATCC 27583 | 0.6 | 1.2 | - | 0.15 | 0.6 | - | - | 0.3 | 1.2 | 0.6 | 0.6 | - | - | 0.6 | ||
| Pseudomonas aeruginosa | 8129 | 0.6 | 1.2 | 1.2 | 0.3 | 0.6 | 1.2 | 1.2 | 0.3 | 1.2 | 1.2 | 1.2 | 1.2 | - | 0.6 | ||
 MIC: 0.6 mg/mL,
MIC: 0.6 mg/mL,  MIC: 0.6 mg/mL ≥ MIC > 0.3 mg/mL,
MIC: 0.6 mg/mL ≥ MIC > 0.3 mg/mL,  MIC ≤ 0.3 mg/mL; (-) no MIC or FIC determined at 1.2 mg/mL; (n = 3; with standard deviation for value: 1.2 ± 0.4 mg/mL; 0.6 ± 0.2 mg/mL; 0.3 ± 0.1 mg/mL; 0.15 ± 0.05 mg/mL; 0.07 ± 0.03 mg/mL).
MIC ≤ 0.3 mg/mL; (-) no MIC or FIC determined at 1.2 mg/mL; (n = 3; with standard deviation for value: 1.2 ± 0.4 mg/mL; 0.6 ± 0.2 mg/mL; 0.3 ± 0.1 mg/mL; 0.15 ± 0.05 mg/mL; 0.07 ± 0.03 mg/mL).| Tested Microorganisms | Syzygium malaccense, MIC (mg/mL) | Curcuma longa, MIC (mg/mL) | FIC Index | Effect | ||
|---|---|---|---|---|---|---|
| Yeast | Candida albicans | 10286 | 0.15 | 0.15 | 1.3 | Indifferent |
| Gram + | Enterococcus faecalis | C159-6 | 1.2 | 0.15 | 0.9 | Additive |
| Staphylococcus aureus | 8146 | 1.2 | 0.3 | 0.8 | Additive | |
| Staphylococcus aureus | 8241 | 0.6 | 0.3 | 0.8 | Additive | |
| Staphylococcus aureus | ATCC 6538 | - | 0.3 | 0.7 | Additive | |
| Staphylococcus aureus | T28-1 | 0.6 | 0.3 | 0.7 | Additive | |
| Staphylococcus aureus | T17-4 | 0.6 | 0.3 | 0.9 | Additive | |
| Staphylococcus epidermidis | T46A1 | 0.15 | 0.15 | 1.1 | Indifferent | |
| Staphylococcus epidermidis | T19A1 | 0.15 | 0.07 | 1.3 | Indifferent | |
| Staphylococcus epidermidis | T21A5 | 0.15 | 0.07 | 1.2 | Indifferent | |
| Staphylococcus warneri | T12A12 | 0.6 | 0.15 | 1.9 | Indifferent | |
| Staphylococcus warneri | T26A1 | 0.6 | 0.6 | 1.5 | Indifferent | |
| Staphylococcus pettenkoferi | T47.A6 | 0.6 | 0.6 | 1.5 | Indifferent | |
| Streptococcus agalactiae | T53C9 | 1.2 | 0.15 | 1.3 | Indifferent | |
| Corynebacterium striatum | T40A3 | 0.6 | 0.15 | 0.9 | Additive | |
| Streptococcus pyogenes | 16138 | 0.3 | - | - | - | |
| Streptococcus pyogenes | 19140 | 0.15 | - | - | - | |
| Gram − | Proteus mirabilis | 11060 | 1.2 | - | - | - |
| Proteus mirabilis | T28-3 | 0.6 | - | - | - | |
| Pseudomonas aeruginosa | 8131 | 1.2 | - | - | - | |
| Pseudomonas aeruginosa | ATCC 27583 | 0.15 | - | - | - | |
| Pseudomonas aeruginosa | 8129 | 0.15 | 0.6 | 0.5 | Synergic | |
 MIC: 0.6 mg/mL,
MIC: 0.6 mg/mL,  MIC: 0.6 mg/mL ≥ MIC > 0.3: mg/mL,
MIC: 0.6 mg/mL ≥ MIC > 0.3: mg/mL,  MIC ≤ 0.3 mg/mL; (-) no MIC or FIC determined at 1.2 mg/mL; (n = 3; with standard deviation for value: 1.2 ± 0.4 mg/mL; 0.6 ± 0.2 mg/mL; 0.3 ± 0.1 mg/mL; 0.15 ± 0.05 mg/mL; 0.07 ± 0.03 mg/mL) with FIC index of mixture.
MIC ≤ 0.3 mg/mL; (-) no MIC or FIC determined at 1.2 mg/mL; (n = 3; with standard deviation for value: 1.2 ± 0.4 mg/mL; 0.6 ± 0.2 mg/mL; 0.3 ± 0.1 mg/mL; 0.15 ± 0.05 mg/mL; 0.07 ± 0.03 mg/mL) with FIC index of mixture.| Comp | 1 | 2 | 3 | 4 | 5 | 6 | 7 | |
|---|---|---|---|---|---|---|---|---|
| C (H) | ||||||||
| 1 | 110.5 | 110.7 | 110.5 | 110.7 | 110.7 | 110.7 | 110.5 | |
| 2 | 163.8 | 163.8 | 163.8 | 163.5 | 163.5 | 163.5 | 163.5 | |
| 3 | 116.4 (6.89, d:8.4 Hz) | 116.4 (6.89, d:8.4 Hz) | 116.4 (6.89, d:8.2 Hz) | 115.7 (6.89, d:8.4 Hz) | 115.7 (6.88, d:8.4 Hz) | 115.7 (6.88, d:8.2 Hz) | 115.8 (6.90, d:7.7 Hz) | |
| 4 | 135.4 (7.38, d:7.3 Hz) | 135.4 (7.38, dd:7.3, 8.4 Hz) | 135.4 (7.38, dd:7.5, 8.2 Hz) | 135.4 (7.38, dd:7.8, 8.4 Hz) | 135.0 (7.38, dd:7.8, 8.4 Hz) | 135.0 (7.38, dd:7.8, 8.2 Hz) | 135.4 (7.39, dd:7.7, 8.0 Hz) | |
| 5 | 122.8 (6.80, d:7.3 Hz) | 122.8 (6.80, d:7.3 Hz) | 122.8 (6.80, d:7.5 Hz) | 122.8 (6.80, d:7.8 Hz) | 122.8 (6.79, d:7.8 Hz) | 122.8 (6.79, d:7.8 Hz) | 122.7 (6.81, d:8.0 Hz) | |
| 6 | 148.0 | 148.0 | 148.0 | 147.5 | 147.5 | 147.5 | 147.8 | |
| 1′ | 36.2 (3.00, t:6.9 Hz) | 36.2 (3.00, t:6.4 Hz) | 36.2 (3.00, t:6.7 Hz) | 36.5 (3.00, t:6.4 Hz) | 36.5 (3.00, t:6.4 Hz) | 36.5 (3.00, t:7.8 Hz) | 36.5 (3.0, m) | |
| 2′ | 31.9 (1.60, m) | 31.9 (1.60, m) | 31.9 (1.60, m) | 31.9 (1.60, m) | 32.3 (1.60, m) | 31.5 (1.60, m) | 31.9 (1.63, m) | |
| 3′–6′ | 29.0–29.7 (1.20–1.40, m) | 29.0–29.7 (1.20–1.40, m) | 29.0–29.7 (1.20–1.40, m) | 29.0–29.8 (1.20–1.40, m) | 29.3–29.7 (1.20–1.40, m) | 29.0–29.7 (1.20–1.40, m) | 29.3–29.8 (1.20–1.40, m) | |
| 7′ | “ | “ | “ | “ | 27.3 (2.09, m) | 27.2 (2.07, m) | “ | |
| 8′ | “ | “ | “ | “ | 127.2–132.5 (5.38, m) | 127.9–130.2 (5.38, m) | “ | |
| 9′ | “ | 27.3 (2.03, m) | “ | “ | 127.2–132.5 (5.38, m) | 127.9–130.2 (5.38, m) | “ | |
| 10′ | “ | 130.3 (5.38, m) | “ | “ | 25.5 (2.82, m) | 25.5 (2.82, t:6.7) | “ | |
| 11′ | “ | 130.3 (5.38, m) | “ | 27.2 (2.09, m) | 127.2–132.5 (5.38, m) | 127.9–130.2 (5.38, m) | “ | |
| 12′ | “ | 27.2 (2.03, m) | “ | 129.9 (5.36, m) | 127.2–132.5 (5.38, m) | 127.9–130.2 (5.38, m) | “ | |
| 13′ | 32.2 (1.20–1.40, m) | 32.2 (1.20–1.40, m) | “ | 129.9 (5.36, m) | 25.6 (2.82, m) | 27.2 (2.07, m) | 27.9 (2.04, m) | |
| 14′ | 22.7 (1.20–1.40, m) | 22.7 (1.20–1.40, m) | “ | 26.9 (1.20–1.40, m) | 127.2–132.5 (5.38, m) | 29.0-29.7 (1.20–1.40, m) | 129.8 (5.38, m) | |
| 15′ | 14.4 (0.90, t:6.9 Hz) | 14.4 (0.91, t:7.6 Hz) | 32.2 (1.20–1.40, m) | 32.3 (1.20–1.40, m) | 127.2-132.5 (5.38, m) | 32.3 (1.20–1.40, m) | 129.8 (5.38, m) | |
| 16′ | - | - | 22.7 (1.20–1.40, m) | 22.6 (1.20–1.40, m) | 20.6 (2.09, m) | 22.6 (1.20–1.40, m) | 27.7 (2.04, m) | |
| 17′ | - | - | 14.1 (0.90, t:6.9 Hz) | 14.0 (0.90, t:6.9 Hz) | 14.7 (0.99, t:7.6) | 14.3 (0.89, t:6.7 Hz) | 32.0 (1.20–1.40, m) | |
| 18′ | - | - | - | - | - | - | 22.3 (1.20–1.40, m) | |
| 19′ | - | - | - | - | - | - | 14.0 (0.93, t:6.8 Hz) | |
| COOH | 175.0 | 175.7 | 175.0 | 175.7 | 175.7 | 175.7 | 176.4 | |
| MIC (µg/mL) | ||||
|---|---|---|---|---|
| Compounds | Aliphatic Chain | Staphylococcus aureus | Streptococcus pyogenes | Pseudomonas aeruginosa |
| 1 | C15:0 | 75.0 | 9.37 | 150 |
| 2 | C15:1, ω5 | 18.75 | 9.37 | 150 |
| 3 | C17:0 | 37.5 | 18.75 | 150 |
| 4 | C17:1, ω5 | 18.75 | 9.37 | 150 |
| 5 | C17:3, ω3; ω6; ω9 | 18.75 | 2.34 | 150 |
| 6 | C17:2, ω6; ω9 | 18.75 | 4.68 | 150 |
| 7 | C19:1, ω5 | 18.75 | 9.37 | 150 |
| Gentamycin | 0.5 | 0.125 | 0.03 | |
| Vancomycin | 1.0 | 0.25 | NA | |
| Amoxicillin | 4.0 | 0.03 | NA | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quenon, C.; Hennebelle, T.; Butaud, J.-F.; Ho, R.; Samaillie, J.; Neut, C.; Lehartel, T.; Rivière, C.; Siah, A.; Bonneau, N.; et al. Antimicrobial Properties of Compounds Isolated from Syzygium malaccense (L.) Merr. and L.M. Perry and Medicinal Plants Used in French Polynesia. Life 2022, 12, 733. https://doi.org/10.3390/life12050733
Quenon C, Hennebelle T, Butaud J-F, Ho R, Samaillie J, Neut C, Lehartel T, Rivière C, Siah A, Bonneau N, et al. Antimicrobial Properties of Compounds Isolated from Syzygium malaccense (L.) Merr. and L.M. Perry and Medicinal Plants Used in French Polynesia. Life. 2022; 12(5):733. https://doi.org/10.3390/life12050733
Chicago/Turabian StyleQuenon, Camille, Thierry Hennebelle, Jean-François Butaud, Raimana Ho, Jennifer Samaillie, Christel Neut, Tamatoa Lehartel, Céline Rivière, Ali Siah, Natacha Bonneau, and et al. 2022. "Antimicrobial Properties of Compounds Isolated from Syzygium malaccense (L.) Merr. and L.M. Perry and Medicinal Plants Used in French Polynesia" Life 12, no. 5: 733. https://doi.org/10.3390/life12050733
APA StyleQuenon, C., Hennebelle, T., Butaud, J.-F., Ho, R., Samaillie, J., Neut, C., Lehartel, T., Rivière, C., Siah, A., Bonneau, N., Sahpaz, S., Anthérieu, S., Lebegue, N., Raharivelomanana, P., & Roumy, V. (2022). Antimicrobial Properties of Compounds Isolated from Syzygium malaccense (L.) Merr. and L.M. Perry and Medicinal Plants Used in French Polynesia. Life, 12(5), 733. https://doi.org/10.3390/life12050733







