A Role for Superficial Heat Therapy in the Management of Non-Specific, Mild-to-Moderate Low Back Pain in Current Clinical Practice: A Narrative Review
Abstract
1. Introduction
2. Causes of Low Back Pain
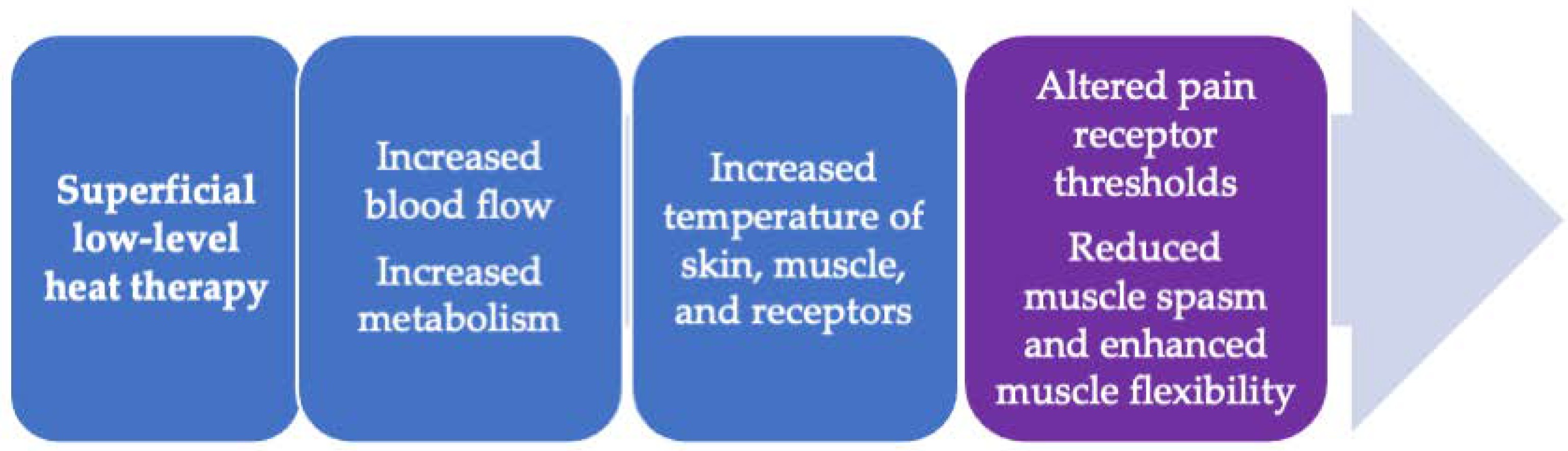
3. Superficial Heat Therapy—How Does It Work?
4. Overview of Superficial Heat Therapy Modalities
5. Evidence of the Effectiveness of Continuous Low-Level Heat Wrap Therapy for Low Back Pain
6. Other Applications for Superficial, Low-Level Heat Therapy
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, A.; March, L.; Zheng, X.; Huang, J.; Wang, X.; Zhao, J.; Blyth, F.M.; Smith, E.; Buchbinder, R.; Hoy, D. Global low back pain prevalence and years lived with disability from 1990 to 2017: Estimates from the Global Burden of Disease Study 2017. Ann. Transl. Med. 2020, 8, 299. [Google Scholar] [CrossRef]
- Fatoye, F.; Gebrye, T.; Odeyemi, I. Real-world incidence and prevalence of low back pain using routinely collected data. Rheumatol. Int. 2019, 39, 619–626. [Google Scholar] [CrossRef]
- Ehrlich, G.E. Low back pain. Bull. World Health Organ. 2003, 81, 671–676. [Google Scholar] [PubMed]
- Nasser, M.J. How to approach the problem of low back pain: An overview. J. Fam. Community Med. 2005, 12, 3–9. [Google Scholar]
- Hoy, D.; March, L.; Brooks, P.; Blyth, F.; Woolf, A.; Bain, C.; Williams, G.; Smith, E.; Vos, T.; Barendregt, J.; et al. The global burden of low back pain: Estimates from the Global Burden of Disease 2010 study. Ann. Rheum. Dis. 2014, 73, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Buchbinder, R.; van Tulder, M.; Oberg, B.; Costa, L.M.; Woolf, A.; Schoene, M.; Croft, P.G. Low Back Pain Series Working Group. Low back pain: A call for action. Lancet 2018, 391, 2384–2388. [Google Scholar] [CrossRef]
- GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Krenn, C.; Horvath, K.; Jeitler, K.; Zipp, C.; Siebenhofer-Kroitzsch, A.; Semlitsch, T. Management of non-specific low back pain in primary care—a systematic overview of recommendations from international evidence-based guidelines. Prim. Health Care Res. Dev. 2020, 21, e64. [Google Scholar] [CrossRef] [PubMed]
- van Tulder, M.W.; Scholten, R.J.; Koes, B.W.; Deyo, R.A. Non-steroidal anti-inflammatory drugs for low back pain. Cochrane Database Syst. Rev. 2000, 2, CD000396. [Google Scholar] [CrossRef]
- Kinkade, S. Evaluation and treatment of acute low back pain. Am. Fam. Physician 2007, 75, 1181–1188. [Google Scholar]
- Coxib and traditional NSAID Trialists’ (CNT) Collaboration; Bhala, N.; Emberson, J.; Merhi, A.; Abramson, S.; Arber, N.; Baron, J.; Bombardier, C.; Cannon, C.; Farkouh, M.; et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. Lancet. 2013, 382, 769–779. [Google Scholar] [CrossRef]
- Gore, M.; Sadosky, A.; Stacey, B.R.; Tai, K.S.; Douglas, L. The burden of chronic low back pain: Clinical comorbidities, treatment patterns, and health care costs in usual care settings. Spine 2012, 37, E668–E677. [Google Scholar] [CrossRef]
- Qaseem, A.; Wilt, T.J.; McLean, M.; Forciea, M.A.; Clinical Guidelines Committee of the American College of Physicians. Noninvasive treatments for acute, subacute, and chronic low back pain: A clinical practice guideline from the American College of Physicians. Ann. Intern. Med. 2017, 166, 514–530. [Google Scholar] [CrossRef]
- Papaioannou, T.G.; Karamanou, M.; Protogerou, A.D.; Tousoulis, D. Heat therapy: An ancient concept re-examined in the era of advanced biomedical technologies. J. Physiol. 2016, 594, 7141–7142. [Google Scholar] [CrossRef]
- Chabal, C.; Dunbar, P.J.; Painter, I.; Young, D.; Chabal, D.C. Properties of thermal analgesia in a human chronic low back pain model. J. Pain Res. 2020, 13, 2083–2092. [Google Scholar] [CrossRef]
- French, S.D.; Cameron, M.; Walker, B.F.; Reggars, J.W.; Esterman, A.J. Superficial heat or cold for low back pain. Cochrane Database Syst. Rev. 2006, CD004750. [Google Scholar] [CrossRef]
- Malanga, G.A.; Yan, N.; Stark, J. Mechanisms and efficacy of heat and cold therapies for musculoskeletal injury. Postgrad. Med. 2015, 127, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Nadler, S.F.; Weingand, K.; Kruse, R.J. The physiologic basis and clinical applications of cryotherapy and thermotherapy for the pain practitioner. Pain Physician 2004, 7, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Hsu, J.R.; Mir, H.; Wally, M.K.; Seymour, R.B.; Orthopaedic Trauma Association Musculoskeletal Pain Task Force. Clinical practice guidelines for pain management in acute musculoskeletal injury. J. Orthop. Trauma 2019, 33, e158–e182. [Google Scholar] [CrossRef]
- Perl, E.R. Cutaneous polymodal receptors: Characteristics and plasticity. Prog. Brain Res. 1996, 113, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Mense, S.; Gerwin, R.D. Functional anatomy of muscle: Muscle, nociceptors and afferent fibers. In Muscle Pain: Understanding the Mechanisms; Springer: Heidelberg, Germany; Dordrecht, The Netherlands; London, UK; New York, NY, USA, 2010; pp. 17–48. [Google Scholar] [CrossRef]
- Pace, M.C.; Passavanti, M.B.; De Nardis, L.; Bosco, F.; Sansone, P.; Pota, V.; Barbarisi, M.; Palagiano, A.; Iannotti, F.A.; Panza, E.; et al. Nociceptor plasticity: A closer look. J. Cell. Physiol. 2018, 233, 2824–2838. [Google Scholar] [CrossRef]
- Schomburg, E.D.; Jankowska, E.; Fernstrom, K.W. Nociceptive input to spinal interneurones in reflex pathways from group II muscle afferents in cats. Neurosci. Res. 2000, 38, 447–450. [Google Scholar] [CrossRef]
- Ueno, M.; Nakamura, Y.; Li, J.; Gu, Z.; Niehaus, J.; Maezawa, M.; Crone, S.A.; Goulding, M.; Baccei, M.L.; Yoshida, Y. Corticospinal circuits from the sensory and motor cortices differentially regulate skilled movements through distinct spinal interneurons. Cell Rep. 2018, 23, 1286–1300.e7. [Google Scholar] [CrossRef] [PubMed]
- Graham, B.A.; Hughes, D.I. Defining populations of dorsal horn interneurons. Pain. 2020, 161, 2434–2436. [Google Scholar] [CrossRef]
- Hughes, D.I.; Todd, A.J. Central nervous system targets: Inhibitory interneurons in the spinal cord. Neurotherapeutics 2020, 17, 874–885. [Google Scholar] [CrossRef]
- Tashima, R.; Koga, K.; Yoshikawa, Y.; Sekine, M.; Watanabe, M.; Tozaki-Saitoh, H.; Furue, H.; Yasaka, T.; Tsuda, M. A subset of spinal dorsal horn interneurons crucial for gating touch-evoked pain-like behavior. Proc. Natl. Acad. Sci. USA 2021, 118, e2021220118. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.; Laymon, M.; Lee, H. Local heating of trigger points reduces neck and plantar fascia pain. J. Back Musculoskelet. Rehabil. 2020, 33, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Freiwald, J.; Hoppe, M.W.; Beermann, W.; Krajewski, J.; Baumgart, C. Effects of supplemental heat therapy in multimodal treated chronic low back pain patients on strength and flexibility. Clin. Biomech. 2018, 57, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Hartvigsen, J.; Hancock, M.J.; Kongsted, A.; Louw, Q.; Ferreira, M.L.; Genevay, S.; Hoy, D.; Karppinen, J.; Pransky, G.; Sieper, J.; et al. Lancet Low Back Pain Series Working Group. What low back pain is and why we need to pay attention. Lancet 2018, 391, 2356–2367. [Google Scholar] [CrossRef]
- Liang, C.Z.; Li, H.; Tao, Y.Q.; Zhou, X.P.; Yang, Z.R.; Li, F.C.; Chen, Q.X. The relationship between low pH in intervertebral discs and low back pain: A systematic review. Arch. Med. Sci. 2012, 8, 952–956. [Google Scholar] [CrossRef]
- Liang, C.; Li, H.; Tao, Y.; Shen, C.; Li, F.; Shi, Z.; Han, B.; Chen, Q. New hypothesis of chronic back pain: Low pH promotes nerve ingrowth into damaged intervertebral disks. Acta Anaesthesiol. Scand. 2013, 57, 271–277. [Google Scholar] [CrossRef]
- Voscopoulos, C.; Lema, M. When does acute pain become chronic? Br. J. Anaesth. 2010, 105 (Suppl. 1), i69–i85. [Google Scholar] [CrossRef]
- Allegri, M.; Montella, S.; Salici, F.; Valente, A.; Marchesini, M.; Compagnone, C.; Baciarello, M.; Manferdini, M.E.; Fanelli, G. Mechanisms of low back pain: A guide for diagnosis and therapy. F1000Res 5 (F1000 Faculty Rev.) 2016, 1530. [Google Scholar] [CrossRef]
- Green, B.G. Temperature perception and nociception. J. Neurobiol. 2004, 61, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.S.; Laymon, M.; Berk, L.; Bains, G. Effect of thermacare heatwraps and icy hot cream/patches on skin and quadriceps muscle temperature and blood flow. J. Chiropr. Med. 2016, 15, 9–18. [Google Scholar] [CrossRef]
- Klingler, W. Temperature effects on fascia. In Fascia—The Tensional Network of the Human Body; Schleip, R., Findley, T.W., Chaitow, L., Huijing, P., Eds.; Churchill Livingstone Elsevier: Edinburgh, UK, 2012; pp. 421–424. [Google Scholar]
- Stecco, A.; Gesi, M.; Stecco, C.; Stern, A. Fascial components of the myofascial pain syndrome. Curr. Pain Headache Rep. 2013, 17, 352. [Google Scholar] [CrossRef]
- Langevin, H.M.; Fox, J.R.; Koptiuch, C.; Badger, G.J.; Greenan-Naumann, A.C.; Bouffard, N.A.; Konofagou, E.E.; Lee, W.N.; Triano, J.J.; Henry, S.M. Reduced thoracolumbar fascia shear strain in human chronic low back pain. BMC Musculoskelet. Disord. 2011, 12, 203. [Google Scholar] [CrossRef] [PubMed]
- Bishop, J.H.; Fox, J.R.; Maple, R.; Loretan, C.; Badger, G.J.; Henry, S.M.; Vizzard, M.A.; Langevin, H.M. Ultrasound evaluation of the combined effects of thoracolumbar fascia injury and movement restriction in a porcine model. PLoS ONE. 2016, 11, e0147393. [Google Scholar] [CrossRef]
- Laymon, M.; Petrofsky, J.; McKivigan, J.; Lee, H.; Yim, J.E. Effect of heat, cold, and pressure on the transverse carpal ligament and median nerve: A pilot study. Med. Sci. Monit. 2015, 21, 446–451. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, K.; Reid, B.A.; Casey, C.A.; Bender, B.E.; Ro, B.; Song, Q.; Trewin, A.J.; Petersen, A.C.; Kuang, S.; Gavin, T.P.; et al. Effects of repeated local heat therapy on skeletal muscle structure and function in humans. J. Appl. Physiol. (1985) 2020, 128, 483–492. [Google Scholar] [CrossRef]
- Bellew, J.W.; Michlovitz, S.L.; Nolan, T.P. Modalities for Therapeutic Intervention; F. A. Davis Company: Philadelphia, PA, USA, 2016. [Google Scholar]
- Batavia, M. Contraindications for superficial heat and therapeutic ultrasound: Do sources agree? Arch. Phys. Med. Rehabil. 2004, 85, 1006–1012. [Google Scholar] [CrossRef]
- Cohen, S.P.; Argoff, C.E.; Carragee, E.J. Management of low back pain. BMJ 2008, 337, a2718. [Google Scholar] [CrossRef] [PubMed]
- Nadler, S.F.; Steiner, D.J.; Erasala, G.N.; Hengehold, D.A.; Hinkle, R.T.; Goodale, M.B.; Abeln, S.B.; Weingand, K.W. Continuous low-level heat wrap therapy provides more efficacy than ibuprofen and acetaminophen for acute low back pain. Spine 2002, 27, 1012–1017. [Google Scholar] [CrossRef] [PubMed]
- Nadler, S.F.; Steiner, D.J.; Erasala, G.N.; Hengehold, D.A.; Abeln, S.B.; Weingand, K.W. Continuous low-level heatwrap therapy for treating acute nonspecific low back pain. Arch. Phys. Med. Rehabil. 2003, 84, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Nadler, S.F.; Steiner, D.J.; Petty, S.R.; Erasala, G.N.; Hengehold, D.A.; Weingand, K.W. Overnight use of continuous low-level heatwrap therapy for relief of low back pain. Arch. Phys. Med. Rehabil. 2003, 84, 335–342. [Google Scholar] [CrossRef][Green Version]
- Lurie-Luke, E.; Neubauer, G.; Lindl, C.; Breitkreutz, H.; Fischer, P.; Hitzeroth, S. An exploratory workplace study to investigate the perceived value of continuous low-level heatwrap therapy in manual workers. Occup. Med. 2003, 53, 173–178. [Google Scholar] [CrossRef][Green Version]
- Tao, X.G.; Bernacki, E.J. A randomized clinical trial of continuous low-level heat therapy for acute muscular low back pain in the workplace. J. Occup. Environ. Med. 2005, 47, 1298–1306. [Google Scholar] [CrossRef]
- Mayer, J.M.; Ralph, L.; Look, M.; Erasala, G.N.; Verna, J.L.; Matheson, L.N.; Mooney, V. Treating acute low back pain with continuous low-level heat wrap therapy and/or exercise: A randomized controlled trial. Spine J. 2005, 5, 395–403. [Google Scholar] [CrossRef]
- Lewis, S.E.; Holmes, P.S.; Woby, S.R.; Hindle, J.; Fowler, N.E. Short-term effect of superficial heat treatment on paraspinal muscle activity, stature recovery, and psychological factors in patients with chronic low back pain. Arch. Phys. Med. Rehabil. 2012, 93, 367–372. [Google Scholar] [CrossRef]
- Cote-Picard, C.; Tittley, J.; Mailloux, C.; Perreault, K.; Mercier, C.; Dionne, C.E.; Roy, J.S.; Masse-Alarie, H. Effect of thermal therapy and exercises on acute low back pain: A protocol for a randomized controlled trial. BMC Musculoskelet. Disord. 2020, 21, 814. [Google Scholar] [CrossRef]
- Kettenmann, B.; Wille, C.; Lurie-Luke, E.; Walter, D.; Kobal, G. Impact of continuous low level heatwrap therapy in acute low back pain patients: Subjective and objective measurements. Clin. J. Pain 2007, 23, 663–668. [Google Scholar] [CrossRef]
- Stark, J.; Petrofsky, J.; Berk, L.; Bains, G.; Chen, S.; Doyle, G. Continuous low-level heatwrap therapy relieves low back pain and reduces muscle stiffness. Phys. Sportsmed. 2014, 42, 39–48. [Google Scholar] [CrossRef]
- Lloyd, A.; Scott, D.A.; Akehurst, R.L.; Lurie-Luke, E.; Jessen, G. Cost-effectiveness of low-level heat wrap therapy for low back pain. Value Health 2004, 7, 413–422. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Petrofsky, J.; Laymon, M.; Alshammari, F.; Khowailed, I.A.; Lee, H. Continuous low level heat wraps; faster healing and pain relief during rehabilitation for back, knee and neck injuries. World J. Prev. Med. 2015, 3, 61–72. [Google Scholar]
- McCarberg, W.; Erasala, G.N.; Goodale, M.; Grender, J.; Hengehold, D.; Donikyan, L. Therapeutic benefits of continuous low-level heat wrap therapy (CLHT) for osteoarthritis (OA) of the knee. J. Pain 2005, 6, 781. [Google Scholar] [CrossRef]
- Draper, D.O.; Hopkins, T.J. Increased intramuscular and intracapsular temperature via ThermaCare knee wrap application. Med. Sci. Monit. 2008, 14, PI7–PI11. [Google Scholar]
- Petrofsky, J.S.; Laymon, M.S.; Alshammari, F.S.; Lee, H. Use of low level of continuous heat as an adjunct to physical therapy improves knee pain recovery and the compliance for home exercise in patients with chronic knee pain: A randomized controlled trial. J. Strength Cond. Res. 2016, 30, 3107–3115. [Google Scholar] [CrossRef] [PubMed]
- Michlovitz, S.; Hun, L.; Erasala, G.N.; Hengehold, D.A.; Weingand, K.W. Continuous low-level heat wrap therapy is effective for treating wrist pain. Arch. Phys. Med. Rehabil. 2004, 85, 1409–1416. [Google Scholar] [CrossRef] [PubMed]
- Mayer, J.M.; Mooney, V.; Matheson, L.N.; Erasala, G.N.; Verna, J.L.; Udermann, B.E.; Leggett, S. Continuous low-level heat wrap therapy for the prevention and early phase treatment of delayed-onset muscle soreness of the low back: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2006, 87, 1310–1317. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.; Batt, J.; Bollinger, J.N.; Jensen, M.C.; Maru, E.H.; Al-Nakhli, H.H. Comparison of different heat modalities for treating delayed-onset muscle soreness in people with diabetes. Diabetes Technol. Ther. 2011, 13, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Heiss, R.; Lutter, C.; Freiwald, J.; Hoppe, M.W.; Grim, C.; Poettgen, K.; Forst, R.; Bloch, W.; Huttel, M.; Hotfiel, T. Advances in delayed-onset muscle soreness (DOMS)—part II, treatment and prevention. Sportverlet. Sportschaden. 2019, 33, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.S.; Khowailed, I.A.; Lee, H.; Berk, L.; Bains, G.S.; Akerkar, S.; Shah, J.; Al-Dabbak, F.; Laymon, M.S. Cold vs. heat after exercise—is there a clear winner for muscle soreness. J. Strength Cond. Res. 2015, 29, 3245–3252. [Google Scholar] [CrossRef] [PubMed]
- Akin, M.D.; Weingand, K.W.; Hengehold, D.A.; Goodale, M.B.; Hinkle, R.T.; Smith, R.P. Continuous low-level topical heat in the treatment of dysmenorrhea. Obstet. Gynecol. 2001, 97, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Navvabi Rigi, S.; Kermansaravi, F.; Navidian, A.; Safabakhsh, L.; Safarzadeh, A.; Khazaian, S.; Shafie, S.; Salehian, T. Comparing the analgesic effect of heat patch containing iron chip and ibuprofen for primary dysmenorrhea: A randomized controlled trial. BMC Womens Health 2012, 12, 25. [Google Scholar] [CrossRef] [PubMed]
- Bissell, J.H. Therapeutic modalities in hand surgery. J. Hand Surg. Am. 1999, 24, 435–448. [Google Scholar] [CrossRef]
| Method of Heat Application | Type of Therapy | |
|---|---|---|
| Superficial heat therapy | Conduction | Heat wrap (wearable) Heat pack (grain) Hot water bottles Hot poultices Hot stone therapy Electric heat pads |
| Convection | Hydrotherapy Hot baths Heat lamp Stream/sauna |
| Author (Year) | N | Study Design | Study Treatment | Comparator(s) | Primary Endpoint Results | Other Endpoints/Outcomes |
|---|---|---|---|---|---|---|
| Acute low back pain | ||||||
| Nadler 2002 [46] | 371 | Prospective, randomized, single-blind, comparative, multicenter study Two days of treatment Two days of post-treatment follow-up | Continuous low-level heat wrap: 8 h/day at 40 °C for two days (n = 113) | Ibuprofen, 1200 mg/day (n = 106) Acetaminophen, 4000 mg/day (n = 113) Oral placebo (n = 20) Unheated back wrap (n = 19) | Pain relief on Day 1: Significantly greater with a heat wrap vs. acetaminophen (p = 0.0001) or ibuprofen (p = 0.0007) | Compared to acetaminophen or ibuprofen, a heat wrap wassignificantly associated with: (a) greater pain relief on Day 2 and extended pain relief (Days 3 and 4) (b) reduced muscle stiffness (Days 1–4) (c) improved flexibility (Days 2 and 4) (d) reduced disability (Days 2 and 4) |
| Nadler 2003 [47] | 219 | Prospective, randomized, parallel, single-blind, placebo-controlled, multicenter study Three days of treatment Two days of post-treatment follow-up | Continuous low-level heat wrap: 8 h/day at 40 °C for three consecutive days (n = 95) | Oral placebo (n = 96) Oral ibuprofen (n = 12) Unheated back wrap (n = 16) | Pain relief on Day 1: Significantly greater with a heat wrap vs. placebo (p < 0.001) | Compared to the placebo, a heat wrap was significantly associated with: (a) greater pain relief on Days 2 and 3 andextended pain relief (Days 4 and 5) (b) reduced muscle stiffness (Days 1–5) (c) improved flexibility (Days 1–5) (d) reduced disability (Days 3 and 5) |
| Nadler 2003 [48] | 76 | Prospective, randomized, parallel, single-blind, placebo-controlled, multicenter study Three nights of treatment Two days of post-treatment follow-up | Continuous low-level heat wrap: 8 h/night at 40 °C for three consecutive nights (n = 33) | Oral placebo (n = 34) Ibuprofen (n = 4) Unheated heat wrap (n = 5) | Morning pain relief on Days 2–4: Significantly greater with a heat wrap vs. placebo(p = 0.00005) | Compared to the placebo, a heat wrap was significantlyassociated with: (a) greater pain relief the following day and extended pain relief (Days 2–5) (b) reduced morning muscle stiffness in the morning and during the day (Days 2–5) (c) reduced disability at the end of treatment and follow-up (d) improved trunk flexibility on Day 4 (e) improved sleep quality and onset of sleep |
| Lurie-Luke 2003 [49] | 52 | Workplace intervention study Two days of treatment Two-week post-treatment follow-up | Continuous low-level heat wrap: 8 h/day at 40 °C for two consecutive days | – | A heat wrap significantly reduced pain intensity and impact of pain on work-related activities and sleep for two weeks post-treatment | A heat wrap was associated with a reduction in the use of other over-the-counter pain relief products A heat wrap was rated as “excellent” or “very good” by 44% of respondents and as “good” by 37% |
| Tao 2005 [50] | 43 | Randomized workplace study Three days of treatment 11 days of post-treatment follow-up | Continuous low-level heat wrap: 8 h/day at 40 °C for three consecutive days plus back pain education (n = 25) | Back pain education alone(n = 18) | Pain intensity and pain relief during treatment and follow-up: Heat wrap + education significantly reduced pain intensity (Days 1–14) and provided improved pain relief (Days 1–4) vs. education alone | Compared to education alone, heat wrap + education wasassociated with: reduced disability on Days 7 and 14 |
| Mayer 2005 [51] | 100 | Randomized, controlled outpatient study Five days of treatment Two days of post-treatment follow-up | Continuous low-level heat wrap: 8 h/day at 40 °C plus exercise for five consecutive days n = 24) | Heat wrap alone (n = 25) Exercise alone (n = 25) Educational booklet (control; n = 26) | Functional ability: Heat wrap + exercise significantly improved functional outcomes vs. exercise alone (p = 0.18), or the control (p = 0.002) on Day 4 and vs. a heat wrap alone (p = 0.0007), exercise alone (p = 0.0003) or the control (p < 0.0001) at Day 7 | Compared to a heat wrap alone, exercise alone, or the control, a heat wrap + exercise was significantly associated with: (a) less deficit from pre-injury function on Day 7 (b) reduced disability on Day 7 (and on Day 4 vs. the control) (c) greater pain relief on Days 4 (vs. the control) and 7 (vs. exercise alone and the control) |
| Kettenmann 2007 [54] | 30 | Randomized, active-controlled, parallel design study Four days of treatment One day of post-treatment follow-up | Continuous low-level heat wrap: ≥4 h/day at 40 °C for four consecutive days plus oral analgesics (as needed; n = 15) | Oral analgesics (as needed; n = 15) | Objective evidence of reduced pain arousal (EEG data): A heat wrap led to significantly greater drops in Beta-1 and -2 frequencies post-treatment vs. the control (Days 2 and 4) | Subjective evidence Compared to the control, a heat wrap was significantlyassociated with: (a) reduced pain (Days 2–4) (b) reduced stress (Day 3) (c) reduced tiredness (Days 2 and 4) (d) improved sleep quality (Day 4) (c) improved concentration (Days 2, 4) A heat wrap was rated as “excellent,” “very good,” or “good” by 86% of respondents |
| Stark 2014 [55] | 61 | Pilot study to evaluate sensitivity of two methods to assess time to onset of pain relief and flexibility | Continuous low-level heat wrap: 8 h at 40 °C (n = 26) | Oral placebo (n = 25) Sham wrap (n = 5) Oral ibuprofen (n = 5) | Median time to first pain and meaningful relief were both significantly shorter for heat wrap vs. placebo (p = 0.046 for both) | Compared to the placebo, a heat wrap was significantlyassociated with: (a) greater pain relief (b) greater change in muscle stiffness |
| Petrofsky 2015 [57] | 145 | Randomized, controlled outpatient study | Continuous low-level heat wrap: 6 h at 40 °C prior to home exercise program over two weeks (n = 71) | Home exercise program over two weeks without prior heat therapy (n = 7) | Compared to the control, a heat wrap was associated with a significantly greater: (a) improvement in strength after the two-week period (p < 0.01) (b) improvement in flexibility after the two-week period (p < 0.01) (c) reduction in disability after the two-week period (p < 0.01) (d) reduction in pain after the two-week period (p < 0.01) (e) compliance in the completion of home exercise (p < 0.01) Similar results were observed in patients with knee (n = 44) and neck (n = 59) pain | |
| Chronic low back pain | ||||||
| Lewis 2012 [52] | 24 | Prospective single-arm study (within-subjects repeated measures design) | Continuous low-level heat wrap: 40 °C applied 2 h prior to assessment | Assessment without prior heat wrap application | Pain ratings were impacted by the fluctuating nature of chronic LBP Heat wrap treatment was associated with a reduction in non-normalized muscle activity and improved short-term well-being | |
| Freiwald 2018 [29] | 176 | Randomized, active controlled, multicenter, single-blind, observational study 12 weeks treatment | Continuous low-level heat wrap: 8 h at 40 °C plus multimodal treatment for 12 weeks (n = 88) | Multimodal treatment only (n = 88) | Muscular strength and flexibility: (a) strength and flexibility improved in both groups (b) significantly greater improvements in extension, and right and left rotation observed in the heat therapy-supplemented group | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freiwald, J.; Magni, A.; Fanlo-Mazas, P.; Paulino, E.; Sequeira de Medeiros, L.; Moretti, B.; Schleip, R.; Solarino, G. A Role for Superficial Heat Therapy in the Management of Non-Specific, Mild-to-Moderate Low Back Pain in Current Clinical Practice: A Narrative Review. Life 2021, 11, 780. https://doi.org/10.3390/life11080780
Freiwald J, Magni A, Fanlo-Mazas P, Paulino E, Sequeira de Medeiros L, Moretti B, Schleip R, Solarino G. A Role for Superficial Heat Therapy in the Management of Non-Specific, Mild-to-Moderate Low Back Pain in Current Clinical Practice: A Narrative Review. Life. 2021; 11(8):780. https://doi.org/10.3390/life11080780
Chicago/Turabian StyleFreiwald, Jürgen, Alberto Magni, Pablo Fanlo-Mazas, Ema Paulino, Luís Sequeira de Medeiros, Biagio Moretti, Robert Schleip, and Giuseppe Solarino. 2021. "A Role for Superficial Heat Therapy in the Management of Non-Specific, Mild-to-Moderate Low Back Pain in Current Clinical Practice: A Narrative Review" Life 11, no. 8: 780. https://doi.org/10.3390/life11080780
APA StyleFreiwald, J., Magni, A., Fanlo-Mazas, P., Paulino, E., Sequeira de Medeiros, L., Moretti, B., Schleip, R., & Solarino, G. (2021). A Role for Superficial Heat Therapy in the Management of Non-Specific, Mild-to-Moderate Low Back Pain in Current Clinical Practice: A Narrative Review. Life, 11(8), 780. https://doi.org/10.3390/life11080780