Origin of Life on Mars: Suitability and Opportunities
Abstract
1. Introduction
2. Materials and Methods
2.1. Organic Molecules and Elements
2.2. Energy Sources
2.3. Planetary Settings
3. Results
3.1. Elements and Molecules on Mars
3.1.1. Organics and CHNOPS Elements
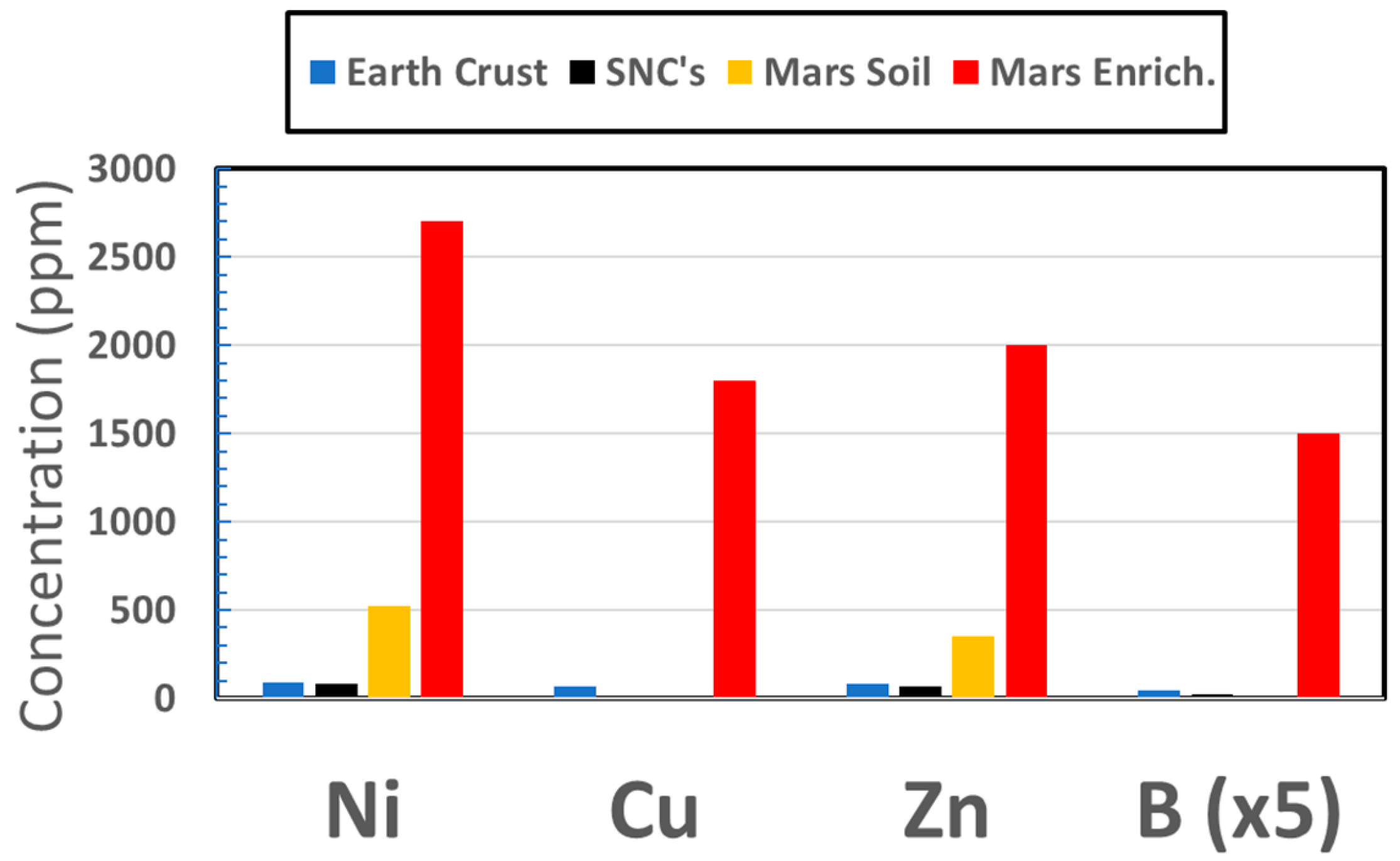
3.1.2. Transition Elements
3.1.3. Other Key Elements
3.1.4. Elements Availability
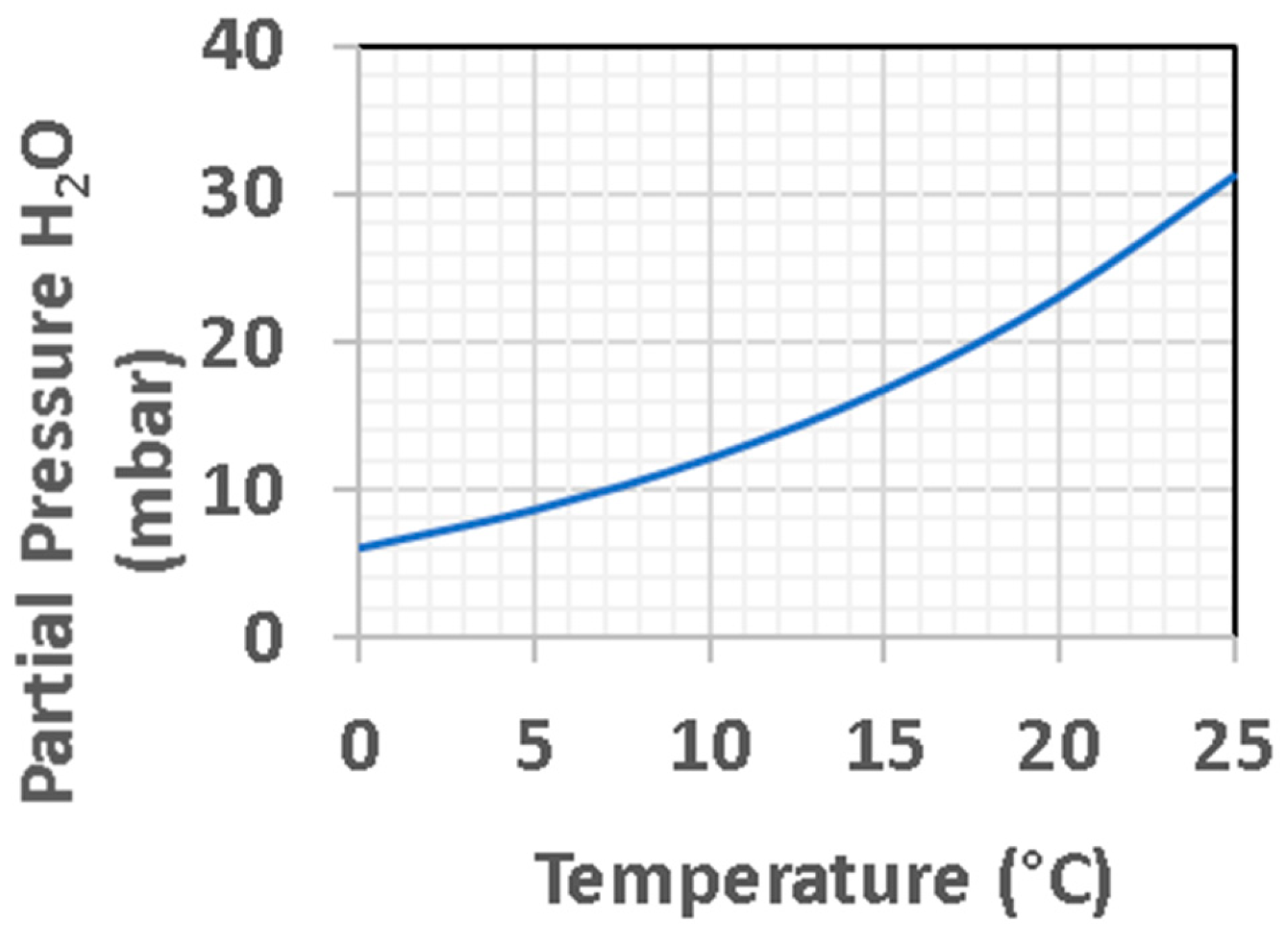
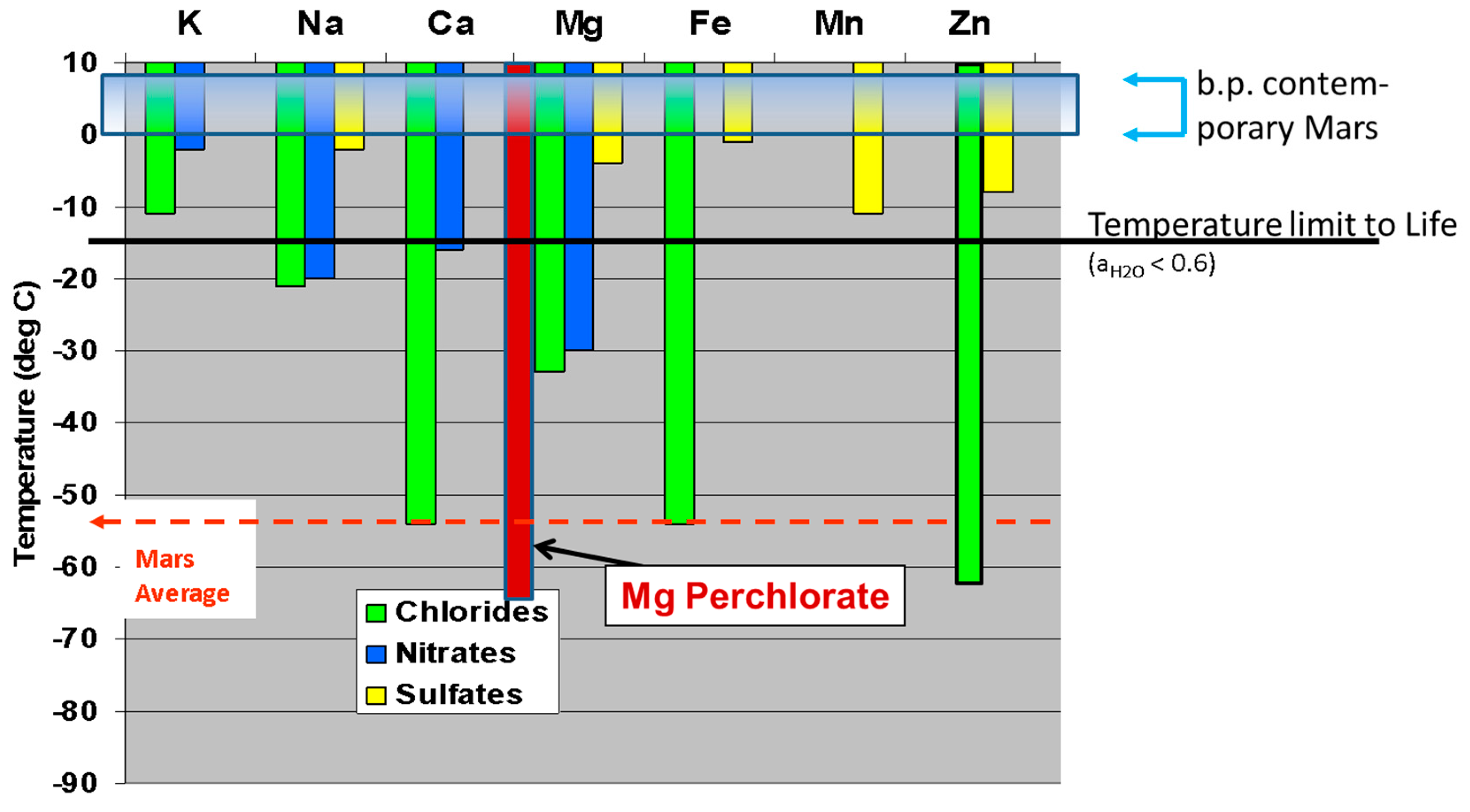
3.2. Energy Sources on Mars
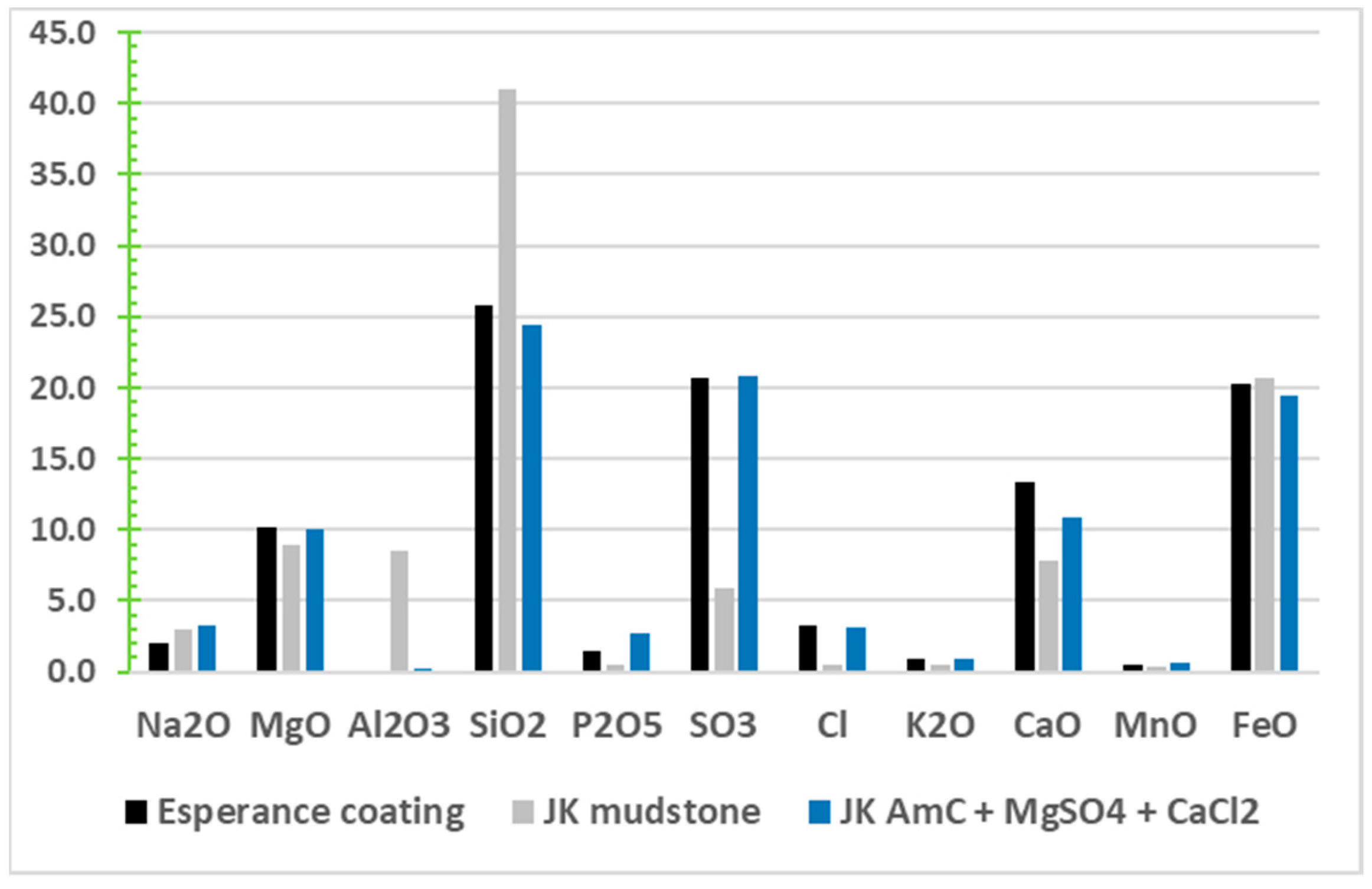
3.3. Settings on Mars
3.3.1. Early Mars as Compared to Early Earth
3.3.2. Subaerial Terrain Proto-Macrobionts
3.3.3. Suboceanic Hydrothermal Proto-Macrobionts
4. Discussion
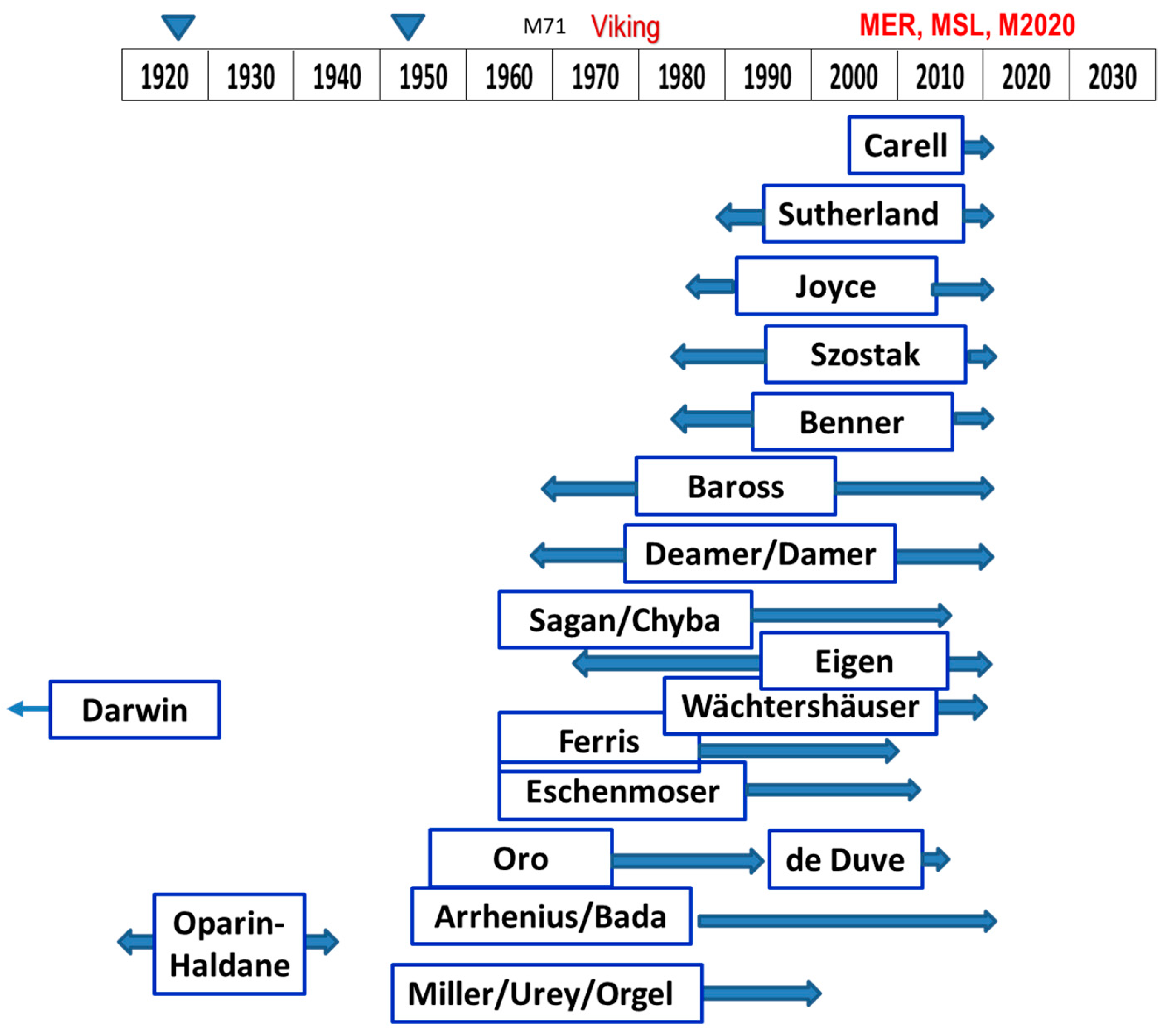
4.1. When Would Be an OoL on Mars: Past, Present, Future
4.2. Where an OoL Would Have Been on Mars
4.3. Likelihood for Origin of Life
4.3.1. Expected Value for the OoL
4.3.2. Lithopanspermia
4.4. Future Research
5. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AmC | Amorphous Component (ChemCam XRD) |
| APXS | Alpha Particle X-ray Spectrometer (XRF on MER, MSL rovers) |
| aw | Chemical activity of H2O |
| CCAM | ChemCam (MSL/LIBS on MSL rover) |
| CheMin | Chemical and Mineralogy instrument (XRD on MSL rover) |
| CRISM | Compact Recon Imaging Spectrometer for Mars (MRO orbiter) |
| OMEGA | Vis/IR Mineralogical Mapping (ESA/Mars Express orbiter) |
| EGA | Evolved Gas Analyzer (part of SAM on MSL rover) |
| GHS | Geothermal Hot Spring |
| GCMSGas | Chromatograph and Mass Spectrometer (Viking, MSL/SAM) |
| GCRGalactic | Cosmic Radiation |
| LAWKI | Life As We Know It (biochemically) |
| LIBS | Laser-Induced Breakdown Spectroscopy |
| MERMars | Exploration Rover (Spirit and Opportunity rovers) |
| MORB | Mid-Ocean Ridge Basalt |
| MS | Mass Spectrometer |
| MSL | Mars Science Laboratory (Curiosity rover) |
| M2020 | Mars 2020 Mission (Perseverance rover) |
| NWA | Northwest Africa (meteorite identifier) |
| OoL | Origin of Life |
| OHV | Oceanic Hydrothermal Vent |
| PAT | Pond in Ambient Temperature |
| PIXL | Planetary Instrument for X-ray Lithochemistry (M2020 rover) |
| PCE | Prebiotic chemical evolution |
| RNAzyme | RNA enzyme (ribozyme) |
| SAM | Sample Analysis at Mars (GCMS, EGA on MSL rover) |
| THEMIS | Thermal Emission Imaging System (Odyssey orbiter) |
| WCL | Wet Chemistry Laboratory (instrument on Phoenix lander) |
| XRD | X-ray Diffraction |
| XRF | X-ray Fluorescence |
References
- Pimentel, G.C.; Forney, P.B.; Herr, K.C. Evidence about hydrate and solid water in the Martian surface from the 1969 Mariner infrared spectrometer. J. Geophys. Res. 1974, 79, 1623–1634. [Google Scholar] [CrossRef]
- Milton, D.J. Water and processes of degradation in the Martian landscape. J. Geophys. Res. 1973, 78, 4037–4047. [Google Scholar] [CrossRef]
- Klein, H.P. The search for life on Mars: What we learned from Viking. J. Geophys. Res. Planets 1998, 103, 28463–284636. [Google Scholar] [CrossRef]
- Wordsworth, R.D. The climate of early Mars. Annu. Rev. Earth Planet. Sci. 2016, 44, 381–408. [Google Scholar] [CrossRef]
- Ehlmann, B.L.; Edwards, C.S. Mineralogy of the Martian surface. Annu. Rev. Earth Planet. Sci. 2014, 42, 291–315. [Google Scholar] [CrossRef]
- Rampe, E.B.; Blake, D.F.; Bristow, T.F.; Ming, D.W.; Vaniman, D.T.; Morris, R.V.; Achilles, C.N.; Chipera, S.J.; Morrison, S.M.; Tu, V.M.; et al. Mineralogy and geochemistry of sedimentary rocks and eolian sediments in Gale crater, Mars: A review after six Earth years of exploration with Curiosity. Geochemistry 2020, 80, 125605. [Google Scholar] [CrossRef]
- McLennan, S.M.; Grotzinger, J.P.; Hurowitz, J.A.; Tosca, N.J. The sedimentary cycle on early Mars. Annu. Rev. Earth Planet. Sci. 2019, 47, 91–118. [Google Scholar] [CrossRef]
- Grotzinger, J.P.; Sumner, D.Y.; Kah, L.C.; Stack, K.; Gupta, S.; Edgar, L.; Rubin, D.; Lewis, K.; Schieber, J.; Mangold, N.; et al. A habitable fluvio-lacustrine environment at Yellowknife Bay, Gale Crater, Mars. Science 2014, 24, 343. [Google Scholar] [CrossRef] [PubMed]
- Knoll, A.H.; Carr, M.; Clark, B.; Marais, D.J.D.; Farmer, J.D.; Fischer, W.W.; Grotzinger, J.P.; McLennan, S.M.; Malin, M.; Schröder, C.; et al. Astrobiological implications of Meridiani sediments. Earth Planet. Sci. Lett. 2005, 240, 179–189. [Google Scholar] [CrossRef]
- Benner, S.A.; Kim, H.J. The Case for a Martian Origin for Earth Life. In Proceedings of the Instruments, Methods, and Missions for Astrobiology XVII, San Diego, CA, USA, 28 September 2015; International Society for Optics and Photonics: Bellingham, WA, USA; Volume 9606, p. 96060C. [Google Scholar] [CrossRef]
- Fornaro, T.; Steele, A.; Brucato, J.R. Catalytic/protective properties of martian minerals and implications for possible origin of life on Mars. Life 2018, 8, 56. [Google Scholar] [CrossRef]
- Sasselov, D.D.; Grotzinger, J.P.; Sutherland, J.D. The origin of life as a planetary phenomenon. Sci. Adv. 2020, 6, eaax3419. [Google Scholar] [CrossRef] [PubMed]
- Clark, B.C.; Kolb, V.M. Macrobiont: Cradle for the origin of life and creation of a biosphere. Life 2020, 10, 278. [Google Scholar] [CrossRef] [PubMed]
- Mojarro, A.; Jin, L.; Szostak, J.W.; Head, J.W.; Zuber, M.T. In search of the RNA world on Mars. Geobiology 2021, 19, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Damer, B.; Deamer, D. The hot spring hypothesis for an origin of life. Astrobiology 2020, 20, 429–452. [Google Scholar] [CrossRef] [PubMed]
- McKay, C.P. An origin of life on Mars. Cold Spring Harb. Perspect. Biol. 2010, 2, a003509. [Google Scholar] [CrossRef]
- Frenkel-Pinter, M.; Rajaei, V.; Glass, J.B.; Hud, N.V.; Williams, L.D. Water and life: The medium is the message. J. Mol. Evol. 2021, 11, 1–10. [Google Scholar] [CrossRef]
- Miller, S.L. A production of amino acids under possible primitive earth conditions. Science 1953, 117, 528–529. [Google Scholar] [CrossRef]
- de Duve, C. Clues from Present-Day Biology: The Thioester World. In The Molecular Origins of Life: Assembling Pieces of the Puzzle; Brack, A., Ed.; Cambridge University Press: Cambridge, UK, 2000; pp. 219–236. [Google Scholar]
- Muchowska, K.B.; Varma, S.J.; Moran, J. Nonenzymatic metabolic reactions and life’s origins. Chem. Rev. 2020, 120, 7708–7744. [Google Scholar] [CrossRef]
- Chevallot-Beroux, E.; Gorges, J.; Moran, J. Energy conservation via thioesters in a non-enzymatic metabolism-like reaction network. ChemRxiv 2019, 9, 8832425. [Google Scholar]
- Goldford, J.E.; Hartman, H.; Marsland, R.; Segrè, D. Environmental boundary conditions for the origin of life converge to an organo-sulfur metabolism. Nat. Ecol. Evol. 2019, 3, 1715–1724. [Google Scholar] [CrossRef]
- Muchowska, K.B.; Moran, J. Peptide synthesis at the origin of life. Science 2020, 370, 767–768. [Google Scholar] [CrossRef] [PubMed]
- Wächtershäuser, G. The origin of life and its methodological challenge. J. Theor. Biol. 1997, 187, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Wächtershäuser, G. From volcanic origins of chemoautotrophic life to Bacteria, Archaea and Eukarya. Philos. Trans. R. B Biol. Sci. 2006, 361, 1787–1808. [Google Scholar] [CrossRef] [PubMed]
- Wächtershäuser, G. On the chemistry and evolution of the pioneer organism. Chem. Biodivers. 2007, 4, 584–602. [Google Scholar] [CrossRef] [PubMed]
- Ragsdale, S.W.; Pierce, E. Acetogenesis and the Wood-Ljungdahl pathway of CO2 fixation. Biochim. Biophys. Acta (BBA) Proteins Proteom. 2008, 1784, 1873–1898. [Google Scholar] [CrossRef]
- Todd, Z.R.; House, C.H. Vesicles protect activated acetic acid. Astrobiology 2014, 14, 859–865. [Google Scholar] [CrossRef]
- Russell, M.J.; Martin, W. The rocky roots of the acetyl-CoA pathway. Trends Biochem. Sci. 2004, 29, 358–363. [Google Scholar] [CrossRef]
- Ferry, J.G.; House, C.H. The stepwise evolution of early life driven by energy conservation. Mol. Biol. Evol. 2005, 23, 1286–1292. [Google Scholar] [CrossRef]
- Patel, B.H.; Percivalle, C.; Ritson, D.J.; Duffy, C.D.; Sutherland, J.D. Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nat. Chem. 2015, 7, 301–307. [Google Scholar] [CrossRef]
- Liu, Z.; Wu, L.F.; Xu, J.; Bonfio, C.; Russell, D.A.; Sutherland, J.D. Harnessing chemical energy for the activation and joining of prebiotic building blocks. Nat. Chem. 2020, 12, 1–6. [Google Scholar] [CrossRef]
- Ritson, D.J.; Battilocchio, C.; Ley, S.V.; Sutherland, J.D. Mimicking the surface and prebiotic chemistry of early Earth using flow chemistry. Nat. Commun. 2018, 9, 1–10. [Google Scholar]
- Ferus, M.; Kubelík, P.; Knížek, A.; Pastorek, A.; Sutherland, J.; Civiš, S. High energy radical chemistry formation of HCN-rich atmospheres on early Earth. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Xu, J.; Ritson, D.J.; Ranjan, S.; Todd, Z.R.; Sasselov, D.D.; Sutherland, J.D. Photochemical reductive homologation of hydrogen cyanide using sulfite and ferrocyanide. Chem. Commun. 2018, 54, 5566–5569. [Google Scholar] [CrossRef]
- Toner, J.D.; Catling, D.C. Alkaline lake settings for concentrated prebiotic cyanide and the origin of life. Geochim. Cosmochim. Acta 2019, 260, 124–132. [Google Scholar] [CrossRef]
- Foden, C.S.; Islam, S.; Fernández-García, C.; Maugeri, L.; Sheppard, T.D.; Powner, M.W. Prebiotic synthesis of cysteine peptides that catalyze peptide ligation in neutral water. Science 2020, 370, 865–869. [Google Scholar] [CrossRef]
- Becker, S.; Feldmann, J.; Wiedemann, S.; Okamura, H.; Schneider, C.; Iwan, K.; Crisp, A.; Rossa, M.; Amatov, T.; Carell, T. Unified prebiotically plausible synthesis of pyrimidine and purine RNA ribonucleotides. Science 2019, 366, 76–82. [Google Scholar] [CrossRef]
- Becker, S.; Schneider, C.; Okamura, H.; Crisp, A.; Amatov, T.; Dejmek, M.; Carell, T. Wet-dry cycles enable the parallel origin of canonical and non-canonical nucleosides by continuous synthesis. Nat. Commun. 2018, 9, 1–9. [Google Scholar] [CrossRef]
- Becker, S.; Thoma, I.; Deutsch, A.; Gehrke, T.; Mayer, P.; Zipse, H.; Carell, T. A high-yielding, strictly regioselective prebiotic purine nucleoside formation pathway. Science 2016, 352, 833–836. [Google Scholar] [CrossRef]
- Becker, S.; Schneider, C.; Crisp, A.; Carell, T. Non-canonical nucleosides and chemistry of the emergence of life. Nat. Commun. 2018, 9, 1–4. [Google Scholar] [CrossRef]
- Silva, J.D.; Franco, A. Boron in prebiological evolution. Angew. Chem. 2020, 133, 10550–10560. [Google Scholar] [CrossRef]
- Ricardo, A.; Carrigan, M.A.; Olcott, A.N.; Benner, S.A. Borate minerals stabilize ribose. Science 2004, 303, 196. [Google Scholar] [CrossRef]
- Kim, H.J.; Furukawa, Y.; Kakegawa, T.; Bita, A.; Scorei, R.; Benner, S.A. Evaporite borate-containing mineral ensembles make phosphate available and regiospecifically phosphorylate ribonucleosides: Borate as a multifaceted problem solver in prebiotic chemistry. Angew. Chem. 2016, 128, 16048–16052. [Google Scholar] [CrossRef]
- Kim, H.J.; Ricardo, A.; Illangkoon, H.I.; Kim, M.J.; Carrigan, M.A.; Frye, F.; Benner, S.A. Synthesis of carbohydrates in mineral-guided prebiotic cycles. J. Am. Chem. Soc. 2011, 133, 9457–9468. [Google Scholar] [CrossRef]
- Kitadai, N.; Maruyama, S. Origins of building blocks of life: A review. Geosci. Front. 2018, 9, 1117–1153. [Google Scholar] [CrossRef]
- Kim, H.J.; Benner, S.A. Abiotic synthesis of nucleoside 5′-triphosphates with nickel borate and cyclic trimetaphosphate (CTMP). Astrobiology 2021, 21, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Kawai, J.; McLendon, D.C.; Kim, H.J.; Benner, S.A. Hydroxymethanesulfonate from volcanic sulfur dioxide: A “mineral” reservoir for formaldehyde and other simple carbohydrates in prebiotic chemistry. Astrobiology 2019, 19, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Ferris, J.P.; Hill, A.R.; Liu, R.; Orgel, L.E. Synthesis of long prebiotic oligomers on mineral surfaces. Nature 1996, 381, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Ferris, J.P. One-step, regioselective synthesis of up to 50-mers of RNA oligomers by montmorillonite catalysis. J. Am. Chem. Soc. 2006, 128, 8914–8919. [Google Scholar] [CrossRef]
- Varma, S.J.; Muchowska, K.B.; Chatelain, P.; Moran, J. Native iron reduces CO2 to intermediates and end-products of the acetyl-CoA pathway. Nat. Ecol. Evol. 2018, 2, 1019–1024. [Google Scholar] [CrossRef]
- Martin, W.F. Older than genes: The acetyl CoA pathway and origins. Front. Microbiol. 2020, 11, 817. [Google Scholar] [CrossRef]
- Deamer, D.W. Membrane Compartments in Prebiotic Evolution. In The Molecular Origins of Life: Assembling Pieces of the Puzzle; Brack, A., Ed.; Cambridge University Press: Cambridge, UK, 2000; pp. 189–205. [Google Scholar]
- Deamer, D.; Damer, B.; Kompanichenko, V. Hydrothermal chemistry and the origin of cellular life. Astrobiology 2019, 19, 1523–1537. [Google Scholar] [CrossRef] [PubMed]
- Cornell, C.E.; Black, R.A.; Xue, M.; Litz, H.E.; Ramsay, A.; Gordon, M.; Mileant, A.; Cohen, Z.R.; Williams, J.A.; Lee, K.K.; et al. Prebiotic amino acids bind to and stabilize prebiotic fatty acid membranes. Proc. Natl. Acad. Sci. USA 2019, 116, 17239–172344. [Google Scholar] [CrossRef]
- Jin, L.; Engelhart, A.E.; Zhang, W.; Adamala, K.; Szostak, J.W. Catalysis of template-directed nonenzymatic RNA copying by Iron (II). J. Am. Chem. Soc. 2018, 140, 15016–15021. [Google Scholar] [CrossRef]
- O’Flaherty, D.K.; Kamat, N.P.; Mirza, F.N.; Li, L.; Prywes, N.; Szostak, J.W. Copying of mixed-sequence RNA templates inside model protocells. J. Am. Chem. Soc. 2018, 140, 5171–5178. [Google Scholar] [CrossRef]
- Li, M.; Huang, X.; Tang, T.D.; Mann, S. Synthetic cellularity based on non-lipid micro-compartments and protocell models. Curr. Opin. Chem. Biol. 2014, 22, 1–11. [Google Scholar] [CrossRef]
- Deamer, D.; Singaram, S.; Rajamani, S.; Kompanichenko, V.; Guggenheim, S. Self-assembly processes in the prebiotic environment. Philos. Trans. R. Soc. B Biol. Sci. 2006, 361, 1809–1818. [Google Scholar] [CrossRef]
- Ross, D.S.; Deamer, D. Dry/wet cycling and the thermodynamics and kinetics of prebiotic polymer synthesis. Life 2016, 6, 28. [Google Scholar] [CrossRef]
- Milshteyn, D.; Damer, B.; Havig, J.; Deamer, D. Amphiphilic compounds assemble into membranous vesicles in hydrothermal hot spring water but not in seawater. Life 2018, 8, 11. [Google Scholar] [CrossRef]
- Tjhung, K.F.; Sczepanski, J.T.; Murtfeldt, E.R.; Joyce, G.F. RNA-catalyzed cross-chiral polymerization of RNA. J. Am. Chem. Soc. 2020, 142, 15331–15339. [Google Scholar] [CrossRef]
- Joyce, G.F.; Szostak, J.W. Protocells and RNA self-replication. Cold Spring Harb. Perspect. Biol. 2018, 10, a034801. [Google Scholar] [CrossRef]
- Horning, D.P.; Joyce, G.F. Amplification of RNA by an RNA polymerase ribozyme. Proc. Natl. Acad. Sci. USA 2016, 113, 9786–9791. [Google Scholar] [CrossRef]
- Joyce, G.F.; Orgel, L.E. Prospects for Understanding of RNA World. In The RNA World: The Nature of Modern RNA Suggests a Prebiotic RNA World; Gesteland, R.F., Atkins, J.F., Eds.; Cold Spring Harbor University Press: Cold Spring Harbor, NY, USA, 1993; pp. 1–25. [Google Scholar]
- Robertson, M.P.; Joyce, G.F. The origins of the RNA World. Cold Spring Harb. Perspect. Biol. 2012, 4, a003608. [Google Scholar] [CrossRef]
- Tjhung, K.F.; Shokhirev, M.N.; Horning, D.P.; Joyce, G.F. An RNA polymerase ribozyme that synthesizes its own ancestor. Proc. Natl. Acad. Sci. USA 2020, 117, 2906–2913. [Google Scholar] [CrossRef]
- Kaim, W.; Schwederski, B.; Klein, A. Bioinorganic Chemistry—Inorganic Elements in the Chemistry of Life: An Introduction and Guide; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Silva, J.F.D.; Williams, R.J. The Biological Chemistry of the Elements: The Inorganic Chemistry of Life; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Morowitz, H.J.; Srinivasan, V.; Smith, E. Ligand field theory and the origin of life as an emergent feature of the periodic table of elements. Biol. Bull. 2010, 219, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Muchowska, K.B.; Varma, S.J.; Chevallot-Beroux, E.; Lethuillier-Karl, L.; Li, G.; Moran, J. Metals promote sequences of the reverse Krebs cycle. Nat. Ecol. Evol. 2017, 1, 1716–1721. [Google Scholar] [CrossRef]
- Degtyarenko, K. Bioinorganic motifs: Towards functional classification of metalloproteins. Bioinformatics 2000, 16, 851–864. [Google Scholar] [CrossRef]
- Hao, J.; Mokhtari, M.; Pedreira-Segade, U.; Michot, L.J.; Daniel, I. Transition metals enhance the adsorption of nucleotides onto clays: Implications for the origin of life. ACS Earth Space Chem. 2018, 3, 109–119. [Google Scholar] [CrossRef]
- Moore, E.K.; Jelen, B.I.; Giovannelli, D.; Raanan, H.; Falkowski, P.G. Metal availability and the expanding network of microbial metabolisms in the Archaean eon. Nat. Geosci. 2017, 10, 629–636. [Google Scholar] [CrossRef]
- Waldron, K.J.; Rutherford, J.C.; Ford, D.; Robinson, N.J. Metalloproteins and metal sensing. Nature 2009, 460, 823–830. [Google Scholar] [CrossRef]
- Frenkel-Pinter, M.; Samanta, M.; Ashkenasy, G.; Leman, L.J. Prebiotic peptides: Molecular hubs in the origin of life. Chem. Rev. 2020, 11, 4707–4765. [Google Scholar] [CrossRef] [PubMed]
- Van Der Gulik, P.; Massar, S.; Gilis, D.; Buhrman, H.; Rooman, M. The first peptides: The evolutionary transition between prebiotic amino acids and early proteins. J. Theor. Biol. 2009, 261, 531–539. [Google Scholar] [CrossRef]
- Presta, L.; Fondi, M.; Emiliani, G.; Fani, R. Molybdenum Availability in the Ecosystems (Geochemistry Aspects, When and How Did It Appear?). In Molybdenum Cofactors and Their role in the Evolution of Metabolic Pathways; Springer: Dordrecht, The Netherlands, 2015. [Google Scholar] [CrossRef]
- Hickman-Lewis, K.; Cavalazzi, B.; Sorieul, S.; Gautret, P.; Foucher, F.; Whitehouse, M.J.; Jeon, H.; Georgelin, T.; Cockell, C.S.; Westall, F. Metallomics in deep time and the influence of ocean chemistry on the metabolic landscapes on Earth’s earliest ecosystems. Sci. Rep. 2020, 10, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Mulkidjanian, A.Y.; Bychkov, A.Y.; Dibrova, D.V.; Galperin, M.Y.; Koonin, E.V. Origin of first cells at terrestrial, anoxic geothermal fields. Proc. Natl. Acad. Sci. USA 2012, 109, E821–E830. [Google Scholar] [CrossRef]
- Lu, G.S.; LaRowe, D.E.; Fike, D.A.; Druschel, G.K.; Gilhooly, W.P., III; Price, R.E.; Amend, J.P. Bioenergetic characterization of a shallow-sea hydrothermal vent system: Milos Island, Greece. PLoS ONE 2020, 15, e0234175. [Google Scholar] [CrossRef] [PubMed]
- Clark, B.C.; Kolb, V.M. Comet pond II: Synergistic intersection of concentrated extraterrestrial materials and planetary environments to form procreative Darwinian ponds. Life 2018, 8, 12. [Google Scholar] [CrossRef]
- Pearce, B.K.; Pudritz, R.E.; Ralph, E.; Semenov, D.A.; Henning, T.K. Origin of the RNA world: The fate of nucleobases in warm little ponds. Proc. Natl. Acad. Sci. USA 2017, 114, 11327–11332. [Google Scholar] [CrossRef] [PubMed]
- Baross, J.A. The rocky road to biomolecules. Nature 2018, 564, 42–43. [Google Scholar] [CrossRef] [PubMed]
- Baross, J.A.; Anderson, R.E.; Stüeken., E.E. The Environmental Roots of the Origin of Life. In Planetary Astrobiology; Meadows, V.S., Arney, G.N., Schmidt, B.E., Marais, D.J.D., Eds.; University of Arizona: Tucson, AZ, USA, 2020; pp. 71–92. [Google Scholar]
- Westall, F.; Hickman-Lewis, K.; Hinman, N.; Gautret, P.; Campbell, K.A.; Bréhéret, J.G.; Foucher, F.; Hubert, A.; Sorieul, S.; Dass, A.V.; et al. A hydrothermal-sedimentary context for the origin of life. Astrobiology 2018, 18, 259–293. [Google Scholar] [CrossRef]
- Sojo, V.; Herschy, B.; Whicher, A.; Camprubi, E.; Lane, N. The origin of life in alkaline hydrothermal vents. Astrobiology 2016, 16, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Egel, R. Primal eukaryogenesis: On the communal nature of precellular states, ancestral to modern life. Life 2012, 2, 170–212. [Google Scholar] [CrossRef]
- Russell, M.J. The alkaline solution to the emergence of life: Energy, entropy and early evolution. Acta Biotheor. 2007, 55, 133–179. [Google Scholar] [CrossRef] [PubMed]
- Russell, M.J.; Barge, L.M.; Bhartia, R.; Bocanegra, D.; Bracher, P.J.; Branscomb, E.; Kidd, R.; McGlynn, S.; Meier, D.H.; Nitschke, W.; et al. The drive to life on wet and icy worlds. Astrobiology 2014, 14, 308–343. [Google Scholar] [CrossRef]
- Russell, M.J.; Hall, A.J. The emergence of life from iron monosulphide bubbles at a submarine hydrothermal redox and pH front. J. Geol. Soc. 1997, 154, 377–402. [Google Scholar] [CrossRef] [PubMed]
- Maslennikov, V.V.; Maslennikova, S.P.; Large, R.R.; Danyushevsky, L.V.; Herrington, R.J.; Ayupova, N.R.; Zaykov, V.V.; Lein, A.Y.; Tseluyko, A.S.; Melekestseva, I.Y.; et al. Chimneys in Paleozoic massive sulfide mounds of the Urals VMS deposits: Mineral and trace element comparison with modern black, grey, white and clear smokers. Ore Geol. Rev. 2017, 85, 64–106. [Google Scholar] [CrossRef]
- Deamer, D.W. Assembling Life: How Can Life Begin on Earth and Other Habitable Planets? Oxford University Press: Oxford, UK, 2018. [Google Scholar]
- Chatterjee, S. A symbiotic view of the origin of life at hydrothermal impact crater-lakes. Phys. Chem. Chem. Phys. 2016, 18, 20033–20046. [Google Scholar] [CrossRef] [PubMed]
- Kring, D.A.; Whitehouse, M.J.; Schmieder, M. Microbial sulfur isotope fractionation in the Chicxulub hydrothermal system. Astrobiology 2020, 21, 103–114. [Google Scholar] [CrossRef]
- Osinski, G.R.; Cockell, C.S.; Pontefract, A.; Sapers, H.M. The role of meteorite impacts in the origin of life. Astrobiology 2020, 20, 1121–1149. [Google Scholar] [CrossRef] [PubMed]
- Benner, S.A.; Bell, E.A.; Biondi, E.; Brasser, R.; Carell, T.; Kim, H.J.; Trail, D. When did life likely emerge on Earth in an RNA-first process? Chem. Syst. Chem. 2019, 2, e1900035. [Google Scholar] [CrossRef]
- Lahav, N.; White, D.; Chang, S. Peptide formation in the prebiotic era: Thermal condensation of glycine in fluctuating clay environments. Science 1978, 201, 67–69. [Google Scholar] [CrossRef]
- Higgs, P.S. The effect of limited diffusion and wet-dry cycling on reversible polymerization reactions: Implications for prebiotic synthesis of nucleic acids. Life 2016, 6, 24. [Google Scholar] [CrossRef]
- Marais, D.J.D.; Walter, M.R. Terrestrial hot spring systems: Introduction. Astrobiology 2019, 19, 1419–1432. [Google Scholar] [CrossRef] [PubMed]
- Joshi, M.P.; Sawant, A.A.; Rajamani, S. Spontaneous emergence of membrane-forming protoamphiphiles from a lipid-amino acid mixture under wet-dry cycles. Chem. Sci. 2021, 12, 2970–2978. [Google Scholar] [CrossRef]
- Qiao, H.; Hu, N.; Bai, J.; Ren, L.; Liu, Q.; Fang, L.; Wang, Z. Encapsulation of nucleic acids into giant unilamellar vesicles by freeze-thaw: A way protocells may form. Orig. Life Evol. Biosph. 2017, 47, 499–510. [Google Scholar] [CrossRef]
- Vlassov, A.V.; Johnston, B.H.; Landweber, L.F.; Kazakov, S.A. Ligation activity of fragmented ribozymes in frozen solution: Implication for the RNAWorld. Nucleic Acids Res. 2004, 32, 2966–2974. [Google Scholar] [CrossRef]
- Mutschler, H.; Wochner, A.; Holliger, P. Freeze-thaw cycles as drivers of complex ribozyme assembly. Nat. Chem. 2015, 7, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Feller, G. Cryosphere and psychrophiles: Insights into a cold origin of life? Life 2017, 7, 25. [Google Scholar] [CrossRef]
- Putnam, A.R.; Palucis, M.C. The hydrogeomorphic history of Garu crater: Implications and constraints on the timing of large late-stage lakes in the Gale crater region. J. Geophys. Res. Planets 2020, e2020JE006688. [Google Scholar] [CrossRef]
- Salese, F.; Kleinhans, M.G.; Mangold, N.; Ansan, V.; McMahon, W.; Haas, T.D.; Dromart, G. Estimated minimum life span of the Jezero fluvial delta (Mars). Astrobiology 2020, 20, 977–993. [Google Scholar] [CrossRef]
- Schwartz, A.W. Evaluating the plausibility of prebiotic multistage syntheses. Astrobiology 2013, 13, 784–789. [Google Scholar] [CrossRef]
- Hunten, D.M. Possible oxidant sources in the atmosphere and surface of Mars. J. Mol. Evol. 1979, 14, 71–78. [Google Scholar] [CrossRef]
- Ranjan, S.; Todd, Z.R.; Sutherland, J.D.; Sasselov, D.D. Sulfidic anion concentrations on early Earth for surficial origins-of-life chemistry. Astrobiology 2018, 18, 1023–1040. [Google Scholar] [CrossRef]
- Achille, G.D.; Hynek, B.M. Ancient ocean on Mars supported by global distribution of deltas and valleys. Nat. Geosci. 2010, 3, 459–463. [Google Scholar] [CrossRef]
- Lasne, J.; Noblet, A.; Szopa, C.; Navarro-González, R.; Cabane, M.; Poch, O.; Stalport, F.; François, P.; Atreya, S.K.; Coll, P. Oxidants at the surface of Mars: A review in light of recent exploration results. Astrobiology 2016, 16, 977–996. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, R.M.; Kopparapu, R.; Zugger, M.E.; Robinson, T.D.; Freedman, R.; Kasting, J.F. Warming early Mars with CO2 and H2. Nat. Geosci. 2014, 7, 59–63. [Google Scholar] [CrossRef]
- Hayworth, B.P.; Kopparapu, R.K.; Haqq-Misra, J.; Batalha, N.E.; Payne, R.C.; Foley, B.J.; Ikwut-Ukwa, M.; Kasting, J.F. Warming early Mars with climate cycling: The effect of CO2-H2 collision-induced absorption. Icarus 2020, 27, 113770. [Google Scholar] [CrossRef]
- Wordsworth, R.; Pierrehumbert, R. Hydrogen-nitrogen greenhouse warming in Earth’s early atmosphere. Science 2013, 339, 64–67. [Google Scholar] [CrossRef]
- Klein, F.; Tarnas, J.D.; Bach, W. Abiotic sources of molecular hydrogen on Earth. Elem. Int. Mag. Mineral. Geoch. Petrol. 2020, 16, 19–24. [Google Scholar] [CrossRef]
- Jakosky, B.M. The CO2 inventory on Mars. Planet. Space Sci. 2019, 175, 52–59. [Google Scholar] [CrossRef]
- Kite, E.S.; Williams, J.P.; Lucas, A.; Aharonson, O. Low palaeopressure of the Martian atmosphere estimated from the size distribution of ancient craters. Nat. Geosci. 2014, 7, 335–339. [Google Scholar] [CrossRef]
- Warren, A.O.; Kite, E.S.; Williams, J.P.; Horgan, B. Through the thick and thin: New constraints on Mars paleopressure history 3.8–4 Ga from small exhumed craters. J. Geophys. Res. Planets. 2019, 124, 2793–2818. [Google Scholar] [CrossRef]
- Batalha, N.E.; Kopparapu, R.K.; Haqq-Misra, J.; Kasting, J.F. Climate cycling on early Mars caused by the carbonate–silicate cycle. Earth Planet. Sci. Lett. 2016, 455, 7–13. [Google Scholar] [CrossRef]
- Falkowski, P.; Scholes, R.J.; Boyle, E.E.; Canadell, J.; Canfield, D.; Elser, J.; Gruber, N.; Hibbard, K.; Högberg, P.; Linder, S.; et al. The global carbon cycle: A test of our knowledge of earth as a system. Science 2000, 290, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Schmitt-Kopplin, P.; Harir, M.; Kanawati, B.; Gougeon, R.; Moritz, F.; Hertkorn, N.; Clary, S.; Gebefügi, I.; Gabelica, Z. Analysis of Extraterrestrial Organic Matter in Murchison Meteorite: A Progress Report. In Astrobiology: An Evolutionary Approach; Kolb, V.M., Ed.; CRC Press/Taylor &Francis: Boca Raton, FL, USA, 2014; pp. 63–82. [Google Scholar]
- Yen, A.S.; Gellert, R.; Schröder, C.; Morris, R.V.; Bell, J.F.; Knudson, A.T.; Clark, B.C.; Ming, D.W.; Crisp, J.A.; Arvidson, R.E.; et al. An integrated view of the chemistry and mineralogy of Martian soils. Nature 2005, 436, 49–54. [Google Scholar] [CrossRef]
- Humayun, M.; Nemchin, A.; Zanda, B.; Hewins, R.H.; Grange, M.; Kennedy, A.; Lorand, J.P.; Göpel, C.; Fieni, C.; Pont, S.; et al. Origin and age of the earliest Martian crust from meteorite NWA 7533. Nature 2013, 503, 513–516. [Google Scholar] [CrossRef]
- Lodders, K. A survey of shergottite, nakhlite and chassigny meteorites whole-rock compositions. Meteorit. Planet. Sci. 1998, 33, A183–A190. [Google Scholar] [CrossRef]
- Airapetian, V.S.; Glocer, A.; Gronoff, G.; Hebrard, E.; Danchi, W. Prebiotic chemistry and atmospheric warming of early Earth by an active young Sun. Nat. Geosci. 2016, 9, 452–455. [Google Scholar] [CrossRef]
- Parkos, D.; Pikus, A.; Alexeenko, A.; Melosh, H.J. HCN production via impact ejecta reentry during the late heavy bombardment. J. Geophys. Res. Planets. 2018, 123, 892–909. [Google Scholar] [CrossRef]
- Matthews, C.N.; Minard, R.D. Hydrogen cyanide polymers, comets and the origin of life. Faraday Discuss. 2006, 133, 393–401. [Google Scholar] [CrossRef]
- Todd, Z.R.; Öberg, K.I. Cometary delivery of hydrogen cyanide to the early Earth. Astrobiology 2020, 20, 1109–1120. [Google Scholar] [CrossRef]
- Milo, R.; Phillips, R. Cell Biology by the Numbers; Garland Science: New York, NY, USA, 2015. [Google Scholar]
- Ramirez, R.M. A warmer and wetter solution for early Mars and the challenges with transient warming. Icarus 2017, 297, 71–82. [Google Scholar] [CrossRef]
- Stern, J.C.; Sutter, B.; Freissinet, C.; Navarro-González, R.; McKay, C.P.; Archer, P.D.; Buch, A.; Brunner, A.E.; Coll, P.; Eigenbrode, J.L.; et al. Evidence for indigenous nitrogen in sedimentary and aeolian deposits from the Curiosity rover investigations at Gale crater, Mars. Proc. Natl. Acad. Sci. USA 2015, 112, 4245–4250. [Google Scholar] [CrossRef]
- Stern, J.C.; Sutter, B.; Jackson, W.A.; Navarro-González, R.; McKay, C.P.; Ming, D.W.; Archer, P.D.; Mahaffy, P.R. The nitrate/(per) chlorate relationship on Mars. Geophys. Res. Lett. 2017, 44, 2643–2651. [Google Scholar] [CrossRef]
- Ranjan, S.; Todd, Z.R.; Rimmer, P.B.; Sasselov, D.D.; Babbin, A.R. Nitrogen oxide concentrations in natural waters on early Earth. Geochem. Geophys. Geosyst. 2019, 20, 2021–2039. [Google Scholar] [CrossRef]
- Kounaves, S.P.; Hecht, M.H.; Kapit, J.; Quinn, R.C.; Catling, D.C.; Clark, B.C.; Ming, D.W.; Gospodinova, K.; Hredzak, P.; McElhoney, K.; et al. Soluble sulfate in the Martian soil at the Phoenix landing site. Geophys. Res. Lett. 2010, 37. [Google Scholar] [CrossRef]
- Stroble, S.T.; McElhoney, K.M.; Kounaves, S.P. Comparison of the Phoenix Mars lander WCL soil analyses with Antarctic Dry Valley soils, Mars meteorite EETA79001 sawdust, and a Mars simulant. Icarus 2013, 225, 933–939. [Google Scholar] [CrossRef]
- Steele, A.; Benning, L.G.; Wirth, R.; Siljeström, S.; Fries, M.D.; Hauri, E.; Conrad, P.G.; Rogers, K.; Eigenbrode, J.; Schreiber, A.; et al. Organic synthesis on Mars by electrochemical reduction of CO. Sci. Adv. 2018, 4, eaat5118. [Google Scholar] [CrossRef]
- Hu, L.; Khaniya, A.; Wang, J.; Chen, G.; Kaden, W.E.; Feng, X. Ambient electrochemical ammonia synthesis with high selectivity on Fe/Fe oxide catalyst. ACS Catal. 2018, 8, 9312–9319. [Google Scholar] [CrossRef]
- Du, H.; Yang, C.; Pu, W.; Zeng, L.; Gong, J. Enhanced electrochemical reduction of N2 to ammonia over pyrite FeS2 with excellent selectivity. ACS Sustain. Chem. Eng. 2020, 8, 10572–10580. [Google Scholar] [CrossRef]
- Fray, N.; Bardyn, A.; Cottin, H.; Baklouti, D.; Briois, C.; Engrand, C.; Fischer, H.; Hornung, K.; Isnard, R.; Langevin, Y.; et al. Nitrogen-to-carbon atomic ratio measured by COSIMA in the particles of comet 67P/Churyumov–Gerasimenko. Mon. Not. R. Astron. Soc. 2017, 469, S506–S516. [Google Scholar] [CrossRef]
- Clifford, S.M.; Parker, T.J. The evolution of the Martian hydrosphere: Implications for the fate of a primordial ocean and the current state of the northern plains. Icarus 2001, 154, 40–79. [Google Scholar] [CrossRef]
- Manning, C.V.; Zahnle, K.J.; McKay, C.P. Impact processing of nitrogen on early Mars. Icarus 2009, 199, 273–285. [Google Scholar] [CrossRef]
- Fox, J.L. The production and escape of nitrogen atoms on Mars. J. Geophys. Res. Planets. 1993, 8, 3297–3310. [Google Scholar] [CrossRef]
- McElroy, M.B.; Yung, Y.L.; Nier, A.O. Isotopic composition of nitrogen: Implications for the past history of Mars’ atmosphere. Science 1976, 194, 70–72. [Google Scholar] [CrossRef]
- Robinson, T.D.; Reinhard, C.T. Earth as an Exoplanet. In Planetary Astrobiology; University of Arizona: Tucson, AZ, USA, 2020; pp. 379–416. [Google Scholar]
- Wordsworth, R.; Knoll, A.H.; Hurowitz, J.; Baum, M.; Ehlmann, B.L.; Head, J.W.; Steakley, K. A coupled model of episodic warming, oxidation and geochemical transitions on early Mars. Nat. Geosci. 2021, 14, 127–132. [Google Scholar] [CrossRef]
- Dreibus, G.; Jagoutz, E.; Spettel, B.; Wanke, H. Phosphate-mobilization on Mars? Implication from leach experiments on SNC’s. LPI 1996, 27, 323. [Google Scholar]
- Adcock, C.T.; Hausrath, E.M.; Forster, P.M. Readily available phosphate from minerals in early aqueous environments on Mars. Nat. Geosci. 2013, 6, 824–827. [Google Scholar] [CrossRef]
- Clark, B.C., III; Arvidson, R.E.; Gellert, R.; Morris, R.V.; Ming, D.W.; Richter, L.; Ruff, S.W.; Michalski, J.R.; Farrand, W.H.; Herkenhoff, K.E.; et al. Evidence for montmorillonite or its compositional equivalent in Columbia Hills, Mars. J. Geophys. Res. Planets 2007, 112. [Google Scholar] [CrossRef]
- Ming, D.W.; Mittlefehldt, D.W.; Morris, R.V.; Golden, D.C.; Gellert, R.; Yen, A.; Clark, B.C.; Squyres, S.W.; Farrand, W.H.; Ruff, S.W.; et al. Geochemical and mineralogical indicators for aqueous processes in the Columbia Hills of Gusev crater, Mars. J. Geophys. Res. Planets 2006, 111, E2. [Google Scholar] [CrossRef]
- McSween, H.Y.; Ruff, S.W.; Morris, R.V.; Bell, J.F., III; Herkenhoff, K.; Gellert, R.; Stockstill, K.R.; Tornabene, L.L.; Squyres, S.W.; Crisp, J.A.; et al. Alkaline volcanic rocks from the Columbia Hills, Gusev crater, Mars. J. Geophys. Res. Planets 2006, 111, E9. [Google Scholar] [CrossRef]
- McSween, H.Y.; Arvidson, R.E.; Bell, J.F.; Blaney, D.; Cabrol, N.A.; Christensen, P.R.; Clark, B.C.; Crisp, J.A.; Crumpler, L.S.; Marais, D.J.D.; et al. Basaltic rocks analyzed by the Spirit rover in Gusev Crater. Science 2004, 305, 842–845. [Google Scholar] [CrossRef] [PubMed]
- Berger, J.A.; Van Bommel, S.J.V.; Clark, B.C.; Gellert, R.; House, C.H.; King, P.L.; McCraig, M.A.; Ming, D.W.; O’Connell-Cooper, C.D.; Schmidt, M.E.; et al. Manganese and Phosphorus-Rich Nodules in Gale Crater, Mars: Results from the Groken Drill Site. In Proceedings of the 52nd Lunar and Planetary Science Conference, The Woodlands, TX, USA, 15–19 March 2021. [Google Scholar]
- Santos, A.R.; Agee, C.B.; McCubbin, F.M.; Shearer, C.K.; Burger, P.V.; Tartese, R.; Anand, M. Petrology of igneous clasts in Northwest Africa 7034: Implications for the petrologic diversity of the Martian crust. Geochim. Cosmochim. Acta 2015, 157, 56–85. [Google Scholar] [CrossRef]
- Liu, Y.; Fischer, W.W.; Ma, C.; Beckett, J.R.; Tschauner, O.; Guan, Y.; Lingappa, U.F.; Webb, S.M.; Prakapenka, V.B.; Lanza, N.L.; et al. Manganese oxides in Martian meteorites Northwest Africa (NWA) 7034 and 7533. Icarus 2021, 364, 114471. [Google Scholar] [CrossRef]
- Wellington, D.F.; Meslin, P.Y.; Van Beek, J.; Johnson, J.R.; Wiens, R.C.; Calef, F.J.; Bell, J.F. Iron Meteorite Finds Across Lower Mt. Sharp, Gale Crater, Mars: Clustering and Implications. In Proceedings of the 50th Lunar and Planetary Society Conference, The Woodlands, TX, USA, 18–22 March 2019. [Google Scholar]
- Fairen, A.G.; Dohm, J.M.; Baker, V.R.; Thompson, S.D.; Mahaney, W.C.; Herkenhoff, K.E.; Rodriguez, J.A.; Davila, A.F.; Schulze-makuch, D.; Maarry, M.R.E.; et al. Meteorites at Meridiani Planum provide evidence for significant amounts of surface and near-surface water on early Mars. Meteorit. Planet. Sci. 2011, 46, 1832–1841. [Google Scholar] [CrossRef]
- Meslin, P.Y.; Wellington, D.; Wiens, R.C.; Johnson, J.R.; Van Beek, J.; Gasnault, O.; Sautter, V.; Maroger, I.; Lasue, J.; Beck, P.; et al. Diversity and Areal Density of Iron-Nickel Meteorites Analyzed by Chemcam in Gale Crater. In Proceedings of the Lunar and Planetary Science Conference, The Woodlands, TX, USA, 18–22 March 2019; No. 2132; p. 3179. [Google Scholar]
- Lang, C.; Lago, J.; Pasek, M. Phosphorylation on the Early Earth: The Role of Phosphorus in Biochemistry and its Bioavailability. In Handbook of Astrobiology; Kolb, V.M., Ed.; CRC Press: Boca Raton, FL, USA, 2019; pp. 361–370. [Google Scholar]
- Gibard, C.; Gorrell, I.B.; Jiménez, E.I.; Kee, T.P.; Pasek, M.A.; Krishnamurthy, R. Geochemical sources and availability of amidophosphates on the early Earth. Angew. Chem. 2019, 131, 8235–8239. [Google Scholar] [CrossRef]
- Burcar, B.; Castañeda, A.; Lago, J.; Daniel, M.; Pasek, M.A.; Hud, N.V.; Orlando, T.M.; Menor-Salván, C. A stark contrast to modern earth: Phosphate mineral transformation and nucleoside phosphorylation in an iron-and cyanide-rich early earth scenario. Angew. Chem. Int. Ed. 2019, 58, 16981–16987. [Google Scholar] [CrossRef]
- Clark, B.C.; Baird, A.K. Is the Martian lithosphere sulfur rich? J. Geophys. Res. Solid Earth 1979, 84, 8395–8403. [Google Scholar] [CrossRef]
- Franz, H.B.; King, P.L.; Gaillard, F. Sulfur on Mars from the Atmosphere to the Core. In Volatiles in the Martian Crust; Elsevier: Amsterdam, The Netherlands, 2019; pp. 119–1483. [Google Scholar] [CrossRef]
- Sutherland, J.D. The origin of life—Out of the blue. Angew. Chem. Int. Ed. 2016, 55, 104–121. [Google Scholar] [CrossRef]
- Bonfio, C.; Valer, L.; Scintilla, S.; Shah, S.; Evans, D.J.; Jin, L.; Szostak, J.W.; Sasselov, D.D.; Sutherland, J.D.; Mansy, S.S. UV-light-driven prebiotic synthesis of iron–sulfur clusters. Nat. Chem. 2017, 9, 1229. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tsanakopoulou, M.; Magnani, C.J.; Szabla, R.; Šponer, J.E.; Šponer, J.; Góra, R.W.; Sutherland, J.D. A prebiotically plausible synthesis of pyrimidine β-ribonucleosides and their phosphate derivatives involving photoanomerization. Nat. Chem. 2017, 9, 303–309. [Google Scholar] [CrossRef]
- Styrt, M.M.; Brackmann, A.J.; Holland, H.D.; Clark, B.C.; Pisutha-Arnond, V.; Eldridge, C.S.; Ohmoto, H. The mineralogy and the isotopic composition of sulfur in hydrothermal sulfide/sulfate deposits on the East Pacific Rise, 21 N latitude. Earth Planet. Sci. Lett. 1981, 53, 382–390. [Google Scholar] [CrossRef]
- Gale, A.; Dalton, C.A.; Langmuir, C.H.; Su, Y.; Schilling, J.G. The mean composition of ocean ridge basalts. Geochem. Geophys. Geosyst. 2013, 14, 489–518. [Google Scholar] [CrossRef]
- Clark, B.C. Geochemical components in Martian soil. Geochim. Cosmochim. Acta 1993, 57, 4575–4581. [Google Scholar] [CrossRef]
- Yen, A.S.; Morris, R.V.; Clark, B.C.; Gellert, R.; Knudson, A.T.; Squyres, S.; Mittlefehldt, D.W.; Ming, D.W.; Arvidson, R.; McCoy, T.; et al. Hydrothermal processes at Gusev Crater: An evaluation of Paso Robles class soils. J. Geophys. Res. Planets 2008, 113, E6. [Google Scholar] [CrossRef]
- Clark, B.C.; Morris, R.V.; McLennan, S.M.; Gellert, R.; Jolliff, B.; Knoll, A.H.; Squyres, S.W.; Lowenstein, T.K.; Ming, D.W.; Tosca, N.J.; et al. Chemistry and mineralogy of outcrops at Meridiani Planum. Earth Planet. Sci. Lett. 2005, 240, 73–94. [Google Scholar] [CrossRef]
- Wang, A.; Haskin, L.A.; Squyres, S.W.; Jolliff, B.L.; Crumpler, L.; Gellert, R.; Schröder, C.; Herkenhoff, K.; Hurowitz, J.; Tosca, N.J.; et al. Sulfate deposition in subsurface regolith in Gusev crater, Mars. J. Geophys. Res. Planets 2006, 111, E2. [Google Scholar] [CrossRef]
- Rapin, W.; Ehlmann, B.L.; Dromart, G.; Schieber, J.; Thomas, N.H.; Fischer, W.W.; Fox, V.K.; Stein, N.T.; Nachon, M.; Clark, B.C.; et al. An interval of high salinity in ancient Gale crater lake on Mars. Nat. Geosci. 2019, 12, 889–895. [Google Scholar] [CrossRef]
- Yen, A.S.; Gellert, R.; Achilles, C.N.; Berger, J.A.; Blake, D.F.; Clark, B.C.; McAdam, C.; Wing, D.M.; Morris, R.V.; Morrison, S.M.; et al. Origin and Speciation of Sulfur Compounds in the Murray Formation, Gale Crater, Mars. In Proceedings of the Lunar and Planetary Science Conference, The Woodlands, TX, USA, 18–22 March 2019; Volume 50, p. 2083. [Google Scholar]
- Crisler, J.D.; Newville, T.M.; Chen, F.; Clark, B.C.; Schneegurt, M.A. Bacterial growth at the high concentrations of magnesium sulfate found in Martian soils. Astrobiology 2012, 12, 98–106. [Google Scholar] [CrossRef]
- Morris, R.V.; Schröder, C.; Klingelhöfer, G.; Agresti, D.G. Mössbauer Spectroscopy at Gusev Crater and Meridiani Planum: Iron Mineralogy, Oxidation State, and Alteration on Mars. In Remote Compositional Analysis: Techniques for Understanding Spectroscopy, Mineralogy, and Geochemistry of Planetary Surfaces; Bishop, J.L., Bell, J.F., Moersch, J.E., Eds.; Cambridge Planetary Science, University Press: Cambridge, UK, 2019; pp. 538–554. [Google Scholar] [CrossRef]
- Squyres, S.W.; Arvidson, R.E.; Bell, J.F.; Calef, F.; Clark, B.C.; Cohen, B.A.; Crumpler, L.A.; Souza, P.A.D.; Farrand, W.H.; Gellert, R.; et al. Ancient impact and aqueous processes at Endeavour Crater, Mars. Science 2012, 336, 570–576. [Google Scholar] [CrossRef]
- Clark, B.C.; Van Hart, D.C. The salts of Mars. Icarus 1981, 45, 370–378. [Google Scholar] [CrossRef]
- Banin, A.; Han, F.X.; Kan, I.; Cicelsky, A. Acidic volatiles and the Mars soil. J. Geophys. Res. Planets 1997, 102, 13341–13356. [Google Scholar] [CrossRef]
- Settle, M. Formation and deposition of volcanic sulfate aerosols on Mars. J. Geophys. Res. Solid Earth 1979, 84, 8343–8354. [Google Scholar] [CrossRef]
- Quinn, R.C.; Chittenden, J.D.; Kounaves, S.P.; Hecht, M.H. The oxidation-reduction potential of aqueous soil solutions at the Mars Phoenix landing site. Geophys. Res. Lett. 2011, 38. [Google Scholar] [CrossRef]
- Bandfield, J.L.; Glotch, T.D.; Christensen, P.R. Spectroscopic identification of carbonate minerals in the Martian dust. Science 2003, 301, 1084–1087. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.M.; Lewis, J.M.; Knudson, C.A.; Millan, M.; McAdam, A.C.; Eigenbrode, J.L.; Andrejkovičová, S.; Gómez, F.; Navarro-González, R.; House, C.H. Detection of reduced sulfur on Vera Rubin ridge by quadratic discriminant analysis of volatiles observed during evolved gas analysis. J. Geophys. Res. Planets 2020, 125, e2019JE006304. [Google Scholar] [CrossRef]
- Biemann, K.; Oro, J.I.; Toulmin, P., III; Orgel, L.E.; Nier, A.O.; Anderson, D.M.; Simmonds, P.G.; Flory, D.; Diaz, A.V.; Rushneck, D.R.; et al. The search for organic substances and inorganic volatile compounds in the surface of Mars. J. Geophys. Res. 1977, 82, 4641–4658. [Google Scholar] [CrossRef]
- Hubbard, J.S.; Hardy, J.P.; Horowitz, N.H. Photocatalytic production of organic compounds from CO and H2O in a simulated martian atmosphere. Proc. Natl. Acad. Sci. USA 1971, 68, 574–578. [Google Scholar] [CrossRef]
- Szopa, C.; Freissinet, C.; Glavin, D.P.; Millan, M.; Buch, A.; Franz, H.B.; Summons, R.E.; Sumner, D.Y.; Sutter, B.; Eigenbrode, J.L.; et al. First detections of dichlorobenzene isomers and trichloromethylpropane from organic matter indigenous to mars mudstone in gale crater, Mars: Results from the sample analysis at Mars instrument onboard the curiosity rover. Astrobiology 2020, 20, 292–306. [Google Scholar] [CrossRef]
- Eigenbrode, J.L.; Summons, R.E.; Steele, A.; Freissinet, C.; Millan, M.; Navarro-González, R.; Sutter, B.; McAdam, A.C.; Franz, H.B.; Glavin, D.P.; et al. Organic matter preserved in 3-billion-year-old mudstones at Gale crater, Mars. Science 2018, 360, 1096–1101. [Google Scholar] [CrossRef]
- Steele, A.; McCubbin, F.M.; Fries, M.; Kater, L.; Boctor, N.Z.; Fogel, M.L.; Conrad, P.G.; Glamoclija, M.; Spencer, M.; Morrow, A.L.; et al. A reduced organic carbon component in martian basalts. Science 2012, 337, 212–215. [Google Scholar] [CrossRef]
- Brown, P.G.; Hildebrand, A.R.; Zolensky, M.E.; Grady, M.; Clayton, R.N.; Mayeda, T.K.; Tagliaferri, E.; Spalding, R.; MacRae, N.D.; Hoffman, E.L.; et al. The fall, recovery, orbit, and composition of the Tagish Lake meteorite: A new type of carbonaceous chondrite. Science 2000, 290, 320–325. [Google Scholar] [CrossRef]
- Mustard, J.F.; Murchie, S.L.; Pelkey, S.M.; Ehlmann, B.L.; Milliken, R.E.; Grant, J.A.; Bibring, J.P.; Poulet, F.; Bishop, J.; Dobrea, E.N.; et al. Hydrated silicate minerals on Mars observed by the Mars Reconnaissance Orbiter CRISM instrument. Nature 2008, 454, 305–309. [Google Scholar] [CrossRef]
- Clark, B.C.; Morris, R.V.; Herkenhoff, K.E.; Farrand, W.H.; Gellert, R.; Jolliff, B.L.; Arvidson., R.E.; Squyres, S.W.; Mittlefehldt, D.W.; Ming, D.W.; et al. Esperance: Multiple episodes of aqueous alteration involving fracture fills and coatings at Matijevic Hill, Mars. Am. Mineral. 2016, 101, 1515–1526. [Google Scholar] [CrossRef]
- Ferris, J.P. Catalyzed RNA synthesis for the RNA world. In The Molecular Origins of Life: Assembling Pieces of the Puzzle; Brack, A., Ed.; Cambridge University Press: Cambridge, UK, 2000; pp. 255–268. [Google Scholar]
- Ming, D.W.; Morris, R.V.; Clark, B.C. Aqueous Alteration on Mars. In The Martian Surface; Bell, J., Ed.; Cambridge University Press: Cambridge, UK, 2008; Chapter 23; pp. 519–540. [Google Scholar]
- Yen, A.S.; Morris, R.V.; Ming, D.W.; Schwenzer, S.P.; Sutter, B.; Vaniman, D.T.; Treiman, A.H.; Gellert, R.; Achilles, C.N.; Berger, J.A.; et al. Formation of tridymite and evidence for a hydrothermal history at gale crater, Mars. J. Geophys. Res. Planets 2021. [Google Scholar] [CrossRef]
- Goetz, W.; Wiens, R.C.; Dehouck, E.; Gasnault, O.; Lasue, J.; Payre, V.; Frydenvang, J.; Clark, B.; Clegg, S.M.; Forni, O.; et al. Tracking of Copper by the ChemCam Instrument in Gale Crater, Mars: Elevated Abundances in Glen Torridon. In Proceedings of the Lunar and Planetary Science Conference, The Woodlands, TX, USA, 18–22 March 2020; Volume 2326, p. 2974. [Google Scholar]
- Payré, V.; Fabre, C.; Sautter, V.; Cousin, A.; Mangold, N.; Deit, L.L.; Forni, O.; Goetz, W.; Wiens, R.C.; Gasnault, O.; et al. Copper enrichments in the Kimberley formation in Gale crater, Mars: Evidence for a Cu deposit at the source. Icarus 2019, 321, 736–751. [Google Scholar] [CrossRef]
- VanBommel, S.J.; Gellert, R.; Berger, J.A.; Yen, A.S.; Boyd, N.I. Mars science laboratory alpha particle X-ray spectrometer trace elements: Situational sensitivity to Co, Ni, Cu, Zn, Ga, Ge, and Br. Acta Astronaut. 2019, 165, 32–42. [Google Scholar] [CrossRef]
- Gasda, P.J.; Haldeman, E.B.; Wiens, R.C.; Rapin, W.; Bristow, T.F.; Bridges, J.C.; Schwenzer, S.P.; Clark, B.; Herkenhoff, K.; Frydenvang, J.; et al. In situ detection of boron by ChemCam on Mars. Geophys. Res. Lett. 2017, 44, 8739–8748. [Google Scholar] [CrossRef]
- Lanza, N.L.; Fischer, W.W.; Wiens, R.C.; Grotzinger, J.; Ollila, A.M.; Cousin, A.; Anderson, R.B.; Clark, B.C.; Gellert, R.; Mangold, N.; et al. High manganese concentrations in rocks at Gale crater, Mars. Geophys. Res. Lett. 2014, 41, 5755–5763. [Google Scholar] [CrossRef]
- Lanza, N.L.; Wiens, R.C.; Arvidson, R.E.; Clark, B.C.; Fischer, W.W.; Gellert, R.; Grotzinger, J.P.; Hurowitz, J.A.; McLennan, S.M.; Morris, R.V.; et al. Oxidation of manganese in an ancient aquifer, Kimberley formation, Gale crater, Mars. Geophys. Res. Lett. 2016, 43, 7398–73407. [Google Scholar] [CrossRef]
- Arvidson, R.E.; Squyres, S.W.; Morris, R.V.; Knoll, A.H.; Gellert, R.; Clark, B.C.; Catalano, J.G.; Jolliff, B.L.; McLennan, S.M.; Herkenhoff, K.E.; et al. High concentrations of manganese and sulfur in deposits on Murray Ridge, Endeavour Crater, Mars. Am. Mineral. 2016, 101, 1389–1405. [Google Scholar] [CrossRef]
- Noda, N.; Imamura, S.; Sekine, Y.; Kurisu, M.; Fukushi, K.; Terada, N.; Uesugi, S.; Numako, C.; Takahashi, Y.; Hartmann, J. Highly oxidizing aqueous environments on early Mars inferred from scavenging pattern of trace metals on manganese oxides. J. Geophys. Res. Planets 2019, 124, 1282–1295. [Google Scholar] [CrossRef]
- Berger, J.A.; King, P.L.; Gellert, R.; Clark, B.C.; O’Connell-Cooper, C.D.; Thompson, L.M.; Van Bommel, S.J.; Yen, A.S. Manganese enrichment pathways relevant to Gale crater, Mars: Evaporative concentration and chlorine-induced precipitation. In Proceedings of the Lunar and Planetary Science Conference, The Woodlands, TX, USA, 18–22 March 2019; Volume 50, p. 2487. [Google Scholar]
- Salminen, R.; Batista, M.J.; Bidovec, M.; Demetriades, A.; Vivo, B.; Vos, W.D.; Duris, M.; Gilucis, A.; Gregorauskiene, V.; Halamić, J.; et al. Geochemical Atlas of Europe; Part 1, Background Information, Methodology and Maps; Geological Survey of Finland: Espoo, Finland, 2005. [Google Scholar]
- Marshall, C.P.; Marshall, A.O.; Aitken, J.B.; Lai, B.; Vogt, S.; Breuer, P.; Steemans, P.; Lay, P.A. Imaging of vanadium in microfossils: A new potential biosignature. Astrobiology 2017, 17, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Carpentier, W.; Sandra, K.; Smet, I.; Brige, A.; Smet, L.D.; Van Beeumen, J. Microbial reduction and precipitation of vanadium by Shewanella oneidensis. Appl. Environ. Microbiol. 2003, 69, 3636–3639. [Google Scholar] [CrossRef] [PubMed]
- Jochum, K.P.; Weis, U.; Schwager, B.; Stoll, B.; Wilson, S.A.; Haug, G.H.; Andreae, M.O.; Enzweiler, J. Reference values following ISO guidelines for frequently requested rock reference materials. Geostand. Geoanalytical Res. 2015, 40, 333–350. [Google Scholar] [CrossRef]
- Campbell, T.D.; Febrian, R.; McCarthy, J.T.; Kleinschmidt, H.E.; Forsythe, J.G.; Bracher, P.J. Prebiotic condensation through wet–dry cycling regulated by deliquescence. Nat. Commun. 2019, 10, 1–7. [Google Scholar] [CrossRef]
- Morris, R.V.; Rampe, E.B.; Vaniman, D.T.; Christoffersen, R.; Yen, A.S.; Morrison, S.M.; Ming, D.W.; Achilles, C.N.; Fraeman, A.A.; Le, L.; et al. Hydrothermal precipitation of sanidine (adularia) having full Al, Si structural disorder and specular hematite at Maunakea volcano (Hawai’i) and at Gale Crater (Mars). J. Geophys. Res. Planets 2020, 125, e2019JE006324. [Google Scholar] [CrossRef]
- Cousin, M.; Desjardins, E.; Dehouck, O.; Forni, G.; David, G.; Berger, G.; Caravaca, P.; Meslin, J.; Lasue, A.; Ollila, W.; et al. Wiens5, K-Rich Rubbly Bedrock at Glen Torridon, Gale Crater, Mars: Investigating the Possible Presence of Illite. In Proceedings of the 52nd Lunar and Planetary Science Conference 2021, The Woodlands, TX, USA, 15–19 March 2021. [Google Scholar]
- Bowman, J.C.; Petrov, A.S.; Frenkel-Pinter, M.; Penev, P.I.; Williams, L.D. Root of the tree: The significance, evolution, and origins of the ribosome. Chem. Rev. 2020, 120, 4848–4878. [Google Scholar] [CrossRef]
- Bray, M.S.; Lenz, T.K.; Haynes, J.W.; Bowman, J.C.; Petrov, A.S.; Reddi, A.R.; Hud, N.V.; Williams, L.D.; Glass, J.B. Multiple prebiotic metals mediate translation. Proc. Natl. Acad. Sci. USA 2018, 115, 12164–12169. [Google Scholar] [CrossRef]
- Kounaves, S.P.; Oberlin, E.A. Volatiles Measured by the Phoenix Lander at the Northern Plains of Mars. In Volatiles in the Martian Crust; Elsevier: Amsterdam, The Netherlands, 2019; pp. 265–283. [Google Scholar] [CrossRef]
- Bibring, J.P.; Langevin, Y.; Mustard, J.F.; Poulet, F.; Arvidson, R.; Gendrin, A.; Gondet, B.; Mangold, N.; Pinet, P.; Forget, F.; et al. Global mineralogical and aqueous Mars history derived from OMEGA/Mars Express data. Science 2006, 312, 400–404. [Google Scholar] [CrossRef]
- Das, D.; Gasda, P.J.; Wiens, R.C.; Berlo, K.; Leveille, R.J.; Frydenvang, J.; Mangold, N.; Kronyak, R.E.; Schwenzer, S.P.; Forni, O.; et al. Boron and lithium in calcium sulfate veins: Tracking precipitation of diagenetic materials in Vera Rubin Ridge, Gale crater. J. Geophys. Res. Planets 2020, 125, e2019JE006301. [Google Scholar] [CrossRef]
- Stephenson, J.D.; Hallis, L.J.; Nagashima, K.; Freeland, S.J. Boron enrichment in martian clay. PLoS ONE 2013, 8, e64624. [Google Scholar] [CrossRef]
- Palmer, M.R.; Sturchio, N.C. The boron isotope systematics of the Yellowstone National Park (Wyoming) hydrothermal system: A reconnaissance. Geochim. Cosmochim. Acta 1990, 54, 2811–2815. [Google Scholar] [CrossRef]
- Anbar, A.D. Elements and evolution. Science 2008, 5, 1481–1483. [Google Scholar] [CrossRef]
- Chemtob, S.M.; Nickerson, R.D.; Morris, R.V.; Agresti, D.G.; Catalano, J.G. Oxidative alteration of ferrous smectites and implications for the redox evolution of early Mars. J. Geophys. Res. Planets 2017, 122, 2469–2488. [Google Scholar] [CrossRef] [PubMed]
- Bullock, M.A.; Moore, J.M. Atmospheric conditions on early Mars and the missing layered carbonates. Geophys. Res. Lett. 2007, 34. [Google Scholar] [CrossRef]
- Baird, A.K.; Clark, B.C. On the original igneous source of Martian fines. Icarus 1981, 45, 113–123. [Google Scholar] [CrossRef]
- Zolotov, M.Y.; Mironenko, M.V. Timing of acid weathering on Mars: A kinetic-thermodynamic assessment. J. Geophys. Res. Planets 2007, 112, E7. [Google Scholar] [CrossRef]
- Thomas, N.H.; Ehlmann, B.L.; Meslin, P.Y.; Rapin, W.; Anderson, D.E.; Rivera-Hernández, F.; Forni, O.; Schröder, S.; Cousin, A.; Mangold, N.; et al. Mars science laboratory observations of chloride salts in Gale crater, Mars. Geophys. Res. Lett. 2019, 46, 10754–10763. [Google Scholar] [CrossRef]
- Smith, R.J.; McLennan, S.M.; Achilles, C.N.; Dehouck, E.; Horgan, B.H.; Mangold, N.; Rampe, E.B.; Salvatore, M.; Siebach, K.L.; Sun, V. X-ray amorphous components in sedimentary rocks of Gale crater, Mars: Evidence for ancient formation and long-lived aqueous activity. J. Geophys. Res. Planets 2021, 126, e2020JE006782. [Google Scholar] [CrossRef]
- Jackson, R.S.; Wiens, R.C.; Vaniman, D.T.; Beegle, L.; Gasnault, O.; Newsom, H.E.; Maurice, S.; Meslin, P.Y.; Clegg, S.; Cousin, A.; et al. ChemCam investigation of the John Klein and Cumberland drill holes and tailings, Gale crater, Mars. Icarus 2016, 277, 330–341. [Google Scholar] [CrossRef]
- Yoshimura, Y. The Search for Life on Mars. In Astrobiology; Yamagishi, A., Kakegawa, T., Usui, T., Eds.; Springer: Singapore, 2019; pp. 367–381. [Google Scholar] [CrossRef]
- Sholes, S.F.; Krissansen-Totton, J.; Catling, D.C. A maximum subsurface biomass on Mars from untapped free energy: CO and H2 as potential antibiosignatures. Astrobiology 2019, 19, 655–668. [Google Scholar] [CrossRef]
- Price, A.; Pearson, V.K.; Schwenzer, S.P.; Miot, J.; Olsson-Francis, K. Nitrate-dependent iron oxidation: A potential Mars metabolism. Front. Microbiol. 2018, 9, 513. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Toril, E.; Martínez-Frías, J.; Gomez, J.M.; Rull, F.; Amils, R. Iron meteorites can support the growth of acidophilic chemolithoauthrophic microorganisms. Astrobiology 2005, 5, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Soare, R.; Conway, S.; Williams, J.-P.; Oehler, D. (Eds.) Mars Geological Enigmas: From the Late Noachian Epoch to the Present Day, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2021; ISBN 9780128202456. [Google Scholar]
- Ernst, W.G. Earth’s thermal evolution, mantle convection, and Hadean onset of plate tectonics. J. Asian Earth Sci. 2017, 145, 334–348. [Google Scholar] [CrossRef]
- Faltys, J.P.; Wielicki, M.M. Inclusions in impact-formed zircon as a tracer of target rock lithology: Implications for Hadean continental crust composition and abundance. Lithos 2020, 376, 105761. [Google Scholar] [CrossRef]
- Bada, J.L.; Korenaga, J. Exposed areas above sea level on Earth >3.5 Gyr ago: Implications for prebiotic and primitive biotic chemistry. Life 2018, 8, 55. [Google Scholar] [CrossRef]
- Marshall, M. The water paradox and the origins of life. Nature 2020, 588, 210–213. [Google Scholar] [CrossRef]
- Carr, M.H.; Head, J.W. Martian surface/near-surface water inventory: Sources, sinks, and changes with time. Geophys. Res. Lett. 2015, 42, 726–732. [Google Scholar] [CrossRef]
- Palumbo, A.M.; Head, J.W.; Wordsworth, R.D. Late Noachian Icy Highlands climate model: Exploring the possibility of transient melting and fluvial/lacustrine activity through peak annual and seasonal temperatures. Icarus 2018, 300, 261–286. [Google Scholar] [CrossRef]
- Morris, R.V.; Klingelhoefer, G.; Schröder, C.; Fleischer, I.; Ming, D.W.; Yen, A.S.; Gellert, R.; Arvidson, R.E.; Rodionov, D.S.; Crumpler, L.S.; et al. Iron mineralogy and aqueous alteration from Husband Hill through Home Plate at Gusev crater, Mars: Results from the Mössbauer instrument on the Spirit Mars Exploration Rover. J. Geophys. Res. Planets 2008, 113, E12. [Google Scholar] [CrossRef]
- Tosca, N.J.; Ahmed, I.A.; Tutolo, B.M.; Ashpitel, A.; Hurowitz, J.A. Magnetite authigenesis and the warming of early Mars. Nat. Geosci. 2018, 11, 635–639. [Google Scholar] [CrossRef]
- Robbins, S.J.; Hynek, B.M. The secondary crater population of Mars. Earth Planet. Sci. Lett. 2014, 400, 66–76. [Google Scholar] [CrossRef]
- Segura, T.L.; Toon, O.B.; Colaprete, A. Modeling the environmental effects of moderate-sized impacts on Mars. J. Geophys. Res. Planets 2008, 113, E11. [Google Scholar] [CrossRef]
- Turbet, M.; Forget, F.; Head, J.W.; Wordsworth, R. 3D modelling of the climatic impact of outflow channel formation events on early Mars. Icarus 2017, 288, 10–36. [Google Scholar] [CrossRef]
- Haberle, R.M.; Zahnle, K.; Barlow, N.G.; Steakley, K.E. Impact degassing of H2 on early Mars and its effect on the climate system. Geophys. Res. Lett. 2019, 46, 13355–13362. [Google Scholar] [CrossRef]
- Treiman, A.H.; Bish, D.L.; Vaniman, D.T.; Chipera, S.J.; Blake, D.F.; Ming, D.W.; Morris, R.V.; Bristow, T.F.; Morrison, S.M.; Baker, M.B.; et al. Mineralogy, provenance, and diagenesis of a potassic basaltic sandstone on Mars: CheMin X-ray diffraction of the Windjana sample (Kimberley area, Gale Crater). J. Geophys. Res. Planets 2016, 121, 75–106. [Google Scholar] [CrossRef]
- Fraeman, A.A.; Edgar, L.A.; Rampe, E.B.; Thompson, L.M.; Frydenvang, J.; Fedo, C.M.; Catalano, J.G.; Dietrich, W.E.; Gabriel, T.S.; Vasavada, A.R.; et al. Evidence for a diagenetic origin of Vera Rubin ridge, Gale crater, Mars: Summary and synthesis of Curiosity’s exploration campaign. J. Geophys. Res. Planets 2020, 125, e2020JE006527. [Google Scholar] [CrossRef] [PubMed]
- David, G.; Cousin, A.; Forni, O.; Meslin, P.Y.; Dehouck, E.; Mangold, N.; L’Haridon, J.; Rapin, W.; Gasnault, O.; Johnson, J.R.; et al. Analyses of high-iron sedimentary bedrock and diagenetic features observed with ChemCam at Vera Rubin ridge, Gale crater, Mars: Calibration and characterization. J. Geophys. Res. Planets 2020, 125, e2019JE006314. [Google Scholar] [CrossRef]
- Achilles, C.N.; Rampe, E.B.; Downs, R.T.; Bristow, T.F.; Ming, D.W.; Morris, R.V.; Vaniman, D.T.; Blake, D.F.; Yen, A.S.; McAdam, A.C.; et al. Evidence for multiple diagenetic episodes in ancient fluvial-lacustrine sedimentary rocks in Gale crater, Mars. J. Geophys. Res. Planets 2020, 125, e2019JE006295. [Google Scholar] [CrossRef] [PubMed]
- Craddock, R.A.; Lorenz, R.D. The changing nature of rainfall during the early history of Mars. Icarus 2017, 293, 172–179. [Google Scholar] [CrossRef]
- Quay, G.S.D.; Goudge, T.A.; Fassett, C.I. Precipitation and aridity constraints from paleolakes on early Mars. Geology 2020, 48, 1189–1193. [Google Scholar] [CrossRef]
- Palumbo, A.M.; Head, J.W.; Wilson, L. Rainfall on Noachian Mars: Nature, timing, and influence on geologic processes and climate history. Icarus 2020, 11, 113782. [Google Scholar] [CrossRef]
- Duran, S.; Coulthard, T.J. The Kasei Valles, Mars: A unified record of episodic channel flows and ancient ocean levels. Sci. Rep. 2020, 10, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Soare, R.J.; Osinski, G.R.; Roehm, C.L. Thermokarst lakes and ponds on Mars in the very recent (late Amazonian) past. Earth Planet. Sci. Lett. 2008, 272, 382–393. [Google Scholar] [CrossRef]
- Grant, J.A.; Wilson, S.A.; Mangold, N.; Calef III, F. and Grotzinger, J.P. The timing of alluvial activity in Gale crater, Mars. Geophysical Res. Lett. 2014, 41, 1142–1149. [Google Scholar] [CrossRef]
- Comte, J.; Monier, A.; Crevecoeur, S.; Lovejoy, C.; Vincent, W.F. Microbial biogeography of permafrost thaw ponds across the changing northern landscape. Ecography 2016, 39, 609–618. [Google Scholar] [CrossRef]
- Kadoya, S.; Krissansen-Totton, J.; Catling, D.C. Probable cold and alkaline surface environment of the Hadean Earth caused by impact ejecta weathering. Geochem. Geophys. Syst. 2019, 21, e2019GC008734. [Google Scholar] [CrossRef]
- Buhler, P.B.; Piqueux, S. Mars Obliquity-Driven CO2 Exchange between the Atmosphere, Regolith, and Polar Cap. J. Geophy. Res. Planets 2021, e2020JE006759. [Google Scholar] [CrossRef]
- Knoll, A.H.; Jolliff, B.L.; Farrand, W.H.; Bell, I.I.I.J.F.; Clark, B.C.; Gellert, R.; Golombek, M.P.; Grotzinger, J.P.; Herkenhoff, K.E.; Johnson, J.R.; et al. Veneers, rinds, and fracture fills: Relatively late alteration of sedimentary rocks at Meridiani Planum, Mars. J. Geophys. Res. Planets 2008, 113, E6. [Google Scholar] [CrossRef]
- Osterloo, M.M.; Hamilton, V.E.; Bandfield, J.L.; Glotch, T.D.; Baldridge, A.M.; Christensen, P.R.; Tornabene, L.L.; Anderson, F.S. Chloride-bearing materials in the southern highlands of Mars. Science 2008, 319, 1651–1654. [Google Scholar] [CrossRef]
- Hynek, B.M.; Osterloo, M.K.; Kierein-Young, K.S. Late-stage formation of Martian chloride salts through ponding and evaporation. Geology 2015, 43, 787–790. [Google Scholar] [CrossRef]
- Rummel, J.D.; Beaty, D.W.; Jones, M.A.; Bakermans, C.; Barlow, N.G.; Boston, P.J.; Chevrier, V.F.; Clark, B.C.; Vera, J.P.D.; Gough, R.V.; et al. A new analysis of Mars “special regions”: Findings of the second MEPAG Special Regions Science Analysis Group (SR-SAG2). Astrobiology 2014, 14, 887–968. [Google Scholar] [CrossRef]
- Downing, J.A.; Prairie, Y.T.; Cole, J.J.; Duarte, C.M.; Tranvik, L.J.; Striegl, R.G.; McDowell, W.H.; Kortelainen, P.; Caraco, N.F.; Melack, J.M.; et al. The global abundance and size distribution of lakes, ponds, and impoundments. Limnol. Oceanogr. 2006, 51, 2388–2397. [Google Scholar] [CrossRef]
- Robbins, S.J.; Hynek, B.M. A new global database of Mars impact craters ≥ 1 km: Database creation, properties, and parameters. J. Geophys. Res. Planets 2012, 117, E5. [Google Scholar] [CrossRef]
- Golombek, M.P.; Warner, N.H.; Ganti, V.; Lamb, M.P.; Parker, T.J.; Fergason, R.L.; Sullivan, R. Small crater modification on Meridiani Planum and implications for erosion rates and climate change on Mars. J. Geophys. Res. Planets 2014, 119, 2522–2547. [Google Scholar] [CrossRef]
- Fairén, A.G.; Dohm, J.M.; Baker, V.R.; Pablo, M.A.D.; Ruiz, J.; Ferris, J.C.; Anderson, R.C. Episodic flood inundations of the northern plains of Mars. Icarus 2003, 165, 53–67. [Google Scholar] [CrossRef]
- Pearce, B.K.; Tupper, A.S.; Pudritz, R.E.; Higgs, P.G. Constraining the time interval for the origin of life on Earth. Astrobiology 2018, 18, 343–364. [Google Scholar] [CrossRef] [PubMed]
- Fassett, C.I.; Head, I.I.I.J.W. Valley network-fed, open-basin lakes on Mars: Distribution and implications for Noachian surface and subsurface hydrology. Icarus 2008, 198, 37–56. [Google Scholar] [CrossRef]
- Goudge, T.A.; Head, J.W.; Mustard, J.F.; Fassett, C.I. An analysis of open-basin lake deposits on Mars: Evidence for the nature of associated lacustrine deposits and post-lacustrine modification processes. Icarus 2012, 219, 211–229. [Google Scholar] [CrossRef]
- Bristow, T.F.; Rampe, E.B.; Achilles, C.N.; Blake, D.F.; Chipera, S.J.; Craig, P.; Crisp, J.A.; Marais, D.J.D.; Downs, R.T.; Gellert, R.; et al. Clay mineral diversity and abundance in sedimentary rocks of Gale crater, Mars. Sci. Adv. 2018, 4, eaar3330. [Google Scholar] [CrossRef] [PubMed]
- Ruff, S.W.; Niles, P.B.; Alfano, F.; Clarke, A.B. Evidence for a Noachian-aged ephemeral lake in Gusev crater, Mars. Geology 2014, 42, 359–362. [Google Scholar] [CrossRef]
- Frydenvang, J.; Mangold, N.; Wiens, R.C.; Fraeman, A.A.; Edgar, L.A.; Fedo, C.M.; L’Haridon, J.; Bedford, C.C.; Gupta, S.; Grotzinger, J.P.; et al. The chemostratigraphy of the Murray formation and role of diagenesis at Vera Rubin ridge in Gale crater, Mars, as observed by the ChemCam instrument. J. Geophys. Res. Planets 2020, 125, e2019JE006320. [Google Scholar] [CrossRef]
- Rapin, W.; Dromart, G.; Rubin, D.; Deit, L.L.; Mangold, N.; Edgar, L.A.; Gasnault, O.; Herkenhoff, K.; Mouélic, S.L.; Anderson, R.B.; et al. Alternating wet and dry depositional environments recorded in the stratigraphy of Mount Sharp at Gale crater, Mars. Geology 2021. [Google Scholar] [CrossRef]
- Martin, P.E.; Farley, K.A.; Malespin, C.A.; Mahaffy, P.R.; Edgett, K.S.; Gupta, S.; Dietrich, W.E.; Malin, M.C.; Stack, K.M.; Vasconcelos, P.M. Billion-year exposure ages in Gale crater (Mars) indicate Mount Sharp formed before the Amazonian period. Earth Planet. Sci. Lett. 2021, 554, 116667. [Google Scholar] [CrossRef]
- Milliken, R.E.; Grotzinger, J.P.; Wiens, R.; Gellert, R.; Thompson, L.M.; Sheppard, R.; Vasavada, A.; Bristow, T.; Mangold, N. The chemistry and mineralogy of an ancient lacustrine sequence on Mars: Lessons learned from integrating rover and orbiter datasets. LPI Contrib. 2019, 2089, 6191. [Google Scholar]
- Hurowitz, J.A.; Grotzinger, J.P.; Fischer, W.W.; McLennan, S.M.; Milliken, R.E.; Stein, N.; Vasavada, A.R.; Blake, D.F.; Dehouck, E.; Eigenbrode, J.L.; et al. Redox stratification of an ancient lake in Gale crater, Mars. Science 2017, 356, 6341. [Google Scholar] [CrossRef]
- Alfreider, A.; Baumer, A.; Bogensperger, T.; Posch, T.; Salcher, M.M.; Summerer, M. CO2 assimilation strategies in stratified lakes: Diversity and distribution patterns of chemolithoautotrophs. Environ. Microbiol. 2017, 19, 2754–2768. [Google Scholar] [CrossRef]
- Stein, N.; Grotzinger, J.P.; Schieber, J.; Mangold, N.; Hallet, B.; Newsom, H.; Stack, K.M.; Berger, J.A.; Thompson, L.; Siebach, K.L.; et al. Desiccation cracks provide evidence of lake drying on Mars, Sutton Island member, Murray formation, Gale Crater. Geology 2018, 46, 515–518. [Google Scholar] [CrossRef]
- Martin, P.E.; Farley, K.A.; Archer, D.P., Jr.; Hogancamp, J.V.; Siebach, K.L.; Grotzinger, J.P.; McLennan, S.M. Reevaluation of perchlorate in Gale crater rocks suggests geologically recent perchlorate addition. J. Geophys. Res. Planets 2020, 125, e2019JE006156. [Google Scholar] [CrossRef]
- Greeley, R.; Schneid, B.D. Magma generation on Mars: Amounts, rates, and comparisons with Earth, Moon, and Venus. Science 1991, 254, 996–998. [Google Scholar] [CrossRef]
- Newsom, H.E. Hydrothermal alteration of impact melt sheets with implications for Mars. Icarus 1980, 44, 207–216. [Google Scholar] [CrossRef]
- Ruff, S.W.; Campbell, K.A.; Van Kranendonk, M.J.; Rice, M.S.; Farmer, J.D. The case for ancient hot springs in Gusev crater, Mars. Astrobiology 2020, 20, 475–499. [Google Scholar] [CrossRef] [PubMed]
- Rapin, W.; Chauviré, B.; Gabriel, T.S.; McAdam, A.C.; Ehlmann, B.L.; Hardgrove, C.; Meslin, P.Y.; Rondeau, B.; Dehouck, E.; Franz, H.B.; et al. In situ analysis of opal in Gale Crater, Mars. J. Geophys. Res. Planets 2018, 123, 1955–1972. [Google Scholar] [CrossRef]
- Sun, V.Z.; Milliken, R.E. Characterizing the mineral assemblages of hot spring environments and applications to Mars orbital data. Astrobiology 2020, 20, 453–474. [Google Scholar] [CrossRef]
- Rossi, A.P.; Neukum, G.; Pondrelli, M.; Van Gasselt, S.; Zegers, T.; Hauber, E.; Chicarro, A.; Foing, B. Large-scale spring deposits on Mars? J. Geophys. Res. Planets 2008, 113, E8. [Google Scholar] [CrossRef]
- Berger, J.A.; Schmidt, M.E.; Gellert, R.; Boyd, N.I.; Desouza, E.D.; Flemming, R.L.; Izawa, M.R.; Ming, D.W.; Perrett, G.M.; Rampe, E.B.; et al. Zinc and germanium in the sedimentary rocks of Gale Crater on Mars indicate hydrothermal enrichment followed by diagenetic fractionation. J. Geophys. Res. Planets 2017, 122, 1747–1772. [Google Scholar] [CrossRef]
- Michalski, J.R.; Onstott, T.C.; Mojzsis, S.J.; Mustard, J.; Chan, Q.H.; Niles, P.B.; Johnson, S.S. The Martian subsurface as a potential window into the origin of life. Nat. Geosci. 2018, 11, 21–26. [Google Scholar] [CrossRef]
- Bridges, J.C.; Schwenzer, S.P. The nakhlite hydrothermal brine on Mars. Earth Planet. Sci. Lett. 2012, 359, 117–123. [Google Scholar] [CrossRef]
- Amador, E.S.; Bandfield, J.L.; Brazelton, W.J.; Kelley, D. The lost city hydrothermal field: A spectroscopic and astrobiological analogue for Nili Fossae, Mars. Astrobiology 2017, 17, 1138–1160. [Google Scholar] [CrossRef]
- Brown, A.J.; Hook, S.J.; Baldridge, A.M.; Crowley, J.K.; Bridges, N.T.; Thomson, B.J.; Marion, G.M.; Filho, C.R.D.S.; Bishop, J.L. Hydrothermal formation of clay-carbonate alteration assemblages in the Nili Fossae region of Mars. Earth Planet. Sci. Lett. 2010, 297, 174–182. [Google Scholar] [CrossRef]
- Turner, S.M.; Bridges, J.C.; Grebby, S.; Ehlmann, B.L. Hydrothermal activity recorded in post Noachian-aged impact craters on Mars. J. Geophys. Res. Planets 2016, 121, 608–625. [Google Scholar] [CrossRef]
- Carrozzo, F.G.; Achille, G.D.; Salese, F.; Altieri, F.; Bellucci, G. Geology and mineralogy of the Auki Crater, Tyrrhena Terra, Mars: A possible post impact-induced hydrothermal system. Icarus 2017, 281, 228–239. [Google Scholar] [CrossRef]
- Hamilton, C.W.; Mouginis-Mark, P.J.; Sori, M.M.; Scheidt, S.P.; Bramson, A.M. Episodes of aqueous flooding and effusive volcanism associated with Hrad Vallis, Mars. J. Geophys. Res. Planets 2018, 123, 1484–1510. [Google Scholar] [CrossRef]
- Michalski, J.R.; Dobrea, E.Z.; Niles, P.B.; Cuadros, J. Ancient hydrothermal seafloor deposits in Eridania basin on Mars. Nat. Commun. 2017, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Azuma, S.; Katayama, I. Evolution of the rheological structure of Mars. Earth Planets Space 2017, 69, 1–3. [Google Scholar] [CrossRef]
- Quarles, B.L.; Lissauer, J.J. Dynamical evolution of the Earth–Moon progenitors–Whence Theia? Icarus 2015, 248, 318–339. [Google Scholar] [CrossRef]
- Morrison, P.R.; Mojzsis, S.J. Tracing the early emergence of microbial sulfur metabolisms. Geomicrobiol. J. 2020, 10, 1–21. [Google Scholar] [CrossRef]
- Bouvier, L.C.; Costa, M.M.; Connelly, J.N.; Jensen, N.K.; Wielandt, D.; Storey, M.; Nemchin, A.A.; Whitehouse, M.J.; Snape, J.F.; Bellucci, J.J.; et al. Evidence for extremely rapid magma ocean crystallization and crust formation on Mars. Nature 2018, 558, 586–589. [Google Scholar] [CrossRef] [PubMed]
- Quantin-Nataf, C.; Craddock, R.A.; Dubuffet, F.; Lozac’h, L.; Martinot, M. Decline of crater obliteration rates during early martian history. Icarus 2019, 317, 427–433. [Google Scholar] [CrossRef]
- Grant, J.A.; Wilson, S.A. Evidence for late alluvial activity in Gale crater, Mars. Geophys. Res. Lett. 2019, 46, 7287–7294. [Google Scholar] [CrossRef]
- Martin, P.E.; Farley, K.A.; Baker, M.B.; Malespin, C.A.; Schwenzer, S.P.; Cohen, B.A.; Mahaffy, P.R.; McAdam, A.C.; Ming, D.W.; Vasconcelos, P.M.; et al. A two-step K-Ar experiment on Mars: Dating the diagenetic formation of jarosite from Amazonian groundwaters. J. Geophys. Res. Planets 2017, 122, 2803–2818. [Google Scholar] [CrossRef]
- Guitreau, M.; Flahaut, J. Record of low-temperature aqueous alteration of Martian zircon during the late Amazonian. Nat. Commun. 2019, 10, 1–9. [Google Scholar] [CrossRef]
- Richardson, M.I.; Mischna, M.A. Long-term evolution of transient liquid water on Mars. J. Geophys. Res. Planets 2005, 110, E3. [Google Scholar] [CrossRef]
- Davila, A.; Kahre, M.A.; Quinn, R.; Marais, D.J.D. The biological potential of present-day Mars. Planetary Astrobiology 2020, 16, 169. [Google Scholar]
- Meadows, V.; Arney, G.; Schmidt, B.; Marais, D.J.D. (Eds.) Planetary Astrobiology; University of Arizona Press: San Diego, CA, USA, 2020. [Google Scholar]
- Malin, M.C.; Edgett, K.S.; Posiolova, L.V.; McColley, S.M.; Dobrea, E.Z. Present-day impact cratering rate and contemporary gully activity on Mars. Science 2006, 314, 1573–1577. [Google Scholar] [CrossRef] [PubMed]
- Dundas, C.M.; Byrne, S.; McEwen, A.S.; Mellon, M.T.; Kennedy, M.R.; Daubar, I.J.; Saper, L. HiRISE observations of new impact craters exposing Martian ground ice. J. Geophys. Res. Planets 2014, 119, 109–127. [Google Scholar] [CrossRef]
- Vaughan, R.G.; Heasler, H.; Jaworowski, C.; Lowenstern, J.B.; Keszthelyi, L.P. Provisional Map of Thermal Areas in Yellowstone National Park Based on Satellite Thermal Infrared Imaging and Field Observations; US Geological Survey Scientific Investigations Report, No. 5137; US Geological Survey: Washington, DC, USA, 2014; p. 2. [Google Scholar] [CrossRef]
- Benner, S.A.; Devine, K.G.; Matveeva, L.N.; Powell, D.H. The missing organic molecules on Mars. Proc. Natl. Acad. Sci. USA 2000, 97, 2425–2430. [Google Scholar] [CrossRef] [PubMed]
- Zent, A. A historical search for habitable ice at the Phoenix landing site. Icarus 2008, 196, 385–408. [Google Scholar] [CrossRef]
- Krasnopolsky, V.A.; Feldman, P.D. Detection of molecular hydrogen in the atmosphere of Mars. Science 2001, 294, 1914–1917. [Google Scholar] [CrossRef] [PubMed]
- Grew, E.S.; Bada, J.L.; Hazen, R.M. Borate minerals and origin of the RNA world. Origins Life Evol. Biosph. 2011, 41, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Horneck, G.; Bäcker, H. Can microorganisms withstand the multistep trial of interplanetary transfer? Considerations and experimental approaches. Origins Life Evol. Biosph. 1986, 16, 414–415. [Google Scholar] [CrossRef]
- Clark, B.C. Barriers to natural interchange of biologically active material between Earth and Mars. Origins Life 1986, 16, 410–411. [Google Scholar] [CrossRef]
- Mileikowsky, C.; Cucinotta, F.A.; Wilson, J.W.; Gladman, B.; Horneck, G.; Lindegren, L.; Melosh, J.; Rickman, H.; Valtonen, M.; Zheng, J.Q. Natural transfer of viable microbes in space: From Mars to Earth and Earth to Mars. Icarus 2000, 145, 391–427. [Google Scholar] [CrossRef]
- Clark, B.C. Planetary interchange of bioactive material: Probability factors and implications. Origins Life Evol. Biosph. 2001, 31, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Kirschvink, J.L.; Weiss, B.P. Mars, panspermia, and the origin of life: Where did it all begin. Palaeontol. Electron. 2002, 4, 8–15. [Google Scholar]
- Melosh, H.J. The rocky road to panspermia. Nature 1988, 332, 687–688. [Google Scholar] [CrossRef] [PubMed]
- Gladman, B.J.; Burns, J.A.; Duncan, M.; Lee, P.; Levison, H.F. The exchange of impact ejecta between terrestrial planets. Science 1996, 271, 1387–1392. [Google Scholar] [CrossRef]
- El Goresy, A.; Gillet, P.; Miyahara, M.; Ohtani, E.; Ozawa, S.; Beck, P.; Montagnac, G. Shock-induced deformation of Shergottites: Shock-pressures and perturbations of magmatic ages on Mars. Geochim. Cosmochim. Acta 2013, 101, 233–262. [Google Scholar] [CrossRef]
- Cabrol, N.A.; Grin, E.A. (Eds.) From Habitability to Life on Mars; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Westall, F.; Foucher, F.; Bost, N.; Bertrand, M.; Loizeau, D.; Vago, J.L.; Kminek, G.; Gaboyer, F.; Campbell, K.A.; Bréhéret, J.G.; et al. Biosignatures on Mars: What, where, and how? Implications for the search for martian life. Astrobiology 2015, 15, 998–1029. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clark, B.C.; Kolb, V.M.; Steele, A.; House, C.H.; Lanza, N.L.; Gasda, P.J.; VanBommel, S.J.; Newsom, H.E.; Martínez-Frías, J. Origin of Life on Mars: Suitability and Opportunities. Life 2021, 11, 539. https://doi.org/10.3390/life11060539
Clark BC, Kolb VM, Steele A, House CH, Lanza NL, Gasda PJ, VanBommel SJ, Newsom HE, Martínez-Frías J. Origin of Life on Mars: Suitability and Opportunities. Life. 2021; 11(6):539. https://doi.org/10.3390/life11060539
Chicago/Turabian StyleClark, Benton C., Vera M. Kolb, Andrew Steele, Christopher H. House, Nina L. Lanza, Patrick J. Gasda, Scott J. VanBommel, Horton E. Newsom, and Jesús Martínez-Frías. 2021. "Origin of Life on Mars: Suitability and Opportunities" Life 11, no. 6: 539. https://doi.org/10.3390/life11060539
APA StyleClark, B. C., Kolb, V. M., Steele, A., House, C. H., Lanza, N. L., Gasda, P. J., VanBommel, S. J., Newsom, H. E., & Martínez-Frías, J. (2021). Origin of Life on Mars: Suitability and Opportunities. Life, 11(6), 539. https://doi.org/10.3390/life11060539







