Cancer-Associated Fibroblasts Regulate the Plasticity of Breast Cancer Stemness through the Production of Leukemia Inhibitory Factor
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Breast Cancer Cell Lines
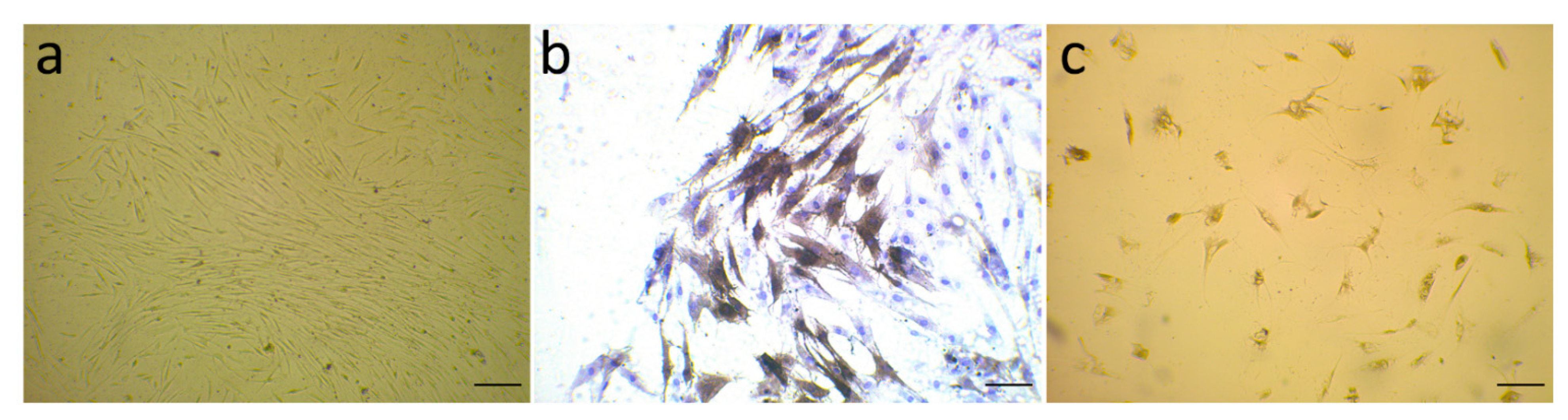
2.3. Isolation and Verification of CAFs
2.4. Immunocytochemistry (ICC)
2.5. Collection of Conditioned Media (CM)
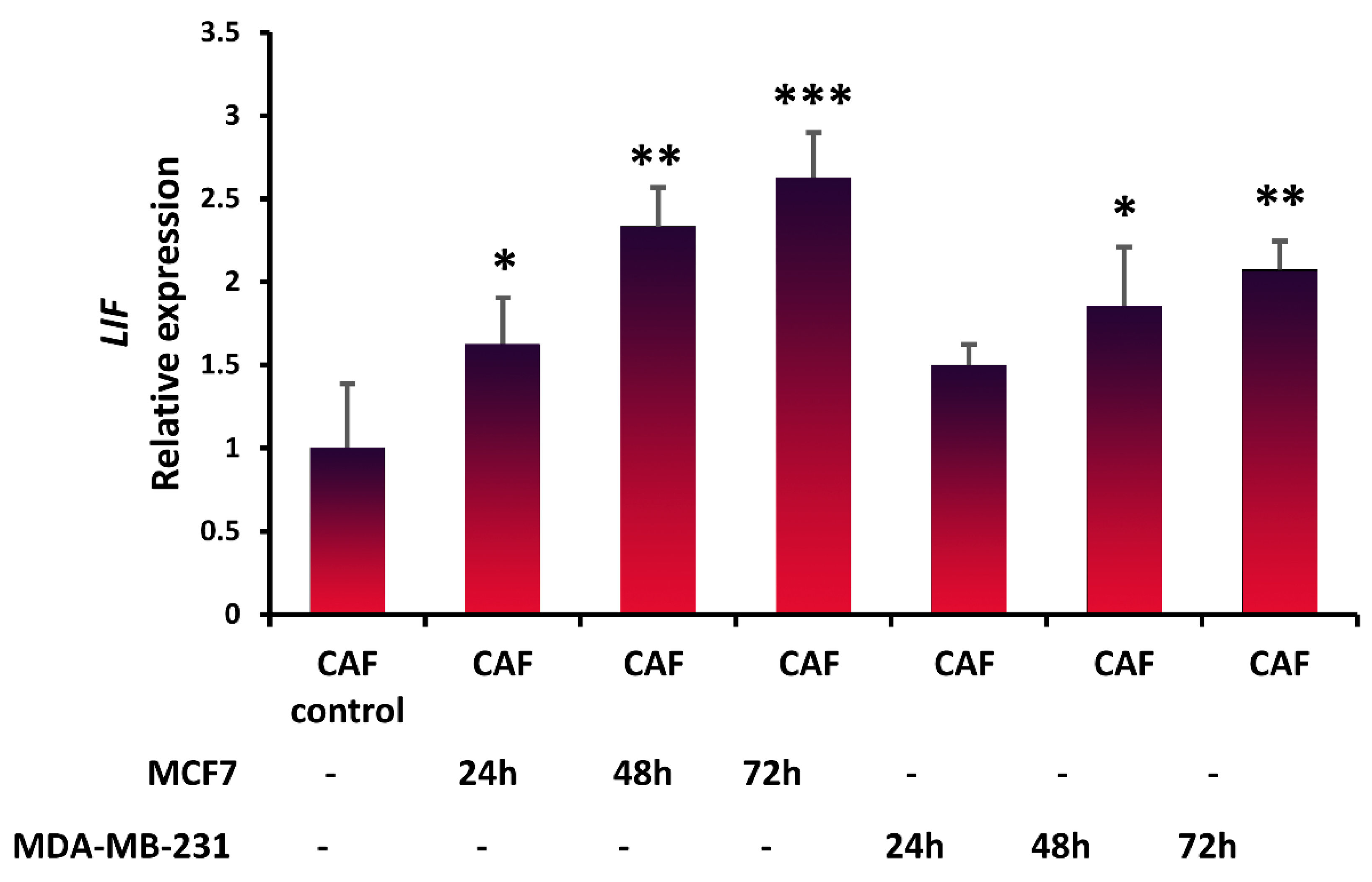
2.6. Evaluation of Cancer Cells’ Effect on LIF Expression of CAFs
2.7. Evaluation of Cancer Cells’ Stemness after Treatments
2.7.1. Cancer Cell Treatment with CAF-CM
2.7.2. Cancer Cell Treatment with LIF
2.7.3. Cancer Cell Treatment with LIF Receptor Inhibitor Antibody
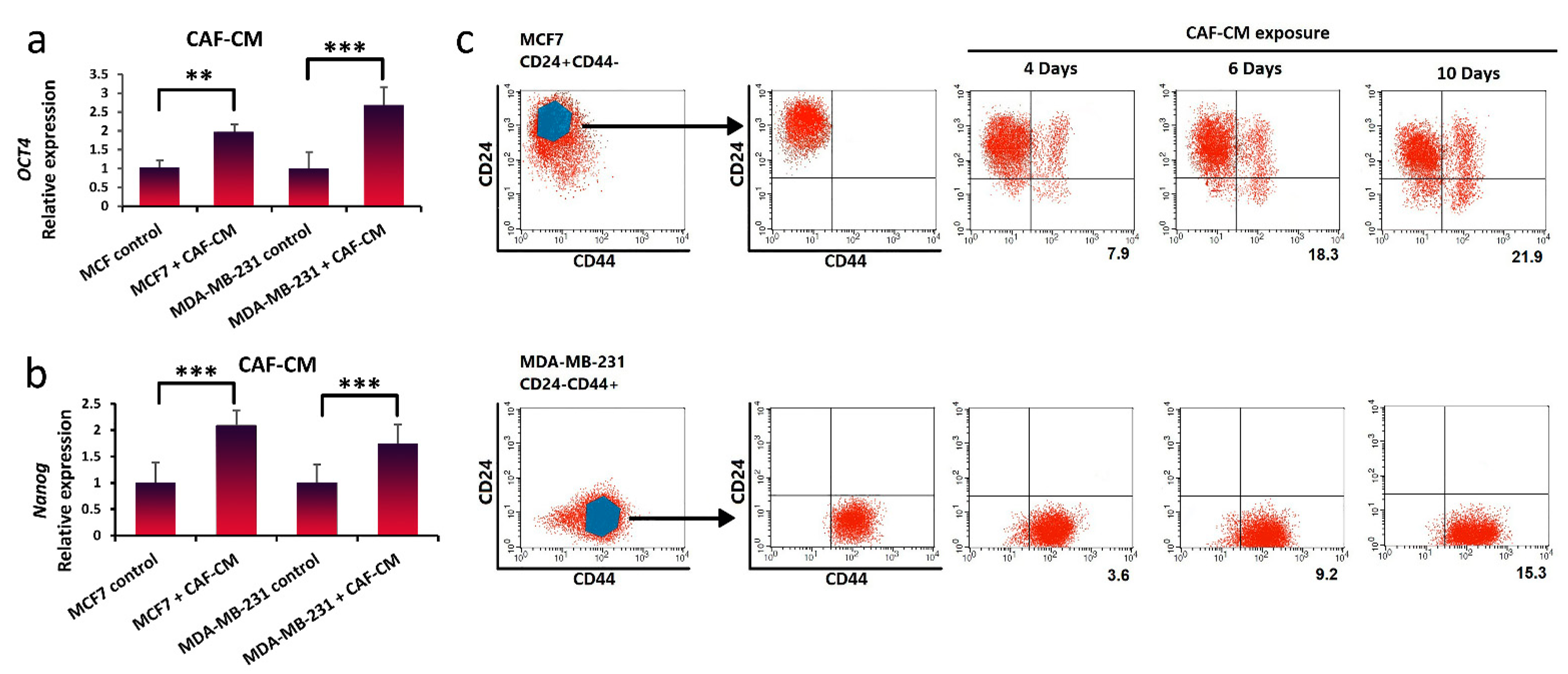
2.8. Flow Cytometry
2.9. Quantitative Real-Time PCR
2.10. Statistical Analyses
3. Results
3.1. Evaluation of α-SMA Markers’ Exhibition by Isolated Fibroblast
3.2. Expression of LIF in Fibroblasts in the Presence of Breast Cancer Cells
3.3. Inducing Stem-Cell-Like Properties in Breast Cancer Cells in the Presence of LIF
3.4. Promotion of Stem-Cell-like Properties in Breast Cancer Cells via Exogenous Exposure with LIF
3.5. Suppression of Breast Cancer Stemness via Blocking LIFR
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CAF | cancer-associated fibroblast |
| CSC | cancer stem cells |
| EMT | epithelial–mesenchymal transition |
| LIF | Leukemia inhibitory factor |
| LIF-R | LIF receptor |
| TME | tumor microenvironment |
| JAk | Janus kinase |
| PI3K | phosphatidylinositol 3-kinase |
| STAT | signal transducer and activator of transcription |
| TGF | transforming growth factor |
| FGF | fibroblast growth factor |
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics. CA A Cancer J. Clin. 2021, 71, 7–33. [Google Scholar]
- Kozłowski, J.; Kozłowska, A.; Kocki, J. Breast cancer metastasis-insight into selected molecular mechanisms of the phenomenon. Adv. Hyg. Exp. Med./Postepy Hig. I Med. Dosw. 2015, 69, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Pantel, K.; Kang, Y. Tumor metastasis: Moving new biological insights into the clinic. Nat. Med. 2013, 19, 1450. [Google Scholar] [CrossRef] [PubMed]
- Nurmik, M.; Ullmann, P.; Rodriguez, F.; Haan, S.; Letellier, E. In search of definitions: Cancer-associated fibroblasts and their markers. Int. J. Cancer 2020, 146, 895–905. [Google Scholar] [CrossRef] [Green Version]
- Surowiak, P.; Murawa, D.; Materna, V.; Maciejczyk, A.; Pudelko, M.; Ciesla, S.; Breborowicz, J.; Murawa, P.; Zabel, M.; Dietel, M. Occurence of stromal myofibroblasts in the invasive ductal breast cancer tissue is an unfavourable prognostic factor. Anticancer. Res. 2007, 27, 2917–2924. [Google Scholar]
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598. [Google Scholar] [CrossRef]
- Garcia-Tunon, I.; Ricote, M.; Ruiz, A.; Fraile, B.; Paniagua, R.; Royuela, M. OSM, LIF, its receptors, and its relationship with the malignance in human breast carcinoma (in situ and in infiltrative). Cancer Investig. 2008, 26, 222–229. [Google Scholar] [CrossRef]
- Karnoub, A.E.; Dash, A.B.; Vo, A.P.; Sullivan, A.; Brooks, M.W.; Bell, G.W.; Richardson, A.L.; Polyak, K.; Tubo, R.; Weinberg, R.A. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 2007, 449, 557. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Xiao, C.; Tan, L.; Wang, Q.; Li, X.; Feng, Y. Cancer-associated fibroblasts induce epithelial–mesenchymal transition of breast cancer cells through paracrine TGF-β signalling. Br. J. Cancer 2014, 110, 724. [Google Scholar] [CrossRef] [Green Version]
- Orimo, A.; Gupta, P.B.; Sgroi, D.C.; Arenzana-Seisdedos, F.; Delaunay, T.; Naeem, R.; Carey, V.J.; Richardson, A.L.; Weinberg, R.A. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 2005, 121, 335–348. [Google Scholar] [CrossRef]
- Reya, T.; Morrison, S.J.; Clarke, M.F.; Weissman, I.L. Stem cells, cancer, and cancer stem cells. Nature 2001, 414, 105–111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCarthy, N. Enforced compliance. Nat. Rev. Cancer 2012, 12, 153. [Google Scholar] [CrossRef] [PubMed]
- Ohshio, Y.; Teramoto, K.; Hanaoka, J.; Tezuka, N.; Itoh, Y.; Asai, T.; Daigo, Y.; Ogasawara, K. Cancer-associated fibroblast-targeted strategy enhances antitumor immune responses in dendritic cell-based vaccine. Cancer Sci. 2015, 106, 134–142. [Google Scholar] [CrossRef]
- Albini, A.; Bruno, A.; Gallo, C.; Pajardi, G.; Noonan, D.M.; Dallaglio, K. Cancer stem cells and the tumor microenvironment: Interplay in tumor heterogeneity. Connect. Tissue Res. 2015, 56, 414–425. [Google Scholar] [CrossRef] [Green Version]
- Economopoulou, P.; Kaklamani, V.G.; Siziopikou, K. The role of cancer stem cells in breast cancer initiation and progression: Potential cancer stem cell-directed therapies. Oncologist 2012, 17, 1394–1401. [Google Scholar] [CrossRef] [Green Version]
- Malanchi, I.; Santamaria-Martínez, A.; Susanto, E.; Peng, H.; Lehr, H.-A.; Delaloye, J.-F.; Huelsken, J. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 2012, 481, 85. [Google Scholar] [CrossRef] [PubMed]
- Guille, A.; Chaffanet, M.; Birnbaum, D. Signaling pathway switch in breast cancer. Cancer Cell Int. 2013, 13, 66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heinrich, P.C.; Behrmann, I.; Muller-Newen, G.; Schaper, F.; Graeve, L. Interleukin-6-type cytokine signalling through the gp130/Jak/STAT pathway. Biochem. J. 1998, 334 Pt 2, 297–314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arthan, D.; Hong, S.K.; Park, J.I. Leukemia inhibitory factor can mediate Ras/Raf/MEK/ERK-induced growth inhibitory signaling in medullary thyroid cancer cells. Cancer Lett. 2010, 297, 31–41. [Google Scholar] [CrossRef]
- Graf, U.; Casanova, E.A.; Cinelli, P. The role of the leukemia inhibitory factor (LIF)—Pathway in derivation and maintenance of murine pluripotent stem cells. Genes 2011, 2, 280–297. [Google Scholar] [CrossRef] [Green Version]
- Metcalf, D. The unsolved enigmas of leukemia inhibitory factor. Stem Cells. 2003, 21, 5–14. [Google Scholar] [CrossRef]
- Hirai, H.; Karian, P.; Kikyo, N. Regulation of embryonic stem cell self-renewal and pluripotency by leukaemia inhibitory factor. Biochem. J. 2011, 438, 11–23. [Google Scholar] [CrossRef] [Green Version]
- Arici, A.; Engin, O.; Attar, E.; Olive, D.L. Modulation of leukemia inhibitory factor gene expression and protein biosynthesis in human endometrium. J. Clin. Endocrinol. Metab. 1995, 80, 1908–1915. [Google Scholar] [CrossRef]
- Dazai, S.; Akita, S.; Hirano, A.; Rashid, M.A.; Naito, S.; Akino, K.; Fujii, T. Leukemia inhibitory factor enhances bone formation in calvarial bone defect. J. Craniofacial Surg. 2000, 11, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, M.K.; Cui, X.; Hu, Z.Y.; Jackson, J.; Sherman, S.; Seiger, A.; Wahlberg, L.U. In vitro expansion of a multipotent population of human neural progenitor cells. Exp. Neurol. 1999, 158, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Spangenburg, E.E.; Booth, F.W. Multiple signaling pathways mediate LIF-induced skeletal muscle satellite cell proliferation. Am. J. Physiol. Cell Physiol. 2002, 283, C204–C211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaziri, N.; Shariati, L.; Javanmard, S.H. Leukemia inhibitory factor: A main controller of breast cancer. J. Biosci. 2020, 45, 1–11. [Google Scholar] [CrossRef]
- Li, X.; Yang, Q.; Yu, H.; Wu, L.; Zhao, Y.; Zhang, C.; Yue, X.; Liu, Z.; Wu, H.; Haffty, B.G.; et al. LIF promotes tumorigenesis and metastasis of breast cancer through the AKT-mTOR pathway. Oncotarget 2014, 5, 788–801. [Google Scholar] [CrossRef] [Green Version]
- Quaglino, A.; Schere-Levy, C.; Romorini, L.; Meiss, R.P.; Kordon, E.C. Mouse mammary tumors display Stat3 activation dependent on leukemia inhibitory factor signaling. Breast Cancer Res. BCR 2007, 9, R69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yue, X.; Zhao, Y.; Zhang, C.; Li, J.; Liu, Z.; Liu, J.; Hu, W. Leukemia inhibitory factor promotes EMT through STAT3-dependent miR-21 induction. Oncotarget 2016, 7, 3777. [Google Scholar] [CrossRef] [Green Version]
- Zeng, H.; Qu, J.; Jin, N.; Xu, J.; Lin, C.; Chen, Y.; Yang, X.; He, X.; Tang, S.; Lan, X. Feedback activation of leukemia inhibitory factor receptor limits response to histone deacetylase inhibitors in breast cancer. Cancer Cell 2016, 30, 459–473. [Google Scholar] [CrossRef] [Green Version]
- Ohata, Y.; Tsuchiya, M.; Hirai, H.; Yamaguchi, S.; Akashi, T.; Sakamoto, K.; Yamaguchi, A.; Ikeda, T.; Kayamori, K. Leukemia inhibitory factor produced by fibroblasts within tumor stroma participates in invasion of oral squamous cell carcinoma. PLoS ONE 2018, 13, e0191865. [Google Scholar] [CrossRef]
- Gangemi, R.M.R.; Griffero, F.; Marubbi, D.; Perera, M.; Capra, M.C.; Malatesta, P.; Ravetti, G.L.; Zona, G.L.; Daga, A.; Corte, G. SOX2 silencing in glioblastoma tumor-initiating cells causes stop of proliferation and loss of tumorigenicity. Stem Cells 2009, 27, 40–48. [Google Scholar] [CrossRef] [Green Version]
- Chiou, S.-H.; Wang, M.-L.; Chou, Y.-T.; Chen, C.-J.; Hong, C.-F.; Hsieh, W.-J.; Chang, H.-T.; Chen, Y.-S.; Lin, T.-W.; Hsu, H.-S. Coexpression of Oct4 and Nanog enhances malignancy in lung adenocarcinoma by inducing cancer stem cell–like properties and epithelial–mesenchymal transdifferentiation. Cancer Res. 2010, 70, 10433–10444. [Google Scholar] [CrossRef] [Green Version]
- Brooks, M.D.; Burness, M.L.; Wicha, M.S. Therapeutic Implications of Cellular Heterogeneity and Plasticity in Breast Cancer. Cell Stem Cell 2015, 17, 260–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doherty, M.R.; Smigiel, J.M.; Junk, D.J.; Jackson, M.W. Cancer Stem Cell Plasticity Drives Therapeutic Resistance. Cancers. 2016, 8, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vermeulen, L.; de Sousa e Melo, F.; Richel, D.J.; Medema, J.P. The developing cancer stem-cell model: Clinical challenges and opportunities. Lancet Oncol. 2012, 13, e83–e89. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bjornson, C.R.; Rietze, R.L.; Reynolds, B.A.; Magli, M.C.; Vescovi, A.L. Turning brain into blood: A hematopoietic fate adopted by adult neural stem cells in vivo. Science 1999, 283, 534–537. [Google Scholar] [CrossRef] [Green Version]
- Xie, G.; Yao, Q.; Liu, Y.; Du, S.; Liu, A.; Guo, Z.; Sun, A.; Ruan, J.; Chen, L.; Ye, C.; et al. IL-6-induced epithelial-mesenchymal transition promotes the generation of breast cancer stem-like cells analogous to mammosphere cultures. Int. J. Oncol. 2012, 40, 1171–1179. [Google Scholar] [CrossRef] [Green Version]
- Zhou, J.; Zhang, C.; Pan, J.; Chen, L.; Qi, S.T. Interleukin-6 induces an epithelial-mesenchymal transition phenotype in human adamantinomatous craniopharyngioma cells and promotes tumor cell migration. Mol. Med. Rep. 2017, 15, 4123–4131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, S.C.; Chang, Y.S. Role of leukemia inhibitory factor in nasopharyngeal carcinogenesis. Mol. Cell. Oncol. 2014, 1, e29900. [Google Scholar] [CrossRef] [PubMed]
- Bolt, R.; Foran, B.; Murdoch, C.; Lambert, D.W.; Thomas, S.; Hunter, K.D. HPV-negative, but not HPV-positive, oropharyngeal carcinomas induce fibroblasts to support tumour invasion through micro-environmental release of HGF and IL-6. Carcinogenesis 2017, 39, 170–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jobe, N.P.; Rösel, D.; Dvořánková, B.; Kodet, O.; Lacina, L.; Mateu, R.; Smetana, K.; Brábek, J. Simultaneous blocking of IL-6 and IL-8 is sufficient to fully inhibit CAF-induced human melanoma cell invasiveness. Histochem. Cell Biol. 2016, 146, 205–217. [Google Scholar] [CrossRef]
- Nagasaki, T.; Hara, M.; Nakanishi, H.; Takahashi, H.; Sato, M.; Takeyama, H. Interleukin-6 released by colon cancer-associated fibroblasts is critical for tumour angiogenesis: Anti-interleukin-6 receptor antibody suppressed angiogenesis and inhibited tumour–stroma interaction. Br. J. Cancer 2014, 110, 469. [Google Scholar] [CrossRef] [Green Version]
- Qiao, Y.; Zhang, C.; Li, A.; Wang, D.; Luo, Z.; Ping, Y.; Zhou, B.; Liu, S.; Li, H.; Yue, D.; et al. IL6 derived from cancer-associated fibroblasts promotes chemoresistance via CXCR7 in esophageal squamous cell carcinoma. Oncogene 2018, 37, 873–883. [Google Scholar] [CrossRef]
- Albrengues, J.; Bourget, I.; Pons, C.; Butet, V.; Hofman, P.; Tartare-Deckert, S.; Feral, C.C.; Meneguzzi, G.; Gaggioli, C. LIF mediates proinvasive activation of stromal fibroblasts in cancer. Cell Rep. 2014, 7, 1664–1678. [Google Scholar] [CrossRef] [Green Version]
- Franco, O.E.; Jiang, M.; Strand, D.W.; Peacock, J.; Fernandez, S.; Jackson, R.S., 2nd; Revelo, M.P.; Bhowmick, N.A.; Hayward, S.W. Altered TGF-beta signaling in a subpopulation of human stromal cells promotes prostatic carcinogenesis. Cancer Res. 2011, 71, 1272–1281. [Google Scholar] [CrossRef] [Green Version]
- Kalluri, R.; Zeisberg, M. Fibroblasts in cancer. Nat. Rev. Cancer 2006, 6, 392–401. [Google Scholar] [CrossRef]
- Wang, J.; Boddupalli, A.; Koelbl, J.; Nam, D.H.; Ge, X.; Bratlie, K.M.; Schneider, I.C. Degradation and remodeling of epitaxially grown collagen fibrils. Cell. Mol. Bioeng. 2019, 12, 69–84. [Google Scholar] [CrossRef]
- Wang, K.; Wu, F.; Seo, B.R.; Fischbach, C.; Chen, W.; Hsu, L.; Gourdon, D. Breast cancer cells alter the dynamics of stromal fibronectin-collagen interactions. Matrix Biol. 2017, 60, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Mannello, F.; Tonti, G.A. Concise review: No breakthroughs for human mesenchymal and embryonic stem cell culture: Conditioned medium, feeder layer, or feeder-free; medium with fetal calf serum, human serum, or enriched plasma; serum-free, serum replacement nonconditioned medium, or ad hoc formula? All that glitters is not gold! Stem Cells 2007, 25, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
- Adorno-Cruz, V.; Kibria, G.; Liu, X.; Doherty, M.; Junk, D.J.; Guan, D.; Hubert, C.; Venere, M.; Mulkearns-Hubert, E.; Sinyuk, M. Cancer stem cells: Targeting the roots of cancer, seeds of metastasis, and sources of therapy resistance. Cancer Res. 2015, 75, 924–929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, W.J.; Ho, C.C.; Chang, Y.L.; Chen, H.Y.; Lin, C.A.; Ling, T.Y.; Yu, S.L.; Yuan, S.S.; Chen, Y.J.; Lin, C.Y.; et al. Cancer-associated fibroblasts regulate the plasticity of lung cancer stemness via paracrine signalling. Nat. Commun. 2014, 5, 3472. [Google Scholar] [CrossRef] [PubMed]


| Gene | Primer Sequence (5′-3′) | Amplicon Size | Annealing Temperature (°C) |
|---|---|---|---|
| GAPDH | Forward 5′-AGTCCACTGGCGTCTTCA-3′ Reverse 5′-GAGGCATTGCTGATGATCT-3′ | 163 | 58 55 |
| Oct4 | Forward 5′-GGGGGTTCTATTTGGGAAG-3′ Reverse 5′-TTGTCAGCTTCCTCCACCC-3′ | 126 | 57 59 |
| Nanog | Forward 5′-CAGCTACAAACAGGTGAAGACC-3′ Reverse 5′-GGTGGTAGGAAGAGTAAAGGC-3′ | 146 | 62 61 |
| LIF | Forward 5′-GCCCTCTTTATTCTCTATTACACAG-3′ Reverse 5′-ACACGACTATGCGGTACAGC-3′ | 151 | 63 60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vaziri, N.; Shariati, L.; Zarrabi, A.; Farazmand, A.; Haghjooy Javanmard, S. Cancer-Associated Fibroblasts Regulate the Plasticity of Breast Cancer Stemness through the Production of Leukemia Inhibitory Factor. Life 2021, 11, 1298. https://doi.org/10.3390/life11121298
Vaziri N, Shariati L, Zarrabi A, Farazmand A, Haghjooy Javanmard S. Cancer-Associated Fibroblasts Regulate the Plasticity of Breast Cancer Stemness through the Production of Leukemia Inhibitory Factor. Life. 2021; 11(12):1298. https://doi.org/10.3390/life11121298
Chicago/Turabian StyleVaziri, Nazanin, Laleh Shariati, Ali Zarrabi, Ali Farazmand, and Shaghayegh Haghjooy Javanmard. 2021. "Cancer-Associated Fibroblasts Regulate the Plasticity of Breast Cancer Stemness through the Production of Leukemia Inhibitory Factor" Life 11, no. 12: 1298. https://doi.org/10.3390/life11121298
APA StyleVaziri, N., Shariati, L., Zarrabi, A., Farazmand, A., & Haghjooy Javanmard, S. (2021). Cancer-Associated Fibroblasts Regulate the Plasticity of Breast Cancer Stemness through the Production of Leukemia Inhibitory Factor. Life, 11(12), 1298. https://doi.org/10.3390/life11121298









