Promoter Demethylation Upregulates STEAP1 Gene Expression in Human Prostate Cancer: In Vitro and In Silico Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Prostate Cell Lines and Treatments
2.2. Patients and Tissue Sample Collection
2.3. DNA Extraction and Bisulfite Conversion
2.4. Polymerase Chain Reaction (PCR) Amplification and Cloning Products
2.5. DNA Sequencing
2.6. Datasets and Bioinformatic Analysis
2.7. Total RNA Extraction, cDNA Synthesis and Quantitative Real-Time PCR (qPCR)
2.8. Statistical Analysis
3. Results
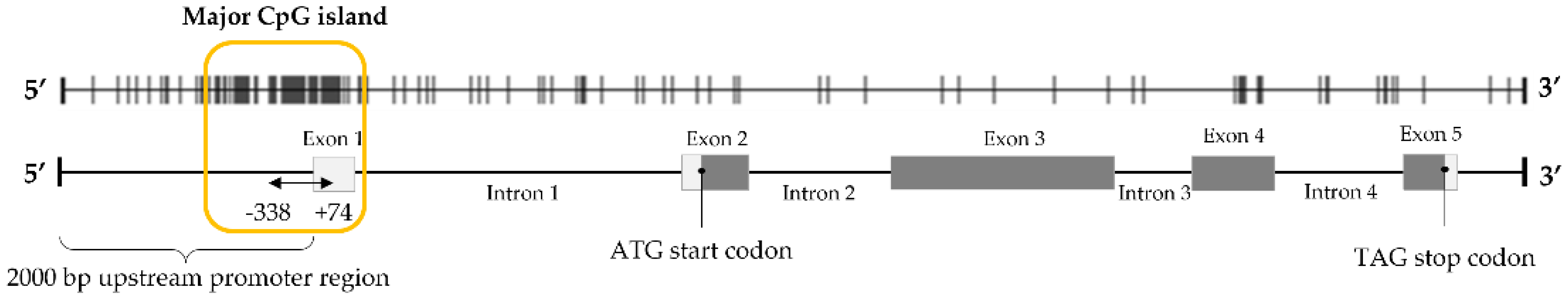
3.1. Methylation Analysis of STEAP1 Gene in Neoplastic Tissue/Cells Compared with Non-Neoplastic Cells
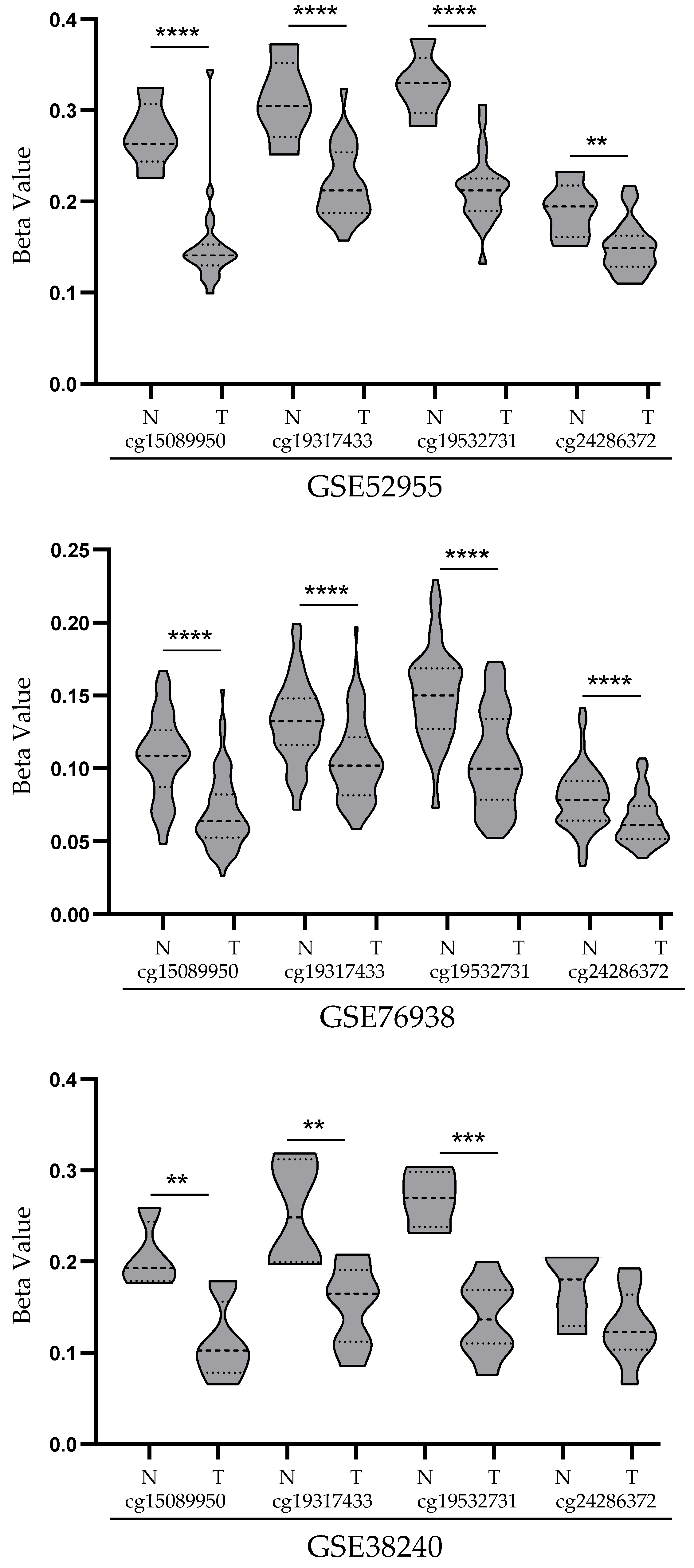
3.2. Analysis of STEAP1 Promoter Methylation Levels in PCa and Normal Prostate Tissues from the GEO Database
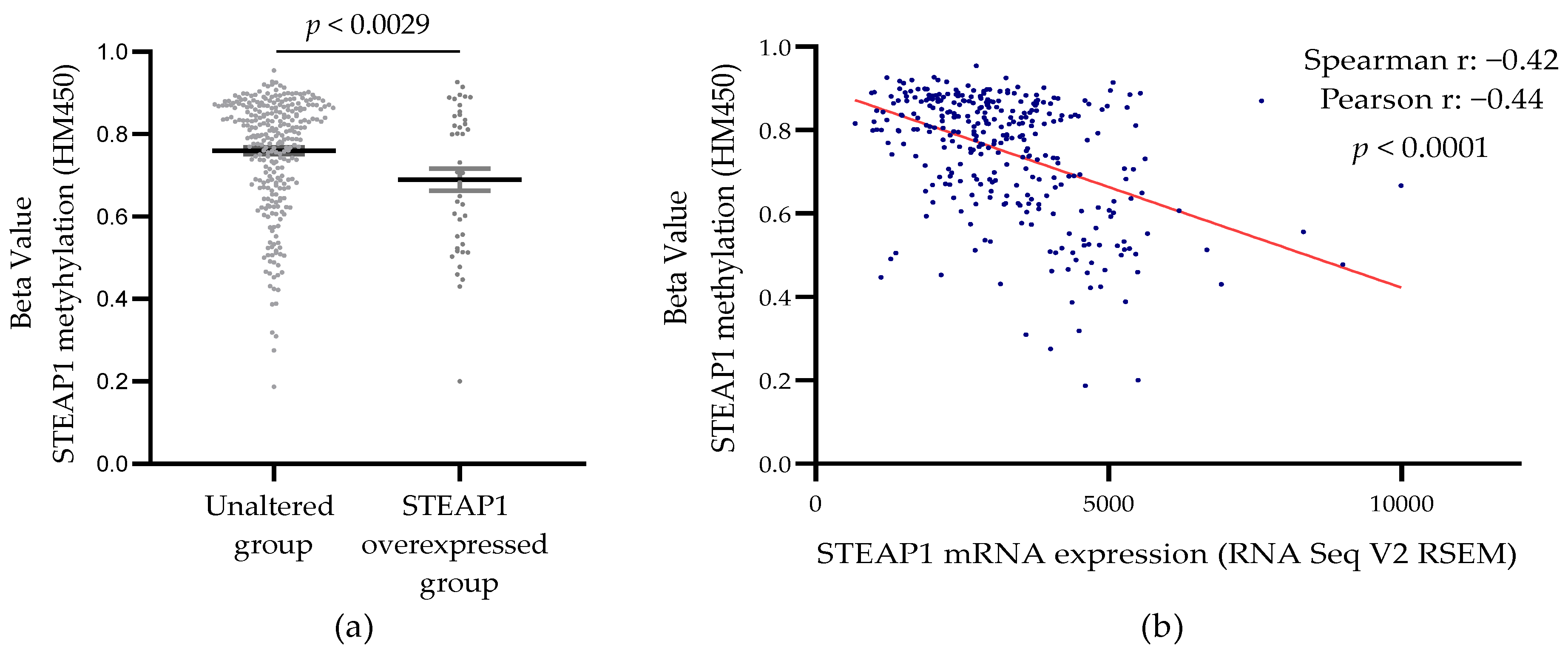
3.3. Correlation between STEAP1 Gene Promoter Demethylation and Its Expression in PCa Tissue from the TCGA Database
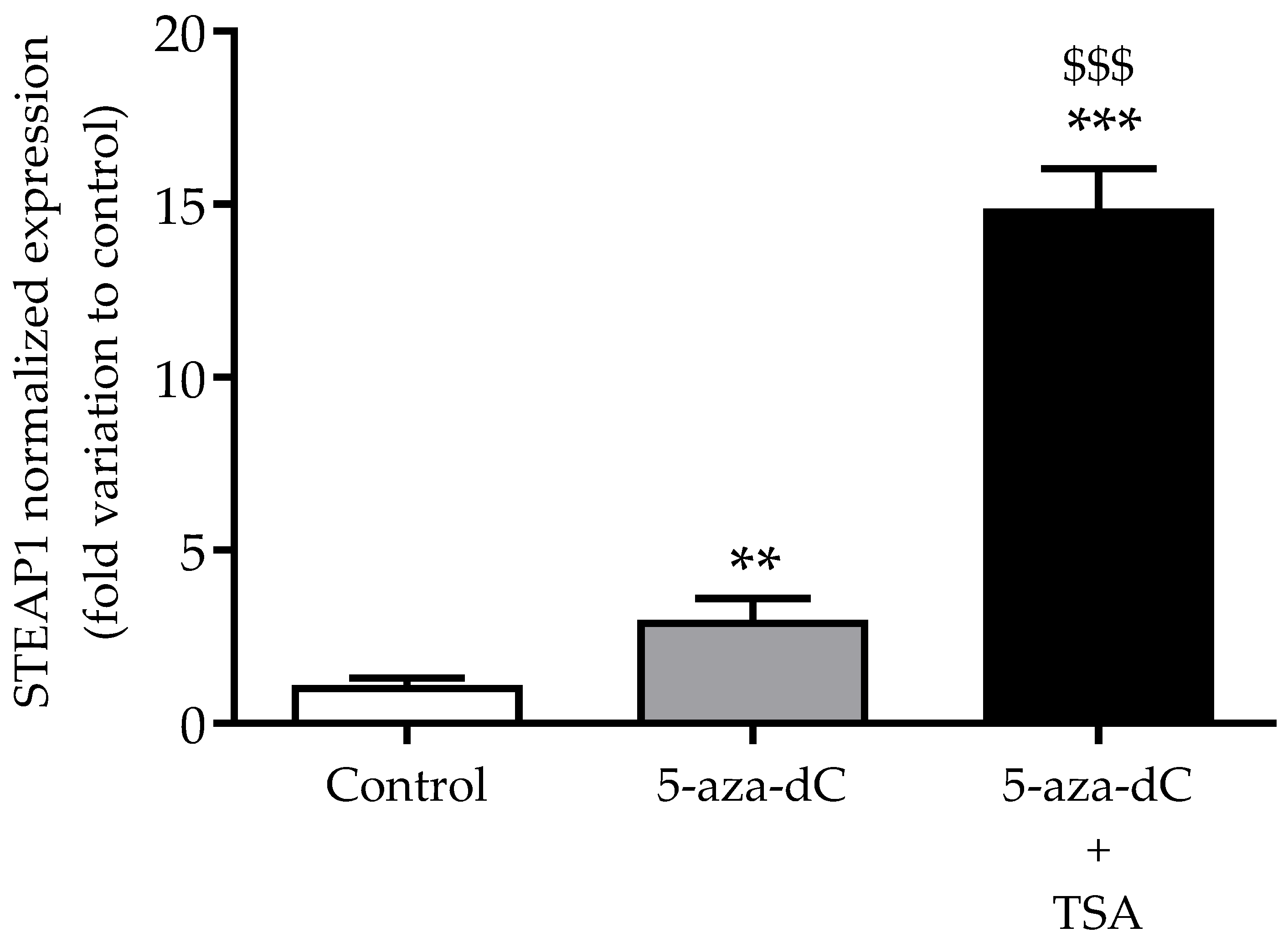
3.4. Effect of Epigenetic-Modulating Drugs in STEAP1 Gene Expression in Non-Neoplastic PNT1A Cells
3.5. Analysis of Co-Expression between STEAP1 mRNA Expression and HDACs
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Culp, M.B.B.; Soerjomataram, I.; Efstathiou, J.A.; Bray, F.; Jemal, A. Recent Global Patterns in Prostate Cancer Incidence and Mor-tality Rates. In European Urology; Elsevier BV: Amsterdam, The Netherlands, 2020; Volume 77, pp. 38–52. [Google Scholar]
- Gonzalgo, M.L.; Isaacs, W.B. Molecular Pathways to Prostate Cancer. J. Urol. 2003, 170, 2444–2452. [Google Scholar] [CrossRef]
- Reynolds, M.A. Molecular alterations in prostate cancer. Cancer Lett. 2008, 271, 13–24. [Google Scholar] [CrossRef]
- Joshua, A.M.; Evans, A.; Van der Kwast, T.; Zielenska, M.; Meeker, A.K.; Chinnaiyan, A.; Squire, J.A. Prostatic preneoplasia and beyond. Biochim. Biophys. Acta. 2008, 1785, 156–181. [Google Scholar] [CrossRef]
- Nelson, W.G.; De Marzo, A.M.; Yegnasubramanian, S. Epigenetic alterations in human prostate cancers. Endocrinology 2009, 150, 3991–4002. [Google Scholar] [CrossRef] [Green Version]
- Wang, R.; Liu, X. Epigenetic regulation of prostate cancer. Genes Dis. 2019, 7, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Kulis, M.; Esteller, M. DNA methylation and cancer. Adv. Genet. 2010, 70, 27–56. [Google Scholar] [PubMed]
- Hubert, R.S.; Vivanco, I.; Chen, E.; Rastegar, S.; Leong, K.; Mitchell, S.C.; Madraswala, R.; Zhou, Y.; Kuo, J.; Raitano, A.B.; et al. STEAP: A prostate-specific cell-surface antigen highly expressed in human prostate tumors. Proc. Natl. Acad. Sci. USA 1999, 96, 14523–14528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamamoto, T.; Tamura, Y.; Kobayashi, J.-I.; Kamiguchi, K.; Hirohashi, Y.; Miyazaki, A.; Torigoe, T.; Asanuma, H.; Hiratsuka, H.; Sato, N. Six-transmembrane epithelial antigen of the prostate-1 plays a role for in vivo tumor growth via intercellular communication. Exp. Cell Res. 2013, 319, 2617–2626. [Google Scholar] [CrossRef]
- Challita-Eid, P.M.; Morrison, K.; Etessami, S.; An, Z.; Morrison, K.J.; Perez-Villar, J.J.; Raitano, A.B.; Jia, X.C.; Gudas, J.M.; Kanner, S.B.; et al. Monoclonal antibodies to six-transmembrane epithelial antigen of the prostate-1 inhibit intercellular communication in vitro and growth of human tumor xenografts in vivo. Cancer Res. 2007, 67, 5798–5805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grunewald, T.G.P.; Diebold, I.; Esposito, I.; Plehm, S.; Hauer, K.; Thiel, U.; Da Silva-Buttkus, P.; Neff, F.; Unland, R.; Müller-Tidow, C.; et al. STEAP1 is associated with the invasive and oxidative stress phenotype of ewing tumors. Mol. Cancer Res. 2012, 10, 52–65. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Hou, W.; Zhang, C.; Tan, Y.; Zhang, D.; An, W.; Pan, S.; Wu, W.; Chen, Q.; Xu, H. A research of STEAP1 regulated gastric cancer cell proliferation, migration and invasion in vitro and in vivos. J. Cell. Mol. Med. 2020, 24, 14217–14230. [Google Scholar] [CrossRef]
- Huo, S.-F.; Shang, W.-L.; Yu, M.; Ren, X.-P.; Wen, H.-X.; Chai, C.-Y.; Sun, L.; Hui, K.; Liu, L.-H.; Wei, S.-H.; et al. STEAP1 facilitates metastasis and epithelial-mesenchymal transition of lung adenocarcinoma via the JAK2/STAT3 signaling pathway. Biosci. Rep. 2020, 40, BSR20193169. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Z.; Huang, L.; Sun, J.; Xie, J.; Wang, T.; Yin, X.; Zhang, H.; Chen, J. Six-transmembrane epithelial antigen of the prostate 1 ex-pression promotes ovarian cancer metastasis by aiding progression of epithelial-to-mesenchymal transition. Histochem. Cell Biol. 2020, 154, 215–230. [Google Scholar] [CrossRef] [PubMed]
- Gomes, I.M.; Rocha, S.; Gaspar, C.; Alvelos, M.I.; Santos, C.R.; Socorro, S.; Maia, C.J. Knockdown of STEAP1 inhibits cell growth and induces apoptosis in LNCaP prostate cancer cells counteracting the effect of androgens. Med. Oncol. 2018, 35, 40. [Google Scholar] [CrossRef]
- Gomes, I.M.; Santos, C.R.; Maia, C.J. Expression of STEAP1 and STEAP1B in prostate cell lines, and the putative regulation of STEAP1 by post-transcriptional and post-translational mechanisms. Genes Cancer 2014, 5, 142–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The Cancer Genome Atlas Research Network. The Molecular Taxonomy of Primary Prostate Cancer. Cell 2015, 163, 1011–1025. [Google Scholar] [CrossRef] [Green Version]
- Ramalho-Carvalho, J.; Gonçalves, C.; Graça, I.; Bidarra, D.; Pereira-Silva, E.; Salta, S.; Godinho, M.I.; Gomez, A.; Esteller, M.; Costa, B.; et al. A multiplatform approach identifies miR-152-3p as a common epigenetically regulated onco-suppressor in prostate cancer targeting TMEM97. Clin. Epigenetics 2018, 10, 40. [Google Scholar] [CrossRef]
- Kirby, M.K.; Ramaker, R.C.; Roberts, B.S.; Lasseigne, B.N.; Gunther, D.S.; Burwell, T.C.; Davis, N.S.; Gulzar, Z.G.; Absher, D.M.; Cooper, S.J.; et al. Genome-wide DNA methylation measurements in prostate tissues uncovers novel prostate cancer diagnostic biomarkers and transcription factor binding patterns. BMC Cancer 2017, 17, 273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aryee, M.J.; Liu, W.; Engelmann, J.C.; Nuhn, P.; Gurel, M.; Haffner, M.C.; Esopi, D.; Irizarry, R.A.; Getzenberg, R.H.; Nelson, W.G.; et al. DNA methylation alterations exhibit intra-individual stability and inter-individual heterogeneity in prostate cancer metastases. Sci. Transl. Med. 2013, 5, 169ra10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
- Abbas, A.; Gupta, S. The role of histone deacetylases in prostate cancer. Epigenetics 2008, 3, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Kelly, T.K.; Jones, P.A. Epigenetics in cancer. Carcinogenesis 2010, 31, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Kukkonen, K.; Taavitsainen, S.; Huhtala, L.; Uusi-Makela, J.; Granberg, K.; Nykter, M.; Urbanucci, A. Chromatin and Epigenetic Dysregulation of Prostate Cancer Development, Progression, and Therapeutic Response. Cancers 2021, 13, 3325. [Google Scholar] [CrossRef]
- Darwiche, N. Epigenetic mechanisms and the hallmarks of cancer: An intimate affair. Am. J. Cancer Res. 2020, 10, 1954–1978. [Google Scholar] [PubMed]
- Wang, Q.; Williamson, M.; Bott, S.; Brookman-Amissah, N.; Freeman, A.; Nariculam, J.; Hubank, M.; Ahmed, A.; Masters, J.R. Hypomethylation of WNT5A, CRIP1 and S100P in prostate cancer. Oncogene 2007, 26, 6560–6565. [Google Scholar] [CrossRef] [Green Version]
- He, Y.; Cui, Y.; Wang, W.; Gu, J.; Guo, S.; Ma, K.; Luo, X. Hypomethylation of the hsa-miR-191 locus causes high expression of hsa-mir-191 and promotes the epithelial-to-mesenchymal transition in hepatocellular carcinoma. Neoplasia 2011, 13, 841–853. [Google Scholar] [CrossRef] [Green Version]
- Yamada, N.; Noguchi, S.; Kumazaki, M.; Shinohara, H.; Miki, K.; Naoe, T.; Akao, Y. Epigenetic regulation of microRNA-128a ex-pression contributes to the apoptosis-resistance of human T-cell leukaemia jurkat cells by modulating expression of fas-associated protein with death domain (FADD). Biochim. Biophys. Acta. 2014, 1843, 590–602. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; Lv, X.; Jiang, Y.; Li, G.; Qiao, Q. Identification of aberrantly methylated differentially expressed genes in glioblastoma multiforme and their association with patient survival. Exp. Ther. Med. 2019, 18, 2140–2152. [Google Scholar] [CrossRef] [Green Version]
- Tamura, T.; Chiba, J. STEAP4 regulates focal adhesion kinase activation and CpG motifs within STEAP4 promoter region are frequently methylatedin DU145, human androgen-independent prostate cancer cells. Int. J. Mol. Med. 2009, 24, 599–604. [Google Scholar] [CrossRef] [Green Version]
- Yamada, N.; Yasui, K.; Dohi, O.; Gen, Y.; Tomie, A.; Kitaichi, T.; Iwai, N.; Mitsuyoshi, H.; Sumida, Y.; Moriguchi, M.; et al. Genome-wide DNA methylation analysis in hepatocellular carcinoma. Oncol. Rep. 2016, 35, 2228–2236. [Google Scholar] [CrossRef] [Green Version]
- Esteller, M. Cancer epigenomics: DNA methylomes and histone-modification maps. Nat. Rev. Genet. 2007, 8, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Di Croce, L.; Raker, V.A.; Corsaro, M.; Fazi, F.; Fanelli, M.; Faretta, M.; Fuks, F.; Lo Coco, F.; Kouzarides, T.; Nervi, C.; et al. Methyltransferase recruitment and DNA hypermethylation of target promoters by an oncogenic transcription factor. Science 2002, 295, 1079–1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Alessio, A.C.; Szyf, M. Epigenetic tête-à-tête: The bilateral relationship between chromatin modifications and DNA methylation. Biochem. Cell Biol. 2006, 84, 463–476. [Google Scholar] [CrossRef]
- Minucci, S.; Pelicci, P.G. Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat. Rev. Cancer 2006, 6, 38–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.-Y.; Kim, J.-S. A short guide to histone deacetylases including recent progress on class II enzymes. Exp. Mol. Med. 2020, 52, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wang, B.; Xu, Y.; Zhang, J. Autotaxin is induced by TSA through HDAC3 and HDAC7 inhibition and antagonizes the TSA-induced cell apoptosis. Mol. Cancer 2011, 10, 18. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Jin, X.; Ma, J.; Ding, D.; Huang, Z.; Sheng, H.; Yan, Y.; Pan, Y.; Wei, T.; Wang, L.; et al. HDAC5 Loss Impairs RB Repression of Pro-Oncogenic Genes and Confers CDK4/6 Inhibitor Resistance in Cancer. Cancer Res. 2021, 81, 1486–1499. [Google Scholar] [CrossRef]
- Yang, Y.; Tse, A.K.-W.; Li, P.; Ma, Q.; Xiang, S.; Nicosia, S.V.; Seto, E.; Zhang, X.; Bai, W. Inhibition of androgen receptor activity by histone deacetylase 4 through receptor SUMOylation. Oncogene 2011, 30, 2207–2218. [Google Scholar] [CrossRef] [Green Version]
- Hontecillas-Prieto, L.; Flores-Campos, R.; Silver, A.; De Álava, E.; Hajji, N.; García-Domínguez, D.J. Synergistic Enhancement of Cancer Therapy Using HDAC Inhibitors: Opportunity for Clinical Trials. Front. Genet. 2020, 11, 1113. [Google Scholar] [CrossRef]
- Pacheco, M.B.; Camilo, V.; Lopes, N.; Moreira-Silva, F.; Correia, M.P.; Henrique, R.; Jerónimo, C. Hydralazine and Panobinostat Attenuate Malignant Properties of Prostate Cancer Cell Lines. Pharmaceuticals 2021, 14, 670. [Google Scholar] [CrossRef]
- Lakshmaiah, K.C.; Jacob, L.A.; Aparna, S.; Lokanatha, D.; Saldanha, S.C. Epigenetic therapy of cancer with histone deacetylase in-hibitors. J. Cancer Res. Ther. 2014, 10, 469–478. [Google Scholar] [PubMed]
- Rana, Z.; Diermeier, S.; Hanif, M.; Rosengren, R.J. Understanding Failure and Improving Treatment Using HDAC Inhibitors for Prostate Cancer. Biomedicines 2020, 8, 22. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Primers | Accession Number | Sequence | Amplicon Size (bp) | Target |
|---|---|---|---|---|
| STEAP1_−338 fw STEAP1_+74 rv | NC_000007.14 | 5′ AAAGTGTGATTTGGGAATGTTTTT 3′ 5′ TTTTAAGTTAGTTGTAGGTTTT 3′ | 412 | Modified DNA |
| hSTEAP1_619 fw hSTEAP1_747 rv | NM_012449.3 | 5′ GGCGATCCTACAGATACAAGTTGC 3′ 5′ CCAATCCCACAATTCCCAGAGAC 3′ | 128 | mRNA |
| hGAPDH_74 fw hGAPDH_149 rv | NM_002046.7 | 5′ CGCCAGCCGAGCCACATC 3′ 5′ CGC CCA ATA CGA CCA AAT CCG 3′ | 75 | mRNA |
| hβ2M_347 fw hβ2M_439 rv | NM_004048.4 | 5′ ATGAGTATGCCTGCCGTGTG 3′ 5′ CAAACCTCCATGATGCTGCTTAC 3′ | 92 | mRNA |
| Dataset | Platform | Sample Type | Disease Condition (n) | Normal Tissue (n) | Reference |
|---|---|---|---|---|---|
| GSE52955 | GLP13534 | Frozen tissue | Cancer (25) | 5 * | [18] |
| GSE76938 | GLP13534 | Cancer (73) | Adjacent Tissue (63) | [19] | |
| GSE38240 | GLP13534 | Cancer Metastasis (8) | 4 # | [20] |
| STEAP1 Correlated | Spearman’s Correlation | p-Value |
|---|---|---|
| HDAC1 | −0.011 | 0.834 |
| HDAC2 | −0.005 | 0.929 |
| HDAC3 | 0.089 | 0.105 |
| HDAC4 | −0.242 | 8.07 × 10−6 |
| HDAC5 | −0.305 | 1.35 × 10−8 |
| HDAC6 | 0.049 | 0.366 |
| HDAC7 | −0.336 | 3.31 × 10−10 |
| HDAC8 | 0.255 | 2.34 × 10−6 |
| HDAC9 | −0.294 | 4.53 × 10−8 |
| HDAC10 | −0.064 | 0.245 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rocha, S.M.; Sousa, I.; Gomes, I.M.; Arinto, P.; Costa-Pinheiro, P.; Coutinho, E.; Santos, C.R.; Jerónimo, C.; Lemos, M.C.; Passarinha, L.A.; et al. Promoter Demethylation Upregulates STEAP1 Gene Expression in Human Prostate Cancer: In Vitro and In Silico Analysis. Life 2021, 11, 1251. https://doi.org/10.3390/life11111251
Rocha SM, Sousa I, Gomes IM, Arinto P, Costa-Pinheiro P, Coutinho E, Santos CR, Jerónimo C, Lemos MC, Passarinha LA, et al. Promoter Demethylation Upregulates STEAP1 Gene Expression in Human Prostate Cancer: In Vitro and In Silico Analysis. Life. 2021; 11(11):1251. https://doi.org/10.3390/life11111251
Chicago/Turabian StyleRocha, Sandra M., Inês Sousa, Inês M. Gomes, Patrícia Arinto, Pedro Costa-Pinheiro, Eduarda Coutinho, Cecília R. Santos, Carmen Jerónimo, Manuel C. Lemos, Luís A. Passarinha, and et al. 2021. "Promoter Demethylation Upregulates STEAP1 Gene Expression in Human Prostate Cancer: In Vitro and In Silico Analysis" Life 11, no. 11: 1251. https://doi.org/10.3390/life11111251











