Super-Earths, M Dwarfs, and Photosynthetic Organisms: Habitability in the Lab
Abstract
1. Introduction
2. Experimental Aims
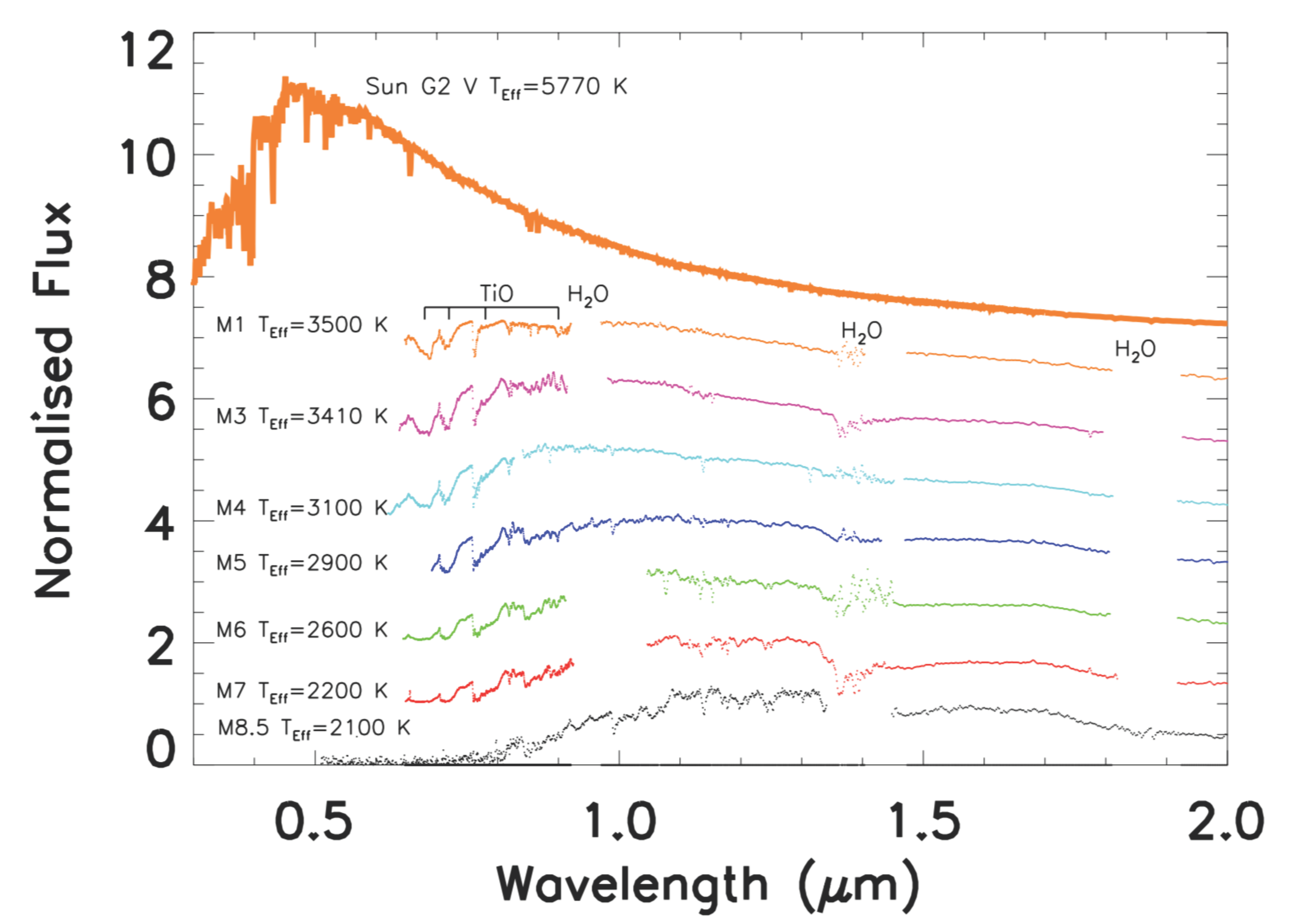
2.1. The Astrophysical Context
2.2. The Experiment Plan
- i
- We set up the laboratory instrumentation and selected the organisms for the tests (see Section 4.1). We built some of the laboratory tools ex-novo. We first built a light source suitable for the purpose of the experiment, simulating the star irradiation (see Section 3.1). Secondly, we built the reaction cell (see Section 3.2).
- ii
- Before conducting the main experiment, we performed a fiducial one considering the terrestrial environment. We irradiated the selected organisms with solar light and within a terrestrial atmosphere environment.
- iii
- Once we checked that the experimental set up functions well in terrestrial condition, we switched to the M star irradiation of organisms, considering a terrestrial atmosphere.
- iv
- Lastly, we are planning to conduct experiments using the M star light to irradiate the cyanobacteria that will be put in a modified atmosphere. The composition is defined using the 1-D model of the atmosphere of super-Earths described by Petralia et al. 2020 [54] and Alei et al. 2020 (submitted).
3. Laboratory Set Up
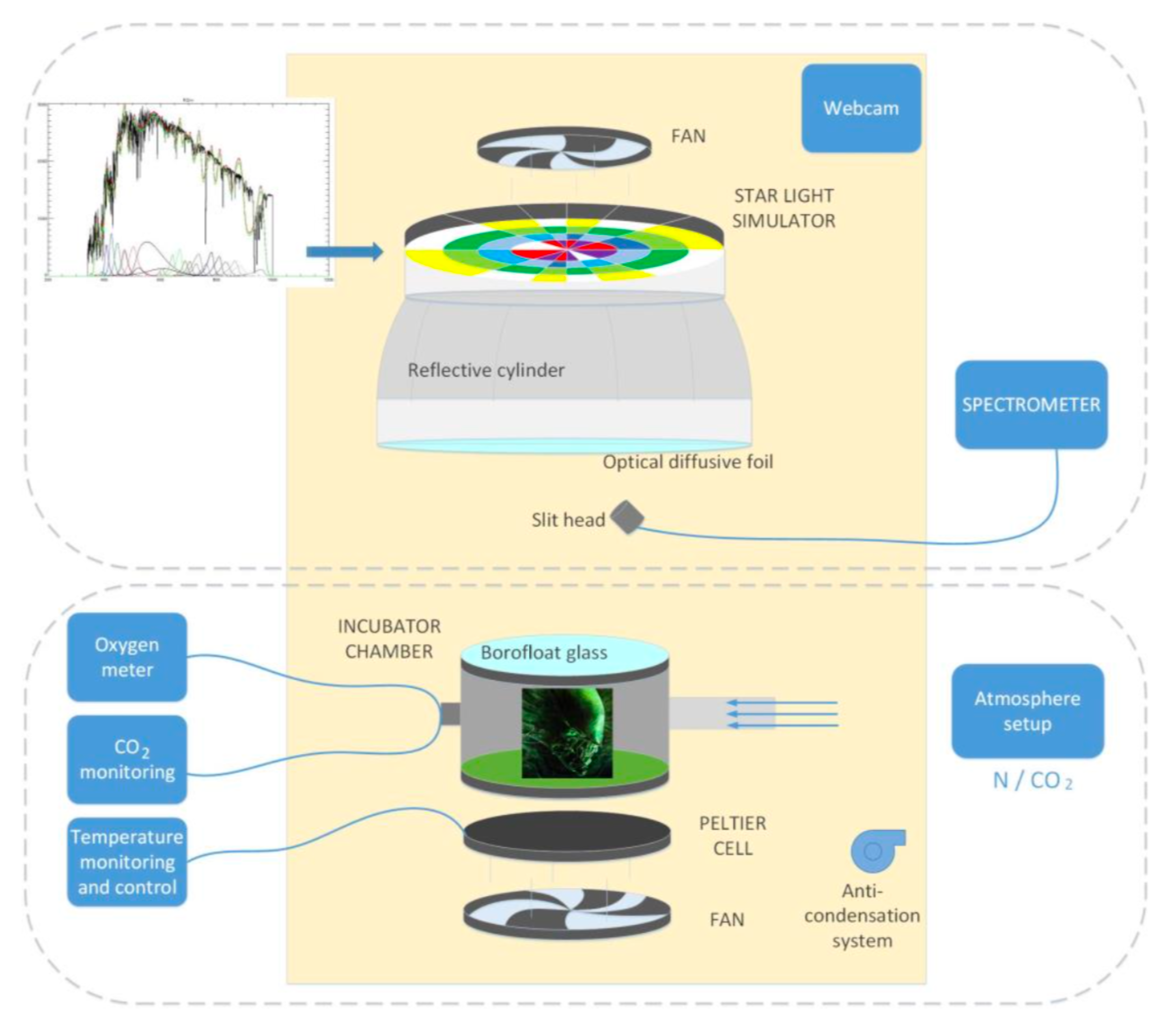
3.1. Star Irradiation Simulator
3.2. The Reaction Cell
3.3. The Control Software
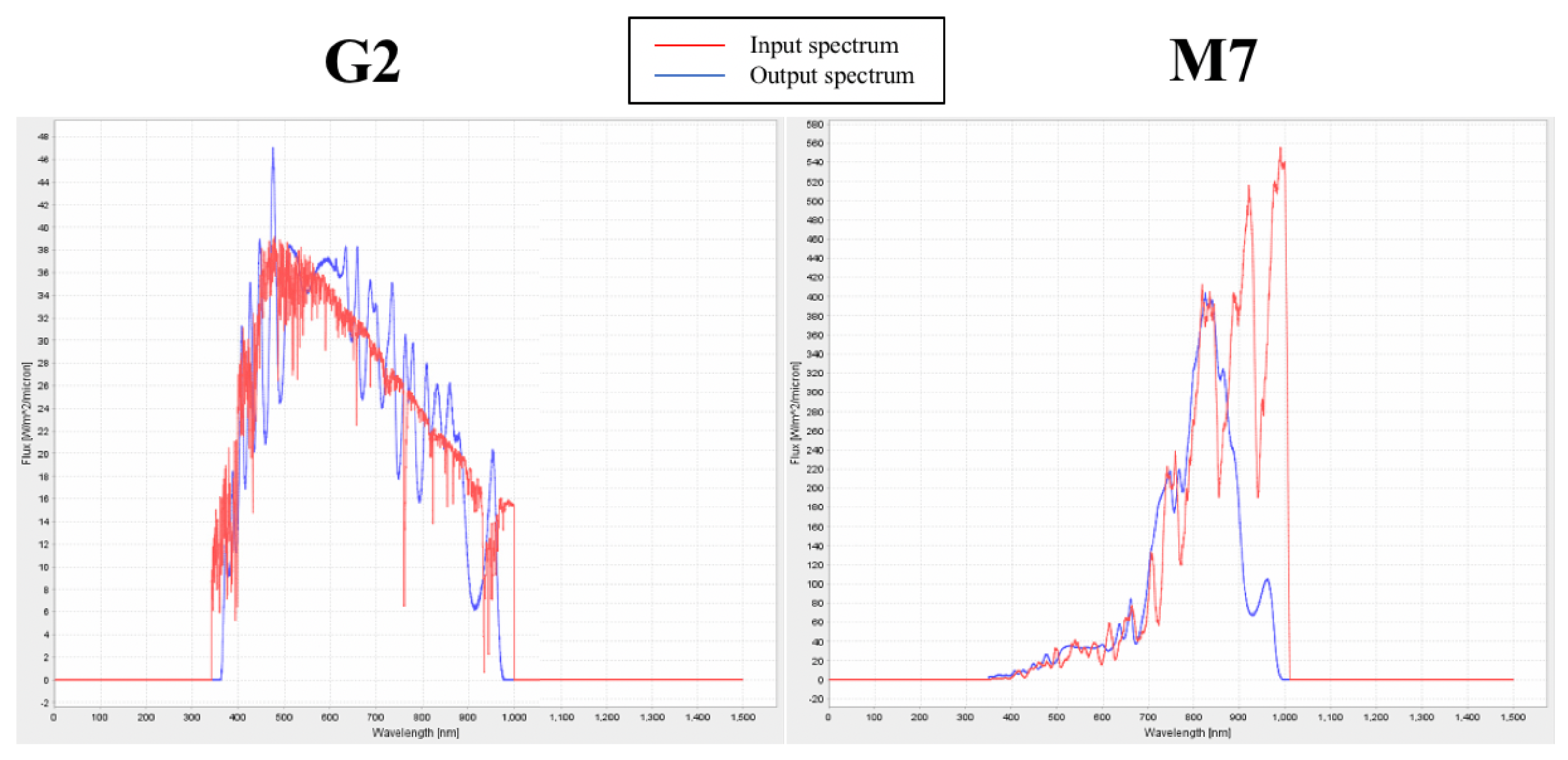
3.4. Validation of the Experimental Set up
4. Biological Experiment as Proof of Concept
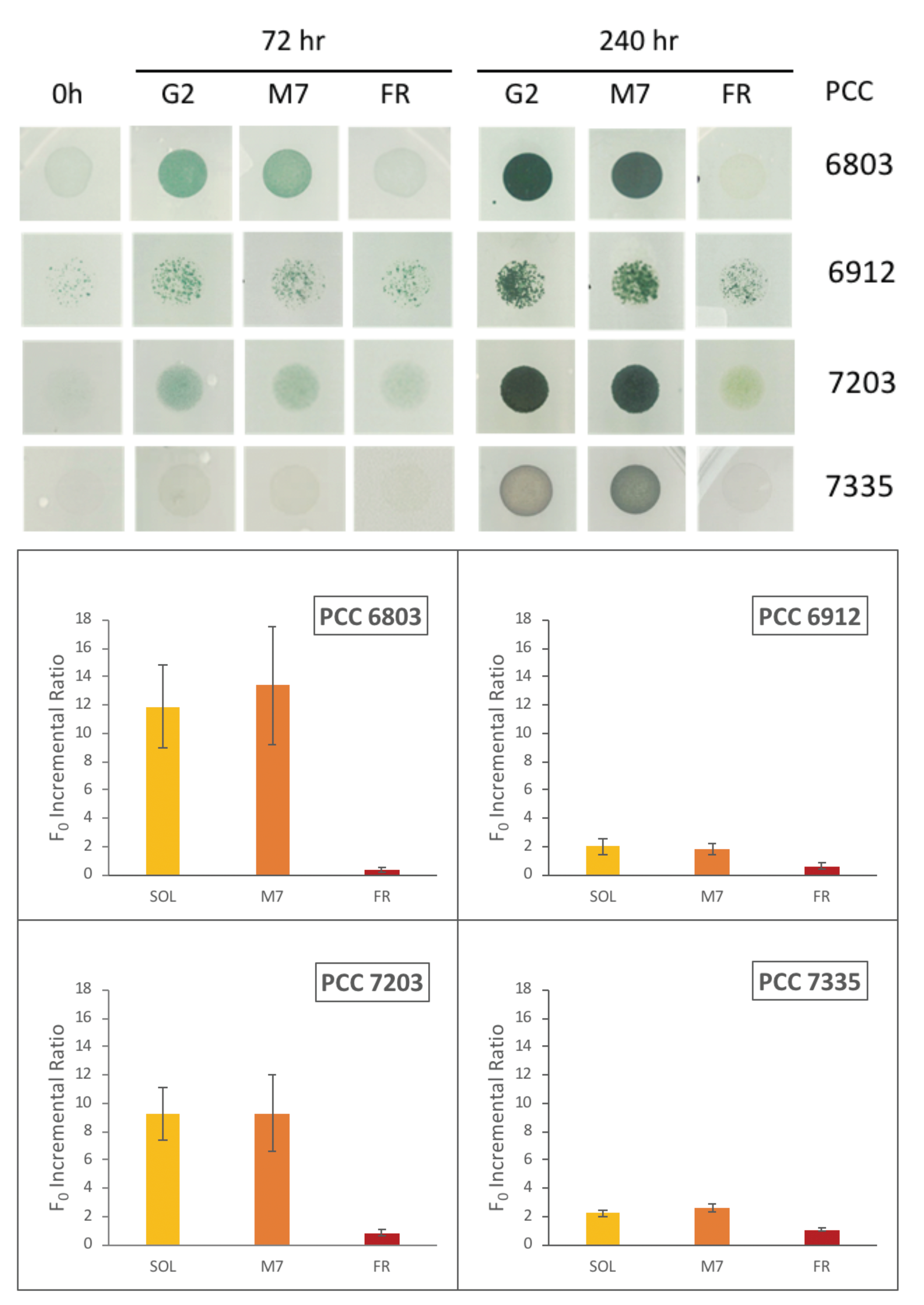
4.1. Selected Organisms
4.2. Growth and Photosynthetic Efficiency
5. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CA | Chromatic Acclimation |
| CCT | Correlated Color Temperature |
| CHL | Chlorophyll |
| COTS | Component Off The Shelf |
| FaRLiP | Far-red light photoacclimation |
| HZ | Habitable Zone |
| JWST | James Webb Space Telescope |
| NIR | near infra-red |
| OD | Optical Density |
| OST | Origin Space Telescope |
| PAM | Pulse-Amplitude Modulation |
| PAR | Photochemically Active Radiation |
| PC | Personal Computer |
| PCC | Pasteur Culture Collection |
| TDLAS | Tunable Diode Laser Absorption Spectroscopy |
| WMS | Wavelength Modulation Spectroscopy |
References
- Dressing, C.D.; Charbonneau, D. The Occurrence Rate of Small Planets around Small Stars. Astrophys. J. 2013, 767, 95. [Google Scholar] [CrossRef]
- Davenport, J.R.A.; Kipping, D.M.; Sasselov, D.; Matthews, J.M.; Cameron, C. MOST Observations of Our Nearest Neighbor: Flares on Proxima Centauri. Astrophys. J. Lett. 2016, 829, L31. [Google Scholar] [CrossRef]
- Mayor, M.; Lovis, C.; Santos, N.C. Doppler spectroscopy as a path to the detection of Earth-like planets. Nature 2014, 513, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Bonfils, X.; Delfosse, X.; Udry, S.; Forveille, T.; Mayor, M.; Perrier, C.; Bouchy, F.; Gillon, M.; Lovis, C.; Pepe, F.; et al. The HARPS search for southern extra-solar planets. XXXI. The M-dwarf sample. Astron. Astrophys. 2013, 549, A109. [Google Scholar] [CrossRef]
- Kopparapu, R.K.; Ramirez, R.; Kasting, J.F.; Eymet, V.; Robinson, T.D.; Mahadevan, S.; Terrien, R.C.; Domagal-Goldman, S.; Meadows, V.; Deshpande, R. Habitable Zones around Main-sequence Stars: New Estimates. Astrophys. J. 2013, 765, 131. [Google Scholar] [CrossRef]
- Seager, S.; Schrenk, M.; Bains, W. An Astrophysical View of Earth-Based Metabolic Biosignature Gases. Astrobiology 2012, 12, 61–82. [Google Scholar] [CrossRef]
- Seager, S.; Bains, W.; Petkowski, J.J. Toward a List of Molecules as Potential Biosignature Gases for the Search for Life on Exoplanets and Applications to Terrestrial Biochemistry. Astrobiology 2016, 16, 465–485. [Google Scholar] [CrossRef]
- Kaltenegger, L. How to Characterize Habitable Worlds and Signs of Life. Annu. Rev. Astron. Astrophys. 2017, 55, 433–485. [Google Scholar] [CrossRef]
- Grenfell, J.L. A review of exoplanetary biosignatures. Phys. Rep. 2017, 713, 1–17. [Google Scholar] [CrossRef]
- Catling, D.C.; Krissansen-Totton, J.; Kiang, N.Y.; Crisp, D.; Robinson, T.D.; DasSarma, S.; Rushby, A.J.; Del Genio, A.; Bains, W.; Domagal-Goldman, S. Exoplanet Biosignatures: A Framework for Their Assessment. Astrobiology 2018, 18, 709–738. [Google Scholar] [CrossRef]
- Schwieterman, E.W.; Kiang, N.Y.; Parenteau, M.N.; Harman, C.E.; DasSarma, S.; Fisher, T.M.; Arney, G.N.; Hartnett, H.E.; Reinhard, C.T.; Olson, S.L.; et al. Exoplanet Biosignatures: A Review of Remotely Detectable Signs of Life. Astrobiology 2018, 18, 663–708. [Google Scholar] [CrossRef] [PubMed]
- Claudi, R.; Alei, E. Biosignatures Search in Habitable Planets. Galaxies 2019, 7, 82. [Google Scholar] [CrossRef]
- Lingam, M.; Loeb, A. Colloquium: Physical constraints for the evolution of life on exoplanets. Rev. Mod. Phys. 2019, 91, 021002. [Google Scholar] [CrossRef]
- Lovelock, J.E. A Physical Basis for Life Detection Experiments. Nature 1965, 207, 568–570. [Google Scholar] [CrossRef]
- Meadows, V.S.; Reinhard, C.T.; Arney, G.N.; Parenteau, M.N.; Schwieterman, E.W.; Domagal-Goldman, S.D.; Lincowski, A.P.; Stapelfeldt, K.R.; Rauer, H.; DasSarma, S.; et al. Exoplanet Biosignatures: Understanding Oxygen as a Biosignature in the Context of Its Environment. Astrobiology 2018, 18, 630–662. [Google Scholar] [CrossRef] [PubMed]
- Lederberg, J. Signs of Life: Criterion-System of Exobiology. Nature 1965, 207, 9–13. [Google Scholar] [CrossRef]
- Seager, S.; Bains, W. The search for signs of life on exoplanets at the interface of chemistry and planetary science. Sci. Adv. 2015, 1, e1500047. [Google Scholar] [CrossRef]
- Harman, C.E.; Schwieterman, E.W.; Schottelkotte, J.C.; Kasting, J.F. Abiotic O2 Levels on Planets around F, G, K, and M Stars: Possible False Positives for Life? Astrophys. J. 2015, 812, 137. [Google Scholar] [CrossRef]
- Gates, D.M.; Gates, H.J.; Gates, J.C.; Gates, V.R. Spectral properties of plants. Appl. Opt. 1965, 4, 11. [Google Scholar] [CrossRef]
- Gan, F.; Bryant, D. Adaptive and acclimative responses of cyanobacteria to far-red light. Environ. Microbiol. 2015, 17, 3450–3465. [Google Scholar] [CrossRef]
- Wolstencroft, R.D.; Raven, J.A. Photosynthesis: Likelihood of Occurrence and Possibility of Detection on Earth-like Planets. Icarus 2002, 157, 535–548. [Google Scholar] [CrossRef]
- Tinetti, G.; Rashby, S.; Yung, Y.L. Detectability of Red-Edge-shifted Vegetation on Terrestrial Planets Orbiting M Stars. Astrophys. J. Lett. 2006, 644, L129–L132. [Google Scholar] [CrossRef]
- Kiang, N.Y.; Segura, A.; Tinetti, G.; Govindjee; Blankenship, R.E.; Cohen, M.; Siefert, J.; Crisp, D.; Meadows, V.S. Spectral Signatures of Photosynthesis. II. Coevolution with Other Stars And The Atmosphere on Extrasolar Worlds. Astrobiology 2007, 7, 252–274. [Google Scholar] [CrossRef] [PubMed]
- Claudi, R.; Erculiani, M.S.; Galletta, G.; Billi, D.; Pace, E.; Schierano, D.; Giro, E.; D’Alessandro, M. Simulating super earth atmospheres in the laboratory. Int. J. Astrobiol. 2016, 15, 35–44. [Google Scholar] [CrossRef]
- Pecaut, M.J.; Mamajek, E.E. Intrinsic Colors, Temperatures, and Bolometric Corrections of Pre-main-sequence Stars. Astrophys. J. Suppl. 2013, 208, 9. [Google Scholar] [CrossRef]
- Leggett, S.K.; Allard, F.; Dahn, C.; Hauschildt, P.H.; Kerr, T.H.; Rayner, J. Spectral Energy Distributions for Disk and Halo M Dwarfs. Astrophys. J. 2000, 535, 965–974. [Google Scholar] [CrossRef]
- Leggett, S.K.; Allard, F.; Berriman, G.; Dahn, C.C.; Hauschildt, P.H. Infrared Spectra of Low-Mass Stars: Toward a Temperature Scale for Red Dwarfs. Astrophys. J. Suppl. 1996, 104, 117. [Google Scholar] [CrossRef]
- Kasting, J.F.; Whitmire, D.P.; Reynolds, R.T. Habitable Zones around Main Sequence Stars. Icarus 1993, 101, 108–128. [Google Scholar] [CrossRef]
- Selsis, F.; Kasting, J.F.; Levrard, B.; Paillet, J.; Ribas, I.; Delfosse, X. Habitable planets around the star Gliese 581? Astron. Astrophys. 2007, 476, 1373–1387. [Google Scholar] [CrossRef]
- Brown, S.P.; Mead, A.J.; Forgan, D.H.; Raven, J.A.; Cockell, C.S. Photosynthetic potential of planets in 3 : 2 spin-orbit resonances. Int. J. Astrobiol. 2014, 13, 279–289. [Google Scholar] [CrossRef]
- Lingam, M.; Loeb, A. Photosynthesis on habitable planets around low-mass stars. Mon. Not. R. Astron. Soc. 2019, 485, 5924–5928. [Google Scholar] [CrossRef]
- Heath, M.J.; Doyle, L.R.; Joshi, M.M.; Haberle, R.M. Habitability of Planets Around Red Dwarf Stars. Orig. Life Evol. Biosph. 1999, 29, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Lissauer, J.J. Planets Formed in Habitable Zones of M Dwarf Stars Probably Are Deficient in Volatiles. Astrophys. J. Lett. 2007, 660, L149–L152. [Google Scholar] [CrossRef]
- Raymond, S.N.; Scalo, J.; Meadows, V.S. A Decreased Probability of Habitable Planet Formation around Low-Mass Stars. Astrophys. J. 2007, 669, 606–614. [Google Scholar] [CrossRef]
- Luger, R.; Barnes, R. Extreme Water Loss and Abiotic O2Buildup on Planets Throughout the Habitable Zones of M Dwarfs. Astrobiology 2015, 15, 119–143. [Google Scholar] [CrossRef]
- Tian, F.; Ida, S. Water contents of Earth-mass planets around M dwarfs. Nat. Geosci. 2015, 8, 177–180. [Google Scholar] [CrossRef]
- Gillon, M.; Jehin, E.; Lederer, S.M.; Delrez, L.; de Wit, J.; Burdanov, A.; Van Grootel, V.; Burgasser, A.J.; Triaud, A.H.M.J.; Opitom, C.; et al. Temperate Earth-sized planets transiting a nearby ultracool dwarf star. Nature 2016, 533, 221–224. [Google Scholar] [CrossRef]
- Bolmont, E.; Libert, A.S.; Leconte, J.; Selsis, F. Habitability of planets on eccentric orbits: Limits of the mean flux approximation. Astron. Astrophys. 2016, 591, A106. [Google Scholar] [CrossRef]
- Scalo, J.; Kaltenegger, L.; Segura, A.G.; Fridlund, M.; Ribas, I.; Kulikov, Y.N.; Grenfell, J.L.; Rauer, H.; Odert, P.; Leitzinger, M.; et al. M Stars as Targets for Terrestrial Exoplanet Searches And Biosignature Detection. Astrobiology 2007, 7, 85–166. [Google Scholar] [CrossRef]
- Segura, A.; Kasting, J.F.; Meadows, V.; Cohen, M.; Scalo, J.; Crisp, D.; Butler, R.A.H.; Tinetti, G. Biosignatures from Earth-Like Planets Around M Dwarfs. Astrobiology 2005, 5, 706–725. [Google Scholar] [CrossRef]
- Segura, A.; Walkowicz, L.M.; Meadows, V.; Kasting, J.; Hawley, S. The Effect of a Strong Stellar Flare on the Atmospheric Chemistry of an Earth-like Planet Orbiting an M Dwarf. Astrobiology 2010, 10, 751–771. [Google Scholar] [CrossRef] [PubMed]
- France, K.; Froning, C.S.; Linsky, J.L.; Roberge, A.; Stocke, J.T.; Tian, F.; Bushinsky, R.; Désert, J.M.; Mauas, P.; Vieytes, M.; et al. The Ultraviolet Radiation Environment around M dwarf Exoplanet Host Stars. Astrophys. J. 2013, 763, 149. [Google Scholar] [CrossRef]
- Cuntz, M.; Guinan, E.F. About Exobiology: The Case for Dwarf K Stars. Astrophys. J. 2016, 827, 79. [Google Scholar] [CrossRef]
- O’Malley-James, J.T.; Kaltenegger, L. UV Surface Environments of M Star Planets: Surface Habitability and Temporal Biosignatures. In Proceedings of the Habitable Worlds 2017: A System Science Workshop, Laramie, WY, USA, 13–17 November 2017; Volume 2042, p. 4128. [Google Scholar]
- O’Malley-James, J.T.; Kaltenegger, L. Biofluorescent Worlds-II. Biological fluorescence induced by stellar UV flares, a new temporal biosignature. Mon. Not. R. Astron. Soc. 2019, 488, 4530–4545. [Google Scholar] [CrossRef]
- Tarter, J.C.; Backus, P.R.; Mancinelli, R.L.; Aurnou, J.M.; Backman, D.E.; Basri, G.S.; Boss, A.P.; Clarke, A.; Deming, D.; Doyle, L.R.; et al. A Reappraisal of The Habitability of Planets around M Dwarf Stars. Astrobiology 2007, 7, 30–65. [Google Scholar] [CrossRef] [PubMed]
- Gale, J.; Wandel, A. The potential of planets orbiting red dwarf stars to support oxygenic photosynthesis and complex life. Int. J. Astrobiol. 2017, 16, 1–9. [Google Scholar] [CrossRef]
- Ranjan, S.; Sasselov, D.D. Influence of the UV Environment on the Synthesis of Prebiotic Molecules. Astrobiology 2016, 16, 68–88. [Google Scholar] [CrossRef]
- Mullan, D.J.; Bais, H.P. Photosynthesis on a Planet Orbiting an M Dwarf: Enhanced Effectiveness during Flares. Astrophys. J. 2018, 865, 101. [Google Scholar] [CrossRef]
- Dressing, C.D.; Charbonneau, D. The Occurrence of Potentially Habitable Planets Orbiting M Dwarfs Estimated from the Full Kepler Dataset and an Empirical Measurement of the Detection Sensitivity. Astrophys. J. 2015, 807, 45. [Google Scholar] [CrossRef]
- Gillon, M.; Triaud, A.H.M.J.; Demory, B.O.; Jehin, E.; Agol, E.; Deck, K.M.; Lederer, S.M.; de Wit, J.; Burdanov, A.; Ingalls, J.G.; et al. Seven temperate terrestrial planets around the nearby ultracool dwarf star TRAPPIST-1. Nature 2017, 542, 456–460. [Google Scholar] [CrossRef]
- Anglada-Escudé, G.; Amado, P.J.; Barnes, J.; Berdiñas, Z.M.; Butler, R.P.; Coleman, G.A.L.; de La Cueva, I.; Dreizler, S.; Endl, M.; Giesers, B.; et al. A terrestrial planet candidate in a temperate orbit around Proxima Centauri. Nature 2016, 536, 437–440. [Google Scholar] [CrossRef] [PubMed]
- Miller-Ricci, E.; Seager, S.; Sasselov, D. The Atmospheric Signatures of Super-Earths: How to Distinguish Between Hydrogen-Rich and Hydrogen-Poor Atmospheres. Astrophys. J. 2009, 690, 1056–1067. [Google Scholar] [CrossRef]
- Petralia, A.; Alei, E.; Aresu, G.; Locci, D.; Cecchi-Pestellini, C.; Micela, G.; Claudi, R.; Ciaravella, A. A systematic study of CO2 planetary atmospheres and their link to the stellar environment. Mon. Not. R. Astron. Soc. 2020, 496, 5350–5359. [Google Scholar] [CrossRef]
- Bula, R.; Morrow, R.; Tibbitts, T.; Barta, D.; Ignatius, R.; Martin, T. Light-emitting Diodes as a Radiation Source for Plants. HortScience 1991, 26, 203–205. [Google Scholar] [CrossRef] [PubMed]
- Trivellin, N.; Barbisan, D.; Ferretti, M.; Erculiani, M.S.; Claudi, R.U.; Giro, E.; Bonato, M.; Cocola, L.; Poletto, L.; Salasnich, B.; et al. Adaptive multi-wavelength LED star simulator for space life studies. In Light-Emitting Diodes: Materials, Devices, and Applications for Solid State Lighting XX; Jeon, H., Tu, L.W., Krames, M.R., Strassburg, M., Eds.; SPIE: Bellingham, WA, USA, 2016; p. 976815. [Google Scholar] [CrossRef]
- Salasnich, B.; Claudi, R.U.; Alei, E.; Barbisan, D.; Baruffolo, A.; Cocola, L.; Erculiani, M.S.; La Rocca, N.; Pace, E.; Poletto, L.; et al. Control software for the Multi-Channel Led starlight simulator. In Software and Cyberinfrastructure for Astronomy V; SPIE: Bellingham, WA, USA, 2018; Volume 10707, p. 107071I. [Google Scholar] [CrossRef]
- Danilović, B.; Cocola, L.; Fedel, M.; Poletto, L.; Savic̀, D. Formation and Cumulation of CO2 in the Bottles of the Fermented Milk Drinks. IPCBEE 2016, 95, 26. [Google Scholar] [CrossRef]
- Battistuzzi, M.; Cocola, L.; Salasnich, B.; Erculiani, M.S.; Alei, E.; Morosinotto, T.; Claudi, R.; Poletto, L.; La Rocca, N. A New Remote Sensing-Based System for the Monitoring and Analysis of Growth and Gas Exchange Rates of Photosynthetic Microorganisms Under Simulated Non-Terrestrial Conditions. Front. Plant Sci. 2020, 11, 182. [Google Scholar] [CrossRef]
- Erculiani, M.S.; Claudi, R.; Barbisan, D.; Giro, E.; Bonato, M.; Cocola, L.; Farisato, G.; Meneghini, M.; Poletto, L.; Salasnich, B.; et al. A tunable integrated system to simulate colder stellar radiation. In Optical Systems Design 2015: Optical Design and Engineering VI; Mazuray, L., Wartmann, R., Wood, A.P., Eds.; SPIE: Bellingham, WA, USA, 2015; Volume 9626, p. 96262D. [Google Scholar] [CrossRef]
- Allakhverdiev, S.; Kreslavskii, V.; Zharmukhamedov, S.; Voloshin, R.; Korol’kova, D.; Tomo, T.; Shen, J.R. Chlorophylls d and f and Their Role in Primary Photosynthetic Processes of Cyanobacteria. Biochemistry 2016, 81, 201–212. [Google Scholar] [CrossRef]
- de Vera, J.P.; Dulai, S.; Kereszturi, A.; Koncz, L.; Lorek, A.; Mohlmann, D.; Marschall, M.; Pocs, T. Results on the survival of cryptobiotic cyanobacteria samples after exposure to Mars-like environmental conditions. Int. J. Astrobiol. 2014, 13, 35–44. [Google Scholar] [CrossRef]
- Billi, D.; Staibano, C.; Verseux, C.; Fagliarone, C.; Mosca, C.; Baqué, M.; Rabbow, E.; Rettberg, P. Dried Biofilms of Desert Strains of Chroococcidiopsis Survived Prolonged Exposure to Space and Mars-like Conditions in Low Earth Orbit. Astrobiology 2019, 19, 1008–1017. [Google Scholar] [CrossRef]
- Billi, D.; Baqué, M.; Smith, H.; McKay, C. Cyanobacteria from Extreme Deserts to Space. Adv. Microbiol. 2013, 3, 80–86. [Google Scholar] [CrossRef]
- McKay, C. Thickness of tropical ice and photosynthesis on a snowball earth. Geophys. Res. Lett. 2000, 27, 2153–2156. [Google Scholar] [CrossRef] [PubMed]
- Cockell, C.S.; Raven, J.A. Zones of photosynthetic potential on Mars and the early Earth. Icarus 2004, 169, 300–310. [Google Scholar] [CrossRef]
- Wolf, B.; Blankenship, R. Far-red light acclimation in diverse oxygenic photosynthetic organisms. Photosynth. Res. 2019, 142, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Airs, R.; Temperton, B.; Sambles, C.; Farnham, G.; Skill, S.; Llewellyn, C. Chlorophyll f and chlorophyll d are produced in the cyanobacterium Chlorogloeopsis fritschii when cultured under natural light and near-infrared radiation. FEBS Lett. 2014, 588, 3770–3777. [Google Scholar] [CrossRef]
- Cumbers, J.; Rothschild, L.J. Salt tolerance and polyphyly in the cyanobacterium Chroococcidiopsis (Pleurocapsales). J. Phycol. 2014, 50, 472–482. [Google Scholar] [CrossRef]
- Zhao, C.; Gan, F.; Shen, G.; Bryant, D.A. RfpA, RfpB, and RfpC are the Master Control Elements of Far-Red Light Photoacclimation (FaRLiP). Front. Microbiol. 2015, 6, 1303. [Google Scholar] [CrossRef]
- Ho, M.; Gan, F.; Shen, G.; Zhao, C.; Bryant, D.A. Far-red light photoacclimation (FaRLiP) in Synechococcus sp. PCC 7335: I. Regulation of FaRLiP gene expression. Photosynth. Res. 2017, 131, 173–186. [Google Scholar] [CrossRef]
- Rippka, R.; Deruelles, J.; Waterbury, J.B.; Herdman, M.; Stanier, R.Y. Generic Assignments, Strain Histories and Properties of Pure Cultures of Cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Misumi, M.; Katoh, H.; Tomo, T.; Sonoike, K. Relationship between Photochemical Quenching and Non-Photochemical Quenching in Six Species of Cyanobacteria Reveals Species Difference in Redox State and Species Commonality in Energy Dissipation. Plant Cell Physiol. 2015, 57, 1510–1517. [Google Scholar] [CrossRef]
- Perin, G.; Bellan, A.; Segalla, A.; Meneghesso, A.; Alboresi, A.; Morosinotto, T. Generation of random mutants to improve light-use efficiency of Nannochloropsis gaditana cultures for biofuel production. Biotechnol. Biofuels 2015, 8, 161. [Google Scholar] [CrossRef]
- Llewellyn, C.A.; Airs, R.L.; Farnham, G.; Greig, C. Synthesis, Regulation and Degradation of Carotenoids Under Low Level UV-B Radiation in the Filamentous Cyanobacterium Chlorogloeopsis fritschii PCC 6912. Front. Microbiol. 2020, 11, 163. [Google Scholar] [CrossRef] [PubMed]
- Kiang, N.Y.; Siefert, J.; Govindjee; Blankenship, R.E. Spectral Signatures of Photosynthesis. I. Review of Earth Organisms. Astrobiology 2007, 7, 222–251. [Google Scholar] [CrossRef]
- Takizawa, K.; Minagawa, J.; Tamura, M.; Kusakabe, N.; Narita, N. Red-edge position of habitable exoplanets around M-dwarfs. Scientific Reports 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Lingam, M.; Loeb, A. Photosynthesis on exoplanets and exomoons from reflected light. Int. J. Astrobiol. 2020, 19, 210–219. [Google Scholar] [CrossRef]
- Miyashita, H.; Ikemoto, H.; Kurano, N. Chlorophyll d as a major pigment. Nature 1996, 383, 402. [Google Scholar] [CrossRef]
- Chen, M.; Schliep, M.; Willows, R.D.; Cai, Z.L.; Neilan, B.A.; Scheer, H. A Red-Shifted Chlorophyll. Science 2010, 329, 1318. [Google Scholar] [CrossRef]
- Gan, F.; Zhang, S.; Rockwell, N.C.; Martin, S.S.; Lagarias, J.C.; Bryant, D.A. Extensive remodeling of a cyanobacterial photosynthetic apparatus in far-red light. Science 2014, 345, 1312–1317. [Google Scholar] [CrossRef]
- Ho, M.Y.; Niedzwiedzki, D.M.; MacGregor-Chatwin, C.; Gerstenecker, G.; Hunter, C.N.; Blankenship, R.E.; Bryant, D.A. Extensive remodeling of the photosynthetic apparatus alters energy transfer among photosynthetic complexes when cyanobacteria acclimate to far-red light. Biochim. Biophys. Acta Bioenerg. 2020, 1861, 148064. [Google Scholar] [CrossRef]
- Nürnberg, D.J.; Morton, J.; Santabarbara, S.; Telfer, A.; Joliot, P.; Antonaru, L.A.; Ruban, A.V.; Cardona, T.; Krausz, E.; Boussac, A.; et al. Photochemistry beyond the red limit in chlorophyll f-containing photosystems. Science 2018, 360, 1210–1213. [Google Scholar] [CrossRef]
- Ho, M.Y.; Bryant, D.A. Global Transcriptional Profiling of the Cyanobacterium Chlorogloeopsis fritschii PCC 9212 in Far-Red Light: Insights Into the Regulation of Chlorophyll d Synthesis. Front. Microbiol. 2019, 10, 465. [Google Scholar] [CrossRef]
- Kurashov, V.; Ho, M.Y.; Shen, G.; Piedl, K.; Laremore, T.N.; Bryant, D.A.; Golbeck, J.H. Energy transfer from chlorophyll f to the trapping center in naturally occurring and engineered Photosystem I complexes. Photosynth. Res. 2019, 141, 151–163. [Google Scholar] [CrossRef]
- Catling, D.C.; Kasting, J.F. Atmospheric Evolution on Inhabited and Lifeless Worlds; Cambridge University Press: Cambridge, UK, 2017. [Google Scholar]
- Kaltenegger, L.; Selsis, F.; Fridlund, M.; Lammer, H.; Beichman, C.; Danchi, W.; Eiroa, C.; Henning, T.; Herbst, T.; Léger, A.; et al. Deciphering Spectral Fingerprints of Habitable Exoplanets. Astrobiology 2010, 10, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Lehmer, O.R.; Catling, D.C.; Parenteau, M.N.; Hoehler, T.M. The Productivity of Oxygenic Photosynthesis around Cool, M Dwarf Stars. Astrophys. J. 2018, 859, 171. [Google Scholar] [CrossRef]

| Nominal (nm) | Measured (nm) | LEDs Number | Tot. Luminosity @0.7A&25 (W) | LED Type |
|---|---|---|---|---|
| 365.0 | 368.0 | 5 | 2.735 | Engin LZI-00U600 |
| 385.0 | 390.0 | 15 | 2.140 | Lumileds LHUV-0380-0200 |
| 405.0 | 404.0 | 10 | 6.030 | Lumileds LHUV-0400-0500 |
| 425.0 | 424.0 | 10 | 8.770 | Lumileds LHUV-0420-0650 |
| 447.5 | 450.0 | 10 | 6.980 | Lumileds LXZ1-PR01 |
| 470.0 | 476.0 | 10 | 4.670 | Lumileds LXZ1-PB01 |
| 485.0 | 469.0 | 7 | 5.470 | Osram CRBP-HXIX-47-1 |
| 505.0 | 499.0 | 25 | 7.400 | Lumileds LXZ1-PE01 |
| 530.0 | 520.0 | 10 | 2.260 | Lumileds LXZ1-PM01 |
| 567.5 | 548.0 | 45 | 22.500 | Lumileds LXZ1-PX01 |
| 590.0 | 604.0 | 20 | 1.530 | Lumileds LXZ1-PL01 |
| 627.0 | 634.0 | 10 | 3.780 | Lumileds LXZ1-PD01 |
| 655.0 | 665.0 | 10 | 4.150 | Lumileds LXZ1-PA01 |
| 680.0 | 689.0 | 14 | 2.814 | Roithner SMB1N-680 |
| 700.0 | 708.0 | 10 | 2.070 | Roithner SMB1N-700 |
| 720.0 | 727.0 | 11 | 2.220 | Roithner SMBIN-720D |
| 740.0 | 738.0 | 8 | 4.100 | Engin LZ1-00R300 |
| 760.0 | 763.0 | 6 | 2.390 | Roithner SMB1N-760D |
| 780.0 | 777.0 | 8 | 3.390 | Roithner SMB1N-780D |
| 810.0 | 807.0 | 8 | 4.060 | Roithner SMB1N-810D |
| 830.0 | 834.0 | 15 | 3.860 | Roithner SMB1N-830N |
| 870.0 | 871.0 | 6 | 3.920 | Osram SFH 47155 |
| 880.0 | 889.0 | 16 | 4.700 | Roithner SMB1N-880 |
| 940.0 | 972.0 | 9 | 5.820 | Osram SFH 4725S |
| white (2200 K) | 605.0 | 14 | 5.820 | Lumileds 997-LXZ1-2280-5-2200 |
| Light Source | PCC | |||
|---|---|---|---|---|
| 6803 | 6912 | 7203 | 7335 | |
| SOL | ||||
| M7 | ||||
| FR | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Claudi, R.; Alei, E.; Battistuzzi, M.; Cocola, L.; Erculiani, M.S.; Pozzer, A.C.; Salasnich, B.; Simionato, D.; Squicciarini, V.; Poletto, L.; et al. Super-Earths, M Dwarfs, and Photosynthetic Organisms: Habitability in the Lab. Life 2021, 11, 10. https://doi.org/10.3390/life11010010
Claudi R, Alei E, Battistuzzi M, Cocola L, Erculiani MS, Pozzer AC, Salasnich B, Simionato D, Squicciarini V, Poletto L, et al. Super-Earths, M Dwarfs, and Photosynthetic Organisms: Habitability in the Lab. Life. 2021; 11(1):10. https://doi.org/10.3390/life11010010
Chicago/Turabian StyleClaudi, Riccardo, Eleonora Alei, Mariano Battistuzzi, Lorenzo Cocola, Marco Sergio Erculiani, Anna Caterina Pozzer, Bernardo Salasnich, Diana Simionato, Vito Squicciarini, Luca Poletto, and et al. 2021. "Super-Earths, M Dwarfs, and Photosynthetic Organisms: Habitability in the Lab" Life 11, no. 1: 10. https://doi.org/10.3390/life11010010
APA StyleClaudi, R., Alei, E., Battistuzzi, M., Cocola, L., Erculiani, M. S., Pozzer, A. C., Salasnich, B., Simionato, D., Squicciarini, V., Poletto, L., & La Rocca, N. (2021). Super-Earths, M Dwarfs, and Photosynthetic Organisms: Habitability in the Lab. Life, 11(1), 10. https://doi.org/10.3390/life11010010






