Diurnal Regulation of In Vivo Localization and CO2-Fixing Activity of Carboxysomes in Synechococcus elongatus PCC 7942
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains, Generation of Constructs, and Culture Conditions
2.2. Carbon-Fixation Assays
2.3. Circadian Bioluminescence Monitoring
2.4. Fluorescence Microscopy and Data Analysis
3. Results
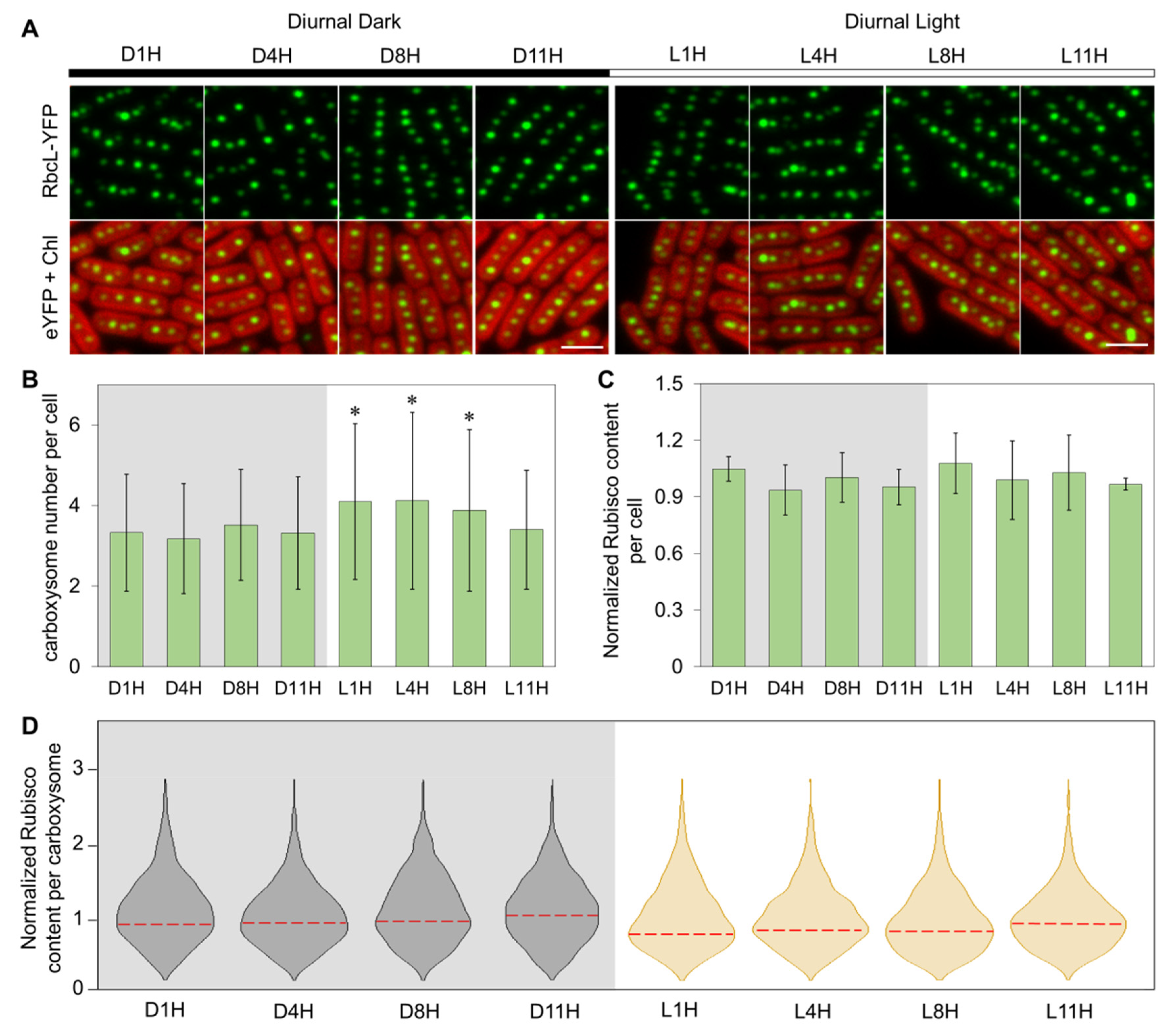
3.1. Carboxysome Biosynthesis Is Regulated during Diurnal Cycles in Syn7942
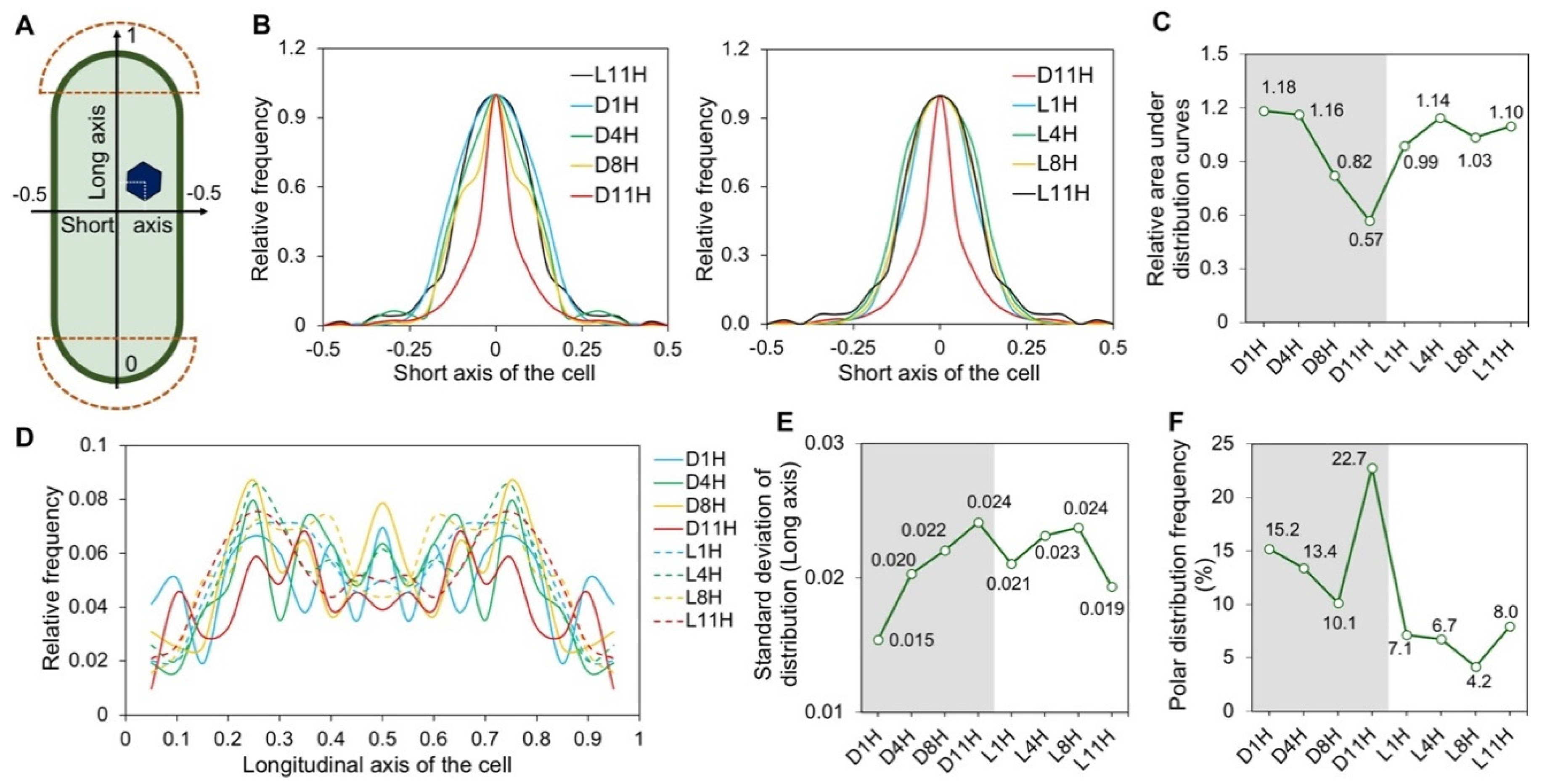
3.2. Subcellular Localization of Carboxysomes Is Diurnally Regulated in Syn7942
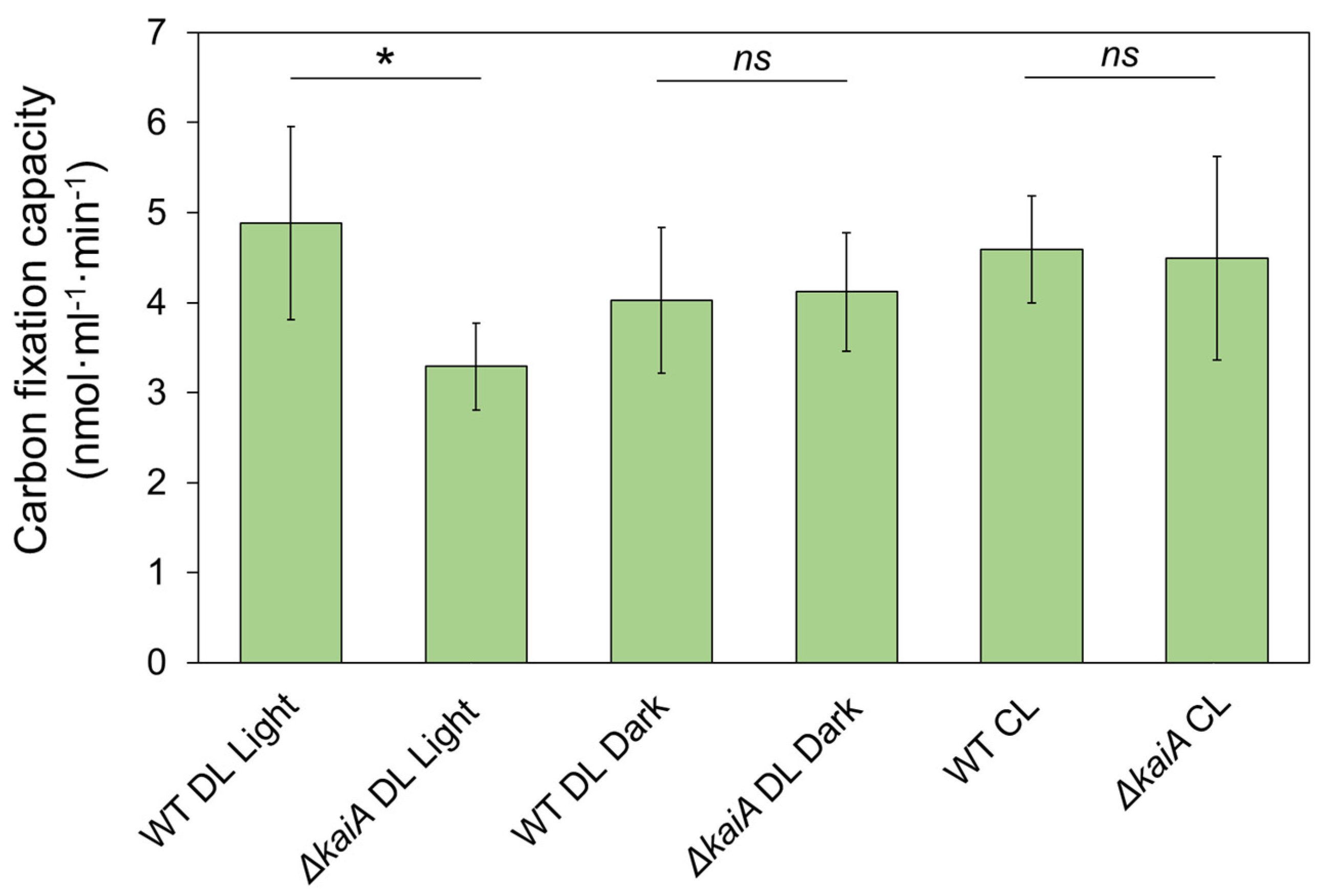
3.3. KaiA Deletion Alters the Carboxysome Localization and Abundance in Syn7942
3.4. Carbon Fixation of Syn7942 Cells Is Rhythmically Alternated during Diurnal Cycles
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dvornyk, V.; Vinogradova, O.; Nevo, E. Origin and evolution of circadian clock genes in prokaryotes. Proc. Natl. Acad. Sci. USA 2003, 100, 2495–2500. [Google Scholar] [CrossRef] [PubMed]
- Casella, S.; Huang, F.; Mason, D.; Zhao, G.-Y.; Johnson, G.N.; Mullineaux, C.W.; Liu, L.-N. Dissecting the Native Architecture and Dynamics of Cyanobacterial Photosynthetic Machinery. Mol. Plant 2017, 10, 1434–1448. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.N.; Bryan, S.J.; Huang, F.; Yu, J.; Nixon, P.J.; Rich, P.R.; Mullineaux, C.W. Control of electron transport routes through redox-regulated redistribution of respiratory complexes. Proc. Natl. Acad. Sci. USA 2012, 109, 11431–11436. [Google Scholar] [CrossRef] [PubMed]
- Wiltbank, L.B.; Kehoe, D.M. Diverse light responses of cyanobacteria mediated by phytochrome superfamily photoreceptors. Nat. Rev. Microbiol. 2019, 17, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Schuergers, N.; Lenn, T.; Kampmann, R.; Meissner, M.V.; Esteves, T.; Temerinac-Ott, M.; Korvink, J.G.; Lowe, A.R.; Mullineaux, C.W.; Wilde, A. Cyanobacteria use micro-optics to sense light direction. Elife 2016, 5, e12620. [Google Scholar] [CrossRef]
- Sanfilippo, J.E.; Garczarek, L.; Partensky, F.; Kehoe, D.M. Chromatic Acclimation in Cyanobacteria: A Diverse and Widespread Process for Optimizing Photosynthesis. Annu. Rev. Microbiol. 2019, 73, 407–433. [Google Scholar] [CrossRef]
- Liu, L.N. Distribution and dynamics of electron transport complexes in cyanobacterial thylakoid membranes. Biochim. Biophys. Acta 2016, 1857, 256–265. [Google Scholar] [CrossRef]
- Mullineaux, C.W.; Liu, L.N. Membrane dynamics in phototrophic bacteria. Annu. Rev. Microbiol. 2020, 74, 633–654. [Google Scholar] [CrossRef]
- Zhao, L.S.; Huokko, T.; Wilson, S.; Simpson, D.M.; Wang, Q.; Ruban, A.V.; Mullineaux, C.W.; Zhang, Y.Z.; Liu, L.N. Structural variability, coordination, and adaptation of a native photosynthetic machinery. Nat. Plants 2020, 6, 869–882. [Google Scholar] [CrossRef]
- Mahbub, M.; Hemm, L.; Yang, Y.; Kaur, R.; Carmen, H.; Engl, C.; Huokko, T.; Riediger, M.; Watanabe, S.; Liu, L.-N.; et al. mRNA localisation, reaction centre biogenesis and thylakoid membrane targeting in cyanobacteria. Nat. Plants 2020, in press. [Google Scholar] [CrossRef]
- Vijayan, V.; Zuzow, R.; O’Shea, E.K. Oscillations in supercoiling drive circadian gene expression in cyanobacteria. Proc. Natl. Acad. Sci. USA 2009, 106, 22564–22568. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Pando, B.F.; Dong, G.; Golden, S.S.; van Oudenaarden, A. Circadian gating of the cell cycle revealed in single cyanobacterial cells. Science 2010, 327, 1522–1526. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.H.; Zhao, C.; Xu, Y.; Mori, T. Timing the day: What makes bacterial clocks tick? Nat. Rev. Microbiol. 2017, 15, 232–242. [Google Scholar] [CrossRef]
- Rust, M.J.; Golden, S.S.; O’Shea, E.K. Light-driven changes in energy metabolism directly entrain the cyanobacterial circadian oscillator. Science 2011, 331, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Golden, S.S.; Canales, S.R. Cyanobacterial circadian clocks—Timing is everything. Nat. Rev. Microbiol. 2003, 1, 191–199. [Google Scholar] [CrossRef]
- Woelfle, M.A.; Ouyang, Y.; Phanvijhitsiri, K.; Johnson, C.H. The adaptive value of circadian clocks: An experimental assessment in cyanobacteria. Curr. Biol. CB 2004, 14, 1481–1486. [Google Scholar] [CrossRef] [PubMed]
- Welkie, D.G.; Rubin, B.E.; Chang, Y.G.; Diamond, S.; Rifkin, S.A.; LiWang, A.; Golden, S.S. Genome-wide fitness assessment during diurnal growth reveals an expanded role of the cyanobacterial circadian clock protein KaiA. Proc. Natl. Acad. Sci. USA 2018, 115, E7174–E7183. [Google Scholar] [CrossRef]
- Welkie, D.G.; Rubin, B.E.; Diamond, S.; Hood, R.D.; Savage, D.F.; Golden, S.S. A Hard Day’s Night: Cyanobacteria in Diel Cycles. Trends Microbiol. 2019, 27, 231–242. [Google Scholar] [CrossRef]
- Turmo, A.; Gonzalez-Esquer, C.R.; Kerfeld, C.A. Carboxysomes: Metabolic modules for CO2 fixation. FEMS Microbiol. Lett. 2017, 364, fnx176. [Google Scholar] [CrossRef]
- Yeates, T.O.; Tsai, Y.; Tanaka, S.; Sawaya, M.R.; Kerfeld, C.A. Self-assembly in the carboxysome: A viral capsid-like protein shell in bacterial cells. Biochem. Soc. Trans. 2007, 35, 508–511. [Google Scholar] [CrossRef]
- Kerfeld, C.A.; Erbilgin, O. Bacterial microcompartments and the modular construction of microbial metabolism. Trends Microbiol. 2015, 23, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Sui, N.; Huang, F.; Liu, L.N. Photosynthesis in Phytoplankton: Insights from the Newly Discovered Biological Inorganic Carbon Pumps. Mol. Plant 2020, 13, 949–951. [Google Scholar] [CrossRef]
- Rae, B.D.; Long, B.M.; Forster, B.; Nguyen, N.D.; Velanis, C.N.; Atkinson, N.; Hee, W.Y.; Mukherjee, B.; Price, G.D.; McCormick, A.J. Progress and challenges of engineering a biophysical CO2-concentrating mechanism into higher plants. J. Exp. Bot. 2017, 68, 3717–3737. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, M.; Rodriguez-Ramos, J.; Dykes, G.F.; Owen, S.V.; Casella, S.; Simpson, D.M.; Beynon, R.J.; Liu, L.-N. Direct characterization of the native structure and mechanics of cyanobacterial carboxysomes. Nanoscale 2017, 9, 10662–10673. [Google Scholar] [CrossRef]
- Long, B.M.; Badger, M.R.; Whitney, S.M.; Price, G.D. Analysis of carboxysomes from Synechococcus PCC7942 reveals multiple Rubisco complexes with carboxysomal proteins CcmM and CcaA. J. Biol. Chem. 2007, 282, 29323–29335. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Huang, F.; Faulkner, M.; Jiang, Q.; Dykes, G.F.; Yang, M.; Liu, L.N. Engineering and Modulating Functional Cyanobacterial CO2-Fixing Organelles. Front. Plant Sci. 2018, 9, 739. [Google Scholar] [CrossRef]
- Huang, F.; Vasieva, O.; Sun, Y.; Faulkner, M.; Dykes, G.F.; Zhao, Z.; Liu, L.N. Roles of RbcX in Carboxysome Biosynthesis in the Cyanobacterium Synechococcus elongatus PCC7942. Plant Physiol. 2019, 179, 184–194. [Google Scholar] [CrossRef]
- Sun, Y.; Wollman, A.J.M.; Huang, F.; Leake, M.C.; Liu, L.N. Single-organelle quantification reveals the stoichiometric and structural variability of carboxysomes dependent on the environment. Plant Cell 2019, 31, 1648–1664. [Google Scholar] [CrossRef]
- Huang, F.; Kong, W.; Sun, Y.; Chen, T.; Dykes, G.F.; Jiang, Y.L.; Liu, L.N. Rubisco accumulation factor 1 (Raf1) plays essential roles in mediating Rubisco assembly and carboxysome biogenesis. Proc. Natl. Acad. Sci. USA 2020, 117, 17418–17428. [Google Scholar] [CrossRef]
- Rae, B.D.; Long, B.M.; Badger, M.R.; Price, G.D. Structural determinants of the outer shell of beta-carboxysomes in Synechococcus elongatus PCC 7942: Roles for CcmK2, K3-K4, CcmO, and CcmL. PLoS ONE 2012, 7, e43871. [Google Scholar] [CrossRef]
- Wang, C.; Sun, B.; Zhang, X.; Huang, X.; Zhang, M.; Guo, H.; Chen, X.; Huang, F.; Chen, T.; Mi, H.; et al. Structural mechanism of the active bicarbonate transporter from cyanobacteria. Nat. Plants 2019, 5, 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- Hennacy, J.H.; Jonikas, M.C. Prospects for Engineering Biophysical CO2 Concentrating Mechanisms into Land Plants to Enhance Yields. Annu. Rev. Plant Biol. 2020, 71, 461–485. [Google Scholar] [CrossRef] [PubMed]
- Faulkner, M.; Szabó, I.; Weetman, S.L.; Sicard, F.; Huber, R.G.; Bond, P.J.; Rosta, E.; Liu, L.-N. Molecular simulations unravel the molecular principles that mediate selective permeability of carboxysome shell protein. bioRxiv 2020. 2020.06.14.151241. Available online: https://www.biorxiv.org/content/10.1101/2020.06.14.151241v1.abstract (accessed on 14 June 2020).
- Burnap, R.L.; Hagemann, M.; Kaplan, A. Regulation of CO2 Concentrating Mechanism in Cyanobacteria. Life 2015, 5, 348–371. [Google Scholar] [CrossRef] [PubMed]
- Savage, D.F.; Afonso, B.; Chen, A.H.; Silver, P.A. Spatially ordered dynamics of the bacterial carbon fixation machinery. Science 2010, 327, 1258–1261. [Google Scholar] [CrossRef] [PubMed]
- Hill, N.C.; Tay, J.W.; Altus, S.; Bortz, D.M.; Cameron, J.C. Life cycle of a cyanobacterial carboxysome. Sci. Adv. 2020, 6, eaba1269. [Google Scholar] [CrossRef] [PubMed]
- MacCready, J.S.; Hakim, P.; Young, E.J.; Hu, L.; Liu, J.; Osteryoung, K.W.; Vecchiarelli, A.G.; Ducat, D.C. Protein gradients on the nucleoid position the carbon-fixing organelles of cyanobacteria. Elife 2018, 7, e39723. [Google Scholar] [CrossRef]
- MacCready, J.S.; Basalla, J.L.; Vecchiarelli, A.G. Origin and Evolution of Carboxysome Positioning Systems in Cyanobacteria. Mol. Biol. Evol. 2020, 37, 1434–1451. [Google Scholar] [CrossRef]
- Sun, Y.; Casella, S.; Fang, Y.; Huang, F.; Faulkner, M.; Barrett, S.; Liu, L.N. Light modulates the biosynthesis and organization of cyanobacterial carbon fixation machinery through photosynthetic electron flow. Plant Physiol. 2016, 171, 530–541. [Google Scholar] [CrossRef]
- Cohen, S.E.; Golden, S.S. Circadian Rhythms in Cyanobacteria. Microbiol. Mol. Biol. Rev. 2015, 79, 373–385. [Google Scholar] [CrossRef]
- Watson, G.M.; Tabita, F.R. Regulation, unique gene organization, and unusual primary structure of carbon fixation genes from a marine phycoerythrin-containing cyanobacterium. Plant Mol. Biol. 1996, 32, 1103–1115. [Google Scholar] [CrossRef]
- Ito, H.; Mutsuda, M.; Murayama, Y.; Tomita, J.; Hosokawa, N.; Terauchi, K.; Sugita, C.; Sugita, M.; Kondo, T.; Iwasaki, H. Cyanobacterial daily life with Kai-based circadian and diurnal genome-wide transcriptional control in Synechococcus elongatus. Proc. Natl. Acad. Sci. USA 2009, 106, 14168–14173. [Google Scholar] [CrossRef] [PubMed]
- Aryal, U.K.; Stockel, J.; Krovvidi, R.K.; Gritsenko, M.A.; Monroe, M.E.; Moore, R.J.; Koppenaal, D.W.; Smith, R.D.; Pakrasi, H.B.; Jacobs, J.M. Dynamic proteomic profiling of a unicellular cyanobacterium Cyanothece ATCC51142 across light-dark diurnal cycles. BMC Syst. Biol. 2011, 5, 194. [Google Scholar] [CrossRef] [PubMed]
- Datsenko, K.A.; Wanner, B.L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl. Acad. Sci. USA 2000, 97, 6640–6645. [Google Scholar] [CrossRef]
- Rippka, R.; Deruelles, J.; Waterbury, J.B.; Herdman, M.; Stanier, R.Y. Generic Assignments, Strain Histories and Properties of Pure Cultures of Cyanobacteria. Microbiology 1979, 111, 1–61. [Google Scholar] [CrossRef]
- Cohen, S.E.; Erb, M.L.; Selimkhanov, J.; Dong, G.; Hasty, J.; Pogliano, J.; Golden, S.S. Dynamic localization of the cyanobacterial circadian clock proteins. Curr. Biol. 2014, 24, 1836–1844. [Google Scholar] [CrossRef] [PubMed]
- Mackey, S.R.; Ditty, J.L.; Clerico, E.M.; Golden, S.S. Detection of rhythmic bioluminescence from luciferase reporters in cyanobacteria. Methods Mol. Biol. 2007, 362, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Barrett, S.D. Image SXM. 2015. Available online: http://www.ImageSXM.org.uk (accessed on 29 August 2020).
- Cameron, J.C.; Wilson, S.C.; Bernstein, S.L.; Kerfeld, C.A. Biogenesis of a bacterial organelle: The carboxysome assembly pathway. Cell 2013, 155, 1131–1140. [Google Scholar] [CrossRef]
- Chen, A.H.; Robinson-Mosher, A.; Savage, D.F.; Silver, P.A.; Polka, J.K. The bacterial carbon-fixing organelle is formed by shell envelopment of preassembled cargo. PLoS ONE 2013, 8, e76127. [Google Scholar] [CrossRef]
- Clerico, E.M.; Cassone, V.M.; Golden, S.S. Stability and lability of circadian period of gene expression in the cyanobacterium Synechococcus elongatus. Microbiology 2009, 155, 635–641. [Google Scholar] [CrossRef][Green Version]
- Diamond, S.; Jun, D.; Rubin, B.E.; Golden, S.S. The circadian oscillator in Synechococcus elongatus controls metabolite partitioning during diurnal growth. Proc. Natl. Acad. Sci. USA 2015, 112, E1916–E1925. [Google Scholar] [CrossRef] [PubMed]
- Waldbauer, J.R.; Rodrigue, S.; Coleman, M.L.; Chisholm, S.W. Transcriptome and Proteome Dynamics of a Light-Dark Synchronized Bacterial Cell Cycle. PLoS ONE 2012, 7, e43432. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro, A.C.L.; Benevento, M.; Lehmann, R.; van Breukelen, B.; Post, H.; Giansanti, P.; Maarten Altelaar, A.F.; Axmann, I.M.; Heck, A.J.R. Daily Rhythms in the Cyanobacterium Synechococcus elongatus Probed by High-resolution Mass Spectrometry–based Proteomics Reveals a Small Defined Set of Cyclic Proteins. Mol. Cell. Proteom. 2014, 13, 2042–2055. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, V.; Jain, I.H.; O’Shea, E.K. A high resolution map of a cyanobacterial transcriptome. Genome Biol. 2011, 12, R47. [Google Scholar] [CrossRef] [PubMed]
- Nassoury, N.; Fritz, L.; Morse, D. Circadian Changes in Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase Distribution Inside Individual Chloroplasts Can Account for the Rhythm in Dinoflagellate Carbon Fixation. Plant Cell 2001, 13, 923. [Google Scholar] [CrossRef]
- Dai, W.; Chen, M.; Myers, C.; Ludtke, S.J.; Pettitt, B.M.; King, J.A.; Schmid, M.F.; Chiu, W. Visualizing Individual RuBisCO and Its Assembly into Carboxysomes in Marine Cyanobacteria by Cryo-Electron Tomography. J. Mol. Biol. 2018, 430, 4156–4167. [Google Scholar] [CrossRef] [PubMed]
- Martins, B.M.C.; Tooke, A.K.; Thomas, P.; Locke, J.C.W. Cell size control driven by the circadian clock and environment in cyanobacteria. Proc. Natl. Acad. Sci. USA 2018, 115, E11415–E11424. [Google Scholar] [CrossRef]
- Saha, R.; Liu, D.; Hoynes-O’Connor, A.; Liberton, M.; Yu, J.; Bhattacharyya-Pakrasi, M.; Balassy, A.; Zhang, F.; Moon, T.S.; Maranas, C.D.; et al. Diurnal Regulation of Cellular Processes in the Cyanobacterium Synechocystis sp. Strain PCC 6803: Insights from Transcriptomic, Fluxomic, and Physiological Analyses. mBio 2016, 7, e00464-16. [Google Scholar] [CrossRef]
- Jain, I.H.; Vijayan, V.; O’Shea, E.K. Spatial ordering of chromosomes enhances the fidelity of chromosome partitioning in cyanobacteria. Proc. Natl. Acad. Sci. USA 2012, 109, 13638–13643. [Google Scholar] [CrossRef]
- Murata, K.; Hagiwara, S.; Kimori, Y.; Kaneko, Y. Ultrastructure of compacted DNA in cyanobacteria by high-voltage cryo-electron tomography. Sci. Rep. 2016, 6, 34934. [Google Scholar] [CrossRef]
- Tamoi, M.; Miyazaki, T.; Fukamizo, T.; Shigeoka, S. The Calvin cycle in cyanobacteria is regulated by CP12 via the NAD(H)/NADP(H) ratio under light/dark conditions. Plant J. 2005, 42, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, B.; Mubarakshina, M.; Khorobrykh, S. Kinetics of the plastoquinone pool oxidation following illumination Oxygen incorporation into photosynthetic electron transport chain. FEBS Lett. 2007, 581, 1342–1346. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.I.; Vinyard, D.J.; Ananyev, G.M.; Dismukes, G.C.; Golden, S.S. Oxidized quinones signal onset of darkness directly to the cyanobacterial circadian oscillator. Proc. Natl. Acad. Sci. USA 2012, 109, 17765–17769. [Google Scholar] [CrossRef]
- Carmo-Silva, A.E.; Keys, A.J.; Andralojc, P.J.; Powers, S.J.; Arrabaca, M.C.; Parry, M.A. Rubisco activities, properties, and regulation in three different C4 grasses under drought. J. Exp. Bot. 2010, 61, 2355–2366. [Google Scholar] [CrossRef]
- Lan, Y.; Woodrow, I.E.; Mott, K.A. Light-dependent changes in ribulose bisphosphate carboxylase activase activity in leaves. Plant Physiol. 1992, 99, 304–309. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yokosho, K.; Liu, S.; Cao, H.R.; Yamaji, N.; Zhu, X.G.; Liao, H.; Ma, J.F.; Chen, Z.C. Diel magnesium fluctuations in chloroplasts contribute to photosynthesis in rice. Nat. Plants 2020, 6, 848–859. [Google Scholar] [CrossRef]
- To, K.Y.; Suen, D.F.; Chen, S.C. Molecular characterization of ribulose-1,5-bisphosphate carboxylase/oxygenase activase in rice leaves. Planta 1999, 209, 66–76. [Google Scholar] [CrossRef]
- Liu, Z.; Taub, C.C.; McClung, C.R. Identification of an Arabidopsis thaliana ribulose-1,5-bisphosphate carboxylase/oxygenase activase (RCA) minimal promoter regulated by light and the circadian clock. Plant Physiol. 1996, 112, 43–51. [Google Scholar] [CrossRef]
- Hartwell, J.; Dever, L.V.; Boxall, S.F. Emerging model systems for functional genomics analysis of Crassulacean acid metabolism. Curr. Opin. Plant Biol. 2016, 31, 100–108. [Google Scholar] [CrossRef]
- Boxall, S.F.; Dever, L.V.; Knerova, J.; Gould, P.D.; Hartwell, J. Phosphorylation of Phosphoenolpyruvate Carboxylase Is Essential for Maximal and Sustained Dark CO2 Fixation and Core Circadian Clock Operation in the Obligate Crassulacean Acid Metabolism Species Kalanchoe fedtschenkoi. Plant Cell 2017, 29, 2519–2536. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.; Huang, F.; Dykes, G.F.; Liu, L.-N. Diurnal Regulation of In Vivo Localization and CO2-Fixing Activity of Carboxysomes in Synechococcus elongatus PCC 7942. Life 2020, 10, 169. https://doi.org/10.3390/life10090169
Sun Y, Huang F, Dykes GF, Liu L-N. Diurnal Regulation of In Vivo Localization and CO2-Fixing Activity of Carboxysomes in Synechococcus elongatus PCC 7942. Life. 2020; 10(9):169. https://doi.org/10.3390/life10090169
Chicago/Turabian StyleSun, Yaqi, Fang Huang, Gregory F. Dykes, and Lu-Ning Liu. 2020. "Diurnal Regulation of In Vivo Localization and CO2-Fixing Activity of Carboxysomes in Synechococcus elongatus PCC 7942" Life 10, no. 9: 169. https://doi.org/10.3390/life10090169
APA StyleSun, Y., Huang, F., Dykes, G. F., & Liu, L.-N. (2020). Diurnal Regulation of In Vivo Localization and CO2-Fixing Activity of Carboxysomes in Synechococcus elongatus PCC 7942. Life, 10(9), 169. https://doi.org/10.3390/life10090169





