Protoenzymes: The Case of Hyperbranched Polymer-Scaffolded ZnS Nanocrystals
Abstract
1. Introduction
2. Materials and Methods
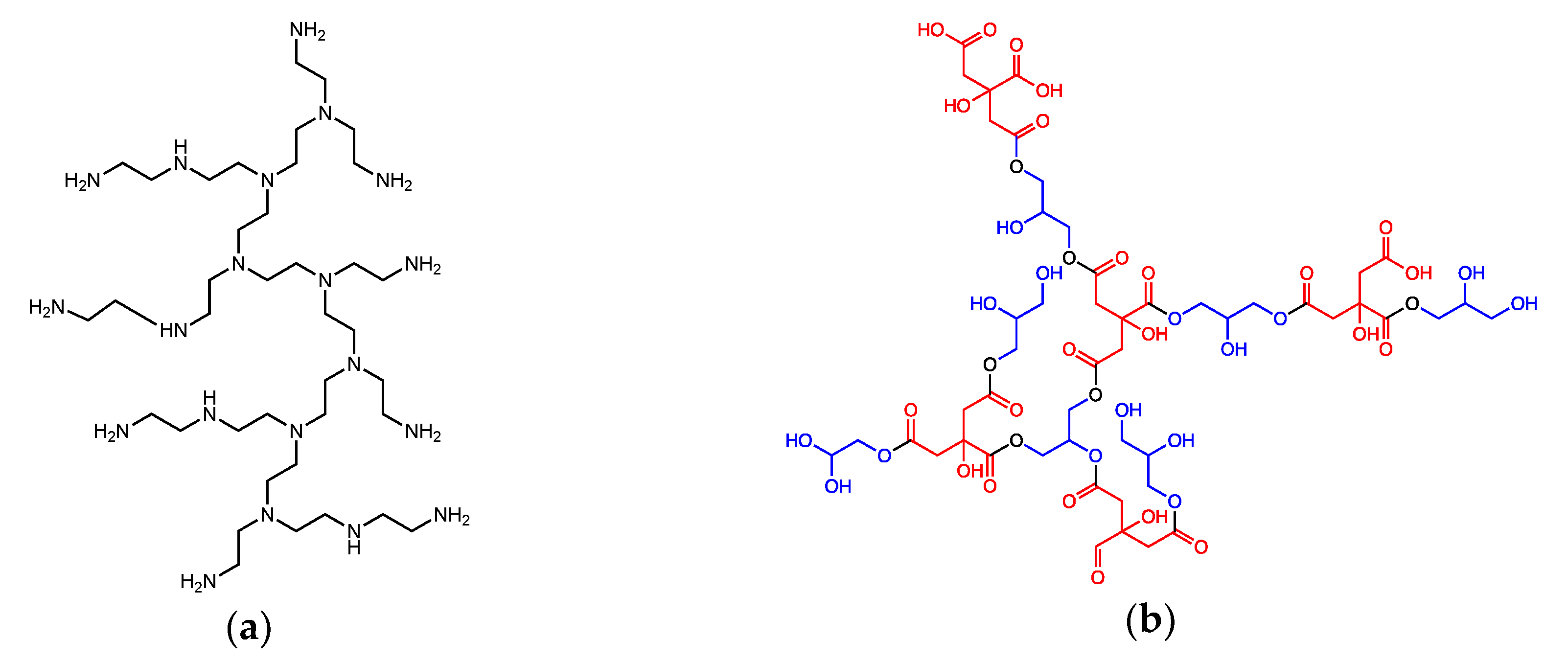
2.1. Synthesis of Hyperbranched Polyethyleneimine (HyPEI)-Supported ZnS Nanocrystals (NCs)
2.2. Synthesis of Glycerol Citrate Polyesters
2.3. Measurement of the Divalent Cation Loading Capacity of the Polymers
2.4. Synthesis of Glycerol Citrate Polyester-Supported ZnS NCs
2.5. Mass Spectrometry
2.6. Transmission Electron Microscopy (TEM)
2.7. Photocatalytic Activity Measurement
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Campbell, M.K.; Farrell, S.O. Biochemistry; Cengage Learning: Boston, MA, USA, 2011. [Google Scholar]
- Radzicka, A.; Wolfenden, R. A Proficient Enzyme. Science 1995, 267, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Richard, J.P.; Amyes, T.L.; Reyes, A.C. Orotidine 5′-Monophosphate Decarboxylase: Probing the Limits of the Possible for Enzyme Catalysis. Acc. Chem. Res. 2018, 51, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Szöke, A.; Scott, W.G.; Hajdu, J. Catalysis, Evolution and Life. FEBS Lett. 2003, 553, 18–20. [Google Scholar] [CrossRef]
- Canepa, C. The Role of Catalysis on the Formation of an Active Proto-Enzyme in the Prebiotic Aqueous Environment. Nat. Sci. 2013, 5, 549–555. [Google Scholar] [CrossRef]
- Kruger, K.; Grabowski, P.J.; Zaug, A.J.; Sands, J.; Gottschling, D.E.; Cech, T.R. Self-Splicing RNA: Autoexcision and Autocyclization of the Ribosomal RNA Intervening Sequence of Tetrahymena. Cell 1982, 31, 147–157. [Google Scholar] [CrossRef]
- Guerrier-Takada, C.; Gardiner, K.; Marsh, T.; Pace, N.; Altman, S. The RNA Moiety of Ribonuclease P Is the Catalytic Subunit of the Enzyme. Cell 1983, 35, 849–857. [Google Scholar] [CrossRef]
- Cech, T.R. The RNA Worlds in Context. Cold Spring Harb. Perspect. Biol. 2012, 4, a006742. [Google Scholar] [CrossRef]
- Orgel, L.E. Some Consequences of the RNA World Hypothesis. Orig. Life Evol. Biosph. 2003, 33, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Morowitz, H.J.; Srinivasan, V.; Smith, E. Ligand Field Theory and the Origin of Life as an Emergent Feature of the Periodic Table of Elements. Biol. Bull. 2010, 219, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, R. Small Molecule Interactions Were Central to the Origin of Life. Q. Rev. Biol. 2006, 81, 105–125. [Google Scholar] [CrossRef] [PubMed]
- Cleaves, H.J.; Scott, A.M.; Hill, F.C.; Leszczynski, J.; Sahai, N.; Hazen, R. Mineral–Organic Interfacial Processes: Potential Roles in the Origins of Life. Chem. Soc. Rev. 2012, 41, 5502–5525. [Google Scholar] [CrossRef] [PubMed]
- Russell, M.J.; Barge, L.M.; Bhartia, R.; Bocanegra, D.; Bracher, P.J.; Branscomb, E.; Kidd, R.; McGlynn, S.; Meier, D.H.; Nitschke, W.; et al. Drive to Life on Wet and Icy Worlds. Astrobiology 2014, 14, 308–343. [Google Scholar] [CrossRef] [PubMed]
- Bruice, T.C.; Benkovic, S.J. Chemical Basis for Enzyme Catalysis. Biochemistry 2000, 39, 6267–6274. [Google Scholar] [CrossRef] [PubMed]
- Hansma, H.G. Possible Origin of Life between Mica Sheets: Does Life Imitate Mica? J. Biomol. Struct. Dyn. 2013, 31, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Ferris, J.P.; Hill, A.R.; Liu, R.; Orgel, L.E. Synthesis of Long Prebiotic Oligomers on Mineral Surfaces. Nature 1996, 381, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Kitadai, N.; Oonishi, H.; Umemoto, K.; Usui, T.; Fukushi, K.; Nakashima, S. Glycine Polymerization on Oxide Minerals. Orig. Life Evol. Biosph. 2017, 47, 123–143. [Google Scholar] [CrossRef]
- Zozulia, O.; Dolan, M.A.; Korendovych, I.V. Catalytic Peptide Assemblies. Chem. Soc. Rev. 2018, 47, 3621–3639. [Google Scholar] [CrossRef]
- Carny, O.; Gazit, E. A Model for the Role of Short Self-Assembled Peptides in the Very Early Stages of the Origin of Life. FASEB J. 2005, 19, 1051–1055. [Google Scholar] [CrossRef]
- Makam, P.; Yamijala, S.S.R.K.C.; Tao, K.; Shimon, L.J.W.; Eisenberg, D.S.; Sawaya, M.R.; Wong, B.M.; Gazit, E. Non-Proteinaceous Hydrolase Comprised of a Phenylalanine Metallo-Supramolecular Amyloid-like Structure. Nat. Catal. 2019, 2, 977–985. [Google Scholar] [CrossRef]
- Scintilla, S.; Bonfio, C.; Belmonte, L.; Forlin, M.; Rossetto, D.; Li, J.; Cowan, J.A.; Galliani, A.; Arnesano, F.; Assfalg, M.; et al. Duplications of an Iron–Sulphur Tripeptide Leads to the Formation of a Protoferredoxin. Chem. Commun. 2016, 52, 13456–134569. [Google Scholar] [CrossRef] [PubMed]
- Bonfio, C.; Valer, L.; Scintilla, S.; Shah, S.; Evans, D.J.; Jin, L.; Szostak, J.W.; Sasselov, D.D.; Sutherland, J.D.; Mansy, S.S. UV-Light-Driven Prebiotic Synthesis of Iron–Sulfur Clusters. Nat. Chem. 2017, 9, 1229–1234. [Google Scholar] [CrossRef] [PubMed]
- Benner, S.A. Paradoxes in the origin of life. Orig. Life Evol. Biosph. 2014, 44, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.W.; Harada, K. Thermal copolymerization of amino acids to a product resembling protein. Science 1958, 128, 1214. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.W.; Harada, K. The thermal copolymerization of amino acids common to protein. J. Am. Chem. Soc. 1960, 82, 3745–3751. [Google Scholar] [CrossRef]
- Fox, S.W.; Krampitz, G. Catalytic decomposition of glucose in aqueous solution by thermal proteinoids. Nature 1964, 203, 1362–1364. [Google Scholar] [CrossRef] [PubMed]
- Durant, D.H.; Fox, S.W. Enhancement of rate of decarboxylation of pyruvic acid and of hydrolysis of adenosine phosphates by thermal polyanhydro-alpha-amino acids. Fed. Proc. 1966, 25, 342. [Google Scholar]
- Rohlfing, D.L.; Fox, S.W. Catalytic activity of thermal polyanhydro-alpha-amino acids for hydrolysis of p-nitrophenyl acetate—Catalysis by thermal polyamino acids. Arch. Biochem. Biophys. 1967, 118, 122–126. [Google Scholar] [CrossRef]
- Ryan, J.W.; Fox, S.W. Activation of glycine by ATP, a divalent cation, and proteinoid microspheres. Biosystems 1973, 5, 115–118. [Google Scholar] [CrossRef]
- Quirk, S. Enhanced catalytic activity from proteinoid microspheres. J Biomed Mater Res. 2013, 101, 1133–1143. [Google Scholar] [CrossRef]
- Gao, C.; Yan, D. Hyperbranched Polymers: From Synthesis to Applications. Prog. Polym. Sci. 2004, 29, 183–275. [Google Scholar] [CrossRef]
- Kirkorian, K.; Ellis, A.; Twyman, L.J. Catalytic Hyperbranched Polymers as Enzyme Mimics; Exploiting the Principles of Encapsulation and Supramolecular Chemistry. Chem. Soc. Rev. 2012, 41, 6138–6159. [Google Scholar] [CrossRef] [PubMed]
- Cheremisinoff, N.P.C. Condensed Encyclopedia of Polymer Engineering Terms; Cheremisinoff, N.P., Ed.; Butterworth-Heinemann: Boston, MA, USA, 2001; pp. 39–81. [Google Scholar]
- Voit, B.I.; Lederer, A. Hyperbranched and Highly Branched Polymer Architectures—Synthetic Strategies and Major Characterization Aspects. Chem. Rev. 2009, 109, 5924–5973. [Google Scholar] [CrossRef] [PubMed]
- Mamajanov, I.; Callahan, M.P.; Dworkin, J.P.; Cody, G.D. Prebiotic Alternatives to Proteins: Structure and Function of Hyperbranched Polyesters. Orig. Life Evol. Biosph. 2015, 45, 123–137. [Google Scholar] [CrossRef] [PubMed]
- Mamajanov, I. Wet-Dry Cycling Delays the Gelation of Hyperbranched Polyesters: Implications to the Origin of Life. Life 2019, 9, 56. [Google Scholar] [CrossRef] [PubMed]
- Mamajanov, I. Selective Synthesis of Hyperbranched Polyesters under Wet-Dry Cycling Conditions. In ALife Conference Proceedings; One Rogers Street: Cambridge, MA, USA, 2018; pp. 580–581. [Google Scholar]
- Mamajanov, I.; Cody, G.D. Protoenzymes: The Case of Hyperbranched Polyesters. Philos. Trans. R. Soc. Math. Phys. Eng. Sci. 2017, 375, 20160537. [Google Scholar] [CrossRef]
- Breslow, R. Biomimetic Chemistry and Artificial Enzymes: Catalysis by Design. Acc. Chem. Res. 1995, 28, 146–153. [Google Scholar] [CrossRef]
- Kuah, E.; Toh, S.; Yee, J.; Ma, Q.; Gao, Z. Enzyme Mimics: Advances and Applications. Chem. Eur. J. 2016, 22, 8404–8430. [Google Scholar] [CrossRef]
- Moshfegh, A.Z. Nanoparticle Catalysts. J. Phys. Appl. Phys. 2009, 42, 233001. [Google Scholar] [CrossRef]
- Liang, M.; Yan, X. Nanozymes: From New Concepts, Mechanisms, and Standards to Applications. Acc. Chem. Res. 2019, 52, 2190–2200. [Google Scholar] [CrossRef]
- Griffin, S.; Masood, M.I.; Nasim, M.J.; Sarfraz, M.; Ebokaiwe, A.P.; Schäfer, K.-H.; Keck, C.M.; Jacob, C. Natural Nanoparticles: A Particular Matter Inspired by Nature. Antioxidants 2017, 7, 3. [Google Scholar] [CrossRef]
- Hochella, M.F.; Lower, S.K.; Maurice, P.A.; Penn, R.L.; Sahai, N.; Sparks, D.L.; Twining, B.S. Nanominerals, Mineral Nanoparticles, and Earth Systems. Science 2008, 319, 1631–1635. [Google Scholar] [CrossRef] [PubMed]
- Wächtershäuser, G. Groundworks for an Evolutionary Biochemistry: The Iron-Sulphur World. Prog. Biophys. Mol. Biol. 1992, 58, 85–201. [Google Scholar] [CrossRef]
- Mulkidjanian, A.Y. On the Origin of Life in the Zinc World: 1. Photosynthesizing, Porous Edifices Built of Hydrothermally Precipitated Zinc Sulfide as Cradles of Life on Earth. Biol. Direct 2009, 4, 26. [Google Scholar] [CrossRef] [PubMed]
- Stroyuk, A.L.; Raevskaya, A.E.; Korzhak, A.V.; Kuchmii, S.Y. Zinc Sulfide Nanoparticles: Spectral Properties and Photocatalytic Activity in Metals Reduction Reactions. J. Nanoparticle Res. 2007, 9, 1027–1039. [Google Scholar] [CrossRef]
- Sarkar, S.; Guibal, E.; Quignard, F.; SenGupta, A.K. Polymer-Supported Metals and Metal Oxide Nanoparticles: Synthesis, Characterization, and Applications. J. Nanoparticle Res. 2012, 14, 715. [Google Scholar] [CrossRef]
- de Donegá, C.M. Synthesis and Properties of Colloidal Heteronanocrystals. Chem. Soc. Rev. 2011, 40, 1512–1546. [Google Scholar] [CrossRef]
- Campisi, S.; Schiavoni, M.; Chan-Thaw, C.E.; Villa, A. Untangling the Role of the Capping Agent in Nanocatalysis: Recent Advances and Perspectives. Catalysts 2016, 6, 185. [Google Scholar] [CrossRef]
- Hassan, M.L.; Ali, A.F. Synthesis of Nanostructured Cadmium and Zinc Sulfides in Aqueous Solutions of Hyperbranched Polyethyleneimine. Cryst. Growth 2008, 310, 5252–5258. [Google Scholar] [CrossRef]
- Hu, J.-S.; Ren, L.-L.; Guo, Y.-G.; Liang, H.-P.; Cao, A.-M.; Wan, L.-J.; Bai, C.-L. Mass Production and High Photocatalytic Activity of ZnS Nanoporous Nanoparticles. Angew. Chem. Int. Ed. 2005, 44, 1269–1273. [Google Scholar] [CrossRef]
- Yin, M.; Li, Z.; Kou, J.; Zou, Z. Mechanism Investigation of Visible Light-Induced Degradation in a Heterogeneous TiO2/Eosin Y/Rhodamine B System. Environ. Sci. Technol. 2009, 43, 8361–8366. [Google Scholar] [CrossRef]
- Jia, J.; Wu, A.; Luan, S. Spectrometry Recognition of Polyethyleneimine towards Heavy Metal Ions. Colloids Surf. Physicochem. Eng. Asp. 2014, 449, 1–7. [Google Scholar] [CrossRef]
- Sang, L.-J.; Wu, Y.-Y.; Wang, H.-F. Polyethyleneimine/Manganese-Doped ZnS Nanocomposites: A Multifunctional Probe for Two-Color Imaging and Three-Dimensional Sensing. ChemPlusChem 2013, 78, 423–429. [Google Scholar] [CrossRef]
- Zhuk, D.S.; Gembitskii, P.A.; Kargin, V.A. Advances in the Chemistry of Polyethyleneimine (Polyaziridine). Russ Chem. Rev. 1965, 34, 515–527. [Google Scholar] [CrossRef]
- Steuerle, U.; Feuerhake, R. Aziridines. In Ullmann’s Encyclopedia of Industrial Chemistry; American Cancer Society: New York, NY, USA, 2006; Available online: https://onlinelibrary.wiley.com/doi/abs/10.1002/14356007.a03_239.pub2 (accessed on 12 November 2019).
- Dickens, J.E.; Irvine, W.M.; Nummelin, A.; Møllendal, H.; Saito, S.; Thorwirth, S.; Hjalmarson, A.; Onishi, M. Searches for New Interstellar Molecules, Including a Tentative Detection of Aziridine and a Possible Detection of Propenal. Spectrochim. Acta Mol. Biomol. Spectrosc. 2001, 57, 643–660. [Google Scholar] [CrossRef]
- Aylward, N. A Plausible Route to a Prebiotic Synthesis of L-Histidine. WSEAS Trans. Biol. Biomed. 2012, 9, 10–23. [Google Scholar]
- Lahav, N.; White, D.; Chang, S. Peptide Formation in the Prebiotic Era: Thermal Condensation of Glycine in Fluctuating Clay Environments. Science 1978, 201, 67–69. [Google Scholar] [CrossRef] [PubMed]
- Chandru, K.; Guttenberg, N.; Giri, C.; Hongo, Y.; Butch, C.; Mamajanov, I.; Cleaves, H.J. Simple prebiotic synthesis of high diversity dynamic combinatorial polyester libraries. Commun. Chem. 2018, 1, 30. [Google Scholar] [CrossRef]
- Flory, P.J. Molecular Size Distribution in Three Dimensional Polymers. I. Gelation1. J. Am. Chem. Soc. 1941, 63, 3083–3090. [Google Scholar] [CrossRef]
- Shapiro, R. A Replicator Was Not Involved in the Origin of Life. IUBMB Life 2000, 49, 173–176. [Google Scholar] [CrossRef]
- Wills, C.; Bada, J. The Spark of Life; Basic Books: New York, NY, USA, 2008. [Google Scholar]
- Harada, K.; Fox, S.W. The Thermal Condensation of Glutamic Acid and Glycine to Linear Peptides. J. Am. Chem. Soc. 1958, 80, 2694–2697. [Google Scholar] [CrossRef]
- Bennett, G.D. A Green Polymerization of Aspartic Acid for the Undergraduate Organic Laboratory. J. Chem. Educ. 2005, 82, 1380–1381. [Google Scholar] [CrossRef]
- Stiefel, E.I.; George, G.N. Ferredoxins, Hydrogenases, and Nitrogenases: Metal-Sulfide Proteins. In Bioorganic Chemistry; Bertini, I., Gray, H.B., Lippard, S.J., Valentine, J.S., Eds.; University Science Books, Mill Valley: Mill Valley, CA, USA, 1994; pp. 365–453. [Google Scholar]
- Pérignon, N.; Marty, J.-D.; Mingotaud, A.-F.; Dumont, M.; Rico-Lattes, I.; Mingotaud, C. Hyperbranched Polymers Analogous to PAMAM Dendrimers for the Formation and Stabilization of Gold Nanoparticles. Macromolecules 2007, 40, 3034–3041. [Google Scholar] [CrossRef]
- Peng, Q.; Zhu, J.; Yu, Y.; Hoffman, L.; Yang, X. Hyperbranched Lysine−Arginine Copolymer for Gene Delivery. J. Biomater. Sci. Polym. Ed. 2015, 26, 1–35. [Google Scholar] [CrossRef] [PubMed]
- Forsythe, J.G.; Yu, S.-S.; Mamajanov, I.; Grover, M.A.; Krishnamurthy, R.; Fernández, F.M.; Hud, N.V. Ester-Mediated Amide Bond Formation Driven by Wet–Dry Cycles: A Possible Path to Polypeptides on the Prebiotic Earth. Angew. Chem. Int. Ed. 2015, 54, 9871–9875. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Garcia, M.; Surman, A.J.; Cooper, G.J.T.; Suárez-Marina, I.; Hosni, Z.; Lee, M.P.; Cronin, L. Formation of Oligopeptides in High Yield under Simple Programmable Conditions. Nat. Commun. 2015, 6, 8385. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, G.; Pino, S.; Ciciriello, F.; Mauro, E.D. Generation of Long RNA Chains in Water. J. Biol. Chem. 2009, 284, 33206–33216. [Google Scholar] [CrossRef] [PubMed]


| Polymer | Co2+ Loading Capacity (μmol Co2+/g Polymer) |
|---|---|
| Hyperbranched Polyethyleneimine Mw = 800 | 736 |
| COOH-terminated oligo-2,2-bis(hydroxymethyl)propionic acid dendrimer, trimethylol propane core, generation 1, C45H62O30 (bis-MPA-COOH) | 4.5 |
| OH-terminated oligo- 2,2-bis(hydroxymethyl)propionic acid dendrimer, trimethylol propane core, generation 1, C21H38O12 (bis-MPA-OH) | 1.6 |
| Citric acid–glycerol polymer, prepared neat | Non-detectable |
| Citric acid–glycerol, prepared in the presence of CoCl2 | 542 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mamajanov, I.; Caudan, M.; Jia, T.Z. Protoenzymes: The Case of Hyperbranched Polymer-Scaffolded ZnS Nanocrystals. Life 2020, 10, 150. https://doi.org/10.3390/life10080150
Mamajanov I, Caudan M, Jia TZ. Protoenzymes: The Case of Hyperbranched Polymer-Scaffolded ZnS Nanocrystals. Life. 2020; 10(8):150. https://doi.org/10.3390/life10080150
Chicago/Turabian StyleMamajanov, Irena, Melina Caudan, and Tony Z. Jia. 2020. "Protoenzymes: The Case of Hyperbranched Polymer-Scaffolded ZnS Nanocrystals" Life 10, no. 8: 150. https://doi.org/10.3390/life10080150
APA StyleMamajanov, I., Caudan, M., & Jia, T. Z. (2020). Protoenzymes: The Case of Hyperbranched Polymer-Scaffolded ZnS Nanocrystals. Life, 10(8), 150. https://doi.org/10.3390/life10080150






