Shed Light in the DaRk LineagES of the Fungal Tree of Life—STRES
Abstract
1. Introduction
Black Fungi Profit from the Era of Genome Consortia
2. The STRES Project
2.1. Available Genomic Data
2.2. Main Objectives
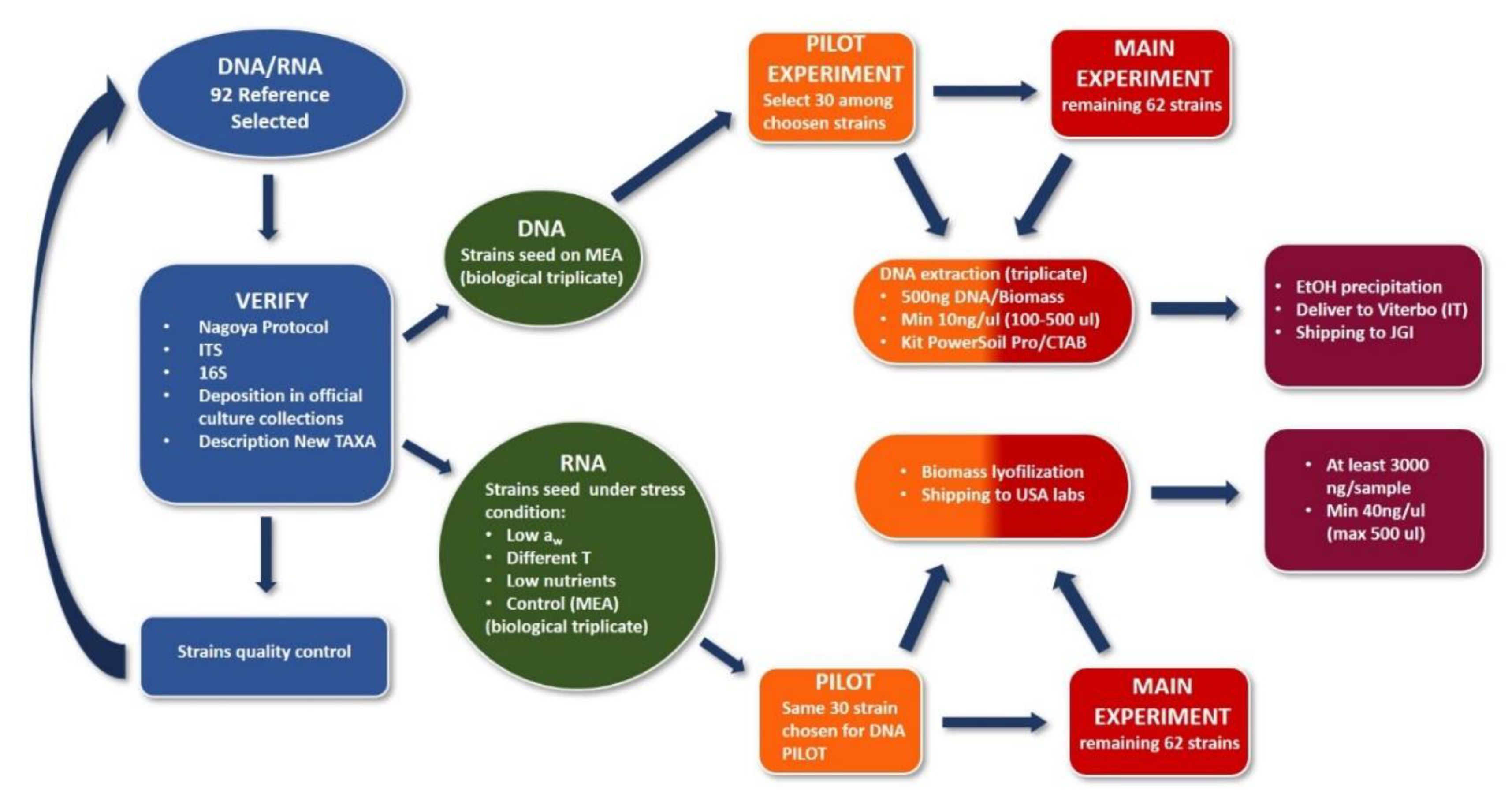
2.3. Sampling to Sequencing
2.4. Methodologies
- Phylogenomic profiling to give insights into the evolutionary history of uncovered clades throughout the FTOL (e.g., the origin of symbioses).
- Single-nucleotide polymorphisms (SNPs) calling to identify genomic regions contributing to local adaptation or even speciation.
- Detection of genes duplication or whole genome duplication as events contributing to the ability to adapt to the extremes.
- Carbohydrate-active enzymes (CAZymes), assuming that predicted metabolic competences vary among different groups of black yeasts according to their phylogenetic affiliation and ecology.
- Hydrocarbon and monoaromatic-active enzymes. Some black fungi, particularly in the order Chaetothyriales, are well known for their ability to degrade pollutants and hydrocarbons. Understanding the distribution and functionality of these genes will also inform us of their possible applications in bioremediation.
- Stress-tolerance involved enzymes. Genes involved in stress responses (e.g., UV and ionizing radiation, osmotic, and thermal stresses) will be characterized.
- Secondary metabolite biosynthetic pathway genes as potential contributors to local adaptation.
- Transcription regulators (TFs) as drivers of adaptation and speciation.
3. Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Anderson, I.C.; Campbell, C.D.; Prosser, J.I. Diversity of fungi in organic soils under a moorland – Scots pine (Pinus sylvestris L.) gradient. Environ. MicroBiol. 2003, 5, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Royer, T.V.; Leff, L.G. Diversity of fungi, bacteria, and actinomycetes on leaves decomposing in a stream. Appl. Environ. MicroBiol. 2007, 73, 756–767. [Google Scholar] [PubMed]
- Shaffer, J.P.; U’Ren, J.M.; Gallery, R.E.; Baltrus, D.A.; Arnold, A.E. An endohyphal bacterium (Chitinophaga, Bacteroidetes) alters carbon source use by Fusarium keratoplasticum (F. solani species complex, Nectriaceae). Front. MicroBiol. 2017, 8, 350. [Google Scholar] [CrossRef] [PubMed]
- Spatafora, J.W.; Aime, M.C.; Grigoriev, I.V.; Martin, F.; Stajich, J.E.; Blackwell, M. The Fungal Tree of Life: From Molecular Systematics to Genome-Scale Phylogenies. MicroBiol. Spectr. 2017, 5. [Google Scholar] [CrossRef]
- Schoch, C.L.; Sung, G.-H.; López-Giráldez, F.; Townsend, J.P.; Miadlikowska, J.; Hofsterrer, V.; Robbertse, B.; Mathenym, B.P.; Hauff, F.; Wang, Z.; et al. The Ascomycota Tree of Life: A phylum-wide phylogeny clarifies the origin and evolution of fundamental reproductive and ecological traits. Syst. Biol. 2009, 58, 224–239. [Google Scholar] [CrossRef]
- Sterflinger, K. Black yeasts and meristematic fungi: Ecology, diversity and identification. In Biodiversity and Ecophysiology of Yeasts; Springer: Berlin, Germany, 2006; pp. 501–514. [Google Scholar]
- Zajc, J.; Kogej, T.; Galinski, E.A.; Ramos, J.; Gunde-Cimerman, N. Osmoadaptation strategy of the most halophilic fungus, Wallemia ichthyophaga, growing optimally at salinities above 15% NaCl. Appl. Environ. MicroBiol. 2014, 80, 247–256. [Google Scholar] [CrossRef]
- Dadachova, E.; Casadevall, A. Ionizing radiation: How fungi cope, adapt, and exploit with the help of melanin. Curr. Opin. MicroBiol. 2008, 11, 525–531. [Google Scholar] [CrossRef]
- Onofri, S.; de Vera, J.P.; Zucconi, L.; Selbmann, L.; Scalzi, G.; Venkateswaran, K.J.; Rabbow, E.; de la Torre, R.; Horneck, G. Survival of Antarctic cryptoendolithic fungi in simulated Martian conditions on-board the International Space Station. AstroBiology 2015, 15, 1052–1059. [Google Scholar]
- Onofri, S.; Selbmann, L.; Pacelli, C.; Zucconi, L.; Rabbow, E.; de Vera, J.P. Survival, DNA, and ultrastructural integrity of a cryptoendolithic Antarctic fungus in Mars and Lunar rock analogs exposed outside the International Space Station. AstroBiology 2019, 19, 170–182. [Google Scholar] [CrossRef]
- Selbmann, L.; Isola, D.; Zucconi, L.; Onofri, S. Resistance to UV-B induced DNA damage in extreme-tolerant cryptoendolithic Antarctic fungi: Detection by PCR assays. Fungal Biol. 2011, 115, 937–944. [Google Scholar] [CrossRef]
- Selbmann, L.; Pacelli, C.; Zucconi, L.; Dadachova, E.; Moeller, R.; de Vera, J.P.; Onofri, S. Resistance of an Antarctic cryptoendolithic black fungus to radiation gives new insights of astrobiological relevance. Fungal Biol. 2018, 122, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Campos, X.; Kinsela, A.S.; Waite, T.D.; Collins, R.N.; Neilan, B.A. Fodinomyces uranophilus gen. nov. sp. nov. and Coniochaeta fodinicola sp. nov.; two uranium mine inhabiting Ascomycota fungi from northern Australia. Mycologia 2014, 106, 1073–1089. [Google Scholar] [PubMed]
- Gorbushina, A.A.; Kotlova, E.R.; Sherstneva, O.A. Cellular responses of microcolonial rock fungi to long-term desiccation and subsequent rehydration. Stud. Mycol. 2008, 61, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Plemenitaš, A.; Gunde-Cimerman, N. Cellular responses in the halophilic black yeast Hortaea werneckii to high environmental salinity. In Adaptation to Life at High Salt Concentrations in Archaea, Bacteria, and Eukarya; Springer: Dordrecht, Germany, 2005; pp. 453–470. [Google Scholar]
- Baker, B.J.; Lutz, M.A.; Dawson, S.C.; Bond, P.L.; Banfield, J.F. Metabolically active eukaryotic communities in extremely acidic mine drainage. Appl. Environ. MicroBiol. 2004, 70, 6264–6271. [Google Scholar] [CrossRef] [PubMed]
- Seyedmousavi, S.; Badali, H.; Chlebicki, A.; Zhao, J.; Prenafeta-Boldu, F.X.; de Hoog, G.S. Exophiala sideris, a novel black yeast isolated from environments polluted with toxic alkyl benzenes and arsenic. Fungal Biol. 2011, 115, 1030–1037. [Google Scholar] [CrossRef]
- Isola, D.; Selbmann, L.; de Hoog, G.S.; Fenice, M.; Onofri, S.; Prenafeta-Boldú, F.X.; Zucconi, L. Isolation and screening of black fungi as degraders of volatile aromatic hydrocarbons. Mycopathologia 2013, 175, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Ruibal, C.; Platas, G.; Bills, G.F. Isolation and characterization of melanized fungi from limestone formations in Mallorca. Mycol. Prog. 2005, 4, 23–38. [Google Scholar] [CrossRef]
- Sert, H.B.; Sümbül, H.; Sterflinger, K. Microcolonial fungi from antique marbles in Perge/side/Termessos (Antalya/Turkey). Antonie Van Leeuwenhoek 2007, 91, 217–227. [Google Scholar] [CrossRef]
- Staley, J.T.; Palmer, F.; Adams, J.B. Microcolonial fungi: Common inhabitants on desert rocks? Science 1982, 215, 1093–1095. [Google Scholar]
- Ruibal, C.; Selbmann, L.; Avci, S.; Martin-Sanchez, P.; Gorbushina, A. Roof-Inhabiting Cousins of Rock-Inhabiting Fungi: Novel Melanized Microcolonial Fungal Species from Photocatalytically Reactive Subaerial Surfaces. Life 2018, 8, 30. [Google Scholar] [CrossRef]
- Martin-Sanchez, P.M.; Gebhardt, C.; Toepel, J.; Barry, J.; Munzke, N.; Günster, J.; Gorbushina, A.A. Monitoring microbial soiling in photovoltaic systems: A qPCR-based approach. Int. Biodeter. Biodegr. 2018, 129, 13–22. [Google Scholar] [CrossRef]
- Selbmann, L.; de Hoog, G.S.; Mazzaglia, A.; Friedmann, E.I.; Onofri, S. Fungi at the edge of life: Cryptoendolithic black fungi from Antarctic deserts. Stud. Mycol. 2005, 51, 1–32. [Google Scholar]
- Selbmann, L.; de Hoog, G.S.; Zucconi, L.; Isola, D.; Ruisi, S.; van den Ende, A.H.G.; Ruibal, C.; De Leo, F.; Urzì, C.; Onofri, S. Drought meets acid: Three new genera in a dothidealean clade of extremotolerant fungi. Stud. Mycol. 2008, 61, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Branda, E.; Turchetti, B.; Diolaiuti, G.; Pecci, M.; Smiraglia, C.; Buzzini, P. Yeast and yeast-like diversity in the southernmost glacier of Europe (Calderone Glacier, Apennines, Italy). FEMS MicroBiol. Ecol. 2010, 72, 354–369. [Google Scholar] [CrossRef] [PubMed]
- Zalar, P.; Gunde-Cimerman, N. Cold-adapted yeasts in Arctic habitats. In Cold-Adapted Yeasts; Buzzini, P., Margesin, R., Eds.; Springer: Berlin, Germany, 2014; pp. 49–74. [Google Scholar]
- Brunner, I.; Plötze, M.; Rieder, S.; Zumsteg, A.; Furrer, G.; Frey, B. Pioneering fungi from the Damma glacier forefield in the Swiss Alps can promote granite weathering. GeoBiology 2011, 9, 266–279. [Google Scholar] [CrossRef] [PubMed]
- Coleine, C.; Stajich, J.E.; Zucconi, L.; Onofri, S.; Pombubpa, N.; Egidi, E.; Franks, A.; Buzzini, P.; Selbmann, L. Antarctic cryptoendolithic fungal communities are highly adapted and dominated by Lecanoromycetes and Dothideomycetes. Front. MicroBiol. 2018, 9, 1392. [Google Scholar] [CrossRef]
- Perini, L.; Gostinčar, C.; Anesio, A.M.; Williamson, C.; Tranter, M.; Gunde-Cimerman, N. Darkening of the Greenland Ice Sheet: Fungal abundance and diversity are associated with algal bloom. Front. MicroBiol. 2019, 10, 557. [Google Scholar] [CrossRef]
- Blasi, B.; Tafer, H.; Tesei, D.; Sterflinger, K. From glacier to sauna: Rna-seq of the human pathogen black fungus Exophiala dermatitidis under varying temperature conditions exhibits common and novel fungal response. PLoS ONE 2015, 10, e0127103. [Google Scholar] [CrossRef]
- Listemann, H.; Freiesleben, H. Exophiala mesophila spec. nov. Mycoses 1996, 39, 1–3. [Google Scholar] [CrossRef]
- Zalar, P.; Novak, M.; De Hoog, G.S.; Gunde-Cimerman, N. Dishwashers–a man-made ecological niche accommodating human opportunistic fungal pathogens. Fungal Biol. 2011, 115, 997–1007. [Google Scholar]
- Babič, M.N.; Gostinčar, C.; Gunde-Cimerman, N. Microorganisms populating the water-related indoor biome. Appl. MicroBiol. Biotechnol. 2020, 104, 6443–6462. [Google Scholar] [CrossRef] [PubMed]
- Moreno, L.F.; Mayer, V.; Voglmayr, H.; Blatrix, R.; Stielow, J.B.; Teixeira, M.M.; Vicente, V.A.; de Hoog, S. Genomic analysis of ant domatia-associated melanized fungi (Chaetothyriales, Ascomycota). Mycol. Prog. 2019, 18, 541–552. [Google Scholar]
- Vicente, V.A.; Attili-Angelis, D.; Pie, M.R.; Queiroz-Telles, F.; Cruz, L.M.; Najafzadeh, M.J.; de Hoog, G.S.; Zhao, J.; Pizzirani-Kleiner, A. Environmental isolation of black yeast-like fungi involved in human infection. Stud. Mycol. 2008, 61, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Vicente, V.A.; Weiss, V.A.; Bombassaro, A.; Moreno, L.F.; Costa, F.F.; Raittz, R.T.; de Castro, R.J. Comparative genomics of sibling species of Fonsecaea associated with human chromoblastomycosis. Front. MicroBiol. 2017, 8, 1924. [Google Scholar] [CrossRef]
- Prenafeta-Boldú, F.X.; Guivernau, M.; Gallastegui, G.; Viñas, M.; de Hoog, G.S.; Elías, A. Fungal/bacterial interactions during the biodegradation of TEX hydrocarbons (toluene, ethylbenzene and p-xylene) in gas biofilters operated under xerophilic conditions. FEMS MicroBiol. Ecol. 2012, 80, 722–734. [Google Scholar] [CrossRef]
- Mohanta, T.K.; Bae, H. Functional Genomics and Signaling Events in Mycorrhizal Symbiosis. J. Plant Interact. 2015, 10, 21–40. [Google Scholar] [CrossRef]
- Gostinčar, C.; Gunde-Cimerman, N. Overview of oxidative stress response genes in selected halophilic fungi. Genes 2018, 9, 143. [Google Scholar]
- Coleine, C.; Masonjones, S.; Sterflinger, K.; Onofri, S.; Selbmann, L.; Stajich, J.E. Peculiar genomic traits in the stress-adapted cryptoendolithic Antarctic fungus Friedmanniomyces endolithicus. Fungal Biol. 2020, 124, 458–467. [Google Scholar] [CrossRef]
- Goffeau, A.; Barrell, B.G.; Bussey, H.; Davis, R.W.; Dujon, B.; Feldmann, H.; Galibert, F.; Hoheisel, J.D.; Jacq, C.; Johnston, M.; et al. Life with 6000 genes. Science 1996, 274, 563–567. [Google Scholar] [CrossRef]
- Grigoriev, I.V.; Nikitin, R.; Haridas, S.; Kuo, A.; Ohm, R.; Otillar, R.; Riley, R.; Salamov, A.; Zhao, X.; Korzeniewski, F.; et al. MycoCosm portal: Gearing up for 1000 fungal genomes. Nucleic Acids Res. 2014, 42, D699–D704. [Google Scholar]
- Kirk, P.; Cannon, P.; Stalpers, J. Dictionary of the Fungi, 10th ed.; CABI: Wallingford, UK, 2008; p. 771. [Google Scholar]
- Choi, J.; Kim, S.H. A genome tree of life for the fungi kingdom. Proc. Natl. Acad. Sci. USA 2017, 114, 9391–9396. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Martinez, D.A.; Gujja, S.; Sykes, S.M.; Zeng, Q.; Szaniszlo, P.J.; Wang, Z.; Cuomo, C.A. Comparative genomic and transcriptomic analysis of Wangiella dermatitidis, a major cause of phaeohyphomycosis and a model black yeast human pathogen. G3 2014, 4, 561–578. [Google Scholar] [CrossRef] [PubMed]
- Gostinčar, C.; Ohm, R.A.; Kogej, T.; Sonjak, S.; Turk, M.; Zajc, J.; Zalar, P.; Grube, M.; Sun, H.; Han, J.; et al. Genome sequencing of four Aureobasidium pullulans varieties: Biotechnological potential, stress tolerance, and description of new species. BMC Genome 2014, 15, 549. [Google Scholar] [CrossRef] [PubMed]
- Lenassi, M.; Gostinčar, C.; Jackman, S.; Turk, M.; Sadowski, I.; Nislow, C.; Jones, S.; Birol, I.; Gunde-Cimerman, N.; Plemenitaš, A. Whole genome duplication and enrichment of metal cation transporters revealed by de novo genome sequencing of extremely halotolerant black yeast Hortaea werneckii. PLoS ONE 2013, 8, e71328. [Google Scholar] [CrossRef] [PubMed]
- Sterflinger, K.; Lopandic, K.; Pandey, R.V.; Blasi, B.; Kriegner, A. Nothing special in the specialist? Draft genome sequence of Cryomyces antarcticus, the most extremophilic fungus from Antarctica. PLoS ONE 2014, 9, e109908. [Google Scholar] [CrossRef] [PubMed]
- Coleine, C.; Masonjones, S.; Selbmann, L.; Zucconi, L.; Onofri, S.; Pacelli, C.; Stajich, J.E. Draft genome sequences of the antarctic endolithic fungi Rachicladosporium antarcticum CCFEE 5527 and Rachicladosporium sp. CCFEE 5018. Genome Announc. 2017, 5, e00397-17. [Google Scholar] [CrossRef]
- Coleine, C.; Selbmann, L.; Masonjones, S.; Onofri, S.; Zucconi, L.; Stajich, J.E. Draft Genome Sequence of an Antarctic Isolate of the Black Yeast Fungus Exophiala mesophila. MicroBiol. Resour. Announc. 2019, 8, e00142-19. [Google Scholar]
- Blasi, B.; Tafer, H.; Kustor, C.; Poyntner, C.; Lopandic, K.; Sterflinger, K. Genomic and transcriptomic analysis of the toluene degrading black yeast Cladophialophora immunda. Sci. Rep. 2017, 7, 11436. [Google Scholar] [CrossRef]
- Teixeira, M.D.M.; Moreno, L.F.; Stielow, B.J.; Muszewska, A.; Hainaut, M.; Gonzaga, L.; Young, S. Exploring the genomic diversity of black yeasts and relatives (Chaetothyriales, Ascomycota). Stud. Mycol. 2017, 86, 1–28. [Google Scholar] [CrossRef]
- Dadachova, E.; Bryan, R.A.; Huang, X.; Moadel, T.; Schweitzer, A.D.; Aisen, P.; Nosanchuk, J.D.; Casadevall, A. Ionizing radiation changes the electronic properties of melanin and enhances the growth of melanized fungi. PLoS ONE 2007, 2, e457. [Google Scholar] [CrossRef]
- Karányi, Z.; Holb, I.; Hornok, L.; Pócsi, I.; Miskei, M. FSRD: Fungal Stress Response Database. Database 2013, 2012, bat037. [Google Scholar] [CrossRef] [PubMed]
- de Vries, R.P.; Riley, R.; Ad Wiebenga, A.; Aguilar-Osorio, G.; Amillis, S.; Uchima, C.A.; Anderluh Asadollahi, M.; Askin, M.; Barry, K.; Battaglia, E.; et al. Comparative genomics reveals high biological diversity and specific adaptations in the industrially and medically important fungal genus Aspergillus. Genome Biol. 2017, 18, 28. [Google Scholar] [CrossRef] [PubMed]
- Emri, T.; Antal, K.; Riley, R.; Karányi, Z.; Miskei, M.; Orosz, E.; Baker, S.E.; Wiebenga, A.; de Vries, R.P.; Pócsi, I. Duplications and losses of genes encoding known elements of the stress defence system of the Aspergilli contribute to the evolution of these filamentous fungi but do not directly influence their environmental stress tolerance. Stud. Mycol. 2018, 91, 23–36. [Google Scholar] [CrossRef] [PubMed]


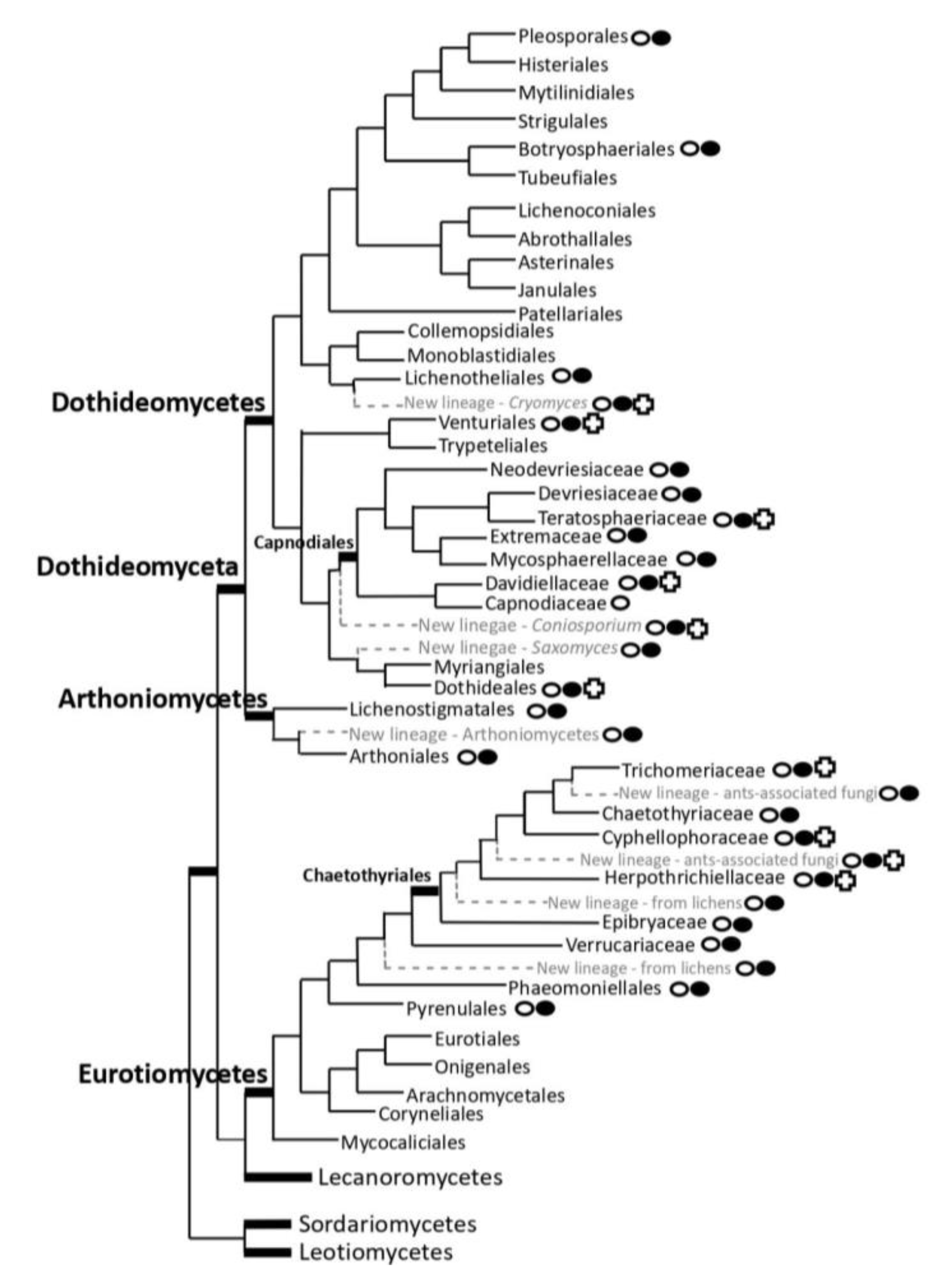
 and lineages represented in the selection proposed
and lineages represented in the selection proposed  in the ‘STRES’ project; lineages where black fungal genomes of some genera are available
in the ‘STRES’ project; lineages where black fungal genomes of some genera are available  .
.
 and lineages represented in the selection proposed
and lineages represented in the selection proposed  in the ‘STRES’ project; lineages where black fungal genomes of some genera are available
in the ‘STRES’ project; lineages where black fungal genomes of some genera are available  .
.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selbmann, L.; Benkő, Z.; Coleine, C.; de Hoog, S.; Donati, C.; Druzhinina, I.; Emri, T.; Ettinger, C.L.; Gladfelter, A.S.; Gorbushina, A.A.; et al. Shed Light in the DaRk LineagES of the Fungal Tree of Life—STRES. Life 2020, 10, 362. https://doi.org/10.3390/life10120362
Selbmann L, Benkő Z, Coleine C, de Hoog S, Donati C, Druzhinina I, Emri T, Ettinger CL, Gladfelter AS, Gorbushina AA, et al. Shed Light in the DaRk LineagES of the Fungal Tree of Life—STRES. Life. 2020; 10(12):362. https://doi.org/10.3390/life10120362
Chicago/Turabian StyleSelbmann, Laura, Zsigmond Benkő, Claudia Coleine, Sybren de Hoog, Claudio Donati, Irina Druzhinina, Tamás Emri, Cassie L. Ettinger, Amy S. Gladfelter, Anna A. Gorbushina, and et al. 2020. "Shed Light in the DaRk LineagES of the Fungal Tree of Life—STRES" Life 10, no. 12: 362. https://doi.org/10.3390/life10120362
APA StyleSelbmann, L., Benkő, Z., Coleine, C., de Hoog, S., Donati, C., Druzhinina, I., Emri, T., Ettinger, C. L., Gladfelter, A. S., Gorbushina, A. A., Grigoriev, I. V., Grube, M., Gunde-Cimerman, N., Karányi, Z. Á., Kocsis, B., Kurbessoian, T., Miklós, I., Miskei, M., Muggia, L., ... Stajich, J. E. (2020). Shed Light in the DaRk LineagES of the Fungal Tree of Life—STRES. Life, 10(12), 362. https://doi.org/10.3390/life10120362















