Outcomes Evaluated in Controlled Clinical Trials on the Management of COVID-19: A Methodological Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Selection and Data Extraction
2.2. Outcome Grouping and Classification
3. Results
3.1. Description of the Included Studies
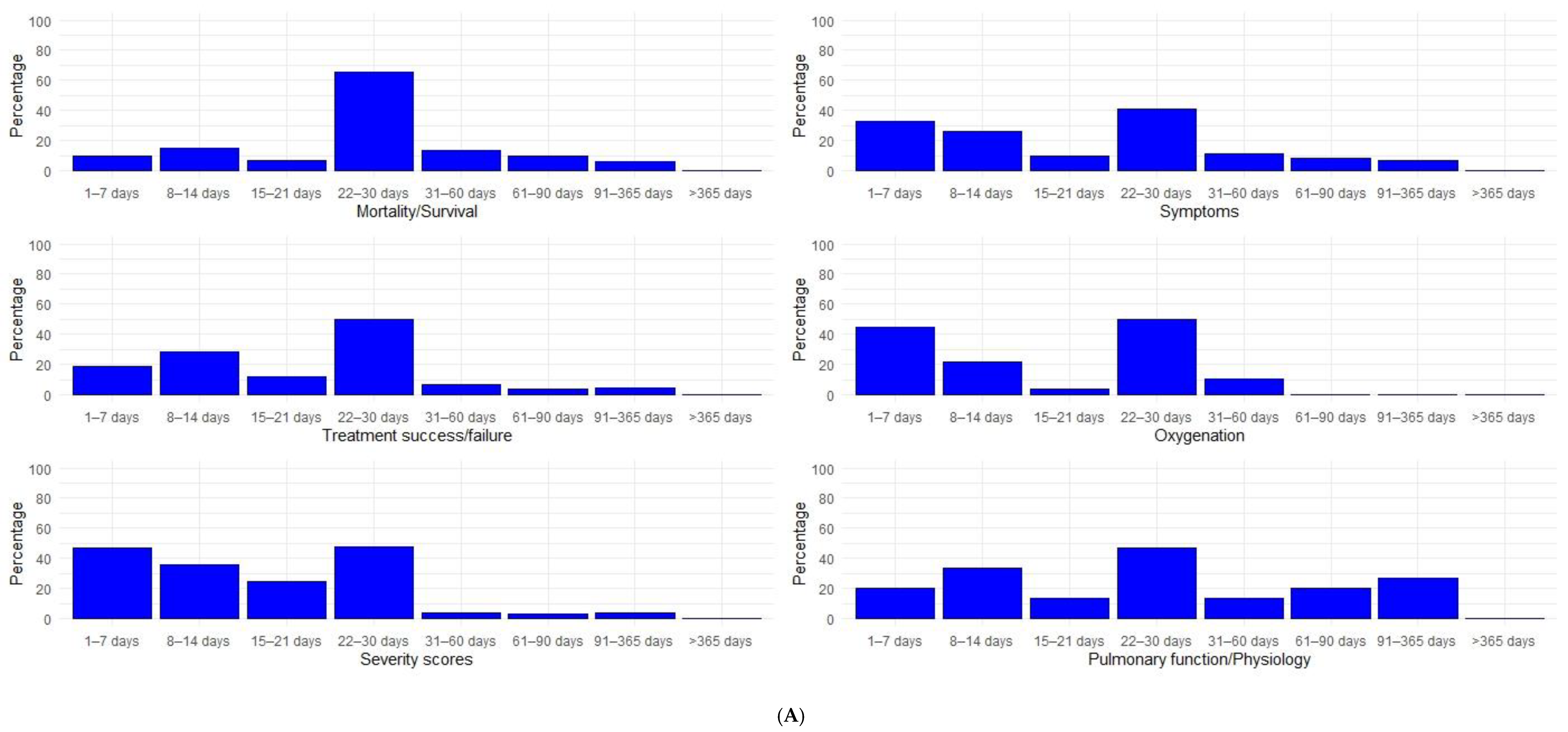
3.2. Outcome Measurement Instruments
3.2.1. Mortality/Survival (Assessed by 284 Outcomes)
3.2.2. Clinical Outcomes
3.2.3. Physiological Outcomes
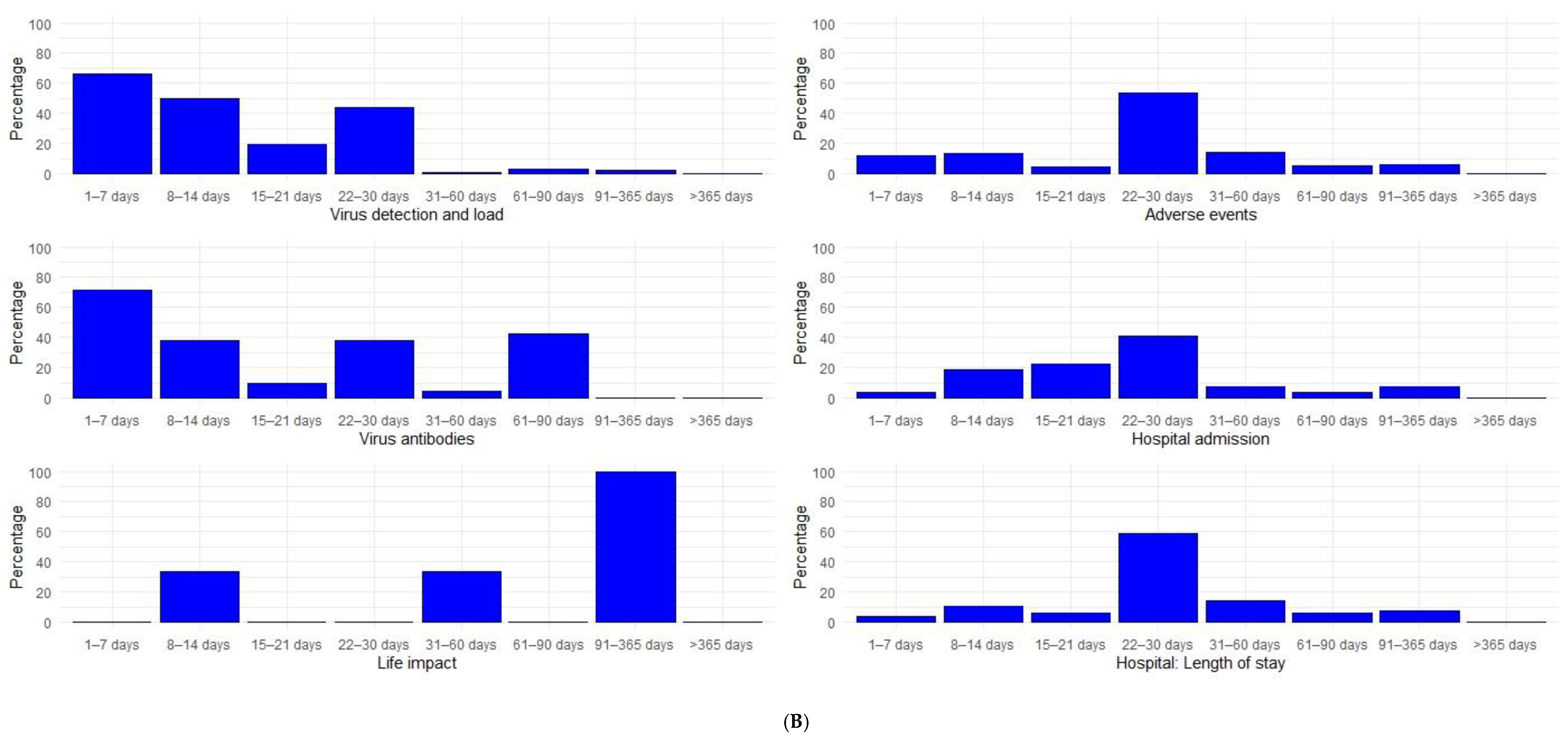
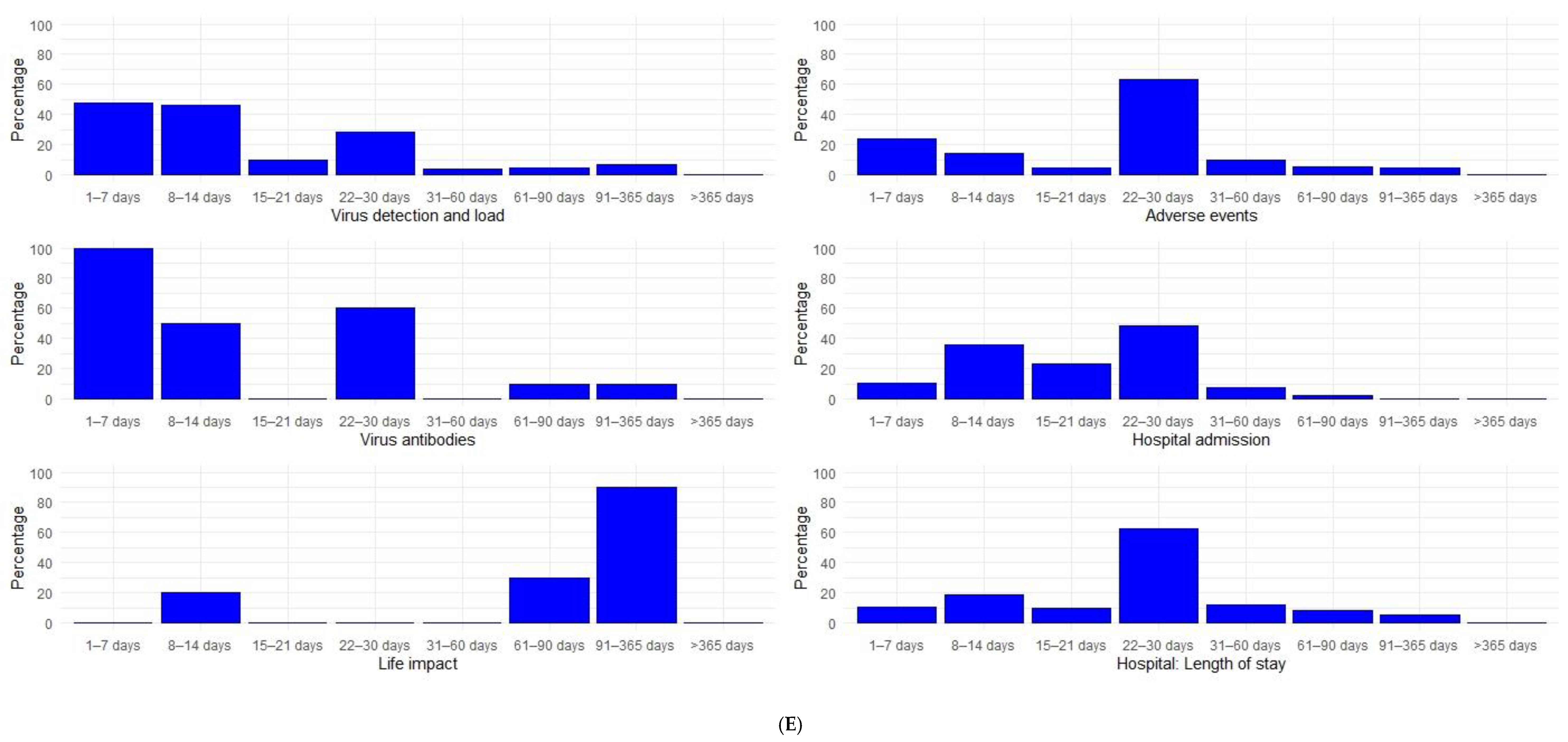
3.2.4. Adverse Events
3.2.5. Life Impact (13 Outcomes)
3.2.6. Resources Use
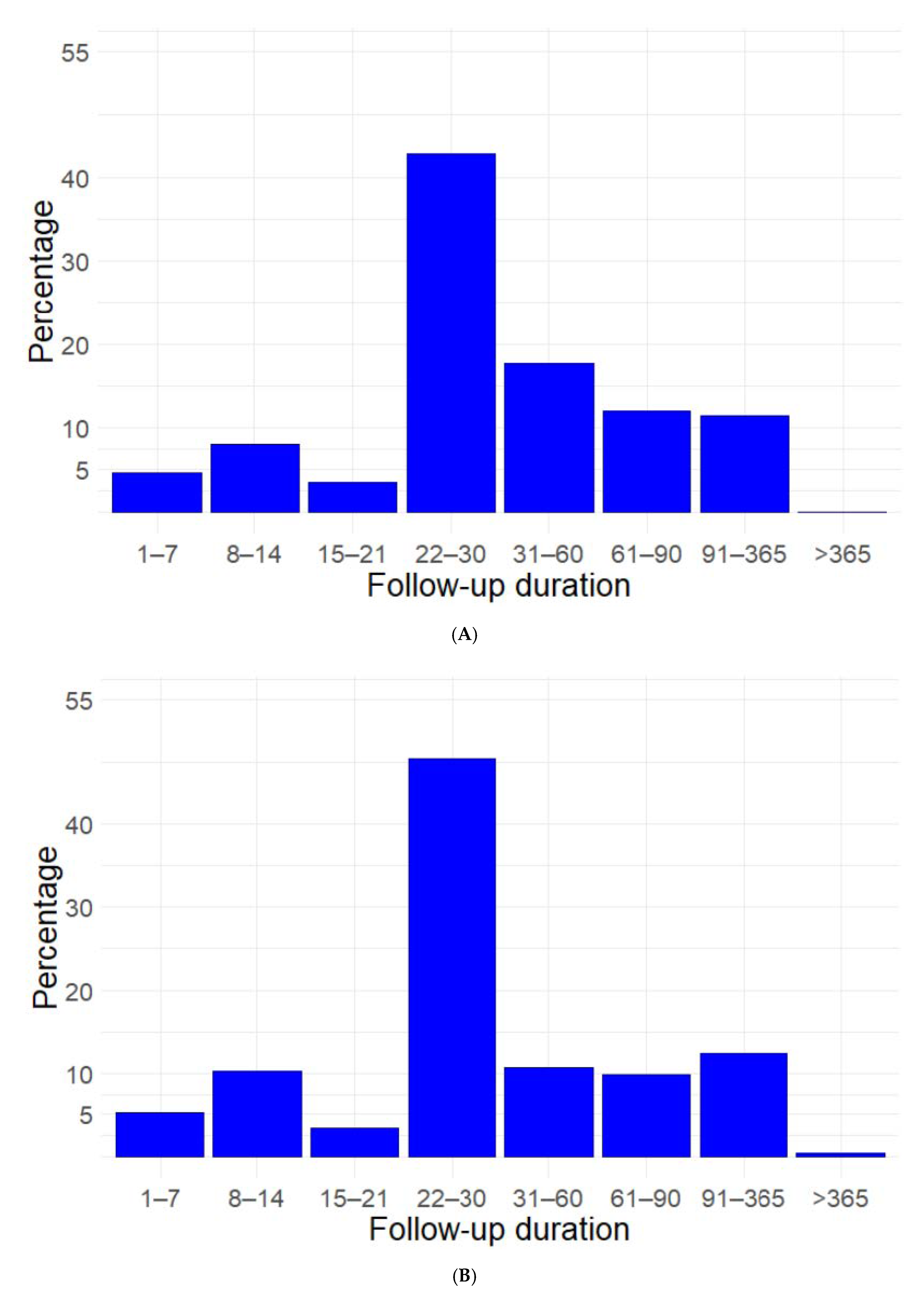
3.3. Study Follow-Up
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A





| NCT Number | N | NCT Number | N | NCT Number | N | NCT Number | N |
|---|---|---|---|---|---|---|---|
| NCT04336904 | 100 | NCT04346147 | 165 | NCT04324528 | 30 | NCT04393038 | 1034 |
| NCT04345445 | 310 | NCT04360876 | 90 | NCT04343651 | 75 | NCT04392141 | 200 |
| NCT04359095 | 1600 | NCT04336332 | 160 | NCT04366908 | 1008 | NCT04405102 | 48 |
| NCT04347915 | 60 | NCT04357444 | 30 | NCT04349618 | 200 | NCT04385043 | 400 |
| NCT04333407 | 3170 | NCT04317040 | 230 | NCT04355962 | 64 | NCT04401579 | 1032 |
| NCT04336462 | 100 | NCT04342897 | 200 | NCT04347239 | 390 | NCT04390594 | 258 |
| NCT04342689 | 1500 | NCT04365231 | 50 | NCT04323800 | 487 | NCT04385940 | 64 |
| NCT04376788 | 15 | NCT04346368 | 20 | NCT04321096 | 580 | NCT04405310 | 80 |
| NCT04333420 | 130 | NCT04371406 | 2770 | NCT04268537 | 120 | NCT04380519 | 372 |
| NCT04360356 | 100 | NCT04288102 | 90 | NCT04332666 | 60 | NCT04382755 | 81 |
| NCT04370262 | 942 | NCT04286503 | 520 | NCT04349098 | 230 | NCT04400890 | 200 |
| NCT04350593 | 900 | NCT04351581 | 215 | NCT04332835 | 80 | NCT04391712 | 20 |
| NCT04372979 | 80 | NCT04351243 | 270 | NCT04361461 | 500 | NCT04394416 | 204 |
| NCT04325633 | 584 | NCT04347512 | 405 | NCT04366271 | 106 | NCT04393311 | 150 |
| NCT04339660 | 30 | NCT04339816 | 240 | NCT04366089 | 152 | NCT04392414 | 60 |
| NCT04362813 | 450 | NCT04347980 | 122 | NCT04373733 | 450 | NCT04398303 | 70 |
| NCT04354389 | 82 | NCT04357808 | 30 | NCT04312009 | 200 | NCT04391127 | 200 |
| NCT04362137 | 402 | NCT04358926 | 30 | NCT04361474 | 120 | NCT04396106 | 180 |
| NCT04359615 | 40 | NCT04293692 | 0 | NCT04315948 | 3100 | NCT04405843 | 400 |
| NCT04359316 | 40 | NCT04362176 | 500 | NCT04311177 | 580 | NCT04381052 | 30 |
| NCT04343768 | 60 | NCT04338828 | 260 | NCT04261426 | 80 | NCT04397562 | 204 |
| NCT04280705 | 800 | NCT04353180 | 45 | NCT04255017 | 400 | NCT04385264 | 800 |
| NCT04329832 | 300 | NCT04371952 | 330 | NCT04254874 | 100 | NCT04385264 | 800 |
| NCT04365257 | 220 | NCT04335305 | 24 | NCT04341935 | 20 | NCT04382586 | 52 |
| NCT04350671 | 40 | NCT04347538 | 90 | NCT04261270 | 60 | NCT04381858 | 500 |
| NCT04350684 | 40 | NCT04372628 | 900 | NCT04342169 | 400 | NCT04379479 | 562 |
| NCT04330586 | 141 | NCT04350320 | 102 | NCT04329195 | 554 | NCT04386616 | 300 |
| NCT04361318 | 100 | NCT04364763 | 252 | NCT04321616 | 700 | NCT04382651 | 120 |
| NCT04361942 | 24 | NCT04344730 | 550 | NCT04311697 | 144 | NCT04393246 | 1407 |
| NCT04315298 | 400 | NCT04341038 | 84 | NCT04357730 | 60 | NCT04390503 | 200 |
| NCT04359953 | 1600 | NCT04328272 | 75 | NCT04367077 | 400 | NCT04394208 | 50 |
| NCT04377620 | 500 | NCT04374487 | 100 | NCT04360096 | 288 | NCT04402866 | 159 |
| NCT04330638 | 342 | NCT04328480 | 2500 | NCT04359810 | 105 | NCT04395170 | 75 |
| NCT04366739 | 40 | NCT04350580 | 138 | NCT03042143 | 75 | NCT04404426 | 100 |
| NCT04369742 | 626 | NCT04323345 | 1000 | NCT04333368 | 40 | NCT04386694 | 30 |
| NCT04363372 | 90 | NCT04366232 | 50 | NCT02735707 | 7100 | NCT04395768 | 200 |
| NCT04326920 | 80 | NCT04342663 | 152 | NCT04348695 | 94 | NCT04402203 | 50 |
| NCT04353284 | 114 | NCT04343001 | 10000 | NCT04347382 | 30 | NCT04391309 | 300 |
| NCT04359277 | 1000 | NCT04356534 | 40 | NCT04358081 | 444 | NCT04389840 | 524 |
| NCT04351763 | 804 | NCT04344288 | 304 | NCT04342650 | 210 | NCT04397718 | 198 |
| NCT04340544 | 2700 | NCT04348383 | 120 | NCT04279197 | 136 | NCT04379076 | 48 |
| NCT04366115 | 126 | NCT04341870 | 27 | NCT04345861 | 7 | NCT04401475 | 510 |
| NCT04366050 | 560 | NCT04352400 | 256 | NCT04376684 | 800 | NCT04401475 | 510 |
| NCT04341675 | 30 | NCT04360824 | 170 | NCT04349592 | 456 | NCT04379271 | 230 |
| NCT04329923 | 400 | NCT04369469 | 270 | NCT04324463 | 4000 | NCT04390061 | 116 |
| NCT04329923 | 400 | NCT04367831 | 100 | NCT04324463 | 4000 | NCT04383535 | 333 |
| NCT04329923 | 400 | NCT04251767 | 0 | NCT04371393 | 300 | NCT04405921 | 200 |
| NCT04361643 | 120 | NCT04368923 | 60 | NCT04351295 | 40 | NCT04382053 | 120 |
| NCT04355143 | 150 | NCT04348513 | 60 | NCT04363840 | 1080 | NCT04398290 | 30 |
| NCT04333628 | 210 | NCT04257656 | 237 | NCT04351347 | 300 | NCT04392128 | 114 |
| NCT04333628 | 210 | NCT04338126 | 60 | NCT03808922 | 250 | NCT04406532 | 100 |
| NCT04334382 | 1550 | NCT04334850 | 194 | NCT04341493 | 86 | NCT04403646 | 140 |
| NCT04357990 | 81 | NCT04335071 | 100 | NCT04362059 | 24 | NCT04392531 | 120 |
| NCT04335136 | 200 | NCT04348305 | 1000 | NCT04346446 | 29 | NCT04385095 | 400 |
| NCT04362189 | 110 | NCT04362111 | 20 | NCT04365582 | 640 | NCT04390464 | 1167 |
| NCT04330690 | 440 | NCT04355364 | 100 | NCT04363437 | 70 | NCT04381871 | 110 |
| NCT04359511 | 210 | NCT04377503 | 40 | NCT04325906 | 346 | NCT04390139 | 30 |
| NCT04351724 | 500 | NCT04373460 | 1344 | NCT04346628 | 120 | NCT04386447 | 145 |
| NCT04344444 | 600 | NCT04343963 | 436 | NCT04327388 | 409 | NCT04395456 | 144 |
| NCT04344236 | 48 | NCT04349410 | 500 | NCT04344535 | 500 | NCT04401527 | 200 |
| NCT04307693 | 150 | NCT04354428 | 630 | NCT04338906 | 334 | NCT04387760 | 150 |
| NCT04331899 | 120 | NCT04351490 | 3140 | NCT04325893 | 1300 | NCT04393948 | 48 |
| NCT04362332 | 950 | NCT04341415 | 60 | NCT04371367 | 108 | NCT04387240 | 22 |
| NCT04336254 | 20 | NCT04374552 | 140 | NCT04374539 | 116 | NCT04390217 | 120 |
| NCT04332094 | 276 | NCT04365153 | 45 | NCT04251871 | 150 | NCT04397510 | 50 |
| NCT04292899 | 6000 | NCT04356937 | 300 | NCT04361253 | 220 | NCT04390022 | 24 |
| NCT04370782 | 750 | NCT04361032 | 260 | NCT04322123 | 630 | NCT04405570 | 44 |
| NCT04312997 | 100 | NCT04364009 | 240 | NCT04363502 | 30 | NCT04399356 | 100 |
| NCT04377711 | 400 | NCT04353271 | 58 | NCT04322396 | 226 | NCT04399980 | 60 |
| NCT04348409 | 50 | NCT04364737 | 300 | NCT04346693 | 320 | NCT04382040 | 50 |
| NCT04347954 | 45 | NCT04355728 | 24 | NCT04344041 | 260 | NCT04401293 | 308 |
| NCT04360551 | 40 | NCT04366245 | 72 | NCT04321278 | 440 | NCT04379492 | 120 |
| NCT04343989 | 90 | NCT04357457 | 212 | NCT04345289 | 1500 | NCT04389580 | 160 |
| NCT04292730 | 1600 | NCT04333914 | 273 | NCT04358783 | 30 | NCT04384445 | 20 |
| NCT04358549 | 50 | NCT04351191 | 400 | NCT04353037 | 850 | NCT04400929 | 30 |
| NCT04345523 | 278 | NCT04358406 | 60 | NCT04260594 | 380 | NCT04391179 | 80 |
| NCT04346615 | 120 | NCT04326790 | 180 | NCT04326426 | 300 | NCT04405739 | 80 |
| NCT04244591 | 80 | NCT04372082 | 480 | NCT04345406 | 60 | NCT04401150 | 800 |
| NCT04329650 | 200 | NCT04331054 | 436 | NCT04366856 | 500 | NCT04397497 | 50 |
| NCT04331470 | 30 | NCT04344184 | 200 | NCT04338802 | 96 | NCT04402957 | 60 |
| NCT04320615 | 330 | NCT04338698 | 500 | NCT04345887 | 60 | NCT04381377 | 394 |
| NCT04372186 | 379 | NCT04335786 | 651 | NCT04374474 | 75 | NCT04403100 | 1968 |
| NCT04358809 | 480 | NCT04335552 | 500 | NCT04322773 | 200 | NCT04385771 | 80 |
| NCT04273529 | 100 | NCT04357860 | 120 | NCT04345419 | 120 | NCT04381936 | 12000 |
| NCT04374279 | 60 | NCT04351516 | 350 | NCT04347031 | 320 | NCT04402060 | 66 |
| NCT04273581 | 40 | NCT04366063 | 60 | NCT04350281 | 60 | NCT04392778 | 30 |
| NCT04374032 | 120 | NCT04374019 | 240 | NCT04343729 | 416 | NCT04394377 | 600 |
| NCT04363866 | 40 | NCT04356495 | 1057 | NCT04261907 | 160 | NCT04403243 | 70 |
| NCT04342221 | 220 | NCT04346667 | 400 | NCT04264533 | 140 | NCT04402944 | 60 |
| NCT04315896 | 500 | NCT04354441 | 600 | NCT04275388 | 426 | NCT04382846 | 80 |
| NCT04355767 | 206 | NCT04347941 | 200 | NCT04322682 | 6000 | NCT04403555 | 40 |
| NCT04338074 | 100 | NCT04328012 | 4000 | NCT04355052 | 250 | NCT04395807 | 120 |
| NCT04368000 | 60 | NCT04338009 | 152 | NCT04341727 | 500 | NCT04404218 | 480 |
| NCT04331600 | 400 | NCT04310228 | 150 | NCT04346927 | 30 | NCT04380935 | 60 |
| NCT04347174 | 40 | NCT04295551 | 80 | NCT04328467 | 1500 | NCT04404361 | 358 |
| NCT04363060 | 104 | NCT04365985 | 500 | NCT03852537 | 90 | NCT04389450 | 140 |
| NCT04332107 | 2271 | NCT04273763 | 18 | NCT04367168 | 174 | NCT04395144 | 346 |
| NCT04273646 | 48 | NCT04369794 | 1000 | NCT04276688 | 127 | NCT04396067 | 360 |
| NCT04349241 | 100 | NCT04371107 | 64 | NCT04346940 | 30 | NCT04383717 | 60 |
| NCT04363203 | 300 | NCT04359862 | 50 | NCT03680274 | 800 | NCT04385186 | 60 |
| NCT04252664 | 308 | NCT04324021 | 54 | NCT04308668 | 1309 | NCT04382391 | 20 |
| NCT04341116 | 144 | NCT04334967 | 1250 | NCT04346979 | 50 | NCT04390152 | 40 |
| NCT04375397 | 46 | NCT04298060 | 280 | NCT04401423 | 100 | NCT04380961 | 270 |
| NCT04358068 | 2000 | NCT04332991 | 510 | NCT04406389 | 186 |
References
- Rothan, H.A.; Byrareddy, S.N. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J. Autoimmun. 2020, 109, 102433. [Google Scholar] [CrossRef]
- Fragkou, P.C.; Belhadi, D.; Peiffer-Smadja, N.; Moschopoulos, C.D.; Lescure, F.X.; Janocha, H.; Karofylakis, E.; Yazdanpanah, Y.; Mentre, F.; Skevaki, C.; et al. Review of trials currently testing treatment and prevention of COVID-19. Clin. Microbiol. Infect. 2020, 26, 988–998. [Google Scholar] [CrossRef] [PubMed]
- Mathioudakis, A.G.; Fally, M.; Hashad, R.; Knight, S.; Felton, T.; Vestbo, J. COVID-19 Clinical Trials: Unravelling a Methodological Gordian Knot. Am. J. Respir. Crit. Care Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, J.J.; Gargon, E.; Clarke, M.; Williamson, P.R. Can a core outcome set improve the quality of systematic reviews?—A survey of the Co-ordinating Editors of Cochrane Review Groups. Trials 2013, 14, 21. [Google Scholar] [CrossRef] [PubMed]
- Roche, N.; Tonia, T.; Bush, A.; Brightling, C.; Kolb, M.; Dinh-Xuan, A.T.; Humbert, M.; Simonds, A.; Adir, Y. Guidance production before evidence generation for critical issues: The example of COVID-19. Eur. Respir. Rev. 2020, 29, 200310. [Google Scholar] [CrossRef]
- Williamson, P.R.; Altman, D.G.; Bagley, H.; Barnes, K.L.; Blazeby, J.M.; Brookes, S.T.; Clarke, M.; Gargon, E.; Gorst, S.; Harman, N.; et al. The COMET Handbook: Version 1.0. Trials 2017, 18, 280. [Google Scholar] [CrossRef]
- Kirkham, J.J.; Gorst, S.; Altman, D.G.; Blazeby, J.M.; Clarke, M.; Tunis, S.; Williamson, P.R.; Group, C.-S. Core Outcome Set-STAndardised Protocol Items: The COS-STAP Statement. Trials 2019, 20, 116. [Google Scholar] [CrossRef]
- Kirkham, J.J.; Gorst, S.; Altman, D.G.; Blazeby, J.M.; Clarke, M.; Devane, D.; Gargon, E.; Moher, D.; Schmitt, J.; Tugwell, P.; et al. Core Outcome Set-STAndards for Reporting: The COS-STAR Statement. PLoS Med. 2016, 13, e1002148. [Google Scholar] [CrossRef]
- Tong, A.; Elliott, J.H.; Azevedo, L.C.; Baumgart, A.; Bersten, A.; Cervantes, L.; Chew, D.P.; Cho, Y.; Cooper, T.; Crowe, S.; et al. Core Outcomes Set for Trials in People With Coronavirus Disease 2019. Crit. Care Med. 2020, 48, 1622. [Google Scholar] [CrossRef]
- Qiu, R.; Zhao, C.; Liang, T.; Hao, X.; Huang, Y.; Zhang, X.; Chen, Z.; Wei, X.; Zhao, M.; Zhong, C.; et al. Core Outcome Set for Clinical Trials of COVID-19 Based on Traditional Chinese and Western Medicine. Front. Pharmacol. 2020, 11, 781. [Google Scholar] [CrossRef]
- Jin, X.; Pang, B.; Zhang, J.; Liu, Q.; Yang, Z.; Feng, J.; Liu, X.; Zhang, L.; Wang, B.; Huang, Y.; et al. Core Outcome Set for Clinical Trials on Coronavirus Disease 2019 (COS-COVID). Engineering 2020, 6, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- WHO Working Group on the Clinical Characterisation and Management of COVID-19 Infection. A minimal common outcome measure set for COVID-19 clinical research. Lancet Infect. Dis. 2020, 20, e192–e197. [Google Scholar] [CrossRef]
- Mathioudakis, A.G.; Moberg, M.; Janner, J.; Alonso-Coello, P.; Vestbo, J. Outcomes reported on the management of COPD exacerbations: A systematic survey of randomised controlled trials. ERJ Open Res. 2019, 5, 00072–02019. [Google Scholar] [CrossRef] [PubMed]
- Mathioudakis, A.G.; Abroug, F.; Agusti, A.; Bakke, P.; Bartziokas, K.; Beghe, B.; Bikov, A.; Bradbury, T.; Brusselle, G. Core Outcome Set for the management of Acute Exacerbations of Chronic Obstructive Pulmonary Disease. The COS-AECOPD ERS Task Force study protocol. ERJ Open Res. 2020, in press. [Google Scholar]
- Alkhaffaf, B.; Blazeby, J.M.; Williamson, P.R.; Bruce, I.A.; Glenny, A.M. Reporting of outcomes in gastric cancer surgery trials: A systematic review. BMJ Open 2018, 8, e021796. [Google Scholar] [CrossRef] [PubMed]
- Mathioudakis, A.G.; Janner, J.; Moberg, M.; Alonso-Coello, P.; Vestbo, J. A systematic evaluation of the diagnostic criteria for COPD and exacerbations used in randomised controlled trials on the management of COPD exacerbations. ERJ Open Res. 2019, 5, 00136–02019. [Google Scholar] [CrossRef] [PubMed]
- Dodd, S.; Clarke, M.; Becker, L.; Mavergames, C.; Fish, R.; Williamson, P.R. A taxonomy has been developed for outcomes in medical research to help improve knowledge discovery. J. Clin. Epidemiol. 2018, 96, 84–92. [Google Scholar] [CrossRef]
- World Health Organization. Novel Coronavirus COVID-19 Therapeutic Trial Synopsis; WHO R&D Blueprint: Geneva, Switzerland, 2020. [Google Scholar]
- Vincent, J.L.; Moreno, R.; Takala, J.; Willatts, S.; De Mendonca, A.; Bruining, H.; Reinhart, C.K.; Suter, P.M.; Thijs, L.G. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996, 22, 707–710. [Google Scholar] [CrossRef]
- McGinley, A.; Pearse, R.M. A national early warning score for acutely ill patients. BMJ 2012, 345, e5310. [Google Scholar] [CrossRef]
- Garrigues, E.; Janvier, P.; Kherabi, Y.; Le Bot, A.; Hamon, A.; Gouze, H.; Doucet, L.; Berkani, S.; Oliosi, E.; Mallart, E.; et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J. Infect. 2020, 81, e4–e6. [Google Scholar] [CrossRef]
- Halpin, S.J.; McIvor, C.; Whyatt, G.; Adams, A.; Harvey, O.; McLean, L.; Walshaw, C.; Kemp, S.; Corrado, J.; Singh, R.; et al. Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation. J. Med. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Carfi, A.; Bernabei, R.; Landi, F.; Gemelli Against, C.-P.-A.C.S.G. Persistent Symptoms in Patients After Acute COVID-19. JAMA 2020, 324, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Mo, X.; Jian, W.; Su, Z.; Chen, M.; Peng, H.; Peng, P.; Lei, C.; Chen, R.; Zhong, N.; Li, S. Abnormal pulmonary function in COVID-19 patients at time of hospital discharge. Eur. Respir. J. 2020, 55, 2001217. [Google Scholar] [CrossRef] [PubMed]
- George, P.M.; Barratt, S.L.; Condliffe, R.; Desai, S.R.; Devaraj, A.; Forrest, I.; Gibbons, M.A.; Hart, N.; Jenkins, R.G.; McAuley, D.F.; et al. Respiratory follow-up of patients with COVID-19 pneumonia. Thorax 2020, 75, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.; Zhang, J.; Qiu, R.; Blackwood, B.; Sinha, I.; Tong, A.; Gargon, L.; Williamson, P. The ‘Meta-Cos’ for Research in COVID-19 Hospitalised Patients. Available online: https://www.comet-initiative.org/assets/downloads/COVID-19%20meta%20COS_Table%201_29th%20October%202020.pdf (accessed on 13 December 2020).




| Study Characteristics | Phase 2 Trials (n = 178) | Later Phase Trials (n = 237) |
|---|---|---|
| Number of participants | ||
| Median (range) | 120 (15–2000) | 253 (7–12,000) |
| Setting | ||
| Community | 25 (14.0%) | 38 (16.0%) |
| Hospital | 137 (77.0%) | 167 (70.5%) |
| Community and Hospital | 3 (1.7%) | 1 (0.4%) |
| ICU | 9 (5.1%) | 18 (7.6%) |
| Other | 0 (0.0%) | 1 (0.4%) |
| Unclear | 4 (2.2%) | 13 (5.5%) |
| Continent | ||
| Africa | 5 (2.8%) | 21 (8.9%) |
| Asia | 29 (16.3%) | 51 (21.5%) |
| Europe | 46 (25.8%) | 94 (39.7%) |
| North America | 90 (50.6%) | 67 (28.3%) |
| Oceania | 1 (0.6%) | 1 (0.4%) |
| South America | 12 (6.7%) | 22 (9.3%) |
| Multiple continents * | 6 (3.4%) | 15 (6.3%) |
| Unclear | 6 (3.4%) | 0 (0.0%) |
| Age range | ||
| Minimum age | ||
| Median (range) | 18 (3–50) | 18 (1–70) |
| Not reported | 2 (1.1%) | 0 (0.0%) |
| Maximum age | ||
| Median (range) | 80 (50–110) | 80 (40–110) |
| Not reported | 115 (64.6%) | 157 (66.0%) |
| Number of interventions | ||
| 2 | 139 (78.1%) | 172 (72.6%) |
| 3 | 25 (14.0%) | 40 (16.9%) |
| 4 | 10 (5.6%) | 11 (4.6%) |
| 5 | 1 (0.6%) | 5 (2.1%) |
| 6 | 3 (1.7%) | 4 (1.7%) |
| 8 | 0 (0.0%) | 3 (1.3%) |
| 11 | 0 (0.0%) | 1 (0.4%) |
| 19 | 0 (0.0%) | 1 (0.4%) |
| Sponsor | ||
| Academic | 124 (69.7%) | 190 (80.2%) |
| Pharmaceutical industry | 54 (30.3%) | 47 (19.8%) |
| Outcome Categories | Definitions |
|---|---|
| Mortality/Survival | Evaluation the survival status. |
| Clinical/Physiological | |
| Treatment success or treatment failure | A clinical evaluation of whether COVID-19 was successfully treated. Usually a composite endpoint based on one or more of the following: survival, symptoms progression or regression, pyrexia regression, oxygen requirements and/or the requirement for ventilation. We only considered in this category binary outcomes describing criteria either for treatment success or treatment failure. Time-to-treatment success or failure is a measurement instrument that could provide more granular information. |
| Severity scores | A quantitative evaluation of disease severity. In this category we included outcomes presenting mean/median scores or change from baseline in a validated score. Outcomes describing predefined score thresholds for treatment success or failure were classified in the previous category. |
| Symptoms | Quantitative or qualitative evaluation of the intensity of one or more symptoms, including but not limited to breathlessness, cough, pyrexia or anosmia. |
| Oxygenation | Physiological measures of oxygenation, including oxygen saturation (SatO2), the partial pressure of oxygen (PaO2) or carbon dioxide (PaCO2). The need for supplementary oxygen or ventilation were summarized in separate outcome categories. |
| Pulmonary function and physiology | Measures of pulmonary functions and lung physiology including the forced expiratory volume in 1 second (FEV1), forced vital capacity (FVC), respiratory muscle strength or the lung compliance. |
| Viral detection and load | Polymerase chain reaction (PCR) to evaluate the presence, persistence and/or load of the severe acute respiratory syndrome coronavirus-2 (SARS-CoV2). |
| Viral antibodies | Detection of the presence and titres of antibodies against SARS-CoV2. |
| Radiological outcomes | Radiological progression in chest x-ray (CXR) or computed tomography (CT) of the chest. |
| Inflammatory biomarkers | The levels and trajectories of any inflammatory biomarkers, including white blood cells count, lymphocytes, neutrophils, eosinophils, monocytes, CD4+ or CD8+ T cell counts, c-reactive protein, interleukins, tumour necrosis factors, or any other inflammatory biomarkers. |
| Other biomarkers | The levels and trajectories of any other biomarkers, including but not limited to kidney function, liver function, haematocrit, coagulation profile, d-dimers, troponin or the brain natriuretic peptide (BNP). |
| Pharmacokinetics/pharmacodynamics | Evaluation of the pharmacokinetics and/or pharmacodynamics of the drug interventions (mainly serum levels over time). |
| Adverse events | Adverse events or grade 3 or more severe adverse events, or serious adverse events, according to the Common Toxicity Criteria for Adverse Events (CTCAE). In this category, we also included outcomes evaluating specific adverse events, such as renal failure, liver failure, pulmonary embolism, myocardial infarction or tachyarrhythmias. Treatment discontinuation was also included in this category. |
| Life impact | Quantitative assessment of the general well-being of participants. |
| Resource use | |
| Need for (higher) level of care | This group of outcomes include the need for (i) hospital admission, (ii) hospital re-admission, (iii) intensive care admission, (iv) invasive ventilation, or need for ECMO. In each category, we also included the composite outcomes consisting of the need for the specific level of care or death. For example: “intensive care admission or death”, as these composite outcomes were developed to account for patients who might have benefitted by the higher level of care but died or patients who were not eligible for the higher level of care due to their baseline clinical status. In studies conducted in the hospital setting, need for hospital admission at a specific follow-up timepoint, refers to the proportion of patients who remain inpatients at that timepoint. Similarly, for studies conducted in the ICU stay and the need for ICU admission. |
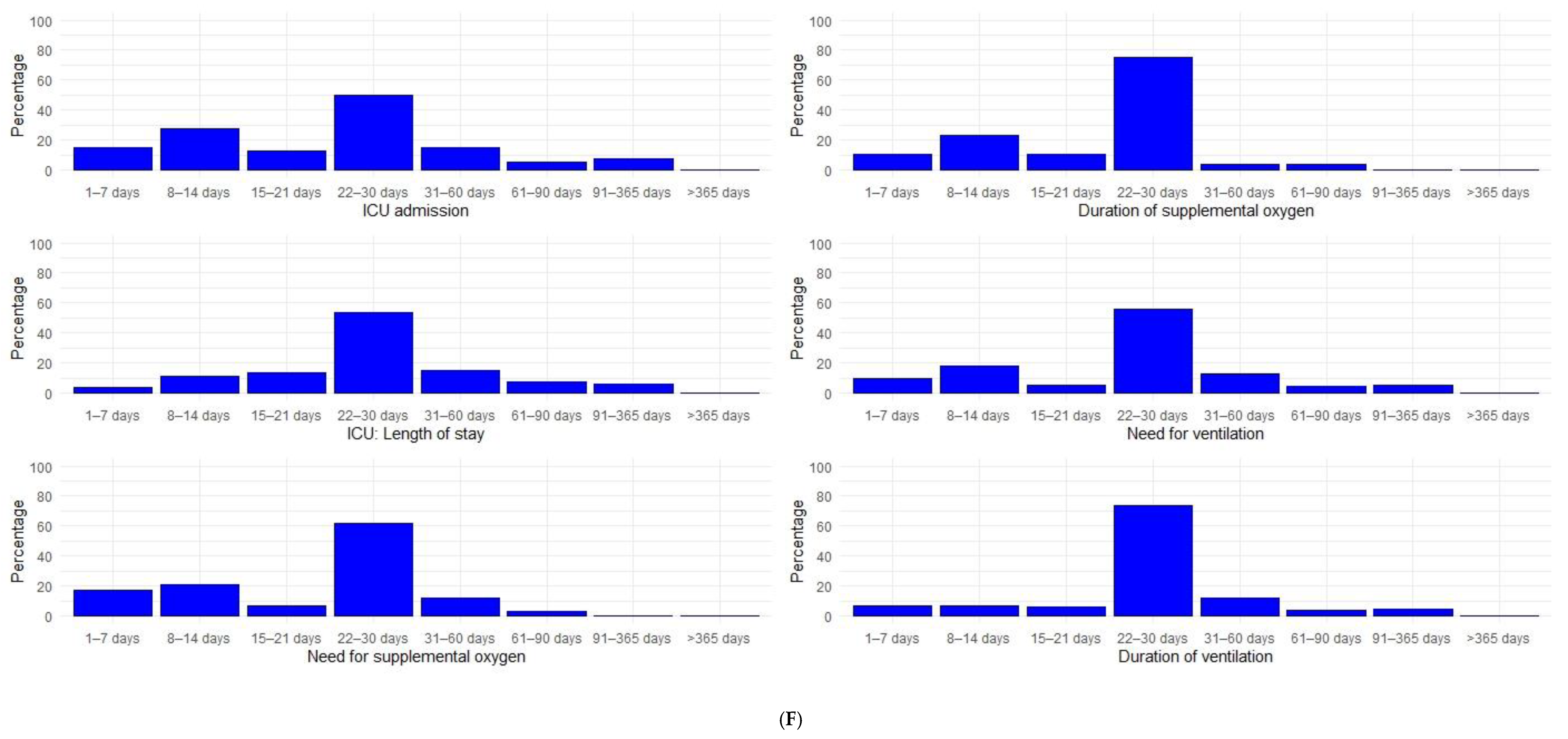
| Duration of stay in a specific level of care | This group of outcomes include length of (i) hospital stay, (ii) intensive care admission, or (iii) mechanical ventilation. The end date was often defined as the last day of stay in a specific level of care, or the last day that the stay was indicated (to account for cases when patients are medically optimized for hospital discharge but remain at hospital for social or other reasons. |
| Need for supplemental oxygen or NIV | An assessment of the need for supplemental oxygen, the required oxygen flow or modality of delivery (e.g., oxygen, continuous positive airways pressure [CPAP], bilevel positive airway pressure [BiPAP], or high flow nasal oxygen). |
| Duration of supplemental oxygen or NIV | An evaluation of the duration of supplemental oxygen needs. |
| Need for other organ support | This category included the need for (a) vasopressors and (b) need for renal replacement therapy. |
| Other outcomes | In this category we summarized outcomes that were reported in less than 10 of all eligible trials. These included changes in activities of daily living, quality of life, pharmacodynamics and pharmacokinetics, drug compliance, feasibility outcomes, use of antibiotics or other drugs, emergency room visits or use of other healthcare resources, the need for prone positioning, need for transfusion and discharge destinations. |
| Outcome Category | Phase 2 Trials (n = 178) | Later Phase Trials (n = 237) | ||
|---|---|---|---|---|
| Any Outcome | Primary Outcome | Any Outcome | Primary Outcome | |
| Mortality/survival | 115 (64.6%) | 24 (13.5%) | 153 (64.6%) | 32 (13.5%) |
| Clinical/physiological outcomes | ||||
| Treatment success or treatment failure | 70 (39.3%) | 31 (17.4%) | 103 (43.5%) | 69 (29.1%) |
| Success | 55 (30.9%) | 19 (10.7%) | 88 (37.1%) | 54 (22.8%) |
| Failure | 23 (12.9%) | 12 (6.7%) | 31 (13.1%) | 14 (5.9%) |
| Subgroup: Time to treatment success or treatment failure | 37 (20.2%) | 12 (6.7%) | 62 (26.2%) | 36 (15.2%) |
| Success | 30 (16.9%) | 9 (5.1%) | 59 (24.9%) | 33 (13.9%) |
| Failure | 8 (4.5%) | 3 (1.7%) | 11 (4.6%) | 3 (1.3%) |
| Severity scores | 76 (42.7%) | 21 (11.8%) | 93 (39.2%) | 25 (10.5%) |
| Symptoms | 43 (24.2%) | 5 (2.8%) | 60 (25.3%) | 7 (3.0%) |
| Oxygenation | 63 (35.4%) | 22 (12.4%) | 72 (30.4%) | 23 (9.7%) |
| Pulmonary function/physiology | 12 (6.7%) | 3 (1.7%) | 9 (3.8%) | 1 (0.4%) |
| Viral detection and load | 59 (33.1%) | 20 (11.2%) | 97 (40.9%) | 36 (15.2%) |
| Viral antibodies | 17 (9.6%) | 0 (0.0%) | 8 (3.4%) | 2 (0.8%) |
| Radiological outcomes | 25 (14.0%) | 3 (1.7%) | 25 (10.5%) | 9 (3.8%) |
| Inflammatory biomarkers | 69 (38.8%) | 7 (3.9%) | 66 (27.8%) | 9 (3.8%) |
| Other biomarkers | 47 (26.4%) | 4 (2.2%) | 51 (21.5%) | 2 (0.8%) |
| Pharmacokinetics/pharmacodynamics | 10 (5.6%) | 0 (0.0%) | 5 (2.1%) | 0 (0.0%) |
| Adverse events | 95 (53.4%) | 18 (10.1%) | 121 (51.1%) | 8 (3.4%) |
| Life impact | 3 (1.7%) | 1 (0.6%) | 10 (4.2%) | 0 (0.0%) |
| Resource use | ||||
| Hospital admission | 21 (11.8%) | 9 (5.1%) | 30 (12.7%) | 18 (7.6%) |
| Hospital re-admission | 6 (3.4%) | 1 (0.6 %) | 3 (1.3%) | 0 (0.0%) |
| Length of hospital stay | 70 (39.3%) | 5 (2.8%) | 103 (43.5%) | 7 (3.0%) |
| ICU admission | 35 (19.7%) | 6 (3.4%) | 38 (16.0%) | 2 (0.8%) |
| Length of ICU stay | 42 (23.6%) | 0 (0.0%) | 49 (20.7%) | 3 (1.3%) |
| Need for supplemental oxygen or NIV | 31 (17.4%) | 12 (6.7%) | 44 (18.6%) | 3 (1.3%) |
| Duration of supplemental oxygen or NIV | 40 (22.5%) | 2 (1.1%) | 39 (16.5%) | 1 (0.4%) |
| Need for invasive ventilation | 62 (34.8%) | 16 (9.0%) | 87 (36.7%) | 27 (11.4%) |
| Duration of invasive ventilation | 65 (36.5%) | 9 (5.1%) | 68 (28.7%) | 9 (3.8%) |
| Need for vasopressors | 11 (6.2%) | 0 (0.0%) | 10 (4.2%) | 0 (0.0%) |
| Need for renal replacement therapy | 6 (3.4%) | 0 (0.0%) | 7 (3.0%) | 0 (0.0%) |
| Other outcomes | 31 (17.4%) | 2 (1.1%) | 42 (17.7%) | 5 (2.1%) |
| Outcome Category | Community (n = 63) | Hospital (n = 304) | ICU (n = 27) | |||
|---|---|---|---|---|---|---|
| Any Outcome | Primary Outcome | Any Outcome | Primary Outcome | Any Outcome | Primary Outcome | |
| Mortality/survival | 19 (30.2%) | 3 (4.8%) | 216 (71.6%) | 44 (14.5%) | 24 (88.9%) | 8 (29.6%) |
| Clinical/Physiological Outcomes | ||||||
| Treatment success or treatment failure | 25 (39.7%) | 15 (23.8%) | 140 (46.2%) | 81 (26.6%) | 2 (7.4%) | 0 (0.0%) |
| Success | 16 (25.4%) | 7 (11.1%) | 121 (39.8%) | 63 (20.7%) | 1 (3.7%) | 0 (0.0%) |
| Failure | 12 (19.0%) | 8 (12.7%) | 41 (13.5%) | 17 (5.6%) | 1 (3.7%) | 0 (0.0%) |
| Subgroup: Time to treatment success or treatment failure | 12 (19.0%) | 5 (7.9%) | 83 (27.3%) | 40 (13.2%) | 0 (0.0%) | 0 (0.0%) |
| Success | 7 (11.1%) | 3 (4.8%) | 79 (26.0%) | 37 (12.2%) | 0 (0.0%) | 0 (0.0%) |
| Failure | 4 (6.3%) | 2 (3.2%) | 13 (4.3) | 3 (0.9%) | 0 (0.0%) | 0 (0.0%) |
| Severity scores | 16 (25.4%) | 5 (7.9%) | 136 (44.7%) | 40 (13.2%) | 12 (44.4%) | 1 (3.7%) |
| Symptoms | 31 (49.2%) | 4 (6.3%) | 61 (20.1%) | 7 (2.3%) | 2 (7.4%) | 0 (0.0%) |
| Oxygenation | 6 (9.5%) | 2 (3.2%) | 110 (36.2%) | 35 (11.5%) | 15 (55.6%) | 7 (25.9%) |
| Pulmonary function/physiology | 1 (1.6%) | 1 (1.6%) | 12 (3.9%) | 1 (0.3%) | 5 (18.6%) | 0 (0.0%) |
| Viral detection and load | 35 (55.6%) | 18 (28.6%) | 107 (35.2%) | 34 (11.1%) | 7 (25.9%) | 0 (0.0%) |
| Viral Antibodies | 4 (6.3%) | 0 (0.0%) | 19 (6.3%) | 2 (0.7%) | 1 (3.7%) | 0 (0.0%) |
| Radiological outcomes | 4 (6.3%) | 3 (4.8%) | 40 (13.2%) | 8 (2.6%) | 3 (11.1%) | 0 (0.0%) |
| Inflammatory biomarkers | 6 (9.5%) | 1 (1.6%) | 114 (37.5%) | 14 (4.6%) | 11 (40.7%) | 1 (3.7%) |
| Other biomarkers | 4 (6.3%) | 0 (0.0%) | 79 (26.0%) | 5 (1.6%) | 10 (37.0%) | 0 (0.0%) |
| Pharmacokinetics / Pharmacodynamics | 2 (3.2%) | 0 (0.0%) | 13 (4.3%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) |
| Adverse events | 25 (39.7%) | 3 (4.8%) | 166 (54.6%) | 21 (6.9%) | 18 (66.7%) | 2 (7.4%) |
| Life Impact | 0 (0.0%) | 0 (0.0%) | 7 (2.3%) | 0 (0.0%) | 3 (11.1%) | 0 (0.0%) |
| Resource Use | ||||||
| Hospital admission | 32 (50.8%) | 21 (33.3%) | 15 (4.9%) * | 4 (1.3%) * | 1 (3.7%) * | 0 (0.0%) * |
| Hospital re-admission | 0 (0.0%) | 0 (0.0%) | 9 (3%) | 1 (0.3%) | 0 (0.0%) | 0 (0.0%) |
| Length of hospital stay | 9 (14.3%) | 1 (1.6%) | 152 (50%) | 11 (36.2%) | 10 (37.0%) | 1 (3.7%) |
| ICU admission | 8 (12.7%) | 0 (0.0%) | 61 (20.1%) * | 8 (2.6%) * | 2 (7.4%) * | 0 (0.0%) * |
| Length of ICU stay | 5 (7.9%) | 1 (1.6%) | 70 (23.0%) | 1 (0.3%) | 14 (51.9%) | 1 (3.7%) |
| Need for supplemental oxygen or NIV | 4 (6.3%) | 0 (0.0%) | 68 (22.4%) | 13 (4.3%) | 1 (3.7%) | 0 (0.0%) |
| Duration of supplemental oxygen or NIV | 3 (4.8%) | 0 (0.0%) | 70 (23.0%) | 3 (0.9%) | 3 (11.1%) | 0 (0.0%) |
| Need for invasive ventilation | 7 (11.1%) | 2 (3.2%) | 130 (42.8%) | 34 (11.2%) | 6 (22.2%) | 4 (14.8%) |
| Duration of invasive ventilation | 5 (7.9%) | 1 (1.6%) | 106 (34.9%) | 10 (3.3%) | 19 (70.4%) | 7 (25.9%) |
| Need for vasopressors | 0 (0.0%) | 0 (0.0%) | 18 (5.9%) | 0 (0.0%) | 2 (7.4%) | 0 (0.0%) |
| Need for renal replacement therapy | 0 (0.0%) | 0 (0.0%) | 10 (3.3%) | 0 (0.0%) | 3 (11.1%) | 0 (0.0%) |
| Other outcomes | 13 (20.6%) | 3 (4.8%) | 44 (14.5%) | 5 (1.6%) | 8 (29.6%) | 0 (0.0%) |
| Patient State | Descriptor | Score |
|---|---|---|
| Uninfected | No clinical or virological evidence of infection | 0 |
| Ambulatory | No limitation of activities | 1 |
| Limitation of activities | 2 | |
| Hospitalized, Mild disease | Hospitalized, no oxygen therapy | 3 |
| Hospitalized, oxygen therapy by mask or nasal prongs | 4 | |
| Hospitalized, Severe disease | Non-invasive ventilation or high-flow oxygen | 5 |
| Intubation and mechanical ventilation | 6 | |
| Ventilation and additional organ support (vasopressors, renal replacement therapy, or ECMO) | 7 | |
| Dead | Death | 8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mathioudakis, A.G.; Fally, M.; Hashad, R.; Kouta, A.; Hadi, A.S.; Knight, S.B.; Bakerly, N.D.; Singh, D.; Williamson, P.R.; Felton, T.; et al. Outcomes Evaluated in Controlled Clinical Trials on the Management of COVID-19: A Methodological Systematic Review. Life 2020, 10, 350. https://doi.org/10.3390/life10120350
Mathioudakis AG, Fally M, Hashad R, Kouta A, Hadi AS, Knight SB, Bakerly ND, Singh D, Williamson PR, Felton T, et al. Outcomes Evaluated in Controlled Clinical Trials on the Management of COVID-19: A Methodological Systematic Review. Life. 2020; 10(12):350. https://doi.org/10.3390/life10120350
Chicago/Turabian StyleMathioudakis, Alexander G., Markus Fally, Rola Hashad, Ahmed Kouta, Ali Sina Hadi, Sean Blandin Knight, Nawar Diar Bakerly, Dave Singh, Paula R. Williamson, Tim Felton, and et al. 2020. "Outcomes Evaluated in Controlled Clinical Trials on the Management of COVID-19: A Methodological Systematic Review" Life 10, no. 12: 350. https://doi.org/10.3390/life10120350
APA StyleMathioudakis, A. G., Fally, M., Hashad, R., Kouta, A., Hadi, A. S., Knight, S. B., Bakerly, N. D., Singh, D., Williamson, P. R., Felton, T., & Vestbo, J. (2020). Outcomes Evaluated in Controlled Clinical Trials on the Management of COVID-19: A Methodological Systematic Review. Life, 10(12), 350. https://doi.org/10.3390/life10120350






