Effects of Heavy Ion Particle Irradiation on Spore Germination of Bacillus spp. from Extremely Hot and Cold Environments
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Areas
2.2. Bacterial Strains and Spore Purification
2.3. Radiation Exposition to HZE
2.4. Spore Survival
2.5. Spectroscopic Analysis
2.6. Germination Assay
3. Results
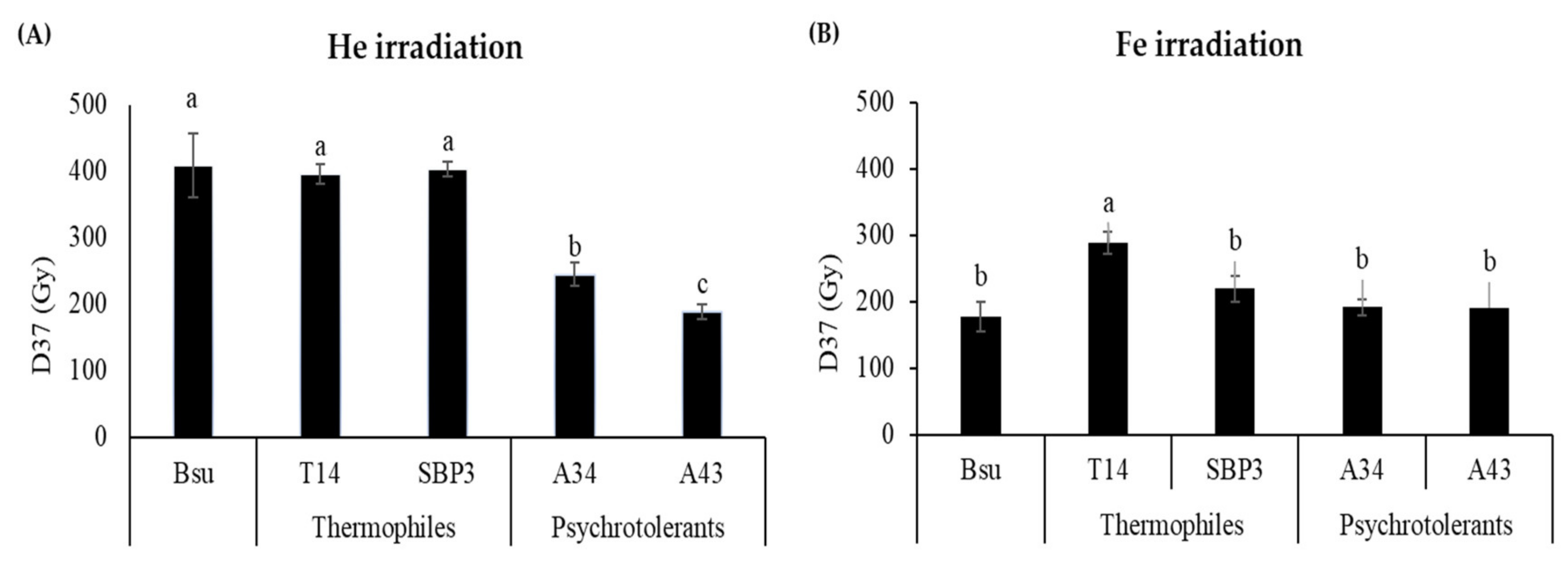
3.1. Spores Survival
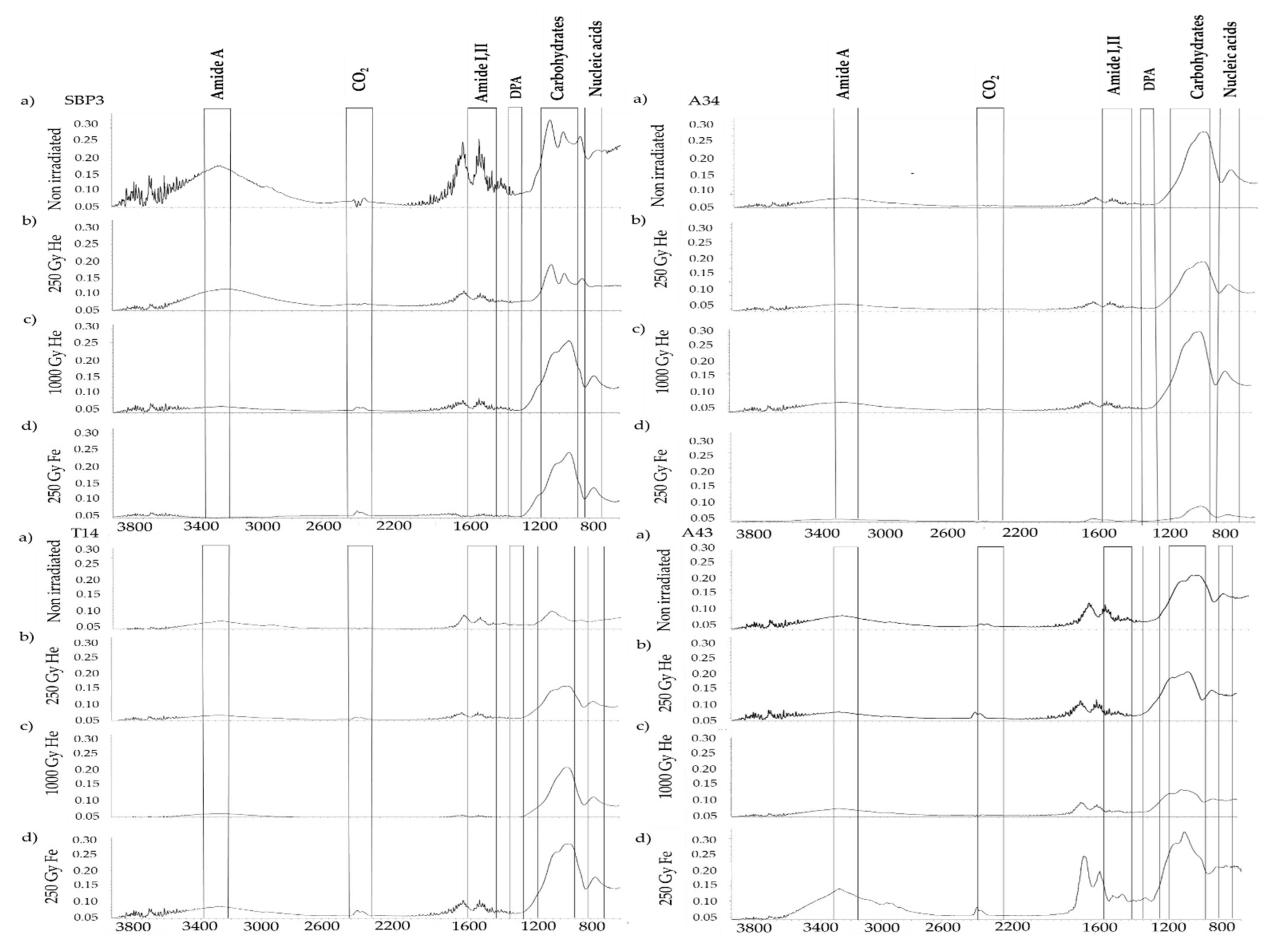
3.2. Spectroscopic Analysis
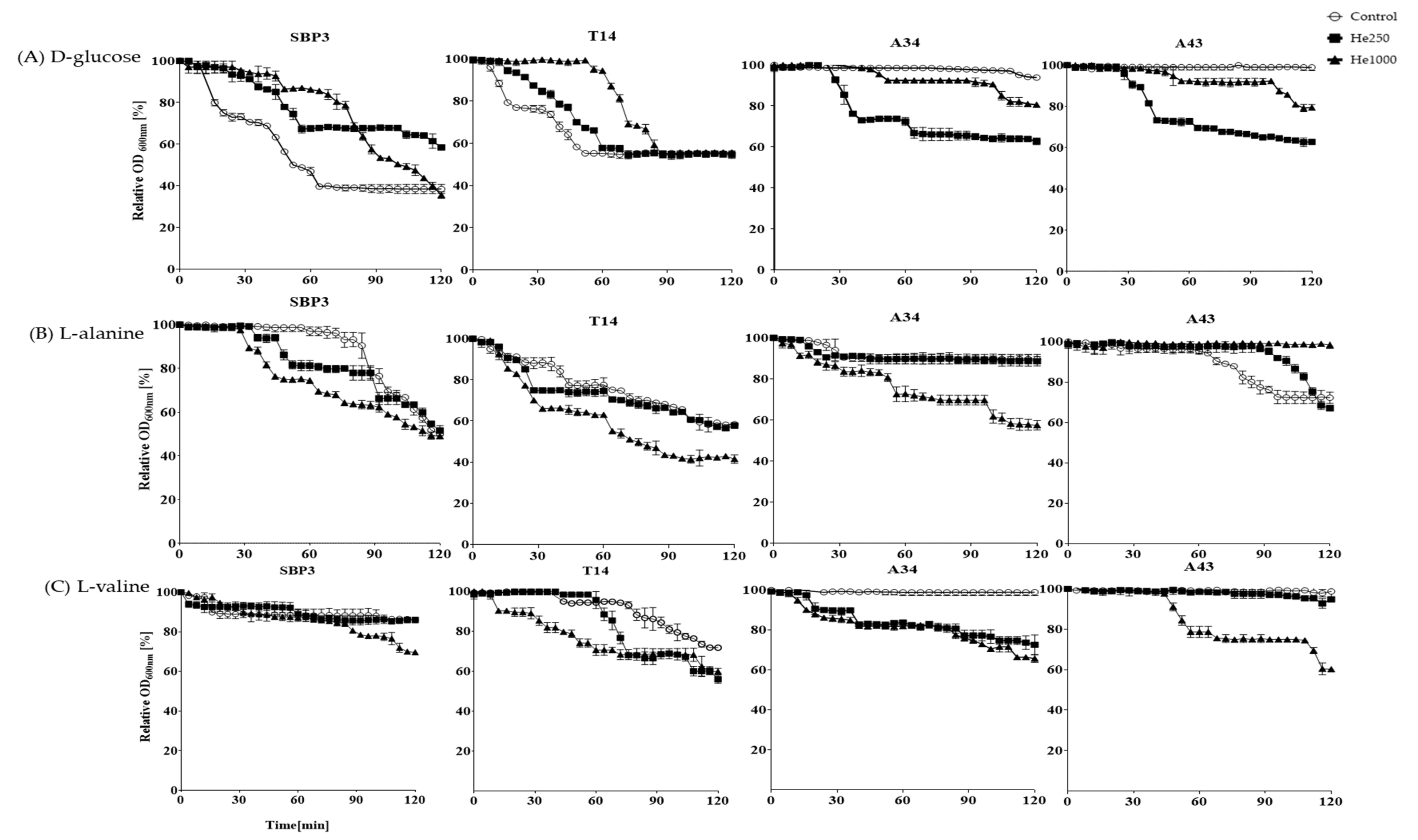
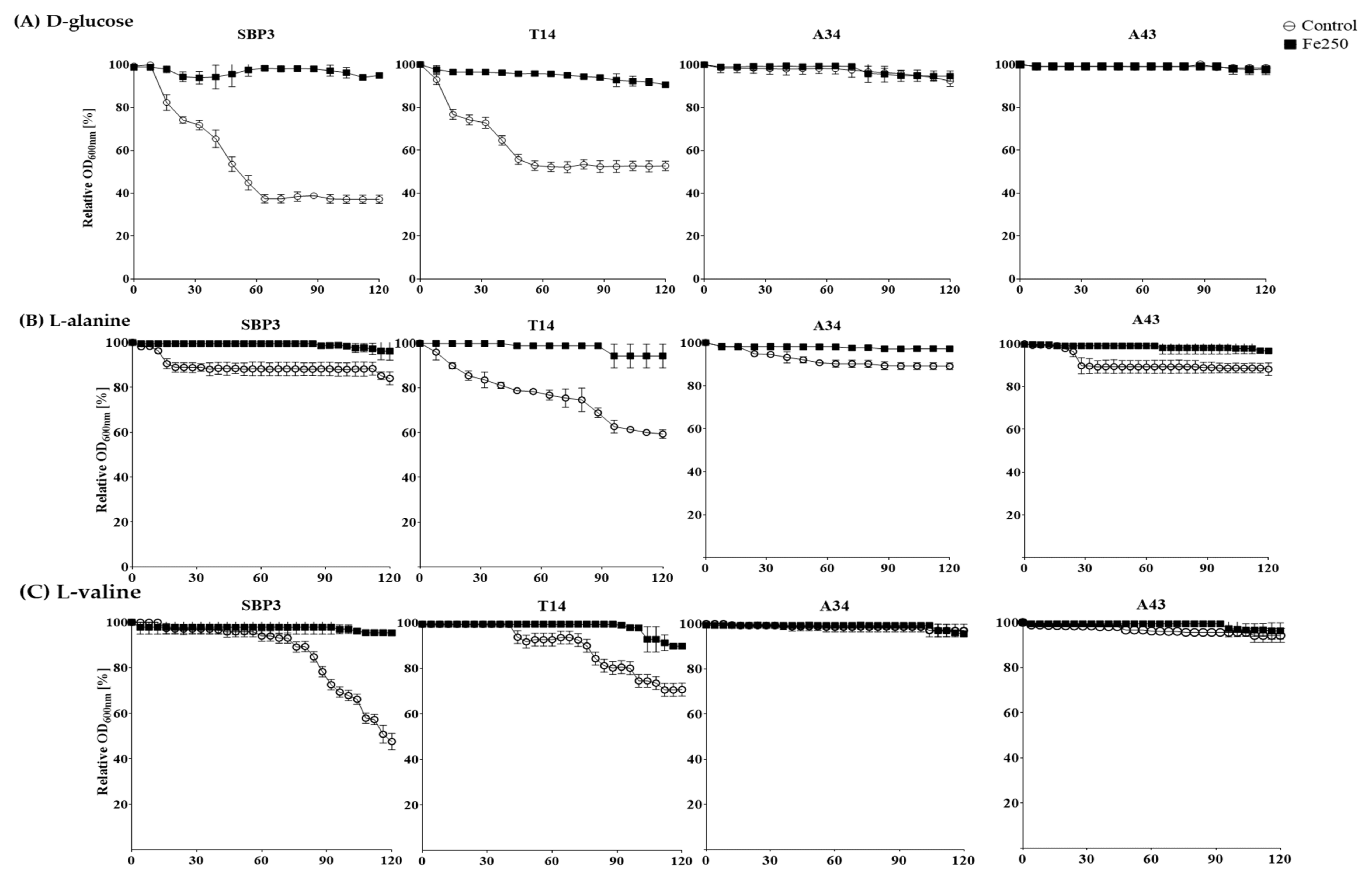
3.3. Germination Assay
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Ala | L-alanine |
| AP | Apurinic/apyrimidinic |
| CFU | Colony forming unit |
| LD90 | Lethal dose required to kill 90% of the spore population |
| FTIR | Fourier transform infrared |
| GCR | Galactic cosmic radiation |
| Glu | D-glucose |
| GRs | Germinant receptors |
| HIMAC | Heavy ion medical accelerator center |
| HZE | Heavy charge energy |
| IR | Ionizing radiation |
| LET | Linear energy transfer |
| NHEJ | Non-homologous end joining |
| NIRS | National Institute for Radiological Sciences |
| OD | Optical density |
| ROS | Radical oxygen species |
| SASP | Small acid soluble proteins |
| SHV | Shallow hydrothermal vents |
| TSA | Tryptone soy agar |
| TSA1 | Tryptone soy agar with 1% NaCl |
| Val | L-valine |
References
- Fairén, A.G.; Davila, A.F.; Lim, D.; Bramall, N.; Bonaccorsi, R.; Zavaleta, J.; Uceda, E.R.; Stoker, C.; Wierzchos, J.; Dohm, J.M.; et al. Astrobiology through the Ages of Mars: The Study of Terrestrial Analogues to Understand the Habitability of Mars. Astrobiology 2010, 10, 821–843. [Google Scholar] [CrossRef] [PubMed]
- Capece, M.C.; Clark, E.; Saleh, J.K.; Halford, D.; Heinl, N.; Hoskins, S.; Rothschild, L.J. Polyextremophiles and the constraints for terrestrial habitability. In Polyextremophiles; Seckbach, J., Oren, A., Stan-Lotter, H., Eds.; Springer: Dordrecht, The Netherlands, 2013; Volume 27, pp. 3–59. [Google Scholar]
- Harrison, J.P.; Gheeraert, N.; Tsigelnitskiy, D.; Cockell, C.S. The limits for life under multiple extremes. Trends Microbiol. 2013, 21, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Carlson, C.; Singh, N.K.; Bibra, M.; Sani, R.K.; Venkateswaran, K. Pervasiveness of UVC254-resistant Geobacillus strains in extreme environments. Appl. Microbiol. Biotechnol. 2018, 102, 1869–1887. [Google Scholar] [CrossRef] [PubMed]
- Merino, N.; Aronson, H.S.; Bojanova, D.; Feyhl-Buska, J.; Wong, M.L.; Zhang, S.; Giovannelli, D. Living at the Extremes: Extremophiles and the Limits of Life in a Planetary Context. Front. Microbiol. 2019, 10, 780. [Google Scholar] [CrossRef] [PubMed]
- Rothschild, L.J.; Mancinelli, R.L. Life in extreme environments. Nat. Cell Biol. 2001, 409, 1092–1101. [Google Scholar] [CrossRef] [PubMed]
- Cosciotti, B.; Balbi, A.; Ceccarelli, A.; Fagliarone, C.; Mattei, E.; Lauro, S.E.; Di Paolo, F.; Pettinelli, E.; Billi, D. Survivability of Anhydrobiotic Cyanobacteria in Salty Ice: Implications for the Habitability of Icy Worlds. Life 2019, 9, 86. [Google Scholar] [CrossRef]
- Rampelotto, P.H. Resistance of Microorganisms to Extreme Environmental Conditions and Its Contribution to Astrobiology. Sustainability 2010, 2, 1602–1623. [Google Scholar] [CrossRef]
- Schulze-Makuch, D.; Airo, A.; Schirmack, J. The Adaptability of Life on Earth and the Diversity of Planetary Habitats. Front. Microbiol. 2017, 8, 2011. [Google Scholar] [CrossRef]
- Horneck, G.; Walter, N.; Westall, F.; Grenfell, J.L.; Martin, W.F.; Gómez, F.; Leuko, S.; Lee, N.; Onofri, S.; Tsiganis, K.; et al. AstRoMap European Astrobiology Roadmap. Astrobiology 2016, 16, 201–243. [Google Scholar] [CrossRef]
- Cortesão, M.; Fuchs, F.M.; Commichau, F.M.; Eichenberger, P.; Schuerger, A.C.; Nicholson, W.L.; Setlow, P.; Moeller, R. Bacillus subtilis Spore Resistance to Simulated Mars Surface Conditions. Front. Microbiol. 2019, 10, 333. [Google Scholar] [CrossRef]
- Cavicchioli, R. Extremophiles and the Search for Extraterrestrial Life. Astrobiology 2002, 2, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, W.L.; Schuerger, A.C.; Race, M.S. Migrating microbes and planetary protection. Trends Microbiol. 2009, 17, 389–392. [Google Scholar] [CrossRef] [PubMed]
- Horneck, G.; Klaus, D.M.; Mancinelli, R.L. Space Microbiology. Microbiol. Mol. Biol. Rev. 2010, 74, 121–156. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, C.D.; Liang, M.-C.; Yung, Y.L.; Kirschivnk, J.L. Habitability of Enceladus: Planetary Conditions for Life. Orig. Life Evol. Biosph. 2008, 38, 355–369. [Google Scholar] [CrossRef]
- Williams, D.M.; Knacke, R.F. Looking for planetary moons in the spectra of distant Jupiters. Astrobiology 2004, 4, 400–403. [Google Scholar] [CrossRef]
- Taubner, R.-S.; Olsson-Francis, K.; Vance, S.D.; Ramkissoon, N.K.; Postberg, F.; De Vera, J.-P.; Antunes, A.; Casas, E.C.; Sekine, Y.; Noack, L.; et al. Experimental and Simulation Efforts in the Astrobiological Exploration of Exooceans. Sp. Sci. Rev. 2020, 216, 1–41. [Google Scholar] [CrossRef]
- Nicholson, W.L.; Munakata, N.; Horneck, G.; Melosh, H.J.; Setlow, P. Resistance of Bacillus Endospores to Extreme Terrestrial and Extraterrestrial Environments. Microbiol. Mol. Biol. Rev. 2000, 64, 548–572. [Google Scholar] [CrossRef] [PubMed]
- Confalonieri, F.; Sommer, S. Bacterial and archaeal resistance to ionizing radiation. J. Phys. Conf. Ser. 2011, 261, 012005. [Google Scholar] [CrossRef]
- Ranawat, P.; Rawat, S. Stress response physiology of thermophiles. Arch. Microbiol. 2017, 199, 391–414. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.C.; Nobre, M.F.; Rainey, F.A.; Silva, M.T.; Wait, R.; Burghardt, J.; Chung, A.P.; Da Costa, M.S. Deinococcus geothermalis sp. nov. and Deinococcus murrayi sp. nov., Two Extremely Radiation-Resistant and Slightly Thermophilic Species from Hot Springs. Int. J. Syst. Bacteriol. 1997, 47, 939–947. [Google Scholar] [CrossRef]
- DiRuggiero, J.; Santangelo, N.; Nackerdien, Z.; Ravel, J.; Robb, F. Repair of extensive ionizing-radiation DNA damage at 95 degrees C in the hyperthermophilic archaeon Pyrococcus furiosus. J. Bacteriol. 1997, 179, 4643–4645. [Google Scholar] [CrossRef]
- Jolivet, E.; L’Haridon, S.; Corre, E.; Forterre, P.; Prieur, D. Thermococcus gammatolerans sp. nov., a hyperthermophilic archaeon from a deep-sea hydrothermal vent that resists ionizing radiation. Int. J. Syst. Evol. Microbiol. 2003, 53, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Jolivet, E.; Corre, E.; L’Haridon, S.; Forterre, P.; Prieur, D. Thermococcus marinus sp. nov. and Thermococcus radiotolerans sp. nov., two hyperthermophilic archaea from deep-sea hydrothermal vents that resist ionizing radiation. Extremophiles 2004, 8, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Setlow, P. Spores of Bacillus subtilis: Their resistance to and killing by radiation, heat and chemicals. J. Appl. Microbiol. 2006, 101, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Caccamo, D.; Gugliandolo, C.; Stackebrandt, E.; Maugeri, T.L. Bacillus vulcani sp. nov., a novel thermophilic species isolated from a shallow marine hydrothermal vent. Int. J. Syst. Evol. Microbiol. 2000, 50, 2009–2012. [Google Scholar] [CrossRef] [PubMed]
- Gugliandolo, C.; Lentini, V.; Spanò, A.; Maugeri, T. New bacilli from shallow hydrothermal vents of Panarea Island (Italy) and their biotechnological potential. J. Appl. Microbiol. 2012, 112, 1102–1112. [Google Scholar] [CrossRef]
- Spanò, A.; Gugliandolo, C.; Lentini, V.; Maugeri, T.L.; Anzelmo, G.; Poli, A.; Nicolaus, B. A Novel EPS-Producing Strain of Bacillus licheniformis Isolated from a Shallow Vent Off Panarea Island (Italy). Curr. Microbiol. 2013, 67, 21–29. [Google Scholar] [CrossRef]
- Spanò, A.; Laganà, P.; Visalli, G.; Maugeri, T.L.; Gugliandolo, C. In Vitro Antibiofilm Activity of an Exopolysaccharide from the Marine Thermophilic Bacillus licheniformis T14. Curr. Microbiol. 2016, 72, 518–528. [Google Scholar] [CrossRef]
- Zammuto, V.; Laue, M.; Fiebrandt, M.; Stapelmann, K.; Ulrich, N.J.; Maugeri, T.L.; Pukall, R.; Gugliandolo, C.; Moeller, R. Comparing Spore Resistance of Bacillus Strains Isolated from Hydrothermal Vents and Spacecraft Assembly Facilities to Environmental Stressors and Decontamination Treatments. Astrobiology 2018, 18, 1425–1434. [Google Scholar] [CrossRef]
- Zammuto, V.; Conte, A.; Michaud, L.; Papale, M.; Guglielmin, M.; Romano, I.; Vizzini, S.; Lo Giudice, A.; Gugliandolo, C. Extremophiles from cold and hot environments resistant to ultraviolet radiation (UV-C) simulating space environment. J. Biol. Res. 2016, 86, 17. [Google Scholar]
- Zammuto, V.; Gugliandolo, C. Resistance to high temperatures of spores from bacilli of shallow hydrothermal vent origins. AAPP Atti della Accademia Peloritana dei Pericolant Classe di Scienze Fisiche, Matematiche e Naturali 2019, 97, 28. [Google Scholar]
- Setlow, P. Observations on research with spores of Bacillales and Clostridiales species. J. Appl. Microbiol. 2018, 126, 348–358. [Google Scholar] [CrossRef]
- Reineke, K.; Ellinger, N.; Berger, D.; Baier, D.; Mathys, A.; Setlow, P.; Knorr, D. Structural analysis of high pressure treated Bacillus subtilis spores. Innov. Food Sci. Emerg. Technol. 2013, 17, 43–53. [Google Scholar] [CrossRef]
- Nagler, K.; Julius, C.; Moeller, R. Germination of Spores of Astrobiologically RelevantBacillusSpecies in High-Salinity Environments. Astrobiology 2016, 16, 500–512. [Google Scholar] [CrossRef]
- He, L.; Chen, Z.; Wang, S.; Wu, M.; Setlow, P.; Li, Y.-Q. Germination, Outgrowth, and Vegetative-Growth Kinetics of Dry-Heat-Treated Individual Spores ofBacillusSpecies. Appl. Environ. Microbiol. 2018, 84, e02618-17. [Google Scholar] [CrossRef]
- Atluri, S.; Ragkousi, K.; Cortezzo, D.E.; Setlow, P. Cooperativity between Different Nutrient Receptors in Germination of Spores of Bacillus subtilis and Reduction of This Cooperativity by Alterations in the GerB Receptor. J. Bacteriol. 2006, 188, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Webb, K.M.; DiRuggiero, J. Role of Mn2+ and Compatible Solutes in the Radiation Resistance of Thermophilic Bacteria and Archaea. Archaea 2012, 2012, 1–11. [Google Scholar] [CrossRef]
- Edwards, H.; Moeller, R.; Jorge-Villar, S.E.; Horneck, G.; Stackebrandt, E. Raman spectroscopic study of the photoprotection of extremophilic microbes against ultraviolet radiation. Int. J. Astrobiol. 2006, 5, 313–318. [Google Scholar] [CrossRef]
- Dartnell, L. Ionizing Radiation and Life. Astrobiology 2011, 11, 551–582. [Google Scholar] [CrossRef]
- Ferrari, F.; Szuszkiewicz, E. Cosmic Rays: A Review for Astrobiologists. Astrobiology 2009, 9, 413–436. [Google Scholar] [CrossRef]
- Durante, M.; Cucinotta, F.A. Heavy ion carcinogenesis and human space exploration. Nat. Rev. Cancer 2008, 8, 465–472. [Google Scholar] [CrossRef]
- De Vera, J.-P.; Alawi, M.; Backhaus, T.; Baqué, M.; Billi, D.; Böttger, U.; Berger, T.; Bohmeier, M.; Cockell, C.; DeMets, R.; et al. Limits of Life and the Habitability of Mars: The ESA Space Experiment BIOMEX on the ISS. Astrobiology 2019, 19, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, A.; Ikeda, H.; Yoshida, Y. Role of High-Linear Energy Transfer Radiobiology in Space Radiation Exposure Risks. Int. J. Part. Ther. 2018, 5, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Moeller, R.; Raguse, M.; Leuko, S.; Berger, T.; Hellweg, C.E.; Fujimori, A.; Okayasu, R.; Horneck, G.; STARLIFE Research Group. STARLIFE—An International Campaign to Study the Role of Galactic Cosmic Radiation in Astrobiological Model Systems. Astrobiology 2017, 17, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Moeller, R.; Setlow, P.; Horneck, G.; Berger, T.; Reitz, G.; Rettberg, P.; Doherty, A.J.; Okayasu, R.; Nicholson, W.L. Roles of the Major, Small, Acid-Soluble Spore Proteins and Spore-Specific and Universal DNA Repair Mechanisms in Resistance of Bacillus subtilis Spores to Ionizing Radiation from X Rays and High-Energy Charged-Particle Bombardment. J. Bacteriol. 2007, 190, 1134–1140. [Google Scholar] [CrossRef]
- Moeller, R.; Reitz, G.; Berger, T.; Okayasu, R.; Nicholson, W.L.; Horneck, G. Astrobiological Aspects of the Mutagenesis of Cosmic Radiation on Bacterial Spores. Astrobiology 2010, 10, 509–521. [Google Scholar] [CrossRef]
- Moeller, R.; Schuerger, A.C.; Reitz, G.; Nicholson, W.L. Protective Role of Spore Structural Components in Determining Bacillus subtilis Spore Resistance to Simulated Mars Surface Conditions. Appl. Environ. Microbiol. 2012, 78, 8849–8853. [Google Scholar] [CrossRef]
- Papale, M.; Conte, A.; Mikkonen, A.; Michaud, L.; La Ferla, R.; Azzaro, M.; Caruso, G.; Paranhos, R.; Anderson, S.C.; Maimone, G.; et al. Prokaryotic assemblages within permafrost active layer at Edmonson Point (Northern Victoria Land, Antarctica). Soil Biol. Biochem. 2018, 123, 165–179. [Google Scholar] [CrossRef]
- Nagler, K.; Setlow, P.; Li, Y.-Q.; Moeller, R. High Salinity Alters the Germination Behavior of Bacillus subtilis Spores with Nutrient and Nonnutrient Germinants. Appl. Environ. Microbiol. 2013, 80, 1314–1321. [Google Scholar] [CrossRef]
- Okayasu, R.; Okada, M.; Okabe, A.; Noguchi, M.; Takakura, K.; Takahashi, S. Repair of DNA Damage Induced by Accelerated Heavy Ions in Mammalian Cells Proficient and Deficient in the Non-homologous End-Joining Pathway. Radiat. Res. 2006, 165, 59–67. [Google Scholar] [CrossRef]
- Orsini, F.; Ami, D.; Villa, A.M.; Sala, G.; Bellotti, M.G.; Doglia, S.M. FT-IR microspectroscopy for microbiological studies. J. Microbiol. Methods 2000, 42, 17–27. [Google Scholar] [CrossRef]
- Caccamo, M.T.; Gugliandolo, C.; Zammuto, V.; Magazù, S. Thermal properties of an exopolysaccharide produced by a marine thermotolerant Bacillus licheniformis by ATR-FTIR spectroscopy. Int. J. Biol. Macromol. 2019, 145, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Vongsvivut, J.; Heraud, P.; Zhang, W.; Kralovec, J.A.; McNaughton, D.; Barrow, C.J. Rapid Determination of Protein Contents in Microencapsulated Fish Oil Supplements by ATR-FTIR Spectroscopy and Partial Least Square Regression (PLSR) Analysis. Food Bioprocess Technol. 2013, 7, 265–277. [Google Scholar] [CrossRef]
- Guillén, M.D.; Cabo, N. Characterization of edible oils and lard by Fourier transform infrared spectroscopy. Relationships between composition and frequency of concrete bands in the fingerprint region. J. Am. Oil Chem. Soc. 1997, 74, 1281–1286. [Google Scholar] [CrossRef]
- Filip, Z.; Herrmann, S.; Kubát, J. FT-IR spectroscopic characteristics of differently cultivated Bacillus subtilis. Microbiol. Res. 2004, 159, 257–262. [Google Scholar] [CrossRef]
- Böcker, U.; Kristoffersen, K.A.; Lindberg, D.; Afseth, N.K. Fourier-transform infrared spectroscopy for characterization of protein chain reductions in enzymatic reactions. Analyst 2017, 142, 2812–2818. [Google Scholar] [CrossRef]
- Serrano, V.; Liu, W.-G.; Franzen, S. An Infrared Spectroscopic Study of the Conformational Transition of Elastin-Like Polypeptides. Biophys. J. 2007, 93, 2429–2435. [Google Scholar] [CrossRef]
- Perkins, D.L.; Lovell, C.R.; Bronk, B.V.; Setlow, B.; Setlow, P.; Myrick, M.L. Fourier transform infrared reflectance micro-spectroscopy study of Bacillus subtilis engineered without dipicolinic acid: The contribution of calcium dipicolinate to the mid-infrared absorbance of Bacillus subtilis endospores. Appl. Spectrosc. 2005, 59, 893–896. [Google Scholar] [CrossRef]
- Bizani, D.; Motta, A.S.; Morrissy, J.A.C.; Terra, R.M.S.; Souto, A.A.; Brandelli, A. Antibacterial activity of cerein 8A, a bacteriocin-like peptide produced by Bacillus cereus. Int. Microbiol. 2005, 8, 125–131. [Google Scholar]
- Rohman, A.; Man, Y.C. Fourier transform infrared (FTIR) spectroscopy for analysis of extra virgin olive oil adulterated with palm oil. Food Res. Int. 2010, 43, 886–892. [Google Scholar] [CrossRef]
- Zelig, U.; Mordechai, S.; Shubinsky, G.; Sahu, R.K.; Huleihel, M.; Leibovitz, E.; Nathan, I.; Kapelushnik, J. Pre-screening and follow-up of childhood acute leukemia using biochemical infrared analysis of peripheral blood mononuclear cells. Biochim. Biophys. Acta (BBA) Gen. Subj. 2011, 1810, 827–835. [Google Scholar] [CrossRef]
- Johnson, P.V.; Hodyss, R.; Chernow, V.F.; Lipscomb, D.M.; Goguen, J.D. Ultraviolet photolysis of amino acids on the surface of icy Solar System bodies. Icarus 2012, 221, 800–805. [Google Scholar] [CrossRef]
- McKenna-Lawlor, S.; Goncalves, P.; Keating, A.; Reitz, G.; Matthiä, D. Overview of energetic particle hazards during prospective manned missions to Mars. Planet. Sp. Sci. 2012, 63, 123–132. [Google Scholar] [CrossRef]
- Tsou, P.; Brownlee, D.E.; McKay, C.P.; Anbar, A.D.; Yano, H.; Altwegg, K.; Beegle, L.W.; Dissly, R.; Strange, N.J.; Kanik, I. LIFE: Life Investigation For EnceladusA Sample Return Mission Concept in Search for Evidence of Life. Astrobiology 2012, 12, 730–742. [Google Scholar] [CrossRef]
- Roth, L.; Saur, J.; Retherford, K.D.; Strobel, D.; Feldman, P.D.; McGrath, M.A.; Nimmo, F. Transient Water Vapor at Europa’s South Pole. Science 2013, 343, 171–174. [Google Scholar] [CrossRef]
- Grießmeier, J.-M.; Zarka, P.; Spreeuw, H. Predicting low-frequency radio fluxes of known extrasolar planets. Astron. Astrophys. 2007, 475, 359–368. [Google Scholar] [CrossRef]
- Medvedev, M.V.; Melott, A.L. Do Extragalactic Cosmic Rays Induce Cycles in Fossil Diversity? Astrophys. J. 2007, 664, 879–889. [Google Scholar] [CrossRef]
- Kiefer, J. (Ed.) Elements of photo-and radiation chemistry. In Biological Radiation Effects; Springer: Berlin, Germany, 1990; pp. 88–103. [Google Scholar]
- Horneck, G. Space Radiation Biology. In Biology in Space and Life on Earth: Effects of Spaceflight on Biological Systems; Brinckmann, E., von Friedeburg, C., Eds.; Wiley-VCH: Berlin, Germany, 2007; pp. 243–273. [Google Scholar]
- Dianov, G.L.; O’Neill, P.; Goodhead, D.T. Securing genome stability by orchestrating DNA repair: Removal of radiation-induced clustered lesions in DNA. BioEssays 2001, 23, 745–749. [Google Scholar] [CrossRef]
- Nicastro, A.J.; Vreeland, R.H.; Rosenzweig, W.D. Limits imposed by ionizing radiation on the long-term survival of trapped bacterial spores: Beta radiation. Int. J. Radiat. Biol. 2002, 78, 891–901. [Google Scholar] [CrossRef]
- Setlow, B.; Cowan, A.E.; Setlow, P. Germination of spores of Bacillus subtilis with dodecylamine. J. Appl. Microbiol. 2003, 95, 637–648. [Google Scholar] [CrossRef]
- Zhang, P.; Liang, J.; Yi, X.; Setlow, P.; Li, Y.-Q. Monitoring of Commitment, Blocking, and Continuation of Nutrient Germination of Individual Bacillus subtilis Spores. J. Bacteriol. 2014, 196, 2443–2454. [Google Scholar] [CrossRef]
- McKay, C.P.; Anbar, A.D.; Porco, C.; Tsou, P. Follow the Plume: The Habitability of Enceladus. Astrobiology 2014, 14, 352–355. [Google Scholar] [CrossRef]
- Driks, A. Maximum shields: The assembly and function of the bacterial spore coat. Trends Microbiol. 2002, 10, 251–254. [Google Scholar] [CrossRef]
- Hullo, M.-F.; Moszer, I.; Danchin, A.; Martin-Verstraete, I. CotA of Bacillus subtilis is a Copper-Dependent Laccase. J. Bacteriol. 2001, 183, 5426–5430. [Google Scholar] [CrossRef]
- Moeller, R.; Setlow, P.; Reitz, G.; Nicholson, W.L. Roles of Small, Acid-Soluble Spore Proteins and Core Water Content in Survival of Bacillus subtilis Spores Exposed to Environmental Solar UV Radiation. Appl. Environ. Microbiol. 2009, 75, 5202–5208. [Google Scholar] [CrossRef]
- Paidhungat, M.; Setlow, P. Role of Ger Proteins in Nutrient and Nonnutrient Triggering of Spore Germination in Bacillus subtilis. J. Bacteriol. 2000, 182, 2513–2519. [Google Scholar] [CrossRef]
- Slieman, T.A.; Nicholson, W.L. Role of Dipicolinic Acid in Survival ofBacillus subtilis Spores Exposed to Artificial and Solar UV Radiation. Appl. Environ. Microbiol. 2001, 67, 1274–1279. [Google Scholar] [CrossRef]
- Setlow, P. I will survive: DNA protection in bacterial spores. Trends Microbiol. 2007, 15, 172–180. [Google Scholar] [CrossRef]
- Moeller, R.; Reitz, G.; Li, Z.; Klein, S.; Nicholson, W.L. Multifactorial Resistance ofBacillus subtilisSpores to High-Energy Proton Radiation: Role of Spore Structural Components and the Homologous Recombination and Non-Homologous End Joining DNA Repair Pathways. Astrobiology 2012, 12, 1069–1077. [Google Scholar] [CrossRef] [PubMed]
- Link, L.; Sawyer, J.; Venkateswaran, K.; Nicholson, W. Extreme Spore UV Resistance of Bacillus pumilus Isolates Obtained from an Ultraclean Spacecraft Assembly Facility. Microb. Ecol. 2003, 47, 159–163. [Google Scholar] [CrossRef]
- Redmond, C.; Baillie, L.W.J.; Hibbs, S.; Moir, A.J.G.; Moir, A. Identification of proteins in the exosporium of Bacillus anthracis. Microbiology 2004, 150, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Alimova, A.; Katz, A.; Gottlieb, P.; Alfano, R.R. Proteins and dipicolinic acid released during heat shock activation of Bacillus subtilis spores probed by optical spectroscopy. Appl. Opt. 2006, 45, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Raguse, M.; Fiebrandt, M.; Denis, B.; Stapelmann, K.; Eichenberger, P.; Driks, A.; Eaton, P.; Awakowicz, P.; Möller, R. Understanding of the importance of the spore coat structure and pigmentation in theBacillus subtilisspore resistance to low-pressure plasma sterilization. J. Phys. D Appl. Phys. 2016, 49, 285401. [Google Scholar] [CrossRef]
- Griffiths, K.; Setlow, P. Effects of modification of membrane lipid composition onBacillus subtilissporulation and spore properties. J. Appl. Microbiol. 2009, 106, 2064–2078. [Google Scholar] [CrossRef]
- Nicolaus, B.; Panico, A.; Manca, M.C.; Lama, L.; Gambacorta, A.; Maugeri, T.; Gugliandolo, C.; Caccamo, D. A Thermophilic Bacillus Isolated From an Eolian Shallow Hydrothermal Vent Able to Produce Exopolysaccharides. Syst. Appl. Microbiol. 2000, 23, 426–432. [Google Scholar] [CrossRef]
- Moeller, R.; Raguse, M.; Reitz, G.; Okayasu, R.; Li, Z.; Klein, S.; Setlow, P.; Nicholson, W.L. Resistance of Bacillus subtilis Spore DNA to Lethal Ionizing Radiation Damage Relies Primarily on Spore Core Components and DNA Repair, with Minor Effects of Oxygen Radical Detoxification. Appl. Environ. Microbiol. 2013, 80, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Borch-Pedersen, K.; Lindbäck, T.; Madslien, E.H.; Kidd, S.W.; O’Sullivan, K.; Granum, P.E.; Aspholm, M. The Cooperative and Interdependent Roles of GerA, GerK, and Ynd in Germination of Bacillus licheniformis Spores. Appl. Environ. Microbiol. 2016, 82, 4279–4287. [Google Scholar] [CrossRef]
- Løvdal, I.S.; From, C.; Madslien, E.H.; Romundset, K.C.S.; Klufterud, E.; Rosnes, J.T.; Granum, P.E. Role of the gerA operon in L-alanine germination of Bacillus licheniformis spores. BMC Microbiol. 2012, 12, 34. [Google Scholar] [CrossRef]
- Feavers, I.M.; Foulkes, J.; Setlow, B.; Sun, D.; Nicholson, W.; Setlow, P.; Moir, A. The regulation of transcription of the gerA spore germination operon of Bacillus subtilis. Mol. Microbiol. 1990, 4, 275–282. [Google Scholar] [CrossRef]

| Strain | Source | Optimal Temperature for Growth (°C) | Spore Resistance to UV-C (LD90) | Reference |
|---|---|---|---|---|
| Bacillus horneckiae SBP3 | Black point, shallow hydrothermal vent (Panarea Island, Italy) | 45 | 139 ± 15 | [30] |
| Bacillus licheniformis T14 | Bottaro, shallow hydrothermal vent (Panarea Island, Italy) | 50 | 127 ± 13 | [31] |
| Bacillus sp. A34 | Antarctic soil (Edmonson point) | 15 | 127 ± 10 | [31] |
| Bacillus sp. A43 | Antarctic soil (Edmonson point) | 15 | 110 ± 10 | [31] |
| Ion | Energy (MeV/n) | LET (keV/µm) | Intensity (Particles/s) |
|---|---|---|---|
| Helium (He) | 150 | 2.2 | 2.0 × 109 |
| Iron (Fe) | 500 | 200 | 2.5 × 108 |
| Wavenumber Values (cm-1) | Band Assignment | References |
|---|---|---|
| 3300–3200 | H-bond and OH group of alcohol, phenols, and organic acid including nucleic acids and proteins amide A | [55] |
| 3100–3000 | n(C–H) of cis C=H bonds | [56] |
| ≈2925 | (C–H) from methylene (–CH) group of lipids | [56] |
| 1700–1750 | Protein and esters of muramic acid and ester fatty acid group | [56] |
| 1660–1628 | Amide I peptidic conformation | [57] |
| 1548 | Amide II peptidic conformation | [58] |
| ≈1380 | CH2 and CH3 bending from lipids, DPA | [59] |
| ≈1310 | amide III | [60] |
| ≈1066 | (R–O–P–O–R) from ring vibrations of carbohydrates | [56] |
| ≈966 | (CH) of conjugated trans, trans isomers | [61] |
| ≈780 | Nucleic acids, sugar-phosphate vibration | [62] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zammuto, V.; Rizzo, M.G.; De Plano, L.M.; Franco, D.; Guglielmino, S.; Caccamo, M.T.; Magazù, S.; Fujimori, A.; Giudice, A.L.; Guglielmin, M.; et al. Effects of Heavy Ion Particle Irradiation on Spore Germination of Bacillus spp. from Extremely Hot and Cold Environments. Life 2020, 10, 264. https://doi.org/10.3390/life10110264
Zammuto V, Rizzo MG, De Plano LM, Franco D, Guglielmino S, Caccamo MT, Magazù S, Fujimori A, Giudice AL, Guglielmin M, et al. Effects of Heavy Ion Particle Irradiation on Spore Germination of Bacillus spp. from Extremely Hot and Cold Environments. Life. 2020; 10(11):264. https://doi.org/10.3390/life10110264
Chicago/Turabian StyleZammuto, Vincenzo, Maria G. Rizzo, Laura M. De Plano, Domenico Franco, Salvatore Guglielmino, Maria T. Caccamo, Salvatore Magazù, Akira Fujimori, Angelina Lo Giudice, Mauro Guglielmin, and et al. 2020. "Effects of Heavy Ion Particle Irradiation on Spore Germination of Bacillus spp. from Extremely Hot and Cold Environments" Life 10, no. 11: 264. https://doi.org/10.3390/life10110264
APA StyleZammuto, V., Rizzo, M. G., De Plano, L. M., Franco, D., Guglielmino, S., Caccamo, M. T., Magazù, S., Fujimori, A., Giudice, A. L., Guglielmin, M., McAlpin, K. R., Moeller, R., & Gugliandolo, C. (2020). Effects of Heavy Ion Particle Irradiation on Spore Germination of Bacillus spp. from Extremely Hot and Cold Environments. Life, 10(11), 264. https://doi.org/10.3390/life10110264
















