Kenaf (Hibiscus cannabinus L.) Leaves and Seed as a Potential Source of the Bioactive Compounds: Effects of Various Extraction Solvents on Biological Properties
Abstract
:1. Introduction
2. Method
2.1. Chemicals
2.2. Plant Material and Extracts Preparation
2.3. Gas Chromatography-Mass Spectroscopy (GC-MS) Analysis of Kenaf (Leaves and Seed) Extracts
2.4. Quantitative Analysis
2.4.1. Total Phenolic Content (TPC)
2.4.2. Total Flavonoid Content (TFC)
2.5. Antioxidant Activity
DPPH Free Radical and Hydrogen Peroxide (H2O2) Scavenging Activity
2.6. Cell Culture, Cytotoxicity, and Anti-Lung Cancer Assay
2.7. Antibacterial Activity
3. Statistical Analysis
4. Results and Discussions
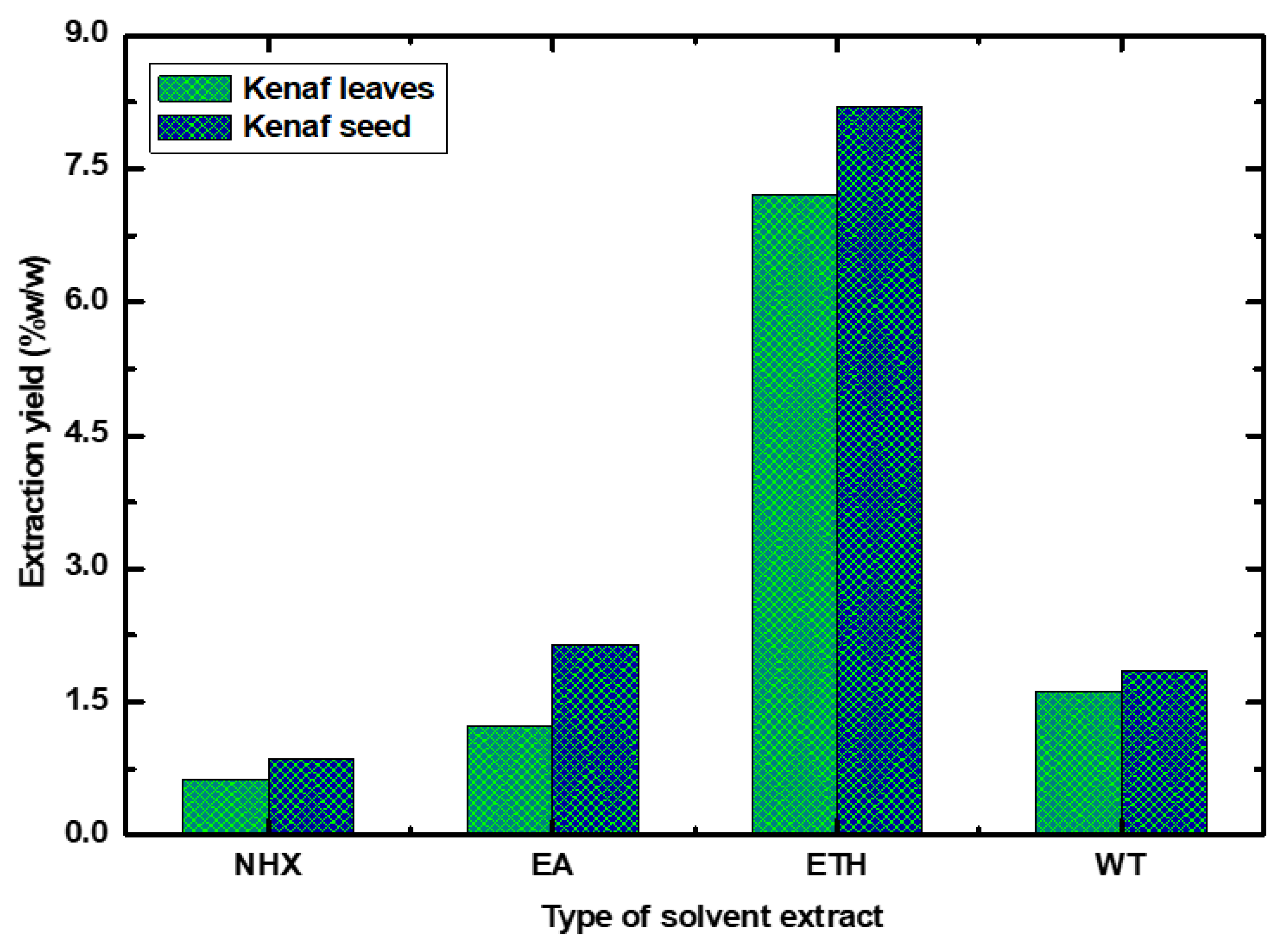
4.1. Extraction Yields
4.2. GC-MS Analysis of Ethanol Extract for Kenaf (Leaves and Seed)
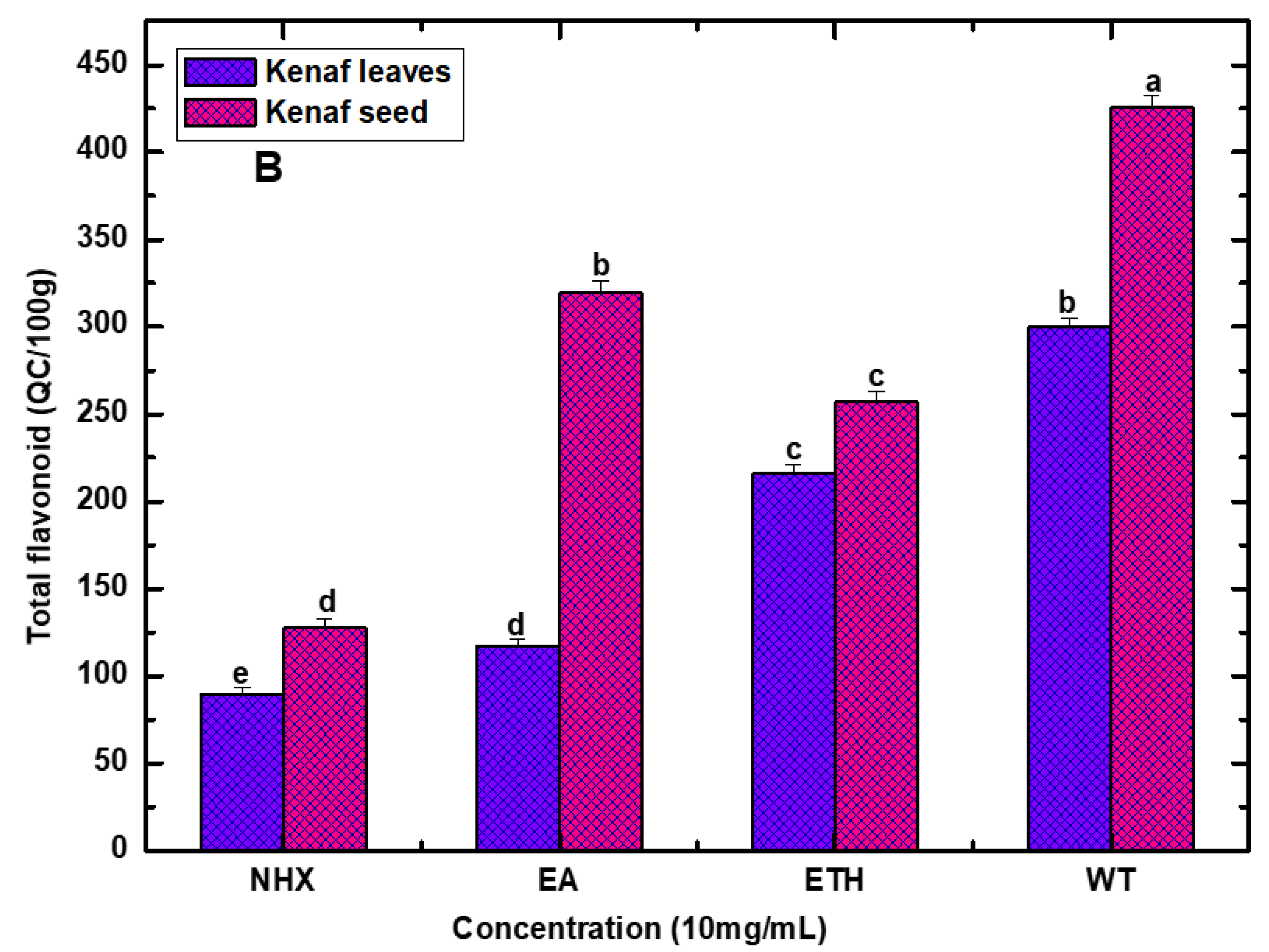
4.3. Total Phenol and Flavonoid Content
4.4. Antioxidant Activity
4.5. Cytotoxicity and Anti-Lung Cancer Activity
4.6. Antibacterial Activity
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Young, I.S.; Woodside, J.V. Antioxidants in health and disease. J. Clin. Pathol. 2001, 54, 176–186. [Google Scholar] [CrossRef] [Green Version]
- Harman, D. Aging: Phenomena and theories. Ann. N. Y. Acad. Sci. 1998, 854, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Adnan, M.; Mohammad, K.I.; Manik, M.E.H. Anticancer agents in combination with statins. J. Bioequiv. Availab. 2017, 9, 463–466. [Google Scholar] [CrossRef]
- Anagnostopoulou, M.A.; Kefalas, P.; Papageorgiou, V.P.; Assimopoulou, A.N.; Boskou, D. Radical scavenging activity of various extracts and fractions of sweet orange peel (Citrus sinensis). Food Chem. 2006, 94, 19–25. [Google Scholar] [CrossRef]
- Adnan, M.; Azad, M.O.K.; Ju, H.S.; Son, J.M.; Park, C.H.; Shin, M.H.; Alle, M.; Cho, D.H. Development of biopolymer-mediated nanocomposites using hot-melt extrusion to enhance the bio-accessibility and antioxidant capacity of kenaf seed flour. Appl. Nanosci. 2019, 2019, 1–13. [Google Scholar] [CrossRef]
- Bergman, M.; Varshavsky, L.; Gottlieb, H.E.; Grossman, S. The antioxidant activity of aqueous spinach extract: Chemical identification of active fractions. Phytochemistry 2001, 58, 143–152. [Google Scholar] [CrossRef]
- Sahreen, S.; Khan, M.R.; Khan, R.A. Evaluation of antioxidant activities of various solvent extracts of Carissa opaca fruits. Food Chem. 2010, 122, 1205–1211. [Google Scholar] [CrossRef]
- Zhao, S.; Li, X.; Cho, D.H.; Arasu, M.V.; Al-Dhabi, N.A.; Park, S.U. Accumulation of kaempferitrin and expression of phenyl-propanoid biosynthetic genes in kenaf (Hibiscus cannabinus). Molecules 2014, 19, 16987–16997. [Google Scholar] [CrossRef] [Green Version]
- Kubmarawa, D.; Andenyang, I.F.H.; Magomya, A.M. Proximate composition and amino acid profile of two non-conventional leafy vegetables (Hibiscus cannabinus and Haematostaphis barteri). Afr. J. Food Sci. 2009, 3, 233–236. [Google Scholar]
- Monti, A.; Alexopoulou, E. Kenaf: A Multi-Purpose Crop for Several Industrial Applications; Springer: Berlin/Heidelberg, Germany, 2013; ISBN 1447150678. [Google Scholar]
- Jin, C.W.; Ghimeray, A.K.; Wang, L.; Xu, M.L.; Piao, J.P.; Cho, D.H. Far infrared assisted kenaf leaf tea preparation and its effect on phenolic compounds, antioxidant and ACE inhibitory activity. J. Med. Plant Res. 2013, 7, 1121–1128. [Google Scholar]
- Ryu, J.; Kwon, S.-J.; Ahn, J.-W.; Jo, Y.D.; Kim, S.H.; Jeong, S.W.; Lee, M.K.; Kim, J.-B.; Kang, S.-Y. Phytochemicals and antioxidant activity in the kenaf plant (Hibiscus cannabinus L.). J. Plant Biotechnol. 2017, 44, 191–202. [Google Scholar] [CrossRef] [Green Version]
- Son, J.M.; Ju, H.S.; Gung, H.J.N.; Azad, M.O.K.; Adnan, M.D.; Cho, D.H. Comparison of antioxidant active and useful components of the leaves extract fractions from Israeli Kenag (Hibisus cannabinus L.). J. Korean Soc. Med. Crop Sci. 2019, 27, 89. [Google Scholar]
- Ryu, J.; Kwon, S.-J.; Ahn, J.-W.; Ha, B.-K.; Jeong, S.W.; Im, S.B.; Kim, J.-B.; Kim, S.H.; Lee, Y.-K.; Kang, S.-Y. Evaluation of nutritive value and identification of fungi in silage from new Kenaf (Hibiscus cannabinus) cultivars. Int. J. Agric. Biol. 2016, 18. [Google Scholar] [CrossRef]
- Adnan, M.; Azad, M.O.K.; Madhusudhan, A.; Saravanakumar, K.; Hu, X.; Wang, M.-H.; Ha, C.D. Simple and cleaner system of silver nanoparticle synthesis using kenaf seed and revealing its anticancer and antimicrobial potential. Nanotechnology 2020, 31, 265101. [Google Scholar] [CrossRef] [PubMed]
- Harborne, A.J. Phytochemical Methods a Guide to Modern Techniques of Plant Analysis; Springer Science & Business Media: Berlin, Germany, 1998; ISBN 0412572702. [Google Scholar]
- Azad, M.O.K.; Park, B.S.; Adnan, M.; Germ, M.; Kreft, I.; Woo, S.H.; Park, C.H. Silicon biostimulant enhances the growth characteristics and fortifies the bioactive compounds in common and Tartary buckwheat plant. J. Crop Sci. Biotechnol. 2020, 2020, 1–9. [Google Scholar] [CrossRef]
- Kalam Azad, M.O.; Jeong, D.I.; Adnan, M.; Salitxay, T.; Heo, J.W.; Naznin, M.T.; Lim, J.D.; Cho, D.H.; Park, B.J.; Park, C.H. Effect of different processing methods on the accumulation of the phenolic compounds and antioxidant profile of broomcorn millet (Panicum miliaceum L.) Flour. Foods 2019, 8, 230. [Google Scholar] [CrossRef] [Green Version]
- Braca, A.; De Tommasi, N.; Di Bari, L.; Pizza, C.; Politi, M.; Morelli, I. Antioxidant principles from bauhinia t arapotensis. J. Nat. Prod. 2001, 64, 892–895. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [Green Version]
- Heffels, P.; Weber, F.; Schieber, A. Influence of accelerated solvent extraction and ultrasound-assisted extraction on the anthocyanin profile of different Vaccinium species in the context of statistical models for authentication. J. Agric. Food Chem. 2015, 63, 7532–7538. [Google Scholar] [CrossRef]
- Adnan, M.; Chy, M.N.U.; Rudra, S.; Tahamina, A.; Das, R.; Tanim, M.A.H.; Siddique, T.I.; Hoque, A.; Tasnim, S.M.; Paul, A.; et al. Evaluation of Bonamia semidigyna (Roxb.) for antioxidant, antibacterial, anthelmintic and cytotoxic properties with the involvement of polyphenols. Orient. Pharm. Exp. Med. 2019, 19, 187–199. [Google Scholar] [CrossRef]
- Pin, K.Y.; Chuah, A.L.; Rashih, A.A.; Mazura, M.P.; Fadzureena, J.; Vimala, S.; Rasadah, M.A. Antioxidant and anti-inflammatory activities of extracts of betel leaves (Piper betle) from solvents with different polarities. J. Trop. For. Sci. 2010, 22, 448–455. [Google Scholar]
- Sim, Y.Y.; Ong, W.T.J.; Nyam, K.L. Effect of various solvents on the pulsed ultrasonic assisted extraction of phenolic compounds from Hibiscus cannabinus L. leaves. Ind. Crops Prod. 2019, 140, 111708. [Google Scholar] [CrossRef]
- Yusri, N.M.; Chan, K.W.; Iqbal, S.; Ismail, M. Phenolic content and antioxidant activity of Hibiscus cannabinus L. seed extracts after sequential solvent extraction. Molecules 2012, 17, 12612–12621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chatha, S.A.S.; Anwar, F.; Manzoor, M. Evaluation of the antioxidant activity of rice bran extracts using different antioxidant assays. Grasas Aceites 2006, 57, 328–335. [Google Scholar]
- Sultana, B.; Anwar, F.; Ashraf, M. Effect of extraction solvent/technique on the antioxidant activity of selected medicinal plant extracts. Molecules 2009, 14, 2167–2180. [Google Scholar] [CrossRef]
- Yi, T.; Li, S.-M.; Fan, J.-Y.; Fan, L.-L.; Zhang, Z.-F.; Luo, P.; Zhang, X.-J.; Wang, J.-G.; Zhu, L.; Zhao, Z.-Z. Comparative analysis of EPA and DHA in fish oil nutritional capsules by GC-MS. Lipids Health Dis. 2014, 13, 190. [Google Scholar] [CrossRef] [Green Version]
- Huertas-Pérez, J.F.; Ernest, M.; Badoud, F. Quantification of folpet and phthalimide in tea and herbal infusions by LC–high-resolution MS and GC–MS/MS. Food Addit. Contam. Part A 2019, 36, 109–119. [Google Scholar] [CrossRef]
- Search List. Available online: https://phytochem.nal.usda.gov/phytochem/search/list (accessed on 3 September 2020).
- Do, Q.D.; Angkawijaya, A.E.; Tran-Nguyen, P.L.; Huynh, L.H.; Soetaredjo, F.E.; Ismadji, S.; Ju, Y.-H. Effect of extraction solvent on total phenol content, total flavonoid content, and antioxidant activity of Limnophila aromatica. J. Food Drug Anal. 2014, 22, 296–302. [Google Scholar] [CrossRef] [Green Version]
- Abarca-Vargas, R.; Pena Malacara, C.F.; Petricevich, V.L. Characterization of chemical compounds with antioxidant and cytotoxic activities in bougainvillea x buttiana holttum and standl, (Var. rose) extracts. Antioxidants 2016, 5, 45. [Google Scholar] [CrossRef] [Green Version]
- Abu, F.; Taib, M.; Norma, C.; Moklas, M.; Aris, M.; Mohd Akhir, S. Antioxidant properties of crude extract, partition extract, and fermented medium of dendrobium sabin flower. Evid.-Based Complement. Altern. Med. 2017, 2017. [Google Scholar] [CrossRef] [Green Version]
- Boeing, J.S.; Barizão, É.O.; e Silva, B.C.; Montanher, P.F.; de Cinque Almeida, V.; Visentainer, J.V. Evaluation of solvent effect on the extraction of phenolic compounds and antioxidant capacities from the berries: Application of principal component analysis. Chem. Cent. J. 2014, 8, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Samarakoon, K.W.; Lakmal, H.H.C.; Kim, S.Y.; Jeon, Y.-J. Electron spin resonance spectroscopic measurement of antioxidant activity of organic solvent extracts derived from the methanolic extracts of Sri Lankan thebu leaves (Costus speciosus). J. Natl. Sci. Found. Sri Lanka 2014, 42, 209–216. [Google Scholar] [CrossRef]
- Adnan, M.; Chy, M.N.U.; Kamal, A.T.M.M.; Chowdhury, K.A.A.; Rahman, M.A.; Reza, A.S.M.A.; Moniruzzaman, M.; Rony, S.R.; Nasrin, M.S.; Azad, M.O.K.; et al. Intervention in neuropsychiatric disorders by suppressing inflammatory and oxidative stress signal and exploration of in silico studies for potential lead compounds from Holigarna caustica (Dennst.) Oken leaves. Biomolecules 2020, 10, 561. [Google Scholar] [CrossRef] [Green Version]
- Danihelová, M.; Veverka, M.; Šturdík, E.; Jantová, S. Antioxidant action and cytotoxicity on HeLa and NIH-3T3 cells of new quercetin derivatives. Interdiscip. Toxicol. 2013, 6, 209–216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guyton, J.R.; Lenz, M.L.; Mathews, B.; Hughes, H.; Karsan, D.; Selinger, E.; Smith, C. V Toxicity of oxidized low density lipoproteins for vascular smooth muscle cells and partial protection by antioxidants. Atherosclerosis 1995, 118, 237–249. [Google Scholar] [CrossRef]
- Wong, Y.H.; Tan, W.Y.; Tan, C.P.; Long, K.; Nyam, K.L. Cytotoxic activity of kenaf (Hibiscus cannabinus L.) seed extract and oil against human cancer cell lines. Asian Pac. J. Trop. Biomed. 2014, 4, S510–S515. [Google Scholar] [CrossRef]
- Chen, V.; Staub, R.E.; Baggett, S.; Chimmani, R.; Tagliaferri, M.; Cohen, I.; Shtivelman, E. Identification and analysis of the active phytochemicals from the anti-cancer botanical extract Bezielle. PLoS ONE 2012, 7, e30107. [Google Scholar] [CrossRef] [Green Version]
- Khan, T.; Ali, M.; Khan, A.; Nisar, P.; Jan, S.A.; Afridi, S.; Shinwari, Z.K. Anticancer Plants: A review of the active phytochemicals, applications in animal models, and regulatory aspects. Biomolecules 2020, 10, 47. [Google Scholar] [CrossRef] [Green Version]
- Vaquero, M.J.R.; Serravalle, L.R.T.; De Nadra, M.C.M.; De Saad, A.M.S. Antioxidant capacity and antibacterial activity of phenolic compounds from argentinean herbs infusions. Food Control. 2010, 21, 779–785. [Google Scholar] [CrossRef] [Green Version]
- Kabir, M.I.; Adnan, M.; Rahman, M.M. Natural sources of tocotrienols: A note on absorption. J. Silico Vitr. Pharmacol. 2017, 3, 1–5. [Google Scholar] [CrossRef]
- Adnan, M.; Chy, N.U.; Mostafa Kamal, A.T.M.; Azad, M.O.K.; Paul, A.; Uddin, S.B.; Barlow, J.W.; Faruque, M.O.; Park, C.H.; Cho, D.H. Investigation of the biological activities and characterization of bioactive constituents of Ophiorrhiza rugosa var. prostrata (D. Don) & Mondal leaves through in vivo, in vitro, and in silico approaches. Molecules 2019, 24, 1367. [Google Scholar]
- Patra, J.K.; Baek, K.-H. Antibacterial activity and action mechanism of the essential oil from Enteromorpha linza L. against foodborne pathogenic bacteria. Molecules 2016, 21, 388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, R.; Islam, B.; Akram, M.; Shakil, S.; Ahmad, A.A.; Ali, S.M.; Siddiqui, M.; Khan, A.U. Antimicrobial activity of five herbal extracts against multi drug resistant (MDR) strains of bacteria and fungus of clinical origin. Molecules 2009, 14, 586–597. [Google Scholar] [CrossRef] [PubMed]





| S/L no | R. Time (min) | PA (%) | Compound Name | Molecular Formula | MW (g/mol) | *** Activity |
|---|---|---|---|---|---|---|
| Leaves | ||||||
| 1 | 3.42 | 1.56 | 2-(Tert-butylamino)-3-methyl-2-pentenenitrile | C10H18N2 | 166.3 | No activity |
| 2 | 3.51 | 1.25 | 5-Methylfurfural | C6H6O2 | 110.1 | Antioxidant, antiproliferative, antibacterial |
| 3 | 4.01 | 1.75 | Cyclopropanecarboxamide | C4H7NO | 85.1 | No activity |
| 4 | 4.24 | 2.36 | 5-Amino-6-nitrosopyrimidine-2,4(1h,3h)-dione | C4H4N4O3 | 156.1 | Antibacterial |
| 5 | 4.47 | 0.25 | Hexyl octanoate | C14H28O2 | 228.4 | Flavouring agent |
| 6 | 4.54 | 1.43 | Pyrrolidin-5-one, 2,3-dedihydro-3-nitro- | C4H3NO4 | 129.1 | No activity |
| 7 | 4.69 | 0.27 | 3-Amino-2-oxazolidinone | C3H6N2O2 | 102.1 | No activity |
| 8 | 4.76 | 3.70 | 4H-Pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- | C6H8O4 | 144.1 | Antibacterial, anti-inflammatory, antiproliferative antioxidant, automatic nerve activity, anticancer |
| 9 | 5.13 | 0.84 | 3,4-Pentadienal | C5H6O | 82.1 | No activity |
| 10 | 5.27 | 0.34 | cyclobut-1-enylmethanol | C5H8O | 84.1 | Antibacterial |
| 11 | 5.32 | 7.20 | 5-Hydroxymethylfurfural | C6H6O3 | 126.1 | Anti-oxidative, anti-allergic, anti-inflammatory, anti-hypoxic, anti-hyperuricemic |
| 12 | 5.89 | 0.21 | 6-O-Acetyl-beta-D-glucose- | C8H14O7 | 222.2 | |
| 13 | 5.93 | 0.43 | 6-Oxa-1-azabicyclo(3.1.0)hexane, 2,2-dimethyl-4,5-diphenyl-, trans- | C18H19NO | 265.3 | Antibacterial |
| 14 | 6.12 | 0.62 | N-(2-Methoxyethyl)alanine | C6H13NO3 | 147.2 | No activity |
| 15 | 6.17 | 1.00 | trans-2-Butenyl acetate | C6H10O2 | No activity | |
| 16 | 6.81 | 0.96 | N-Acetyl-d-serine | C5H9NO4 | 147.1 | No activity |
| 17 | 6.84 | 1.03 | alpha-D-Mannopyranoside, methyl 3,6-anhydro- | C7H12O5 | 176.2 | Antibacterial |
| 18 | 7.19 | 0.27 | 2(4H)-Benzofuranone, 5,6,7,7a-tetrahydro-6-hydroxy-4,4,7a-trimethyl- | C11H16O3 | 196.2 | Antibacterial |
| 19 | 7.24 | 0.46 | 2-Mercaptopyridine-4-ol | C5H5NOS | 127.17 | No activity |
| 20 | 7.71 | 3.5 | 1,3,4,5-Tetrahydroxycyclohexanecarboxylic acid | C7H12O6 | 192.2 | Hepatoprotective |
| 21 | 8.15 | 1.00 | d-Glycero-d-galacto-heptose | C7H14O7 | 210.1 | Antioxidant |
| 22 | 8.27 | 0.30 | 7-Hydroxy-3-(1,1-dimethylprop-2-enyl)coumarin | C14H14O3 | 230.3 | Antibacterial, antitumor |
| 23 | 8.30 | 1.11 | N-Acetyl-D-Glucosamine | C8H15NO6 | 221.1 | Antibacterial, antitumor, antioxidant, anticoagulant, wound healing |
| 24 | 8.39 | 0.64 | Z-8-Methyl-9-tetradecen-1-ol acetate | C17H32O2 | 268.4 | No activity |
| 25 | 8.5 | 0.21 | 3-Methyl-4-(phenylthio)-2-prop-2-enyl-2,5-dihydrothiophene 1,1-dioxide | C14H16O2S2 | 280.4 | No activity |
| 26 | 8.75 | 0.69 | Methyl tricosanoate | C24H48O2 | 368.6 | Antibacterial |
| 27 | 8.93 | 2.05 | Palmitic acid | C16H32O2 | 256.4 | Antioxidant, antitumor, anti-inflammatory, antibacterial, antifungal |
| 28 | 9.14 | 0.77 | Pentadecanoic acid | C15H30O2 | 242.4 | Adhesives, agricultural chemicals (non-pesticidal), lubricants |
| 29 | 9.19 | 0.41 | 26-Hydroxycholesterol | C27H46O2 | 402.7 | No activity |
| 30 | 9.26 | 0.50 | Hexacosanoic acid | C26H52O2 | 396.7 | No activity |
| 31 | 9.32 | 0.60 | Palmitic acid | C16H32O2 | 256.4 | Antioxidant, antitumor, anti-inflammatory, antibacterial, antifungal |
| 32 | 9.43 | 1.78 | 9,12,15-Octadecatrienoic acid, methyl ester | C16H32O2 | 292.5 | No activity |
| 33 | 9.47 | 1.24 | Phytol | C20H40O | 296.5 | Antioxidant, analgesic, antibacterial, anti-inflammatory, anticancer, and neuroprotective |
| 34 | 9.61 | 2.96 | 1,5,9,13-Tetradecatetraene | C14H22 | 190.3 | No activity |
| 35 | 9.80 | 3.40 | 2-Linoleoylglycerol | C21H38O4 | 354.5 | Antibacterial |
| 36 | 9.94 | 0.42 | Stearic acid | C18H36O2 | 284.5 | Antioxidant, antibacterial |
| 37 | 9.98 | 0.50 | Epoxycholesterol | C27H46O2 | 402.7 | No activity |
| 38 | 10.06 | 3.06 | Hexadecanoate | C16H31O2 | 255.42 | Anti-inflammatory |
| 39 | 10.21 | 1.56 | 4alpha,5alpha-Epoxycholestane | C27H46O | 386.7 | No activity |
| 40 | 10.70 | 2.83 | 6,10,14-Trimethylpentadecan-2-one | C18H36O | 268.5 | antibacterial |
| 41 | 10.80 | 1.05 | 1-Cinnamyl-3-methylindole-2-carbaldehyde | C19H17NO | 275.1 | Antioxidant, antibacterial |
| 42 | 10.85 | 0.92 | 3,5-Bis-(trichloromethyl)-benzoyl chloride | C9H3Cl7O | 375.3 | No activity |
| 43 | 10.92 | 5.44 | 2-Stearoylglycerol | C21H42O4 | 358.6 | No activity |
| 44 | 11.60 | 1.58 | Tricyclo[10.2.2.2(5,8)]octadeca-5,7,12,14,15,17-hexaene | C18H20 | 236.3 | Antifungal |
| 45 | 11.70 | 2.17 | 9,12,15-Octadecatrien-1-ol | C18H32O | 264.4 | Antioxidant, antibacterial |
| 46 | 11.74 | 4.50 | 1,5,9,13-Tetradecatetraene | C14H22 | 190.3 | No activity |
| 47 | 11.79 | 3.16 | 5 beta-Coprostanol | C27H48O | 388.7 | No activity |
| 48 | 12.24 | 1.64 | Curan-17-oic acid, 2,16-didehydro-20-hydroxy-19-oxo-, methyl ester | C20H22N2O4 | 354.4 | Antibacterial, antifungal |
| 49 | 14.65 | 4.45 | Vitamin E | C29H50O2 | 430.7 | Antioxidant, antibacterial, Analgesic, anti-inflammatory, anxiolytic and antidepressant, |
| 50 | 15.28 | 1.52 | 3-{[(3,5-Dichlorophenyl)imino]methyl}-1,2-benzenediol | C13H9Cl2NO2 | 282.1 | Antibacterial, antifungal |
| 51 | 16.05 | 0.85 | Methyl 4-oxo-4,5,6,7-tetrahydro-1H-indole-2-carboxylate | C10H11NO3 | 193.2 | Antidiabetic |
| 52 | 16.26 | 1.07 | Stigmasta-5,22-dien-3-ol | C29H48O | 412.7 | Antibacterial |
| 53 | 17.05 | 3.93 | Clionasterol | C29H50O | 414.7 | Antibacterial |
| 54 | 17.81 | 1.71 | Hexadecahydropyrene | C16H26 | 218.3 | No activity |
| 55 | 18.56 | 3.76 | alpha-Amyrin | C30H50O | 426.7 | Analgesic, anti-inflammatory, anxiolytic and antidepressant |
| Seed | ||||||
| 1 | 4.00 | 0.92 | Glycerine | C3H8O3 | 92.1 | Antibacterial |
| 2 | 5.29 | 0.03 | Benzofuran, 2, 3-dihydro- | C15H14OS | 242.3 | Antidepressant, anticancer, antiviral, antifungal, antioxidant, anti-psychotic |
| 3 | 6.89 | 0.16 | N-Acetylethylenediamine | C4H10NO2 | 102.1 | Antibacterial |
| 4 | 7.76 | 0.08 | d-Mannitol, 1, 4-anhydro- | C6H12O5 | 164.2 | No activity |
| 5 | 8.08 | 0.03 | Octadecanoic acid | C18H36O2 | 284.5 | Antibacterial |
| 6 | 8.75 | 0.05 | Hexadecanoic acid, methyl ester | C17H34O2 | 270.5 | Antibacterial, anticancer, anti-inflammatory, anti-diuretic |
| 7 | 9.05 | 8.83 | Hexadecanoic acid | C16H32O2 | 256.4 | Antioxidant, antitumor, anti-inflammatory, antibacterial, antifungal |
| 8 | 9.43 | 1.42 | Hexadecanoic acid | C16H32O2 | 256.4 | Antioxidant, antitumor, anti-inflammatory, antibacterial, antifungal |
| 9 | 9.81 | 77.46 | 9-octadecanoic acid (z)- | C18H34O2 | 282.5 | Antibacterial, antifungal |
| 10 | 10.71 | 1.58 | Linoleic acid | C18H32O2 | 280.4 | Antibacterial |
| 11 | 10.95 | 2.85 | Linoleic acid | C18H32O2 | 280.4 | Antibacterial |
| 12 | 11.75 | 6.21 | 9, 12-octadecadienoic acid (z, z)-, 2-hydroxy-1-(hydroxymethyl)ethyl ester | C21H38O4 | 354.5 | Antioxidant, antibacterial |
| 13 | 13.97 | 0.33 | 17-(acetyloxy)-4, 4-dimethyl-7-oxoandrost-5-en-3-yl acetate | C25H36O5 | 416.5 | No activity |
| 14 | 17.07 | 0.04 | .beta.-Sitosterol | C29H50O | 414.7 | Antidiabetic, antibacterial |
| Zone of Inhibition (mm) | ||||||
|---|---|---|---|---|---|---|
| Bacterial Strain | Gram Negative | Gram Positive | ||||
| Name of Bacteria | Escherichia Coli | Bacillus Cereus | Bacillus Subtilis | |||
| Concentration (50 mg/mL) | Leaves | Seed | Leaves | Seed | Leaves | Seed |
| NHX | - | - | - | - | - | - |
| EA | - | 15.2 ± 0.72 | - | 12.8 ± 0.78 | - | 11.6 ± 0.59 |
| ETH | 9.8 ± 1.41 | 13.1 ± 0.34 | - | 11.2 ± 0.37 | - | 10.1 ± 0.41 |
| WT | 11.3 ± 0.38 | 13.7 ± 0.56 | 9.7 ± 0.73 | 12.5 ± 0.82 | - | 9.3 ± 0.28 |
| Standard Ampicillin (25 ug/mL) | 29.4 ± 0.26 | 27.9 ± 0.48 | 26.4 ± 0.52 | 28.4 ± 0.47 | 30.1 ± 0.18 | 28.4 ± 0.47 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adnan, M.; Oh, K.K.; Azad, M.O.K.; Shin, M.H.; Wang, M.-H.; Cho, D.H. Kenaf (Hibiscus cannabinus L.) Leaves and Seed as a Potential Source of the Bioactive Compounds: Effects of Various Extraction Solvents on Biological Properties. Life 2020, 10, 223. https://doi.org/10.3390/life10100223
Adnan M, Oh KK, Azad MOK, Shin MH, Wang M-H, Cho DH. Kenaf (Hibiscus cannabinus L.) Leaves and Seed as a Potential Source of the Bioactive Compounds: Effects of Various Extraction Solvents on Biological Properties. Life. 2020; 10(10):223. https://doi.org/10.3390/life10100223
Chicago/Turabian StyleAdnan, Md., Ki Kwang Oh, Md Obyedul Kalam Azad, Myung Hwan Shin, Myeong-Hyeon Wang, and Dong Ha Cho. 2020. "Kenaf (Hibiscus cannabinus L.) Leaves and Seed as a Potential Source of the Bioactive Compounds: Effects of Various Extraction Solvents on Biological Properties" Life 10, no. 10: 223. https://doi.org/10.3390/life10100223
APA StyleAdnan, M., Oh, K. K., Azad, M. O. K., Shin, M. H., Wang, M.-H., & Cho, D. H. (2020). Kenaf (Hibiscus cannabinus L.) Leaves and Seed as a Potential Source of the Bioactive Compounds: Effects of Various Extraction Solvents on Biological Properties. Life, 10(10), 223. https://doi.org/10.3390/life10100223








