Multivariate Analysis Based on Geochemical, Isotopic, and Mineralogical Compositions of Uranium-Rich Samples
Abstract
:1. Introduction
2. Materials and Methods
2.1. Deposit Type and Sample Descriptions
2.2. Analytical Methods
3. Results and Discussion
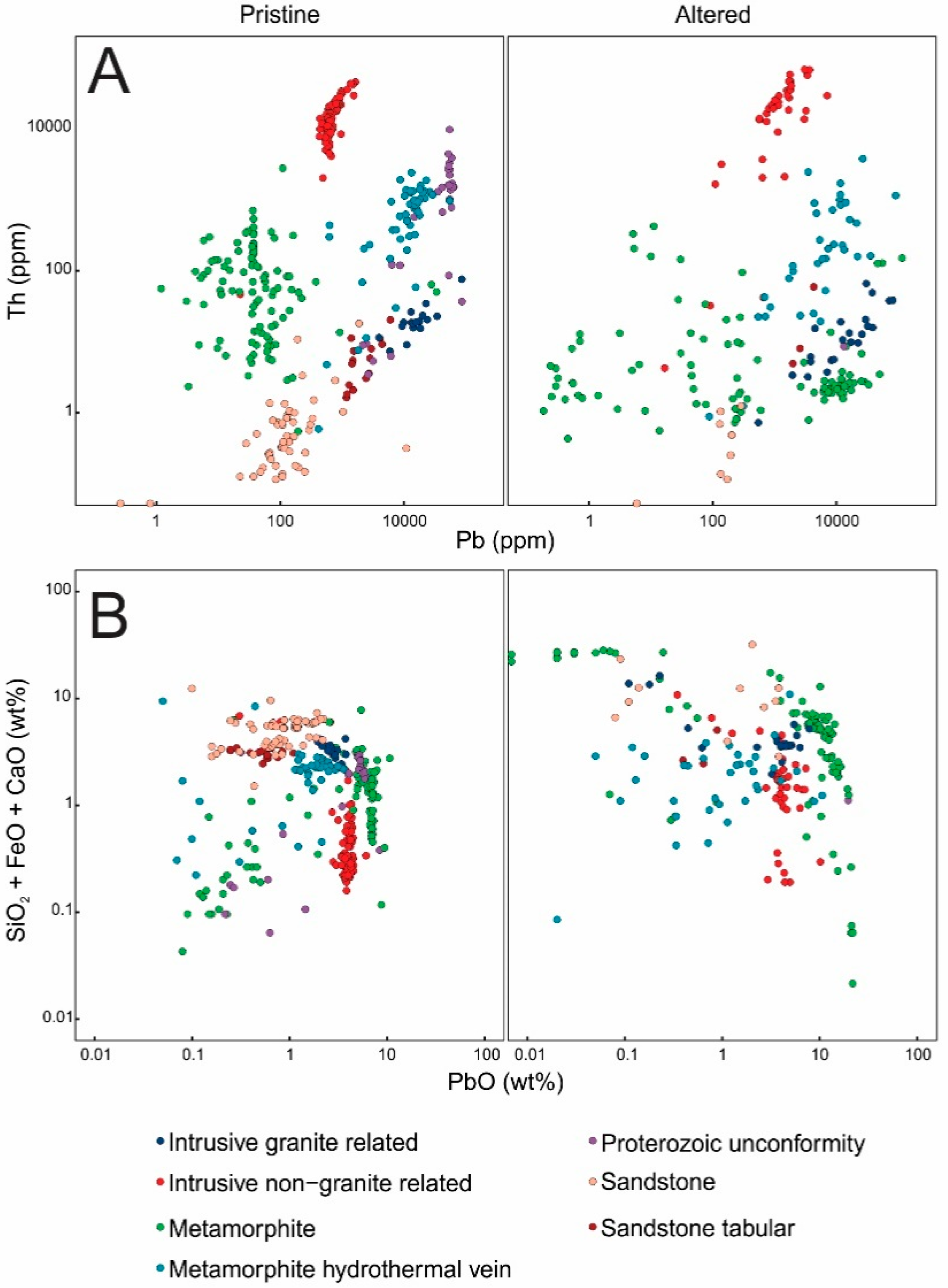
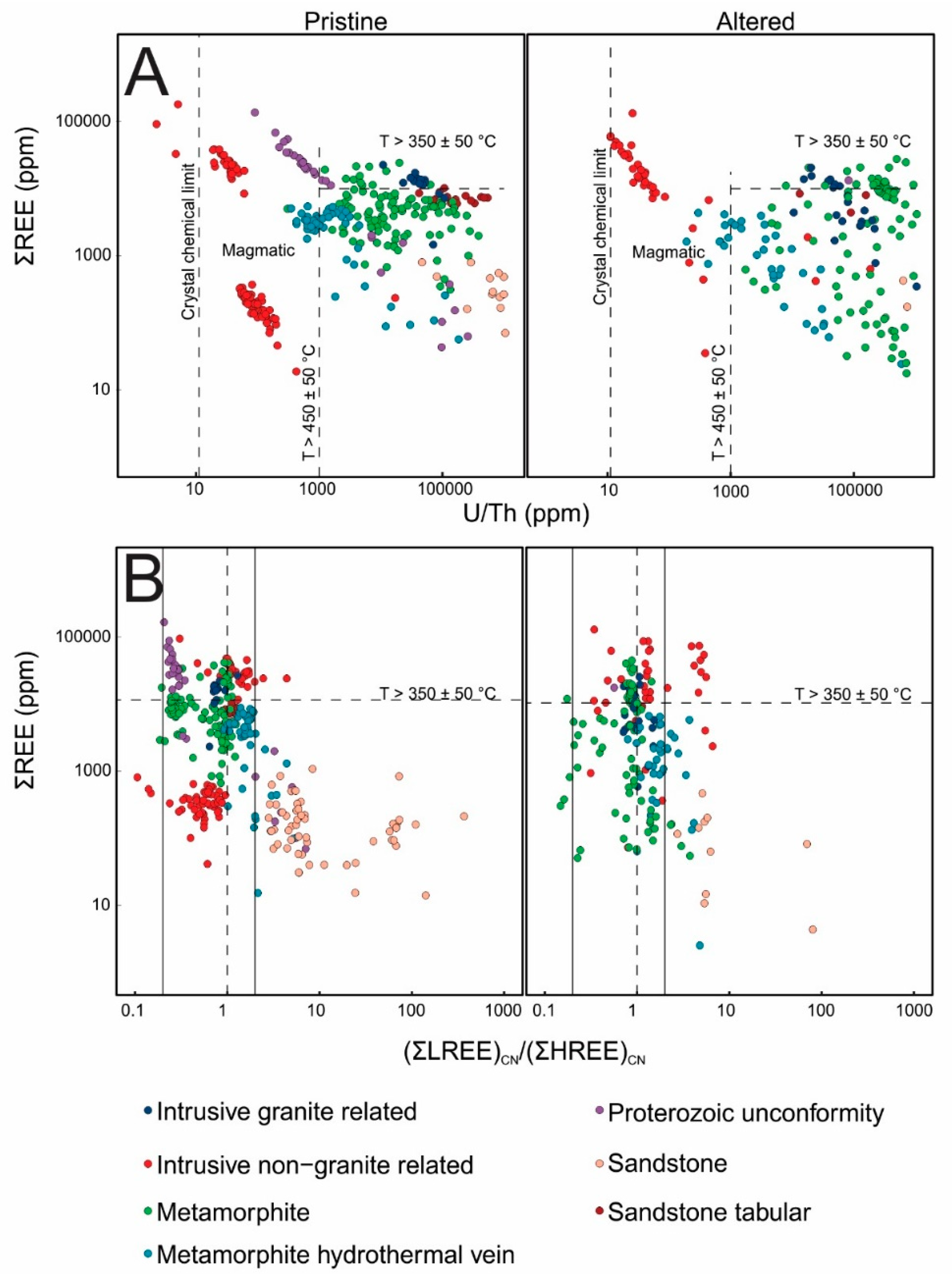
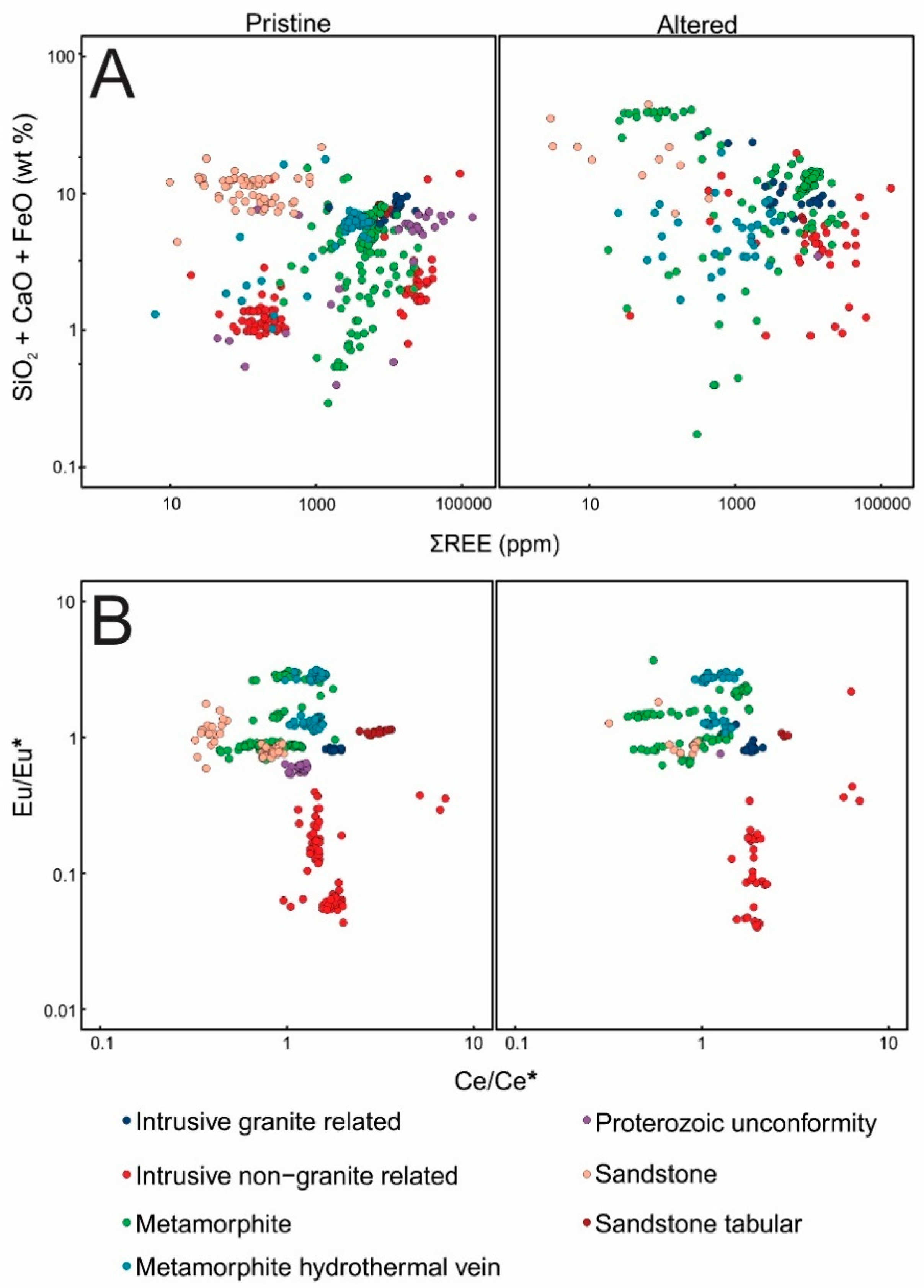
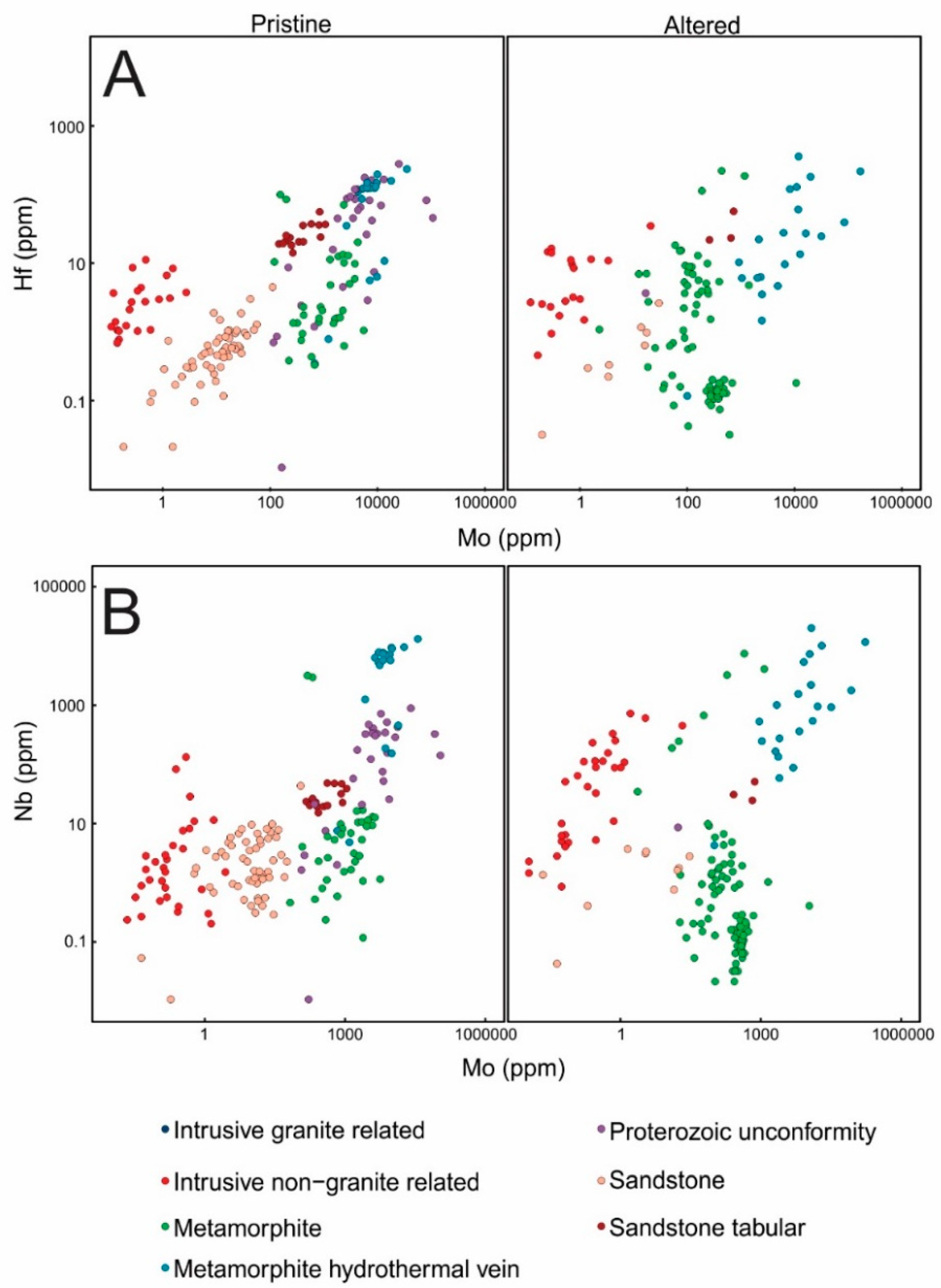
3.1. Major and Trace Element Compositions and PXRD Results
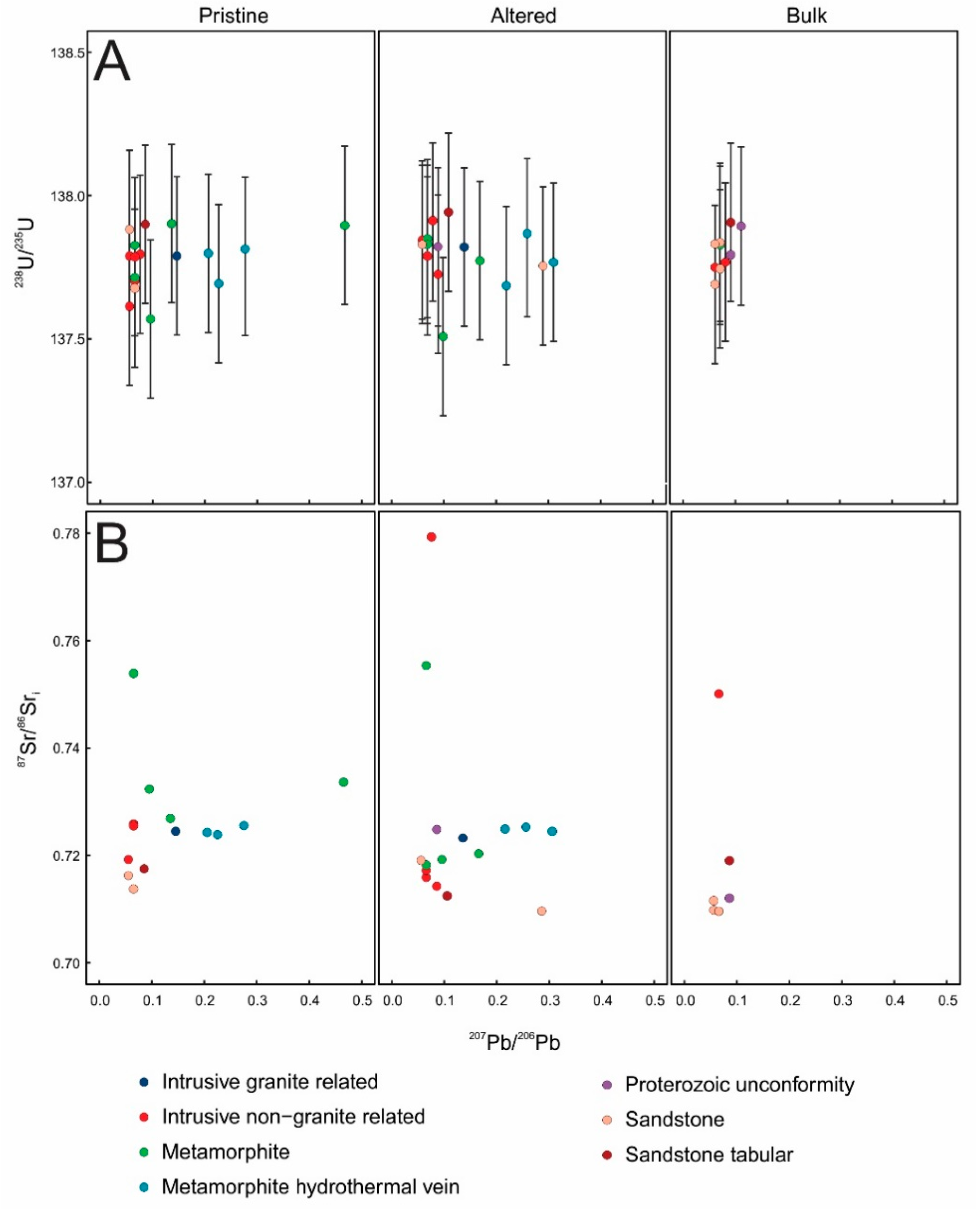
3.2. Radiogenic Isotopes
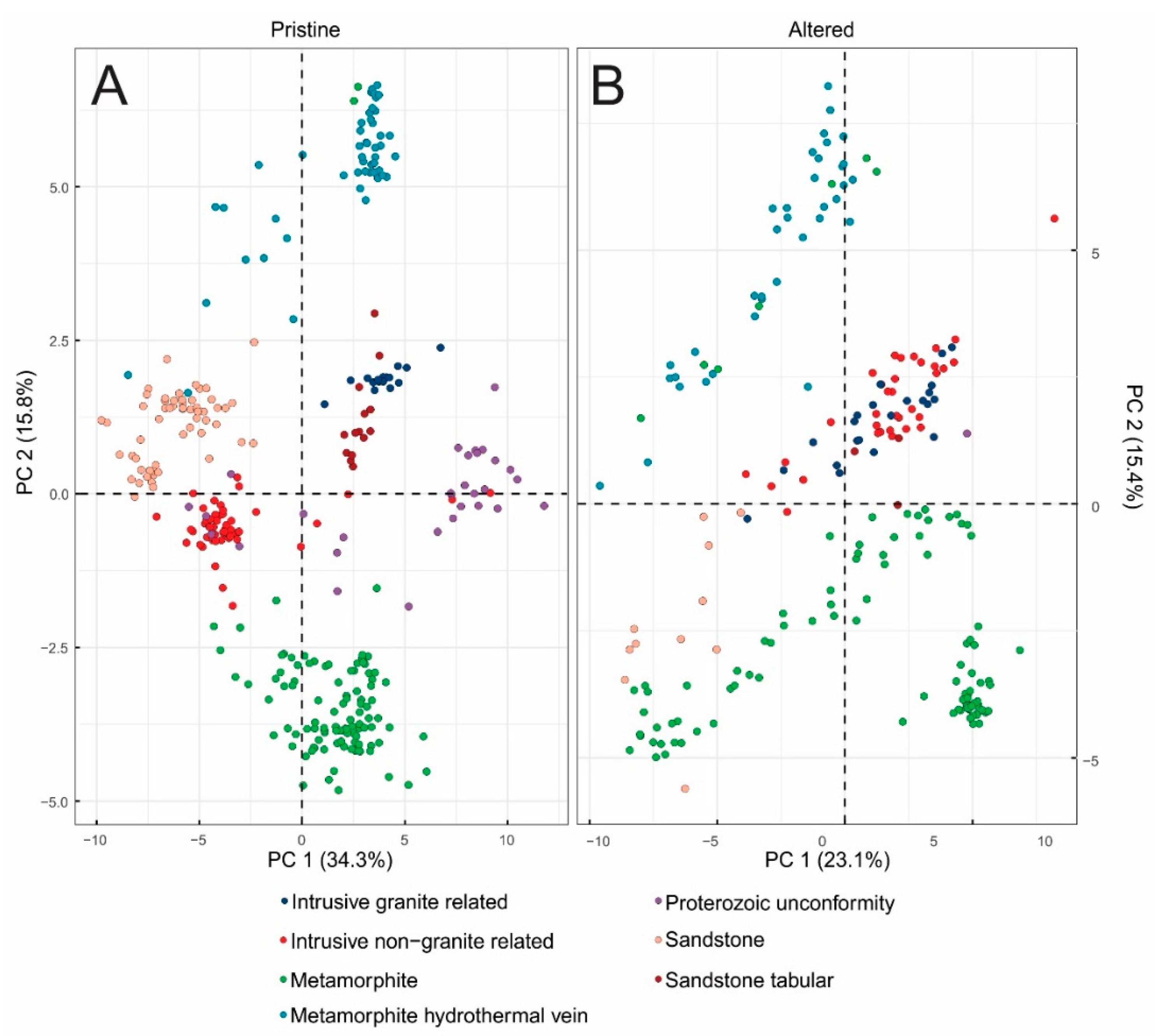
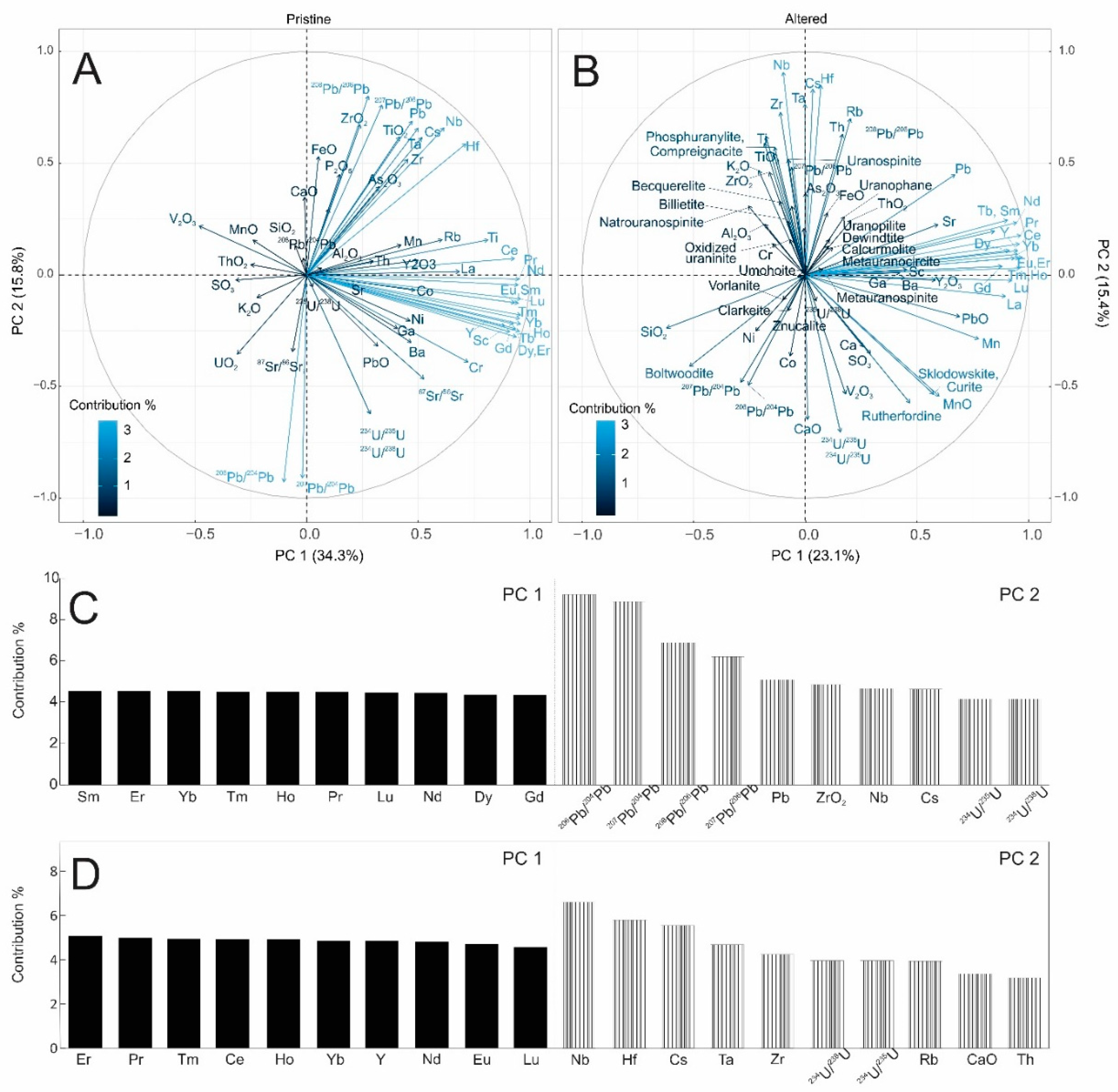
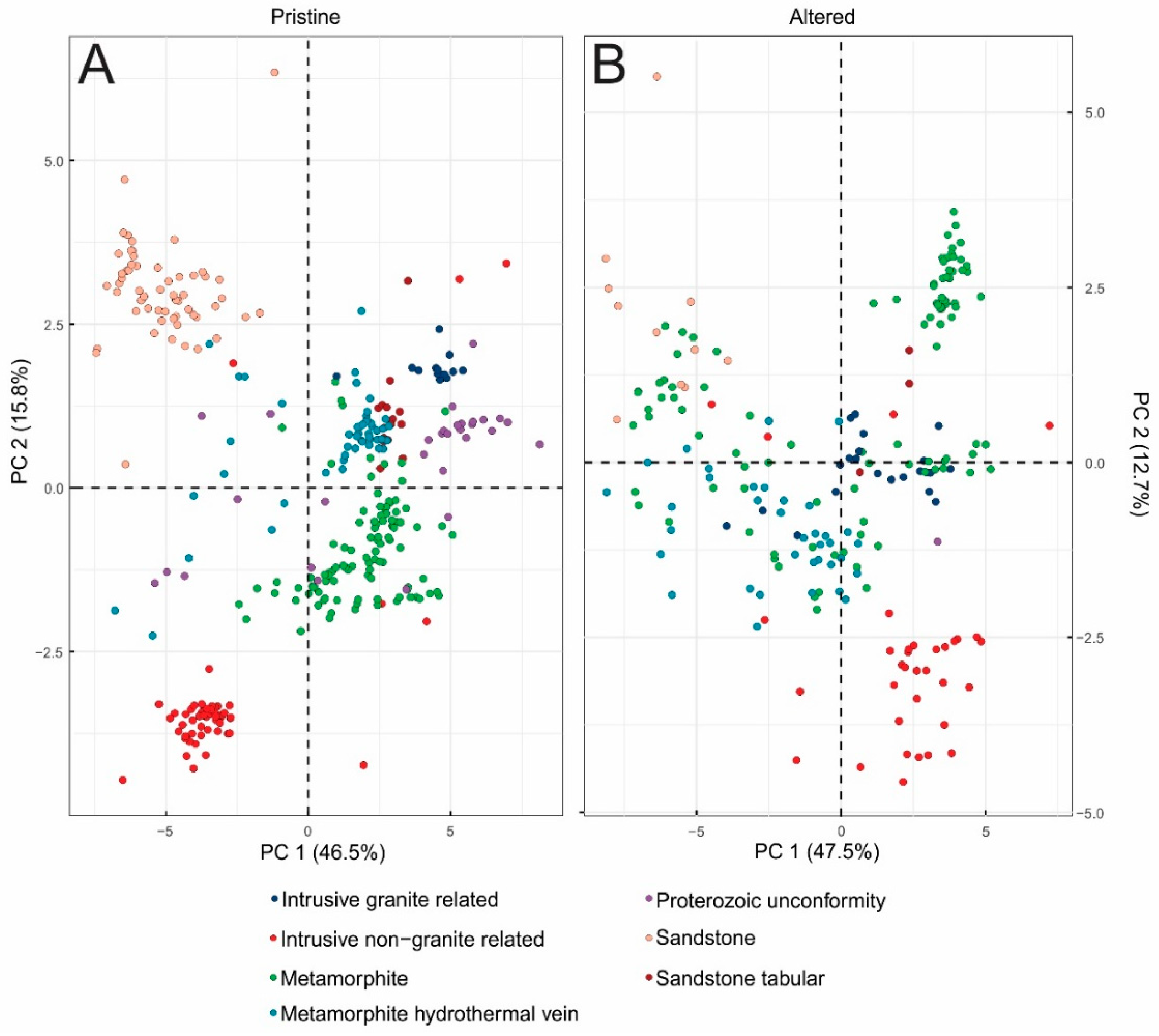
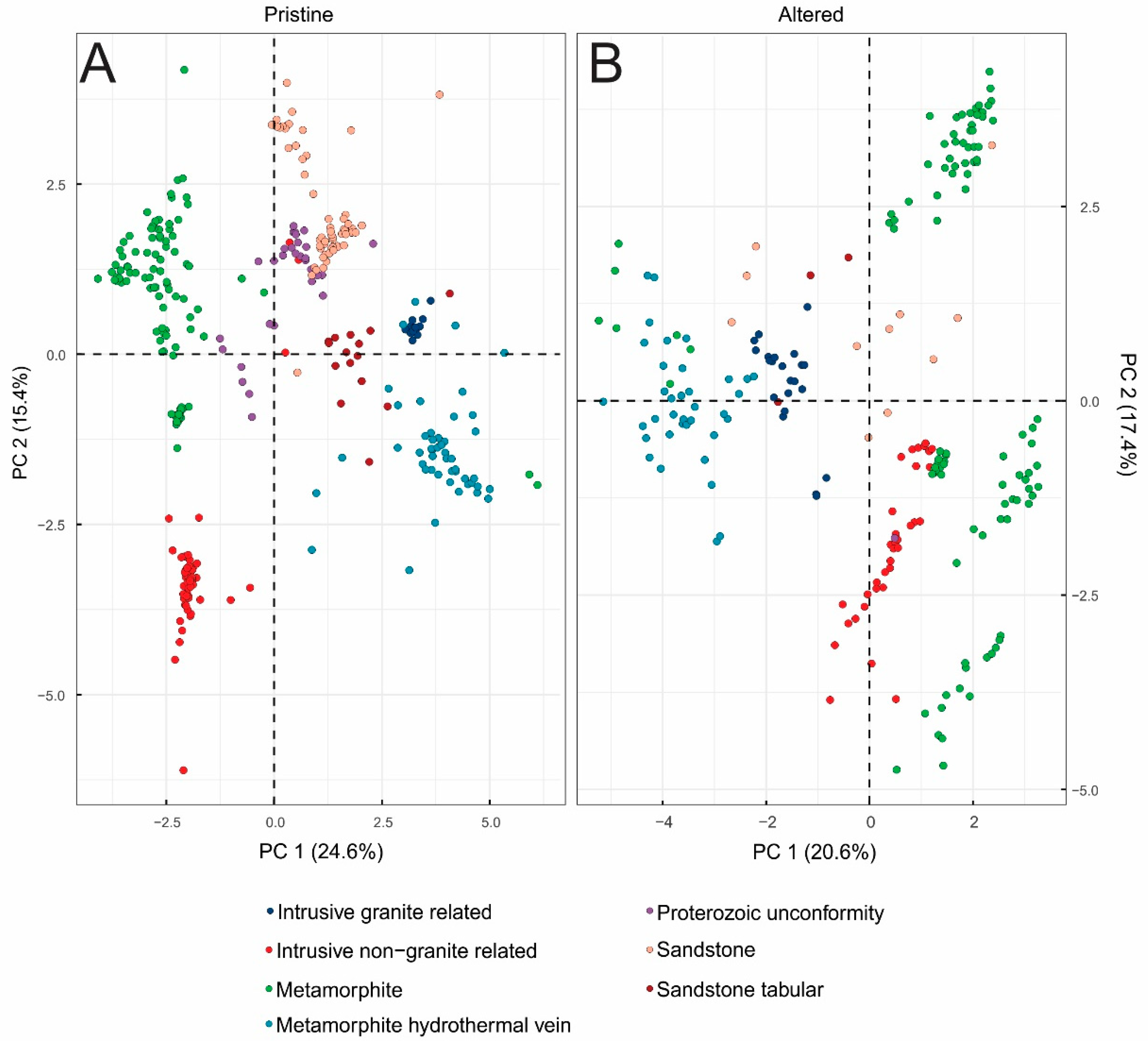
3.3. Principal Component Analysis (PCA) Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Atomic Energy Agency (2019). Incident and Trafficking Database (ITDB). Incidents of Nuclear and Other Radioactive Material Out of Regulatory Control. IAEA, 2019; p. 6. Available online: http://www-ns.iaea.org/downloads/security/itdb-fact-sheet.pdf (accessed on 23 May 2019).
- Balboni, E.; Jones, N.; Spano, T.; Simonetti, A.; Burns, P.C. Chemical and Sr isotopic characterization of North America uranium ores: Nuclear forensic applications. Appl. Geochem. 2016, 74, 24–32. [Google Scholar] [CrossRef] [Green Version]
- Varga, Z.; Wallenius, M. Analysis of uranium ore concentrates for origin assessment. Proc. Radiochem. Suppl. Radiochim. Acta 2011, 1, 27–30. [Google Scholar]
- Varga, Z.; Krajkó, J.; Peńkin, M.; Novák, M.; Eke, Z.; Wallenius, M.; Mayer, K. Identification of uranium signatures relevant for nuclear safeguards and forensics. J. Radioanal. Nucl. Chem. 2017, 312, 639–654. [Google Scholar] [CrossRef] [Green Version]
- Cuney, M. Evolution of uranium fractionation processes through time: Driving the secular variation of uranium deposit types. Econ. Geol. 2010, 105, 553–569. [Google Scholar] [CrossRef]
- Eglinger, A.; André-Mayer, A.S.; Vanderhaeghe, O.; Mercadier, J.; Cuney, M.; Decrée, S.; Feybesse, J.L.; Milesi, J.P. Geochemical signatures of uranium oxides in the Lufilian belt: From unconformity-related to syn-metamorphic uranium deposits during the Pan-African orogenic cycle. Ore Geol. Rev. 2013, 54, 197–213. [Google Scholar] [CrossRef]
- Frimmel, H.E.; Schedel, S.; Brätz, H. Uraninite chemistry as forensic tool for provenance analysis. Appl. Geochem. 2014, 48, 104–121. [Google Scholar] [CrossRef]
- Janeczek, J.; Ewing, R.C. Mechanisms of lead release from uraninite in the natural fission reactors in Gabon. Geochim. et Cosmochim. Acta 1995, 59, 1917–1931. [Google Scholar] [CrossRef]
- Keegan, E.; Richter, S.; Kelly, I.; Wong, H.; Gadd, P.; Kuehn, H.; Alonso-Munoz, A. The provenance of Australian uranium ore concentrates by elemental and isotopic analysis. Appl. Geochem. 2008, 23, 765–777. [Google Scholar] [CrossRef]
- Keegan, E.; Wallenius, M.; Mayer, K.; Varga, Z.; Rasmussen, G. Attribution of uranium ore concentrates using elemental and anionic data. Appl. Geochem. 2012, 27, 1600–1609. [Google Scholar] [CrossRef]
- Mercadier, J.; Cuney, M.; Cathelineau, M.; Lacorde, M. U redox fronts and kaolinisation in basement-hosted unconformity-related U ores of the Athabasca Basin (Canada): Late U remobilisation by meteoric fluids. Miner. Depos. 2011, 46, 105–135. [Google Scholar] [CrossRef]
- Spano, T.L.; Simonetti, A.; Wheeler, T.; Carpenter, G.; Freet, D.; Balboni, E.; Dorais, C.; Burns, P.C. A novel nuclear forensic tool involving deposit type normalized rare earth element signatures. Terra Nova 2017, 29, 294–350. [Google Scholar] [CrossRef]
- International Atomic Energy Agency. Geological Classification of Uranium Deposits and Description of Selected Examples, IAEA-TECDOC-1842; IAEA: Vienna, Austria, 2018. [Google Scholar]
- Alexandre, P.; Kyser, T.K. Effects of cationic substitutions and alteration in uraninite, and implications for the dating of uranium deposits. Can. Mineral. 2005, 43, 1005–1017. [Google Scholar] [CrossRef]
- Janeczek, J.; Ewing, R.C. Structural formula of uraninite. J. Nucl. Mater. 1992, 190, 128–132. [Google Scholar] [CrossRef]
- Plášil, J. Oxidation-hydration weathering of uraninite: The current state-of-knowledge. J. Geosci. 2014, 59, 99–114. [Google Scholar] [CrossRef]
- Balboni, E.; Simonetti, A.; Spano, T.; Cook, N.D.; Burns, P.C. Rare-earth element fractionation in uranium ore and its U(VI) alteration minerals. Appl. Geochem. 2017, 87, 84–92. [Google Scholar] [CrossRef]
- Alexandre, P.; Kyser, K.; Layton-Matthews, D.; Joy, B.; Uvarova, Y. Chemical compositions of natural uraninite. Can. Mineral. 2015, 53, 595–622. [Google Scholar] [CrossRef]
- Chen, S.; Grunsky, E.C.; Hattori, K.; Liu, Y. Principal Component Analysis of Geochemical Data from the REE-Rich Maw Zone, Athabasca Basin, Canada; Natural Resources Canada: Ottawa, ON, Canada, 2015. [Google Scholar]
- Jolliffe, I.T. Principal Component Analysis, 2nd ed.; Springer: Berlin, Germany, 2002. [Google Scholar]
- Brobst, D.A. Geology of the Spruce Pine District Avery, Mitchell, and Yancey Counties North Carolina; US Government Printing Office: Washington, DC, USA, 1962.
- Desbarats, A.J.; Percival, J.B.; Venance, K.E. Trace element mobility in mine waters from granitic pegmatite U-Th-REE deposits, Bancroft area, Ontario. Appl. Geochem. 2016, 67, 153–167. [Google Scholar] [CrossRef]
- Lewis, S.R.; Simonetti, A.; Corcoran, L.; Spano, T.L.; Chung, B.W.; Teslich, N.E.; Burns, P.C. Characterization of uraninite using a FIB–SEM approach and its implications for LA-ICP-MS analyses. J. Radioanal. Nucl. Chem. 2018, 318, 1389–1400. [Google Scholar] [CrossRef]
- Mawdsley, J. Uraninite-bearing deposits, Charlebois Lake area, northeastern Saskatchewan. Can. Inst. Mining Metall. Bull. 1952, 482, 366–375. [Google Scholar]
- Mckeough, M.A.; Lentz, D.R.; Brown, J.A. Geology and Associated Pegmatite- and Vein-hosted Uranium Mineralization of the Kulyk, Eagle, and Karin Lakes Regions, Wollaston Domain, Northern Saskatchewan. Sask. Geol. Surv. 2010, 2, 1–23. [Google Scholar]
- McKeough, M.A.; Lentz, D.R.; Mcfarlane, C.R.M.; Brown, J. Geology and evolution of pegmatite-hosted U-Th ± REE-Y-Nb mineralization, Kulyk, Eagle, and Karin Lakes region, Wollaston Domain, northern Saskatchewan, Canada: Examples of the dual role of extreme fractionation and hybridization processes. J. Geosci. 2013, 58, 321–346. [Google Scholar] [CrossRef]
- Decrée, S.; Deloule, É.; De Putter, T.; Dewaele, S.; Mees, F.; Yans, J.; Marignac, C. SIMS U-Pb dating of uranium mineralization in the Katanga Copperbelt: Constraints for the geodynamic context. Ore Geol. Rev. 2011, 40, 81–89. [Google Scholar] [CrossRef]
- Dahlkamp, F.J. The time related occurrence of uranium deposits. Miner. Depos. 1980, 15, 69–79. [Google Scholar] [CrossRef]
- Miller, R.G. The geochronology of uranium deposits in the Great Bear batholith, Northwest Territories. Can. J. Earth Sci. 1982, 19, 1428–1448. [Google Scholar] [CrossRef]
- Lewis, S.R. Geochemical and Isotopic Constraints for Uraninite Formation: Implications for Nuclear Forensic Analyses. Ph.D. Thesis, University of Notre Dame, Notre Dame, IN, USA, 2018. [Google Scholar]
- Deditius, A.P.; Utsunomiya, S.; Ewing, R.C. Fate of trace elements during alteration of uraninite in a hydrothermal vein-type U-deposit from Marshall Pass, Colorado, USA. Geochim. et Cosmochim. Acta 2007, 71, 4954–4973. [Google Scholar] [CrossRef]
- Hoeve, J.; Sibbald, T.I.I. On the Genesis of Rabbit Lake and Other Unconformity-type Uranium Deposits in Northern Saskatchewan, Canada. Econ. Geol. 1978, 73, 1450–1473. [Google Scholar] [CrossRef]
- Corey, A.S. Mineralogy and Petrology of the Uranium Deposits of Cane Springs Canyon, San Juan and Grand Counties, Utah; RME-128 (open-file report); US Atomic Energy Commission: Washington, DC, USA, 1959. [Google Scholar]
- Chenoweth, W.L. The Geology and Production History of the Uranium Deposits in the White Canyon Mining District, San Juan County, Utah; Utah Geological Survey: Salt Lake City, UT, USA, 1993. [Google Scholar]
- Trites, A.F.; Chew, I.I.I.R. Geology of the Happy Jack Mine White Canyon Area San Juan County, Utah; 1009-H (open-file report); United States. Government Printing Office: Washington, DC, USA, 1955.
- Belluci, J.J.; Simonetti, A.; Koeman, E.C.; Wallace, C.; Burns, P.C. A detailed geochemical investigation of post-nuclear detonation trinitite glass at high spatial resolution: Delineating anthropogenic vs. natural components. Chem. Geol. 2014, 365, 69–86. [Google Scholar] [CrossRef]
- Donohue, P.H.; Simonetti, A.; Neal, C.R. Chemical Characterisation of Natural Ilmenite: A Possible New Reference Material. Geostand. Geoanalytical Res. 2011, 36, 61–73. [Google Scholar] [CrossRef]
- Schurr, M.R.; Donohue, P.H.; Simonetti, A.; Dawson, E.L. Multi-element and lead isotope characterization of early nineteenth century pottery sherds from Native American and Euro-American sites. J. Archaeol. Sci. Rep. 2018, 20, 390–399. [Google Scholar] [CrossRef]
- Simonetti, A.; Neal, C. In-situ chemical, U-Pb dating, and Hf isotope investigation of megacrystic zircons, Malaita (Solomon Islands): Evidence for multi-stage alkaline magmatic activity beneath the Ontong Java Plateau. Earth Planet. Sci. Lett. 2010, 295, 251–261. [Google Scholar] [CrossRef]
- Pearce, N.J.G.; Perkins, W.T.; Westgate, J.A.; Gorton, M.P.; Jackson, S.E.; Neal, C.R.; Chenery, S.P. A compilation of new and published major and trace element data for NIST SRM 610 and NIST SRM 612 glass reference materials. Geostand Geoanal. Res. 1997, 21, 115–144. [Google Scholar] [CrossRef]
- Van Achterberg, E.; Ryan, C.; Jackson, S.; Griffin, W. Data reduction software for LA-ICP-MS. Laser Ablation ICP-MS. Earth Sci. 2001, 29, 239–243. [Google Scholar]
- Dustin, M.K.; Koeman, E.C.; Simonetti, A.; Torrano, Z.; Burns, P.C. Comparative investigation between in situ laser ablation versus bulk sample (solution mode) inductively coupled plasma mass spectrometry (ICP-MS) analysis of trinitite post-detonation materials. Appl. Spectrosc. 2016, 70, 1446–1455. [Google Scholar] [CrossRef]
- Jenner, G.A.; Longerich, H.P.; Jackson, S.E.; Fryer, B.J. ICP-MS—A powerful tool for high-precision trace-element analysis in Earth sciences: Evidence from analysis of selected U.S.G.S reference samples. Chem. Geol. 1990, 83, 133–148. [Google Scholar] [CrossRef]
- Manhes, G.; Minster, J.F.; Allègre, C.J. Comparative uranium-thorium-lead and rubidium-strontium study of the Saint Sèverin amphoterite: Consequences for early solar system chronology. Earth Planet Sci. Lett. 1978, 39, 14–24. [Google Scholar] [CrossRef]
- Simonetti, A.; Gariépy, C.; Banic, C.M.; Tanabe, R.; Wong, H.K. Pb isotopic investigation of aircraft-sampled emissions from the Horne smelter (Rouyn, Québec): Implications for atmospheric pollution in northeastern North America. Geochim. Cosmochim. Acta 2004, 68, 3285–3294. [Google Scholar] [CrossRef]
- Pollington, A.D.; Kinman, W.S.; Hanson, S.K.; Steiner, R.E.F. Polyatomic interferences on high precision uranium isotope ratio measurements by MC-ICP-MS: Applications to environmental sampling for nuclear safeguards. J. Radioanal. Nucl. Chem. 2015, 307, 2109–2115. [Google Scholar] [CrossRef]
- Crock, J.G.; Lichte, F.E.; Wildeman, T.R. The group separation of the rare-earth elements and yttrium from geologic materials by cation-exchange chromatography. Chem. Geol. 1984, 45, 149–163. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Vienna, Austria. Available online: https://www.r-project.org/ (accessed on 7 December 2017).
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R Package for Multivariate Analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef]
- Wickham, H. Ggplots2: Elegant Graphics for Data Analysis; Springer-Verlag: New York, NY, USA, 2016. [Google Scholar]
- GGally: Extension to “ggplot2”. Available online: https://cran.r-project.org/package=GGally (accessed on 7 January 2018).
- ggsci: Scientific Journal and Sci-Fi Themed Color Palettes for “ggplot2”. 2018. Available online: https://cran.r-project.org/package=ggsci (accessed on 7 January 2018).
- Varga, Z.; Katona, R.; Stefánka, Z.; Wallenius, M.; Mayer, K.; Nicholl, A. Determination of rare-earth elements in uranium-bearing materials by inductively coupled plasma mass spectrometry. Talanta 2010, 80, 1744–1749. [Google Scholar] [CrossRef]


| Sample | Mine Location | State | Country | IAEA Classification |
|---|---|---|---|---|
| 333 | Yancey County | North Carolina | USA | Intrusive non-granite-related |
| 334 | Mitchell County | North Carolina | USA | Intrusive non-granite-related |
| 338 | Mitchell County | North Carolina | USA | Intrusive non-granite-related |
| 340 | Bancroft | Ontario | Canada | Intrusive non-granite-related |
| 344 | Ruggles Mine | New Hampshire | USA | Intrusive non-granite-related |
| 348 | Foster Lake | Saskatchewan | Canada | Intrusive non-granite-related |
| 353 | Happy Jack, San Juan County | Utah | USA | Sandstone tabular |
| 423 | Great Bear Lake | Northern Territories | Canada | Metamorphite |
| 437 | Shinkolobwe | Katanga | D.R. Congo | Metamorphite |
| 511 | Rabbit Lake | Saskatchewan | Canada | Proterozoic unconformity |
| 516 | Australia | Northern Territories | Australia | Metamorphite |
| 522 | Billiken Lode, Jefferson County | Colorado | USA | Metamorphite |
| 531 | Marshall Pass, Gunnison County | Colorado | USA | Metamorphite hydrothermal vein |
| 564 | Jachymov | Karlovy Vary | Czech Republic | Intrusive granite-related |
| 604 | Australia | Northern Territories | Australia | Proterozoic unconformity |
| 623 | Marshall Pass | Colorado | USA | Metamorphite hydrothermal vein |
| 626 | Echo Mine, Great Bear Lake | Northern Territories | Canada | Metamorphite |
| 662 | Shinkolobwe | Katanga | D.R. Congo | Metamorphite |
| 809 | Marshall Pass | Colorado | USA | Metamorphite hydrothermal vein |
| 1232 | Big Indian Wash | Utah | USA | Sandstone |
| 1237 | Adair Mine, Cane Spring Canyon | Utah | USA | Sandstone |
| 1262 | Adair Mine, Cane Spring Canyon | Utah | USA | Sandstone |
| 1303 | Big Indian Wash | Utah | USA | Sandstone |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corcoran, L.; Simonetti, A.; Spano, T.L.; Lewis, S.R.; Dorais, C.; Simonetti, S.; Burns, P.C. Multivariate Analysis Based on Geochemical, Isotopic, and Mineralogical Compositions of Uranium-Rich Samples. Minerals 2019, 9, 537. https://doi.org/10.3390/min9090537
Corcoran L, Simonetti A, Spano TL, Lewis SR, Dorais C, Simonetti S, Burns PC. Multivariate Analysis Based on Geochemical, Isotopic, and Mineralogical Compositions of Uranium-Rich Samples. Minerals. 2019; 9(9):537. https://doi.org/10.3390/min9090537
Chicago/Turabian StyleCorcoran, Loretta, Antonio Simonetti, Tyler L. Spano, Stefanie R. Lewis, Corinne Dorais, Stefanie Simonetti, and Peter C. Burns. 2019. "Multivariate Analysis Based on Geochemical, Isotopic, and Mineralogical Compositions of Uranium-Rich Samples" Minerals 9, no. 9: 537. https://doi.org/10.3390/min9090537






