Mineralogy and Leachability of Natural Rocks–A Comparison to Electric Arc Furnace Slags
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Chemical Composition
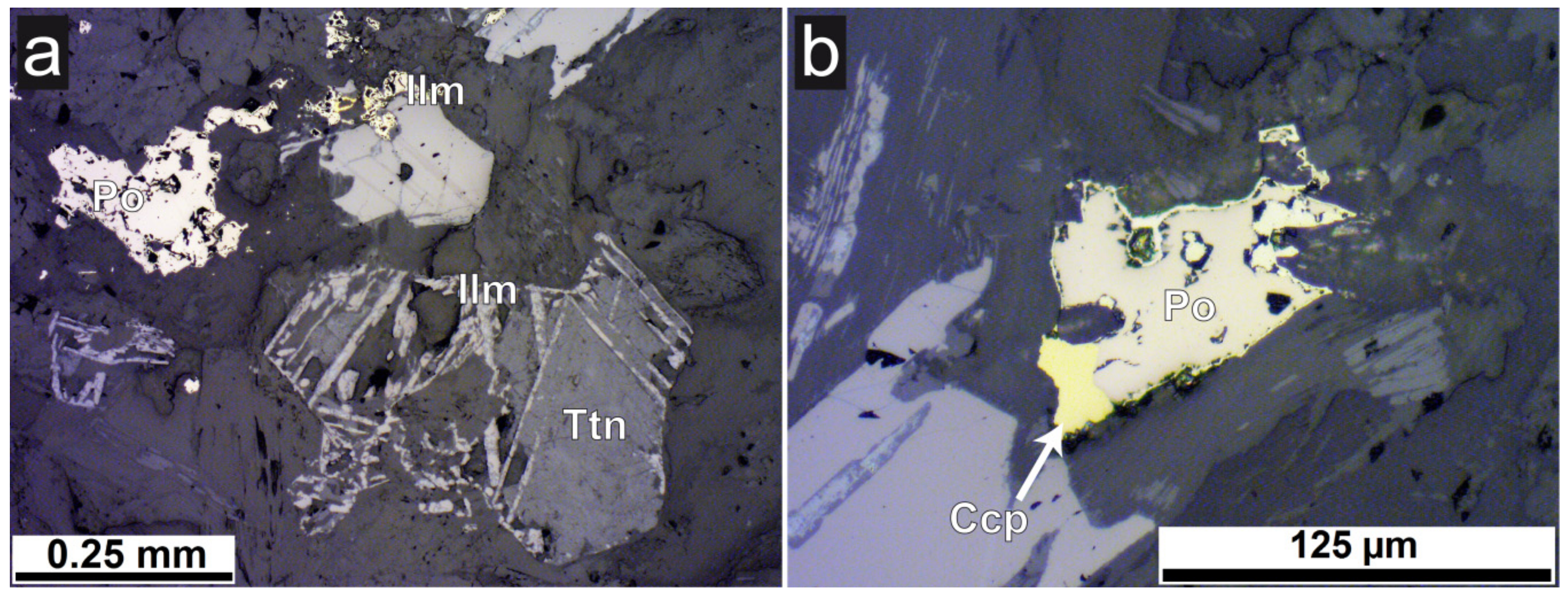
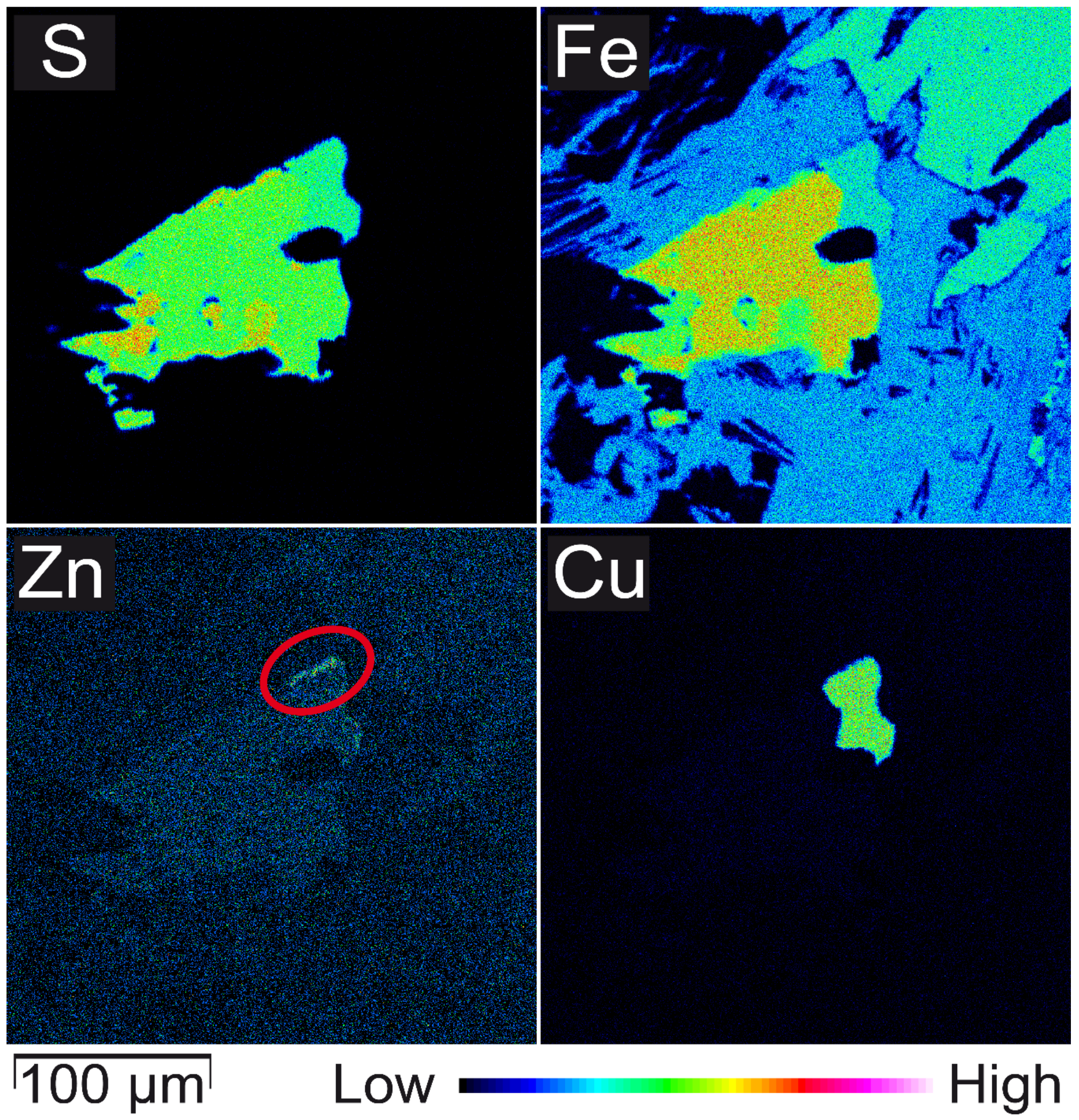
3.2. Mineralogical Composition
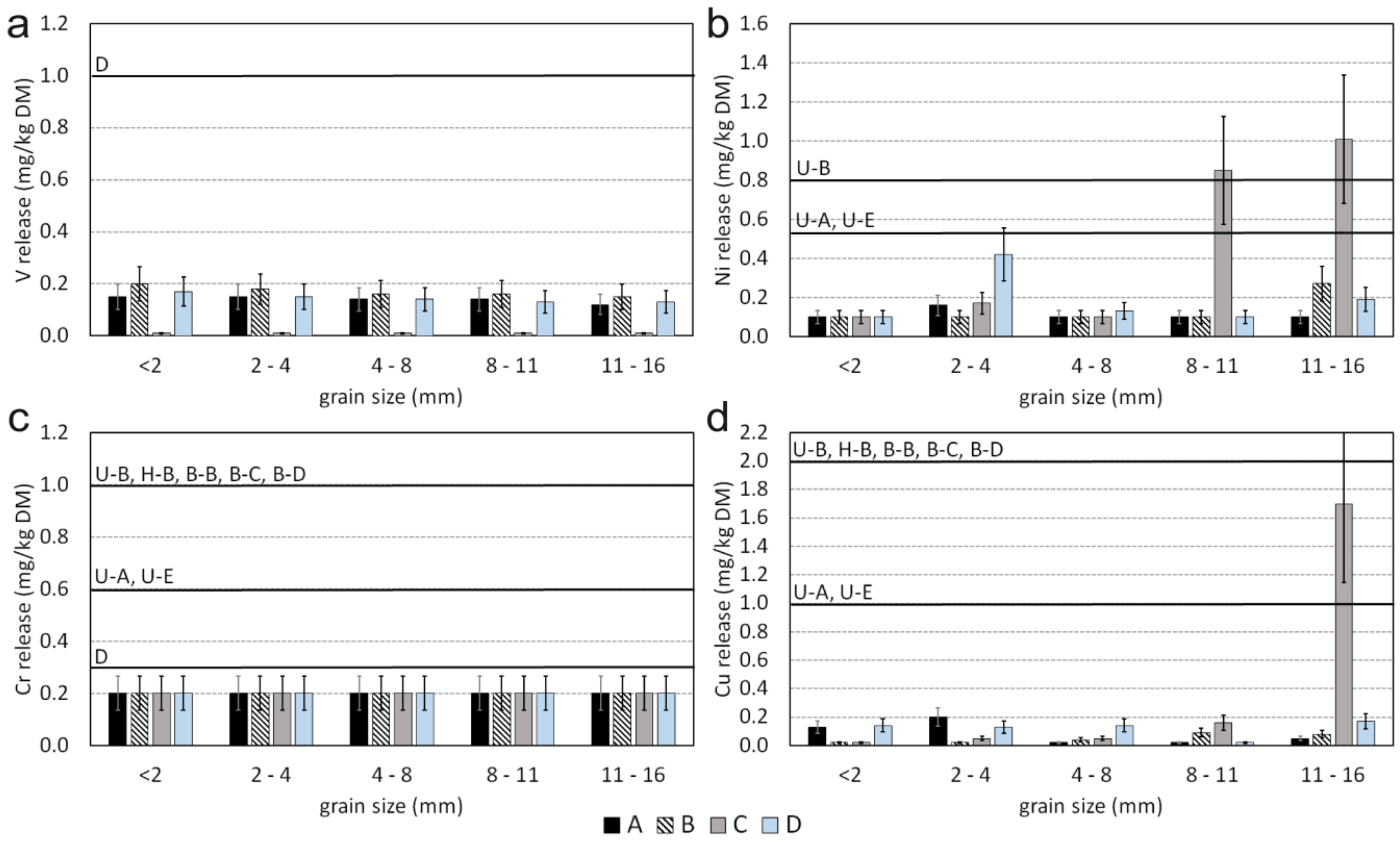
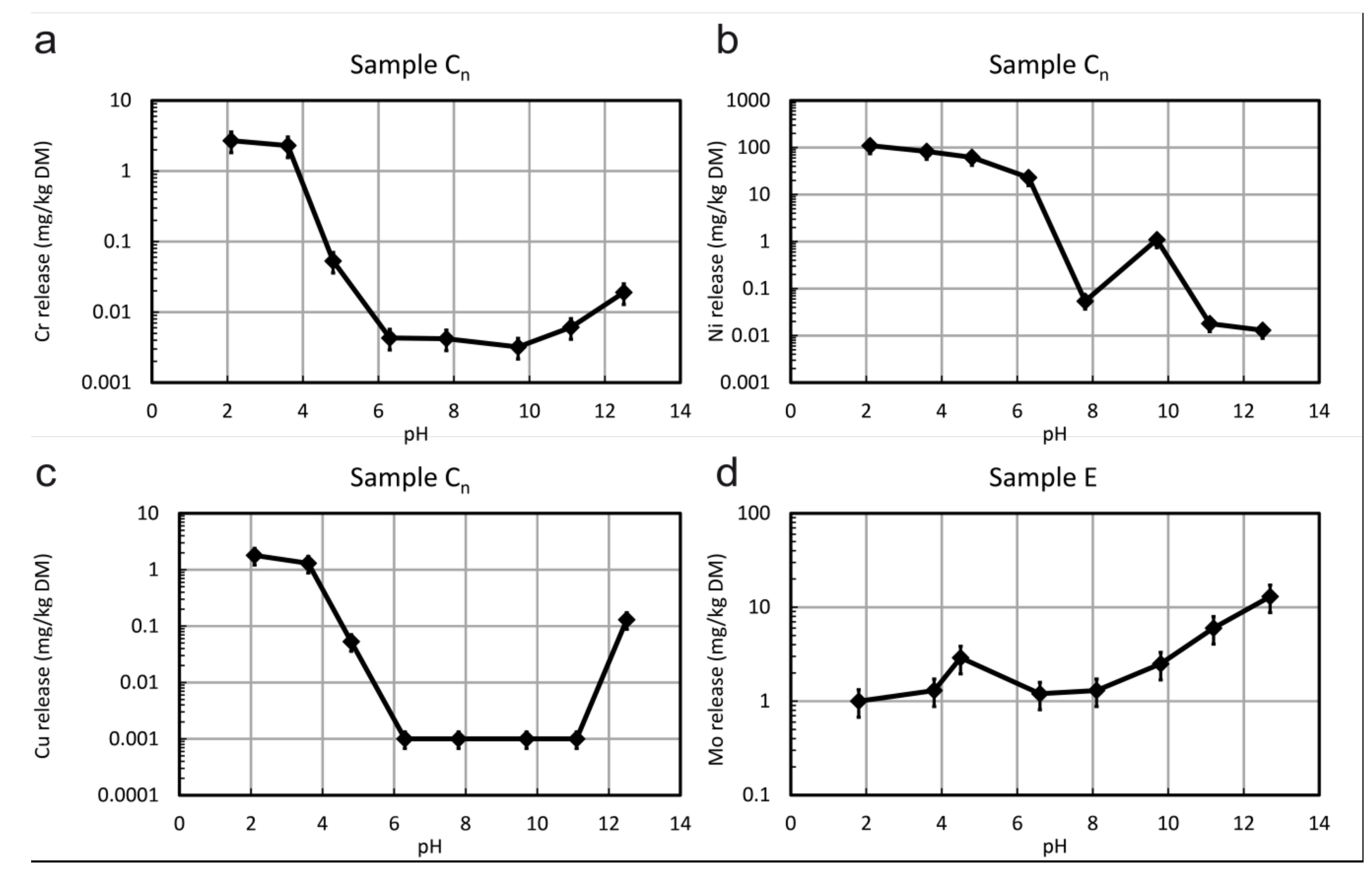
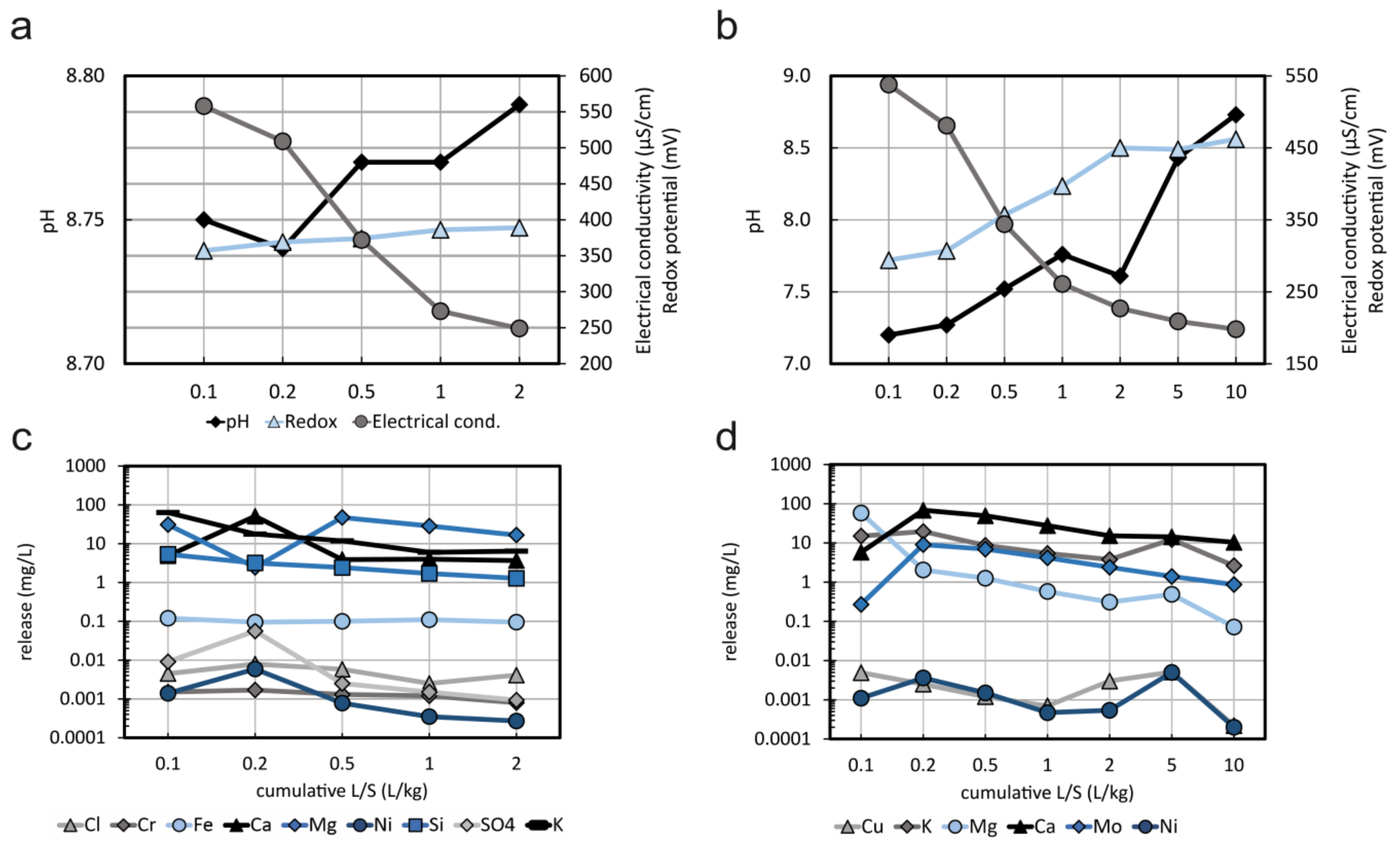
3.3. Leaching Behaviour
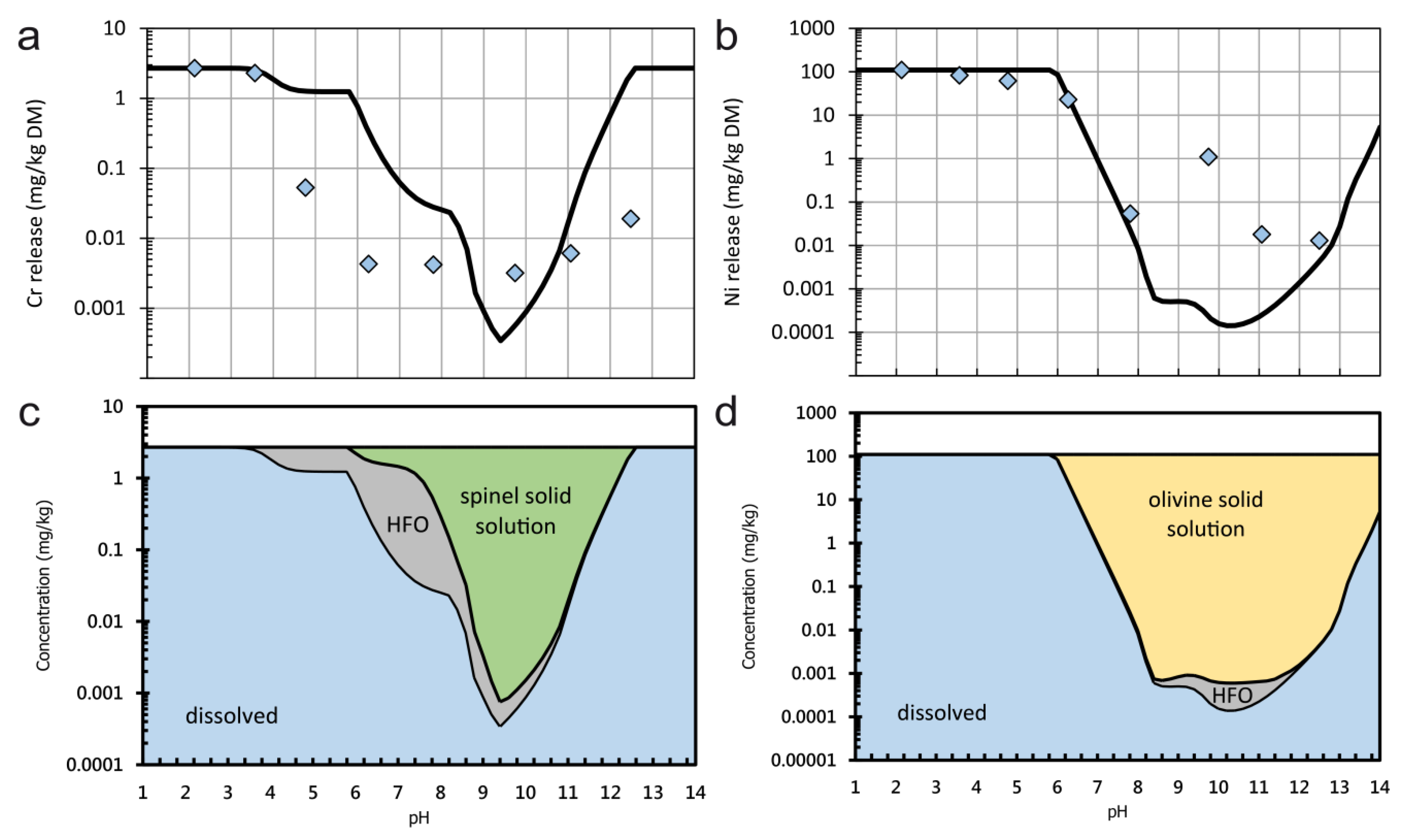
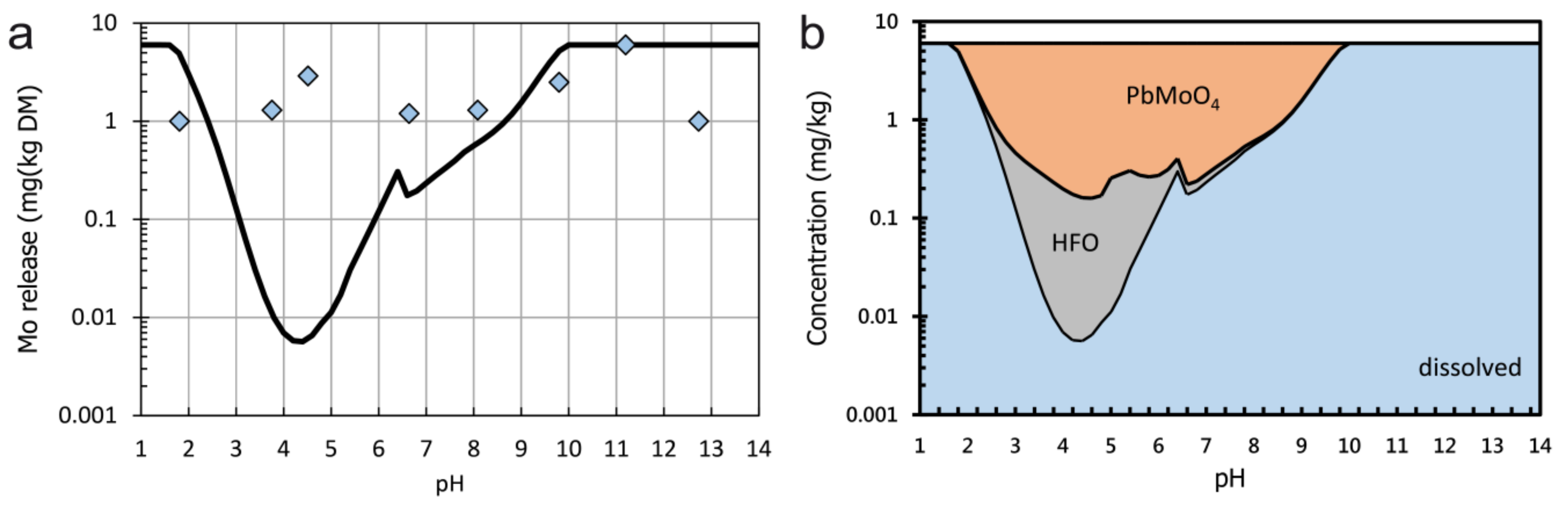
3.4. Hydrogeochemical Modelling
4. Discussion
5. Conclusions
- (1)
- Five rocks—four of them are quarried in Austria for commercial purposes—have been investigated regarding the leachability of potentially environmentally problematic elements (PEPE: Cr, Ni, V, and Mo etc.) depending on mineralogy. The motivation of this study was to get data for the comparison with synthetic materials (slags, recycled materials etc.) which can be used in similar commercial applications but for which limit values are defined in national regulations.
- (2)
- Only two samples have considerable higher total concentrations of Ni and Cr. Sample C, a meta-ultramafic rock (“serpentinite”), has up to 1033 mg/kg Cr which is bound in chromite and subordinately in silicates (chlorite). Higher Ni contents in the same sample are bound to olivine.
- (3)
- It is demonstrated that Cr leaching is very low which can be explained by the very low solubility of chromite; in contrast the leaching of Ni is higher which can be explained with the higher solubility of Ni in olivine and/or other Ni phases.
- (4)
- Sample E, a molybdenite-bearing orthogneiss from the Hohe Tauern area, contains up to 49,500 mg/kg Mo, which is bound to molybdenite. This mineralized rock is not used as a construction material but was included in order to investigate the behaviour of Mo during leaching processes. Only small amounts of Mo (<6mg/kg) are leached but the solubility of molybenite is modelled to be even lower. Thus the leaching of Mo must be controlled by other phases. Feldspars containing traces of Mo or secondary minerals like wulfenite could be alternative sources of Mo.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baurohstoffe. Available online: https://www.bmnt.gv.at/energie-bergbau/bergbau/mineralische-rohstoffe/Baurohstoffe.html (accessed on 25 February 2019).
- Material Flow Accounts. Non-Metallic Minerals, Domestic Material Consumption. Available online: http://appsso.eurostat.ec.europa.eu/nui/submitViewTableAction.do (accessed on 4 June 2019).
- Austrian Standards. ÖNORM B 3130: 2016 08 01. Gesteinskörnungen für Asphalte und Oberflächenbehandlungen für Straßen, Flugplätze und andere Verkehrsflächen-Regeln zur Umsetzung der ÖNORM EN 13043; Austrian Standards International: Vienna, Austria, 2016. [Google Scholar]
- Federal Ministry for Agriculture and Forestry, Environment and Waste Management. Recycling Baustoffverordnung. Available online: https://www.ris.bka.gv.at/GeltendeFassung.wxe?Abfrage=Bundesnormen&Gesetzesnummer=20009212 (accessed on 19 August 2019).
- Thalmann, F.; Schermann, O.; Schroll, E.; Hausberger, G. Geochemical Atlas of the Republic of Austria 1:1,000.000; Geological Survey of Austria: Vienna, Austria, 1989. [Google Scholar]
- Pohl, W. Economic Geology Principles and Practice-Metals, Minerals, Coal and Hydrocarbons-Introduction to Formation and Sustainable Exploitation of Mineral Deposits; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2001. [Google Scholar]
- Edgar, A.; Pizzolato, L.; Sheen, J. Flourine in igeneous rocks and minerals with emphasis on ultrapotassic mafic and ultramafic magmas and their mantle source regions. Miner. Mag. 1996, 60, 243–257. [Google Scholar] [CrossRef]
- European Committee for Standardization (CEN). DIN EN 1744–3. Tests for Chemical Properties of Aggregates—Part 3: Preparation of Eluates by Leaching of Aggregates; Beuth Verlag GmbH: Berlin, Germany, 2002. [Google Scholar]
- Deutsches Institut für Normung. DIN 19529:2015-12. Leaching of Solid Materials-Batch Test for the Examination of the Leaching Behaviour of Inorganic and Organic Substances at a Liquid to Solid Ratio of 2 l/kg; Beuth Verlag GmbH: Berlin, Germany, 2015. [Google Scholar]
- European Committee for Standardization (CEN). DIN EN 12457. Characterization of Waste-Leaching; Compliance Test for Leaching of Granular and Sludges; Beuth Verlag GmbH: Berlin, Germany, 2003. [Google Scholar]
- European Committee for Standardization (CEN). DIN EN 14405. Characterization of Waste-Leaching Behaviour Test-Up-Flow Percolation Test (Under Specified Conditions); Beuth Verlag GmbH: Berlin, Germany, 2017. [Google Scholar]
- Deutsches Institut für Normung. DIN 19528. Leaching of Solid Materials-Percolation Method for the Joint Examination of the Leaching Behaviour of Inorganic and Organic Substances; Beuth Verlag GmbH: Berlin, Germany, 2009. [Google Scholar]
- European Committee for Standardization. DIN EN 14429. Characterization of Waste-Leaching Behaviour Test-Influence of ph on Leaching with Initial Acid/Base Addition; Beuth Verlag GmbH: Berlin, Germany, 2015. [Google Scholar]
- Potysz, A.; van Hullebusch, E.; Kierczak, J. Perspectives regarding the use of metallurgical slags as secondary metal resources–A review of bioleaching approaches. J. Environ. Manag. 2018, 219, 138–152. [Google Scholar] [CrossRef] [PubMed]
- Forschungsgesellschaft für Straßen- und Verkehrswesen (FGSV). Technische Lieferbedingungen für Gesteinskörnungen im Straßenbau: TL Gestein-Stb 04, Köln; FGSV-Verlag: Cologne, Germany, 2008. [Google Scholar]
- Van Houdt, J.; Wolf, E.; Duzijn, R. Composition and leaching characteristics of road construction materials. In Waste Materials in Construction, Proceedings of the International Conference on Environmental Implications of Construction with Waste Materials, Maastricht, The Netherlands, 10–14 November 1991; Elsevier: New York, NY, USA.
- Tossavainen, M.; Forssberg, E. The potential leachability from natural road construction materials. Sci. Total Env. 1999, 239, 31–47. [Google Scholar] [CrossRef]
- Rivera, J.; Reich, M.; Schoenberg, R.; González-Jiménez, J.; Barra, F.; Aiglsperger, T.; Proenza, J.; Carretier, S. Platinum-group element and gold enrichment in soils monitored by chromium stable isotopes during weathering of ultramafic rocks. Chem. Geol. 2018, 499, 84–99. [Google Scholar] [CrossRef]
- Ratié, G.; Jouvin, D.; Garnier, J.; Rouxel, O.; Miska, S.; Guimaraes, E.; Cruz Viera, L.; Sivry, Y.; Zelano, I.; Montarges-Pelletier, E.; et al. Nickel isotope fractionation during tropical weathering of ultramafic rocks. Chem. Geol. 2015, 402, 68–76. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, J.; Li, J.; Wei, G.; Zeng, T.; Li, L.; Zhang, L.; Deng, W.; Xie, L.; Liu, Z. Fe (hydro)oxide controls Mo isotope fractionation during the weathering of granite. Geochim. Cosmochim. Acta 2018, 22, 1–17. [Google Scholar] [CrossRef]
- European Committee for Standardization (CEN). DIN EN 13656. Characterization of Waste—Microwave Assisted Digestion with Hydrofluoric (HF), Nitric (HNO₃) and Hydrochloric (HCl) acid Mixture for Subsequent Determination of Elements in Waste; Beuth Verlag GmbH: Berlin, Germany, 2002. [Google Scholar]
- European Committee for Standardization (CEN). DIN EN ISO 17294-2. Water Quality-Application of Inductively Coupled Plasma Mass Spectrometry (ICP-MS)—Part 2: Determination of 62 Elements; Beuth Verlag GmbH: Berlin, Germany, 2017. [Google Scholar]
- European Committee for Standardization (CEN). DIN EN ISO 11885. Water Quality—Determination of Selected Elements by Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES); Beuth Verlag GmbH: Berlin, Germany, 2007. [Google Scholar]
- European Committee for Standardization. EN ISO 10304-1. Water Quality—Determination of Dissolved Anions by Liquid Chromatography of Ions—Part 1: Determination of Bromide, Chloride, Fluoride, Nitrate, Nitrite, Phosphate and Sulfate; Beuth Verlag GmbH: Berlin, Germany, 2009. [Google Scholar]
- Deutsches Institut für Normung (DIN). DIN 38405-24. German Standard Methods for the Examination of Water, Waste Water and Sludge; Anions (group D); Photometric Determination of Chromium(VI) Using 1,5-Diphenylcarbonohydrazide (D 24); Beuth Verlag GmbH: Berlin, Germany, 1987. [Google Scholar]
- European Committee on Standardization (CEN). DIN EN 1484. Water Analysis—Guidelines for the Determination of Total Organic Carbon (TOC) and Dissolved Organic Carbon (DOC); Beuth Verlag GmbH: Berlin, Germany, 1997. [Google Scholar]
- Meeussen, J. ORCHESTRA: An object-oriented framework for implementing chemical equilibrium models. Env. Sci. Technol. 2003, 37, 1175–1182. [Google Scholar] [CrossRef] [PubMed]
- US-EPA. National Exposure Research Laboratory, Ecosystems Research Division. In MINTEQA2/PRODEFA2, A Geochemcial Assessment Model for Environmental Systems: User Manual Supplement for Version 4.0; HydroGeoLogic, Inc.: Herndon, VA, USA, 1999. [Google Scholar]
- Delany, J.; Lundeen, S. The LNLL Thermodynamic Database; Lawrence Livermore National Laboratory: Livermore, CA, USA, 1990. [Google Scholar]
- Blanc, P.; Lassin, A.; Piantone, P.; Azaroual, M.; Jacquemet, N.; Fabbri, A.; Gaucher, E.C. Thermoddem: A geochemical database focused on low temperature water/rock interactions and waste materials. Appl. Geochem. 2012, 27, 2107–2116. [Google Scholar] [CrossRef]
- Loncnar, M.; van der Sloot, H.; Mladenovič, M.; Zupančič, L.; Kobal, L.; Bukovec, P. Study of the leaching behaviour of ladle slags by means of leaching tests combined with geochemical modelling and mineralogical investigations. J. Hazard. Mater. 2016, 317, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Rudnick, R.; Gao, S. Composition of the continental crust. In Treatise on Geochemistry, 3rd ed.; Elsevier-Pergamon: Amsterdam, The Netherlands, 2003; pp. 1–64. [Google Scholar]
- Pan, Y.; Fleet, M. Mineral chemistry and geochemistry of vanadian silicates in the Hemlo gold deposit, Ontario, Canada. Contrib. Mineral. Petr. 1992, 109, 511–525. [Google Scholar] [CrossRef]
- Dalou, C.; Koga, K.; Shimzu, N.; Boulon, J.; Devidal, J. Experimental determination of F and Cl partitioning between lherzolite and basaltic melt. Contrib. Mineral. Petr. 2012, 163, 591–609. [Google Scholar] [CrossRef]
- Steiner, T. Aplite hosted Reichenspitz-type molybdenite molybdenite mineralization in the Central Tauern Window, Salzburg/Tyrol. Master Thesis, Montanuniversität Leoben, Leoben, Styria, Austria, 2018. [Google Scholar]
- Greaney, A.; Rudnick, N.; Gaschnig, R.; Wahlen, J.; Luais, B.; Clemens, J. Geochemistry of molybdenum in the continental crust. Geochim. Cosmochim. Acta 2018, 238, 36–54. [Google Scholar] [CrossRef]
- Dijkstra, J.; van der Sloot, H.; Comans, R. The leaching of major and trace elements from MSWI bottom ash as a function of pH and time. Appl. Geochem. 2006, 21, 335–351. [Google Scholar] [CrossRef]
- Dijkstra, J.; Meeussen, J.; van der Sloot, H.; Comans, R. A consistent geochemical modelling approach for the leaching and reactive transport of major and trace elements in MSWI bottom ash. Appl. Geochem. 2008, 23, 1544–1562. [Google Scholar] [CrossRef]
- Villanova-de-Benavent, C.; Proenza, J.A.; Galí, S.; García-Casco, A.; Tauler, E.; Lewis, J.F.; Longo, F. Garnierites and garnierites: Textures, mineralogy and geochemistry of garnierites in the Falcondo Ni-laterite deposit. Ore. Geol. Rev. 2014, 59, 91–109. [Google Scholar] [CrossRef]
- Arnórsson, S.; Óskarsson, N. Molybdenum and tungsten in volcanic rocks and in surface and <100 °C ground waters in Iceland. Geochim. Cosmochim. Acta 2007, 71, 284–304. [Google Scholar]
- Neuhold, S.; Vollprecht, D.; Berger, M.; Raith, J.G.; Schüler, S.; Mudersbach, D. Auslaugverhalten von Stahlwerksschlacken und natürlichen Gesteinen—Ein Vergleich. In Schlacken-Symposium der Max Aicher Unternehmensgruppe; Max Aicher Unternehmensgruppe: Meitingen, Germany, 2018; pp. 179–189. [Google Scholar]
- Proctor, D.; Fehling, K.; Shay, E.; Wittenborn, J.; Green, J.; Avent, C.; Bigham, R.; Connolly, M.; Lee, B.; Shepker, T.; et al. Physical and chemical characteristics of blast furnace, basic oxygen furnace and electric arc furnace steel industry slags. Environ. Sci. Technol. 2000, 34, 1576–1582. [Google Scholar] [CrossRef]
- Wu, X.; Wang, P.; Li, L.; Wu, Z.; Chen, R. Distribution and enrichment of phosphorous in solidified BOF steelmaking slag. Ironmak. Steelmak. 2011, 38, 185–188. [Google Scholar] [CrossRef]
- Aldrian, A.; Raith, J.G.; Höllen, D.; Pomberger, R. Influence of chormium containing spinels in electric arc furnace slag on the leaching behaviour. J. Solid Waste Technol. Manag. 2015, 41, 357–365. [Google Scholar] [CrossRef]
- Höllen, D.; Neuhold, S.; Mudersbach, D.; Schüler, S.; Sommerauer, H.; Griessacher, T.; Dijkstra, J.; van Zomeren, A.; Presoly, P.; Schenk, J.; et al. Möglichkeiten und Grenzen von Modellen zur Vorhersage der Auslaugbarkeit von Stahlwerksschlacken. In Proceedings of the Mineralische Nebenprodukte und Abfälle, Berlin, Germany, 12–13 June 2017; TK Verlag: Neuruppin, Germany, 2017; pp. 205–222. [Google Scholar]
- Deer, W.; Howie, R.; Zussman, J. An Introduction to the Rock-Forming Minerals; The Mineralogical Society: Twickenham, UK, 2013. [Google Scholar]
- Okrusch, M.; Matthes, S. Mineralogie: Eine Einführung in die spezielle Mineralogie, Petrologie und Lagerstättenkunde, 8th ed.; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Thalhammer, O.; Ebner, F.; Horkel, K.; Mali, H. Der Ultramafit-Komplex von Kraubath. In Proceedings of the PANGEO 2010, Leoben, Austria, 15–19 September 2010. [Google Scholar]
- Kindness, A.; Lachowski, E.; Minocha, A.; Glasser, F. Immobilisation and fixation of molybdenum(VI) by Portland cement. Waste Manag. 1994, 14, 97–102. [Google Scholar] [CrossRef]



| Element group | Element | Unit | A | B | C | Cn | D | E | Limit value range | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Particle size (mm) | <2 | 2–4 | 4–8 | 8–11 | 11–16 | <2 | 2–4 | 4–8 | 8–11 | 11–16 | <2 | 2–4 | 4–8 | 8–11 | 11–16 | <2 | 2–4 | 4–8 | 8–11 | 11–16 | |||||
| Major Elements | Na | g/kg DM | 8 | 8 | 7 | 7 | 4 | 12 | 13 | 12 | 12 | 11 | 0.7 | 0.5 | 0.7 | 0.9 | 1.3 | 0.5 | 14 | 14 | 14 | 14 | 13 | 37 | - |
| Mg | 24 | 13 | 23 | 24 | 21 | 19 | 19 | 15 | 18 | 24 | 77 | 61 | 120 | 123 | 139 | 229 | 27 | 24 | 27 | 33 | 24 | 0.4 | - | ||
| Al | 42 | 43 | 44 | 45 | 38 | 57 | 59 | 47 | 58 | 61 | 1.3 | 0.6 | 1.2 | 1.2 | 6.3 | 1.9 | 51 | 45 | 50 | 56 | 53 | 31 | - | ||
| Si | 125 | 200 | 155 | 172 | 91 | 191 | 209 | 154 | 202 | 182 | 127 | 137 | 123 | 112 | 90 | 159 | 147 | 150 | 148 | 165 | 141 | 263 | - | ||
| K | 5 | 9 | 9 | 10 | 9 | 11 | 11 | 10 | 12 | 13 | 0.08 | 0.09 | 0.03 | 0.03 | 0.95 | <0.01 | 3 | 3 | 2 | 3 | 7 | 11 | - | ||
| Ca | 82 | 73 | 102 | 118 | 161 | 13 | 14 | 15 | 8 | 18 | <0.3 | <0.3 | 0.3 | <0.3 | 4.5 | 7 | 60 | 50 | 58 | 59 | 65 | 0.9 | - | ||
| Ti | 5 | 3 | 4 | 4 | 3 | 9 | 10 | 9 | 9 | 10 | 0.2 | 0.1 | 0.1 | 0.1 | 0.3 | 0.07 | 8 | 8 | 8 | 9 | 7 | 0.2 | - | ||
| Fe | 47 | 27 | 41 | 35 | 30 | 58 | 58 | 55 | 57 | 63 | 43 | 49 | 48 | 43 | 41 | 55 | 76 | 72 | 79 | 84 | 63 | 2 | - | ||
| Trace Elements | Li | mg/kg DM | 20 | 25 | 31 | 31 | 27 | 32 | 32 | 33 | 34 | 37 | 0.5 | <0.3 | 0.3 | 0.4 | 3.3 | 1.4 | 13 | 12 | 13 | 15 | 21 | 4 | - |
| P | 410 | 542 | 635 | 572 | 648 | 1056 | 1050 | 951 | 1046 | 1019 | 19 | 19 | 5 | 5 | 31 | 33 | 404 | 402 | 421 | 462 | 457 | 100 | - | ||
| V | 23 | 11 | 28 | 23 | 45 | 59 | 55 | 53 | 54 | 81 | <1 | <1 | 8 | 5 | 20 | 32 | 266 | 256 | 277 | 299 | 194 | 4 | - | ||
| Cr | 75 | 60 | 73 | 98 | 56 | 46 | 45 | 55 | 53 | 55 | 582 | 548 | 946 | 755 | 1033 | 780 | 107 | 93 | 100 | 140 | 124 | <3 | 90–2500 | ||
| Mn | 897 | 725 | 1116 | 854 | 872 | 850 | 871 | 832 | 798 | 1049 | 752 | 764 | 775 | 679 | 650 | 960 | 1375 | 1292 | 1416 | 1559 | 1312 | 77 | - | ||
| Co | 24 | 13 | 16 | 17 | 12 | 21 | 21 | 23 | 22 | 25 | 83 | 89 | 95 | 85 | 78 | 100 | 42 | 40 | 43 | 53 | 32 | 1 | 50 | ||
| Ni | 39 | 47 | 56 | 62 | 22 | 19 | 20 | 25 | 24 | 23 | 1478 | 1718 | 1780 | 1563 | 1395 | 2580 | 63 | 58 | 62 | 85 | 83 | <3 | 60–500 | ||
| Cu | 36 | 10 | 12 | 18 | 8 | 16 | 13 | 19 | 15 | 8 | 7 | 11 | 9 | 6 | 6 | 40 | 77 | 58 | 60 | 50 | 49 | 7 | 90–500 | ||
| Zn | 103 | 93 | 55 | 75 | 62 | 172 | 158 | 159 | 132 | 126 | 22 | 23 | 25 | 21 | 31 | 28 | 98 | 96 | 99 | 110 | 93 | 6 | 450–1000 | ||
| As | 9 | 16 | 22 | 16 | 19 | 10 | 12 | 11 | 10 | 8 | 5 | 4 | <3 | <3 | 5 | 8 | 10 | 10 | 10 | 12 | 9 | <3 | 50–200 | ||
| Sr | 165 | 238 | 246 | 257 | 295 | 161 | 176 | 142 | 138 | 159 | 8 | 5 | 4 | 4 | 11 | 2 | 144 | 133 | 141 | 150 | 209 | 16 | - | ||
| Mo | <3 | <3 | <3 | <3 | <3 | <3 | <3 | <3 | <3 | <3 | <3 | <3 | <3 | <3 | <3 | 3 | <3 | <3 | <3 | <3 | <3 | 49500 | 50 | ||
| Cd | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | 0.5 | <0.5 | 0.5 | 0.7 | 0.5 | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 | 0.2 | <0.5 | <0.5 | <0.5 | 0.7 | <0.5 | <0.5 | 1.1–4 | ||
| Ba | 165 | 305 | 307 | 327 | 251 | 612 | 575 | 489 | 606 | 674 | 16 | <1 | 5 | 3 | 32 | 3 | 66 | 67 | 66 | 76 | 225 | 150 | - | ||
| W | 0.6 | 0.5 | 0.7 | 0.9 | 0.6 | 0.6 | 0.7 | 0.7 | 0.8 | 0.6 | <0.3 | <0.3 | <0.3 | <0.3 | 0.4 | 8 | <0.5 | <0.5 | <0.5 | <0.5 | 1.3 | 10 | 450 | ||
| Hg | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | n.a. | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | 0.7–2 | ||
| Tl | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | <0.3 | 0.7 | <0.3 | <0.3 | <0.3 | <0.3 | 0.4 | <0.3 | 50 | ||
| Pb | 15 | 19 | 14 | 20 | 17 | 18 | 13 | 16 | 16 | 16 | <0.5 | 0.5 | <0.5 | 0.7 | 1.0 | <0.03 | 4 | 3 | 4 | 26 | 13 | 13 | 150–500 | ||
| Sample | Stoichometry | A | B | C | D | ||||
|---|---|---|---|---|---|---|---|---|---|
| Particle Size (mm) | <2 | 11–16 | <2 | 11–16 | <2 | 11–16 | <2 | 11–16 | |
| Quartz (Qz) | SiO2 | 38 | 17 | 24 | 21 | n.i. | 5 | 7 | 8 |
| Albite (Ab) | NaAlSi3O8 | 20 | 5 | 32 | 22 | n.i. | n.i. | 24 | 27 |
| Calcite (Cc) | CaCO3 | 13 | 43 | 3 | 0 | n.i. | n.i. | n.i. | n.i. |
| Illite (Ilt) / Muscovite (Ms) | (K,H3O)Al3Si3O10(OH)2 | 9 | 14 | 11 | 14 | n.i. | n.i. | n.i. | n.i. |
| Annite (Ann) | KFe3AlSi3O10(OH,F)2 | 7 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| Chlorite (Chl) | (Mg,Fe2+)5Al(Si3Al)O10(OH)8 | 6 | 4 | 23 | 17 | 28 | 26 | 18 | 12 |
| Diopside (Di) | CaMgSi2O6 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | 12 | 9 |
| Ankerite (Ank) | Ca(Fe2+,Mg)(CO3)2 | 6 | n.i. | n.i. | 6 | n.i. | n.i. | n.i. | n.i. |
| Dolomite (Dol) | CaMg(CO3)2 | n.i. | 8 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| Epidote (Ep) | Ca2(Fe,Al)Al2Si3O12(OH) | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | 9 |
| Magnesite (Mgs) | MgCO3 | n.i. | n.i. | n.i. | n.i. | 6 | n.i. | 4 | n.i. |
| Actinolite (Act) | Ca2(Mg2.5–4.5,Fe2+0.5–2.5)Si8O22(OH)2 | 2 | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. | n.i. |
| Hornblende (Hbl) | Ca2(Na,K)0.5–1(Mg,Fe)3–4(Fe3+, Al)1–2Al2Si6O22(O,OH,F)2 | n.i. | 10 | 5 | 20 | n.i. | n.i. | 34 | 36 |
| Tremolite (Tr) | Ca2Mg5Si8O22(OH)2 | n.i. | n.i. | n.i. | n.i. | n.i. | 5 | n.i. | n.i. |
| Serpentine (Srp) | Mg6Si4O10(OH)8 | n.i. | n.i. | n.i. | n.i. | 44 | 43 | n.i. | n.i. |
| Talc (Tlc) | Mg3Si4O10(OH)2 | n.i. | n.i. | n.i. | n.i. | 9 | 8 | n.i. | n.i. |
| Olivine (Ol) | (Fe,Mg)2SiO4 | n.i. | n.i. | n.i. | n.i. | 13 | 12 | n.i. | n.i. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vollprecht, D.; Berger, M.; Altenburger-Junker, I.; Neuhold, S.; Sedlazeck, K.P.; Aldrian, A.; Dijkstra, J.J.; van Zomeren, A.; Raith, J.G. Mineralogy and Leachability of Natural Rocks–A Comparison to Electric Arc Furnace Slags. Minerals 2019, 9, 501. https://doi.org/10.3390/min9080501
Vollprecht D, Berger M, Altenburger-Junker I, Neuhold S, Sedlazeck KP, Aldrian A, Dijkstra JJ, van Zomeren A, Raith JG. Mineralogy and Leachability of Natural Rocks–A Comparison to Electric Arc Furnace Slags. Minerals. 2019; 9(8):501. https://doi.org/10.3390/min9080501
Chicago/Turabian StyleVollprecht, Daniel, Max Berger, Ingrid Altenburger-Junker, Simone Neuhold, Klaus Philipp Sedlazeck, Alexia Aldrian, Joris J. Dijkstra, André van Zomeren, and Johann G. Raith. 2019. "Mineralogy and Leachability of Natural Rocks–A Comparison to Electric Arc Furnace Slags" Minerals 9, no. 8: 501. https://doi.org/10.3390/min9080501
APA StyleVollprecht, D., Berger, M., Altenburger-Junker, I., Neuhold, S., Sedlazeck, K. P., Aldrian, A., Dijkstra, J. J., van Zomeren, A., & Raith, J. G. (2019). Mineralogy and Leachability of Natural Rocks–A Comparison to Electric Arc Furnace Slags. Minerals, 9(8), 501. https://doi.org/10.3390/min9080501






