Experiments on Rare-Earth Element Extractions from Umber Ores for Optimizing the Grinding Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Experiments
2.2.1. Sample Preparation
2.2.2. Whole Rock Chemical Analyses
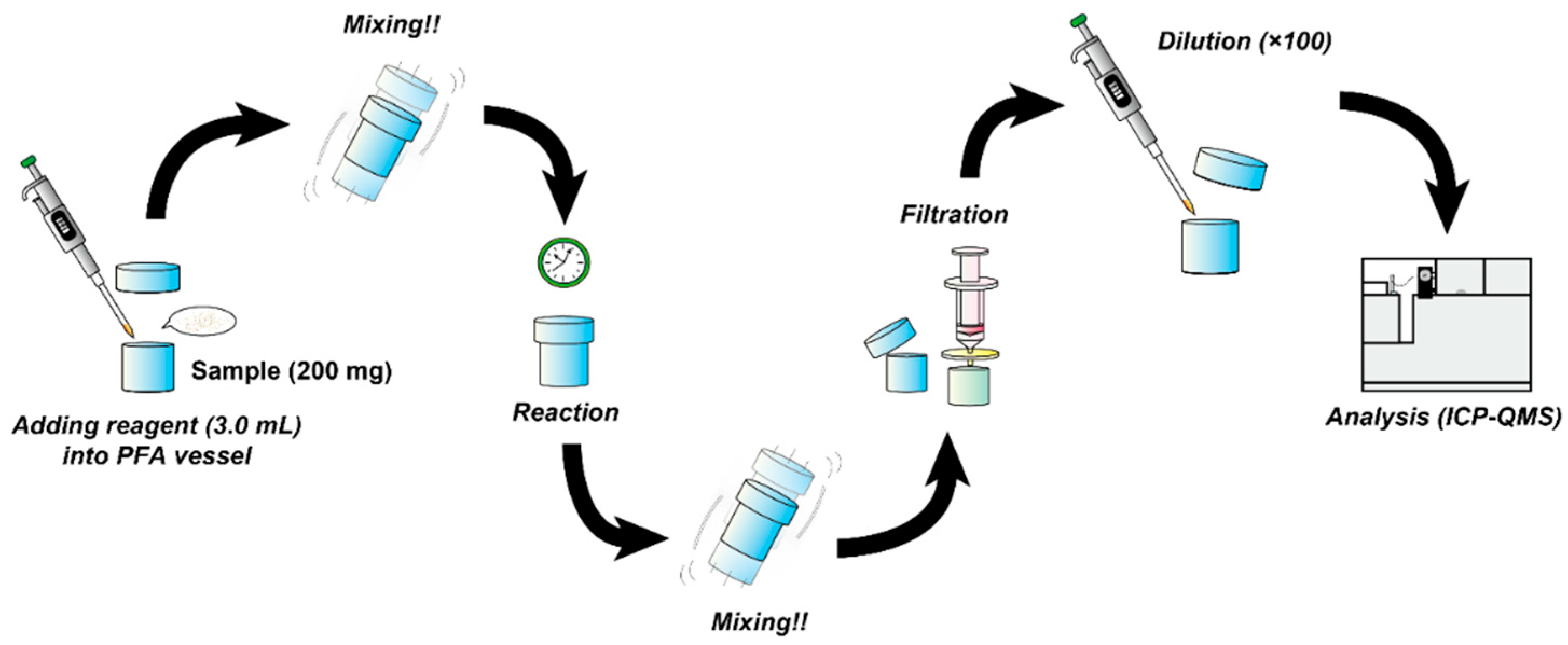
2.2.3. Chemical Leaching Experiments
3. Results
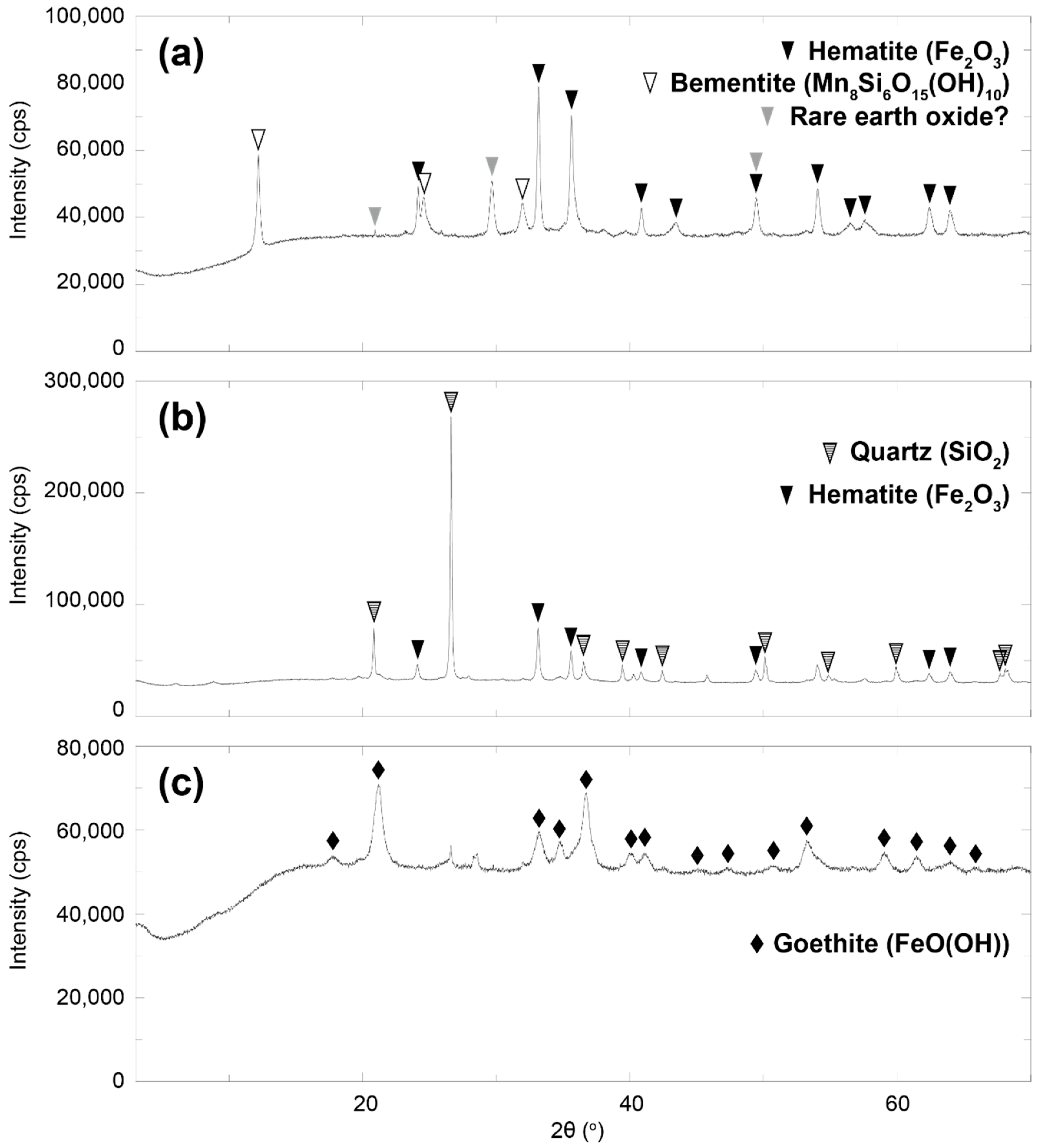
3.1. Chemical Composition of Umber Samples
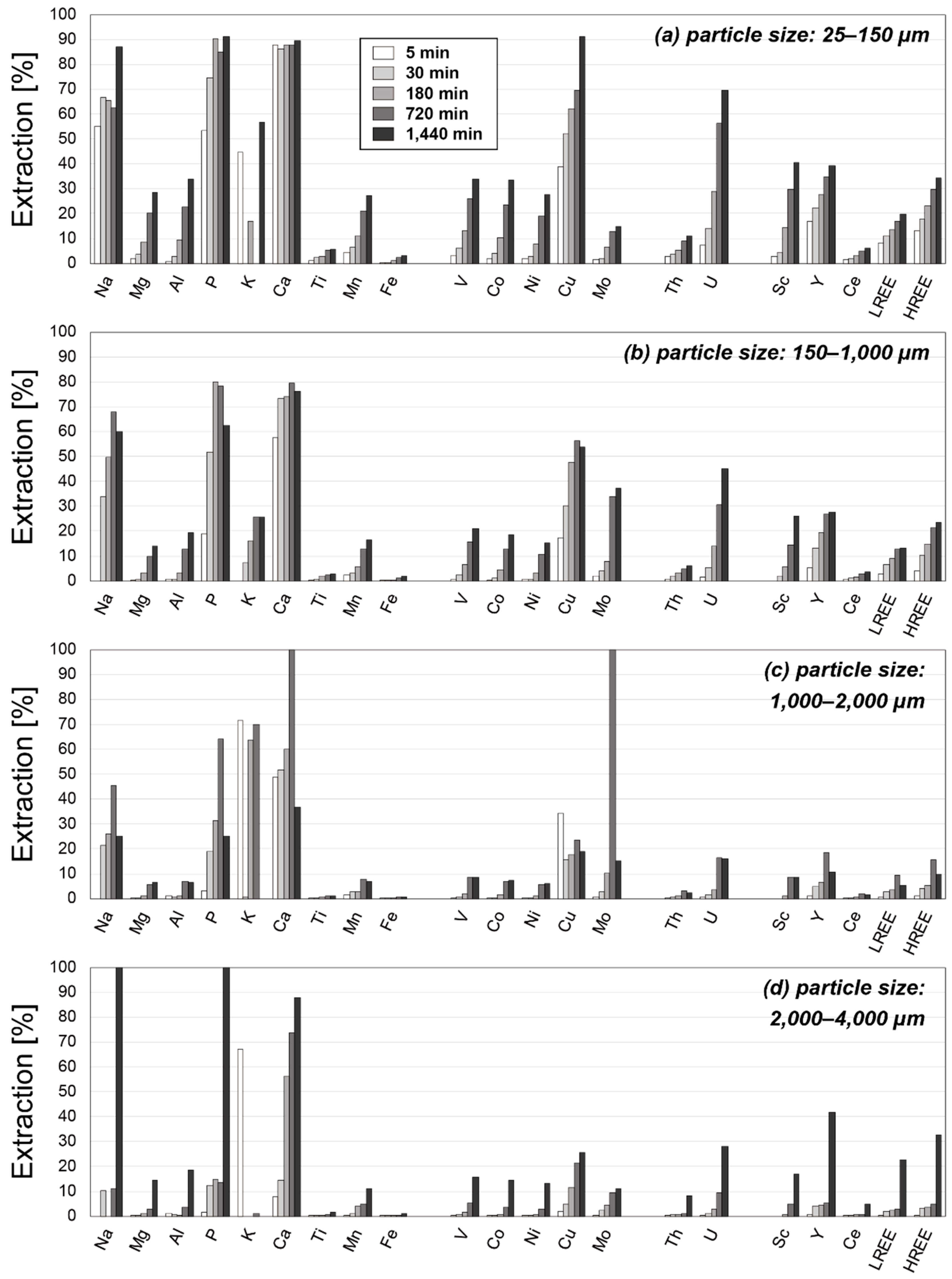
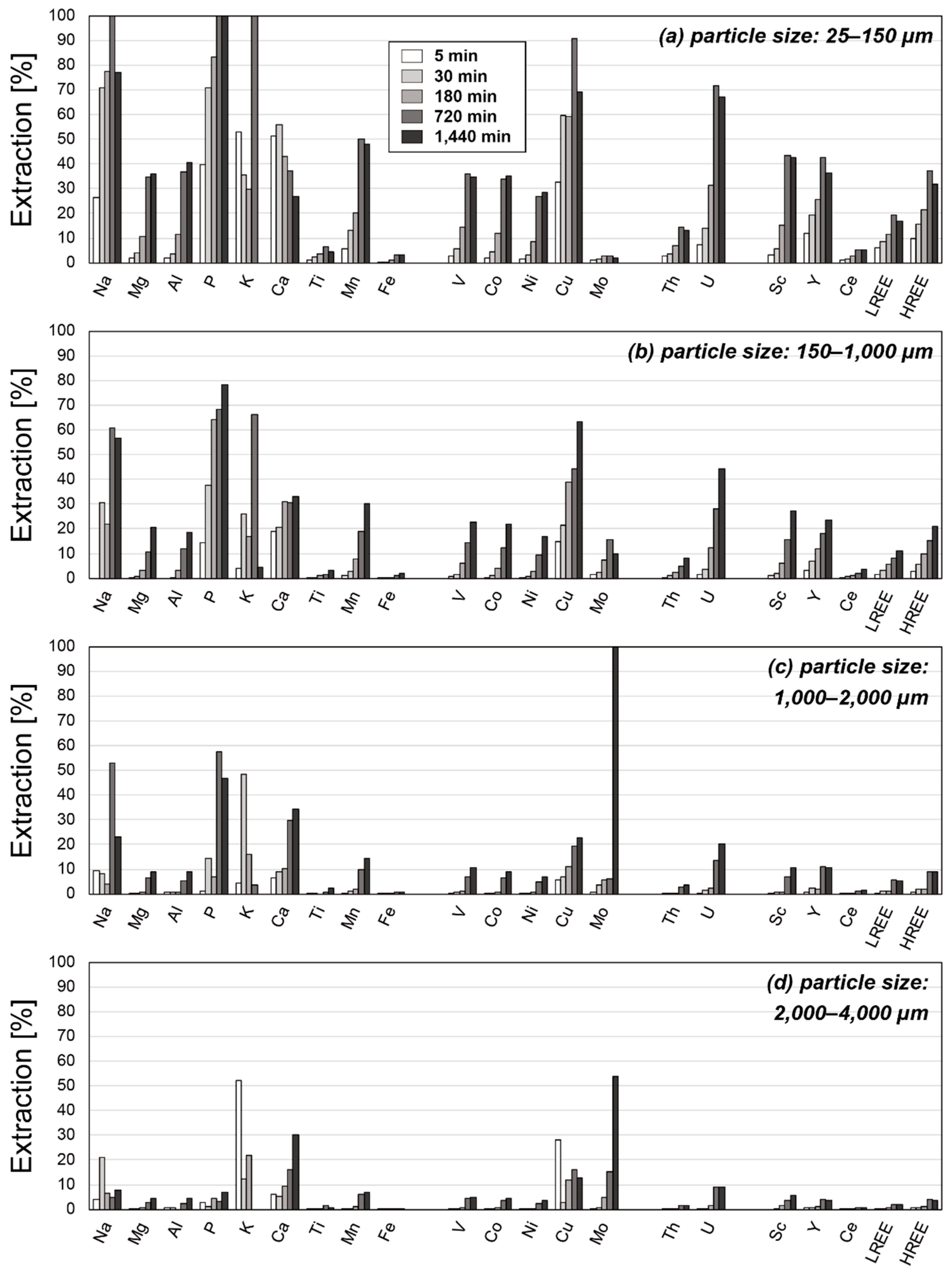
3.2. Results of Leaching Experiments
4. Discussion
4.1. Host Minerals of REE
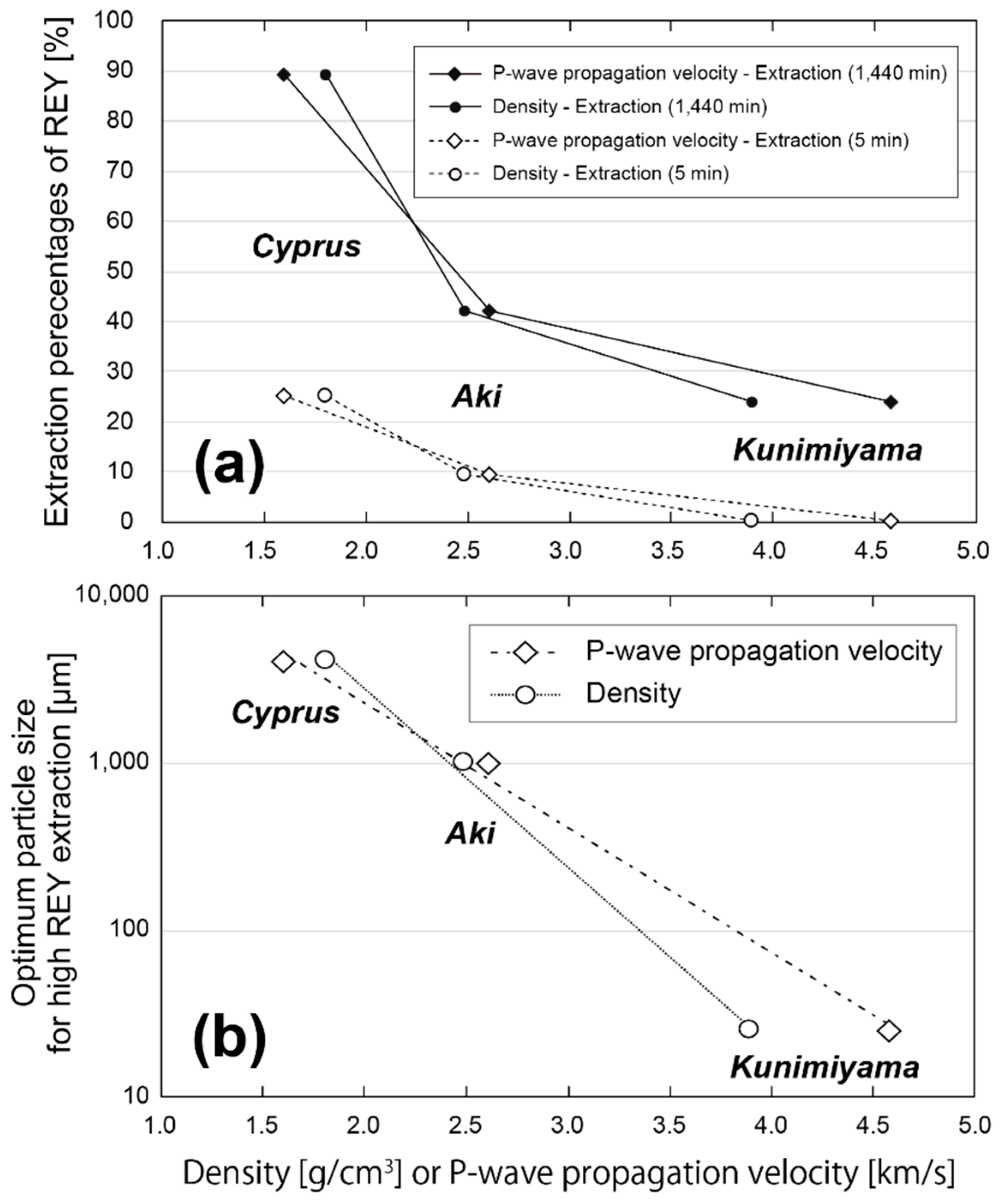
4.2. Factors Affecting the Extraction Behaviors of REE
4.3. Optimum Conditions for REY Extraction from Umber Samples
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kato, Y.; Fujinaga, K.; Nakamura, K.; Takaya, Y.; Kitamura, K.; Ohta, J.; Toda, R.; Nakashima, T.; Iwamori, H. Deep-sea mud in the Pacific Ocean as a potential resource for rare-earth elements. Nat. Geosci. 2011, 4, 535–539. [Google Scholar] [CrossRef]
- Alonso, E.; Sherman, A.M.; Wallington, T.J.; Everson, M.P.; Field, F.R.; Roth, R.; Kirchain, R.E. Evaluating rare earth element availability: A case with revolutionary demand from clean technologies. Environ. Sci. Tech. 2012, 46, 3406–3414. [Google Scholar] [CrossRef]
- Goodenough, K.M.; Wall, F.; Merriman, D. The rare earth elements: Demand, global resources, and challenges for resourcing future generations. Nat. Resour. Res. 2018, 27, 201–216. [Google Scholar] [CrossRef]
- Ober, J.A. Mineral Commodity Summaries 2018; U.S. Geological Survey: Reston, VA, USA, 2018; 200p.
- Takaya, Y.; Yasukawa, K.; Kawasaki, T.; Fujinaga, K.; Ohta, J.; Usui, Y.; Nakamura, K.; Kimura, J.-I.; Chang, Q.; Hamada, M.; et al. The tremendous potential of deep-sea mud as a source of rare-earth elements. Sci. Rep. 2018, 8, 5763. [Google Scholar] [CrossRef] [PubMed]
- Yasukawa, K.; Ohta, J.; Mimura, K.; Tanaka, E.; Takaya, Y.; Usui, Y.; Fujinaga, K.; Machida, S.; Nozaki, T.; Iijima, K.; et al. A new and prospective resource for scandium: Evidence from the geochemistry of deep-sea sediment in the western North Pacific Ocean. Ore Geol. Rev. 2018, 102, 260–267. [Google Scholar] [CrossRef]
- Kato, Y.; Fujinaga, K.; Nozaki, T.; Osawa, H.; Nakamura, K.; Ono, R. Rare earth, major and trace elements in the Kunimiyama ferromanganese deposit in the Northern Chichibu Belt, central Shikoku, Japan. Resour. Geol. 2005, 55, 291–299. [Google Scholar] [CrossRef]
- Fujinaga, K.; Nozaki, T.; Nakayama, K.; Kato, Y. Rare earth resource potential of the Aki strata-bound Fe-Mn deposit in the Northern Shimanto Belt, central Shikoku, Japan. Shigen Chishitsu 2011, 61, 1–11, (In Japanese with English Abstract). [Google Scholar]
- Josso, P.; Roberts, S.; Teagle, D.A.; Pourret, O.; Herrington, R.; de Leon Albarran, C.P. Extraction and separation of rare earth elements from hydrothermal metalliferous sediments. Miner. Eng. 2018, 118, 106–121. [Google Scholar] [CrossRef]
- German, C.R.; Klinkhammer, G.P.; Edmond, J.M.; Mura, A.; Elderfield, H. Hydrothermal scavenging of rare-earth elements in the ocean. Nature 1990, 345, 516–518. [Google Scholar] [CrossRef]
- German, C.R.; Fleer, A.P.; Bacon, M.P.; Edmond, J.M. Hydrothermal scavenging at the Mid-Atlantic Ridge: Radionuclide distributions. Earth Planet. Sci. Lett. 1991, 105, 170–181. [Google Scholar] [CrossRef]
- German, C.R.; Campbell, A.C.; Edmond, J.M. Hydrothermal scavenging at the Mid-Atlantic Ridge: Modification of trace element dissolved fluxes. Earth Planet. Sci. Lett. 1991, 107, 101–114. [Google Scholar] [CrossRef]
- Golev, A.; Scott, M.; Erskine, P.D.; Ali, S.H.; Ballantyne, G.R. Rare earths supply chains: Current status, constraints and opportunities. Resour. Policy 2014, 41, 52–59. [Google Scholar] [CrossRef]
- Kolo, M.T.; Aziz, S.A.B.A.; Khandaker, M.U.; Asaduzzaman, K.; Amin, Y.M. Evaluation of radiological risks due to natural radioactivity around Lynas Advanced Material Plant environment, Kuantan, Pahang, Malaysia. Environ. Sci. Pollut. Res. 2014, 22, 13127–13136. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Fujinaga, K.; Kato, Y. Rare earth element geochemistry of in-situin situ basalts from the Upper Cretaceous Shimanto Belt and its implication for their origin. Jpn. Mag. Mineral. Petrol. Sci. 2000, 29, 175–190, (In Japanese with English Abstract). [Google Scholar]
- Nozaki, T.; Nakamura, K.; Fujinaga, K.; Moriguchi, E.; Kato, Y. Geochemistry of oceanic crust remnant and associated stratified ferromanganese deposit from the Hayachine Belt, northeast Japan. Shigen Chishitsu 2004, 54, 77–89, (In Japanese with English Abstract). [Google Scholar]
- Nozaki, T.; Nakamura, K.; Osawa, H.; Fujinaga, K.; Kato, Y. Geochemical features and tectonic setting of greenstones from Kunimiyama, Northern Chichibu Belt, central Shikoku, Japan. Resour. Geol. 2005, 55, 301–310. [Google Scholar] [CrossRef]
- Fujinaga, K.; Kato, Y.; Kiminami, K.; Miura, K.; Nakamura, K. Geochemistry of red shale from the Mugi Formation in the Upper Cretaceous Shimanto Belt, Shikoku, Japan. Mem. Geol. Soc. Jpn. 1999, 52, 205–216, (In Japanese with English Abstract). [Google Scholar]
- Fujinaga, K.; Kato, Y. Geochemistry of ferromanganese sediment formed by a ridge collision in the late Cretaceous southwestern Japan: Geochemical analogy to a modern hydrothermal sediment at a mid-ocean ridge. Shigen Chishitsu 2001, 51, 29–40, (In Japanese with English Abstract). [Google Scholar]
- Kato, Y.; Fujinaga, K.; Suzuki, K. Major and trace element geochemistry and Os isotopic composition of metalliferous umbers from the Late Cretaceous Japanese accretionary complex. Geochem. Geophys. Geosyst. 2005, 7, Q07004. [Google Scholar] [CrossRef]
- Kato, Y.; Fujinaga, K.; Suzuki, K. Marine Os isotopic fluctuations in the early Eocene greenhouse interval as recorded by metalliferous umbers from a Tertiary ophiolite in Japan. Gondwana Res. 2011, 20, 594–607. [Google Scholar] [CrossRef]
- Gass, I.G. Is the Troodos massif of Cyprus a fragment of Mesozoic ocean floor? Nature 1968, 220, 39–42. [Google Scholar] [CrossRef]
- Searle, M.; Cox, J. Tectonic setting, origin, and obduction of the Oman ophiolite. Geol. Soc. Am. Bull. 1999, 111, 104–122. [Google Scholar] [CrossRef]
- Ravizza, G.; Sherrell, R.M.; Field, M.P.; Pickett, E.A. Geochemistry of the Margi umbers, Cyprus, and the Os isotope composition of Cretaceous seawater. Geology 1999, 27, 971–974. [Google Scholar] [CrossRef]
- Sawamura, T.; Yoshinaga, M. Iron-manganese deposits of Kunimiyama mine, Kochi prefecture. Min. Geol. 1953, 3, 207–219, (In Japanese with English Abstract). [Google Scholar]
- Yoshimura, T. Supplement to manganese ore deposits of Japan. Part II. Sci. Rept. Fac. Sci. Kyushu Univ. Geol. 1969, 9, 487–1004. (In Japanese) [Google Scholar]
- Shimazaki, Y. Mineralogy of the iron-manganese ores of the Kunimiyama mine, Shikoku, Japan. In Volcanism and Ore Genesis; University of Tokyo Press: Tokyo, Japan, 1970; pp. 303–308. [Google Scholar]
- Nozaki, T.; Fujinaga, K.; Kato, Y. Ore deposit formed on a paleo-seafloor in the Japanese accretionary complex. J. Geol. Soc. Jpn. 2018, 124, 995–1020, (In Japanese with English Abstract). [Google Scholar] [CrossRef]
- Kamikawa, Y.; Hisada, K.; Sashida, K.; Igo, H. Geology of the Nanmoku area in the Chichibu Terrane, the northwestern part of the Kanto Mountains, central Japan. Sci. Rept. Inst. Geosci. Univ. Tsukuba Sec. B 1997, 18, 19–38. [Google Scholar]
- Matsuoka, A.; Yamakita, S.; Sakakibara, M.; Hisada, K. Unit division for the Chichibu Composite Belt from a view point of accretionary tectonics and geology of western Shikoku, Japan. J. Geol. Soc. Japan 1998, 104, 634–653, (In Japanese with English Abstract). [Google Scholar] [CrossRef]
- Fujinaga, K.; Kato, Y. Radiolarian age of red chert from the Kunimiyama ferromanganese deposit in the Northern Chichibu Belt, central Shikoku, Japan. Resour. Geol. 2005, 55, 353–356. [Google Scholar] [CrossRef]
- Taira, A.; Katto, J.; Tashiro, M.; Okamura, M.; Kodama, K. The Shimanto belt in Shikoku, Japan-evolution of Cretaceous to Miocene accretionary prism. Modern Geol. 1988, 12, 5–46. [Google Scholar]
- Hall, J.M.; Yang, J.S. A preferred environment of preservation for volcanic massive sulfide deposits in the Troodos Ophiolite (Cyprus). Econ. Geol. 1994, 89, 851–857. [Google Scholar] [CrossRef]
- Josso, P. Investigating the Potential Recovery of REY from Metalliferous Sediments in a Seafloor Analogue: The Troodos Ophiolite, Cyprus. Ph.D. Thesis, University of Southampton, Southampton, UK, 2016. [Google Scholar]
- Morag, N.; Haviv, I.; Katzir, Y. From ocean depths to mountain tops: Uplift of the Troodos ophiolite (Cyprus) constrained by low-temperature thermochronology and geomorphic analysis. Tectonics 2016, 35, 622–637. [Google Scholar] [CrossRef]
- McCallum, J.E.; Robertson, A.H.F. Late Pliocene-early Pleistocene Athalassa Formation, north central Cyprus: Carbonate sand bodies in a shallow seaway between two emerging landmasses. Terra Nova 1995, 7, 265–277. [Google Scholar] [CrossRef]
- Takaya, Y.; Hiraide, T.; Fujinaga, K.; Nakamura, K.; Kato, Y. A study on the recovery method of rare-earth elements from REY-rich mud toward the development and the utilizationuse of REY-rich mud. J. Min. Mater. Inst. Japan 2014, 130, 104–114, (In Japanese with English Abstract). [Google Scholar]
- Takaya, Y.; Fujinaga, K.; Yamagata, N.; Araki, S.; Maki, R.; Nakamura, K.; Iijima, K.; Kato, Y. Chemical leaching of rare earth elements from highly REY-rich mud. Geochem. J. 2015, 49, 637–652. [Google Scholar] [CrossRef]
- Takabe, M. Carriers of rare earth elements in Pacific deep-sea sediments. J. Geol. 2005, 113, 205–215. [Google Scholar] [CrossRef]
- Yasukawa, K.; Nakamura, K.; Fujinaga, K.; Iwamori, H.; Kato, Y. Tracking the spatiotemporal variations of statistically independent components involving enrichment of rare-earth elements in deep-sea sediments. Sci. Rep. 2016, 6, 29603. [Google Scholar] [CrossRef] [PubMed]
- Kashiwabara, T.; Toda, R.; Nakamura, K.; Yasukawa, K.; Fujinaga, K.; Kubo, S.; Nozaki, T.; Takahashi, Y.; Suzuki, K.; Kato, Y. Synchrotron X-ray spectroscopic perspective on the formation mechanism of REY-rich muds in the Pacific Ocean. Geochim. Cosmochim. Acta 2018, 240, 274–292. [Google Scholar] [CrossRef]
- De Baar, H.J.; Bacon, M.P.; Brewer, P.G.; Bruland, K.W. Rare earth elements in the Pacific and Atlantic Oceans. Geochim. Cosmochim. Acta 1985, 49, 1943–1959. [Google Scholar] [CrossRef]
- Alibo, D.S.; Nozaki, Y. Rare earth elements in seawater: Particle association, shale-normalization, and Ce oxidation. Geochim. Cosmochim. Acta 1999, 63, 363–372. [Google Scholar] [CrossRef]
- Chosa-tokeibu, T.S.D.K. Yearbook of Current Production Statistics (Chemical Industry); Research and Statistics Department, Minister’s Secretariat, the Ministry of Economy, Trade and Industry (METI): Tokyo, Japan, 2017; 238p.
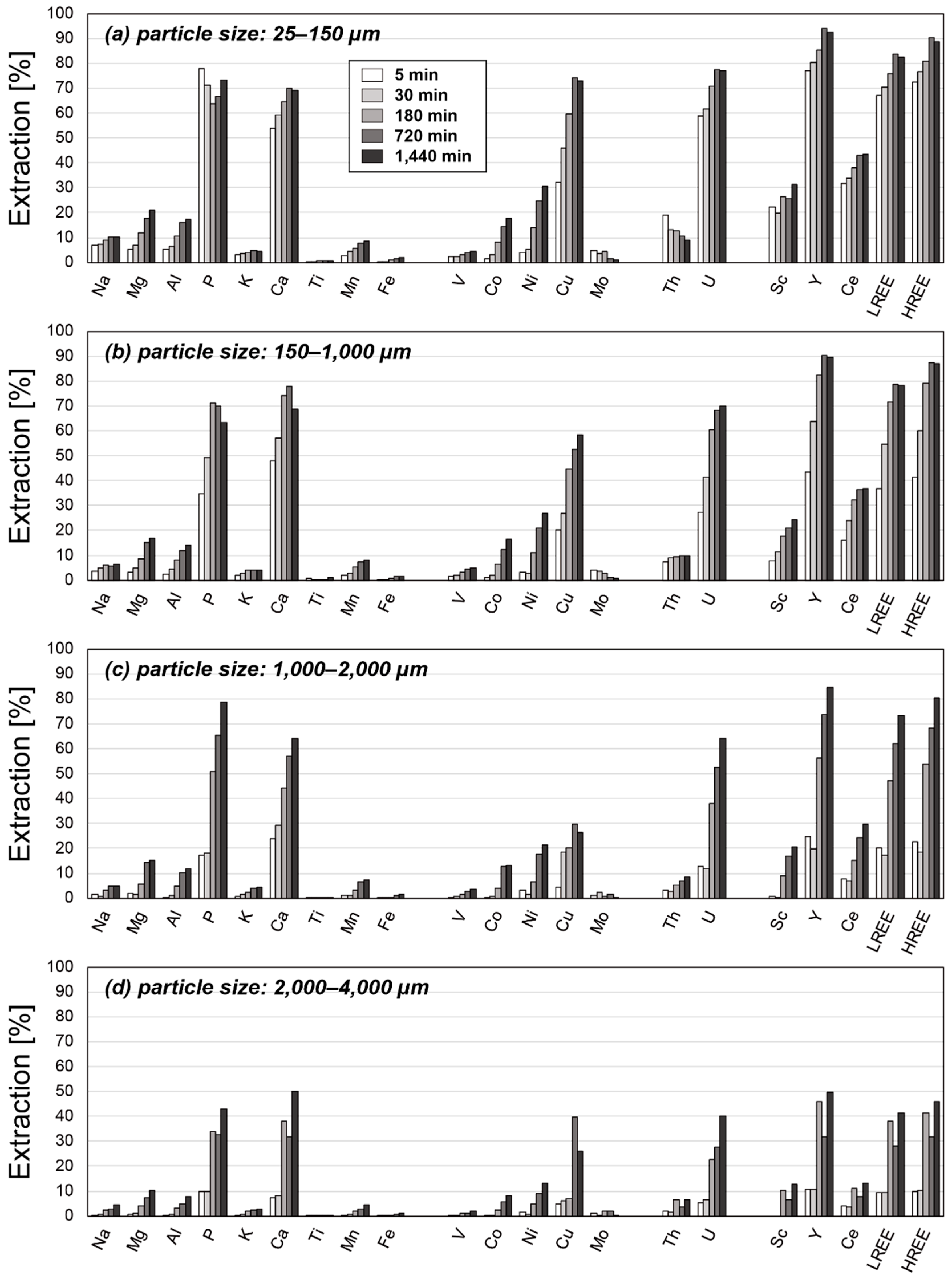
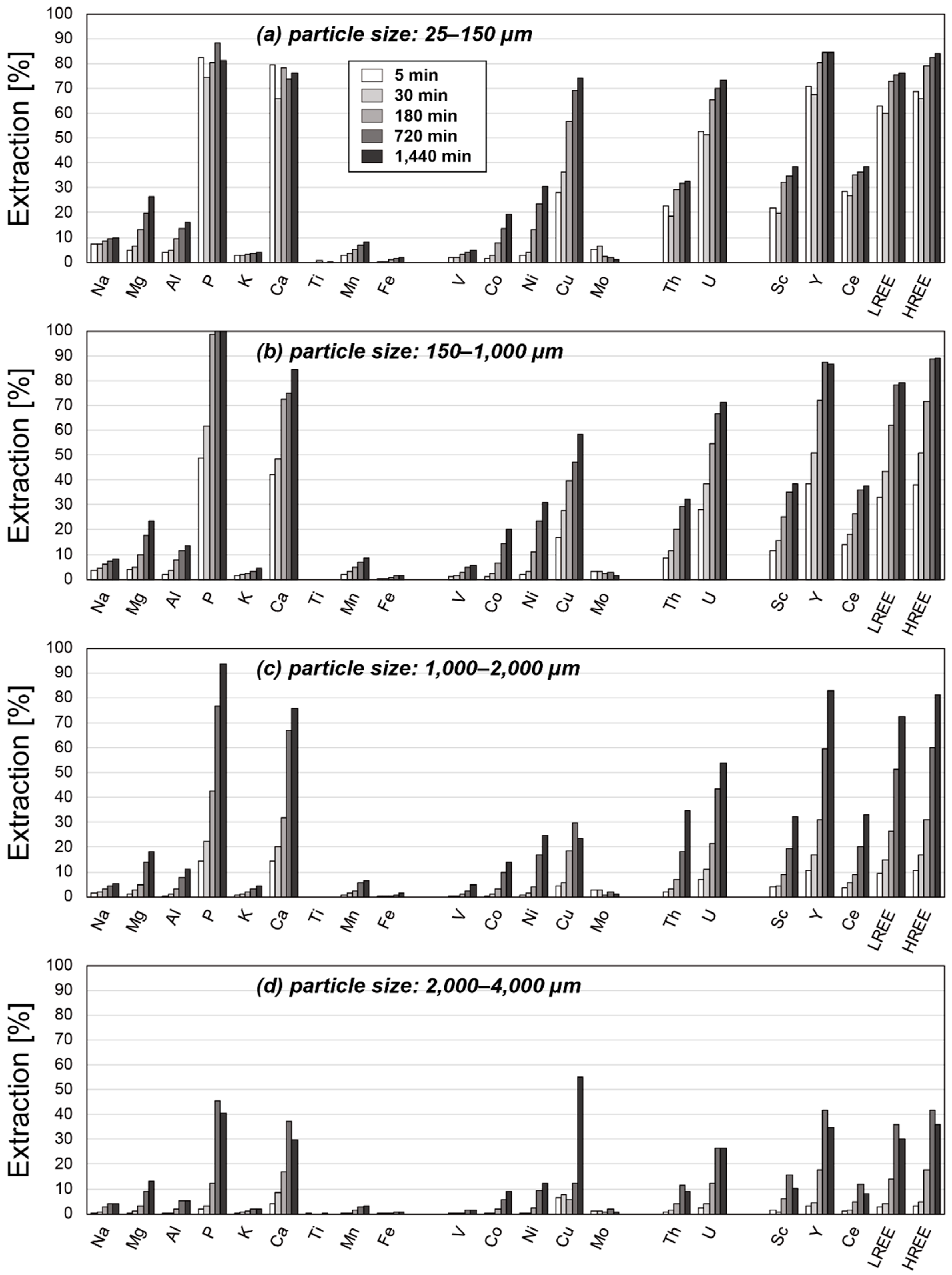
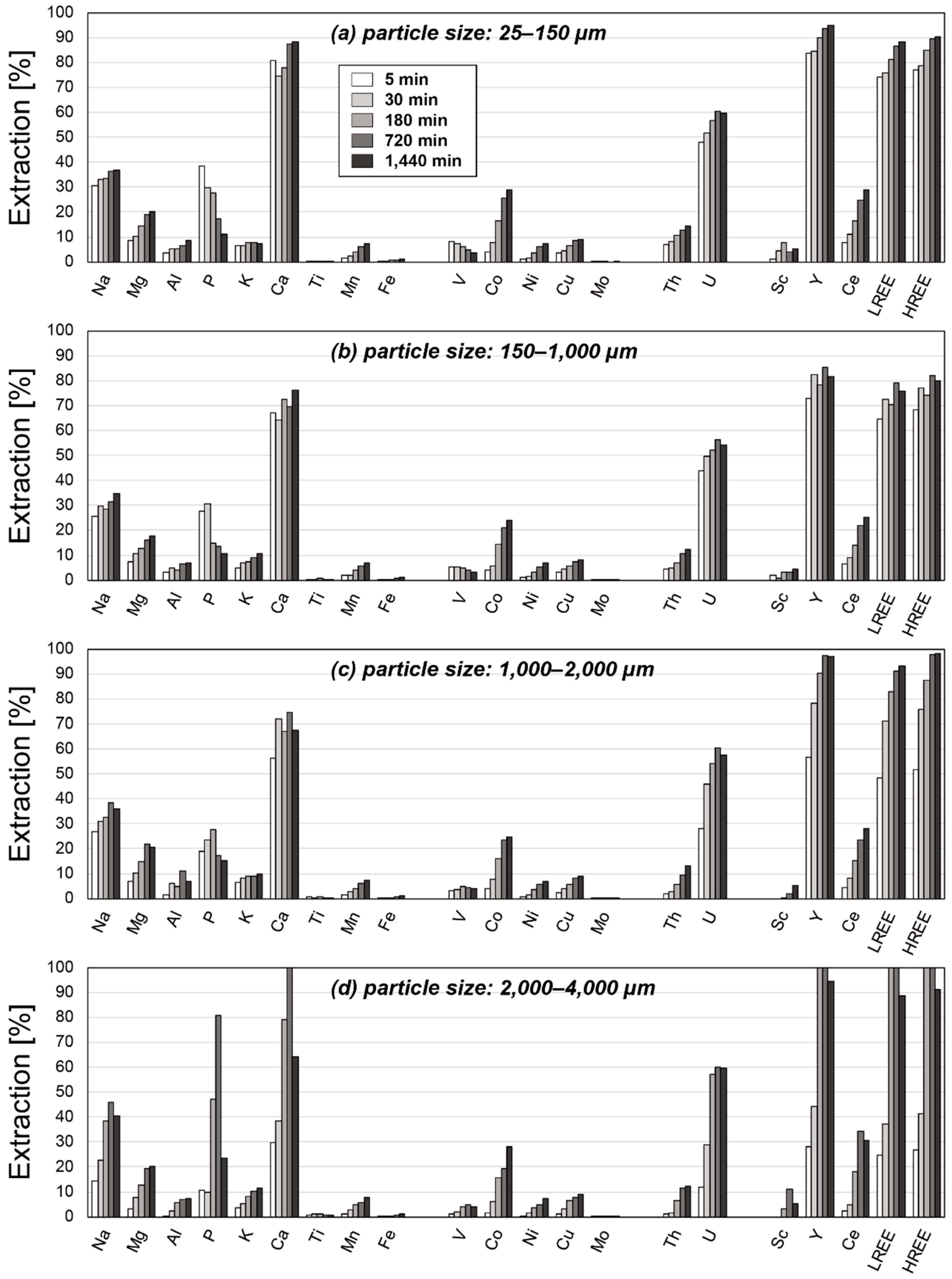
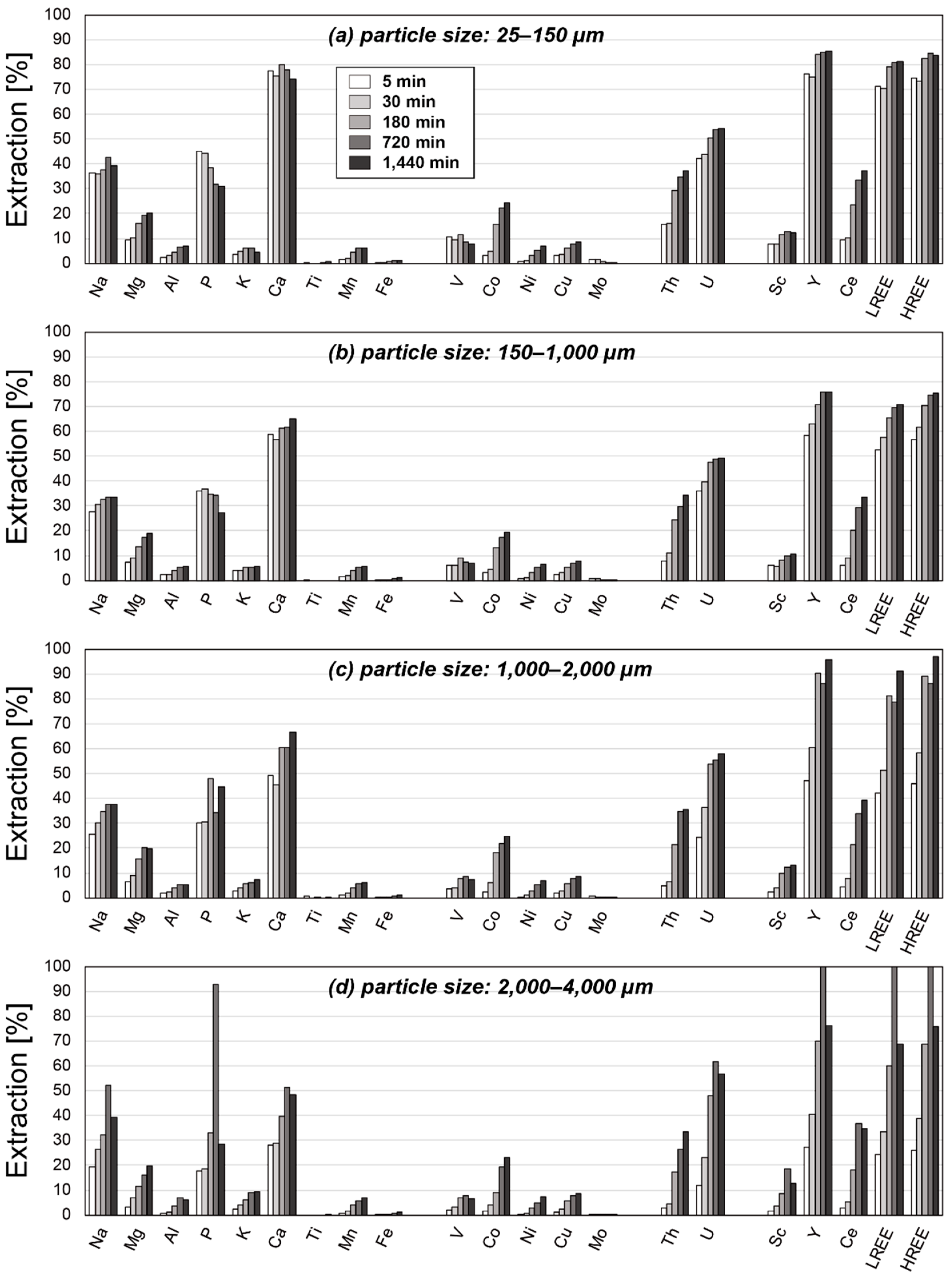
| Particle Size [µm] | 25–150 | 150–1000 | 1000–2000 | 2000–4000 | ||||
|---|---|---|---|---|---|---|---|---|
| Average | 1δ | Average | 1δ | Average | 1δ | Average | 1δ | |
| Na (%) | 0.01 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 |
| Mg | 1.10 | 0.04 | 1.26 | 0.04 | 1.25 | 0.06 | 1.23 | 0.10 |
| Al | 1.08 | 0.07 | 1.29 | 0.03 | 1.23 | 0.05 | 1.09 | 0.00 |
| P | 0.39 | 0.01 | 0.38 | 0.08 | 0.42 | 0.13 | 0.38 | 0.22 |
| K | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Ca | 3.17 | 0.14 | 2.40 | 0.19 | 1.76 | 0.35 | 1.85 | 0.79 |
| Sc (ppm) | 21.89 | 0.70 | 24.46 | 0.22 | 25.10 | 0.71 | 25.46 | 2.45 |
| Ti (%) | 0.30 | 0.02 | 0.35 | 0.02 | 0.37 | 0.01 | 0.35 | 0.01 |
| V (ppm) | 1206.40 | 39.04 | 1299.16 | 25.90 | 1308.36 | 61.25 | 1196.71 | 54.74 |
| Cr | 20.80 | 0.75 | 21.66 | 0.81 | 24.38 | 3.77 | 23.52 | 3.31 |
| Mn (%) | 18.01 | 0.58 | 18.88 | 0.38 | 18.71 | 0.76 | 17.99 | 0.53 |
| Fe | 25.66 | 0.85 | 28.43 | 0.15 | 28.59 | 0.57 | 27.22 | 1.31 |
| Co (ppm) | 287.57 | 7.09 | 313.46 | 5.87 | 317.28 | 11.57 | 316.08 | 12.10 |
| Ni | 1408.46 | 30.48 | 1522.99 | 16.40 | 1542.80 | 64.20 | 1613.62 | 78.31 |
| Cu | 317.78 | 11.70 | 240.21 | 28.59 | 201.89 | 59.97 | 148.55 | 10.79 |
| Zn | 675.09 | 23.55 | 736.27 | 13.53 | 728.55 | 18.87 | 722.51 | 30.04 |
| As | 138.52 | 11.22 | 152.42 | 15.23 | 171.24 | 23.70 | 150.54 | 68.06 |
| Rb | 0.53 | 0.03 | 0.62 | 0.03 | 0.61 | 0.02 | 0.57 | 0.14 |
| Sr | 141.77 | 4.85 | 137.41 | 6.87 | 117.79 | 5.11 | 121.77 | 7.57 |
| Y | 240.16 | 8.71 | 243.56 | 11.30 | 273.22 | 22.34 | 258.28 | 69.84 |
| Zr | 344.77 | 20.16 | 421.93 | 73.88 | 419.31 | 27.38 | 397.43 | 57.84 |
| Nb | 35.54 | 1.01 | 38.91 | 0.16 | 40.86 | 1.64 | 38.61 | 3.64 |
| Mo | 273.92 | 34.30 | 99.17 | 49.43 | 48.78 | 8.36 | 49.22 | 10.73 |
| Cd | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| In | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| Sn | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| Sb | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| Cs | 0.43 | 0.01 | 0.44 | 0.01 | 0.43 | 0.02 | 0.47 | 0.05 |
| Ba | 247.32 | 10.77 | 274.72 | 40.81 | 200.42 | 7.87 | 203.92 | 96.32 |
| La | 397.37 | 11.97 | 421.66 | 9.04 | 429.54 | 17.23 | 418.61 | 14.85 |
| Ce | 241.47 | 8.12 | 259.51 | 5.63 | 256.43 | 3.02 | 252.38 | 15.96 |
| Pr | 93.55 | 3.13 | 97.92 | 2.31 | 100.85 | 3.54 | 97.91 | 6.61 |
| Nd | 378.95 | 10.68 | 387.80 | 10.26 | 403.44 | 13.29 | 391.54 | 34.37 |
| Sm | 83.53 | 2.47 | 84.48 | 2.33 | 88.35 | 4.27 | 84.94 | 10.37 |
| Eu | 19.16 | 0.59 | 19.17 | 0.51 | 20.13 | 1.20 | 19.49 | 2.88 |
| Gd | 81.04 | 2.38 | 81.01 | 2.80 | 85.24 | 5.89 | 84.23 | 15.77 |
| Tb | 12.32 | 0.43 | 12.37 | 0.40 | 13.08 | 0.84 | 12.90 | 2.37 |
| Dy | 70.71 | 2.51 | 71.65 | 2.23 | 75.82 | 4.58 | 74.77 | 14.41 |
| Ho | 13.34 | 0.46 | 13.43 | 0.35 | 14.18 | 0.80 | 13.81 | 2.75 |
| Er | 36.30 | 1.28 | 36.84 | 1.02 | 39.29 | 1.43 | 38.24 | 7.50 |
| Tm | 5.02 | 0.16 | 5.15 | 0.11 | 5.43 | 0.22 | 5.23 | 0.91 |
| Yb | 31.66 | 1.23 | 32.62 | 0.56 | 34.24 | 0.90 | 32.91 | 5.22 |
| Lu | 4.54 | 0.14 | 4.68 | 0.08 | 4.93 | 0.10 | 4.76 | 0.77 |
| Hf | 4.72 | 0.11 | 5.59 | 0.72 | 5.65 | 0.26 | 5.37 | 0.70 |
| Ta | 0.21 | 0.00 | 0.22 | 0.01 | 0.22 | 0.01 | 0.25 | 0.03 |
| W | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Tl | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| Pb | 274.51 | 17.42 | 223.77 | 13.29 | 199.01 | 32.64 | 185.83 | 17.69 |
| Bi | 0.00 | 0.00 | 1.70 | 2.94 | 0.00 | 0.00 | 0.00 | 0.00 |
| Th | 8.83 | 0.30 | 9.93 | 0.69 | 10.14 | 0.60 | 9.76 | 1.10 |
| U | 1.42 | 0.03 | 1.52 | 0.03 | 1.59 | 0.12 | 1.52 | 0.16 |
| ΣREY | 1709.11 | 44.04 | 1771.85 | 32.44 | 1844.17 | 62.28 | 1790.01 | 144.75 |
| Particle Size [µm] | 25–150 | 150–1000 | 1000–2000 | 2000–4000 | ||||
|---|---|---|---|---|---|---|---|---|
| Average | 1δ | Average | 1δ | Average | 1δ | Average | 1δ | |
| Na (%) | 0.17 | 0.00 | 0.16 | 0.01 | 0.13 | 0.01 | 0.15 | 0.11 |
| Mg | 0.91 | 0.02 | 0.75 | 0.08 | 0.59 | 0.06 | 0.82 | 0.26 |
| Al | 2.52 | 0.07 | 2.14 | 0.19 | 1.70 | 0.11 | 2.21 | 1.27 |
| P | 0.35 | 0.01 | 0.27 | 0.03 | 0.19 | 0.01 | 0.31 | 0.30 |
| K | 1.03 | 0.03 | 0.95 | 0.07 | 0.68 | 0.09 | 0.87 | 0.65 |
| Ca | 1.17 | 0.07 | 0.80 | 0.07 | 0.58 | 0.09 | 0.90 | 0.42 |
| Sc (ppm) | 11.00 | 0.30 | 9.01 | 0.68 | 6.88 | 0.50 | 7.99 | 7.12 |
| Ti (%) | 0.13 | 0.01 | 0.11 | 0.01 | 0.08 | 0.01 | 0.12 | 0.12 |
| V (ppm) | 259.46 | 3.44 | 185.38 | 16.92 | 147.80 | 24.86 | 216.14 | 94.67 |
| Cr | 31.66 | 2.49 | 27.94 | 2.61 | 19.14 | 1.77 | 22.97 | 19.42 |
| Mn (%) | 3.35 | 0.09 | 2.20 | 0.26 | 1.54 | 0.22 | 2.05 | 0.78 |
| Fe | 17.45 | 0.31 | 15.52 | 1.09 | 10.96 | 0.77 | 14.26 | 12.22 |
| Co (ppm) | 40.40 | 0.94 | 29.37 | 3.05 | 22.12 | 2.51 | 32.36 | 11.38 |
| Ni | 127.35 | 3.02 | 90.89 | 11.97 | 66.29 | 6.54 | 93.90 | 43.10 |
| Cu | 536.53 | 24.44 | 342.47 | 53.91 | 505.02 | 61.53 | 291.14 | 199.91 |
| Zn | 297.09 | 6.62 | 214.14 | 24.67 | 153.16 | 17.94 | 235.77 | 90.92 |
| As | 98.64 | 1.16 | 69.73 | 5.19 | 56.61 | 5.31 | 81.73 | 42.95 |
| Rb | 51.78 | 0.73 | 49.12 | 3.79 | 35.39 | 6.10 | 46.81 | 36.81 |
| Sr | 123.28 | 2.50 | 90.33 | 9.17 | 68.74 | 3.78 | 86.46 | 36.17 |
| Y | 148.71 | 2.01 | 118.92 | 10.38 | 85.88 | 7.72 | 119.44 | 104.31 |
| Zr | 129.21 | 1.24 | 111.64 | 9.81 | 84.22 | 10.92 | 99.00 | 85.93 |
| Nb | 6.73 | 0.13 | 5.72 | 0.52 | 4.16 | 0.38 | 5.24 | 4.71 |
| Mo | 3.00 | 0.05 | 2.29 | 0.26 | 1.78 | 0.13 | 2.41 | 1.34 |
| Cd | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| In | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| Sn | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| Sb | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| Cs | 5.54 | 0.10 | 5.05 | 0.41 | 3.66 | 0.40 | 4.81 | 4.59 |
| Ba | 540.63 | 2.17 | 293.20 | 52.34 | 165.13 | 36.01 | 228.54 | 172.79 |
| La | 161.03 | 2.46 | 135.50 | 11.29 | 96.85 | 3.93 | 134.28 | 113.94 |
| Ce | 52.82 | 0.61 | 41.66 | 3.39 | 30.73 | 1.34 | 41.61 | 30.78 |
| Pr | 45.76 | 0.47 | 36.42 | 2.82 | 26.70 | 2.27 | 36.14 | 29.37 |
| Nd | 186.81 | 2.68 | 147.63 | 11.45 | 108.19 | 10.49 | 147.39 | 122.02 |
| Sm | 41.58 | 0.60 | 32.47 | 2.78 | 24.04 | 2.61 | 32.23 | 26.64 |
| Eu | 9.57 | 0.12 | 7.56 | 0.63 | 5.61 | 0.51 | 7.61 | 6.33 |
| Gd | 40.45 | 0.45 | 31.79 | 2.77 | 23.29 | 2.21 | 31.75 | 26.66 |
| Tb | 5.91 | 0.09 | 4.67 | 0.38 | 3.43 | 0.36 | 4.64 | 3.92 |
| Dy | 34.85 | 0.44 | 27.24 | 2.50 | 20.05 | 2.17 | 27.18 | 23.15 |
| Ho | 6.41 | 0.08 | 5.08 | 0.46 | 3.72 | 0.39 | 5.13 | 4.44 |
| Er | 17.74 | 0.17 | 14.01 | 1.23 | 10.13 | 0.97 | 14.09 | 12.33 |
| Tm | 2.44 | 0.02 | 1.91 | 0.16 | 1.40 | 0.13 | 1.92 | 1.68 |
| Yb | 14.97 | 0.19 | 11.73 | 1.06 | 8.46 | 0.75 | 11.82 | 10.18 |
| Lu | 2.07 | 0.03 | 1.62 | 0.15 | 1.16 | 0.11 | 1.63 | 1.42 |
| Hf | 1.75 | 0.02 | 1.57 | 0.08 | 1.11 | 0.07 | 1.29 | 1.13 |
| Ta | 0.31 | 0.01 | 0.28 | 0.00 | 0.19 | 0.01 | 0.23 | 0.21 |
| W | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Tl | b.d.l. | - | b.d.l. | - | b.d.l. | - | b.d.l. | - |
| Pb | 89.04 | 2.16 | 72.57 | 5.93 | 56.62 | 6.79 | 67.43 | 51.82 |
| Bi | 0.00 | 0.00 | 0.00 | 0.00 | 1.72 | 2.98 | 1.80 | 3.12 |
| Th | 3.84 | 0.05 | 3.51 | 0.09 | 2.39 | 0.10 | 2.90 | 2.52 |
| U | 2.09 | 0.02 | 1.69 | 0.12 | 1.31 | 0.15 | 1.60 | 0.97 |
| ΣREY | 771.12 | 7.83 | 618.21 | 41.84 | 449.62 | 25.71 | 616.86 | 422.22 |
| Particle Size [µm] | 25–150 | 150–1000 | 1000–2000 | 2000–4000 | ||||
|---|---|---|---|---|---|---|---|---|
| Average | 1δ | Average | 1δ | Average | 1δ | Average | 1δ | |
| Na (%) | 0.10 | 0.00 | 0.11 | 0.01 | 0.10 | 0.01 | 0.11 | 0.01 |
| Mg | 0.94 | 0.01 | 0.94 | 0.06 | 0.91 | 0.02 | 1.01 | 0.01 |
| Al | 1.27 | 0.01 | 1.25 | 0.12 | 1.27 | 0.04 | 1.36 | 0.02 |
| P | 0.54 | 0.01 | 0.51 | 0.08 | 0.47 | 0.12 | 0.57 | 0.22 |
| K | 0.36 | 0.01 | 0.37 | 0.02 | 0.36 | 0.03 | 0.35 | 0.01 |
| Ca | 1.21 | 0.09 | 1.33 | 0.04 | 1.33 | 0.33 | 1.64 | 0.51 |
| Sc (ppm) | 6.41 | 0.08 | 6.38 | 0.35 | 6.50 | 0.25 | 6.47 | 0.41 |
| Ti (%) | 0.08 | 0.00 | 0.08 | 0.00 | 0.07 | 0.01 | 0.06 | 0.01 |
| V (ppm) | 839.23 | 6.53 | 843.49 | 38.25 | 822.70 | 63.74 | 806.32 | 8.71 |
| Cr | 28.07 | 3.01 | 17.22 | 0.65 | 14.94 | 1.47 | 14.84 | 0.99 |
| Mn (%) | 7.45 | 0.05 | 7.63 | 0.27 | 7.58 | 0.50 | 6.82 | 0.20 |
| Fe | 38.31 | 0.37 | 37.01 | 2.06 | 36.91 | 2.12 | 36.33 | 0.71 |
| Co (ppm) | 155.78 | 1.64 | 181.50 | 5.91 | 156.22 | 3.33 | 183.07 | 37.13 |
| Ni | 326.87 | 5.25 | 331.94 | 13.23 | 313.00 | 18.51 | 300.47 | 8.26 |
| Cu | 1079.80 | 8.74 | 1075.19 | 59.80 | 1056.16 | 53.53 | 1023.40 | 9.74 |
| Zn | 404.56 | 6.70 | 401.92 | 17.12 | 389.40 | 20.59 | 382.99 | 3.56 |
| As | 384.11 | 1.61 | 395.46 | 24.70 | 407.02 | 21.08 | 377.68 | 8.46 |
| Rb | 14.05 | 0.24 | 14.00 | 1.04 | 13.70 | 1.48 | 13.23 | 0.17 |
| Sr | 321.66 | 3.93 | 326.28 | 12.84 | 320.29 | 25.29 | 349.82 | 17.82 |
| Y | 105.38 | 1.80 | 97.79 | 6.30 | 93.99 | 16.21 | 116.15 | 26.68 |
| Zr | 73.06 | 0.67 | 71.06 | 4.96 | 70.18 | 4.40 | 70.03 | 0.80 |
| Nb | 2.99 | 0.02 | 2.95 | 0.23 | 2.87 | 0.17 | 2.91 | 0.02 |
| Mo | 12.65 | 0.08 | 13.25 | 0.47 | 12.03 | 0.71 | 12.72 | 0.76 |
| Cd | - | - | - | - | - | - | - | - |
| In | - | - | - | - | - | - | - | - |
| Sn | - | - | - | - | - | - | - | - |
| Sb | - | - | - | - | - | - | - | - |
| Cs | 0.91 | 0.01 | 0.93 | 0.05 | 0.91 | 0.07 | 0.90 | 0.01 |
| Ba | 876.95 | 0.72 | 955.40 | 16.64 | 902.53 | 46.64 | 992.35 | 115.35 |
| La | 93.48 | 0.53 | 92.94 | 8.77 | 84.52 | 26.27 | 114.49 | 43.59 |
| Ce | 31.03 | 0.11 | 30.38 | 1.94 | 30.10 | 2.13 | 31.53 | 1.01 |
| Pr | 24.04 | 0.05 | 22.83 | 1.76 | 21.06 | 4.62 | 26.99 | 7.77 |
| Nd | 101.53 | 0.74 | 94.46 | 6.97 | 86.97 | 17.87 | 110.83 | 29.17 |
| Sm | 22.22 | 0.16 | 20.56 | 1.46 | 19.22 | 3.37 | 23.39 | 5.07 |
| Eu | 5.55 | 0.01 | 5.15 | 0.37 | 4.84 | 0.86 | 5.99 | 1.29 |
| Gd | 23.87 | 0.23 | 21.98 | 1.61 | 20.53 | 3.72 | 25.66 | 5.42 |
| Tb | 3.51 | 0.03 | 3.29 | 0.24 | 3.07 | 0.58 | 3.82 | 0.82 |
| Dy | 21.23 | 0.13 | 19.97 | 1.43 | 18.75 | 3.50 | 23.37 | 5.27 |
| Ho | 4.21 | 0.02 | 3.95 | 0.27 | 3.69 | 0.73 | 4.63 | 1.06 |
| Er | 11.65 | 0.13 | 10.96 | 0.77 | 10.35 | 2.06 | 12.98 | 3.12 |
| Tm | 1.60 | 0.01 | 1.53 | 0.10 | 1.43 | 0.27 | 1.78 | 0.43 |
| Yb | 9.82 | 0.04 | 9.43 | 0.60 | 8.91 | 1.76 | 10.90 | 2.56 |
| Lu | 1.38 | 0.00 | 1.33 | 0.10 | 1.25 | 0.24 | 1.53 | 0.35 |
| Hf | 0.88 | 0.01 | 0.87 | 0.07 | 0.85 | 0.07 | 0.87 | 0.02 |
| Ta | 0.11 | 0.00 | 0.11 | 0.01 | 0.11 | 0.01 | 0.11 | 0.00 |
| W | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Tl | - | - | - | - | - | - | - | - |
| Pb | 140.39 | 0.46 | 137.75 | 6.73 | 136.71 | 10.46 | 140.02 | 2.76 |
| Bi | 1.74 | 3.02 | 1.69 | 2.92 | 0.00 | 0.00 | 0.00 | 0.00 |
| Th | 1.29 | 0.01 | 1.27 | 0.08 | 1.26 | 0.10 | 1.27 | 0.02 |
| U | 1.58 | 0.01 | 1.55 | 0.08 | 1.53 | 0.12 | 1.61 | 0.05 |
| ΣREY | 460.50 | 2.75 | 436.55 | 26.05 | 408.68 | 68.28 | 514.02 | 108.83 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takaya, Y.; Wang, M.; Fujinaga, K.; Uchida, E.; Nozaki, T.; Kato, Y. Experiments on Rare-Earth Element Extractions from Umber Ores for Optimizing the Grinding Process. Minerals 2019, 9, 239. https://doi.org/10.3390/min9040239
Takaya Y, Wang M, Fujinaga K, Uchida E, Nozaki T, Kato Y. Experiments on Rare-Earth Element Extractions from Umber Ores for Optimizing the Grinding Process. Minerals. 2019; 9(4):239. https://doi.org/10.3390/min9040239
Chicago/Turabian StyleTakaya, Yutaro, Meiqi Wang, Koichiro Fujinaga, Etsuo Uchida, Tatsuo Nozaki, and Yasuhiro Kato. 2019. "Experiments on Rare-Earth Element Extractions from Umber Ores for Optimizing the Grinding Process" Minerals 9, no. 4: 239. https://doi.org/10.3390/min9040239
APA StyleTakaya, Y., Wang, M., Fujinaga, K., Uchida, E., Nozaki, T., & Kato, Y. (2019). Experiments on Rare-Earth Element Extractions from Umber Ores for Optimizing the Grinding Process. Minerals, 9(4), 239. https://doi.org/10.3390/min9040239





