Characterization of Carbonate Crust from a Recently Discovered Methane Seep on the North Atlantic Continental Margin of the USA
Abstract
1. Introduction
2. Materials and Methods
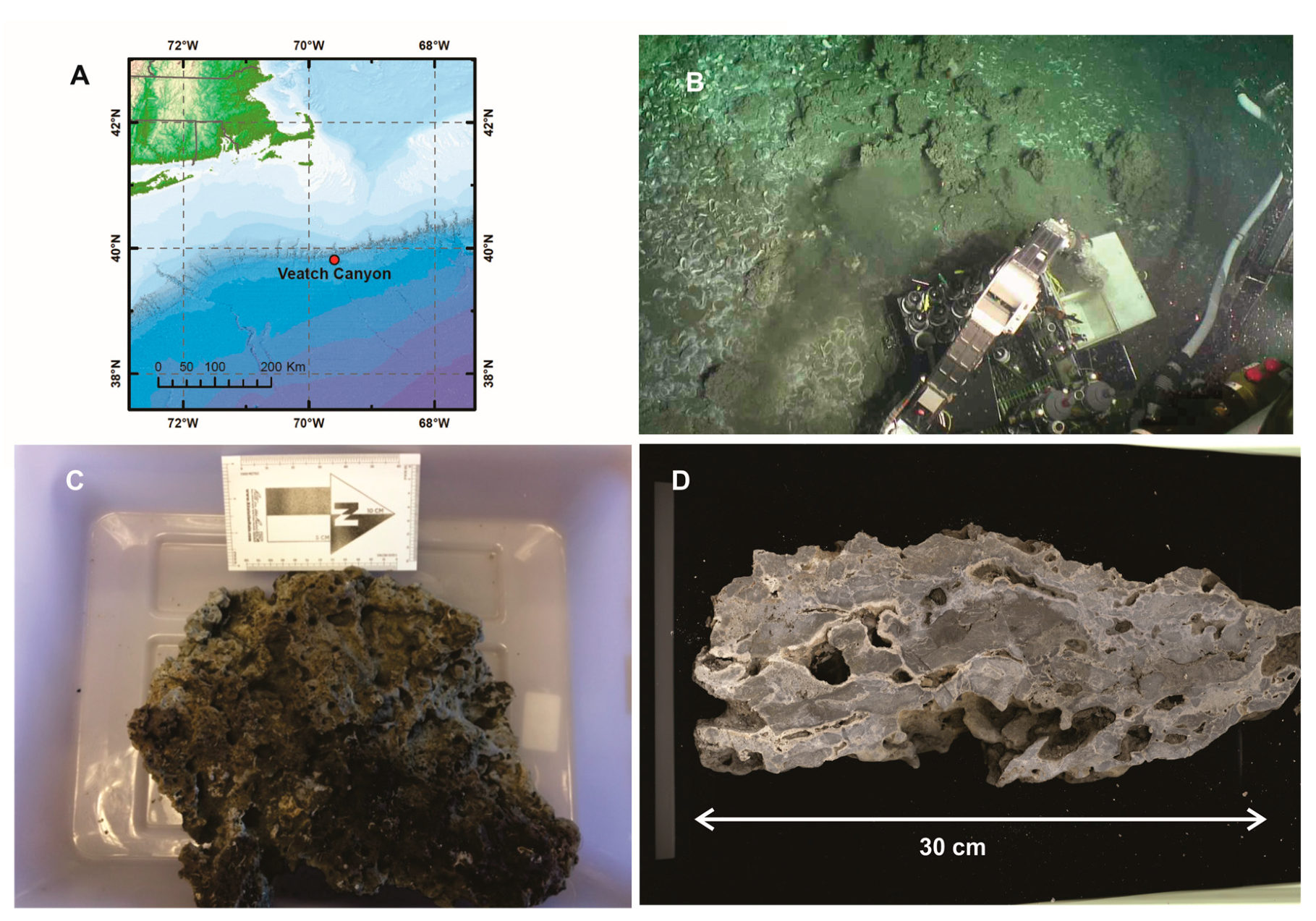
2.1. Sample Collection and Preparation
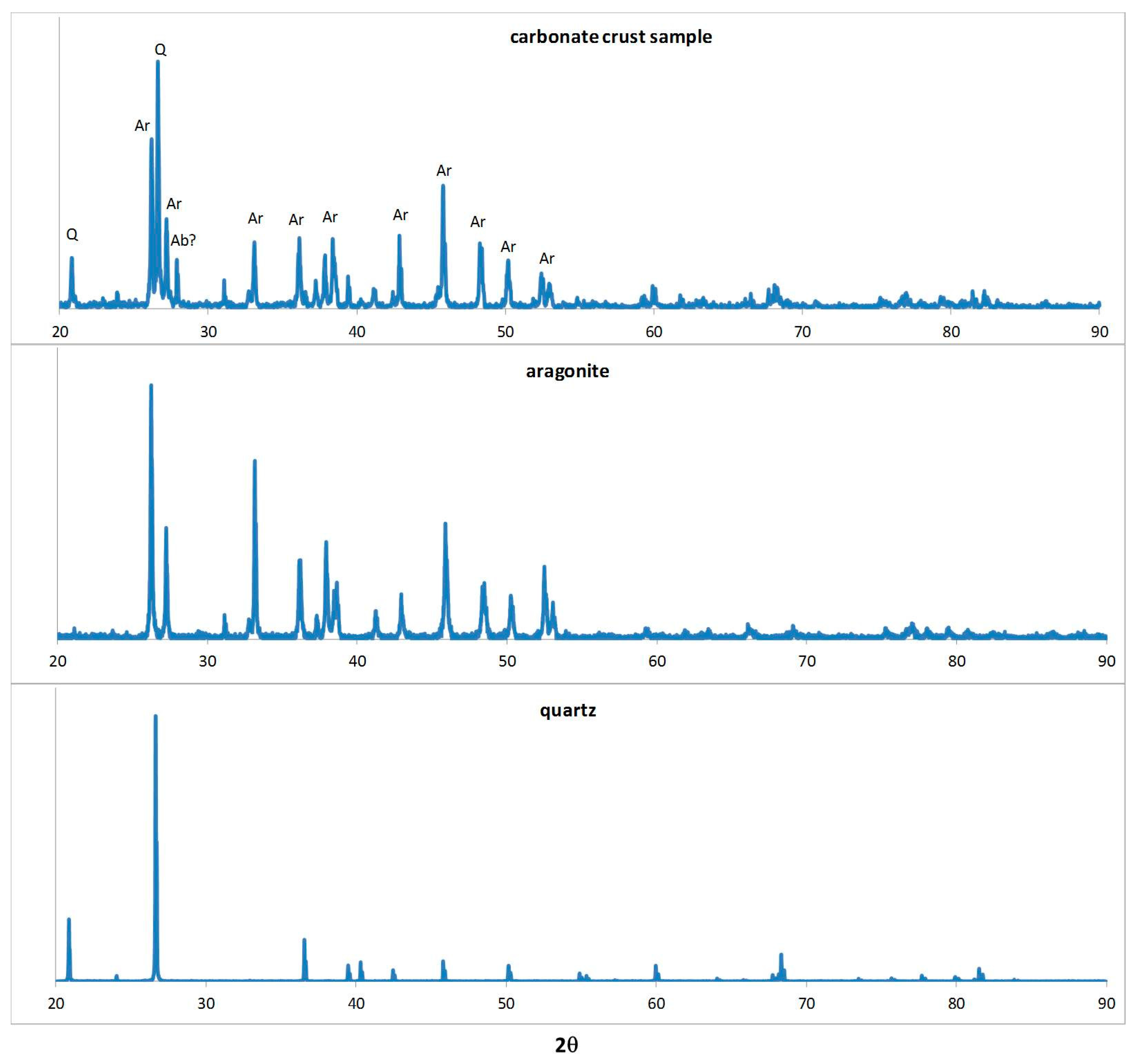
2.2. X-ray Diffraction (XRD), Scanning Electron Microscopy (SEM), and Optical Microscopy
2.3. Carbonate Standard Development
2.4. Elemental Analysis with Laser Ablation Inductively Coupled Plasma Mass Spectrometry (LA-ICP-MS)
2.5. Stable Isotope Analysis with Isotope Ratio Mass Spectrometry (IRMS)
3. Results
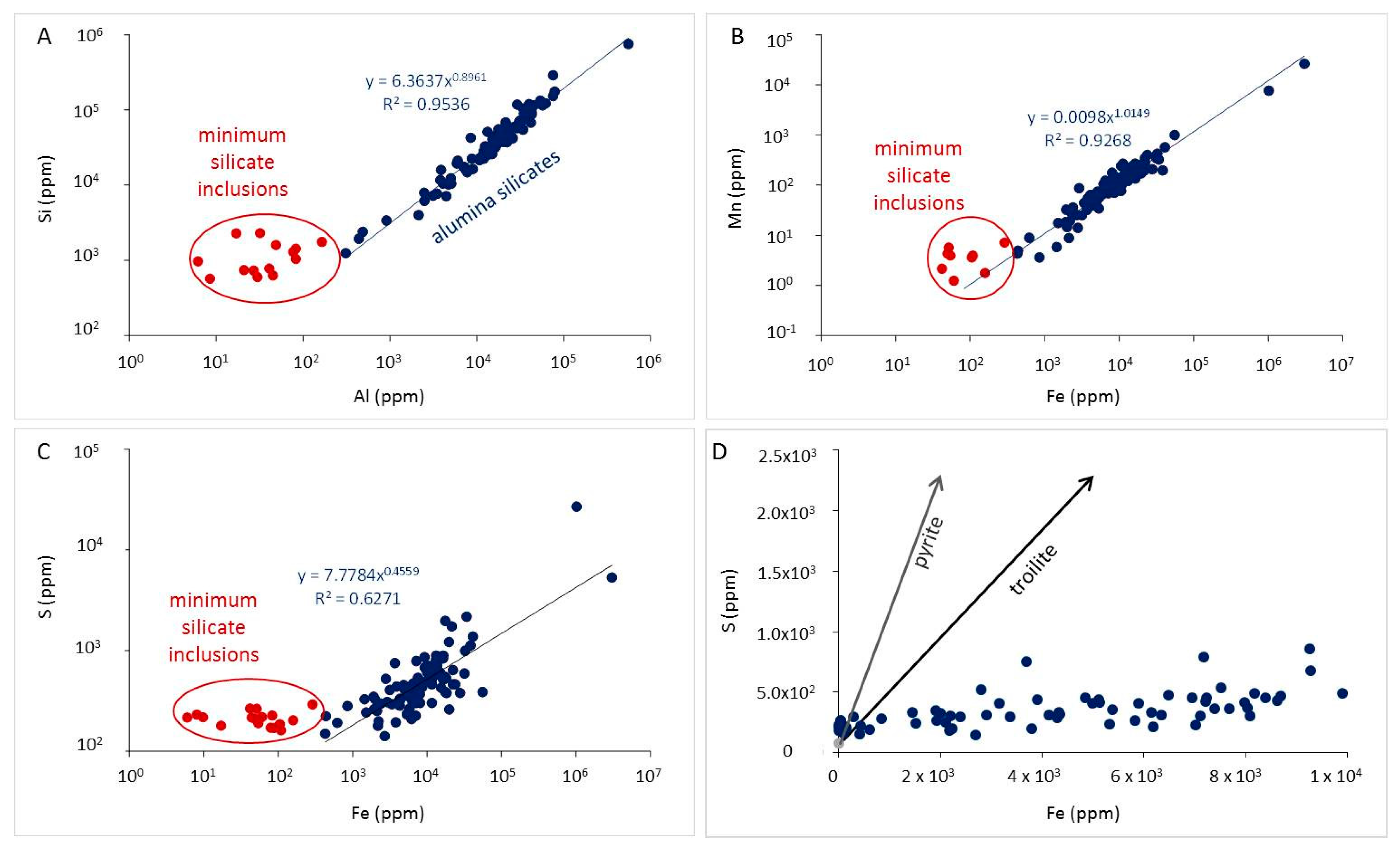
3.1. Comparison of LA-ICP-MS Data Reduction for Silicate and Carbonate Standards
3.2. Carbonate Crust
4. Discussion
4.1. Carbonate Standard for LA-ICP-MS
4.2. Stable Isotopes
4.3. Mineralogy
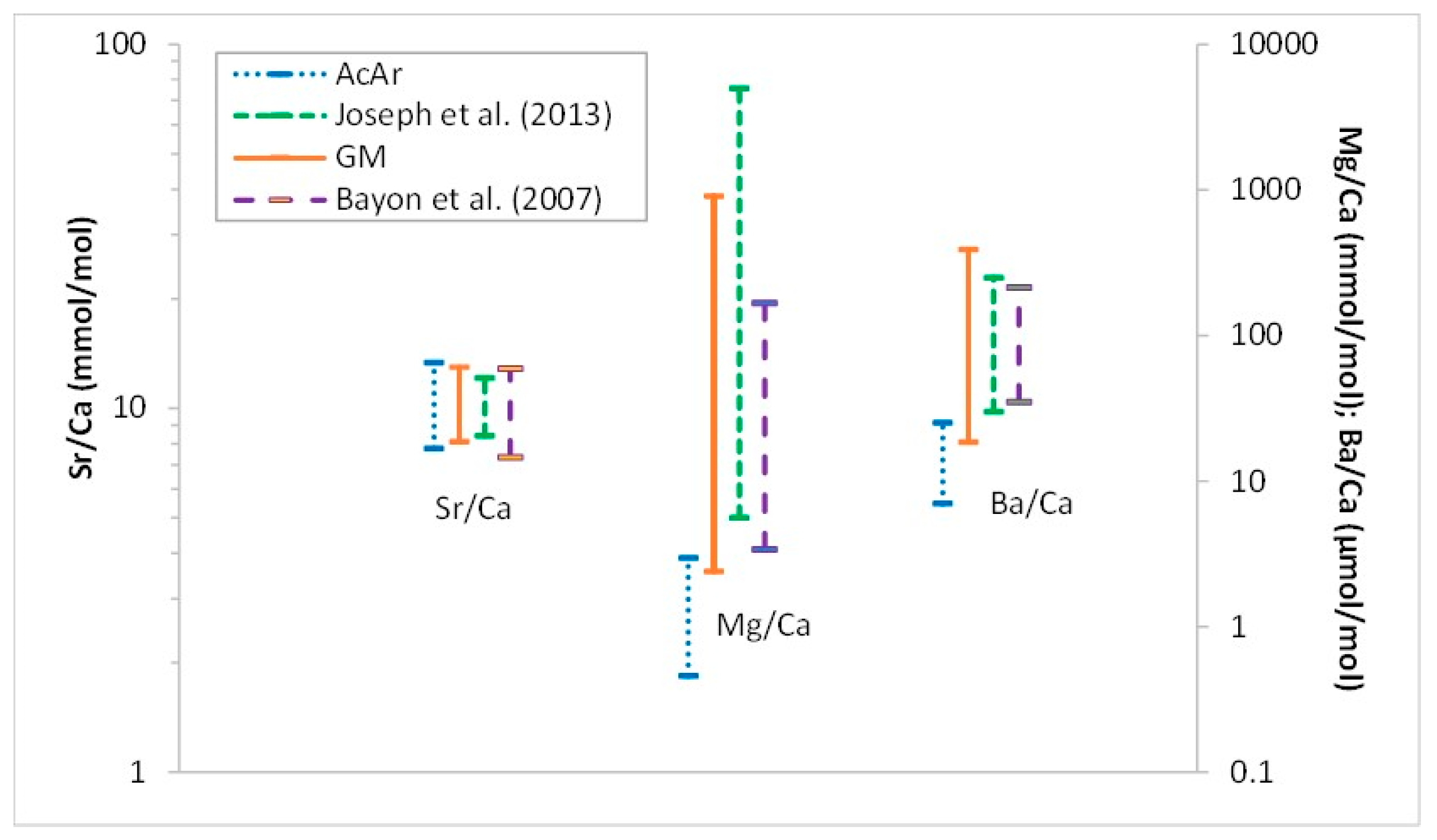
4.4. Minor and Trace Elements
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Greinert, J.; Bohrmann, G.; Suess, E. Gas hydrate-associated carbonates and methane-venting at Hydrate Ridge: Classification, distribution, and origin of authigenic lithologies. Geophys. Monogr. Am. Geophys. Union 2001, 124, 99–113. [Google Scholar]
- Sun, X.; Turchin, A.V. Significant contribution of authigenic carbonate to marine carbon burial. Nat. Geosci. 2014, 7, 201–204. [Google Scholar] [CrossRef]
- Suess, E.; Torres, M.E.; Bohrmann, G.; Collier, R.W.; Greinert, J.; Linke, P.; Rehder, G.; Trehu, A.; Wallmann, K.; Winckler, G.; et al. Gas hydrate destabilization: Enhanced dewatering, benthic material turnover and large methane plumes at the Cascadia convergent margin. Earth Planet. Sci. Lett. 1999, 170, 1–15. [Google Scholar] [CrossRef]
- Boetius, A.; Ravenschlag, K.; Schubert, C.J.; Rickert, D.; Widdel, F.; Gieseke, A.; Amann, R.; Jùrgensen, B.B.; Witte, U.; Pfannkuche, O. A marine microbial consortium apparently mediating anaerobic oxidation of methane. Nature 2000, 407, 623–626. [Google Scholar] [CrossRef] [PubMed]
- McVeigh, D.; Skarke, A.; Dekas, A.E.; Borrelli, C.; Hong, W.-L.; Marlow, J.J.; Pasulka, A.; Jungbluth, S.P.; Barco, R.A.; Djurhuus, A. Characterization of benthic biogeochemistry and ecology at three methane seep sites on the Northern U.S. Atlantic margin. Deep Sea Res. Part II Top. Stud. Oceanogr. 2018, 150, 41–56. [Google Scholar] [CrossRef]
- Bayon, G.; Henderson, G.M.; Bohn, M. U–Th stratigraphy of a cold seep carbonate crust. Chem. Geol. 2009, 260, 47–56. [Google Scholar] [CrossRef]
- Pierre, C.; Bayon, G.; Blanc-Valleron, M.-M.; Mascle, J.; Dupré, S. Authigenic carbonates related to active seepage of methane-rich hot brines at the Cheops mud volcano, Menes caldera (Nile deep-sea fan, eastern Mediterranean Sea). Geo-Mar. Lett. 2014, 34, 253–267. [Google Scholar] [CrossRef]
- Donda, F.; Forlin, E.; Gordini, E.; Panieri, G.; Buenz, S.; Volpi, V.; Civile, D.; De Santis, L. Deep-sourced gas seepage andmethane-derived carbonates in the Northern Adriatic Sea. Basin Res. 2015, 27, 531–545. [Google Scholar] [CrossRef]
- Himmler, T.; Birgel, D.; Bayon, G.; Pape, T.; Ge, L.; Bohrmann, G.; Peckmann, J. Formation of seep carbonates along the Makran convergent margin, northern Arabian Sea and a molecular and isotopic approach to constrain the carbon isotopic composition of parent methane. Chem. Geol. 2015, 415, 102–117. [Google Scholar] [CrossRef]
- Rongemaille, E.; Bayon, G.; Pierre, C.; Bollinger, C.; Chu, N.C.; Fouquet, Y.; Riboulot, V.; Voisset, M. Rare earth elements in cold seep carbonates from the Niger delta. Chem. Geol. 2011, 286, 196–206. [Google Scholar] [CrossRef]
- Feng, D.; Chen, D. Authigenic carbonates from an active cold seep of the northern South China Sea: New insights into fluid sources and past seepage activity. Deep-Sea Res. II 2015, 122, 74–83. [Google Scholar] [CrossRef]
- Loyd, S.J.; Sample, J.; Tripati, R.E.; Defliese, W.F.; Brooks, K.; Hovland, M.; Torres, M.; Marlow, J.; Hancock, L.G.; Martin, R.; et al. Methane seep carbonates yield clumped isotope signatures out of equilibrium with formation temperatures. Nat. Commun. 2016, 7, 12274. [Google Scholar] [CrossRef] [PubMed]
- Skarke, A.; Ruppel, C.; Kodis, M.; Brothers, D.; Lobecker, E. Widespread methane leakage from the seafloor on the northern US Atlantic margin. Nat. Geosci. 2014, 7, 657–661. [Google Scholar] [CrossRef]
- Prouty, N.G.; Sahy, D.; Ruppel, C.D.; Roark, E.B.; Condon, D.; Brooke, S.; Ross, S.W.; Demopoulos, A.W.J. Insights into methane dynamics from analysis of authigenic carbonates and chemosynthetic mussels at newly-discovered Atlantic Margin seeps. Earth Planet. Sci. Lett. 2016, 449, 332–344. [Google Scholar] [CrossRef]
- Fallon, S.J.; White, J.C.; McCulloch, M.T. Porites corals as recorders of mining and environmental impacts: Misima Island, Papua New Guinea. Geochim. Cosmochim. Acta 2002, 66, 45–62. [Google Scholar] [CrossRef]
- Hathorne, E.C.; Alard, O.; James, R.H.; Rogers, N.W. Determination of intratest variability of trace elements in foraminifera by laser ablation inductively coupled plasma-mass spectrometry. Geochem. Geophy. Geosyst. 2003, 4, 8408. [Google Scholar] [CrossRef]
- Barats, A.; Pécheyran, C.; Amouroux, D.; Debascoux, S.; Chauvaud, L.; Donard, O.F.X. Matrix-matched quantitative analysis of trace-elements in calcium carbonate shells by laser-ablation ICP–MS: Application to the determination of daily scale profiles in scallop shell (Pecten maximus). Anal. Bioanal. Chem. 2007, 387, 1131–1140. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Takahashi, Y.; Shimizu, H. Determination of rare earth element in carbonate using laser-ablation inductively-coupled plasma mass spectrometry: An examination of the influence of the matrix on laser-ablation inductively-coupled plasma mass spectrometry analysis. Anal. Chim. Acta 2007, 583, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Wark, D.A.; Watson, E.B. Grain-scale permeabilities of texturally equilibrated, monomineralic rocks. Earth Planet. Sci. Lett. 1998, 164, 591–605. [Google Scholar] [CrossRef]
- Woodhead, J.D.; Hellstrom, J.; Hergt, J.M.; Greig, A.; Maas, R. Isotopic and elemental imaging of geological materials by Laser Ablation Inductively Coupled Plasma-Mass Spectrometry. Geostand. Geoanal. Res. 2007, 31, 331–343. [Google Scholar] [CrossRef]
- Paton, C.; Hellstrom, J.; Paul, B.; Woodhead, J.; Hergt, J. Iolite: Freeware for the visualisation and processing of mass spectrometric data. J. Anal. Atom. Spectrom. 2011, 26, 2508–2518. [Google Scholar] [CrossRef]
- Outridge, P.M.; Doherty, W.; Gregoire, D.C. Ablative and transport fractionation of trace elements during laser sampling of glass and copper. Spectrochim. Acta B 1997, 52, 2093–2102. [Google Scholar] [CrossRef]
- Guillong, M.; Horn, I.; Günter, D. A comparison of 266 nm, 213 nm and 193 nm produced from a single solid state Nd:YAG laser for laser ablation ICP-MS. J. Anal. At. Spectrom. 2003, 18, 230. [Google Scholar] [CrossRef]
- Kim, S.-T.; O’Neil, J.R.; Hillaire-Marcel, C.; Mucci, A. Oxygen isotope fractionation between synthetic aragonite and water: Influence of temperature and Mg2+ concentration. Geochim. Cosmochim. Acta 2007, 71, 4704–4715. [Google Scholar] [CrossRef]
- Hesse, R.; Harrison, W.E. Gashydrates (clathrates) causing pore-water freshening and oxygen isotope fractionation in deep-water sedimentary sections of terrigenous continental margins. Earth Planet. Sci. Lett. 1981, 55, 453–462. [Google Scholar] [CrossRef]
- Sheppard, S.M.F.; Gilg, H.A. Stable isotope geochemistry of clay minerals. Clay Miner. 1996, 31, 1–24. [Google Scholar] [CrossRef]
- Tryon, M.D.; Henry, P.; Çağatay, M.N.; Zitter, T.A.C.; Géli, L.; Gasperini, L.; Burnard, P.; Bourlange, S.; Grall, C. Pore fluid chemistry of the North Anatolian Fault Zone in the Sea of Marmara: A diversity of sources and processes. Geochem. Geophys. Geosyst. 2010, 11, Q0AD03. [Google Scholar] [CrossRef]
- Crémière, A.; Bayon, G.; Ponzevera, E.; Pierre, C. Paleo-environmental controls on cold seep carbonate authigenesis in the Sea of Marmara. Earth Planet. Sci. Lett. 2013, 376, 200–211. [Google Scholar] [CrossRef]
- Testa, M. Imaging Calcium Carbonate Crystallization in Association with Organic Compounds. Ph.D. Thesis, Mississippi State University, Starkville, MS, USA, 2017. [Google Scholar]
- Burton, E.A. Controls on marine carbonate cement mineralogy: Review and reassessment. Chem. Geol. 1993, 105, 163–179. [Google Scholar] [CrossRef]
- Aloisi, G.; Bouloubassi, I.; Heijs, S.K.; Pancost, R.D.; Pierre, C.; Sinninghe Damsté, J.S.; Gottschal, J.C.; Forney, L.J.; Rouchy, J.-M. CH4-consuming microorganisms and the formation of carbonate crusts at cold seeps. Earth Planet. Sci. Lett. 2002, 203, 195–203. [Google Scholar] [CrossRef]
- Luff, R.; Wallmann, K. Fluid flow, methane fluxes, carbonate precipitation and biogeochemical turnover in gas hydrate-bearing sediments at Hydrate Ridge, Cascadia Margin: Numerical modeling and mass balances. Geochim. Cosmochim. Acta 2003, 3403–3421. [Google Scholar] [CrossRef]
- Nöthen, K.; Kasten, S. Reconstructing changes in seep activity by means of pore water and solid phase Sr/Ca and Mg/Ca ratios in pockmark sediments of the Northern Congo Fan. Mar. Geol. 2011, 287, 1–13. [Google Scholar] [CrossRef]
- Zhang, F.; Xu, H.; Konishi, H.; Kemp, J.M.; Roden, E.E.; Shen, Z. Dissolved sulfide-catalyzed precipitation of disordered dolomite: Implications for the formation mechanism of sedimentary dolomite. Geochim. Cosmochim. Acta 2012, 97, 148–165. [Google Scholar] [CrossRef]
- Zhang, F.; Yan, C.; Teng, H.H.; Roden, E.E.; Xu, H. In situ AFM observations of Ca–Mg carbonate crystallization catalyzed by dissolved sulfide: Implications for sedimentary dolomite formation. Geochim. Cosmochim. Acta 2013, 105, 44–55. [Google Scholar] [CrossRef]
- Canfield, D.E.; Farquhar, J. Animal evolution, bioturbation, and the sulfate concentration of the oceans. PNAS 2009, 106, 8123–8127. [Google Scholar] [CrossRef] [PubMed]
- Tostevin, R.; Turchyn, A.V.; Farquhar, J.; Johnston, D.T.; Eldridge, D.L.; Bishop, J.K.B.; McIlvin, M. Multiple sulfur isotope constraints on the modern sulfur cycle. Earth Planet. Sci. Lett. 2014, 396, 14–21. [Google Scholar] [CrossRef]
- Joseph, C.; Campbell, K.A.; Torres, M.E.; Martin, R.A.; Pohlman, J.W.; Riedel, M.; Rose, K. Methane-derived authigenic carbonates from modern and paleoseeps on the Cascadia margin: Mechanisms of formation and diagenetic signals. Palaeogeogr. Palaeoclim. Palaeoecol. 2003, 390, 52–67. [Google Scholar] [CrossRef]
- Bayon, G.; Pierre, C.; Etoubleau, J.; Voisset, M.; Cauquil, E.; Marsset, T.; Sultan, N.; Le Drezen, E.; Fouquet, Y. Sr/Ca and Mg/Ca ratios in Niger Delta sediments: Implications for authigenic carbonate genesis in cold seep environments. Mar. Geol. 2007, 241, 93–109. [Google Scholar] [CrossRef]
- Crémière, A.; Lepland, A.; Chand, S.; Sahy, D.; Kirsimäe, K.; Bau, M.; Whitehouse, M.J.; Noble, S.R.; Martma, T.; Thorsnes, T.; et al. Fluid source and methane-related diagenetic processes recorded in cold seep carbonates from the Alvheim channel, central North Sea. Chem. Geol. 2016, 432, 16–33. [Google Scholar] [CrossRef]
- Campbell, K.A. Hydrocarbon seep and hydrothermal vent paleoenvironments and paleontology: Past developments and future research directions. Palaeogeogr. Palaeoclim. Palaeoecol. 2006, 232, 362–407. [Google Scholar] [CrossRef]
- McGinnis, D.F.; Greinert, J.; Artemov, Y.; Beaubien, S.E.; Wüest, A. Fate of rising methane bubbles in stratified waters: How much methane reaches the atmosphere? J. Gephys. Res. 2006, 111, C09007. [Google Scholar] [CrossRef]
- Biastoch, A.; Treude, T.; Rüpke, L.H.; Riebesell, U.; Roth, C.; Burwicz, E.B.; Park, W.; Latif, M.; Böning, C.W.; Madec, G.; et al. Rising Arctic Ocean temperatures cause gas hydrate destabilization and ocean acidification. Geophys. Res. Lett. 2011, 38, L08602. [Google Scholar] [CrossRef]
- Naehr, T.H.; Eichhubl, P.; Orphan, V.J.; Hovland, M.; Paull, C.K.; Ussler, W.; Lorenson, T.D.; Greene, H.G. Authigenic carbonate formation at hydrocarbon seeps in continental margin sediments: A comparative study. Deep-Sea Res. II Top. Stud. Oceanogr. 2007, 54, 1268–1291. [Google Scholar] [CrossRef]
- Marlow, J.J.; Steele, J.A.; Ziebis, W.; Thurber, A.R.; Levin, L.A.; Orphan, V.J. Carbonate-hosted methanotrophy represents an unrecognized methane sink in the deep sea. Nat. Commun. 2014, 5, 5094. [Google Scholar] [CrossRef] [PubMed]

| ICP-MS in Laser-Ablation Mode | |
| Instrument | Aglient 7900 |
| Carrier gas flow rate | Ar: 1.3 L/min |
| Gas flow in the collision reaction cell | He: 0.5 mL/min |
| Isotopes | 23Na, 24,25,26Mg, 27Al, 29Si, 33,34S, 39K, 43,44Ca |
| Monitored | 54,56,58Fe, 55Mn, 88Sr, 90,91Zr, 137,138Ba, 235,238U |
| Background acquisition time | 60 s |
| Data acquisition time | 20 s |
| Laser-Ablation System | |
| Instrument | CETAC |
| Beam | 193 nm excimer |
| Fluence | 4.72 J/cm2 |
| Repetition rate | 5 Hz |
| Shot count | 150 |
| He gas flow rate | 0.6 L/min (MFC1) |
| 0.2 L/min (MFC2) | |
| Laser spot diameter | 40 µm |
| Spot # * | Mg/Ca | S/Ca | Sr/Ca | Ba/Ca | U/Ca |
|---|---|---|---|---|---|
| Slab-6 | |||||
| 42 | 0.791 | 0.986 | 13.366 | 25.292 | 2.195 |
| 43 | 0.531 | 0.765 | 11.264 | 13.703 | 0.870 |
| 56 | 2.965 | 1.258 | 11.367 | 13.776 | 2.994 |
| 57 | 0.914 | 0.812 | 11.081 | 15.379 | 1.783 |
| 58 | 0.610 | 0.734 | 9.253 | 8.747 | 0.690 |
| 59 | 2.039 | 1.133 | 11.355 | 16.254 | 2.044 |
| 67 | 1.849 | 0.908 | 11.652 | 14.432 | 2.271 |
| 70 | 1.771 | 0.833 | 10.487 | 13.484 | 1.114 |
| 75 | 2.092 | 0.599 | 10.624 | 13.994 | 1.817 |
| average | 1.507 | 0.892 | 11.161 | 15.007 | 1.753 |
| s.d. | 0.834 | 0.206 | 1.094 | 4.381 | 0.743 |
| Slab-7 | |||||
| 24 | 0.684 | 0.599 | 8.933 | 6.997 | 0.891 |
| 40 | 0.459 | 0.656 | 10.453 | 8.747 | 0.673 |
| 41 | 0.659 | 0.699 | 7.768 | 9.694 | 1.165 |
| 42 | 0.618 | 0.668 | 7.974 | 11.662 | 1.034 |
| 43 | 0.457 | 0.640 | 10.396 | 9.038 | 0.576 |
| 44 | 0.651 | 0.659 | 10.293 | 10.496 | 0.984 |
| 45 | 0.675 | 0.652 | 8.134 | 10.715 | 1.009 |
| average | 0.600 | 0.653 | 9.136 | 9.621 | 0.905 |
| s.d. | 0.099 | 0.030 | 1.220 | 1.534 | 0.210 |
| Samples | δ13CVPDB | 1σ | δ18OVPDB | 1σ |
|---|---|---|---|---|
| LR-1 | −50.25 | 0.08 | 4.53 | 0.1 |
| LR-2 | −49.81 | 0.08 | 4.50 | 0.1 |
| LR-3 | −50.19 | 0.08 | 4.53 | 0.1 |
| average | −50.09 | 4.52 | ||
| 1s.d. | 0.24 | 0.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gabitov, R.; Borrelli, C.; Buettner, J.; Kirkland, B.; Skarke, A.; Trail, D.; Garner, B.; Testa, M.; Wahidi, M.; Hoff, C.; et al. Characterization of Carbonate Crust from a Recently Discovered Methane Seep on the North Atlantic Continental Margin of the USA. Minerals 2019, 9, 138. https://doi.org/10.3390/min9030138
Gabitov R, Borrelli C, Buettner J, Kirkland B, Skarke A, Trail D, Garner B, Testa M, Wahidi M, Hoff C, et al. Characterization of Carbonate Crust from a Recently Discovered Methane Seep on the North Atlantic Continental Margin of the USA. Minerals. 2019; 9(3):138. https://doi.org/10.3390/min9030138
Chicago/Turabian StyleGabitov, Rinat, Chiara Borrelli, Jacob Buettner, Brenda Kirkland, Adam Skarke, Dustin Trail, Brittany Garner, Maurice Testa, Mahnaz Wahidi, Christopher Hoff, and et al. 2019. "Characterization of Carbonate Crust from a Recently Discovered Methane Seep on the North Atlantic Continental Margin of the USA" Minerals 9, no. 3: 138. https://doi.org/10.3390/min9030138
APA StyleGabitov, R., Borrelli, C., Buettner, J., Kirkland, B., Skarke, A., Trail, D., Garner, B., Testa, M., Wahidi, M., Hoff, C., Khasanov, S., Panieri, G., Thirumalai, R., Thomas, J., Weremeichik, J., & Zverkova, I. (2019). Characterization of Carbonate Crust from a Recently Discovered Methane Seep on the North Atlantic Continental Margin of the USA. Minerals, 9(3), 138. https://doi.org/10.3390/min9030138





