Exceptional Preservation of Fungi as H2-Bearing Fluid Inclusions in an Early Quaternary Paleo-Hydrothermal System at Cape Vani, Milos, Greece
Abstract
1. Introduction
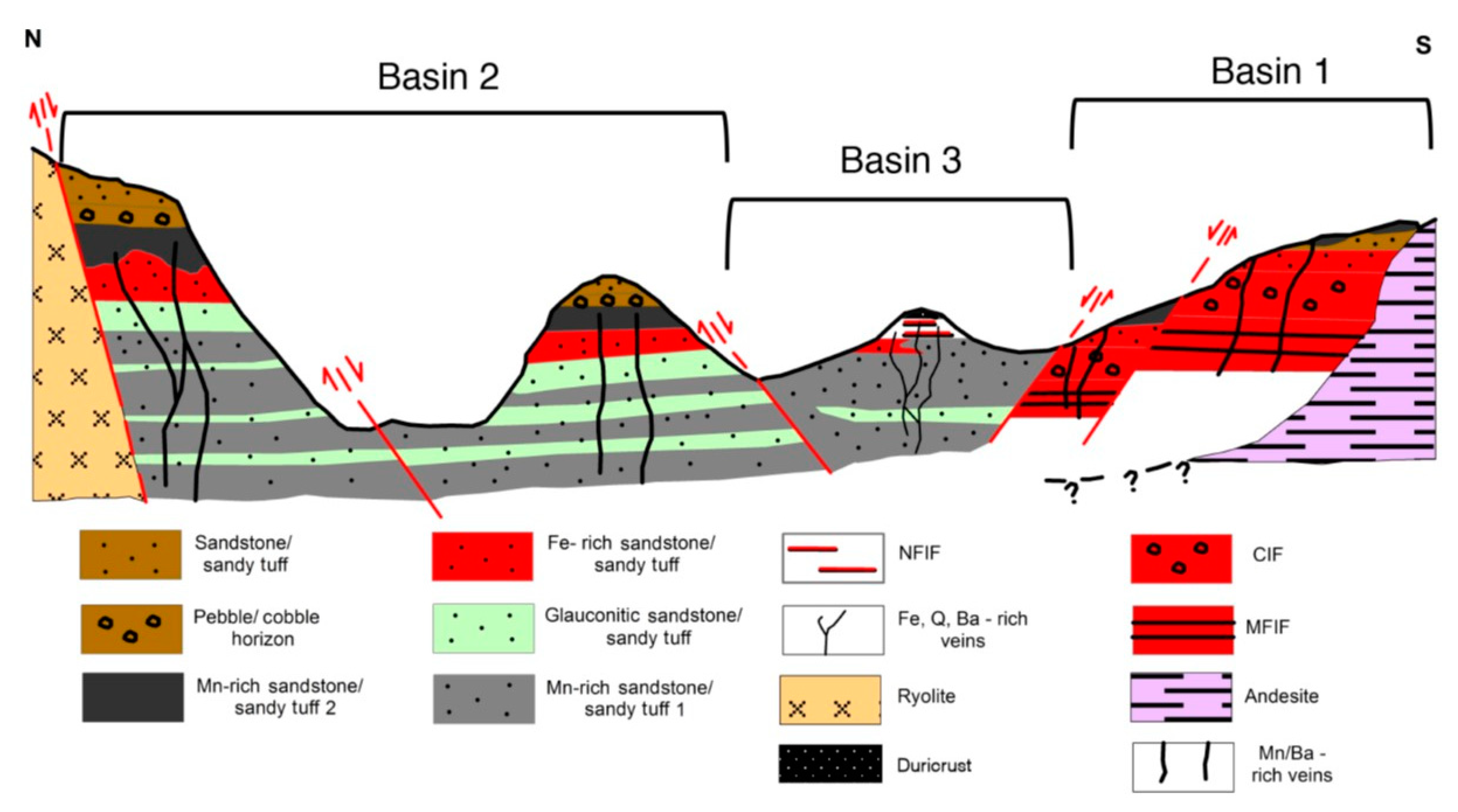
Geology and Mineralization of Milos
- (i)
- disseminated crystals or cement in Mn-mineralized sandstone/sandy tuff;
- (ii)
- quartz-barite (chalcedony, Mn oxide, K-feldspar) hydrothermal veins showing epithermal textures. These occur throughout the stratigraphy, crosscutting both the dacite/andesite basement and the overlying Mn-mineralized sediments, where they become more rich in barite and Mn-oxide;
- (iii)
- bedding-conformable barite (±Mn oxide, silica)-rich horizons underlain by mineralogically similar discordant pipe-like bodies; and
- (iv)
2. Materials and Methods
2.1. Microthermometry
2.2. Raman Spectroscopy
2.3. Staining with WGA-FITC and Fluorescence Microscopy
2.4. Stable Isotopes
3. Results
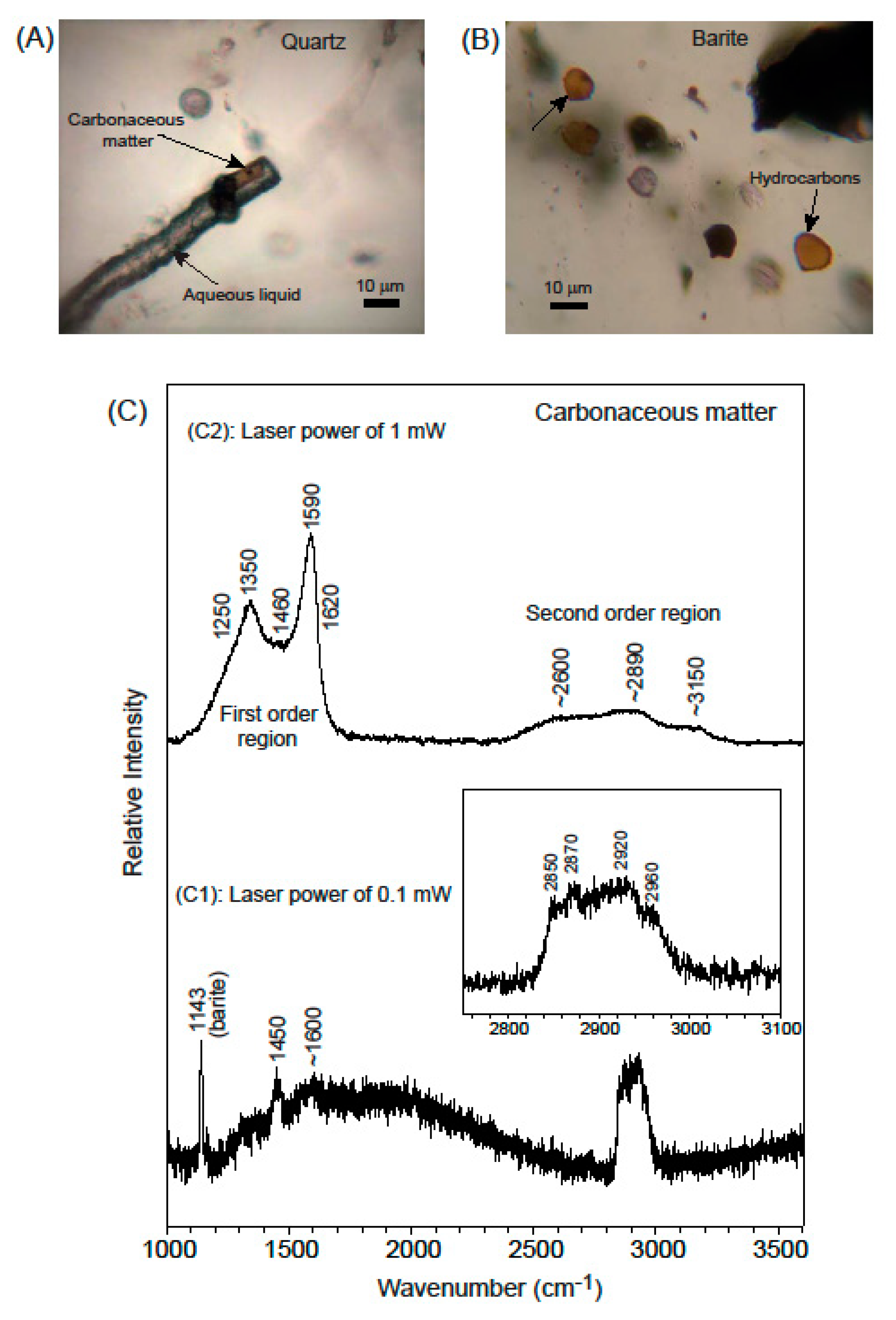
3.1. Mineralogy
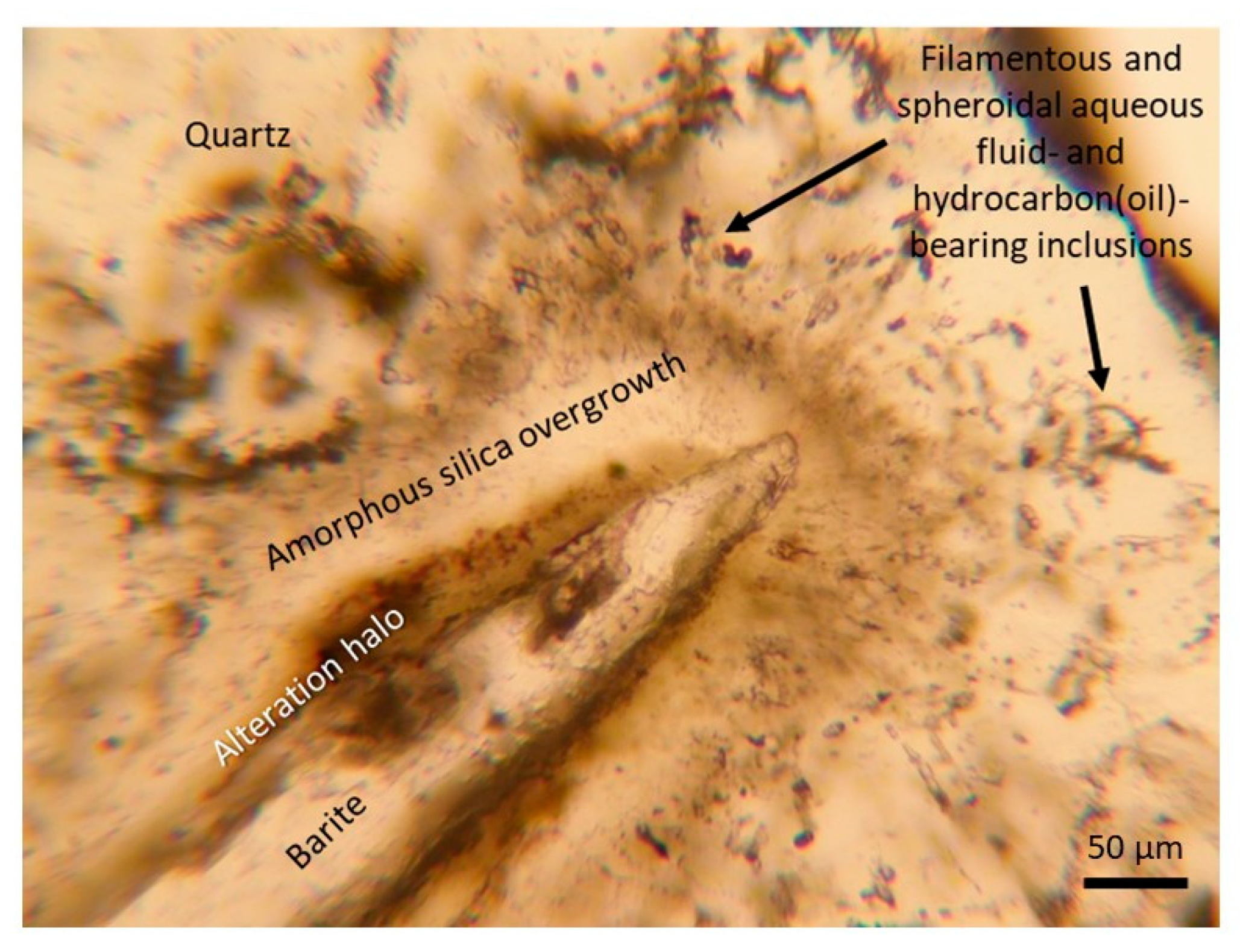
3.2. Fluid Inclusion Petrography
3.2.1. Type I
3.2.2. Type II
3.2.3. Type III
3.3. Fluid Inclusion Analyses
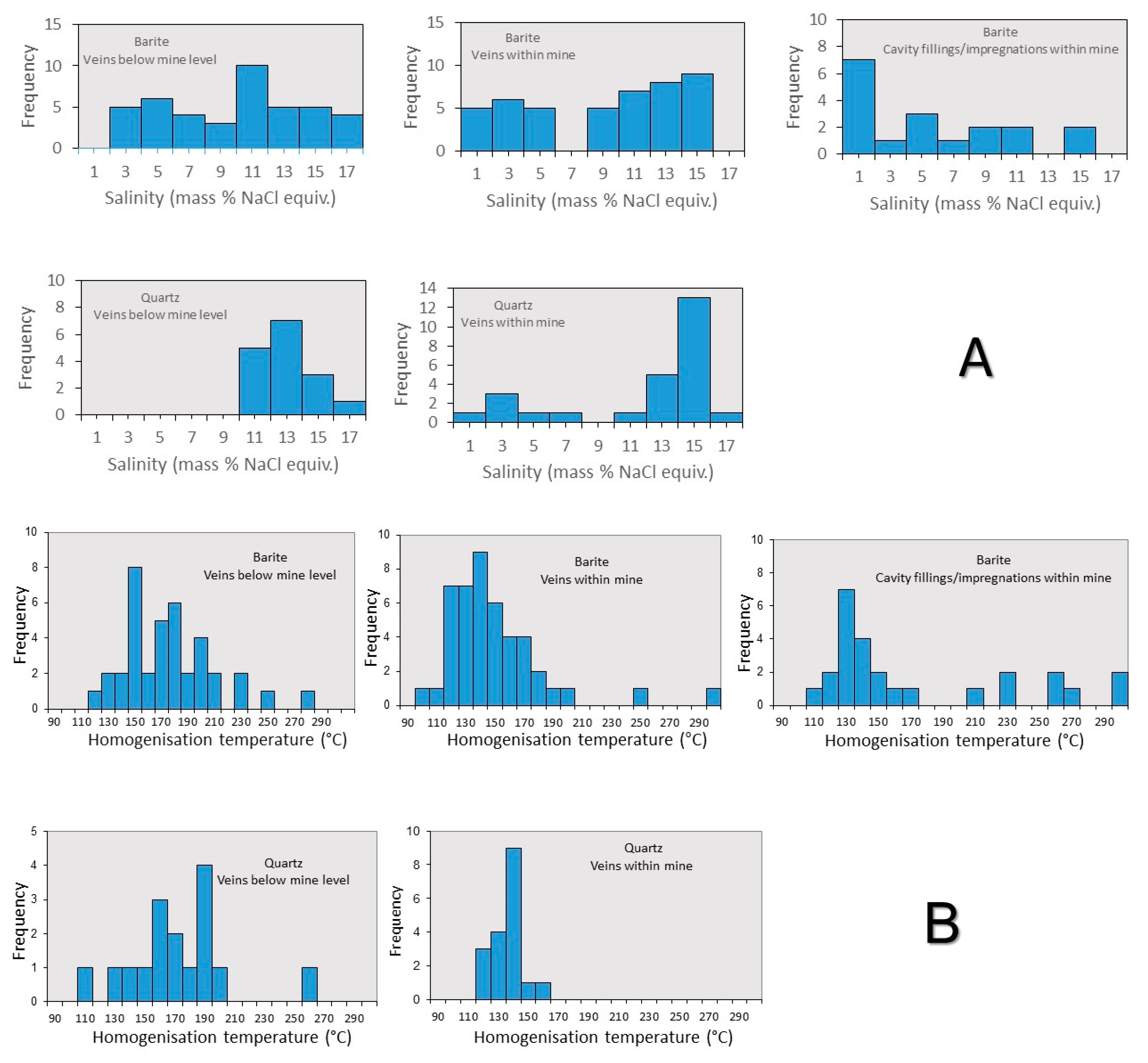
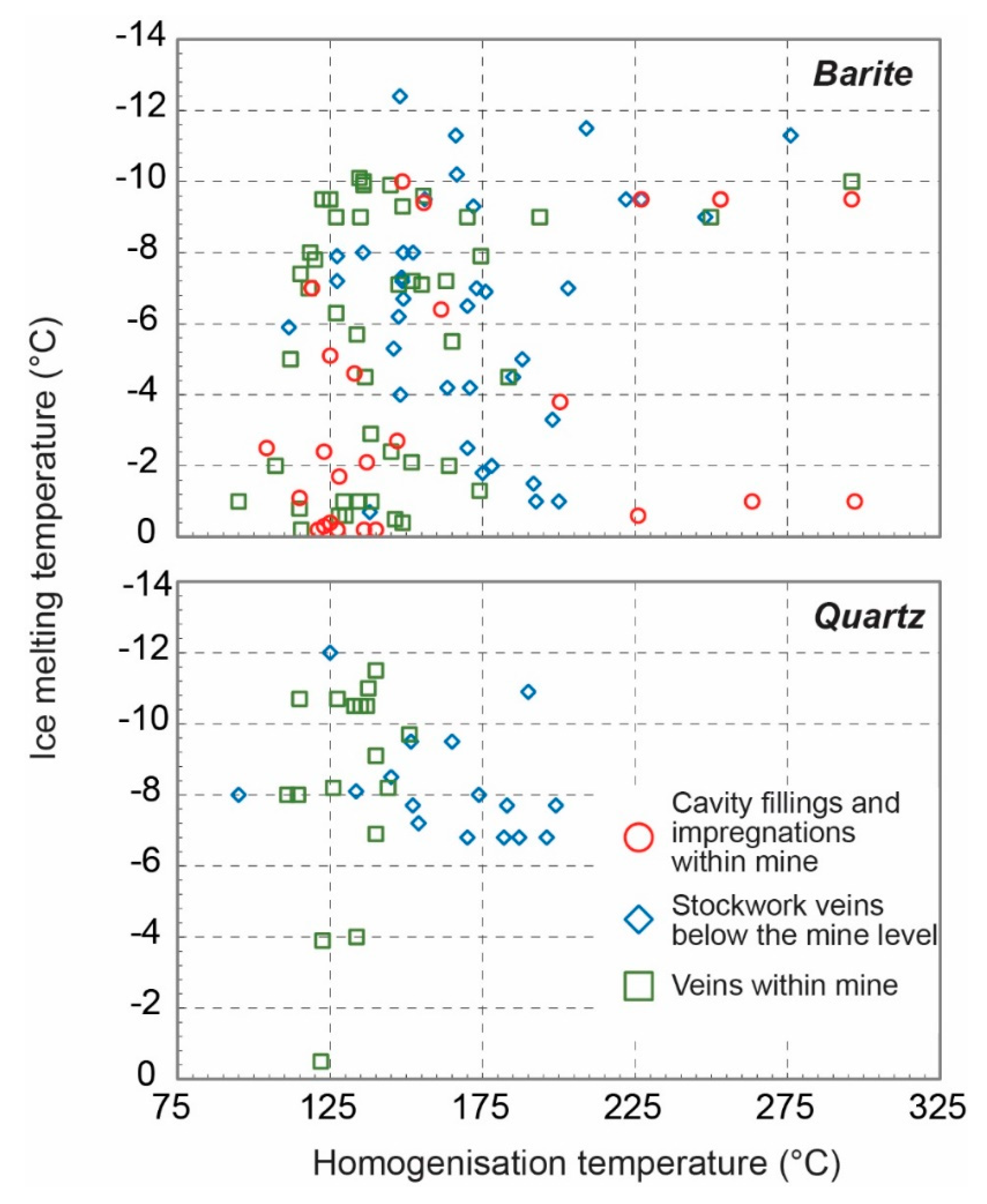
3.3.1. Microthermometry
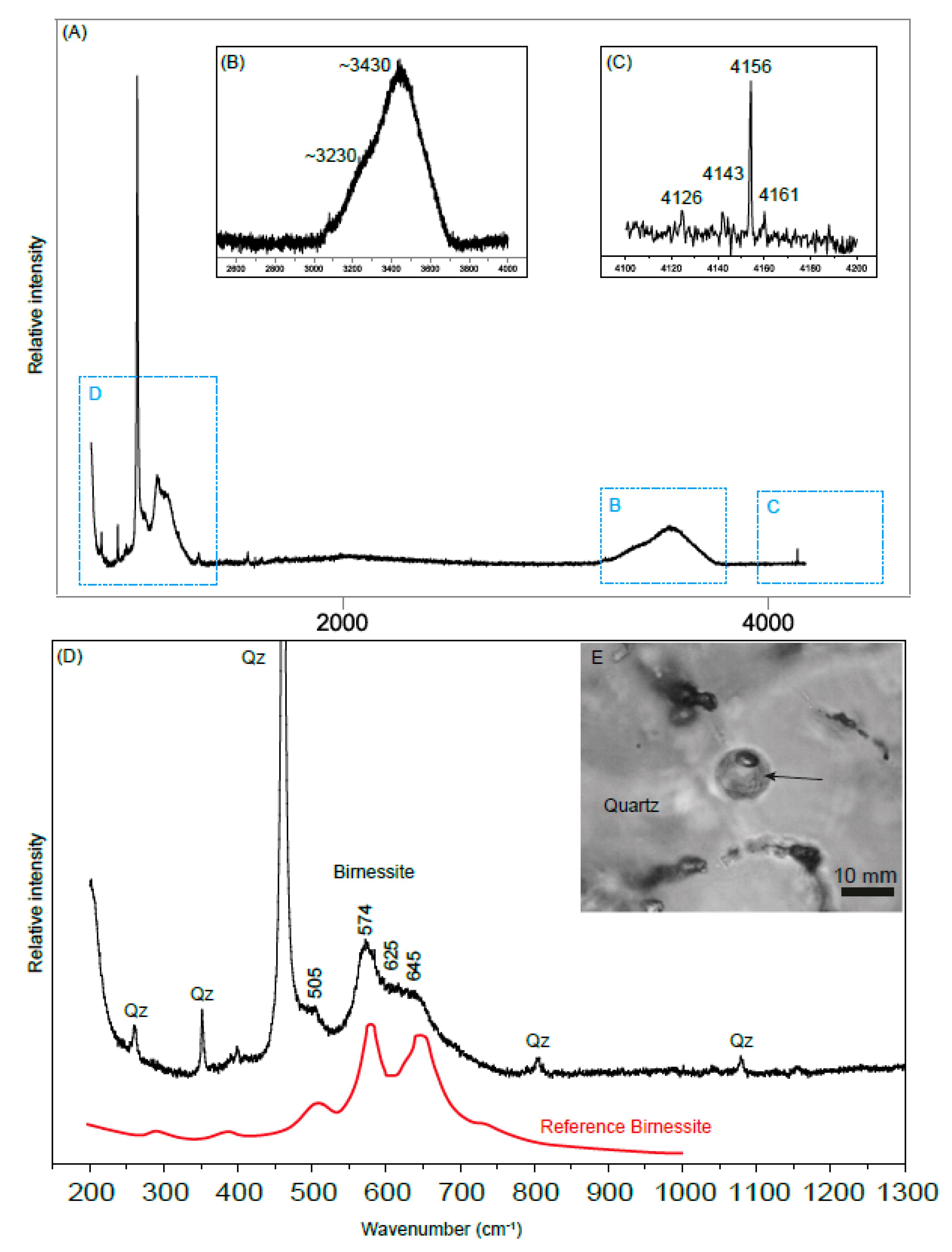
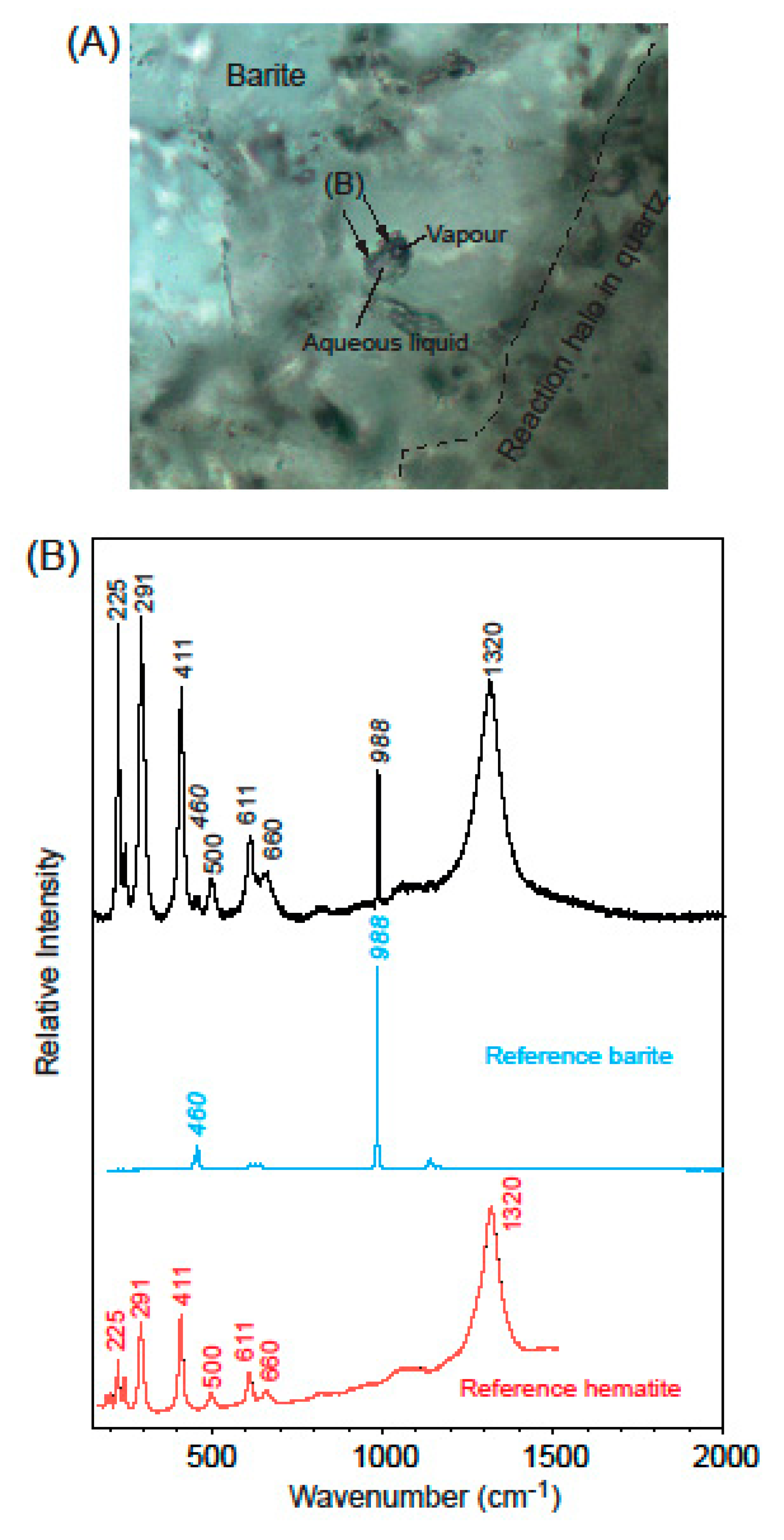
3.3.2. Raman Spectroscopy
- (i)
- (ii)
- funnel-shaped Type II inclusions in barite, that are filled with a pale yellow-amber to yellow-brownish phase (Figure 8B).
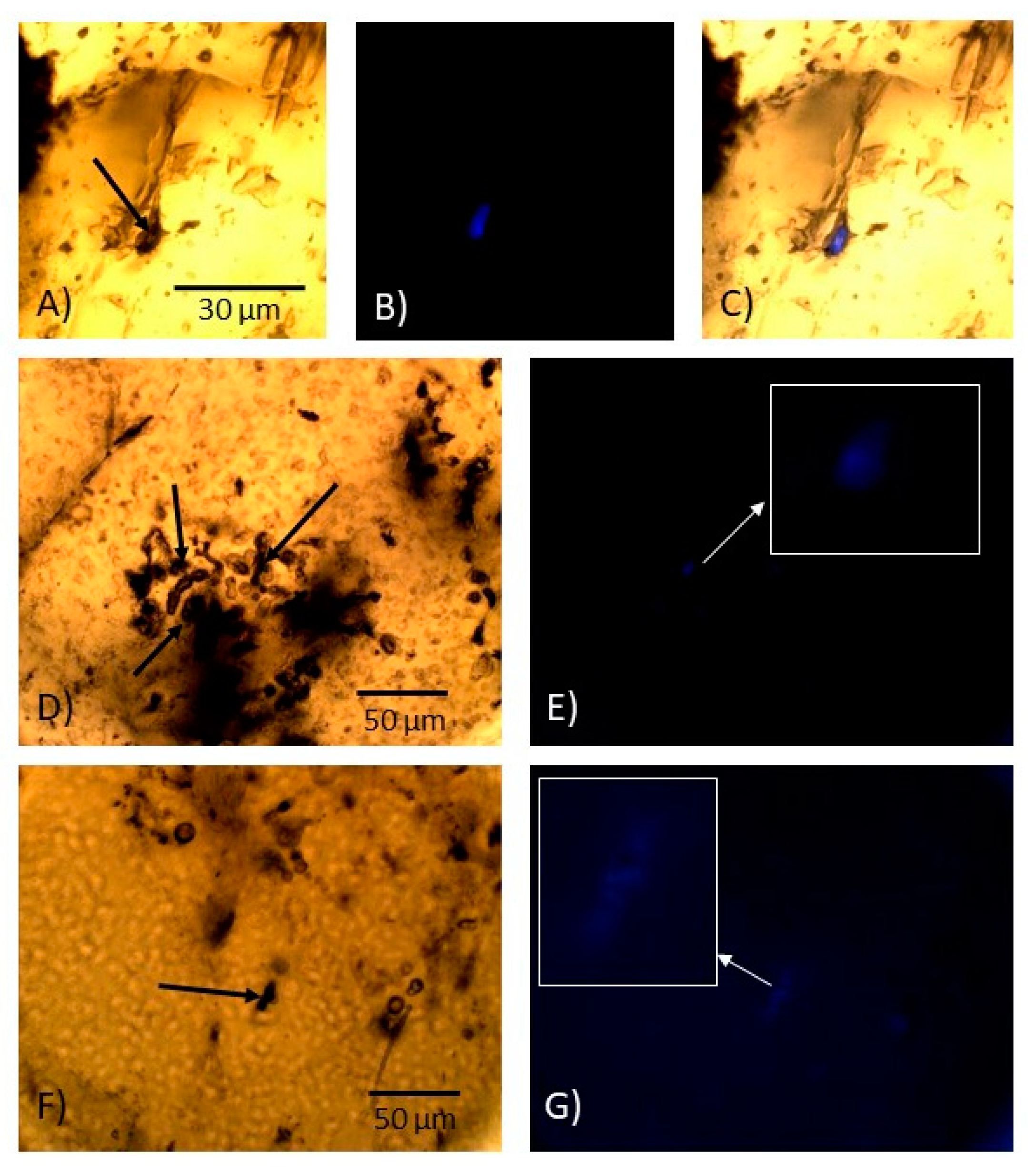
3.3.3. Staining with WGA-FITC and Fluorescence Microscopy
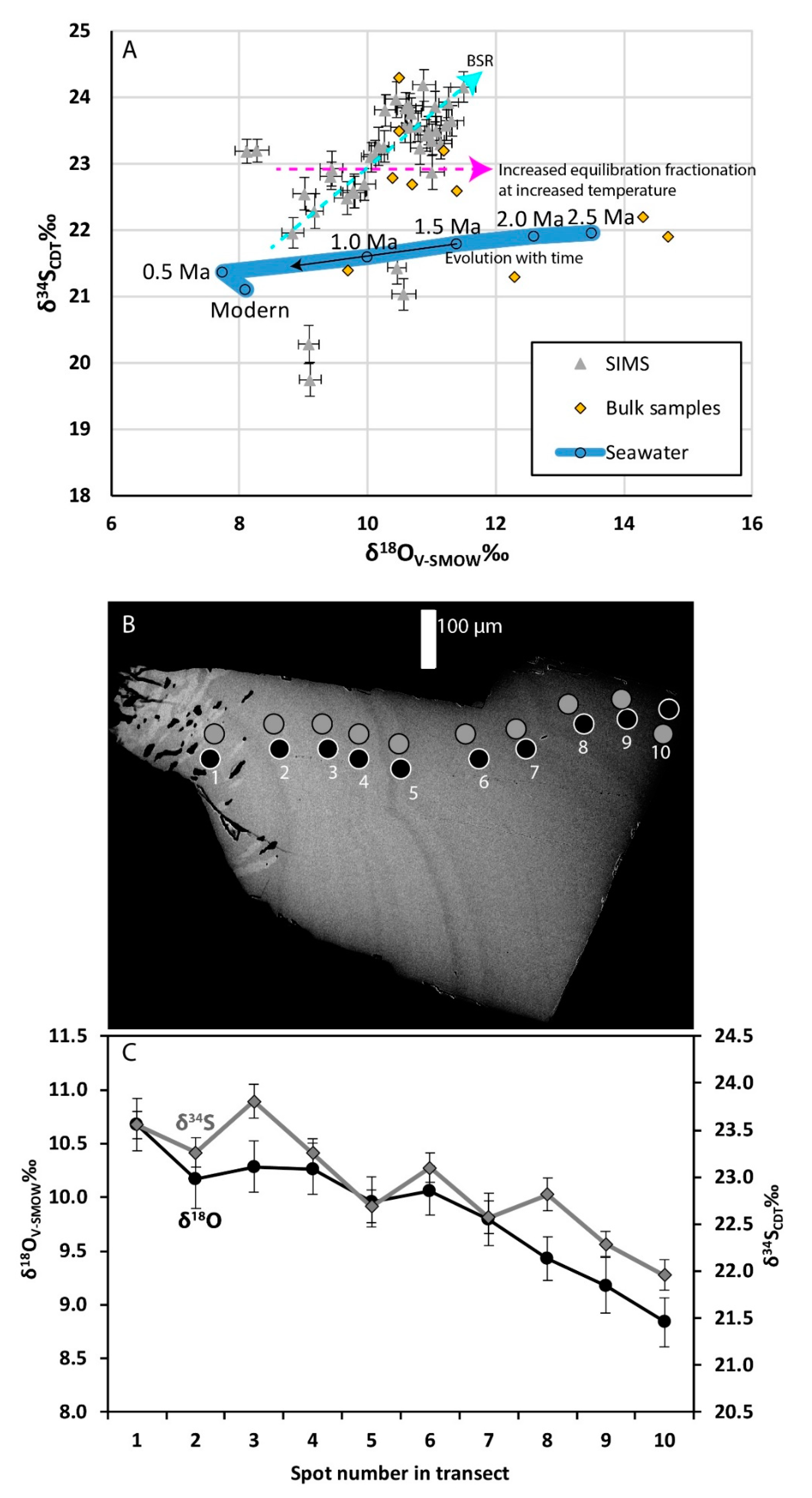
3.4. Stable Isotopes
3.4.1. Carbon Isotopes
3.4.2. Sulfur and Oxygen Isotopes in Barite
4. Discussion
4.1. The Paleoenvironment of the Hydrothermal System
4.1.1. Interpretation of the Fluid Inclusion Data
- (i)
- Homogenization data in barite show significant scatter towards high temperatures, most likely due to some post trapping modification and leakage during heating measurements. Notwithstanding this, there is a general trend of high (−1 °C) to low (−10 °C) Tm-ice at relatively constant Th (125–150 °C). This is much clearer in the quartz-hosted fluid inclusions from the mine samples.
- (ii)
- Comparing mine and stockwork fluid inclusion data samples, in spite of the scatter of the barite data it would seem that fluid inclusions in the stockwork samples have higher Th (25–50 °C) than fluid inclusions in samples from the mine.
- (iii)
- The trend of increasing salinity at relatively constant or falling Th is analogous to that recorded in the nearby Profitis Ilias and Chondro Vouno Au-Ag deposits, where it is interpreted to be the result of extensive open system boiling [34,68] and the Triades Pb-Zn-(Ag) deposit [36]. In these deposits, the highest homogenization temperatures and lowest salinities were interpreted to represent the composition of the hydrothermal fluid at initial boiling with the higher salinity lower Th inclusions characterizing the boiled residual fluid.
- (iv)
- Though most of the data exhibit salinities in excess of seawater (Tm-ice < −2.1 °C), some inclusions exhibit Tm-ice above −1.0 °C (1.5 wt.% NaCl eq.). This is somewhat like the low salinity–high Th inclusions recorded at Profitis Ilias [68]. At Profitis Ilias, this was interpreted to result for the trapping of condensed vapor, and a similar origin is thought to be responsible for these fluid inclusions at Cape Vani.
4.1.2. Pressure–Temperature Considerations
4.1.3. Interpretation of the S and O Isotopes in Barite
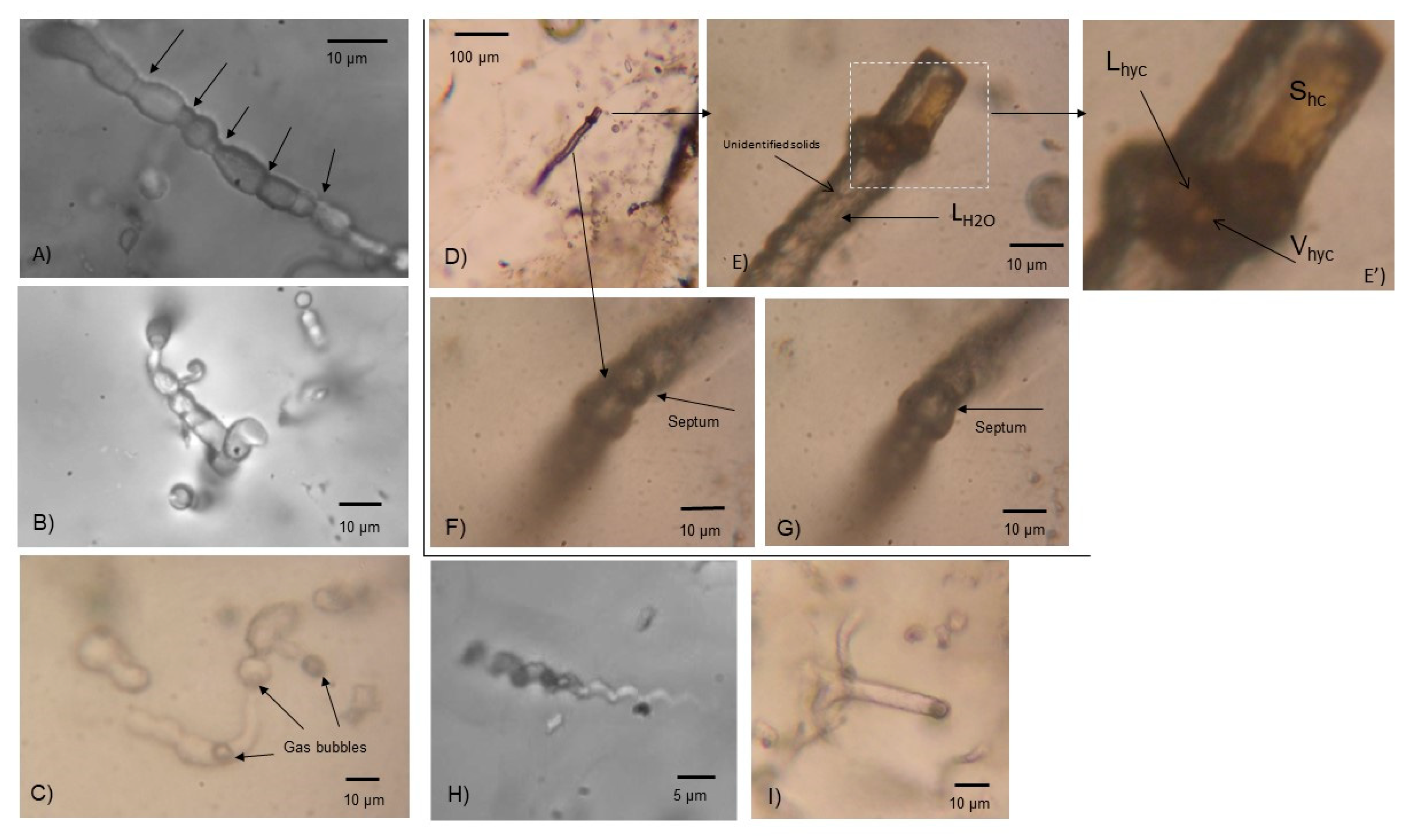
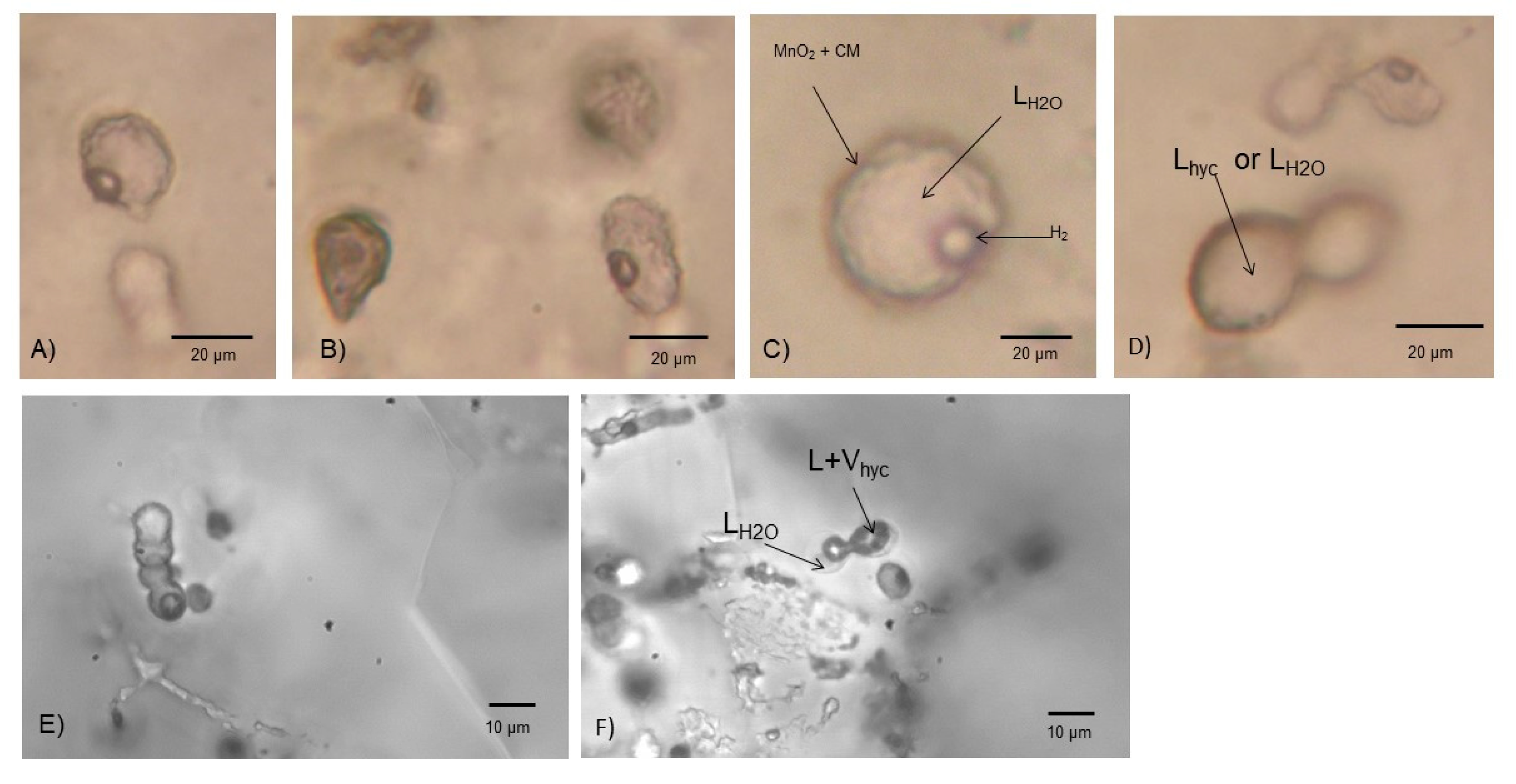
4.2. Microbial Casts
4.3. Mn-Coating of Spheriodal Type III Inclusions
4.4. H2-Content of Spheriodal Type III Inclusions
4.5. Biological Production of H2 in Hydrothermal Environments
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Orcutt, B.N.; Sylvan, J.B.; Knab, N.J.; Edwards, K.J. Microbial ecology of the dark ocean above, at, and below the seafloor. Microbiol. Mol. Biol. Rev. 2011, 75, 361–422. [Google Scholar] [CrossRef] [PubMed]
- Edgcomb, V.P.; Kysela, D.T.; Teske, A.; de vera Gomez, A.; Sogin, M.L. Benthic eukaryotic diversity in the Guaymas Basin hydrothermal vent environment. Proc. Natl. Acad. Sci. USA 2002, 99, 7658–7662. [Google Scholar] [CrossRef] [PubMed]
- López-García, P.; Vereshchaka, A.; Moreira, D. Eukaryotic diversity associated with carbonates and fluid-seawater interface in Lost-City hydrothermal field. Environ. Microbiol. 2007, 9, 546–554. [Google Scholar] [CrossRef] [PubMed]
- López-García, P.; Phillipe, H.; Gaill, F.; Moreira, D. Autochthonus eukaryotic diversity in hydrothermal sediment and experimental micro-colonizers at the Mid-Atlantic Ridge. Proc. Natl. Acad. Sci. USA 2003, 100, 697–702. [Google Scholar] [CrossRef]
- Nagano, Y.; Nagahama, T. Fungal diversity in deep-sea extreme environments. Fung. Ecol. 2012, 5, 463–471. [Google Scholar] [CrossRef]
- Hirayama, H.; Abe, M.; Miyazaki, J.; Sakai, S.; Nagano, Y.; Takai, K. Data Report: Cultivation of microorganisms from basaltic rock and sediment cores from the North Pond on the Western Flank of the Mid-Atlantic Ridge, IODP Expedition 336. Proc. Int. Ocean Drill. Prog. 2015, 336. [Google Scholar] [CrossRef]
- Ivarsson, M.; Holm, N.G.; Neubeck, A. The deep biosphere of the subseafloor igneous crust. In Trace Metal Biogeochemsitry and Ecology of Deep-Sea Hydrothermal Vent Systems; Demina, L.L., Galkin, S.V., Eds.; Springer: Berlin, Germany, 2015; pp. 143–166. [Google Scholar]
- Ivarsson, M.; Bengtson, S.; Drake, H.; Francis, W. Fungi in deep subsurface environments. In Advances in Applied Microbiology; Sariaslani, S., Gadd, G., Eds.; Academic Press: London, UK; Elsevier: London, UK, 2018; Volume 102, pp. 83–116. [Google Scholar]
- Bengtson, S.; Rasmussen, B.; Ivarsson, M.; Muhling, J.; Broman, C.; Marone, F.; Stampanoni, M.; Bekker, A. Fungus-like mycelial fossils in 2.4 billion-year-old vesicular basalt. Nat. Ecol. Evol. 2017, 1, 0141. [Google Scholar] [CrossRef]
- Ivarsson, M.; Bengtson, S.; Belivanova, V.; Stampanoni, M.; Marone, F.; Tehler, A. Fossilized fungi in subseafloor Eocene basalts. Geology 2012, 40, 163–166. [Google Scholar] [CrossRef]
- Drake, H.; Ivarsson, M. The role of anaerobic fungi in fundamental biogeochemical cycles in the deep biosphere. Fungal Biol. Rev. 2018, 32, 20–25. [Google Scholar] [CrossRef]
- Webster, J.; Weber, R.W.S. Introduction to Fungi, 3rd ed.; Cambridge University: Cambridge, UK, 2007; p. 875. [Google Scholar]
- Gadd, G. Geomycology: Biogeochemical transformations of rocks, minerals, metals and radionuclides by fungi, bioweathering and bioremediation. Mycol. Res. 2007, 111, 3–49. [Google Scholar] [CrossRef]
- Bengtson, S.; Ivarsson, M.; Astolfo, A.; Belivanova, V.; Broman, C.; Marone, F.; Stampanoni, M. Deep-biosphere consortium of fungi and prokaryotes in Eocene sub-seafloor basalts. Geobiology 2014, 12, 489–496. [Google Scholar] [CrossRef]
- Ivarsson, M.; Broman, C.; Gustafsson, H.; Holm, N.G. Biogenic Mn-oxides in subseafloor basalts. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Ivarsson, M.; Peckmann, J.; Tehler, A.; Broman, C.; Bach, W.; Behrens, K.; Reitner, J.; Böttcher, M.E.; Norbäck-Ivarsson, L. Zygomycetes in vesicular basanites from Vesteris Seamount, Greenland Basin—A new type of cryptoendolithic fungi. PLoS ONE 2015, 10. [Google Scholar] [CrossRef]
- Liggenstoffer, A.S.; Youssef, N.H.; Couger, M.B.; Elsahed, M.S. Phylogenetic diversity and community structure of anaerobic gut fungi (phylum Neocallimastigomycota) in ruminant and non-ruminant herbivores. ISME J. 2010, 4, 1225–1235. [Google Scholar] [CrossRef]
- Khejornsart, P.; Wanapat, M. Diversity of rumen anaerobic fungi and methanogenic archaea in swamp buffalo influenced by various diets. J. Anim. Vet. Adv. 2010, 9, 3062–3069. [Google Scholar]
- Mountfort, D.O.; Asher, R.A.; Bauchop, T. Fermentation of cellulose to methane and carbon dioxide by a rumen anaerobic fungus in a triculture with Methanobrevibacter sp. Strain RA1 and Methanosarcina barkeri. Appl. Environ. Microbiol. 1982, 44, 128–134. [Google Scholar] [PubMed]
- Hook, S.E.; Wright, A.-D.G.; McBride, B.W. Methanogens: Methane producers of the rumen and mitigation strategies. Archaea 2010, 945785. [Google Scholar] [CrossRef] [PubMed]
- Ivarsson, M.; Schnürer, A.; Bengtson, S.; Neubeck, A. Anaerobic fungi: A potential source of biological H2 in the oceanic crust. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Drake, H.; Ivarsson, M.; Bengtson, M.; Heim, C.; Siljeström, S.; Whitehouse, M.; Broman, C.; Belivanova, V.; Åström, M.E. Anaerobic consortia of fungi and sulfate reducing bacteria in deep granite fractures. Nat. Commun. 2017, 8. [Google Scholar] [CrossRef]
- Stevens, T.O.; McKinley, J.P. Lithoautotrophic microbial ecosystems in deep basalt aquifers. Science 1995, 270, 450–454. [Google Scholar] [CrossRef]
- Lin, L.-H.; Slater, G.F.; Sherwood Lollar, B.; Lacrampe-Couloume, G.; Onstott, T.C. The yield and isotopic composition of radiolytic H2, a potential energy source for the deep subsurface biosphere. Geochim. Cosmochim. Acta 2005, 69, 893–903. [Google Scholar] [CrossRef]
- Pedersen, K. Metabolic activity of subterranean microbial communities in deep granitic groundwater supplemented with methane and H2. ISME J. 2012, 7, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Lau, M.C.; Kieft, T.L.; Kuloyo, O.; Linage-Alvarez, B.; van Heerden, E.; Lindsay, M.R.; Magnabosco, C.; Wang, W.; Wiggins, J.B.; Guo, L.; et al. An oligotrophic deep-subsurface community dependent on syntrophy is dominated by sulfur-driven autotrophic denitrifiers. Proc. Natl Acad. Sci. USA 2016, 113, E7927–E7936. [Google Scholar] [CrossRef] [PubMed]
- McCollom, T.M. Abiotic methane formation during experimental serpentinization of olivine. Proc. Natl Acad. Sci. USA 2016, 113, 13965–13970. [Google Scholar] [CrossRef]
- McCollom, T.M. Geochemical constraints on sources of metabolic energy for chemolithoautotrophy in ultramafic-hosted deep-sea hydrothermal systems. Astrobiology 2007, 7, 933–950. [Google Scholar] [CrossRef]
- Anderson, R.T.; Chapelle, F.H.; Lovley, D.R. Evidence against hydrogen-based microbial ecosystems in basalt aquifers. Science 1998, 281, 976–977. [Google Scholar] [CrossRef]
- Gregory, S.P.; Barnett, M.J.; Field, L.P.; Milodowski, A.E. Subsurface microbial hydrogen cycling: Natural occurrence and implications for industry. Microorganisms 2019, 7, 53. [Google Scholar] [CrossRef]
- Fytikas, M.; Innocenti, F.; Kolios, N.; Manetti, P.; Mazzuoli, R.; Poli, G.; Rita, F.; Villari, L. Volcanology and petrology of volcanic products from the island of Milos and Neighbouring islets. J. Vol. Geotherm. Res. 1986, 28, 297–317. [Google Scholar] [CrossRef]
- Stewart, A.L.; McPhie, J. Facies architecture and Late Ploicene—Pleistocene evolution of a felsic volcanic island, Milo, Greece. Bull. Volcanol. 2006, 68, 703–726. [Google Scholar] [CrossRef]
- Hannington, M.D.; de Ronde, C.E.J.; Petersen, S. Sea-floor tectonics and submarine hydrothermal systems. Econ. Geol. 100th Anniv. Vol. 2005, 100, 111–142. [Google Scholar]
- Naden, J.; Kilias, S.P.; Darbyshire, D.P.F. Active geothermal systems with entrained seawater as modern analogs for transitional volcanic-hosted massive sulphide and continental magmato-hydrothermal mineralization: The example of Milos Island, Greece. Geology 2005, 33, 541–544. [Google Scholar] [CrossRef]
- Alfieris, D.; Voudouris, P.; Spry, P.G. Shallow submarine epithermal Pb–Zn–Cu–Au–Ag–Te mineralization on western Milos Island, Aegean Volcanic Arc, Greece: Mineralogical, geological and geochemical constraints. Ore Geol. Rev. 2013, 53, 159–180. [Google Scholar] [CrossRef]
- Smith, D.J.; Naden, J.; Miles, A.-J.; Bennet, H.; Bicknell, S.H. Mass wasting events and their impact on the formation and preservation of submarine deposits. Ore Geol. Rev. 2018, 7, 143–151. [Google Scholar] [CrossRef]
- Hein, J.R.; Stamatakis, M.G.; Dowling, J.S. Trace metal-rich Quaternary hydrothermal manganese oxide and barite deposit, Milos Island, Greece. Appl. Earth Sci. 2000, 109, 67–76. [Google Scholar] [CrossRef]
- Liakopoulos, A.; Glasby, G.P.; Papavassiliou, C.T.; Boulegue, J. Nature and origin of the Vani manganese deposit, Milos, Greece: An overview. Ore Geol. Rev. 2001, 18, 181–209. [Google Scholar] [CrossRef]
- Chi Fru, E.; Kilias, S.; Ivarsson, M.; Rattray, J.E.; Gkika, K.; McDonald, I.; He, Q.; Broman, C. Sedimentary mechanisms of a modern banded iron formation on Milos Island, Greece. Solid Earth 2018, 9, 573–598. [Google Scholar] [CrossRef]
- Dando, P.R.; Hughes, J.A.; Leahy, Y.; Niven, S.J.; Taylor, L.J.; Smith, C. Gas venting from submarine hydrothermal areas around island of Milos, Hellenic Volcanic Arc. Cont. Shelf Res. 1995, 15, 913–929. [Google Scholar] [CrossRef]
- Kilias, S.P. Microbial-mat related structures in the Quaternary Cape Vani manganese oxide (-barite) deposit, NW Milos island-Greece. In Microbial Mats in Siliciclastic Depositional Systems through Time; SEPM Special Publication: Athen, Greece, 2012; Volume 101, pp. 97–110. [Google Scholar]
- Kilias, S.P.; Detsi, K.; Godelitsas, A.; Typas, M.; Naden, J.; Marrantos, Y. Evidence of Mn-oxide biomineralization, Vani Mn deposit, Milos, Greece. In Proceedings of the Ninth Bienniela SGA Meeting, Dublin, UK, 20–23 August 2007; Andrew, C.J., Ed.; Digging Deeper: Dublin, UK, 2007; pp. 1069–1072. [Google Scholar]
- Skarpelis, N.; Koutles, T. Geology of epithermal mineralization of the NW part of Milos Island, Greece. In Proceedings of the 5th International Symposium on Eastern Mediterranean Geology, Thessaloniki, Greece, 14–20 April 2004; Chatzipetros, A., Pavlides, S., Eds.; School of Geology, Aristotelian University of Thessaloniki: Thessaloniki, Greece, 2004; pp. 1449–1452. [Google Scholar]
- Papavassilliou, K.; Voudouris, P.; Kanellopoulos, C.; Glasby, G.; Alfieris, D.; Mitsis, I. New geochemical and mineralogical constraints on the genesis of the Vani hydrothermal manganese deposit at NW Milos island, Greece: Comparison with the Aspro Gialoudi deposit and implications for the formation of the Milos manganese mineralization. Ore Geol. 2017, 80, 594–611. [Google Scholar] [CrossRef]
- Chi Fru, E.; Ivarsson, M.; Kilias, S.P.; Frings, S.; Hemmingsson, C.; Broman, C.; Bengtson, S.; Chatzitheodoridis, C. Biogenicity of an early Quaternary iron formation, Milos Island, Greece. Geobiology 2015, 13, 225–244. [Google Scholar] [CrossRef]
- Bonfante-Fasolo, P.; Faccio, A.; Perotto, S.; Schubert, A. Correlation between chitin distribution and cell wall morphology in the mycorrhizal fungus Glomus versiforme. Mycol. Res. 1990, 94, 157–165. [Google Scholar] [CrossRef]
- Coleman, M.; Moore, M. Direct reduction of sulphates to sulphur dioxide for isotopic analysis. Anal. Chem. 1978, 50, 1594–1595. [Google Scholar] [CrossRef]
- Hall, A.J.; Boyce, A.J.; Fallick, A.E.; Hamilton, P.J. Isotopic evidence of the depositional environment of Late Proterozoic stratiform barite mineralization, Aberfeldy, Scotland. Chem. Geol. 1991, 87, 99–114. [Google Scholar]
- Whitehouse, M.J. Multiple sulfur isotope analysis by SIMS: Evaluation of reference sulfides for Δ33S with observations and a case study on determination of Δ36S. Geostand. Geoanal. Res. 2013, 37. [Google Scholar] [CrossRef]
- Heinonen, A.; Andersen, T.; Rämö, O.T.; Whitehouse, M. The source of Proterozoic anorthosite and rapakivi granite magmatism: Evidence from combined in situ Hf–O isotopes of zircon in the Ahvenisto complex, southeastern Finland. J. Geol. Soc. 2015, 172, 103–112. [Google Scholar] [CrossRef]
- Stern, R.A.; (University of Alberta, Edmonton, Canada). Personal communication, 2019.
- Ding, T.; Valkires, S.; Kipphardt, H.; De Bievre, P.; Taylor, P.D.P.; Gonfiantini, R.; Krouse, R. Calibrated sulfur isotope abundance ratios of three IAEA sulfur isotope reference materials and V-CDT with a reassessment of the atomic weight of sulfur. Geochim. Cosmochim. Acta 2001, 65, 2433–2437. [Google Scholar] [CrossRef]
- Coplen, T.B. Reporting of stable hydrogen, carbon, and oxygen isotopic abundances. Pure Appl. Chem. 1994, 66, 273–276. [Google Scholar] [CrossRef]
- Ivarsson, M.; Kilias, S.P.; Broman, C.; Naden, J.; Detsi, K. Fossilized microorganisms preserved as fluid inclusions in epithermal veins, Vani Mn-Ba deposit, Milos Island, Greece. In Proceedings of the XIX CBGA Congress, Thessaloniki, Greece, 23–26 September 2010; Volume 100, pp. 297–307. [Google Scholar]
- Bodnar, R.J. Revised equation and table for determining the freezing point depression of H2O-NaCl solutions. Geochim. Cosmochim. Acta 1993, 57, 683–684. [Google Scholar] [CrossRef]
- Orange, D.; Knittle, E.; Farber, D.; Williams, Q. Raman spectroscopy of crude oils and hydrocarbon fluid inclusions: A feasibility study. In Mineral Spectroscopy: A tribute to Roger G. Burns; Dyar, M.D., McCammon, C., Schaefer, M.J.W., Eds.; The Geochemical Society, Special Publications: Washington DC, USA, 1996; Volume 5, pp. 65–81. [Google Scholar]
- Rahl, J.M.; Anderson, K.M.; Brandon, M.T.; Fassoulas, C. Raman spectroscopic carbonaceous material thermometry of low-grade metamorphic rocks: Calibration and application to tectonic exhumation in Crete, Greece. Earth Planet. Sci. Lett. 2005, 240, 339–354. [Google Scholar] [CrossRef]
- Lahfid, A.; Beyssac, O.; Deville, E.; Negro, F.; Chopin, C.; Goffé, B. Evolution of the Raman spectrum of carbonaceous material in low-grade metasediments of the Glarus Alps (Switzerland). Terra Nova 2010, 22, 354–360. [Google Scholar] [CrossRef]
- Julien, C.; Massot, M.; Baddour-Hadjean, R.; Franger, S.; Bach, S.; Pereira-Ramos, J.P. Raman spectra of birnessite manganese dioxides. Sol. Stn. Ion. 2003, 159, 345–356. [Google Scholar] [CrossRef]
- Frezzotti, M.L.; Tecce, F.; Casagli, A. Raman spectroscopy for fluid inclusion analysis. J. Geochem. Expl. 2012, 112, 1–20. [Google Scholar] [CrossRef]
- Downs, R.T. The RRUFF Project: An integrated study of the chemistry, crystallography, Raman and infrared spectroscopy of minerals. In Proceedings of the Program and Abstracts of the 19th General Meeting of the International Mineralogical Association, Kobe, Japan, 23–28 July 2006; pp. 3–13. [Google Scholar]
- Lehmann, M.F.; Bernasconi, S.M.; Barbieri, A.; McKenzie, J.A. Preservation of organic matter and alteration of its carbon and nitrogen isotope composition during simulated and in situ early sedimentary diagenesis. Geochim. Cosmochem. Acta 2002, 66, 3572–3584. [Google Scholar] [CrossRef]
- Paytan, A.; Martinez-Ruiz, F.; Eagle, M.; Ivy, A.; Wankel, S.D. Using sulfur isotopes to elucidate the origin of barite associated with high organic matter accumulation events in marine sediments. In Sulfur Biogeochemistry—Past and Present; Amend, J.P., Edwards, K.J., Lyons, T.W., Eds.; Geological Society of America Special Paper: Boulder, CO, USA, 2004; Volume 379, pp. 151–160. [Google Scholar]
- Paytan, A.; Kastner, M.; Campbell, D.; Thiemens, M.H. Sulfur isotopic composition of Cenozoic seawater sulfate. Science 1998, 282, 1459–1462. [Google Scholar] [CrossRef] [PubMed]
- Turchyn, A.V.; Schrag, D.P. Oxygen isotope constraints on the sulphur cycle over the past 10 million years. Science 2004, 303, 2004–2007. [Google Scholar] [CrossRef] [PubMed]
- Turchyn, A.V.; Schrag, D.P. Cenozoic evolution of the sulphur cycle: Insight from oxygen isotopes in marine sulfate. Earth Planet. Sci. Lett. 2006, 241, 763–779. [Google Scholar] [CrossRef]
- Aharon, P.; Fu, B. Microbial sulfate reduction rates and sulphur and oxygen isotope fractionations at oil and gas seeps in deepwater Gulf of Mexico. Geochim. Cosmochim Acta 2000, 64, 233–246. [Google Scholar] [CrossRef]
- Kilias, S.; Naden, J.; Cheliotis, I.; Shepherd, T.J.; Constandinidous, H.; Crossing, J.; Simos, J. Epithermal gold mineralization in the active Aegean Volcanic Arc: The Profitis Ilias deposit, Milos Island, Greece. Miner. Depos. 2001, 36, 32–44. [Google Scholar] [CrossRef]
- Bakker, R.J. Package FLUIDS 1. Computer programs for analysis of fluid inclusion data and for modelling bulk fluid properties. Chem. Geol. 2003, 194, 3–23. [Google Scholar] [CrossRef]
- Papanikolaou, D.; Lekkas, E.; Syskakis, D. Tectonic analysis of the geothermal field of Milos Island. Bull. Geol. Soc. Greece 1990, 24, 27–46. [Google Scholar]
- Peter, J.M.; Peltonen, P.; Scott, S.D.; Simoneit, B.R.T.; Kawka, O.E. 14C ages of hydrothermal petroleum and carbonate in Guaymas Basin, Gulf of California: Implications for oil generation, expulsion, and migration. Geology 1991, 19, 253–256. [Google Scholar] [CrossRef]
- Simoneit, B.R. Hydrothermal petroleum: Genesis, migration, and deposition in Guaymas Basin, Gulf of California. Can. J. Earth Sci. 1985, 22, 1919–1929. [Google Scholar] [CrossRef]
- Simoneit, B.R.T.; Leif, R.N.; Sturz, A.A.; Sturdivant, A.E.; Gieskes, J.M. Geochemistry of shallow sediments in Guaymas Basin, Gulf of California: Hydrothermal gas and oil migration and effects of mineralogy. Org. Geochem. 1992, 18, 765–784. [Google Scholar] [CrossRef]
- Simoneit, B.R. Hydrothermal Petroleum. In Hydrocarbons, Oils and Lipids: Diversity, Origin, Chemistry and Fate; Springer: Berlin, Germany, 2018; pp. 1–35. [Google Scholar]
- Yamanaka, T.; Ishibashi, J.; Hashimoto, J. Organic geochemistry of hydrothermal petroleum generated in the submarine Wakamiko caldera, southern Kyushu, Japan. Org. Geochem. 2000, 31, 1117–1132. [Google Scholar] [CrossRef]
- Teske, A.; De Beer, D.; McKay, L.J.; Tivey, M.K.; Biddle, J.F.; Hoer, D.; Lloyd, K.G.; Lever, M.A.; Røy, H.; Albert, D.B.; et al. The Guaymas Basin hiking guide to hydrothermal mounds, chimneys, and microbial mats: Complex seafloor expressions of subsurface hydrothermal circulation. Front. Microbial. 2016, 7, 75. [Google Scholar] [CrossRef]
- Daskalopoulou, K.; Gagliano, A.L.; Calabrese, S.; Longo, M.; Hantzis, K.; Kyriakopoulos, K.; D’Alessandro, W. Gas geochemistry and CO2 output estimation at the island of Milos, Greece. J. Volcanol. Geotherm. Res. 2018, 365, 13–22. [Google Scholar] [CrossRef]
- Feng, D.; Roberts, H.H. Geochemical characteristics of the barite deposits at cold seeps from the northern Gulf of Mexico continental slope. Earth Planet. Sci. Lett. 2011, 309, 89–99. [Google Scholar] [CrossRef]
- Aquilina, L.; Pauwels, H.; Genter, A.; Fouillac, C. Water-rock interaction processes in the Triassic sandstone and the granitic basement of the Rhine Graben: Geochemical investigation of a geothermal reservoir. Geochim. Cosmochim. Acta 1997, 61, 4281–4295. [Google Scholar] [CrossRef]
- Richards, J.P. A shake-up in the porphyry world? Econ. Geol. 2018, 118, 1225–1233. [Google Scholar] [CrossRef]
- Bodnar, R.J. Introduction to aqueous-electrolyte fluid inclusions. In Fluid Inclusions: Analysis and Interpretation; Samson, I., Anderson, A., Marshall, D., Eds.; Mineralogical Association of Canada: Quebec City, QC, Cananda, 2003; Volume 32, pp. 81–99. [Google Scholar]
- Ehrlich, H.L. Geomicrobiology; Marcel Dekker: New York, NY, USA, 2002. [Google Scholar]
- Puente-Sáncheza, F.; Arce-Rodríguez, A.; Oggerind, M.; García-Villadangosa, M.; Moreno-Paza, M.; Blancoa, Y.; Rodríguez, N.; Birde, L.; Lincolne, S.A.; Tornos, F.; et al. Viable cyanobacteria in the deep continental subsurface. Proc. Natl. Acad. Sci. USA 2018, 115, 10702–10707. [Google Scholar] [CrossRef]
- Salman, V.; Bailey, J.V.; Teske, A. Phylogenetic and morphologic complexity of giant sulphur bacteria. Antonie van Leeuwenhoek 2013, 104, 169–186. [Google Scholar] [CrossRef]
- Schulz, H.N.; Jørgensen, B.B. Big Bacteria. Annu. Rev. Microbiol. 2001, 55, 105–137. [Google Scholar] [CrossRef]
- Cunningham, J.A.; Thomas, C.-W.; Bengtson, S.; Marone, F.; Stampanoni, M.; Turner, F.R.; Bailey, J.V.; Raff, R.A.; Raff, E.C.; Donoghue, P.C.J. Experimental taphonomy of giant Sulphur bacteria: Implications for the interpretation of the embryo-like Ediacaran Doushantuo fossils. Proc. R. Soc. B. 2012, 279, 1857–1864. [Google Scholar] [CrossRef]
- Carrera, M.; Zandomeni, R.O.; Fitzgibbon, J.; Sagripanti, J.-L. Difference between the spore sizes of Bacillus anthracis and other Bacillus species. J. Appl. Microbiol. 2007, 102, 303–312. [Google Scholar] [CrossRef]
- Perretto, R.; Bettini, V.; Favaron, F.; Alghisi, P.; Bonfante, P. Polygalacturonase activity and location in arbuscular mycorrhizal roots of Allium porrum L. Mycorrhiza 1995, 5, 157–163. [Google Scholar] [CrossRef]
- Fife, D.J.; Bruhn, D.F.; Miller, K.S.; Stoner, D.L. Evaluation of a fluorescent lectin-based staining technique for some acidophilic mining bacteria. Appl. Environ. Microbiol. 2000, 66, 2208–2210. [Google Scholar] [CrossRef]
- Montgomery, M.T.; Welschmeyer, N.A.; Kirchman, D.L. A simple assay for chitin: Application to sediment trap samples from the subarctic Pacific. Mar. Ecol. Prog. Ser. 1990, 64, 301–308. [Google Scholar] [CrossRef]
- Rawat, R.; Rawat, S. Colorless sulfur oxidizing bacteria from diverse habitats. Adv. Appl. Sci. Res. 2015, 6, 230–235. [Google Scholar]
- Wohl, D.L.; McArthur, J.V. Aquatic Actinomycete-fungal interactions and their effects on organic matter decomposition: A microcosm study. Microbial. Ecol. 2001, 42, 446–457. [Google Scholar] [CrossRef]
- Orsi, W.D.; Edgcomb, V.D.; Chrsitman, G.D.; Biddle, J.F. Gene expression in the deep biosphere. Nature 2013, 499, 205–208. [Google Scholar] [CrossRef]
- Orsi, W.D.; Biddle, J.F.; Edgcomb, V.D. Deep sequencing of subseafloor eukaryotic rRNA reveals active fungi across marine subsurface provinces. PLoS ONE 2013, 8, 1–10. [Google Scholar] [CrossRef]
- Maheshwani, R.; Bharadwaj, G.; Bhat, M.K. Thermophilic fungi: Their physiology and enzymes. Microbiol. Mol. Rev. 2000, 64, 461–488. [Google Scholar] [CrossRef]
- Ivarsson, M.; Broman, C.; Lindblom, S.; Holm, N.G. Fluid inclusions as a tool to constrain the preservation conditions of sub-seafloor cryptoendoliths. Planet. Space Sci. 2009, 57, 477–490. [Google Scholar] [CrossRef]
- Tebo, B.M.; Johnson, H.A.; McCarthy, J.K.; Templeton, A.S. Geomicrobiology of manganese (II) oxidation. Trends Microbiol. 2005, 13, 421–428. [Google Scholar] [CrossRef]
- Ehrlich, H.L.; Newman, D.K. Geomicrobiology of manganese. In Geomicrobiology; Ehrlich, H.L., Newman, D.K., Eds.; CRC Press: Boca Raton, FL, USA, 2009; pp. 347–420. [Google Scholar]
- Hansel, C.M.; Zeiner, C.A.; Santelli, C.M.; Webb, S.M. Mn (II) oxidation by anascomycete fungus is linked to superoxide production during asexual reproduction. Proc. Natl. Acad. Sci. USA 2012, 109, 12621–12625. [Google Scholar] [CrossRef]
- Francis, C.A.; Tebo, B.M. Enzymatic manganese (II) oxidation by metabolically dormant spores of diverse Bacillus species. Appl. Environ. Microbiol. 2002, 68, 874–880. [Google Scholar] [CrossRef]
- Santelli, C.M.; Pfister, D.H.; Lazarus, D.; Sun, L.; Burgos, W.D.; Hansel, C.M. Promotion of Mn (II) oxidation and remediation of coal mine drainage in passive treatment systems by diverse fungal and bacterial communities. Appl. Environ. Microbiol. 2010, 76, 4871–4875. [Google Scholar] [CrossRef]
- Hansel, C.M.; Francis, C.A. Coupled photochemical and enzymatic Mn (II) oxidation pathways of a planktonic Roseobacter-like bacterium. Appl. Environ. Microbiol. 2006, 72, 3543–3549. [Google Scholar] [CrossRef]
- Dubessy, J.; Pagel, M.; Beny, J.-M.; Christensen, H.; Hickel, B.; Kosztolanyi, C.; Poty, B. Radiolysis evidence by H2-O2 and H2-bearing fluid inclusions in three uranium deposits. Geochim. Cosmochim. Acta 1988, 52, 1155–1167. [Google Scholar] [CrossRef]
- Li, J.; Chou, I.-M. Hydrogen in silicate melt inclusions in quartz from granite detected with Raman spectroscopy. J. Ram. Spectrophotometer 2015, 46, 983–986. [Google Scholar] [CrossRef]
- Haruta, M.; Yamada, N.; Kobayashi, T.; Iijima, S. Gold catalysts prepared by coprecipitation for low-temperature oxidation of hydrogen and carbon monoxide. J. Catal. 1989, 115, 301–309. [Google Scholar] [CrossRef]
- Torres Sanchez, R.M.; Ueda, A.; Tanaka, K.; Haruta, M. Selective oxidation of CO in hydrogen over gold supported on manganese oxides. J. Catal. 1997, 168, 125–127. [Google Scholar] [CrossRef]
- Botz, R.; Stüben, D.; Winckler, G.; Bayer, R.; Schmitt, M.; Faber, E. Hydrothermal gases offshore Milos Island, Greece. Chem. Geol. 1996, 130, 161–173. [Google Scholar] [CrossRef]
- Daskalopoulou, K.; Calabrese, S.; Grassa, F.; Kyriakopoulos, K.; Parello, F.; Tassi, F.; D’Alessandro, W. Origin of methane and light hydrocarbons in natural fluid emissions: A key study from Greece. Chem. Geol. 2018, 479, 286–301. [Google Scholar] [CrossRef]
- Sherwood Lollar, B.; Lacrampe-Couloume, G.; Slater, G.F.; Ward, J.; Moser, D.P.; Gihring, T.M.; Lin, L.-H.; Onstott, T.C. Unravelling abiogenic and biogenic sources of methane in the Earth’s deep subsurface. Chem. Geol. 2006, 226, 328–339. [Google Scholar] [CrossRef]
- Nandi, R.; Sengupta, S. Microbial production of hydrogen: An overview. Crit. Rev. Microbiol. 1998, 24, 61–84. [Google Scholar] [CrossRef]
- Gray, C.T.; Gest, H. Biological formation of molecular hydrogen. Science 1965, 148, 186–192. [Google Scholar] [CrossRef]
- Horner, D.S.; Foster, P.G.; Embley, T.M. Iron hydrogenases and the evolution of anaerobic eukaryotes. Mol. Biol. Evol. 2000, 17, 1695–1709. [Google Scholar] [CrossRef]
- Thorsen, M.S. Abundance and biomass of the gut-living microorganisms (bacteria, protozoa and fungi) in the irregular sea urchin Echinocardium cordatum (Spatangoida: Echinodermata). Mar. Ecol. 1999, 133, 353–360. [Google Scholar] [CrossRef]
- Mackie, R.I.; Rycyk, M.; Ruemmler, R.L.; Aminov, R.I.; Wikelski, M. Biochemical and microbiological evidence for fermentative digestion in free-living land iguanas (Conolophus pallidus) and marine ignuanas (Amblyrhynchus cristatus) on the Galapagos archipelago. Phys. Biochem. Zool. 2004, 77, 127–138. [Google Scholar] [CrossRef]
- Lockhart, R.J.; Van Dyke, M.I.; Beadle, I.R.; Humphryes, P.; McCarthy, A.J. Molecular biological detection of anaerobic gut fungi (Neocallimastigales) from landfill sites. Appl. Environ. Microbiol. 2006, 72, 5659–5661. [Google Scholar] [CrossRef]
- Wurzbacher, C.; Warthmann, N.; Bourne, E.; Attermeyer, K.; Allgaier, M.; Powell, J.R.; Detering, H.; Mbedi, S.; Grossart, H.-P.; Monaghan, M.T. High habitat-specificity in fungal communities in oligo-mesotrophic, temperate Lake Stechlin (North-East Germany). MycoKeys 2016, 16, 17–44. [Google Scholar] [CrossRef]
- Mohamed, D.J.; Martiny, J.B. Patterns of fungal diversity and composition along a salinity gradient. ISME J. 2011, 5, 379–388. [Google Scholar] [CrossRef]
- Picard, K.T. Coastal marine habitats harbour novel early-diverging fungal diversity. Fungal Ecol. 2017, 25, 1–13. [Google Scholar] [CrossRef]
- Müller, M.; Mentel, M.; van Hellemod, J.J.; Henze, K.; Woehle, C.; Goild, S.B.; Yu, R.-Y.; van der Giezen, M.; Tielens, A.G.M.; Martin, W.F. Biocemistry and evolution of anaerobic energy metabolism in eukaryotes. Microbiol. Mol. Biol. Rev. 2012, 76, 444–495. [Google Scholar] [CrossRef]
- Hjorth, K.; Goldberg, A.V.; Tsaousis, A.D.; Hirt, R.P.; Embley, T.M. Diversity and reductive evolution of mitochondria among microbial eukaryotes. Phil. Trans. R. Soc. B Biol. Sci. 2010, 365, 713–727. [Google Scholar] [CrossRef]
- Embley, T.M.; O’Neill, A.H.; Thomsson, E.; Pedersen, K. Characterisation of yeasts isolated from deep igneous rock aquifers of the Fennoscandian shield. Microb. Ecol. 2003, 46, 416–428. [Google Scholar]
- Baranenko, V.I.; Kirov, V.S. Solubility of hydrogen in water in a broad temperature and pressure range. Sov. At. Energy 1989, 66, 30–34. [Google Scholar] [CrossRef]



| Sample | Δ13C TOT vs PDB (‰) | %C |
|---|---|---|
| VA0518 | −26.69 | 0.13 |
| VA0520 | −30.24 | 0.52 |
| VA0522 | −27.79 | 0.25 |
| MI0344 | −28.25 | 1.05 |
| Sample | δ18OV-SMOW (‰) | δ34SCDT (‰) |
|---|---|---|
| MI-45B | 11.2 | 23.2 |
| MI-04-29 | 10.7 | 22.7 |
| VA-19 | 10.5 | 23.5 |
| VA-21 | 14.3 | 22.2 |
| VA-01 | 14.7 | 21.9 |
| SMO-bar | 9.7 | 21.4 |
| MI-52 | 10.5 | 24.3 |
| VA-26 | 12.3 | 21.3 |
| VA-05-02 | 11.4 | 22.6 |
| VA-18 | 10.4 | 22.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivarsson, M.; Kilias, S.P.; Broman, C.; Neubeck, A.; Drake, H.; Chi Fru, E.; Bengtson, S.; Naden, J.; Detsi, K.; Whitehouse, M.J. Exceptional Preservation of Fungi as H2-Bearing Fluid Inclusions in an Early Quaternary Paleo-Hydrothermal System at Cape Vani, Milos, Greece. Minerals 2019, 9, 749. https://doi.org/10.3390/min9120749
Ivarsson M, Kilias SP, Broman C, Neubeck A, Drake H, Chi Fru E, Bengtson S, Naden J, Detsi K, Whitehouse MJ. Exceptional Preservation of Fungi as H2-Bearing Fluid Inclusions in an Early Quaternary Paleo-Hydrothermal System at Cape Vani, Milos, Greece. Minerals. 2019; 9(12):749. https://doi.org/10.3390/min9120749
Chicago/Turabian StyleIvarsson, Magnus, Stephanos P. Kilias, Curt Broman, Anna Neubeck, Henrik Drake, Ernest Chi Fru, Stefan Bengtson, Jonathan Naden, Kleopatra Detsi, and Martin J. Whitehouse. 2019. "Exceptional Preservation of Fungi as H2-Bearing Fluid Inclusions in an Early Quaternary Paleo-Hydrothermal System at Cape Vani, Milos, Greece" Minerals 9, no. 12: 749. https://doi.org/10.3390/min9120749
APA StyleIvarsson, M., Kilias, S. P., Broman, C., Neubeck, A., Drake, H., Chi Fru, E., Bengtson, S., Naden, J., Detsi, K., & Whitehouse, M. J. (2019). Exceptional Preservation of Fungi as H2-Bearing Fluid Inclusions in an Early Quaternary Paleo-Hydrothermal System at Cape Vani, Milos, Greece. Minerals, 9(12), 749. https://doi.org/10.3390/min9120749








