Cold-Water Corals in Gas Hydrate Drilling Cores from the South China Sea: Occurrences, Geochemical Characteristics and Their Relationship to Methane Seepages
Abstract
1. Introduction
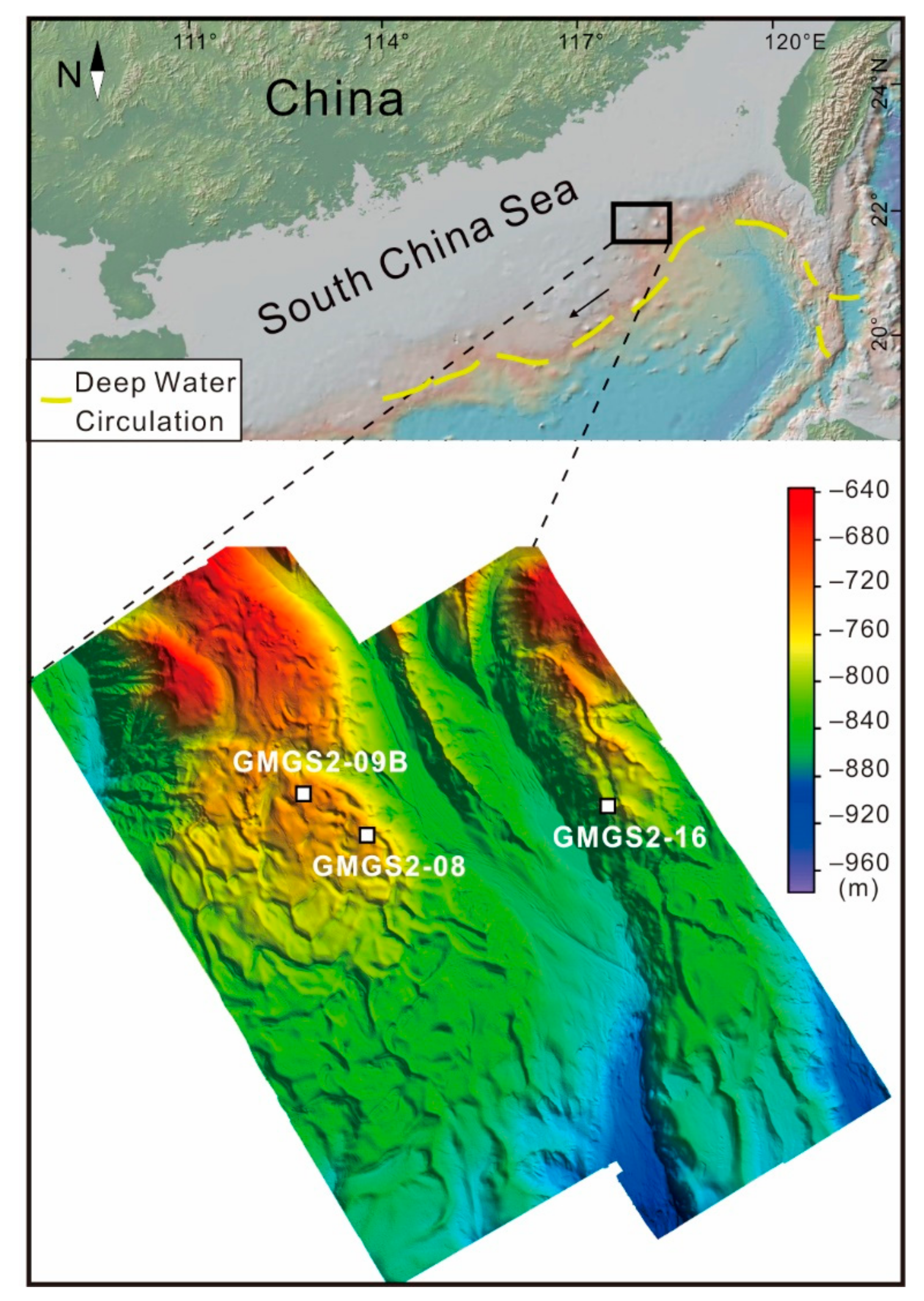
2. Geological Background
3. Samples and Methods
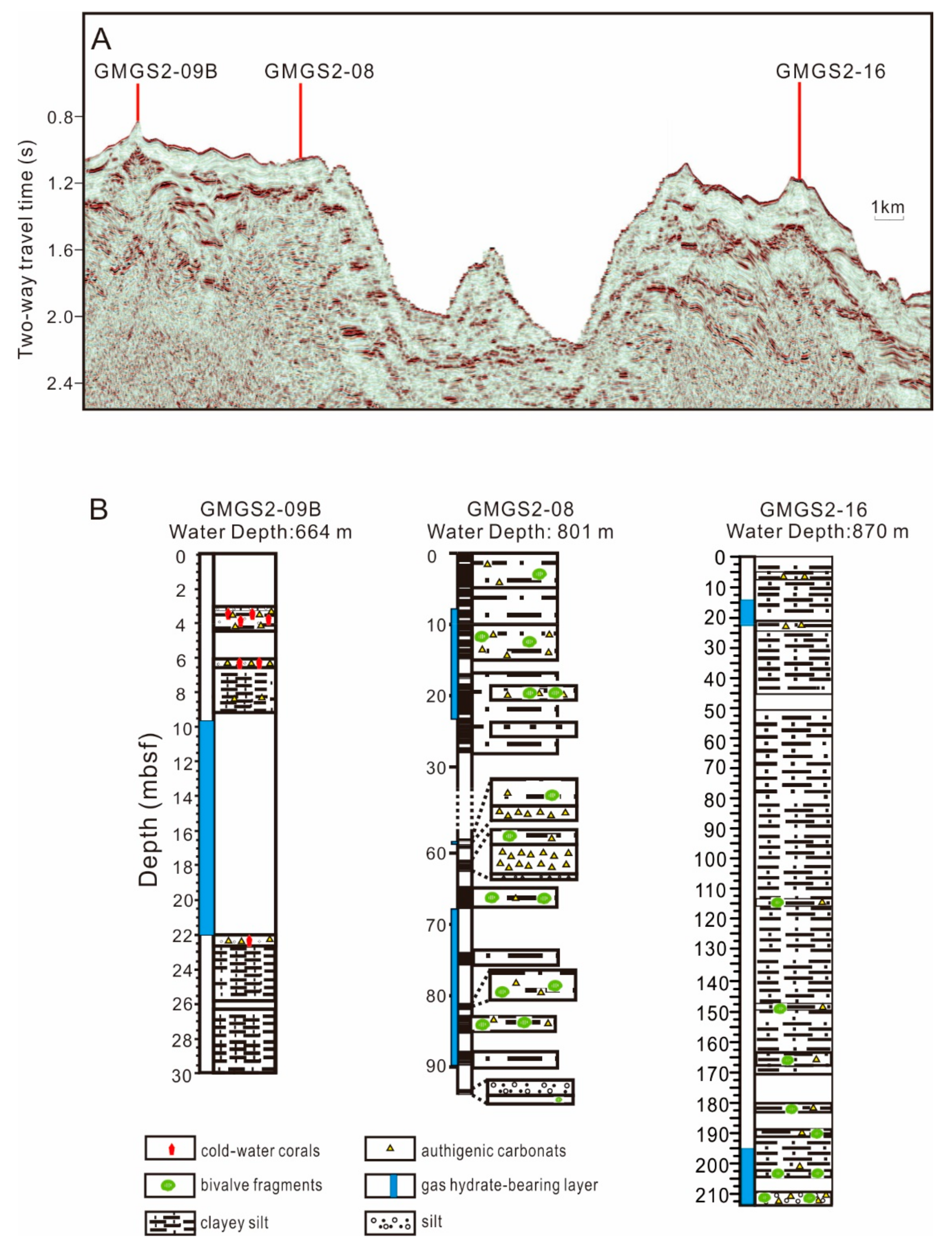
3.1. Samples
3.2. Analytical Methods
4. Results
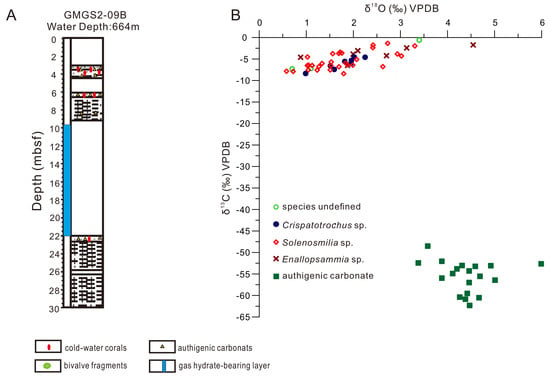
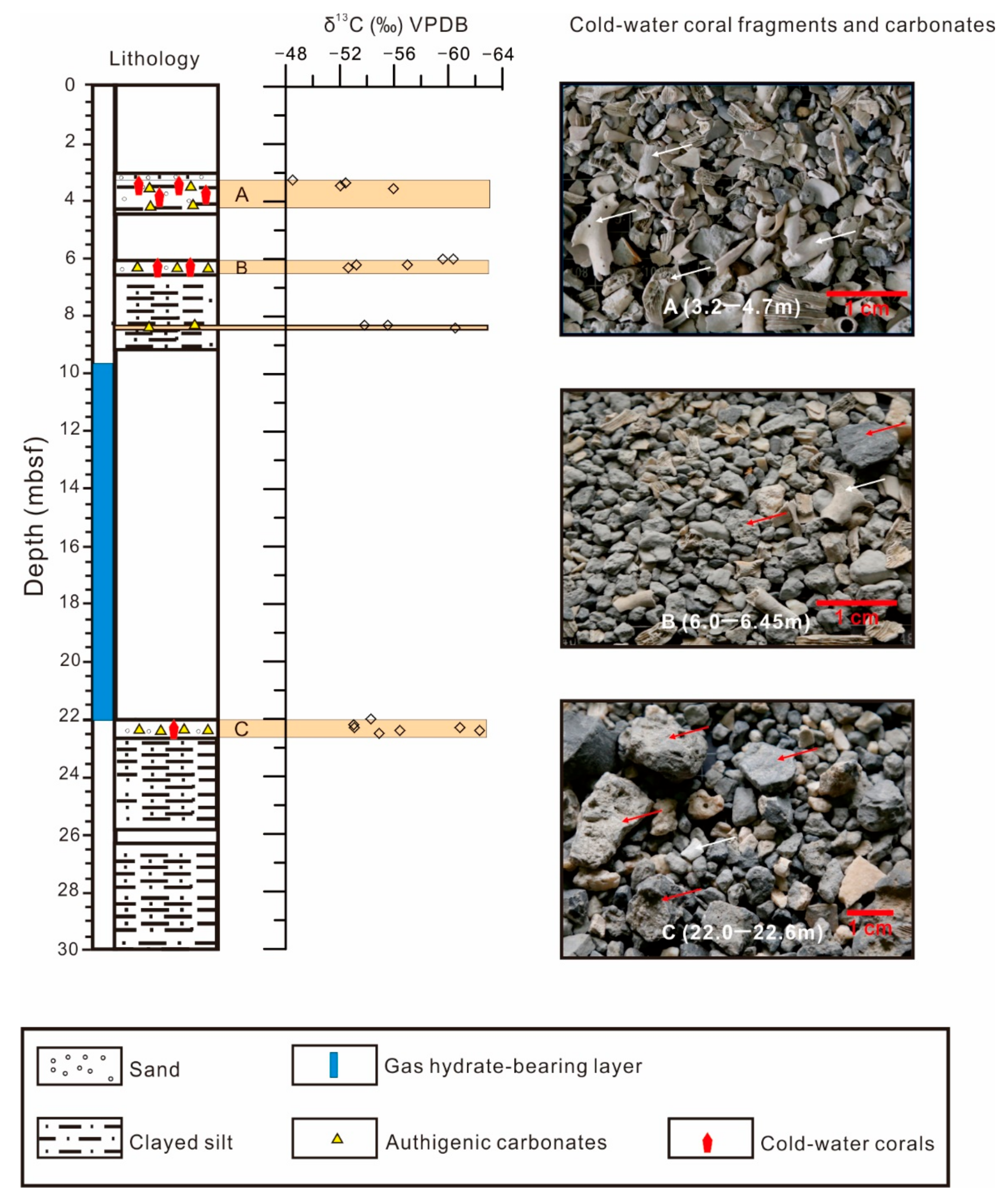
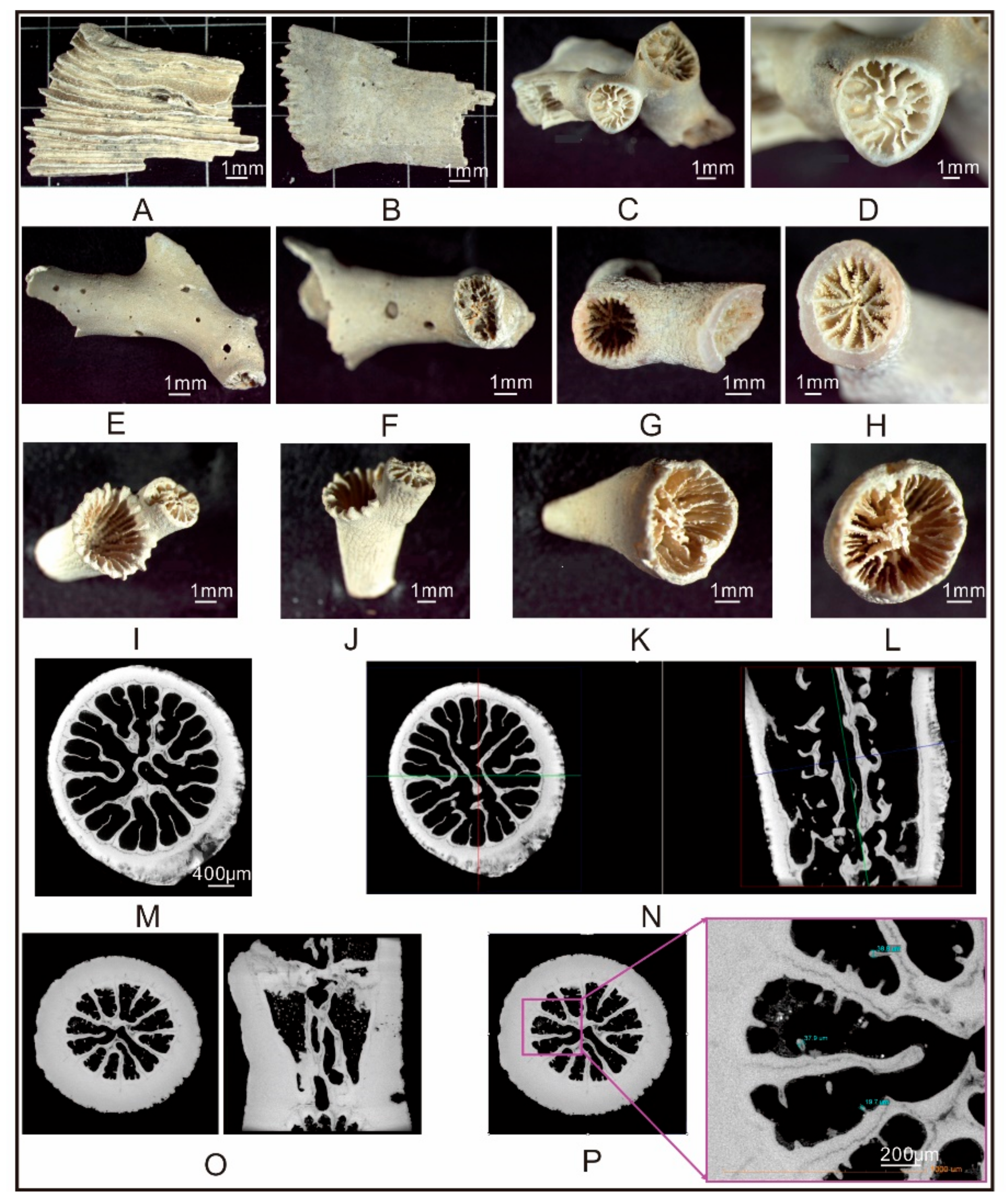
4.1. Distribution and Species of CWCs
4.2. CWC Mineralogy
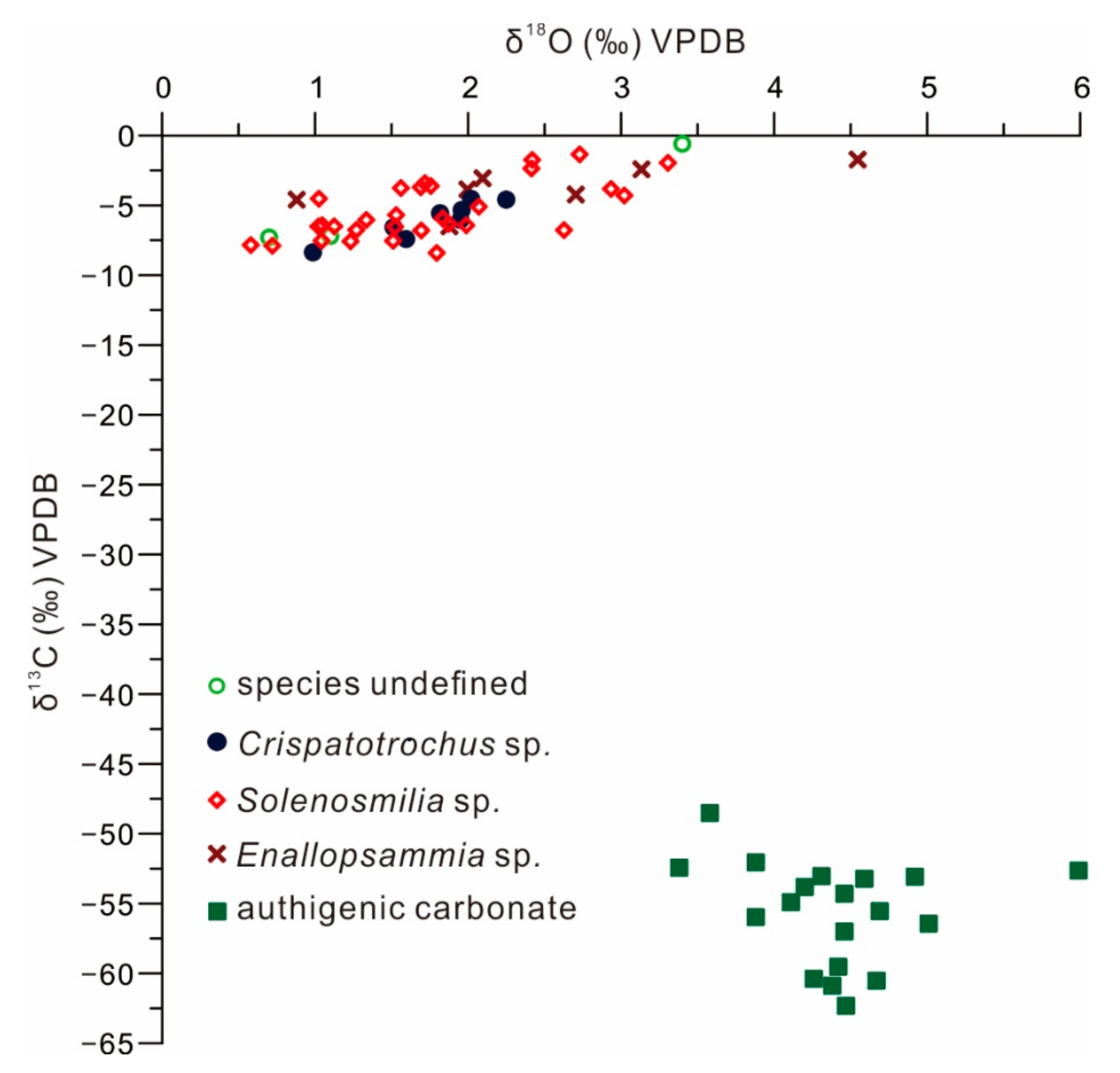
4.3. Carbon and Oxygen Isotopes of CWCs and Carbonates
4.4. CWC Element Contents
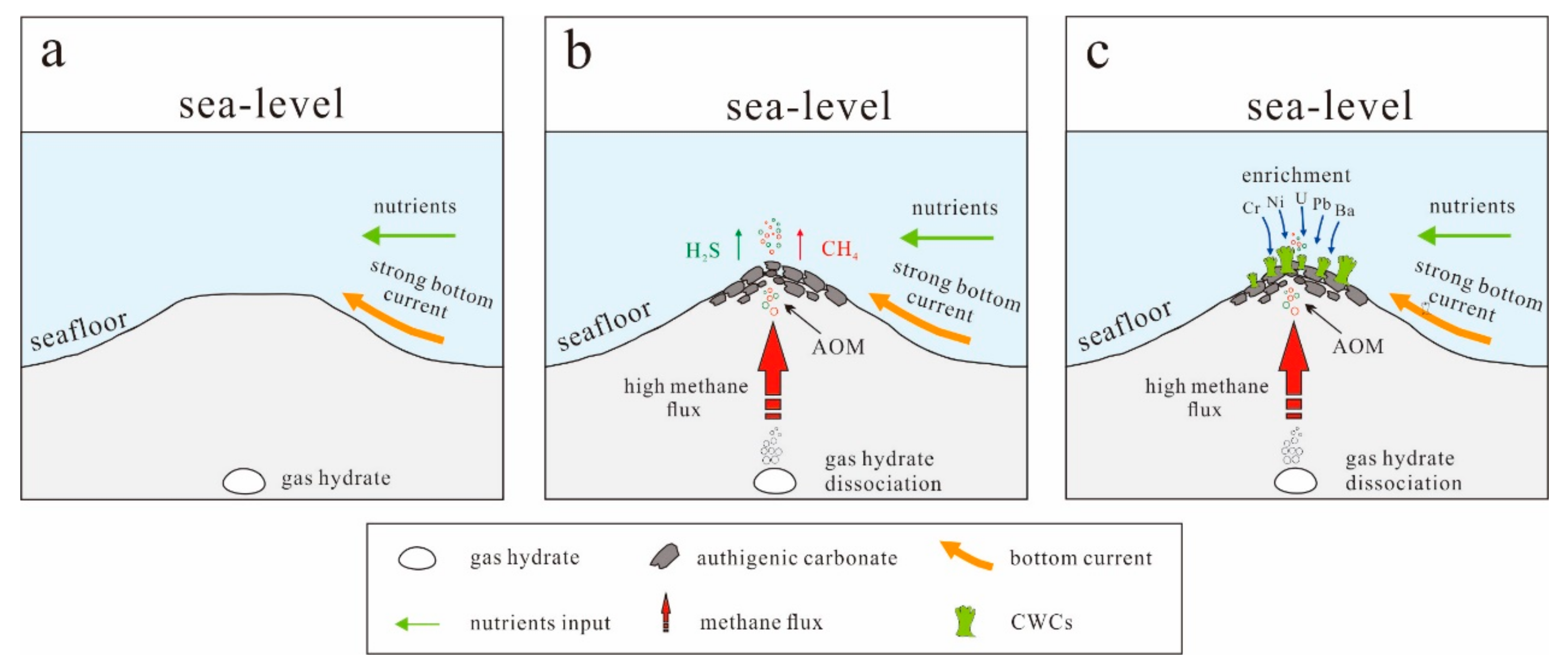
5. Discussion
5.1. CWCs Distribution and Factors Affect Their Growth
5.2. Bottom-Water Chemistry and Fluid Seepage Archived in CWCs
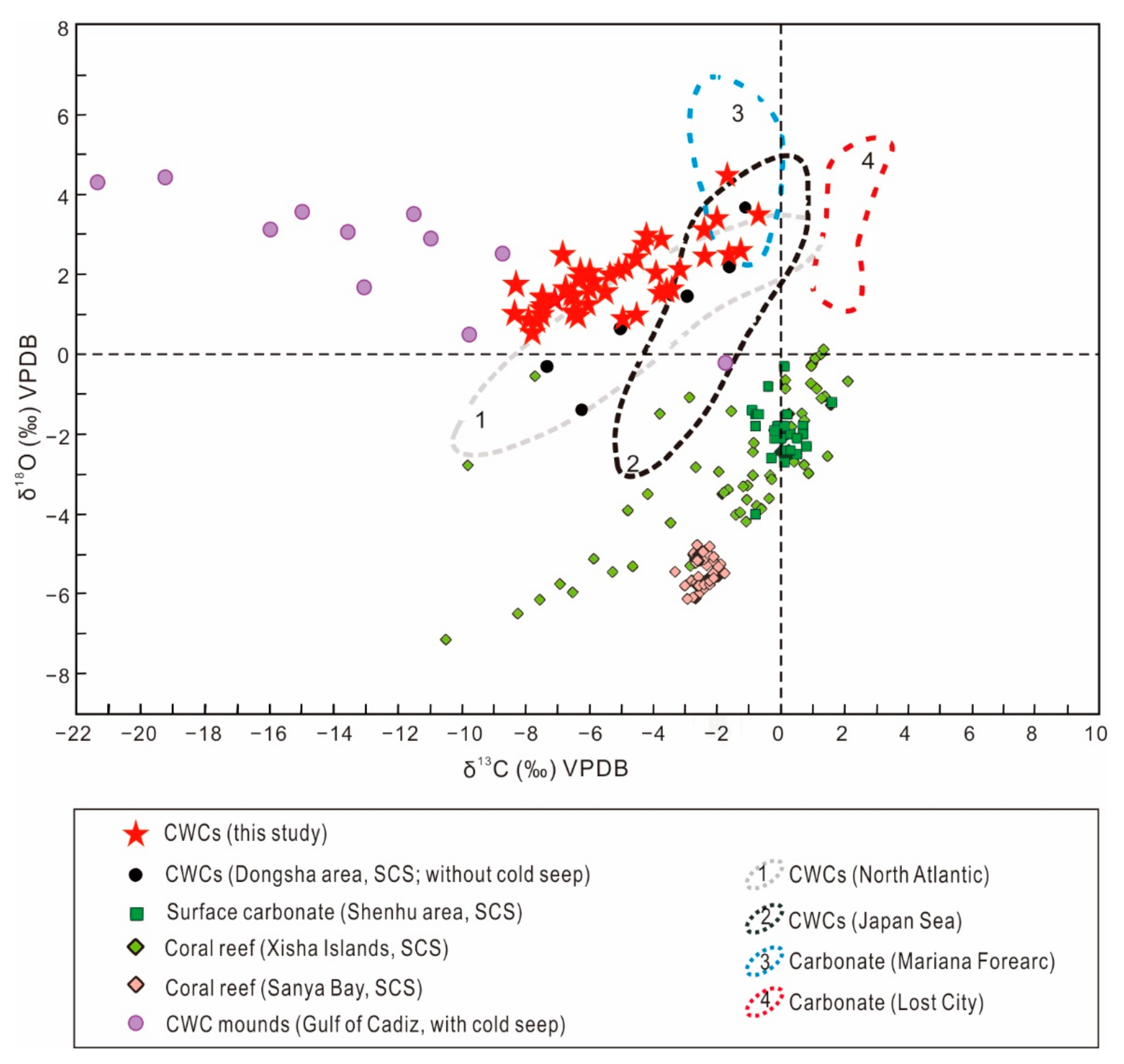
5.2.1. Comparison of C and O Isotope Signatures
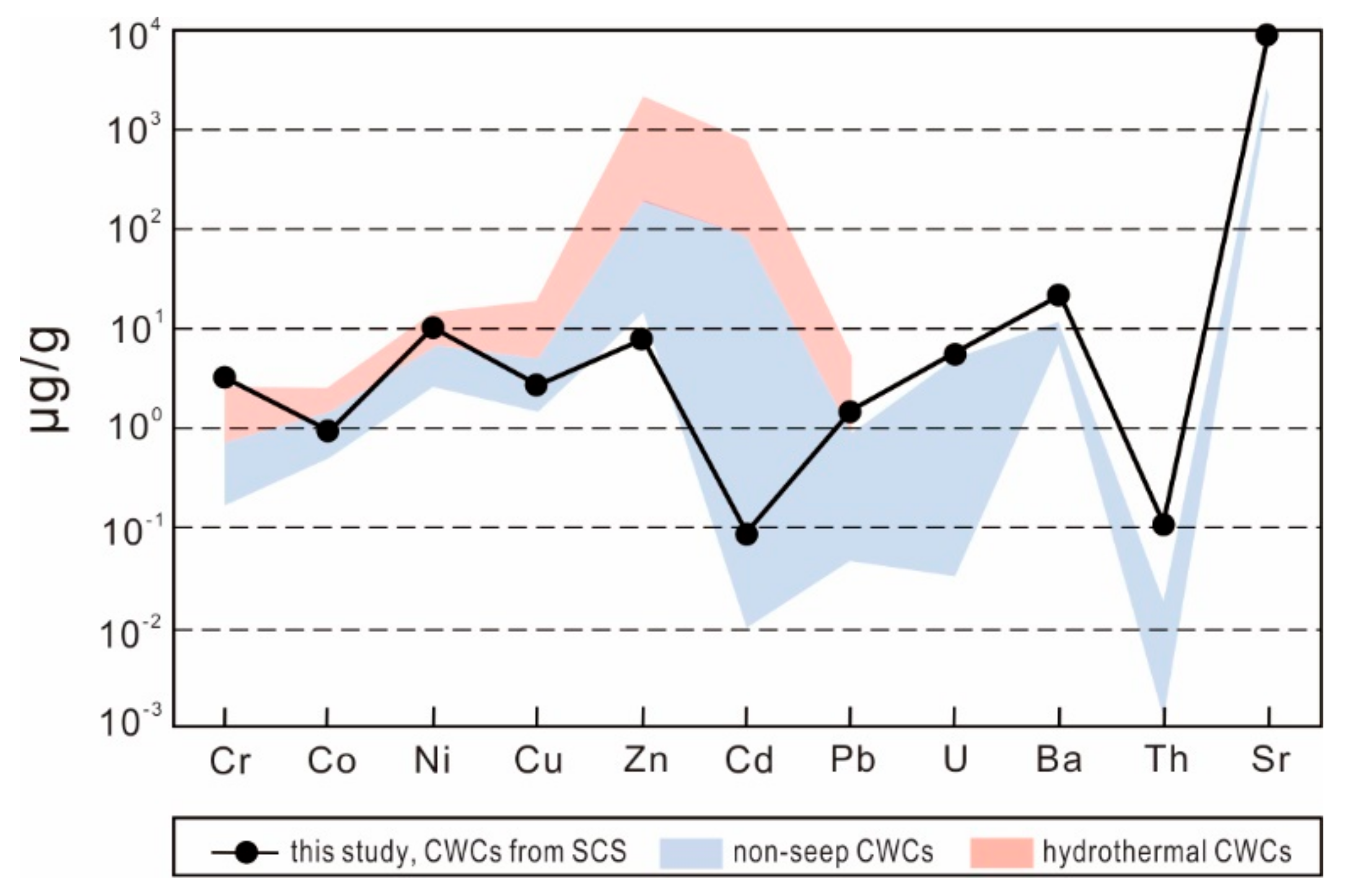
5.2.2. CWC Trace-Element Contents
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Depth (cmbsf) | Sample | Ara | Cal |
|---|---|---|---|
| 335–345 | Cri. | 99 | 1 |
| 345–355 | Cri. | 97 | 3 |
| 345–355 | Sol. | 99 | 1 |
| 345–355 | Sol. | 95 | 5 |
| 345–355 | Sol. | 98 | 2 |
| 355–365 | Sol. | 99 | 1 |
| 355–365 | Ena. | 97 | 3 |
| 630–645 | Ena. | 99 | 1 |
| Core Number | Depth (cmbsf) | Sample Type | δ13C (‰) | δ18O (‰) |
|---|---|---|---|---|
| 09B-2M-1a | 325–335 | Grey nodular carbonate | −48.5 | 3.6 |
| 09B-2M-1a | 335–345 | Grey nodular carbonate | −52.4 | 3.4 |
| 09B-2M-1a | 345–355 | Grey nodular carbonate | −52.0 | 3.9 |
| 09B-2M-1a | 355–365 | Grey nodular carbonate | −56.0 | 3.9 |
| 09B-2M-1a | 600–615 | Grey nodular carbonate | −59.5 | 4.4 |
| 09B-2M-1a | 620–630 | Grey nodular carbonate | −57.0 | 4.5 |
| 09B-2M-1a | 620–630 | Opalescent nodular carbonate | −53.2 | 4.6 |
| 09B-2M-1a | 630–645 | Grey nodular carbonate | −52.6 | 6.0 |
| 09B-2M-1a | 630–645 | Opalescent nodular carbonate | −60.4 | 4.3 |
| 09B-2M-1a | 830–840 | Opalescent nodular carbonate | −53.8 | 4.2 |
| 09B-2M-1a | 830–840 | Grey nodular carbonate | −55.5 | 4.7 |
| 09B-2M-1a | 840–845 | Grey nodular carbonate | −60.5 | 4.7 |
| 09B-5H-1 | 2200–2215 | Grey nodular carbonate | −54.3 | 4.5 |
| 09B-5H-1 | 2220–2230 | Grey nodular carbonate | −53.0 | 4.3 |
| 09B-5H-1 | 2230–2240 | Opalescent nodular carbonate | −60.9 | 4.4 |
| 09B-5H-1 | 2230–2240 | Grey nodular carbonate | −53.1 | 4.9 |
| 09B-5H-1 | 2240–2250 | Opalescent nodular carbonate | −62.3 | 4.5 |
| 09B-5H-1 | 2240–2250 | Grey nodular carbonate | −56.4 | 5.0 |
| 09B-5H-1 | 2250–2260 | Grey nodular carbonate | −54.9 | 4.1 |
| 09B-2M-1a | 325–335 | Crispatotrochus species | −5.6 | 1.8 |
| 09B-2M-1a | 325–335 | Crispatotrochus species | −7.4 | 1.6 |
| 09B-2M-1a | 325–335 | Crispatotrochus species | −5.3 | 2.0 |
| 09B-2M-1a | 335–345 | Crispatotrochus species | −6.6 | 1.5 |
| 09B-2M-1a | 335–345 | Crispatotrochus species | −8.4 | 1.0 |
| 09B-3M-1 | 630–645 | Crispatotrochus species | −4.6 | 2.2 |
| 09B-5H-1 | 2230–2240 | Crispatotrochus species | −6.0 | 2.0 |
| 09B-2M-1a | 345–355 | Crispatotrochus species | −4.5 | 2.0 |
| 09B-5H-1 | 2200–2215 | Enallopsammia species | −3.1 | 2.1 |
| 09B-5H-1 | 2200–2215 | Enallopsammia species | −6.5 | 1.9 |
| 09B-5H-1 | 2230–2240 | Enallopsammia species | −3.9 | 2.0 |
| 09B-5H-1 | 2220–2230 | Enallopsammia species | −4.2 | 2.7 |
| 09B-3M-1 | 630–645 | Enallopsammia species | −2.4 | 3.1 |
| 09B-3M-1 | 630–645 | Enallopsammia species | −1.7 | 4.5 |
| 09B-2M-1a | 2220–2230 | Enallopsammia species | −4.6 | 0.9 |
| 09B-2M-1a | 335–345 | Solenosmilia species | −1.3 | 2.7 |
| 09B-2M-1a | 335–345 | Solenosmilia species | −4.5 | 1.0 |
| 09B-2M-1a | 335–345 | Solenosmilia species | −6.5 | 1.1 |
| 09B-2M-1a | 335–345 | Solenosmilia species | −7.6 | 1.2 |
| 09B-2M-1a | 335–345 | Solenosmilia species | −7.5 | 1.5 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −5.9 | 1.8 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −6.0 | 1.3 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −6.8 | 2.6 |
| 09B-2M-1a | 335–345 | Solenosmilia species | −6.4 | 2.0 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −1.7 | 2.4 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −3.8 | 1.6 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −6.3 | 1.9 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −3.7 | 1.7 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −6.5 | 1.5 |
| 09B-2M-1a | 335–345 | Solenosmilia species | −5.1 | 2.1 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −6.5 | 1.0 |
| 09B-2M-1a | 345–355 | Solenosmilia species | −8.4 | 1.8 |
| 09B-2M-1a | 345–355 | Solenosmilia species | -−7.9 | 0.7 |
| 09B-2M-1a | 355–365 | Solenosmilia species | -−2.0 | 3.3 |
| 09B-2M-1a | 355–365 | Solenosmilia species | −7.8 | 0.6 |
| 09B-3M-1 | 600–615 | Solenosmilia species | −3.8 | 2.9 |
| 09B-3M-1 | 600–615 | Solenosmilia species | −3.4 | 1.7 |
| 09B-3M-1 | 630–645 | Solenosmilia species | −2.4 | 2.4 |
| 09B-5H-1 | 2200–2215 | Solenosmilia species | −3.6 | 1.8 |
| 09B-5H-1 | 2230–2240 | Solenosmilia species | −4.3 | 3.0 |
| 09B-5H-1 | 2230–2240 | Solenosmilia species | −6.4 | 1.0 |
| 09B-5H-1 | 2220–2230 | Solenosmilia species | −7.5 | 1.0 |
| 09B-5H-1 | 2220–2230 | Solenosmilia species | −6.8 | 1.7 |
| 09B-2M-2a | 345–355 | Solenosmilia species | −6.8 | 1.3 |
| 09B-2M-1a | 610–615 | Solenosmilia species | −5.7 | 1.5 |
| 09B-2M-1a | 335–345 | coral (species undefined) | −7.2 | 1.1 |
| 09B-2M-1a | 345–355 | coral (species undefined) | −7.3 | 0.7 |
| 09B-2M-1a | 620–630 | coral (species undefined) | −0.6 | 3.4 |
| Depth | (cmbsf) | 345–355 | 345–355 | 610–615 | 2220–2230 |
|---|---|---|---|---|---|
| Al | (%) | 0.021 | 0.028 | 0.076 | 0.036 |
| Fe | (%) | 0.003 | 0.011 | 0.026 | 0.002 |
| Mn | (%) | 0.0003 | 0.001 | 0.002 | 0.006 |
| P | (%) | 0.009 | 0.023 | 0.012 | 0.006 |
| Ti | (%) | 0.0001 | 0.0002 | 0.0003 | 0.0005 |
| Ca | (%) | 37.89 | 37.09 | 36.34 | 38.82 |
| Mg | (%) | bl | bl | 0.046 | bl |
| Na | (%) | 1.52 | 1.47 | 1.22 | 2.06 |
| K | (%) | 0.021 | 0.045 | 0.048 | 0.1 |
| Cr | (μg/g) | 2.5 | 2.6 | 2.9 | 5.3 |
| Co | (μg/g) | 0.7 | 0.8 | 0.9 | 1.4 |
| Ni | (μg/g) | 9.3 | 10.6 | 10.2 | 12.0 |
| Cu | (μg/g) | 1.3 | 3.1 | 2.7 | 3.8 |
| Zn | (μg/g) | 5.0 | 8.0 | 7.0 | 12.3 |
| Cd | (μg/g) | 0.1 | 0.1 | 0.1 | 0.1 |
| Pb | (μg/g) | 0.8 | 1.0 | 1.1 | 3.3 |
| U | (μg/g) | 4.6 | 4.4 | 6.9 | 7.0 |
| Ba | (μg/g) | 16.2 | 23.4 | 20.4 | 29.5 |
| Th | (μg/g) | 0.1 | 0.1 | 0.2 | 0.1 |
| Sr | (μg/g) | 9218.0 | 9248.0 | 9706.0 | 9853.0 |
References
- Roberts, J.M.; Wheeler, A.J.; Freiwald, A. Reefs of the deep: The biology and geology of cold-water coral ecosystems. Science 2006, 312, 543–547. [Google Scholar] [CrossRef] [PubMed]
- Van de Flierdt, T.; Robinson, L.F.; Adkins, J.F. Deep-sea coral aragonite as a recorder for the neodymium isotopic composition of seawater. Geochim. Cosmochim. Acta 2010, 74, 6014–6032. [Google Scholar] [CrossRef]
- Frank, N.; Paterne, M.; Ayliffe, L.; van Weering, T.; Henriet, J.P.; Blamart, D. Eastern North Atlantic deep-seacorals: Tracing up per intermediate water Delta C-14 during the Holocene. Earth Planet. Sci. Lett. 2004, 219, 297–309. [Google Scholar] [CrossRef]
- Davies, A.J.; Guinotte, J.M. Global habitat suitability for framework-forming cold-water corals. PLoS ONE 2011, 6, e18483. [Google Scholar] [CrossRef] [PubMed]
- Margolin, A.R.; Robinson, L.F.; Burke, A.; Waller, R.G.; Scanlon, K.M.; Roberts, M.L.; Auro, M.E.; Van de Flierdt, T. Temporal and spatial distributions of cold-water corals in the Drake Passage: Insights from the last 35,000 years. Deep-Sea Res. Part II 2014, 99, 237–248. [Google Scholar] [CrossRef]
- Hovland, M.; Risk, M. Do Norwegian deep-water coral reefs rely on seeping fluids? Mar. Geol. 2003, 198, 83–96. [Google Scholar] [CrossRef]
- Raimundo, J.; Vale, C.; Caetano, M.; Anes, B.; Carreiro-Silva, M.; Martins, I.; de Matos, V.; Porteiro, F.M. Element concentrations in cold-water gorgonians and black coral from Azores region. Deep-Sea Res. Part II 2013, 98, 129–136. [Google Scholar] [CrossRef]
- Edwards, R.; Chen, J.H.; Wasserburg, G.J. 238U 234U 230Th 232Th systematics and the precise measurement of time over the past 500,000 years. Earth Planet. Sci. Lett. 1987, 81, 175–192. [Google Scholar] [CrossRef]
- Mortlock, R.A.; Fairbanks, R.G.; Chiu, T.C.; Rubenstone, J. Th-230/U-234/U-238 and Pa-231/U-235 ages from a single fossil coral fragment by multi-collector magnetic-sector inductively coupled plasma mass spectrometry. Geochim. Cosmochim. Acta 2005, 69, 649–657. [Google Scholar] [CrossRef]
- Robinson, L.F.; Adkins, J.F.; Fernandez, D.P.; Burnett, D.S.; Wang, S.L.; Gagnon, A.C.; Krakauer, N. Primary U distribution in scleractinian corals and its implications for U series dating. Geochem. Geophy. Geosy. 2006, 7, Q05022. [Google Scholar] [CrossRef]
- Chen, T.Y.; Robinson, L.F.; Burke, A.; Southon, J.; Spooner, P.; Morris, P.J.; Ng, H.C. Synchronous centennial abrupt events in the ocean and atmosphere during the last deglaciation. Science 2015, 349, 1537–1541. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.Y.; Robinson, L.F.; Beasley, M.P.; Claxton, L.M.; Andersen, M.B.; Gregoire, L.J.; Wadham, J.; Fornari, D.J.; Harpp, K.S. Ocean mixing and ice-sheet control of seawater U-234/U-238 during the last deglaciation. Science 2016, 354, 626–629. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Eltgroth, S.F.; Boyle, E.A.; Adkins, J.E. The transfer of bomb radiocarbon and anthropogenic lead to the deep North Atlantic Ocean observed from a deep sea coral. Earth Planet. Sci. Lett. 2017, 458, 223–232. [Google Scholar] [CrossRef]
- Burke, A.; Robinson, L.F. The Southern Ocean’s Role in Carbon Exchange during the Last Deglaciation. Science 2012, 335, 557–561. [Google Scholar] [CrossRef]
- Hines, S.K.V.; Southon, J.R.; Adkins, J.F. A high-resolution record of Southern Ocean intermediate water radiocarbon over the past 30,000 years. Earth Planet. Sci. Lett. 2015, 432, 46–58. [Google Scholar] [CrossRef]
- Robinson, L.F.; van de Flierdt, T. Southern Ocean evidence for reduced export of North Atlantic Deep Water during Heinrich event 1. Geology 2009, 37, 195–198. [Google Scholar] [CrossRef]
- Copard, K.; Colin, C.; Henderson, G.M.; Scholten, J.; Douville, E.; Sicre, M.A.; Frank, N. Late Holocene intermediate water variability in the northeastern Atlantic as recorded by deep-sea corals. Earth Planet. Sci. Lett. 2012, 313, 34–44. [Google Scholar] [CrossRef]
- Montero-Serrano, J.C.; Frank, N.; Tisnerat-Laborde, N.; Colin, C.; Wu, C.C.; Lin, K.; Shen, C.C.; Copard, K.; Orejas, C.; Gori, A.; et al. Decadal changes in the mid-depth water mass dynamic of the Northeastern Atlantic margin (Bay of Biscay). Earth Planet. Sci. Lett. 2013, 364, 134–144. [Google Scholar] [CrossRef]
- Mitsuguchi, T.; Matsumoto, E.; Abe, O.; Uchida, T.; Isdale, P.J. Mg/Ca thermometry in coral-skeletons. Science 1996, 274, 961–963. [Google Scholar] [CrossRef]
- Turley, C.M.; Roberts, J.M.; Guinotte, J.M. Corals in deep-water: Will the unseen hand of ocean acidification destroy cold-water ecosystems? Coral Reefs 2007, 26, 445–448. [Google Scholar] [CrossRef]
- Tittensor, D.P.; Baco, A.R.; Brewin, P.E.; Clark, M.R.; Consalvey, M.; Hall-Spencer, J.; Rowden, A.A.; Schlacher, T.; Stocks, K.I.; Rogers, A.D. Predicting global habitat suitability for stony corals on seamounts. J. Biogeogr. 2009, 36, 1111–1128. [Google Scholar] [CrossRef]
- Hovland, M.; Thomsen, E. Cold-water corals—Are they hydrocarbon seep related? Mar. Geol. 1997, 137, 159–164. [Google Scholar] [CrossRef]
- Naeth, J.; di Primio, R.; Horsfield, B.; Schaefer, R.G.; Shannon, P.M.; Bailey, W.R.; Henriet, J.P. Hydrocarbon seepage and carbonate mound formation: A basin modelling study from the porcupine basin (offshore Ireland). J. Pet. Geol. 2005, 28, 147–165. [Google Scholar] [CrossRef]
- Hovland, M. Deep-Water Coral Reefs: Unique Biodiversity Hot–Spots; Praxis Publishing: Chichester, UK, 2008; p. 278. [Google Scholar]
- Lindberg, B.; Berndt, C.; Mienert, J. The Fugløy Reef at 701 N; acoustic signature, geologic, geomorphologic and oceanographic setting. Int. J. Earth Sci. 2007, 96, 201–213. [Google Scholar] [CrossRef]
- Liebetrau, V.; Eisenhauer, A.; Linke, P. Cold seep carbonates and associated cold-water corals at the Hikurangi Margin, New Zealand: New insights into fluid pathways, growth structures and geochronology. Mar. Geol. 2010, 272, 307–318. [Google Scholar] [CrossRef]
- Jones, A.T.; Greinert, J.; Bowden, D.A.; Klaucke, I.; Petersen, C.J.; Netzeband, G.L.; Weinrebe, W. Acoustic and visual characterisation of methane-rich seabed seeps at Omakere Ridge on the Hikurangi Margin, New Zealand. Mar. Geol. 2010, 272, 154–169. [Google Scholar] [CrossRef]
- León, R.; Somoza, L.; Medialdea, T.; González, F.J.; Diaz-del-Río, V.; Fernández-Puga, M.C.; Maestro, A.; Mata, M.P. Sea-floor features related to hydrocarbon seeps in deep water carbonate-mud mounds of the Gulf of Cádiz: From mud flows to carbonate precipitates. Geo.-Mar. Lett. 2007, 27, 237–247. [Google Scholar] [CrossRef]
- Schroeder, W.W. Observations of Lophelia pertusa and the surficial geology at a deep-water site in the northeastern Gulf of Mexico. Hydrobiologia 2002, 471, 29–33. [Google Scholar] [CrossRef]
- Pirlet, H.; Wehrmann, L.M.; Foubert, A.; Brunner, B.; Blamart, D.; De Mol, L.; Van Rooij, D.; Dewanckele, J.; Cnudde, V.; Swennen, R.; et al. Unique authigenic mineral assemblages reveal different diagenetic histories in two neighbouring cold-water coral mounds on Pen Duick Escarpment, Gulf of Cadiz. Sedimentology 2012, 59, 578–604. [Google Scholar] [CrossRef]
- Hovland, M.; Mortensen, P.B.; Brattegard, T.; Strass, P.; Rokoengen, K. Ahermatypic coral banks off Mid-Norway: Evidence for a link with seepage of light hydrocarbons. Palaios 1998, 13, 189–200. [Google Scholar] [CrossRef]
- Hovland, M.; Jensen, S.; Indreiten, T. Unit pockmarks associated with Lophelia coral reefs off mid-Norway: More evidence of control by ‘fertilizing’ bottom currents. Geo.-Mar. Lett. 2012, 32, 545–554. [Google Scholar] [CrossRef]
- Rincon-Tomas, B.; Duda, J.P.; Somoza, L.; Gonzalez, F.J.; Schneider, D.; Medialdea, T.; Santofimia, E.; Lopez-Pamo, E.; Madureira, P.; Hoppert, M.; et al. Cold-water corals and hydrocarbon-rich seepage in Pompeia Province (Gulf of Cadiz)-living on the edge. Biogeosciences 2019, 16, 1607–1627. [Google Scholar] [CrossRef]
- Thiel, V.; Peckmann, J.; Seifert, R.; Wehrung, P.; Reitner, J.; Michaelis, W. Highly isotopically depleted isoprenoids: Molecular markers for ancient methane venting. Geochim. Cosmochim. Acta 1999, 63, 3959–3966. [Google Scholar] [CrossRef]
- Boetius, A.; Ravenschlag, K.; Schubert, C.J.; Rickert, D.; Widdel, F.; Gieseke, A.; Amann, R.; Jorgensen, B.B.; Witte, U.; Pfannkuche, O. A marine microbial consortium apparently mediating anaerobic oxidation of methane. Nature 2000, 407, 623–626. [Google Scholar] [CrossRef] [PubMed]
- Peckmann, J.; Gischler, E.; Oschmann, W.; Reitner, J. An early carboniferous seep community and hydrocarbon-derived carbonates from the Harz mountains, Germany. Geology 2001, 29, 471. [Google Scholar] [CrossRef]
- Peckmann, J.; Thiel, V. Carbon cycling at ancient methane-seeps. Chem. Geol. 2004, 205, 443–467. [Google Scholar] [CrossRef]
- Reeburgh, W.S. Oceanic methane biogeochemistry. Chem. Rev. 2007, 107, 486–513. [Google Scholar] [CrossRef]
- Feng, D.; Lin, Z.J.; Bian, Y.Y.; Chen, D.F.; Peckmann, J.; Bohrmann, G.; Roberts, H.H. Rare earth elements of seep carbonates: Indication for redox variations and microbiological processes at modern seep sites. J. Asian Earth Sci. 2013, 65, 27–33. [Google Scholar] [CrossRef]
- Van Rooij, D.; Blamart, D.; De Mol, L.; Mienis, F.; Pirlet, H.; Wehrmann, L.M.; Barbieri, R.; Maignien, L.; Templer, S.P.; de Haas, H.; et al. Cold-water coral mounds on the Pen Duick Escarpment, Gulf of Cadiz: The MICROSYSTEMS project approach. Mar. Geol. 2011, 282, 102–117. [Google Scholar] [CrossRef]
- Magalhaes, V.H.; Pinheiro, L.M.; Ivanov, M.K.; Kozlova, E.; Blinova, V.; Kolganova, J.; Vasconcelos, C.; McKenzie, J.A.; Bernasconi, S.M.; Kopf, A.J.; et al. Formation processes of methane-derived authigenic carbonates from the Gulf of Cadiz. Sediment. Geol. 2012, 243, 155–168. [Google Scholar] [CrossRef]
- Le Bris, N.; Arnaud-Haond, S.; Beaulieu, S.; Cordes, E.E.; Hilario, A.; Rogers, A.; van de Gaever, S.; Watanabe, H. Hydrothermal Vents and Cold Seeps. In The First Global Integrated Marine Assessment, United Nations; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- Vandorpe, T.; Martins, I.; Vitorino, J.; Hebbeln, D.; Garcia, M.; Van Rooij, D. Bottom currents and their influence on the sedimentation pattern in the El Arraiche mud volcano province, southern Gulf of Cadiz. Mar. Geol. 2016, 378, 114–126. [Google Scholar] [CrossRef]
- Roberts, H.H.; Kohl, B. Temperature control of cold-water coral (Lophelia) mound growth by climate-cycle forcing, Northeast Gulf of Mexico. Deep-Sea Res. Part II 2018, 140, 142–158. [Google Scholar] [CrossRef]
- Bassett-Smith, P.W. XLIII.—Report on the corals from the Tizard and Macclesfield Banks, China Sea. J. Nat. Hist. Ser. 1890, 6, 353–374. [Google Scholar] [CrossRef]
- Zou, R.L. Studies on the deep water scleractinia from South China Sea—II. Record and narration of species as well as time-spatial distributional characteristics. Trop. Oceanol. 1988, 7, 74–83, (In Chinese with English Abstract). [Google Scholar]
- Liu, Z.F.; Zhao, Y.L.; Colin, C.; Stattegger, K.; Wiesner, M.G.; Huh, C.A.; Zhang, Y.W.; Li, X.J.; Sompongchaiyakul, P.; You, C.F.; et al. Source-to-sink transport processes of fluvial sediments in the South China Sea. Earth-Sci. Rev. 2016, 153, 238–273. [Google Scholar] [CrossRef]
- Zhang, G.X.; Liang, J.Q.; Lu, J.A.; Yang, S.X.; Zhang, M.; Holland, M.; Schultheiss, P.; Su, X.; Sha, Z.B.; Xu, H.N.; et al. Geological features, controlling factors and potential prospects of the gas hydrate occurrence in the east part of the Pearl River Mouth Basin, South China Sea. Mar. Pet. Geol. 2015, 67, 356–367. [Google Scholar] [CrossRef]
- Suess, E. RV SONNE cruise report SO 177, Sino-German cooperative project, South China Sea Continental Margin: Geological methane budget and environmental effects of methane emissions and gas hydrates. IFM-GEOMAR Reports 2005. Available online: http://store.pangaea.de/documentation/Reports/SO177.pdf (accessed on 29 November 2019).
- Han, X.Q.; Suess, E.; Huang, Y.Y.; Wu, N.Y.; Bohrrnann, G.; Su, X.; Eisenhauer, A.; Rehder, G.; Fang, Y.X. Jiulong methane reef: Microbial mediation of seep carbonates in the South China Sea. Mar. Geol. 2008, 249, 243–256. [Google Scholar] [CrossRef]
- Feng, D.; Cheng, M.; Kiel, S.; Qiu, J.W.; Yang, Q.H.; Zhou, H.Y.; Peng, Y.B.; Chen, D.F. Using Bathymodiolus tissue stable carbon, nitrogen and sulfur isotopes to infer biogeochemical process at a cold seep in the South China Sea. Deep-Sea Res. Part II 2015, 104, 52–59. [Google Scholar] [CrossRef]
- Sha, Z.B.; Liang, J.Q.; Zhang, G.X.; Yang, S.X.; Lu, J.G.; Zhang, Z.J.; McConnell, D.R.; Humphrey, G. A seepage gas hydrate system in northern South China Sea: Seismic and well log interpretations. Mar. Geol. 2015, 366, 69–78. [Google Scholar] [CrossRef]
- Chen, F.; Hu, Y.; Feng, D.; Zhang, X.; Cheng, S.H.; Cao, J.; Lu, H.F.; Chen, D.F. Evidence of intense methane seepages from molybdenum enrichments in gas hydrate-bearing sediments of the northern South China Sea. Chem. Geol. 2016, 443, 173–181. [Google Scholar] [CrossRef]
- Hu, Y.; Feng, D.; Peckmann, J.; Roberts, H.H.; Chen, D.F. New insights into cerium anomalies and mechanisms of trace metal enrichment in authigenic carbonate from hydrocarbon seeps. Chem. Geol. 2014, 381, 55–66. [Google Scholar] [CrossRef]
- Foubert, A.; Depreiter, D.; Beck, T.; Maignien, L.; Pannemans, B.; Frank, N.; Blamart, D.; Henriet, J.P. Carbonate mounds in a mud volcano province off north-west Morocco: Key to processes and controls. Mar. Geol. 2008, 248, 74–96. [Google Scholar] [CrossRef]
- Somoza, L.; Ercilla, G.; Urgorri, V.; Leon, R.; Medialdea, T.; Paredes, M.; Gonzalez, F.J.; Nombela, M.A. Detection and mapping of cold-water coral mounds and living Lophelia reefs in the Galicia Bank, Atlantic NW Iberia margin. Mar. Geol. 2014, 349, 73–90. [Google Scholar] [CrossRef]
- De Mol, B.; Van Rensbergen, P.; Pillen, S.; Van Herreweghe, K.; Van Rooij, D.; McDonnell, A.; Huvenne, V.; Ivanov, M.; Swennen, R.; Henriet, J.P. Large deep-water coral banks in the Porcupine Basin, southwest of Ireland. Mar. Geol. 2002, 188, 193–231. [Google Scholar] [CrossRef]
- Zoccola, D.; Ganot, P.; Bertucci, A.; Caminiti-Segonds, N.; Techer, N.; Voolstra, C.R.; Aranda, M.; Tambutte, E.; Allemand, D.; Casey, J.R.; et al. Bicarbonate transporters in corals point towards a key step in the evolution of cnidarian calcification. Sci. Rep. 2015, 5, 9983. [Google Scholar] [CrossRef]
- Nakamura, T.; Nadaoka, K.; Watanabe, A.; Yamamoto, T.; Miyajima, T.; Blanco, A.C. Reef-scale modeling of coral calcification responses to ocean acidification and sea-level rise. Coral Reefs 2018, 37, 37–53. [Google Scholar] [CrossRef]
- Sherwood, O.A.; Jamieson, R.E.; Edinger, E.N.; Wareham, V.E. Stable C and N isotopic composition of cold-water corals from the Newfoundland and Labrador continental slope: Examination of trophic, depth and spatial effects. Deep-Sea Res. Part II 2008, 55, 1392–1402. [Google Scholar] [CrossRef]
- Levin, L.A. Ecology of cold seep sediments: Interactions of fauna with flow, chemistry and microbes. Oceanogr. Mar. Biol. 2005, 43, 1–46. [Google Scholar]
- Whiticar, M.J. Carbon and hydrogen isotope systematics of bacterial formation and oxidation of methane. Chem. Geol. 1999, 161, 291–314. [Google Scholar] [CrossRef]
- Sackett, W.M. Carbon and hydrogen isotope effects during the thermocatalytic production of hydrocarbons in laboratory simulation experiments. Geochim. Cosmochim. Acta 1978, 42, 571–580. [Google Scholar] [CrossRef]
- Zhang, M.S.; He, Q.X. A study on quaternary stratigraphy of reef deposits on Xisha Islands. Quat. Sci. 1989, 9, 143–154, (In Chinese with English Abstract). [Google Scholar]
- Ke, T.; Wei, G.J.; Liu, Y.; Xie, L.H.; Deng, W.F.; Wang, G.Q.; Xu, J.F. High resolution boron isotopic compositions of a coral from the northern South China Sea and their implications for reconstruction of seawater pH. Geochimica 2015, 44, 1–8, (In Chinese with English Abstract). [Google Scholar]
- Tran, T.H.; Kato, K.; Wada, H.; Fujioka, K.; Matsuzaki, H. Processes involved in calcite and aragonite precipitation during carbonate chimney formation on Conical Seamount, Mariana Forearc: Evidence from geochemistry and carbon, oxygen, and strontium isotopes. J. Geochem. Explor. 2014, 137, 55–64. [Google Scholar] [CrossRef]
- Fruh-Green, G.L.; Kelley, D.S.; Bernasconi, S.M.; Karson, J.A.; Ludwig, K.A.; Butterfield, D.A.; Boschi, C.; Proskurowski, G. 30,000 years of hydrothermal activity at the Lost City vent field. Science 2003, 301, 495–498. [Google Scholar] [CrossRef]
- Blamart, D.; Rollion-Bard, C.; Cuif, J.P.; Juillet-Leclerc, A.; Lutringer, A.; van Weering, T.C.E.; Henriet, J.P. C and O Isotopes in A Deep-Sea Coral (Lophelia Pertusa) Related to Skeletal Microstructure. In Cold-Water Corals and Ecosystems; Freiwald, A., Roberts, J.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 1005–1020. [Google Scholar]
- Shirai, K.; Kusakabe, M.; Nakai, S.; Ishii, T.; Watanabe, T.; Hiyagon, H.; Sano, Y. Deep-sea coral geochemistry: Implication for the vital effect. Chem. Geol. 2005, 224, 212–222. [Google Scholar] [CrossRef]
- Chen, Z.; Mo, A.; Zhao, M.X.; Zhong, Y.; Yan, W. The discovery of deep-water corals from cold seep area in the northern South China Sea and their characteristics. J. Trop. Oceanogr. 2018, 37, 64–71, (In Chinese with English Abstract). [Google Scholar]
- Xu, A.; Chen, Z.; Qu, Y.; Tian, Y.; Shu, C.; Zheng, X.; Li, G.; Yan, W.; Zhao, M. Cold-water corals in a cold seep area on the northern continental slopes of the South China sea and their isotopic characteristics. Deep-Sea Res. I 2019, 153, 103043. [Google Scholar] [CrossRef]
- Becker, E.L.; Cordes, E.E.; Macko, S.A.; Fisher, C.R. Importance of seep primary production to Lophelia pertusa and associated fauna in the Gulf of Mexico. Deep-Sea Res. Part I 2009, 56, 786–800. [Google Scholar] [CrossRef]
- Fallon, S.J.; White, J.C.; McCulloch, M.T. Porites corals as recorders of mining and environmental impacts: Misima Island, Papua New Guinea. Geochim. Cosmochim. Acta 2002, 66, 45–62. [Google Scholar] [CrossRef]
- Robinson, L.F.; Adkins, J.F.; Frank, N.; Gagnon, A.C.; Prouty, N.G.; Roark, E.B.; van de Flierdt, T. The geochemistry of deep-sea coral skeletons: A review of vital effects and applications for palaeoceanography. Deep-Sea Res. Part II 2014, 105, 118. [Google Scholar] [CrossRef]
- Ramos, A.A.; Inoue, Y.; Ohde, S. Metal contents in Porites corals: Anthro- pogenic input of river run-off into a coral reef from an urbanized area, Okinawa. Mar. Pollut. Bull. 2004, 48, 281–294. [Google Scholar] [CrossRef]
- Sinclair, D.J.; Williams, B.; Allard, G.; Ghaleb, B.; Fallon, S.; Ross, S.W.; Risk, M. Reproducibility of trace element profiles in a specimen of the deep-water bamboo coral Keratoisis sp. Geochim. Cosmochim. Acta 2011, 75, 5101–5121. [Google Scholar] [CrossRef]
- Crocket, K.C.; Lambelet, M.; de Flierdt, T.V.; Rehkamper, M.; Robinson, L.F. Measurement of fossil deep-sea coral Nd isotopic compositions and concentrations by TIMS as NdO+, with evaluation of cleaning protocols. Chem. Geol. 2014, 374, 128–140. [Google Scholar] [CrossRef]
- Torres, M.E.; McManus, J.; Huh, C.A. Fluid seepage along the San Clemente Fault scarp: Basin-wide impact on barium cycling. Earth Planet. Sci. Lett. 2002, 203, 181–194. [Google Scholar] [CrossRef]
- Feng, D.; Roberts, H.H. Geochemical characteristics of the barite deposits at cold seeps from the northern Gulf of Mexico continental slope. Earth Planet. Sci. Lett. 2011, 309, 89–99. [Google Scholar] [CrossRef]
- Hu, Y.; Feng, D.; Liang, Q.Y.; Xia, Z.; Chen, L.Y.; Chen, D.F. Impact of anaerobic oxidation of methane on the geochemical cycle of redox-sensitive elements at cold-seep sites of the northern South China Sea. Deep-Sea Res. Part II 2015, 122, 84–94. [Google Scholar] [CrossRef]
- Liang, Q.Y.; Hu, Y.; Feng, D.; Peckmann, J.; Chen, L.Y.; Yang, S.X.; Liang, J.Q.; Tao, J.; Chen, D.F. Authigenic carbonates from newly discovered active cold seeps on the northwestern slope of the South China Sea: Constraints on fluid sources, formation environments, and seepage dynamics. Deep-Sea Res. Part II 2017, 124, 31–41. [Google Scholar] [CrossRef]
- Peckmann, J.; Birgel, D.; Kiel, S. Molecular fossils reveal fluid composition and flow intensity at a Cretaceous seep. Geology 2009, 37, 847–850. [Google Scholar] [CrossRef]
- Roberts, H.H.; Feng, D.; Joye, S.B. Cold-seep carbonates of the middle and lower continental slope, northern Gulf of Mexico. Deep-Sea Res. Part II 2010, 57, 2040–2054. [Google Scholar] [CrossRef]
- Bayon, G.; Dupre, S.; Ponzevera, E.; Etoubleau, J.; Cheron, S.; Pierre, C.; Mascle, J.; Boetius, A.; de Lange, G.J. Formation of carbonate chimneys in the Mediterranean Sea linked to deep-water oxygen depletion. Nat. Geosci. 2013, 6, 755–760. [Google Scholar] [CrossRef]
- Feng, D.; Qiu, J.W.; Hu, Y.; Peckmann, J.; Guan, H.X.; Tong, H.P.; Chen, C.; Chen, J.X.; Gong, S.G.; Li, N.; et al. Cold seep systems in the South China Sea: An overview. J. Asian Earth Sci. 2018, 168, 3–16. [Google Scholar] [CrossRef]
- Teichert, B.M.A.; Eisenhauer, A.; Bohrmann, G.; Haase-Schramm, A.; Bock, B.; Linke, P. U/Th systematics and ages of authigenic carbonates from Hydrate Ridge, Cascadia Margin: Recorders of fluid flow variations. Geochim. Cosmochim. Acta 2003, 67, 3845–3857. [Google Scholar] [CrossRef]
- Watanabe, Y.; Nakai, S.; Hiruta, A.; Matsumoto, R.; Yoshida, K. U-Th dating of carbonate nodules from methane seeps off Joetsu, Eastern Margin of Japan Sea. Earth Planet. Sci. Lett. 2008, 272, 89–96. [Google Scholar] [CrossRef]
- Cremiere, A.; Lepland, A.; Chand, S.; Sahy, D.; Condon, D.J.; Noble, S.R.; Martma, T.; Thorsnes, T.; Sauer, S.; Brunstad, H. Timescales of methane seepage on the Norwegian margin following collapse of the Scandinavian Ice Sheet. Nat. Commun. 2016, 7, 11509. [Google Scholar] [CrossRef]
- Wu, D.D.; Sun, T.T.; Xie, R.; Pan, M.D.; Chen, X.G.; Ye, Y.; Liu, L.H.; Wu, N.Y. Characteristics of Authigenic Minerals around the Sulfate-Methane Transition Zone in the Methane-Rich Sediments of the Northern South China Sea: Inorganic Geochemical Evidence. Int. J. Env. Res. Public Health 2019, 16, 2299. [Google Scholar] [CrossRef]
- Xie, R.; Wu, D.D.; Liu, J.; Sun, T.T.; Liu, L.H.; Wu, N.Y. Geochemical Evidence of Metal-Driven Anaerobic Oxidation of Methane in the Shenhu Area, the South China Sea. Int. J. Env. Res. Public Health 2019, 16, 3559. [Google Scholar] [CrossRef]
- Chen, D.F.; Huang, Y.Y.; Yuan, X.L.; Cathles, L.M. Seep carbonates and preserved methane oxidizing archaea and sulfate reducing bacteria fossils suggest recent gas venting on the seafloor in the northeastern South China Sea. Mar. Pet. Geol. 2005, 22, 613–621. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Deng, Y.; Chen, F.; Li, N.; Jin, M.; Cao, J.; Chen, H.; Zhou, Y.; Wu, C.; Zhuang, C.; Zhao, Y.; et al. Cold-Water Corals in Gas Hydrate Drilling Cores from the South China Sea: Occurrences, Geochemical Characteristics and Their Relationship to Methane Seepages. Minerals 2019, 9, 742. https://doi.org/10.3390/min9120742
Deng Y, Chen F, Li N, Jin M, Cao J, Chen H, Zhou Y, Wu C, Zhuang C, Zhao Y, et al. Cold-Water Corals in Gas Hydrate Drilling Cores from the South China Sea: Occurrences, Geochemical Characteristics and Their Relationship to Methane Seepages. Minerals. 2019; 9(12):742. https://doi.org/10.3390/min9120742
Chicago/Turabian StyleDeng, Yinan, Fang Chen, Niu Li, Meng Jin, Jun Cao, Hong Chen, Yang Zhou, Cong Wu, Chang Zhuang, Yi Zhao, and et al. 2019. "Cold-Water Corals in Gas Hydrate Drilling Cores from the South China Sea: Occurrences, Geochemical Characteristics and Their Relationship to Methane Seepages" Minerals 9, no. 12: 742. https://doi.org/10.3390/min9120742
APA StyleDeng, Y., Chen, F., Li, N., Jin, M., Cao, J., Chen, H., Zhou, Y., Wu, C., Zhuang, C., Zhao, Y., & Cheng, S. (2019). Cold-Water Corals in Gas Hydrate Drilling Cores from the South China Sea: Occurrences, Geochemical Characteristics and Their Relationship to Methane Seepages. Minerals, 9(12), 742. https://doi.org/10.3390/min9120742