Ore-Forming Processes at the Xiajinbao Gold Deposit in Eastern Hebei Province: Constraints from EPMA and LA-ICPMS Analysis
Abstract
1. Introduction
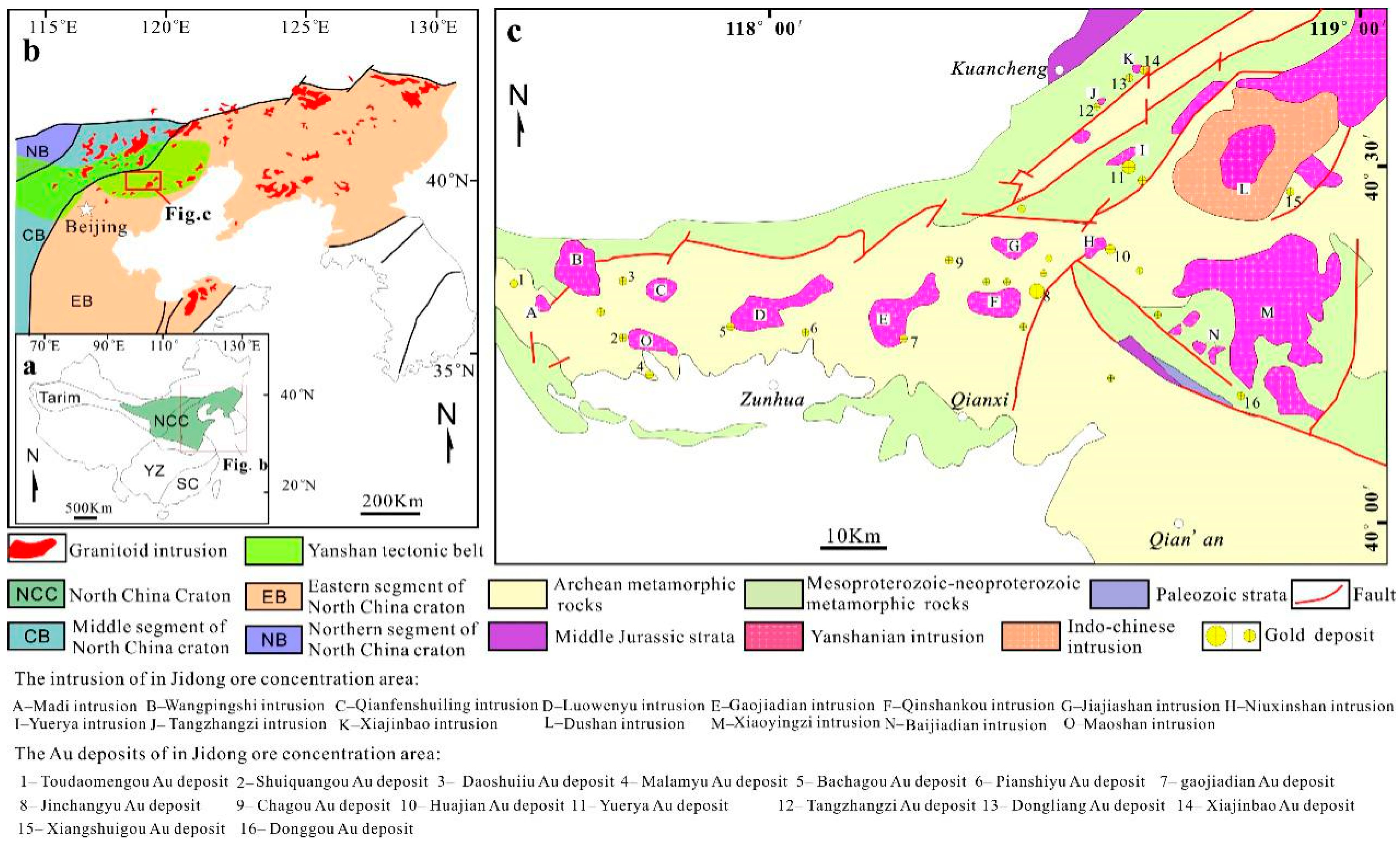
2. Geological Setting
3. Ore deposit Geology
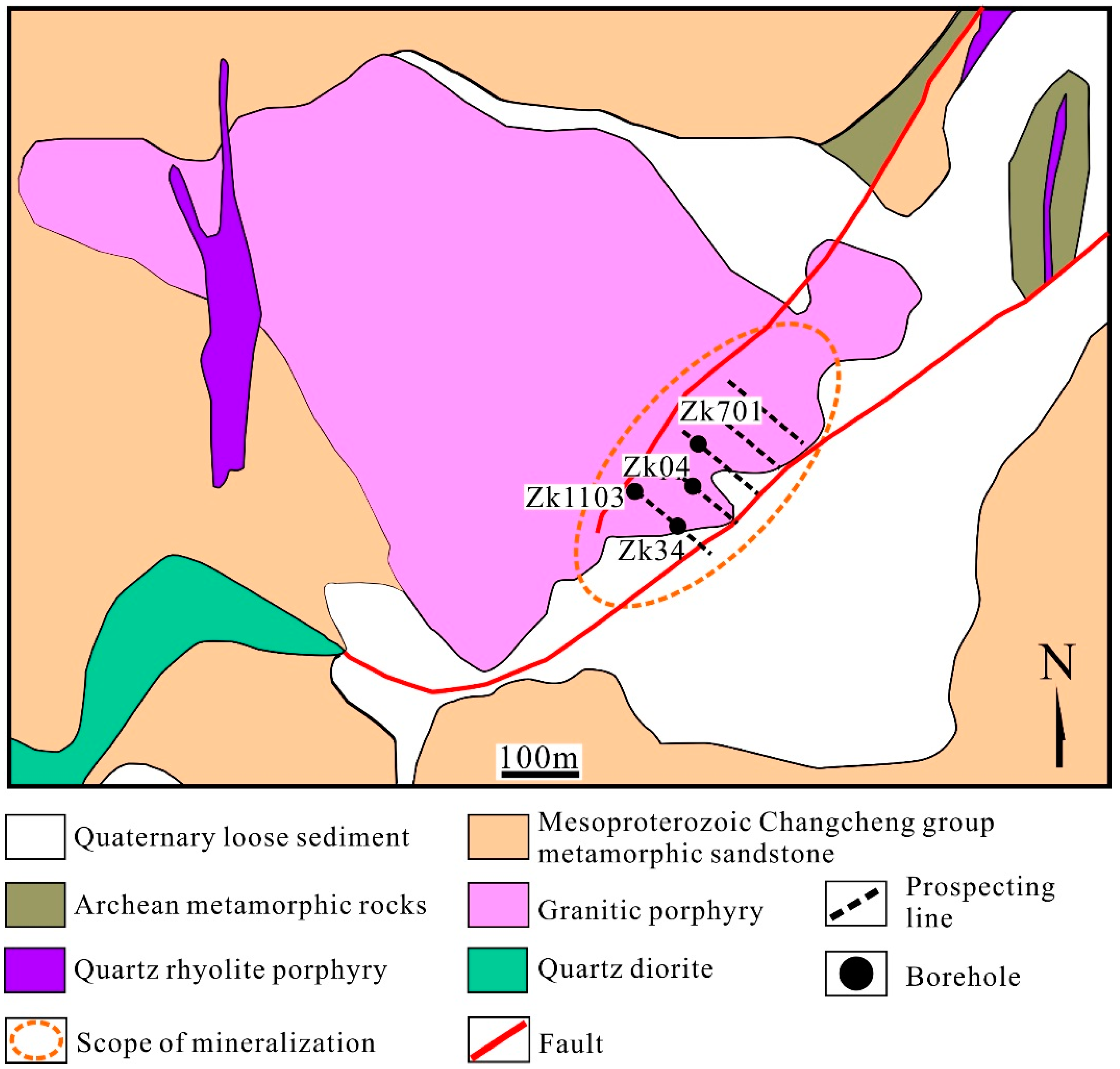
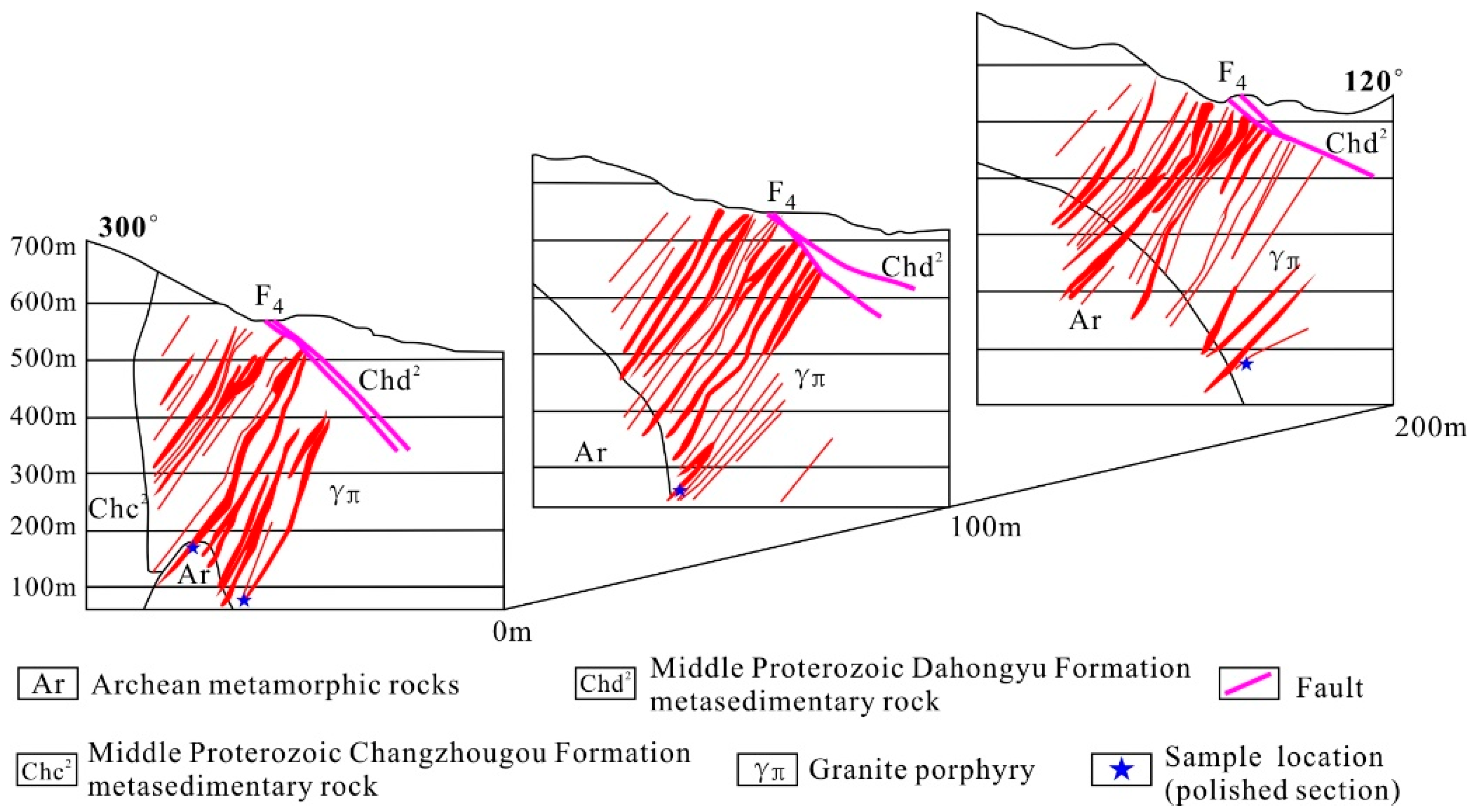
3.1. Host Rocks and Structures
3.2. Ore Bodies, Ore Types, and Mineralogy
3.3. Alteration Assemblages
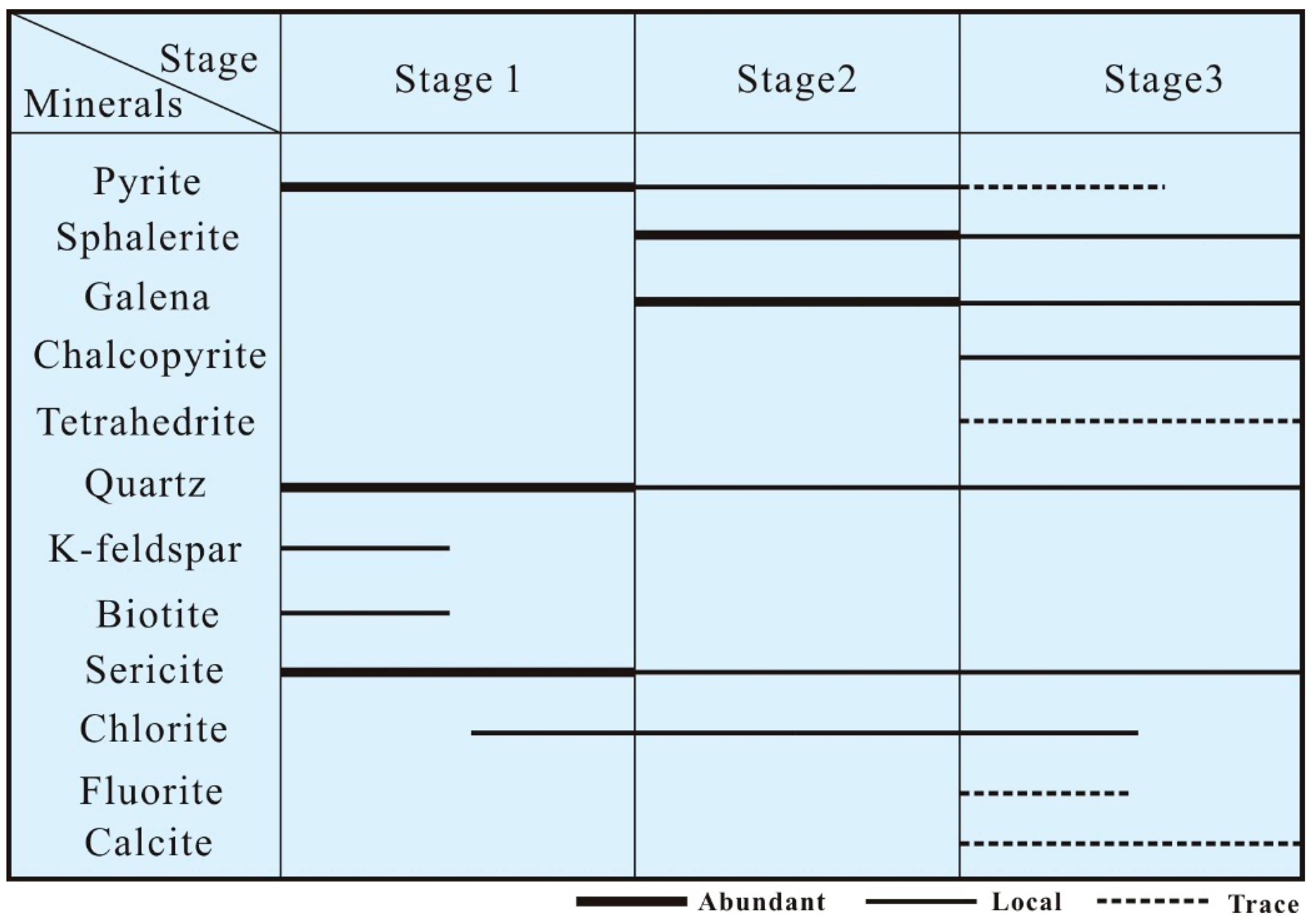
4. Paragenetic Sequences of Mineralization
5. Sampling and Analyses
5.1. Sample Sites
5.2. Microscopic Observations and Electron-Probe Microanalyses
5.3. LA-ICP-MS In-Situ Trace Element Analysis
6. Results
6.1. Petrography and Mineralogy
6.2. Occurrence of Electrum and Gold-Bearing Minerals
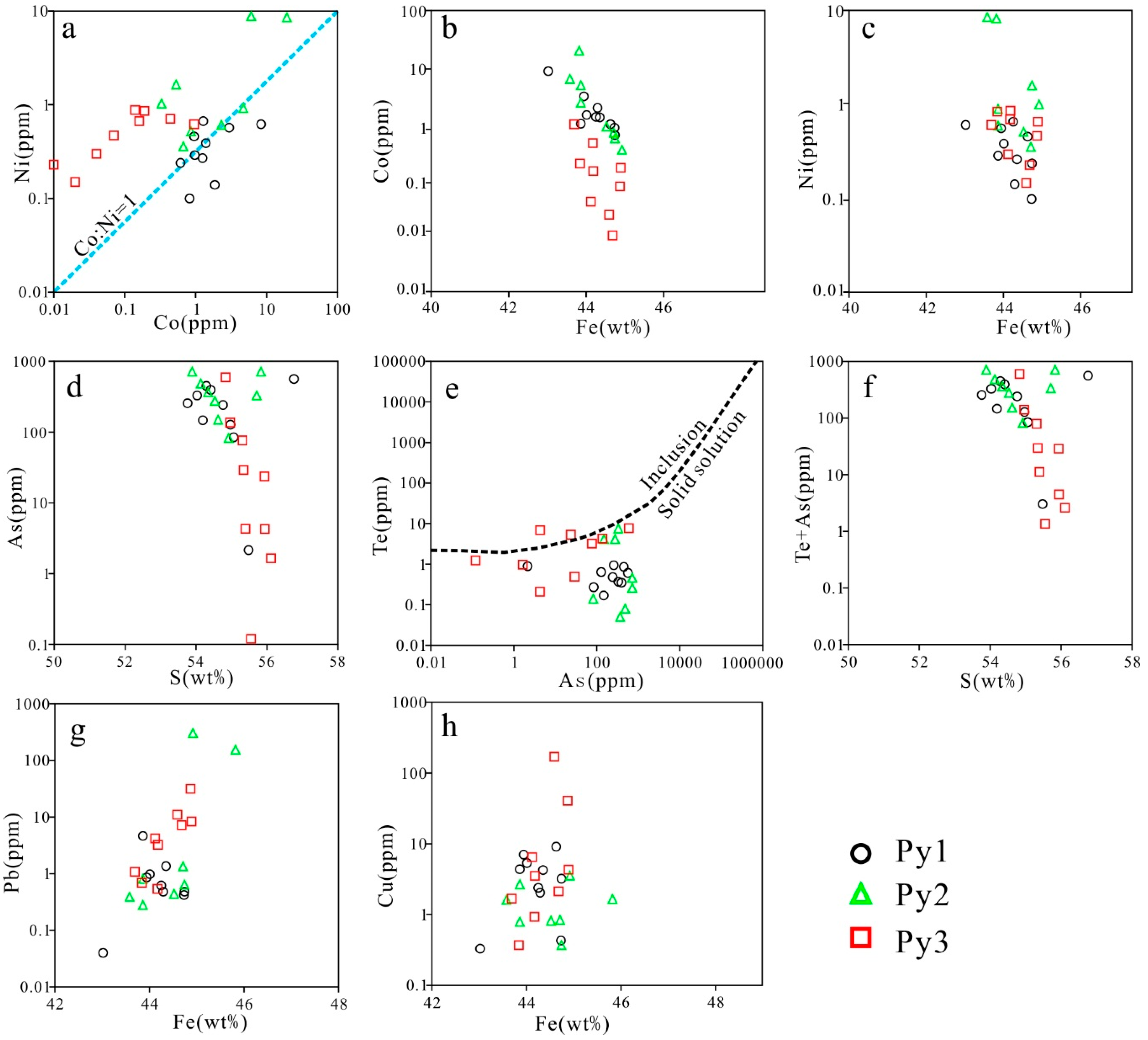
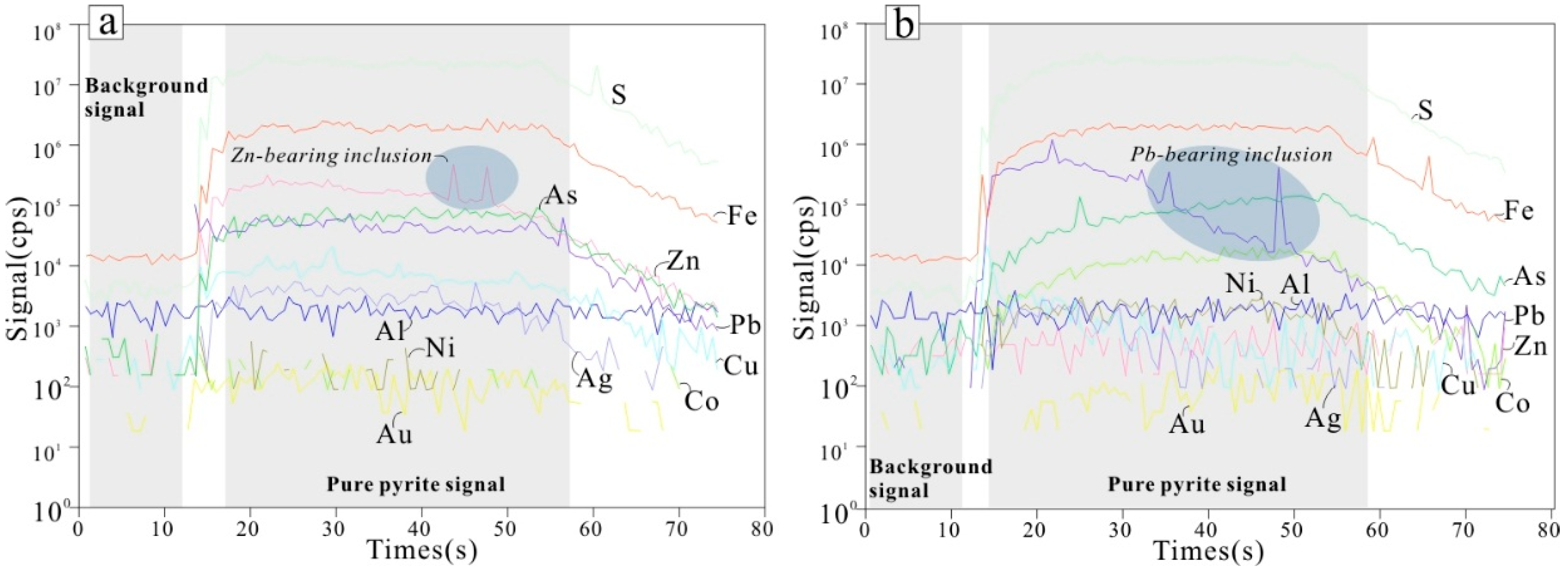
6.3. LA-ICP-MS Trace Element Microscopic in Situ Analysis
7. Discussion
7.1. Trace Element Distributions in Pyrite
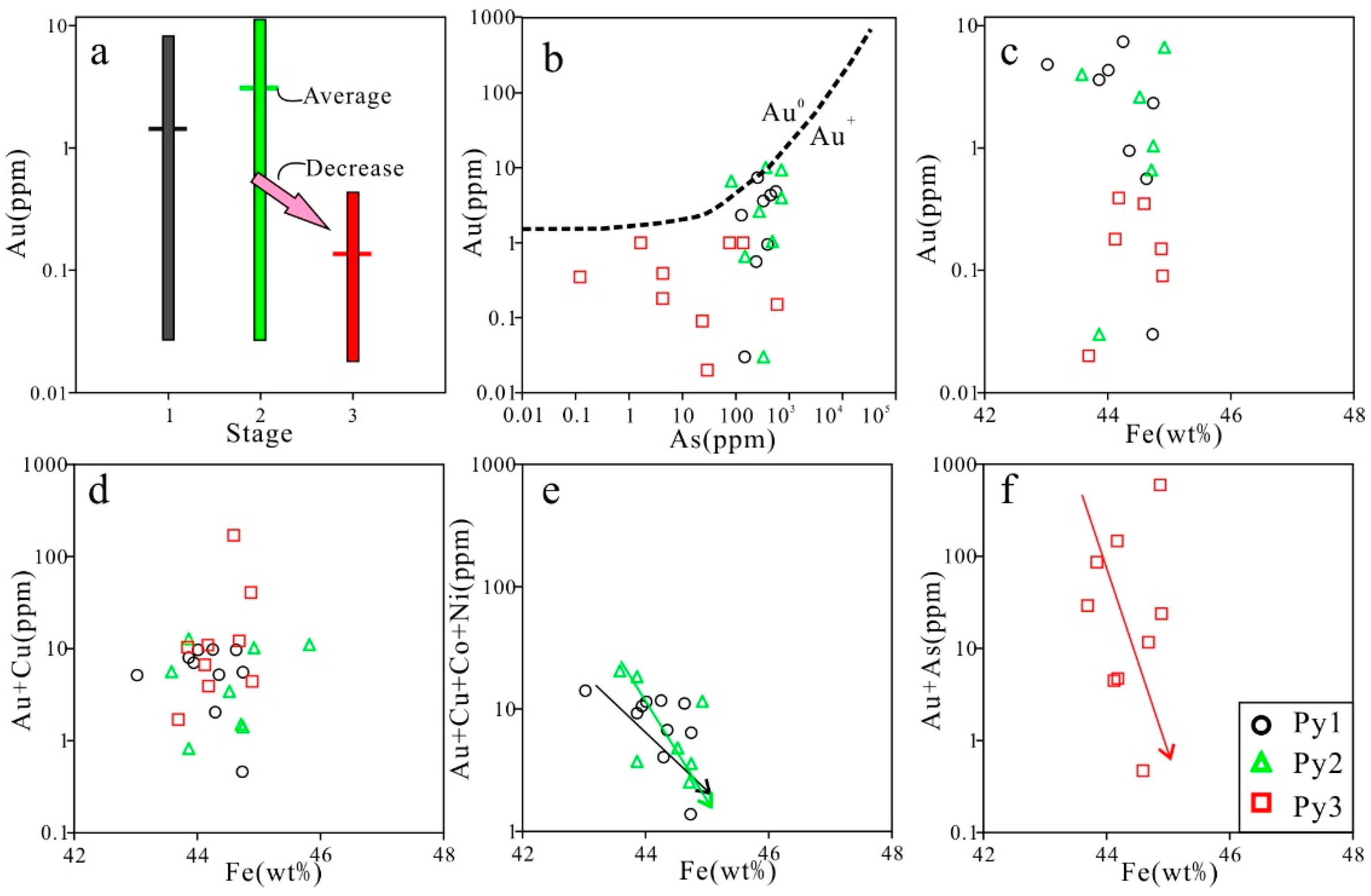
7.2. Pyrite Genesis
7.3. Ore-Forming Process
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hart, C.J.R.; Goldfarb, R.J.; Qiu, Y.; Snee, L.; Miller, L.D.; Miller, M.L. Gold deposits of the northern margin of the North China Craton: Multiple late Paleozoic-Mesozoic mineralizing events. Miner. Depos. 2002, 37, 326–351. [Google Scholar] [CrossRef]
- Wang, W.; Liu, S.; Santosh, M.; Deng, Z.; Guo, B.; Zhao, Y.; Zhang, S.; Yang, P.; Bai, X.; Guo, R. Late Paleoproterozoic geodynamics of the North China Craton: Geochemical and zircon U–Pb–Hf records from a volcanic suite in the Yanliao rift. Gondwana Res. 2015, 27, 300–325. [Google Scholar] [CrossRef]
- Li, H.B.; Ishiyama, D.; Zhang, Y.; Shao, Y.J. Geology and geochemical characteristics of the Xiajinbao gold deposit in the Hebei province, China. J. Miner. Petrol. Sci. 2018, 113, 24–40. [Google Scholar] [CrossRef]
- Mei, Y.X. The metallogenic Characteristic and Evolution of Gold Deposits in Jidong Region, Hebei Province, China. Ph.D. Thesis, Chinese Academy of Geological Sciences, Beijing, China, 1997. (In Chinese). [Google Scholar]
- Song, Y.; Jiang, S.H.; Bagas, L.; Li, C.; Hu, J.Z.; Zhang, Q.; Zhou, W.; Ding, H.Y. The geology and geochemistry of Jinchangyu gold deposit, north China Craton: Implications for metallogenesis and geodynamic setting. Ore Geol. Rev. 2016, 73, 313–329. [Google Scholar] [CrossRef]
- Yuan, W.M.; Wang, S.C.; Wang, L.F. Fission track study on the metallogenetic age of Xiayingfang gold deposit in eastern, Hebei. Nucl. Technol. 1999, 22, 411–413. (In Chinese) [Google Scholar]
- Zou, T.; Wang, Y.W.; Wang, J.B.; Zhang, H.Q.; Zhao, L.T.; Xie, H.J.; Shi, Y.; Liu, Y.Z.; Liu, G.Q. Geochronology of the Xiayingfang Au Deposit in Eastern Hebei Province. Geol. Explor. 2016, 52, 84–97. (In Chinese) [Google Scholar]
- Luan, W.L.; Yu, Y.X. Geochemistry of fluid inclusions of Xiayingfang gold deposit in Pingquan of Hebei province and ore-searching significance. J. Precious Met. Geol. 1995, 4, 161–167. (In Chinese) [Google Scholar]
- Yang, T.D. The study of complex mineralization and metallogenic series of Xiayingfang gold deposit, Hebei province. Earth Sci. 1989, 12, 405–413. (In Chinese) [Google Scholar]
- Shao, J.L.; Mei, J.M. Study on pyrite in a volcanic gold deposit in Pingquan, Hebei province. Gold 1984, 5, 3–8. (In Chinese) [Google Scholar]
- Jia, S.; Wang, E.; Fu, J. Geological differences and mineralization unity of the key gold ore concentrated regions in eastern Hebei and Western Liaoning provinces. Acta Geol. Sin. 2011, 85, 1493–1506. (In Chinese) [Google Scholar]
- Winderbaum, L.; Ciobanu, C.L.; Cook, N.J.; Paul, M.; Metcalfe, A.; Gilbert, S. Multivariate analysis of an LA-ICP-MS trace element dataset for pyrite. Math. Geosci. 2012, 44, 823–842. [Google Scholar] [CrossRef]
- Zhao, H.X.; Frimmel, H.E.; Jiang, S.Y.; Dai, B.Z. LA-ICP-MS trace element analysis of pyrite from the Xiaoqinling gold district, China: Implications for ore genesis. Ore Geol. Rev. 2011, 43, 142–153. [Google Scholar] [CrossRef]
- Zhang, J.; Deng, J.; Chen, H.Y.; Yang, L.Q.; David, C.; Leonid, D.; Gong, Q.J. LA-ICP-MS trace element analysis of pyrite from the Chang’an gold deposit, Sanjiang region, China: Implication for ore-forming process. Gondwana Res. 2014, 26, 557–575. [Google Scholar] [CrossRef]
- Liu, D.Y.; Nutman, A.P.; Compston, W.; Wu, J.S.; Shen, Q.H. Remnants of ≥3800 Ma crust in the Chinese part of the Sino-Korean craton. Geology 1992, 20, 339–342. [Google Scholar] [CrossRef]
- Ma, Y.S.; Zeng, Q.L.; Song, B.; Du, J.J.; Yang, F.Q.; Zhao, Y. SHRTMP U–Pb dating of zircon from Panshan granitoid pluton in Yanshan orogenic belt and its tectonic implications. Acta Petrol. Sin. 2007, 23, 547–556. (In Chinese) [Google Scholar]
- Niu, S.Y.; Li, F.Y.; Chen, H.S.; Sun, A.Q.; Wang, B.D.; Wang, J.Z.; Ma, B.J. The exploration and prognosis in the depth and the periphery of the Jinchangyu gold deposit in eastern Hebei. Chin. Geol. 2012, 39, 999–1006. (In Chinese) [Google Scholar]
- Zhang, G.R.; Xu, J.H.; Wei, H.; Song, G.C.; Zhang, Y.B.; Zhao, J.K.; He, B.; Chen, D.L. Structure, alteration, and fluid inclusion study on deep and surrounding area of the Dongping gold deposit, northern Hebei, China. Acta Petrol. Sin. 2012, 28, 637–651. (In Chinese) [Google Scholar]
- Huang, X.; Ziwei, B.; DePaolo, D.J. Sm–Nd isotope study of early Archean rocks, Qian’an, Hebei Province, China. Geochim. Cosmochim. Acta. 1986, 50, 625–631. [Google Scholar] [CrossRef]
- Jahn, B.M.; Auvray, B.; Cornichet, J.; Bai, Y.L.; Shen, Q.H.; Liu, D.Y. 3.5 Ga old amphibolites from eastern Hebei Province, China: Field occurrence, petrography, Sm–Nd isochron age and REE geochemistry. Precambr. Res. 1986, 34, 311–346. [Google Scholar] [CrossRef]
- Sun, D.Z. The Early Precambrian Geology of the Eastern Hebei; Tianjin Science and Technology Press: Tianjin, China, 1984; pp. 1–273. (In Chinese) [Google Scholar]
- Luo, Z.K.; Miao, L.C.; Guan, F.; Qiu, Y.S.; Qiu, Y.M.; McNaughton, N.J.; Groves, D.I. SHRIMP U–Pb zircon dating of the Dushan granitic batholith and related granite-porphyry dyke, eastern Hebei Province, China, and their geological significance. Geochimica 2003, 32, 173–180. (In Chinese) [Google Scholar]
- Yang, F.L.; Niu, B.G.; Ren, J.S.; Li, S. Zircon U–Pb ages and geochemical characteristics of the Mesozoic intrusive bodies along the core of the Malanyu anticline and their tectonic significances. Acta Geosci. Sin. 2015, 36, 455–465. (In Chinese) [Google Scholar]
- Guo, S.F.; Tang, Z.L.; Luo, Z.H.; Zhao, W.H. Zircon SHRIMP U–Pb dating and geological significance from granite bodies in Tangzhangzi and Niuxinshan, eastern Hebei Province, China. Geol. Bull. China 2009, 28, 1458–1464. (In Chinese) [Google Scholar]
- Zhang, J.G.; Shao, Y.J.; Wang, C.; Liu, Z.F.; Xiong, Y.Q. Characteristics of gold-bearing minerals in Xiajinbao gold deposit of Hebei and their metallogenic indicating significance. J. Cent. South Univ. 2018, 49, 901–909. (In Chinese) [Google Scholar]
- Ciobanu, C.L.; Cook, N.J.; Utsunomiyas, S.; Kogagwa, M.; Green, L.; Gilbert, S.; Wade, B. Gold–telluride nanoparticles revealed in arsenic-free pyrite. Am. Miner. 2012, 97, 1515–1518. [Google Scholar] [CrossRef]
- Thomas, H.V.; Large, R.R.; Bull, S.W.; Malennikov, V.; Berry, R.F.; Fraaser, R.; Froud, S.; Moye, R. Pyrite and pyrrhotite textures and composition in sediments, laminated quartz veins, and reefs at Bendigo gold mine, Australia: Insights for ore genesis. Econ. Geol. 2011, 106, 1–31. [Google Scholar] [CrossRef]
- Fleet, M.E.; Mumin, A.H. Gold-bearing arsenian pyrite and marcasite and arsenopyrite from Carlin Trend gold deposits and laboratory synthesis. Am. Mineral. 1997, 82, 182–193. [Google Scholar] [CrossRef]
- Reich, M.; Kesler, S.E.; Utsunomiya, S.; Palenik, C.S.; Chryssoulis, S.L.; Ewing, R.C. Solubility of gold in arsenian pyrite. Geochim. Cosmochim. Acta 2005, 69, 2781–2796. [Google Scholar] [CrossRef]
- Keith, M.; Smith, D.J.; Jenkin, G.R.T.; Holwell, D.A.; Dye, M.D. A review of Te and Se systematics in hydrothermal pyrite from precious metal deposits: Insights into ore-forming processes. Ore Geol. Rev. 2017, 96, 269–282. [Google Scholar] [CrossRef]
- Koglin, N.; Frimmel, H.E.; Minter, W.E.L.; Bratz, H. Trace-element characteristics of different pyrite types in Meso-archaean to Palaeo-proterozoic placer deposits. Miner. Depos. 2010, 45, 259–280. [Google Scholar] [CrossRef]
- Morse, J.W.; Luther, G.W. Chemical influences on trace metal-sulphide interactions in anoxic sediments. Geochim. Cosmochim. Acta 1999, 63, 3373–3378. [Google Scholar] [CrossRef]
- Cook, N.J.; Ciobanu, C.L.; Mao, J.W. Textural control on gold distribution in As-free pyrite from the Dongping, Huangtuliang and Hougou gold deposits, North China Craton (Hebei Province, China). Chem. Geol. 2009, 264, 101–121. [Google Scholar] [CrossRef]
- Chouinard, A.; Paquette, J.; Williams-Jones, A.E. Crystallographic controls on trace-element incorporation in auriferous pyrite from the Pascua epithermal high-sulfidation deposit, Chile-Argentina. Can. Miner. 2005, 43, 951–963. [Google Scholar] [CrossRef]
- Simmons, S.F.; White, N.C.; John, D.A. Geological characteristics of epithermal precious and base metal deposits. Econ. Geol. 2005, 100, 485–522. [Google Scholar]
- Cook, N.J.; Ciobanu, C.L.; Meria, D.; Silcock, D.; Wade, B. Arsenopyrite-pyrite association in an orogenic gold ore: Tracing mineralization history from textures and trace elements. Econ. Geol. 2013, 108, 1273–1283. [Google Scholar] [CrossRef]
- Pokrovski, G.S.; Kara, S.; Roux, J. Stability and solubility of arsenopyrite, FeAsS, in crustal fluids. Geochim. Cosmochim. Acta 2002, 66, 2361–2378. [Google Scholar] [CrossRef]
- Bralia, A.; Sabatini, G.; Troja, F. A revaluation of the Co/Ni ratio in pyrite as geochemical tool in ore genesis problems. Miner. Depos. 1979, 14, 353–374. [Google Scholar] [CrossRef]
- Chen, Y.J.; Zhai, M.G.; Jiang, S.Y. Significant achievements and open issues in study of orogenesis and metallogenesis surrounding the North China continent. Acta Petrol. Sin. 2009, 25, 2695–2726. (In Chinese) [Google Scholar]
- Li, H.B.; Zeng, F.Z. The pyrite’s typomorphic characteristics in gold deposit. Contrib. Mineral. Petrol. 2005, 20, 199–203. (In Chinese) [Google Scholar]
- Mao, S.D.; Yang, R.S.; Qin, Y.; Guo, J.H. Characteristics of gold bearing mineral and occurrence of gold in the Yangshan gold deposit field, Gansu Province. Acta Petrol. Sin. 2009, 25, 2776–2790. (In Chinese) [Google Scholar]
- Wu, F.Y.; Yang, J.H.; Lo, C.H. The Heilongjiang group: A Jurassic accretionary complex in the Jiamusi massif at the western Pacific margin of northeastern China. Island Arc 2007, 16, 156–172. [Google Scholar] [CrossRef]
- Kojma, S.; Kemlcim, I.V.; Kametaka, M. A correlation of accretionary complexes of southern SiKhote–Alin of Russia and the Inner Zone of Southwest Japan. Geosci. J. 2000, 4, 175–185. [Google Scholar] [CrossRef]
- Tomurtogoo, O.; Windley, B.F.; Kroner, A. Zircon age and occurrence of the Adaatsag ophiolite and Muron shear zone, central Mongolia: Constraints on the evolution of the Mongol-Okhotsk Ocean, suture and orogen. J. Geol. Soc. 2005, 162, 125–134. [Google Scholar] [CrossRef]
- Zorin, Y.A. Geodynamics of the western part of the Mongolia-Okhotsk collisional belt, Trans–Baikal region (Russia) and Mongolia. Tectonophysics 1999, 306, 33–56. [Google Scholar] [CrossRef]
- Ma, Q. Triassic–Jurassic Volcanic Rocks in Western Liaoning: Implications for Lower Crustal Reworking and Lithospheric Destruction in the North Part of Eastern North China Craton; China University of Geosciences: Wuhang, China, 2013. (In Chinese) [Google Scholar]
- Johson, J.W.; Oekers, E.H.; Helgeso, H.C. A software package for calculating the standard molal thermodynamic properties of minerals, gases, aqueous species, and reaction from 1 to 5000 bars and 0 to 1000 °C. Comput. Geosci. 1992, 18, 899–947. [Google Scholar] [CrossRef]
- Seward, T.M. Thio complexes of gold and the transport of gold in hydrothermal ore solutions. Geochim. Cosmochim. Acta 1973, 37, 379–399. [Google Scholar] [CrossRef]
- Stefánsson, A.; Seward, T.M. Gold (I) complexing in aqueous sulphide solutions to 500 °C at 500 bar. Geochim. Cosmochim. Acta 2004, 68, 4121–4143. [Google Scholar] [CrossRef]
- Reed, M.H.; Spycher, N.F. Boiling, cooling and oxidation in epithermal systems: A numerical modeling approach. Rev. Econ. Geol. 1985, 2, 249–272. [Google Scholar]
- Sprcher, N.F.; Reed, M.H. Evolution of a Broadlands–type epithermal ore fluid along alternative P–T paths: Implication for the transport and deposition of base, precious and volatile metals. Econ. Geol. 1989, 84, 328–359. [Google Scholar] [CrossRef]
- Heinrich, C.A.; Eadington, P.J. Thermodynamic predictions of the hydrothermal chemistry of arsenic and their significance for the paragenetic sequence of some cassiterite-arsenopyrite-base metal sulfide deposits. Econ. Geol. 1986, 81, 511–522. [Google Scholar] [CrossRef]
- Czamanske, G.K. The FeS content of sphalerite along the chalcopyrite-pyrite-bornite sulfur fugacity buffer. Econ. Geol. 1974, 69, 1328–1334. [Google Scholar] [CrossRef]
- Einaudi, M.T.; Hedenquist, J.W.; Inan, E. Sulfidation State of Hydrothermal fluids: The Porphyry-Epithermal Transition and Beyond. In Volcanic, Geothermal and Ore-Forming Fluids: Rulers and Witnesses of Processes within the Earth; Sommons, S.F., Graham, J., Eds.; Society of Economic Geologists: Littleton, CO, USA, 2003; Volume 10, pp. 285–313. [Google Scholar]
- Huston, D.L.; Sie, S.H.; Suter, G.F.; Cooke, D.R.; Both, R.A. Trace elements in sulfide minerals from eastern australian volcanic-hosted massive sulfide deposits; part I, proton microprobe analyses of pyrite, chalcopyrite, and sphalerite, and part II, selenium levels in pyrite; comparison with δ34S values and implic. Econ. Geol. 1995, 90, 1167–1196. [Google Scholar] [CrossRef]
- Wang, L.; Zhu, Y.F. Multi-stage pyrite and hydrothermal mineral assemblage of the Hatu gold district (West Junggar, Xinjiang, NW China): Implications for metallogenic evolution. Ore Geol. Rev. 2015, 69, 243–267. [Google Scholar] [CrossRef]



| Sample | Location | Ore Types | Stage | Description |
|---|---|---|---|---|
| Z41-3 | 179.5 m, borehole ZK1103 | Quartz pyrite veins | Ι | Quartz, pyrite, electrum, native gold |
| Z49-3 | 84 m, borehole ZK34 | |||
| K7 | Open-pit mine | |||
| K1-3 | Open-pit mine | Quartz–galena–sphalerite–veins | ΙΙ | Quartz, pyrite, galena, sphalerite, electrum, native gold |
| K10-3 | ||||
| K35 | Open-pit mine | Quartz–polymetallic sulfide veins | ΙΙΙ | Quartz, pyrite, chalcopyrite, galena, sphalerite, tetrahedrite |
| Z4 | 98m, borehole ZK04 | |||
| Z168 | 164m, borehole ZK701 |
| Grain Type | Size Range (mm) | No. of Grains | Ratio (%) | Total Ratio (%) |
|---|---|---|---|---|
| Micro–fine | <0.005 | 8 | 2.74 | 33.65 |
| 0.005–0.01 | 90 | 30.82 | ||
| Fine | 0.01–0.037 | 145 | 49.65 | 49.65 |
| Medium | 0.037–0.053 | 34 | 11.64 | 13.69 |
| 0.053–0.074 | 6 | 2.05 | ||
| Coarse | 0.074–0.1 | 3 | 1.03 | 2.74 |
| 0.1–0.3 | 5 | 1.71 | ||
| Huge | >0.3 | 1 | 0.34 | 0.34 |
| Style | Position | Ratio (%) | Total Ratio |
|---|---|---|---|
| Fissure gold | Pyrite fissure | 7.88 | 9.25 |
| Sphalerite fissure | 1.37 | ||
| Intra-granule gold | Pyrite–quartz | 3.08 | 33.24 |
| Pyrite–sphalerite | 3.8 | ||
| Pyrite–galena | 0.34 | ||
| Pyrite–galena–quartz | 0.34 | ||
| Pyrite–pyrite | 0.68 | ||
| Sphalerite–sphalerite | 3.08 | ||
| Quartz–quartz | 0.68 | ||
| Sphalerite–quartz | 20.21 | ||
| Pyrite–sphalerite–quartz | 1.03 | ||
| Inclusion gold | Sphalerite | 29.45 | 31.86 |
| Pyrite | 1.05 | ||
| Galena | 0.68 | ||
| Quartz | 0.68 | ||
| Independent gold | Quartz | 25.65 | 25.65 |
| Sample No. | Stage | S | Fe | Co | Ni | Cu | Zn | As | Ag | Au | Pb | Bi | Te |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| wt % | wt % | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ppm | ||
| Z51-4 | Ι | 54.19 | 44.73 | 0.82 | 0.1 | 0.43 | 0.31 | 146.87 | 0.05 | 0.03 | 0.42 | 0.02 | 0.17 |
| 54.76 | 44.63 | 0.95 | 0.46 | 9.13 | 1.03 | 241.46 | 180.4 | 0.56 | 2574.78 | 360.81 | 0.48 | ||
| 55.48 | 44.29 | 1.86 | 0.14 | 2.04 | 0.32 | 2.15 | 0.08 | - | 0.48 | 0.38 | 0.89 | ||
| Z49 | 54.03 | 43.86 | 0.97 | 0.29 | 4.38 | 3.24 | 329.37 | 0.39 | 3.61 | 4.65 | 0.01 | 0.37 | |
| 56.76 | 43.02 | 8.34 | 0.62 | 0.33 | 0.95 | 562.34 | 35.26 | 4.82 | 0.04 | 0.06 | 0.61 | ||
| 55.06 | 43.94 | 2.97 | 0.57 | 7.03 | 0.27 | 84.31 | 0.01 | - | 0.86 | - | 0.27 | ||
| 54.97 | 44.74 | 0.61 | 0.24 | 3.22 | 2.16 | 127.69 | 0.89 | 2.33 | 0.48 | 0.01 | 0.64 | ||
| K7 | 54.41 | 44.35 | 1.25 | 0.27 | 4.26 | 0.35 | 394.36 | 0.96 | 0.95 | 1.36 | 0.01 | 0.35 | |
| 53.76 | 44.25 | 1.28 | 0.67 | 2.39 | 0.51 | 256.32 | 1.69 | 7.39 | 0.62 | - | 0.94 | ||
| 54.29 | 44.01 | 1.39 | 0.39 | 5.36 | 4.63 | 449.34 | 0.04 | 4.33 | 0.98 | 0.11 | 0.86 | ||
| Z12 | ΙΙ | 53.89 | 45.82 | 19.29 | 8.55 | 1.66 | 1.47 | 715.98 | 1.64 | 9.35 | 154.57 | 0.05 | 0.26 |
| 54.53 | 44.52 | 0.86 | 0.52 | 0.82 | 1.7 | 275.62 | 33.98 | 2.61 | 0.44 | - | 4.14 | ||
| 54.92 | 44.92 | 0.33 | 1.03 | 3.55 | 2.23 | 82.4 | 3.14 | 6.65 | 305.22 | - | 0.14 | ||
| 55.71 | 43.86 | 2.3 | 0.61 | 0.79 | 1.51 | 330.17 | 0.08 | 0.03 | 0.81 | 0.01 | 7.56 | ||
| Z29 | 55.83 | 43.58 | 6.05 | 8.83 | 1.62 | 0.56 | 716.21 | 69.35 | 3.99 | 0.39 | 0.38 | 0.46 | |
| 54.34 | 43.86 | 4.69 | 0.92 | 2.67 | 0.34 | 365.18 | 2.67 | 10.08 | 0.28 | 0.01 | 0.05 | ||
| 54.62 | 44.71 | 0.67 | 0.36 | 0.84 | 2.69 | 149.27 | 0.25 | 0.66 | 1.35 | - | 4.14 | ||
| 54.13 | 44.74 | 0.53 | 1.64 | 0.37 | 0.22 | 485.32 | 4.63 | 1.04 | 0.64 | 0.57 | 0.08 | ||
| K35 | ΙΙΙ | 54.83 | 44.87 | 0.07 | 0.47 | 40.57 | 1204.53 | 594.97 | 6.52 | 0.15 | 31.61 | 0.01 | 7.71 |
| 55.55 | 44.59 | 0.02 | 0.15 | 170.06 | 4011.92 | 0.12 | 22.21 | 0.35 | 11.02 | 0.01 | 1.24 | ||
| 54.96 | 44.17 | 0.44 | 0.71 | 0.93 | 1.22 | 136.77 | - | - | 0.54 | 0.01 | 4.26 | ||
| Z4 | 55.93 | 44.89 | 0.16 | 0.67 | 4.32 | 0.47 | 23.69 | 4.68 | 0.09 | 8.36 | - | 5.32 | |
| 55.94 | 44.12 | 0.04 | 0.3 | 6.48 | 9.67 | 4.28 | 9.34 | 0.18 | 4.21 | - | 0.21 | ||
| 55.31 | 43.84 | 0.19 | 0.86 | 0.37 | 0.27 | 76.37 | 0.62 | - | 0.69 | 0.01 | 3.23 | ||
| 55.34 | 43.69 | 0.95 | 0.62 | 1.68 | 2.38 | 29.17 | 1.2 | 0.02 | 1.08 | 0.03 | 0.49 | ||
| Z168 | 55.39 | 44.18 | 0.14 | 0.88 | 3.52 | 10.64 | 4.31 | 5.37 | 0.39 | 3.24 | - | 6.89 | |
| 56.11 | 44.68 | 0.01 | 0.23 | 2.13 | 5.96 | 1.65 | 1.87 | - | 7.19 | 1.39 | 0.97 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Shao, Y.; Huang, K.; Zhou, H.; Zhang, J.; Liu, Z.; Liu, Q. Ore-Forming Processes at the Xiajinbao Gold Deposit in Eastern Hebei Province: Constraints from EPMA and LA-ICPMS Analysis. Minerals 2018, 8, 388. https://doi.org/10.3390/min8090388
Wang C, Shao Y, Huang K, Zhou H, Zhang J, Liu Z, Liu Q. Ore-Forming Processes at the Xiajinbao Gold Deposit in Eastern Hebei Province: Constraints from EPMA and LA-ICPMS Analysis. Minerals. 2018; 8(9):388. https://doi.org/10.3390/min8090388
Chicago/Turabian StyleWang, Cheng, Yongjun Shao, Kuanxin Huang, Haodi Zhou, Jianguo Zhang, Zhongfa Liu, and Qingquan Liu. 2018. "Ore-Forming Processes at the Xiajinbao Gold Deposit in Eastern Hebei Province: Constraints from EPMA and LA-ICPMS Analysis" Minerals 8, no. 9: 388. https://doi.org/10.3390/min8090388
APA StyleWang, C., Shao, Y., Huang, K., Zhou, H., Zhang, J., Liu, Z., & Liu, Q. (2018). Ore-Forming Processes at the Xiajinbao Gold Deposit in Eastern Hebei Province: Constraints from EPMA and LA-ICPMS Analysis. Minerals, 8(9), 388. https://doi.org/10.3390/min8090388






