Tsygankoite, Mn8Tl8Hg2(Sb21Pb2Tl)Σ24S48, a New Sulfosalt from the Vorontsovskoe Gold Deposit, Northern Urals, Russia
Abstract
:1. Introduction
2. Occurrence
3. Physical and Optical Properties
4. Chemical Composition and Chemical Properties
5. X-Ray Crystallography
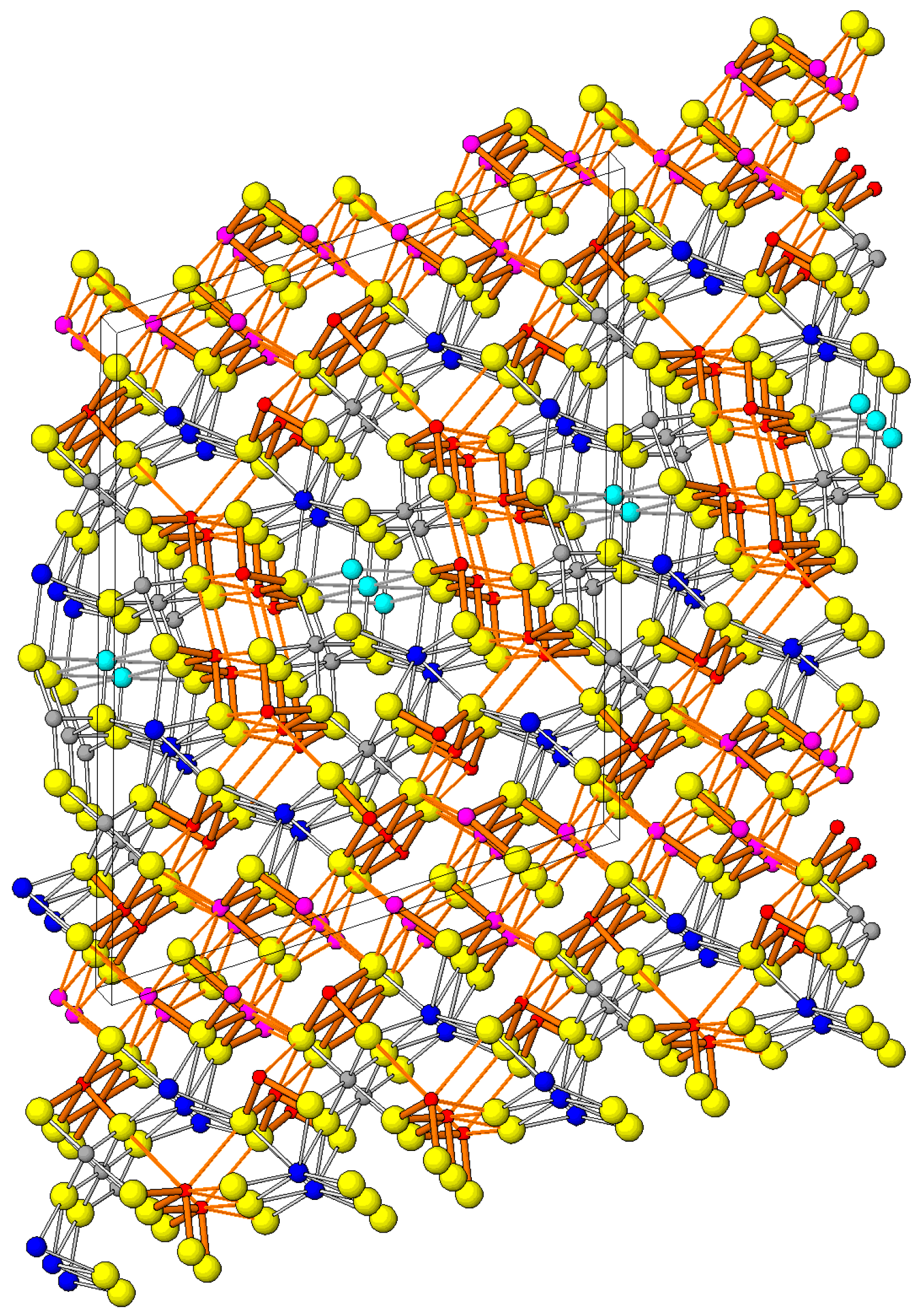
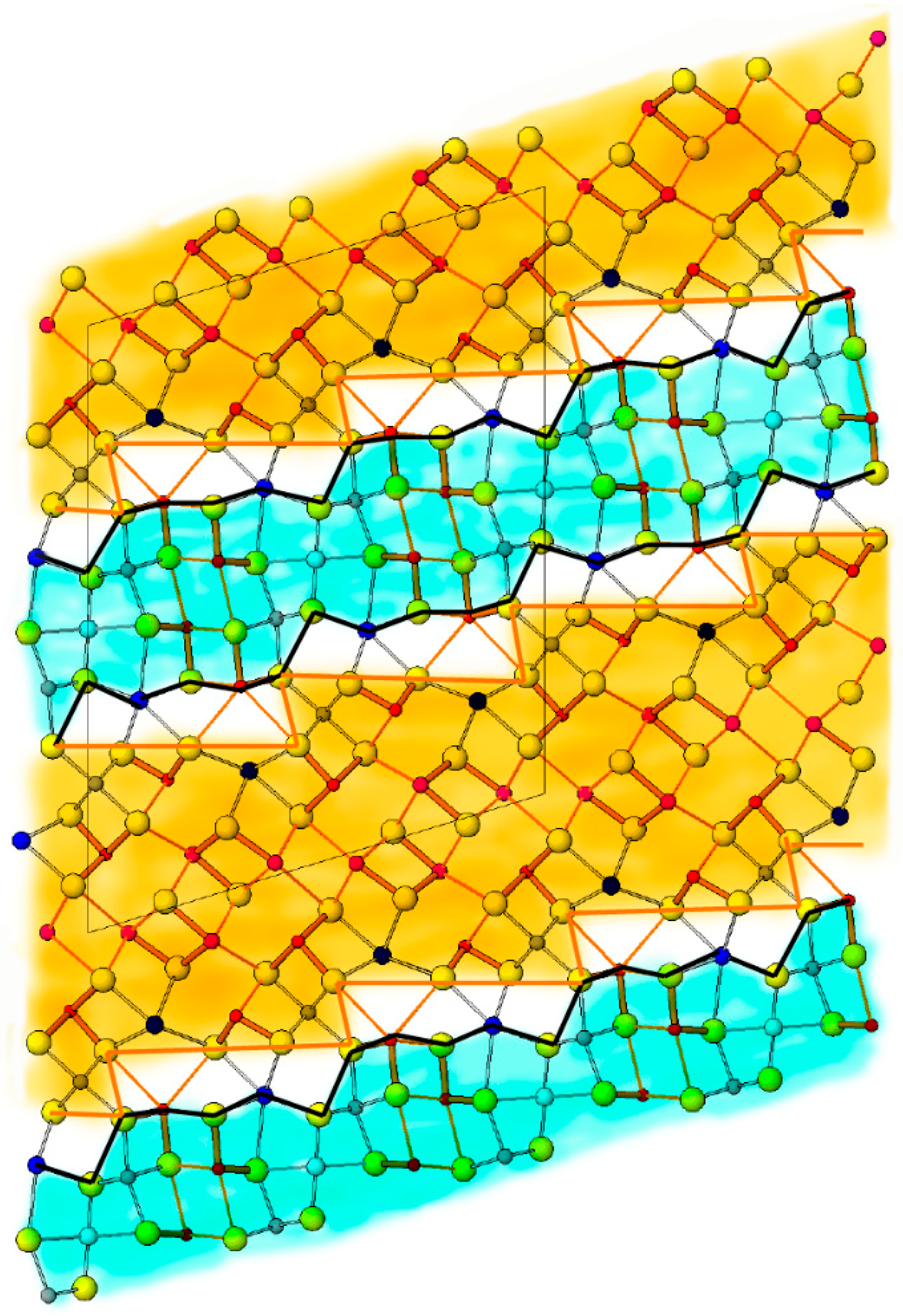
6. Description of Crystal Structure and Discussion
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Tsyganko, M.V. Kamennyi Uzor Zemli Vagranskoi. Mineraly Severouralskogo Rayona (Stone Design of Vagranskaya Earth. Minerals of Northern Urals); “Kvist”: Yekaterinburg, Russia, 2015; 120p. (In Russian) [Google Scholar]
- Sazonov, V.N.; Murzin, V.V.; Grigor’ev, N.A. Vorontsovsk gold deposit: An example of carlin-type mineralization in the Urals, Russia. Geol. Ore Depos. 1998, 40, 139–151. [Google Scholar]
- Murzin, V.V.; Naumov, E.A.; Azovskova, O.B.; Varlamov, D.A.; Rovnushkin, M.Y.; Pirajno, F. The Vorontsovskoe Au-Hg-As ore deposit (Northern Urals, Russia): Geological setting, ore mineralogy, geochemistry, geochronology and genetic model. Ore Geol. Rev. 2017, 85, 271–298. [Google Scholar] [CrossRef]
- Minina, O.V. The Auerbahovskiy complex ore-magmatic system of the Middle Ural. Nativ. Geol. 1994, 7, 17–23. (In Russian) [Google Scholar]
- Stepanov, S.Y.; Sharpenok, L.N.; Antonov, A.V. Fluid-exposive breccias of the Vorontsovskoe gold deposit (The North Urals). Zapiski RMO 2017, 1, 29–43. (In Russian) [Google Scholar]
- Kasatkin, A.V.; Nestola, F.; Agakhanov, A.A.; Škoda, R.; Karpenko, V.Y.; Tsyganko, M.V.; Plášil, J. Vorontsovite, (Hg5Cu)Σ6TlAs4S12, and ferrovorontsovite, (Fe5Cu)Σ6TlAs4S12: The Tl- and Tl-Fe-analogues of Galkhaite from the Vorontsovskoe gold deposit, Northern Urals, Russia. Minerals 2018, 8, 185. [Google Scholar] [CrossRef]
- Kraus, W.; Nolze, G. POWDER CELL—A program for the representation and manipulation of crystal structures and calculation of the resulting X-ray powder patterns. J. Appl. Cryst. 1996, 29, 301–303. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Petříček, V.; Dušek, M.; Palatinus, L. Crystallographic computing system Jana2006: General features. Z. Kristallogr. 2014, 229, 345–352. [Google Scholar] [CrossRef]
- Makovicky, E. The building principles and classification of sulphosalts based on the SnS archetype. Fortschr. Mineral. 1985, 63, 45–89. [Google Scholar]
- Makovicky, E. Rod-based sulphosalt structures derived from the SnS and PbS archetypes. Eur. J. Mineral. 1993, 5, 545–591. [Google Scholar] [CrossRef]
- Orlandi, P.; Meerschaut, A.; Moëlo, Y.; Palvadeau, P.; Léone, P. Lead-antimony sulfosalts from Tuscany (Italy). VIII. Rouxelite, Cu2HgPb22Sb28S64(O,S)2, a new sulfosalt from Buca della Vena mine, Apuan Alps: definition and crystal structure. Can. Mineral. 2005, 43, 919–933. [Google Scholar] [CrossRef]
- Auvray, P.; Genet, F. Affinement de la structure cristalline du cinabre, alpha HgS. Bull. Soc. Fr. Min. Cristallogr. 1973, 96, 218–219. [Google Scholar]
- Biagioni, C.; Bindi, L. Ordered distribution of Cu and Ag in the crystal structure of balkanite, Cu9Ag5HgS8. Eur. J. Mineral. 2017, 29, 279–285. [Google Scholar] [CrossRef]
- Bente, K.; Edenharter, A. Roentgenographische Strukturanalyse von MnSb2S4 und Strukturverfeinerung von Berthierit, FeSb2S4. Z. Kristallogr. 1989, 186, 31–33. [Google Scholar]
- Pfitzner, A.; Kurowski, D. A new modification of MnSb2S4 crystallizing in the HgBi2S4 structure type. Z. Kristallogr. 2000, 215, 373–376. [Google Scholar] [CrossRef]


| λ (nm) | R1 (%) | R2 (%) |
|---|---|---|
| 400 | 33.85 | 30.53 |
| 420 | 33.64 | 30.94 |
| 440 | 32.39 | 30.72 |
| 460 | 32.88 | 31.56 |
| 470 | 33.16 | 31.67 |
| 480 | 33.44 | 31.78 |
| 500 | 33.22 | 31.71 |
| 520 | 32.84 | 31.49 |
| 540 | 32.53 | 31.26 |
| 546 | 32.41 | 31.11 |
| 560 | 32.26 | 31.00 |
| 580 | 31.89 | 30.54 |
| 589 | 31.58 | 30.18 |
| 600 | 31.51 | 30.14 |
| 620 | 30.95 | 29.83 |
| 640 | 30.27 | 29.10 |
| 650 | 29.83 | 28.73 |
| 660 | 29.37 | 28.39 |
| 680 | 28.29 | 27.58 |
| 700 | 27.41 | 26.74 |
| Constituent | Mean | Range | SD |
|---|---|---|---|
| Mn | 6.29 | 6.15–6.40 | 0.11 |
| Hg | 5.42 | 5.31–5.58 | 0.09 |
| Tl | 26.05 | 25.52–26.53 | 0.35 |
| Pb | 5.84 | 5.01–6.34 | 0.59 |
| As | 3.39 | 3.15–3.72 | 0.21 |
| Sb | 30.89 | 30.32–31.39 | 0.34 |
| S | 21.87 | 21.40–22.40 | 0.30 |
| Total | 99.75 |
| Irel. (%) | dcalc. (Å) | h | k | l | Irel. (%) | dcalc. (Å) | h | k | l |
|---|---|---|---|---|---|---|---|---|---|
| 11 | 12.98 | 0 | 0 | 2 | 99 | 2.786 | –5 | 1 | 4 |
| 29 | 9.483 | –2 | 0 | 2 | 22 | 2.731 | 5 | 1 | 1 |
| 6 | 7.822 | –2 | 0 | 3 | 12 | 2.711 | –2 | 0 | 10 |
| 27 | 5.310 | –4 | 0 | 2 | 7 | 2.688 | –3 | 1 | 7 |
| 6 | 5.109 | 4 | 0 | 0 | 28 | 2.604 | –5 | 1 | 6 |
| 11 | 5.097 | –4 | 0 | 3 | 7 | 2.586 | 2 | 0 | 9 |
| 9 | 4.480 | –2 | 0 | 6 | s6 | 2.582 | 1 | 1 | 7 |
| 16 | 4.342 | 4 | 0 | 2 | 8 | 2.534 | –1 | 1 | 8 |
| 14 | 4.326 | 0 | 0 | 6 | 5 | 2.530 | 6 | 0 | 5 |
| 23 | 3.927 | 4 | 0 | 3 | 8 | 2.525 | 5 | 1 | 3 |
| 25 | 3.911 | –4 | 0 | 6 | 7 | 2.490 | –5 | 1 | 7 |
| 38 | 3.866 | –2 | 0 | 7 | 12 | 2.473 | 8 | 0 | 1 |
| 14 | 3.722 | 1 | 1 | 1 | 5 | 2.435 | –6 | 0 | 10 |
| 20 | 3.708 | 0 | 0 | 7 | 17 | 2.368 | –7 | 1 | 1 |
| 100 | 3.587 | 1 | 1 | 2 | 23 | 2.358 | –1 | 1 | 9 |
| 44 | 3.559 | –6 | 0 | 2 | 13 | 2.328 | 7 | 1 | 0 |
| 8 | 3.543 | 4 | 0 | 4 | 5 | 2.275 | –6 | 0 | 11 |
| 25 | 3.540 | –6 | 0 | 3 | 13 | 2.268 | –8 | 0 | 9 |
| 13 | 3.511 | –6 | 0 | 1 | 7 | 2.238 | 1 | 1 | 9 |
| 12 | 3.459 | –6 | 0 | 4 | 26 | 2.198 | –1 | 1 | 10 |
| 68 | 3.391 | –2 | 0 | 8 | 13 | 2.182 | 3 | 1 | 8 |
| 70 | 3.353 | –1 | 1 | 4 | 17 | 2.163 | –8 | 0 | 10 |
| 6 | 3.327 | –6 | 0 | 5 | 12 | 2.116 | –10 | 0 | 2 |
| 88 | 3.204 | 4 | 0 | 5 | 11 | 2.085 | –10 | 0 | 1 |
| 10 | 3.161 | –6 | 0 | 6 | 9 | 2.061 | –7 | 1 | 9 |
| 40 | 3.144 | –1 | 1 | 5 | 9 | 2.054 | –1 | 1 | 11 |
| 7 | 3.142 | 3 | 1 | 2 | 13 | 2.049 | 5 | 1 | 7 |
| 6 | 3.024 | –3 | 1 | 5 | 29 | 2.047 | 3 | 1 | 9 |
| 28 | 3.015 | –2 | 0 | 9 | 9 | 2.021 | –9 | 1 | 3 |
| 45 | 2.988 | 3 | 1 | 3 | 9 | 2.014 | –10 | 0 | 8 |
| 7 | 2.978 | –6 | 0 | 7 | 8 | 1.9608 | 7 | 1 | 5 |
| 41 | 2.910 | 4 | 0 | 6 | 6 | 1.9389 | 5 | 1 | 8 |
| 13 | 2.895 | 6 | 0 | 3 | 53 | 1.9290 | 0 | 2 | 0 |
| 8 | 2.884 | 0 | 0 | 9 | 5 | 1.9241 | 3 | 1 | 10 |
| 19 | 2.863 | –5 | 1 | 2 | 37 | 1.8915 | –7 | 1 | 11 |
| 64 | 2.858 | –3 | 1 | 6 | 7 | 1.8705 | 10 | 0 | 3 |
| 41 | 2.851 | –5 | 1 | 1 | 32 | 1.8670 | 9 | 1 | 2 |
| 72 | 2.841 | –5 | 1 | 3 | 8 | 1.8649 | 8 | 0 | 7 |
| 6 | 2.821 | 3 | 1 | 4 | 15 | 1.8602 | 2 | 0 | 13 |
| 60 | 2.805 | 5 | 1 | 0 | 15 | 1.8358 | –3 | 1 | 13 |
| Formula | Mn8Tl8.00Hg2.00(Sb18.74·As2.18Tl1.67Pb1.44)Σ24.03S48 |
|---|---|
| Crystal system | monoclinic |
| Space group | C2/m |
| Unit-cell parameters: a, b, c [Å] | 21.362(4), 3.8579(10), 27.135(4) |
| β [°] | 106.944(14) |
| Unit-cell volume [Å3] | 2139.19(17) |
| Z | 1 |
| Calculated density [g/cm3] | 5.450 (from empirical formula) |
| Crystal size [mm] | 0.032 × 0.017 × 0.007 |
| Diffractometer | Rigaku SuperNova with Atlas S2 CCD |
| Temperature [K] | 295 |
| Radiation, wavelength [Å] | MoKα, 0.71073 (50 kV, 0.8 mA) |
| θ range for data collection [°] | 3.50–27.14 |
| Limiting Miller indices | h = −26 → 26, k = −4 → 4, l = −34 → 17 |
| Axis, frame width (°), time per frame (s) | ω, 1.0, 500 |
| Total reflections collected | 4686 |
| Unique reflections | 2403 |
| Unique observed reflections, criterion | 957, [I > 3σ(I)] |
| Absorption coefficient [mm−1], type | 33.37; empirical |
| Tmin/Tmax | 0.782/1 |
| Data completeness to θmax (%), Rint | 89.00, 0.105 |
| Structure refinement | Full-matrix least-squares on F2 |
| No. of param., restraints, constraints | 143, 0, 24 |
| R, wR (obs) | 0.0607, 0.0943 |
| R, wR (all) | 0.1757, 0.1452 |
| GOF obs/all | 1.14, 1.04 |
| Weighting scheme, weights | σ, w = 1/(σ2(I) + 0.0001800964I2) |
| Largest diffraction peak and hole (e−/Å3) | 7.32 (0.48 Å to Sb6), −7.76 |
| Atom | x/a | y/b | z/c | Ueq | U11 | U22 | U33 | U12 | U13 | U23 |
|---|---|---|---|---|---|---|---|---|---|---|
| Hg1 | 0.5 | 1 | 0.5 | 0.0243(9) | 0.0275(16) | 0.0240(17) | 0.0240(16) | 0 | 0.0111(13) | 0 |
| Tl1 | 0.61255(10) | −0.5 | 0.64328(7) | 0.0360(8) | 0.0448(13) | 0.0319(13) | 0.0361(12) | 0 | 0.0194(11) | 0 |
| Tl2 | 0.35428(9) | −0.5 | 0.81477(8) | 0.0362(8) | 0.0324(12) | 0.0313(13) | 0.0454(14) | 0 | 0.0122(10) | 0 |
| Sb1 | 0.28499(15) | 0 | 0.45391(11) | 0.0259(11) | 0.0308(19) | 0.0223(19) | 0.0231(16) | 0 | 0.0058(15) | 0 |
| Sb2 | 0.33573(19) | 0 | 0.67105(12) | 0.0415(14) | 0.077(3) | 0.019(2) | 0.0299(19) | 0 | 0.018(2) | 0 |
| Sb3/Pb3 | 0.4110(2) | −1 | 0.97915(15) | 0.081(2) | 0.121(4) | 0.049(3) | 0.112(4) | 0 | 0.094(3) | 0 |
| Sb4/As3 | 0.53848(18) | 0 | 0.87986(13) | 0.0442(17) | 0.044(3) | 0.057(3) | 0.025(2) | 0 | 0.001(2) | 0 |
| Sb5/As5 | 0.67441(18) | 0 | 0.78772(15) | 0.0427(17) | 0.026(2) | 0.050(3) | 0.046(3) | 0 | 0.000(2) | 0 |
| Sb6/Tl6 | 0.2295(2) | −1.5 | 0.92414(15) | 0.091(2) | 0.173(5) | 0.034(2) | 0.112(3) | 0 | 0.116(3) | 0 |
| Mn1 | 0.4134(3) | 0.5 | 0.5777(2) | 0.023(2) | 0.024(4) | 0.022(4) | 0.022(4) | 0 | 0.006(3) | 0 |
| Mn2 | 0.5162(3) | 0.5 | 0.7581(2) | 0.022(2) | 0.023(4) | 0.026(4) | 0.017(3) | 0 | 0.006(3) | 0 |
| S1 | 0.4924(5) | 1 | 0.5836(4) | 0.021(4) | 0.031(6) | 0.023(7) | 0.015(6) | 0 | 0.015(5) | 0 |
| S2 | 0.5745(5) | 0 | 0.7221(4) | 0.020(4) | 0.025(6) | 0.020(6) | 0.015(5) | 0 | 0.007(5) | 0 |
| S3 | 0.4225(5) | 0.5 | 0.6753(4) | 0.024(4) | 0.018(6) | 0.029(7) | 0.021(6) | 0 | −0.002(5) | 0 |
| S4 | 0.3232(5) | 0 | 0.5780(4) | 0.020(4) | 0.033(7) | 0.018(6) | 0.010(5) | 0 | 0.007(5) | 0 |
| S5 | 0.3698(5) | 0.5 | 0.4759(4) | 0.023(4) | 0.019(6) | 0.022(7) | 0.033(7) | 0 | 0.012(6) | 0 |
| S6 | 0.2750(5) | 0 | 0.3625(4) | 0.024(4) | 0.033(7) | 0.021(7) | 0.018(6) | 0 | 0.005(6) | 0 |
| S7 | 0.4610(6) | 0 | 0.7970(4) | 0.026(4) | 0.036(7) | 0.020(7) | 0.021(6) | 0 | 0.005(6) | 0 |
| S8 | 0.7220(6) | −0.5 | 0.7473(4) | 0.037(5) | 0.025(7) | 0.053(9) | 0.031(7) | 0 | 0.004(6) | 0 |
| S9 | 0.6079(7) | 0.5 | 0.8437(4) | 0.044(5) | 0.049(9) | 0.053(10) | 0.019(6) | 0 | −0.006(7) | 0 |
| S10 | 0.3047(7) | −1 | 0.8969(5) | 0.047(6) | 0.045(9) | 0.058(11) | 0.033(7) | 0 | 0.001(7) | 0 |
| S11 | 0.3311(8) | –1.5 | 1.0129(5) | 0.062(6) | 0.096(13) | 0.029(9) | 0.035(8) | 0 | −0.021(8) | 0 |
| S12 | 0.4678(9) | −0.5 | 0.9152(6) | 0.080(8) | 0.104(14) | 0.016(8) | 0.079(11) | 0 | −0.037(11) | 0 |
| Hg1–S1 | 2.321(11) (2×) | Sb3–S11 | 2.896(14) (2×) |
| Tl1–S1 | 3.242(8) (2×) | Sb4–S7 | 2.373(10) |
| Tl1–S2 | 3.158(10) (2×) | Sb4–S9 | 2.781(11) (2×) |
| Tl1–S5 | 3.363(12) | Sb4–S12 | 2.786(15) (2×) |
| Tl1–S6 | 3.119(9) (2×) | Sb5–S2 | 2.347(10) (2×) |
| Tl1–S8 | 3.096(10) (2×) | Sb5–S8 | 2.570(9) (2×) |
| Tl2–S7 | 3.129(11) (2×) | Sb6–S10 | 2.745(12) (2×) |
| Tl2–S8 | 3.470(9) (2×) | Sb6–S11 | 2.733(13) |
| Tl2–S10 | 3.344(13) (2×) | Mn1–S1 | 2.573(8) (2×) |
| Tl2–S12 | 3.075(15) | Mn1–S3 | 2.598(12) |
| Sb1–S4 | 2.940(8) (2×) | Mn1–S4 | 2.728(9) (2×) |
| Sb1–S5 | 2.594(7) (2×) | Mn1–S5 | 2.645(11) |
| Sb1–S6 | 2.427(11) | Mn2–S2 | 2.633(10) (2×) |
| Sb2–S3 | 2.665(8) (2×) | Mn2–S3 | 2.536(10) |
| Sb2–S4 | 2.459(11) | Mn2–S7 | 2.636(10) (2×) |
| Sb2–S6 | 2.981(8) (2×) | Mn2–S9 | 2.565(12) |
| Sb3–S10 | 2.679(13) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kasatkin, A.V.; Makovicky, E.; Plášil, J.; Škoda, R.; Agakhanov, A.A.; Karpenko, V.Y.; Nestola, F. Tsygankoite, Mn8Tl8Hg2(Sb21Pb2Tl)Σ24S48, a New Sulfosalt from the Vorontsovskoe Gold Deposit, Northern Urals, Russia. Minerals 2018, 8, 218. https://doi.org/10.3390/min8050218
Kasatkin AV, Makovicky E, Plášil J, Škoda R, Agakhanov AA, Karpenko VY, Nestola F. Tsygankoite, Mn8Tl8Hg2(Sb21Pb2Tl)Σ24S48, a New Sulfosalt from the Vorontsovskoe Gold Deposit, Northern Urals, Russia. Minerals. 2018; 8(5):218. https://doi.org/10.3390/min8050218
Chicago/Turabian StyleKasatkin, Anatoly V., Emil Makovicky, Jakub Plášil, Radek Škoda, Atali A. Agakhanov, Vladimir Y. Karpenko, and Fabrizio Nestola. 2018. "Tsygankoite, Mn8Tl8Hg2(Sb21Pb2Tl)Σ24S48, a New Sulfosalt from the Vorontsovskoe Gold Deposit, Northern Urals, Russia" Minerals 8, no. 5: 218. https://doi.org/10.3390/min8050218
APA StyleKasatkin, A. V., Makovicky, E., Plášil, J., Škoda, R., Agakhanov, A. A., Karpenko, V. Y., & Nestola, F. (2018). Tsygankoite, Mn8Tl8Hg2(Sb21Pb2Tl)Σ24S48, a New Sulfosalt from the Vorontsovskoe Gold Deposit, Northern Urals, Russia. Minerals, 8(5), 218. https://doi.org/10.3390/min8050218





