Metal Ion Release in Bastnaesite Flotation System and Implications for Flotation
Abstract
:1. Introduction
2. Materials and Methods
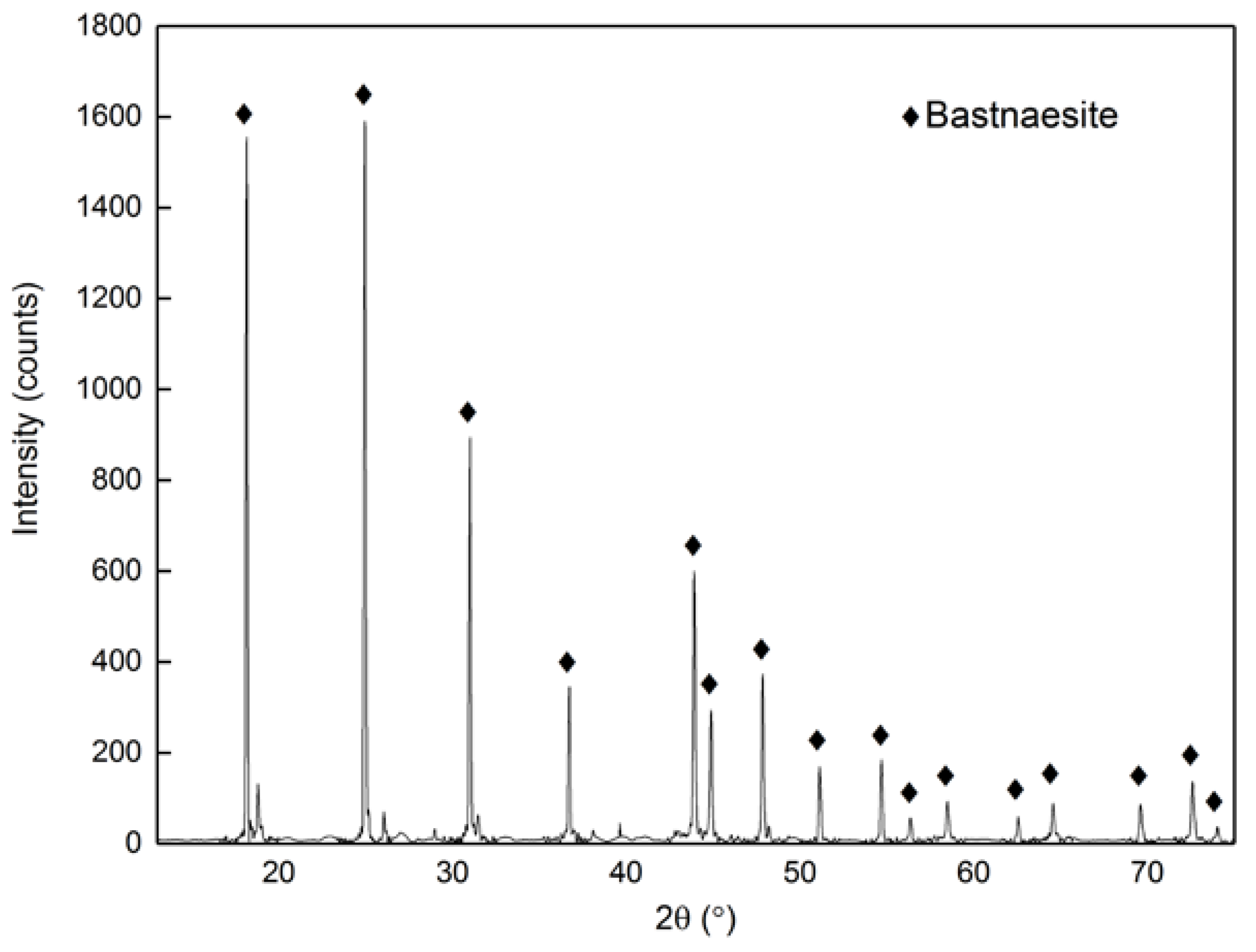
2.1. Materials
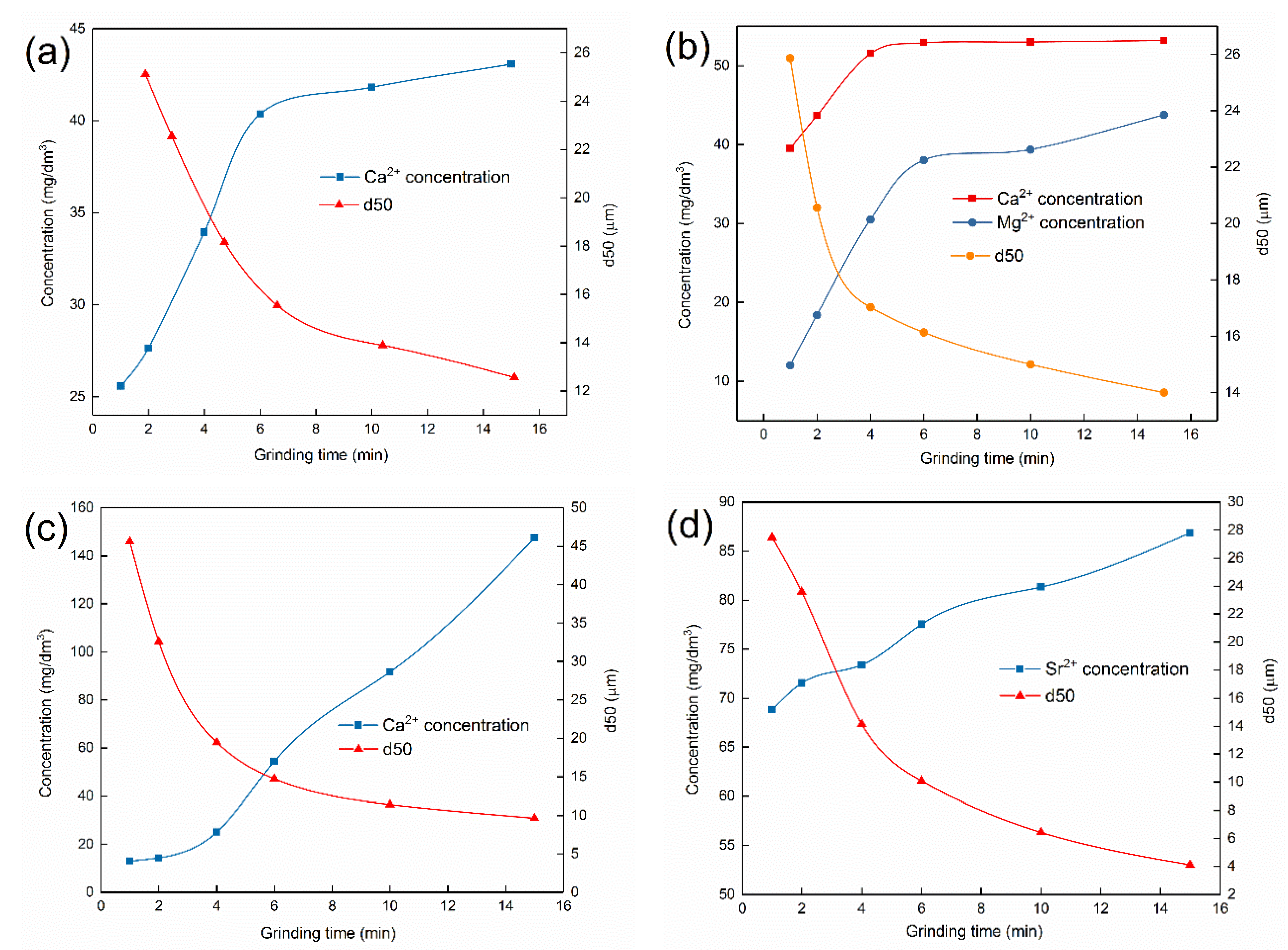
2.2. Metal Ions Released from Minerals
2.3. Flotation Experiments
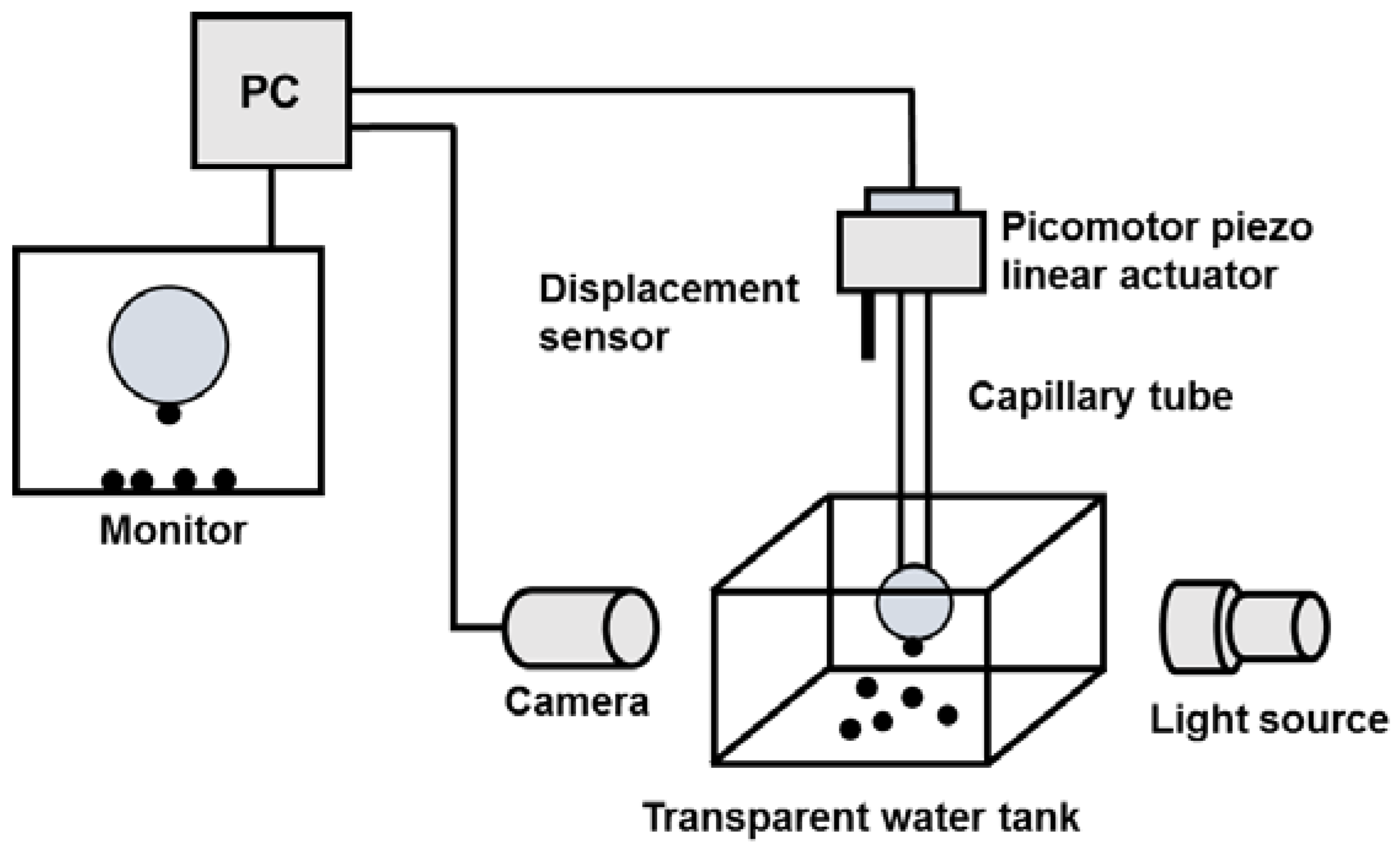
2.4. Induction Time Measurements
2.5. Adsorption Experiments
2.6. Zeta-Potential Determination
3. Results and Discussion
3.1. Determination of Metal Ions in Practical Flotation Pulp
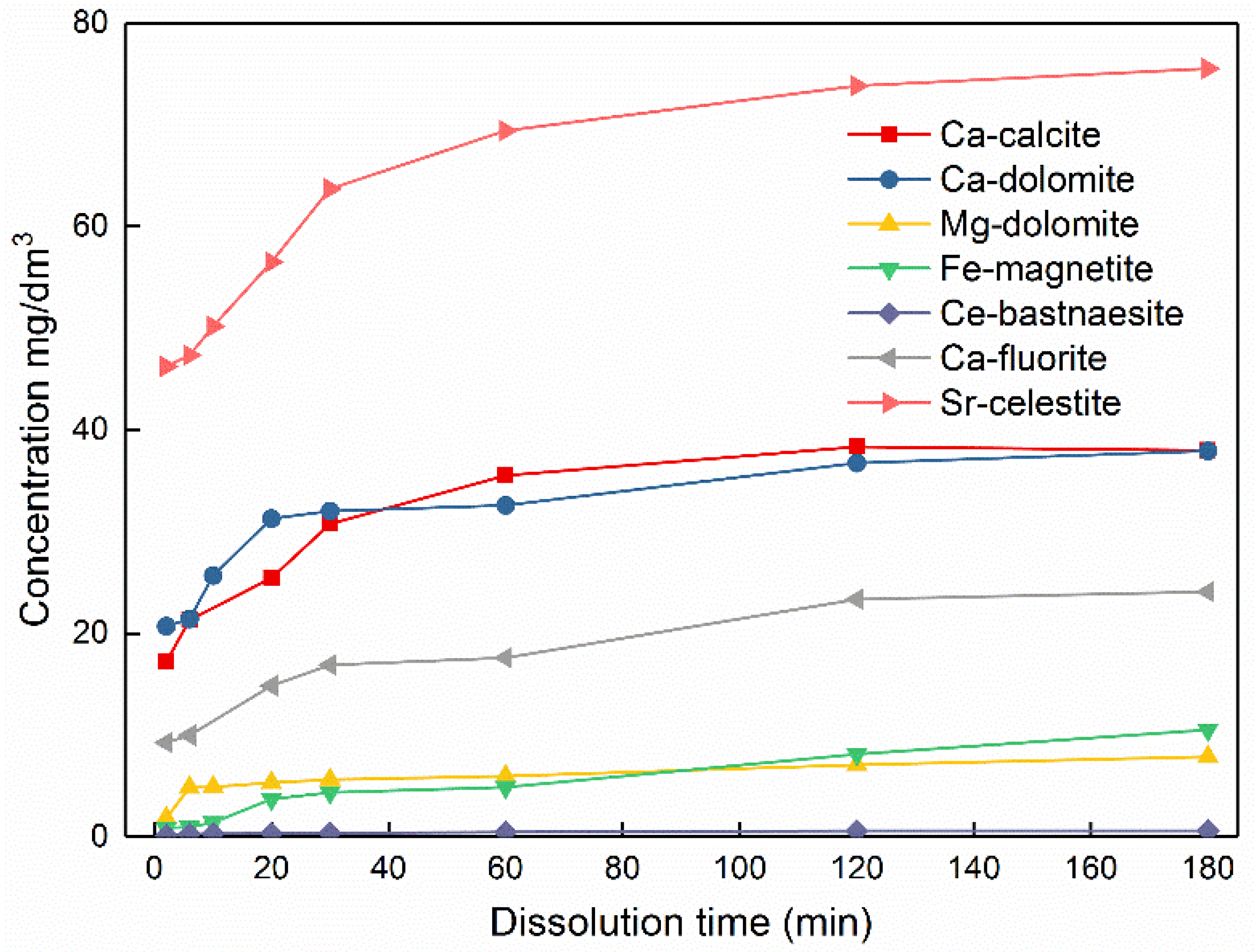
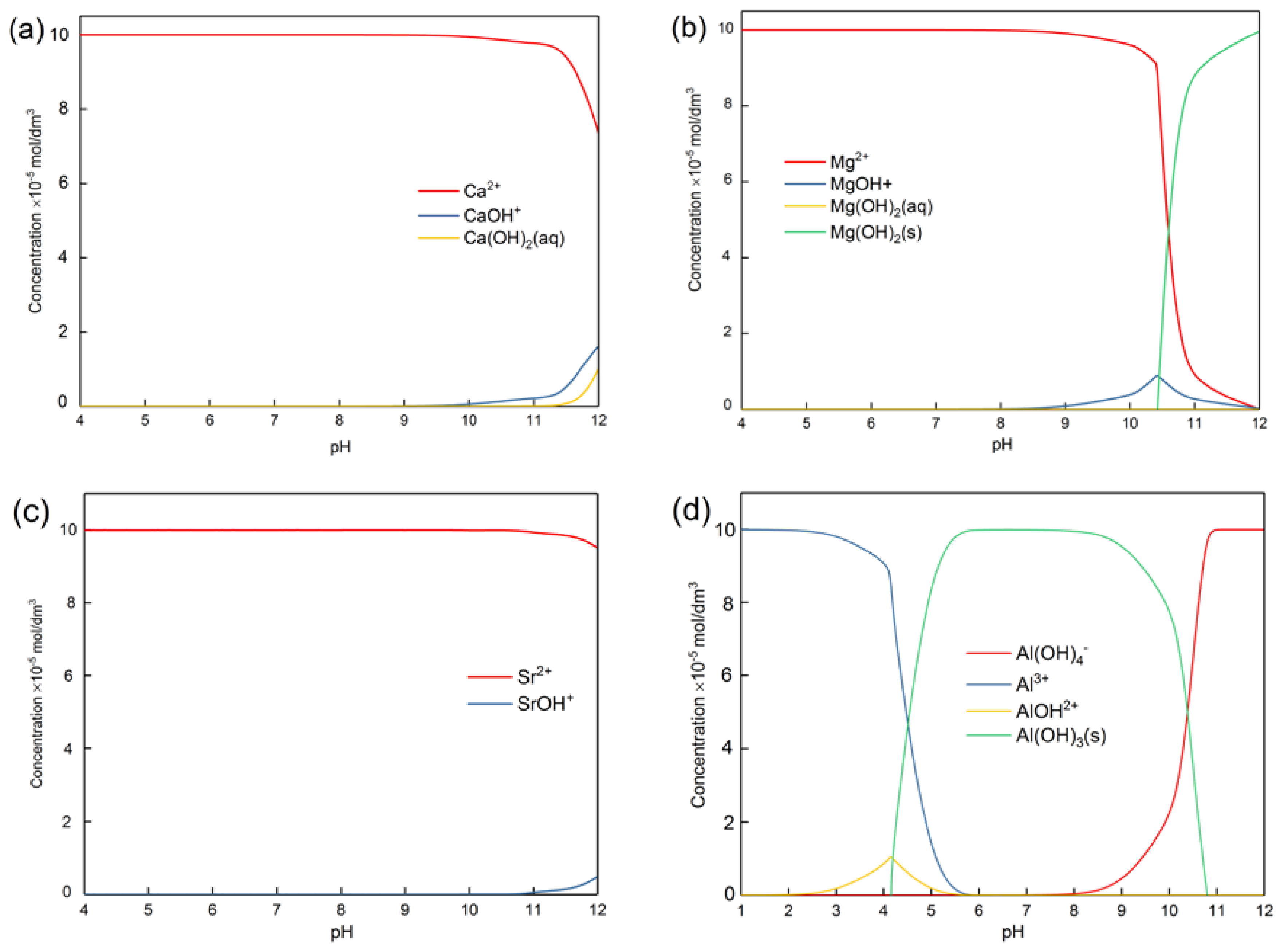
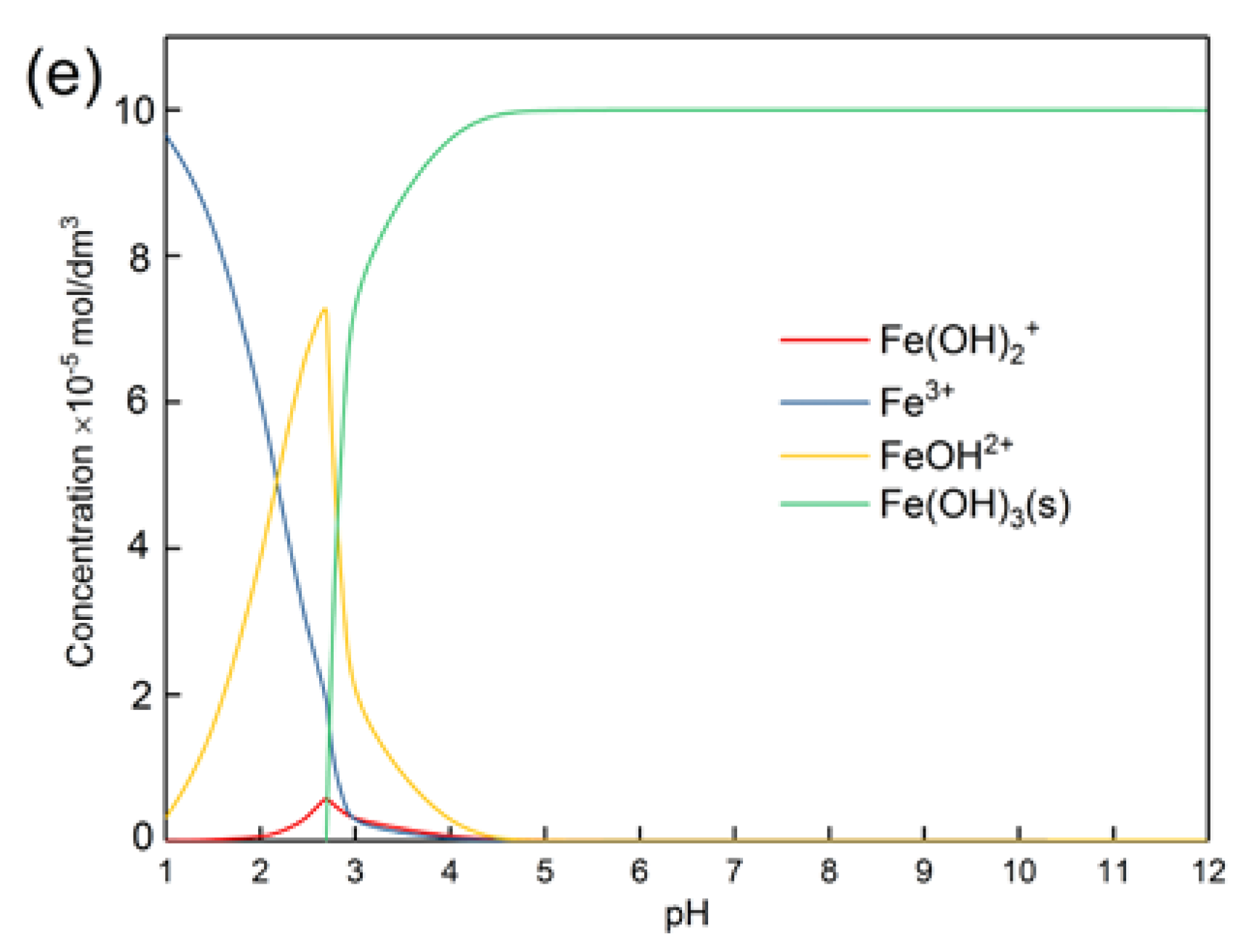
3.2. Concentration of Metal Ions Released from Pure Minerals
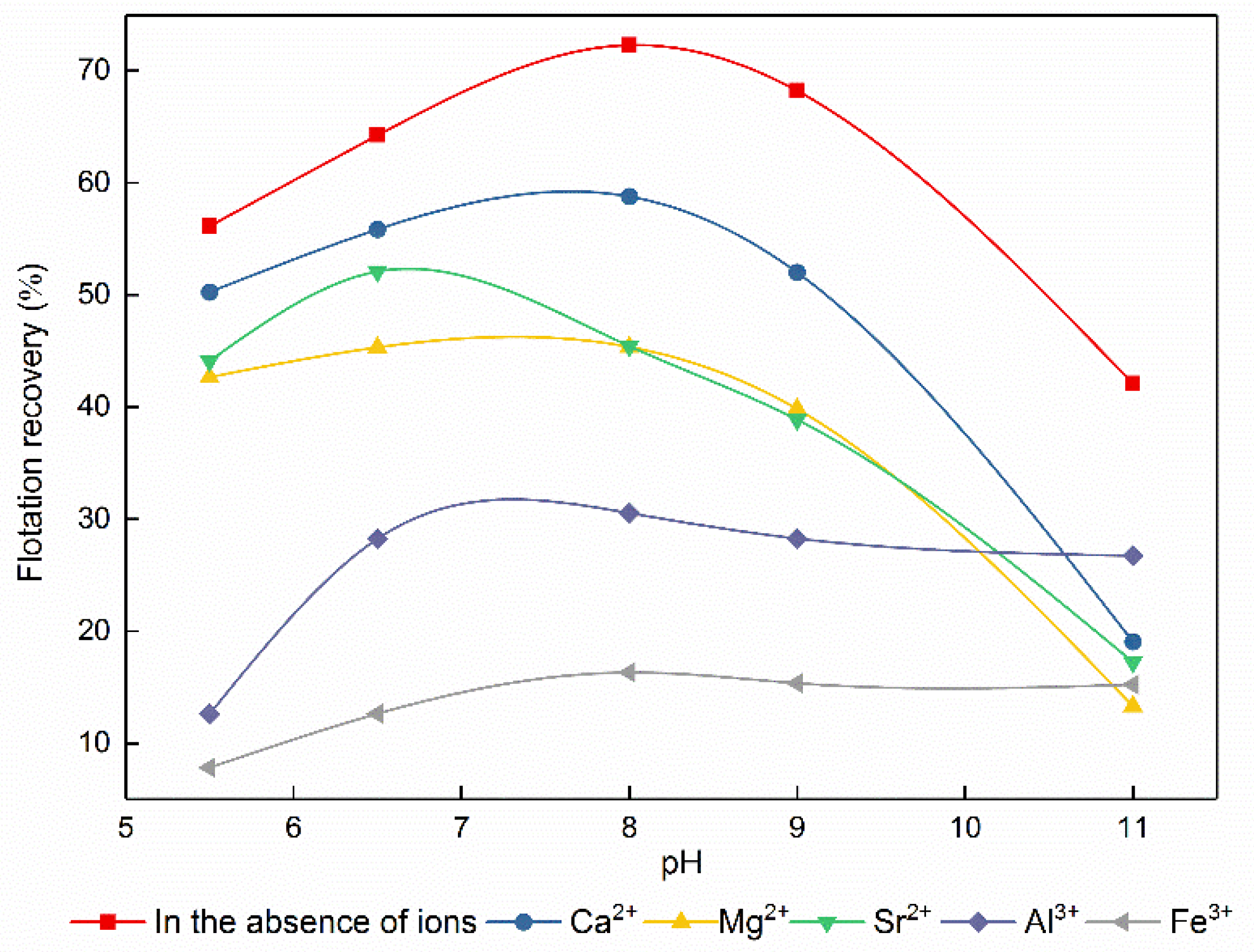
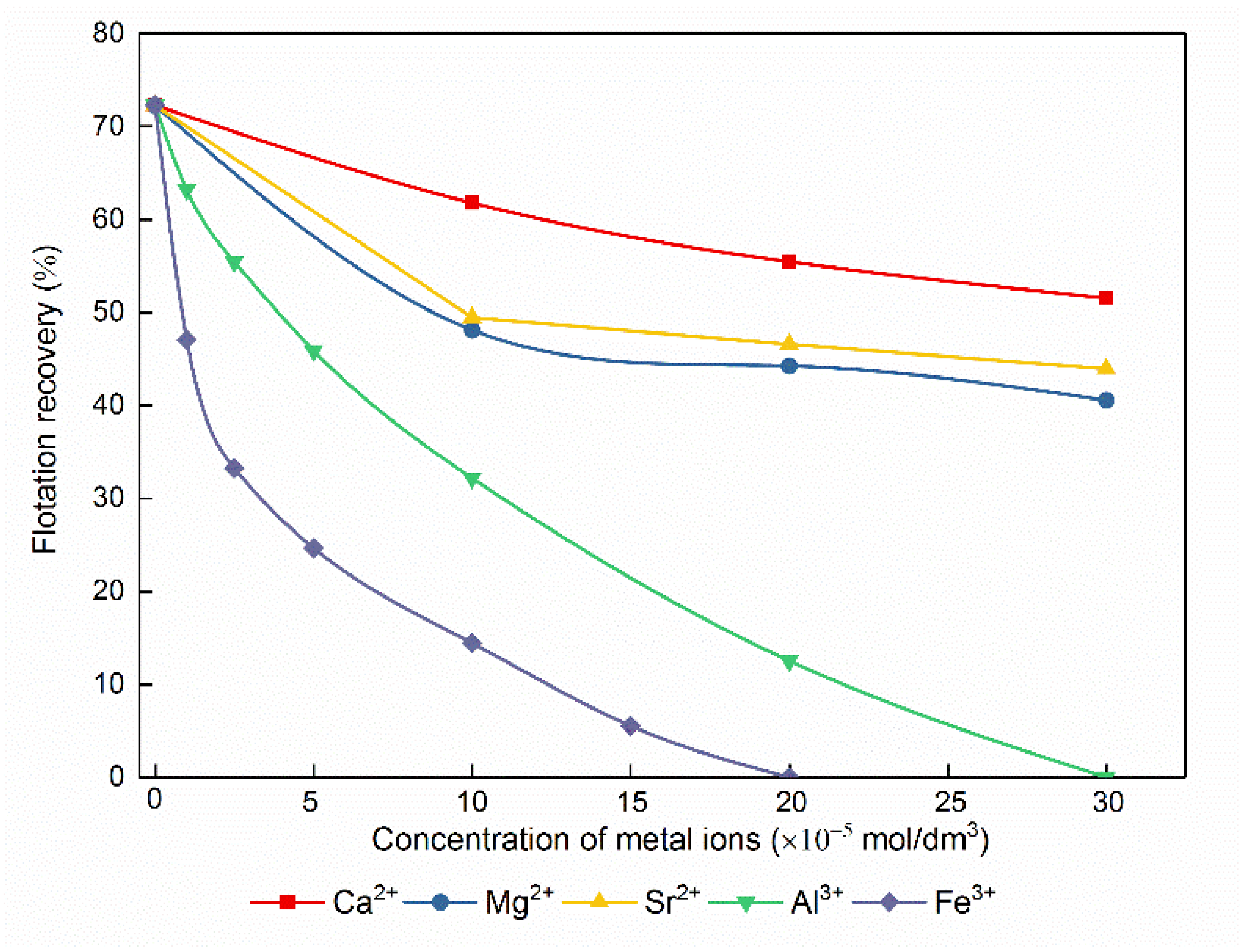
3.3. Influence of Metal Ions on Bastnaesite Flotation
3.4. The Influence Mechanism of Metal Ions on Bastnaesite Flotation
3.4.1. Induction Time Measurements
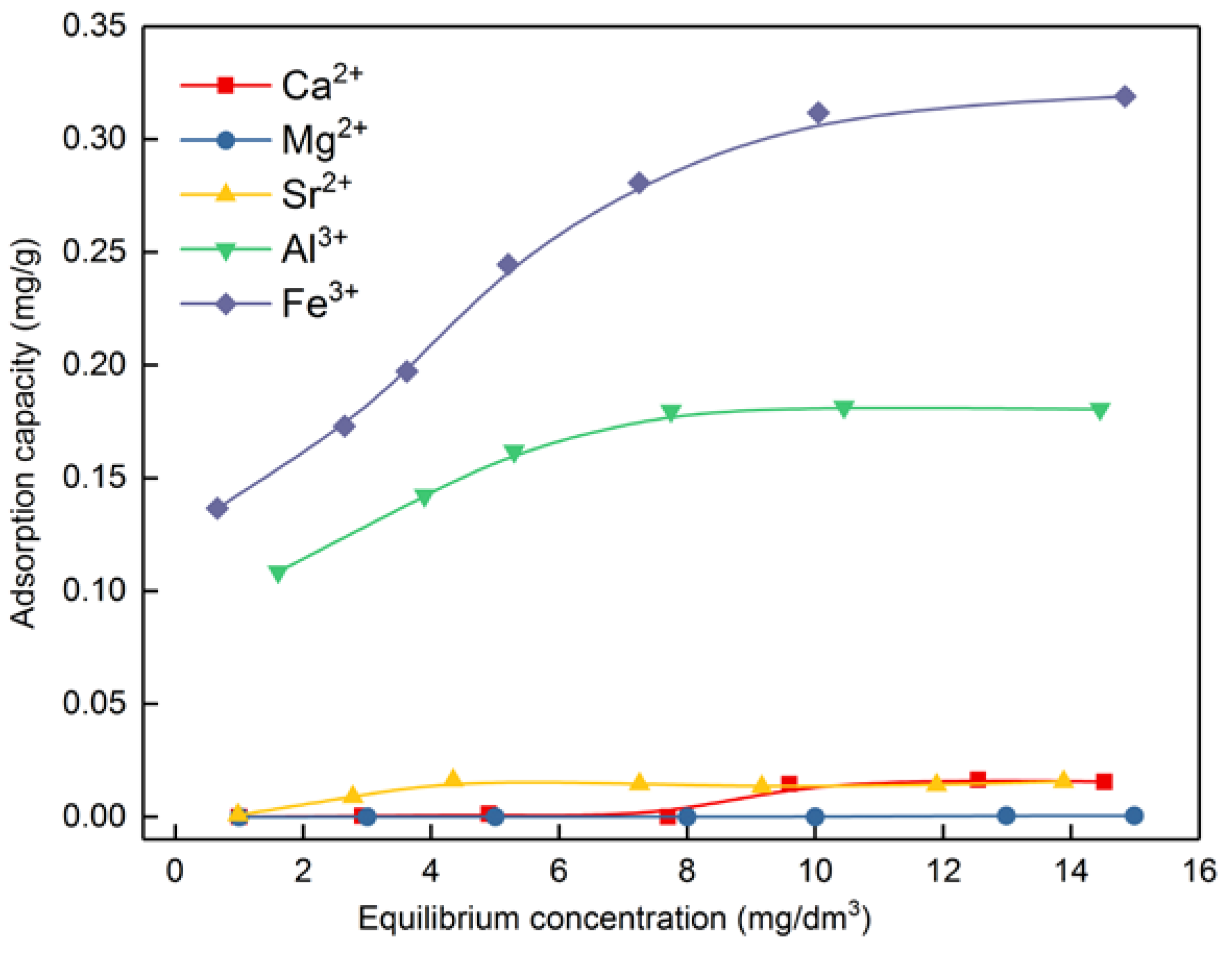
3.4.2. The Adsorption of Metal Ions on Bastnaesite Surfaces
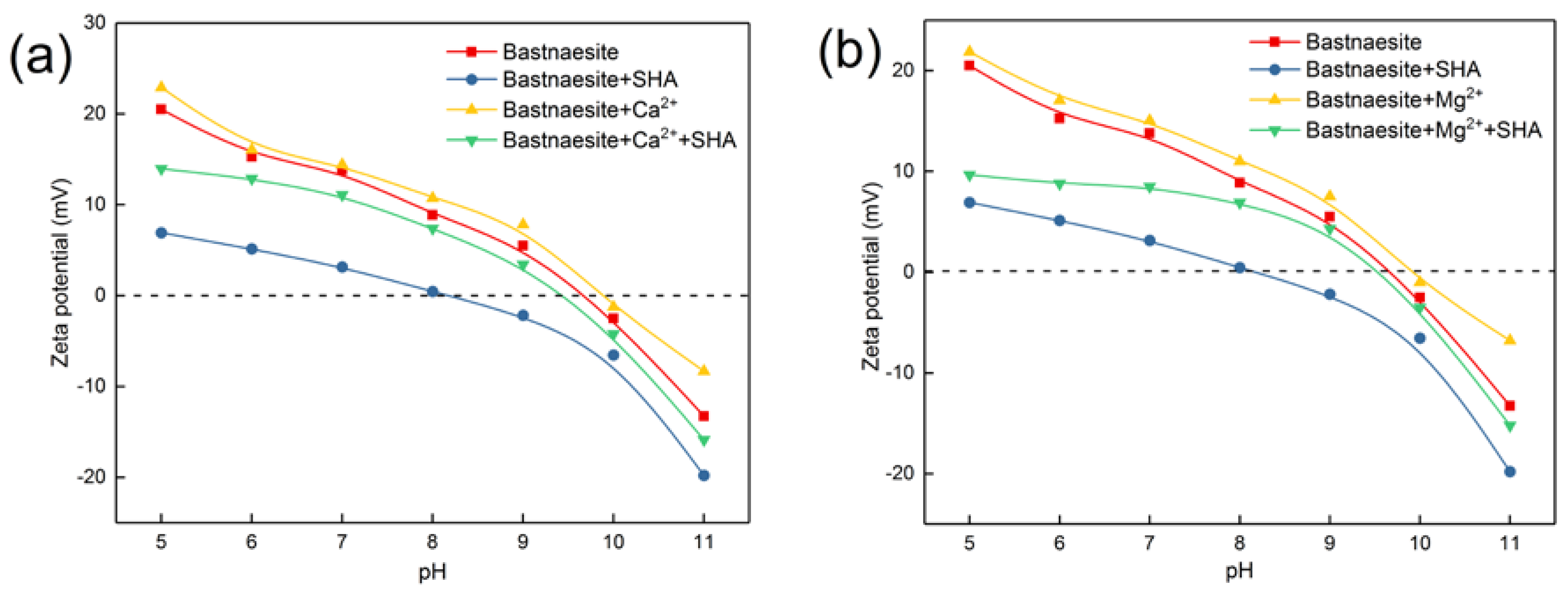
3.4.3. Effect of Metal Ions on Zeta Potential of Bastnaesite
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chi, R.; Wang, D. Mineral Processing of Rare Earth Minerals; Science Press: Beijing, China, 2014; pp. 33–37. [Google Scholar]
- Dolg, M.; Stoll, H. Handbook on the Physics and Chemistry of Rare Earths; Elsevier: Amsterdam, The Netherlands, 1978. [Google Scholar]
- Jordens, A.; Marion, C.; Kuzmina, O.; Waters, K.E. Surface chemistry considerations in the flotation of bastnäsite. Miner. Eng. 2014, 66–68, 119–129. [Google Scholar] [CrossRef]
- Jordens, A.; Cheng, Y.; Waters, K.E. A review of the beneficiation of rare earth element bearing minerals. Miner. Eng. 2013, 41, 97–114. [Google Scholar] [CrossRef]
- Long, K.R.; Gosen, B.S.V.; Foley, N.K.; Cordier, D. The Principal Rare Earth Elements Deposits of the United States: A Summary of Domestic Deposits and a Global Perspective; Springer: Dordrecht, The Netherlands, 2012; pp. 131–155. [Google Scholar]
- Martińez, A.L.; Uribe, A.S. Interfacial properties of celestite and strontianite in aqueous solutions. Miner. Eng. 1995, 8, 1009–1022. [Google Scholar] [CrossRef]
- Sonderegger, J.L.; Brower, K.R.; Lefebre, V.G. A preliminary investigation of strontianite dissolution kinetics. Am. J. Sci. 1976, 276, 997–1022. [Google Scholar] [CrossRef]
- Deng, R.; Hu, Y.; Ku, J.; Ma, Y.; Yang, Z. Ion migration law in flotation pulp and its influence on the separation of smithsonite and quartz. Sep. Sci. Technol. 2017, 2, 1–9. [Google Scholar] [CrossRef]
- Deng, J.; Wen, S.; Liu, J.; Xian, Y.; Wu, D.; Bai, S. New source of unavoidable ions in bornite Flotation Aqueous Solution: Fluid Inclusions. Ind. Eng. Chem. Res. 2013, 52, 4895–4901. [Google Scholar] [CrossRef]
- Liu, J.; Wen, S.; Wu, D.; Bai, S.; Liu, D. Determination of the concentrations of calcium and magnesium released from fluid inclusions of sphalerite and quartz. Miner. Eng. 2013, 45, 41–43. [Google Scholar] [CrossRef]
- Ren, J.; Song, S. Selective flotation of bastnaesite from monazite in rare earth concentrates using potassium alum as depressant. Int. J. Miner. Process. 2000, 59, 237–245. [Google Scholar] [CrossRef]
- Marinakis, K.I.; Shergold, H.L. The mechanism of fatty acid adsorption in the presence of fluorite, calcite and barite. Int. J. Miner. Process. 1985, 14, 161–176. [Google Scholar] [CrossRef]
- Fuerstenau, D.W. The adsorption of hydroxamate on semi-soluble minerals. Part I: Adsorption on barite, Calcite and Bastnaesite. Colloids Surf. 1983, 8, 103–119. [Google Scholar]
- Cui, M. The Influence and Its Elimination of Unavoidable Ions in the Flotation Separation of Smithsonite and Quartz. Master’s Thesis, Central South University, Changsha, China, 2012. [Google Scholar]
- Mackenzie, J.M.W. Zeta potential of quartz in the presence of ferric iron. Trans. AIME 1966, 235, 82. [Google Scholar]
- Deng, R.; Hu, Y.; Ku, J.; Zuo, W.; Yang, Z. Adsorption of Fe (III) on smithsonite surfaces and implications for flotation. Colloids Surf. A Physicochem. Eng. Asp. 2017, 533, 308–315. [Google Scholar] [CrossRef]
- Feng, Q.; Zhao, W.; Wen, S.; Cao, Q. Activation mechanism of lead ions in cassiterite flotation with salicylhydroxamic acid as collector. Sep. Purif. Technol. 2017, 178, 193–199. [Google Scholar] [CrossRef]
- Zeng, Q.; Zhao, H.; Wang, D. Influence of metal cations on cassiterite flotation. Trans. Nonferr. Met. Soc. 2000, 10, 98–101. [Google Scholar]
- Lascelles, D.; Finch, J.A.; Sui, C. Depressant action of Ca and Mg on flotation of Cu activated sphalerite. Can. Metall. Q. 2003, 42, 133–140. [Google Scholar] [CrossRef]
- Yu, F.; Wang, Y.; Wang, J.; Xie, Z.; Zhang, L. First-principle investigation on mechanism of Ca ion activating flotation of spodumene. Rare Met. 2014, 33, 358–362. [Google Scholar] [CrossRef]
- Celik, M.S. Effect of Ultrasonic Treatment on the Floatability of Coal and Galena. Sep. Sci. 1989, 24, 1159–1166. [Google Scholar] [CrossRef]
- Chen, J.; Hou, Y.; Che, L. Making rational multi-purpose use of resources of RE in Baiyunebo deposit. Chin. Rare Earths 2007, 1, 70–74. [Google Scholar]
- Yin, W. Physical Separation of Baotou Rare Earth Tailings. Master’s Thesis, Northeastern University, Shenyang, China, 2013. [Google Scholar]
- Lu, J.; Gao, H.; Jin, J. Effects of Metal Ions on Flotation of Kyanite with Sodium Dodecyl Sulfate. Met. Min. 2015, 44, 73–76. [Google Scholar]
- Deng, R. The Presence Behavior and Adsorption Mechanism of Metal Ions in the Flotation Pulp of Zinc Oxide Ore. Ph.D. Thesis, Kunming University of Science and Technology, Kunming, China, 2015. [Google Scholar]
- Welham, N.J.; Llewellyn, D.J. Mechanical enhancement of the dissolution of ilmenite. Miner. Eng. 1998, 11, 827–841. [Google Scholar] [CrossRef]
- Song, Z. Influence of Physicochemical Factors during Grinding on the Flotation of Several Carbonate Minerals. Ph.D. Thesis, Northeastern University, Shenyang, China, 2009. [Google Scholar]
- Guy, P.J.; Trahar, W.J. The influence of grinding and flotation environments on the laboratory batch flotation of galena. Int. J. Miner. Process. 1984, 12, 15–38. [Google Scholar] [CrossRef]
- Yoon, R.H.; Yordan, J.L. Induction time measurements for the quartz—Amine flotation system. J. Colloid Interface Sci. 1991, 141, 374–383. [Google Scholar] [CrossRef]
- Cao, S.; Cao, Y.; Liao, Y.; Ma, Z. Depression Mechanism of Strontium Ions in Bastnaesite Flotation with Salicylhydroxamic Acid as Collector. Minerals 2018, 8, 66. [Google Scholar] [CrossRef]
- Hirajima, T.; Suyantara, G.P.W.; Ichikawa, O.; Elmahdy, A.; Miki, H.; Sasaki, K. Effect of Mg2+ and Ca2+ as divalent seawater cations on the floatability of molybdenite and chalcopyrite. Miner. Eng. 2016, 96, 83–93. [Google Scholar] [CrossRef]
- Yu, Y.; Song, L.; Zhou, S.; Sun, C. Influence of multivalent metal ions on the floatability of several calcium salt minerals. Ind. Miner. Process. 2015, 9, 9–13. [Google Scholar]
- Ren, J.; Lu, S. Mechanism of aluminum salt in flotation of bastnaestite and monazite. Nonferr. Met. Eng. 1997, 2, 30–35. [Google Scholar]
- Gao, Y.; Gao, Z.; Sun, W. Research progress on the influence of metal ions on mineral flotation behavior. Trans. Nonferr. Met. Soc. 2017, 4, 859–868. [Google Scholar]
- Ou, L.; Ye, J.; Zeng, W.; Wan, L. Influence and Mechanism of Ferric and Ferrous Ions on Flotation of Smithsonite and Quartz. Nonferr. Met. 2012, 6, 79–82. [Google Scholar]
- Ai, G.; Cai, X.; Bi, K.; Li, J.; Tian, Y.; Zhang, R. Research progress on the effect of metal ions on mineral flotation behavior. Nonferr. Met. Sci. Eng. 2017, 6, 70–74. [Google Scholar]
- Parks, G.A. The isoelectric points of solid oxides, solid hydroxides, and aqueous hydroxo complex systems. Chem. Rev. 1965, 65, 177–198. [Google Scholar] [CrossRef]
- Zhao, Y. Flotation Separation of Hematite and Apatite. Master’s Thesis, Central South University, Changsha, China, 2012. [Google Scholar]



| Ions | Ca2+ | Mg2+ | Sr2+ | Cu2+ | Zn2+ | Pb2+ | Ba2+ |
| Concentration | 32.44 | 19.71 | 18.58 | 0.20 | 0.33 | 0.15 | 0.74 |
| Ions | Fe3+ | Al3+ | Na+ | K+ | Ce3+ | La3+ | Nd3+ |
| Concentration | 1.40 | <0.001 | 2.89 | 3.11 | 0.071 | 0.043 | 0.025 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, S.; Cao, Y.; Ma, Z.; Liao, Y. Metal Ion Release in Bastnaesite Flotation System and Implications for Flotation. Minerals 2018, 8, 203. https://doi.org/10.3390/min8050203
Cao S, Cao Y, Ma Z, Liao Y. Metal Ion Release in Bastnaesite Flotation System and Implications for Flotation. Minerals. 2018; 8(5):203. https://doi.org/10.3390/min8050203
Chicago/Turabian StyleCao, Shiming, Yijun Cao, Zilong Ma, and Yinfei Liao. 2018. "Metal Ion Release in Bastnaesite Flotation System and Implications for Flotation" Minerals 8, no. 5: 203. https://doi.org/10.3390/min8050203
APA StyleCao, S., Cao, Y., Ma, Z., & Liao, Y. (2018). Metal Ion Release in Bastnaesite Flotation System and Implications for Flotation. Minerals, 8(5), 203. https://doi.org/10.3390/min8050203






