Structure Simulation and Host–Guest Interaction of Histidine-Intercalated Hydrotalcite–Montmorillonite Complex
Abstract
1. Introduction
2. Calculation Methods
3. Results and Discussion
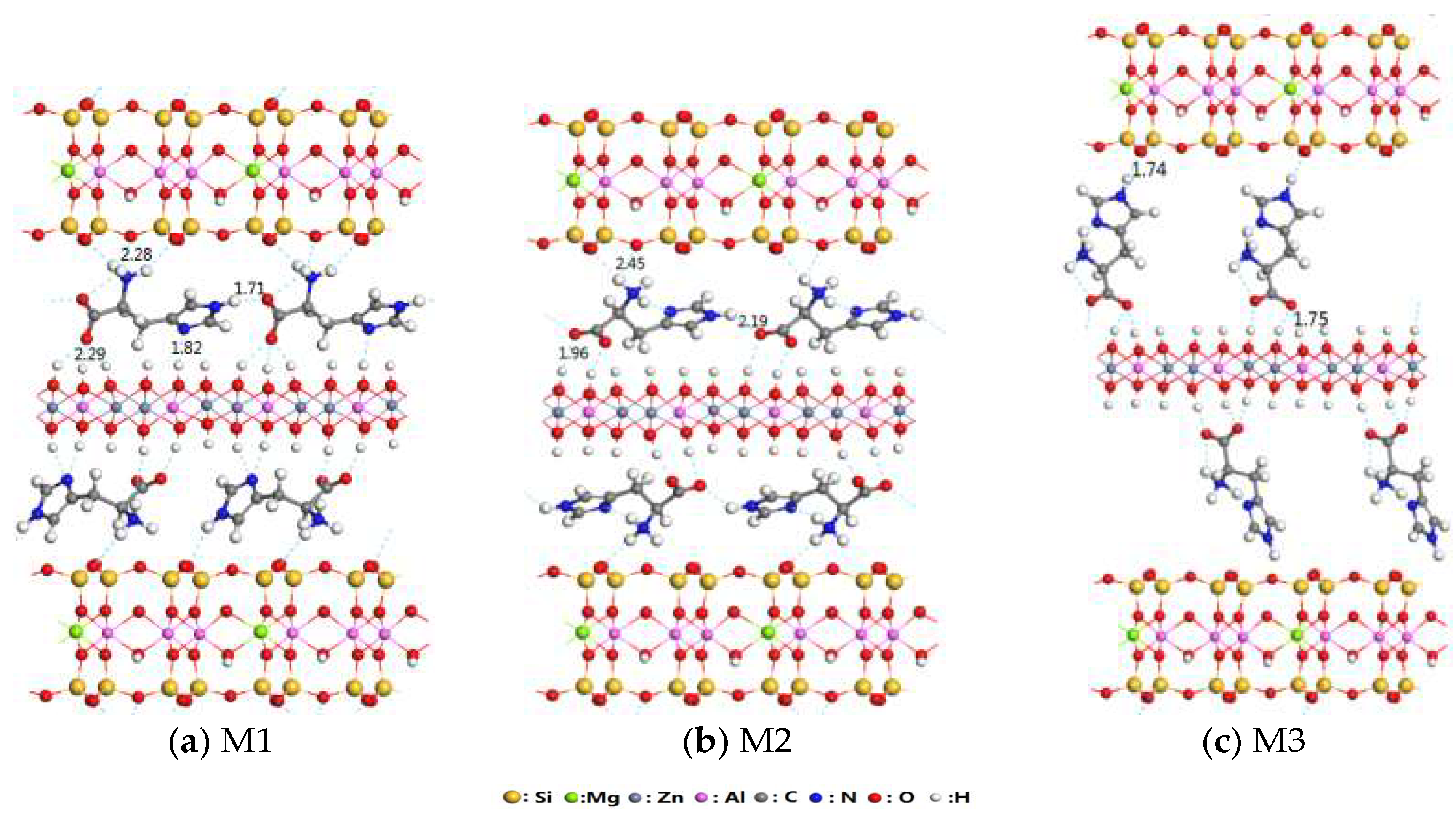
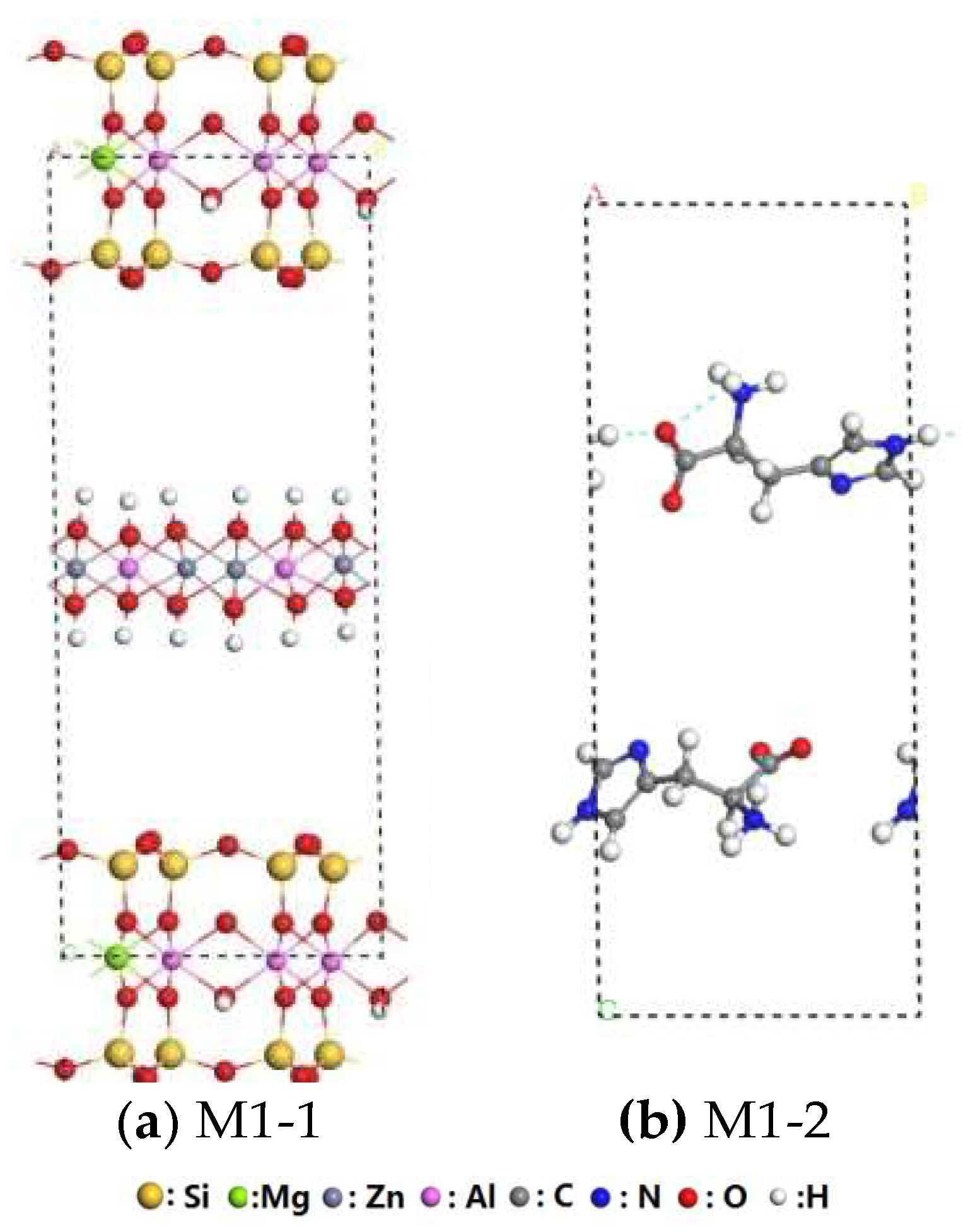
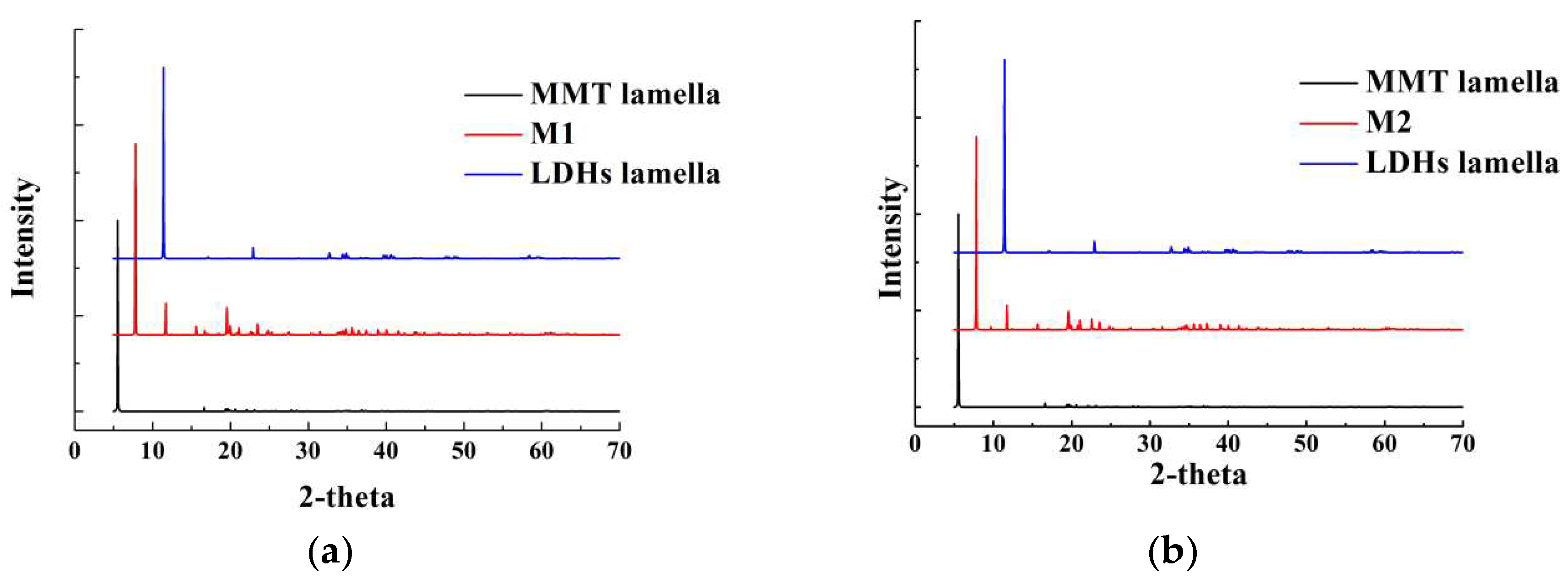
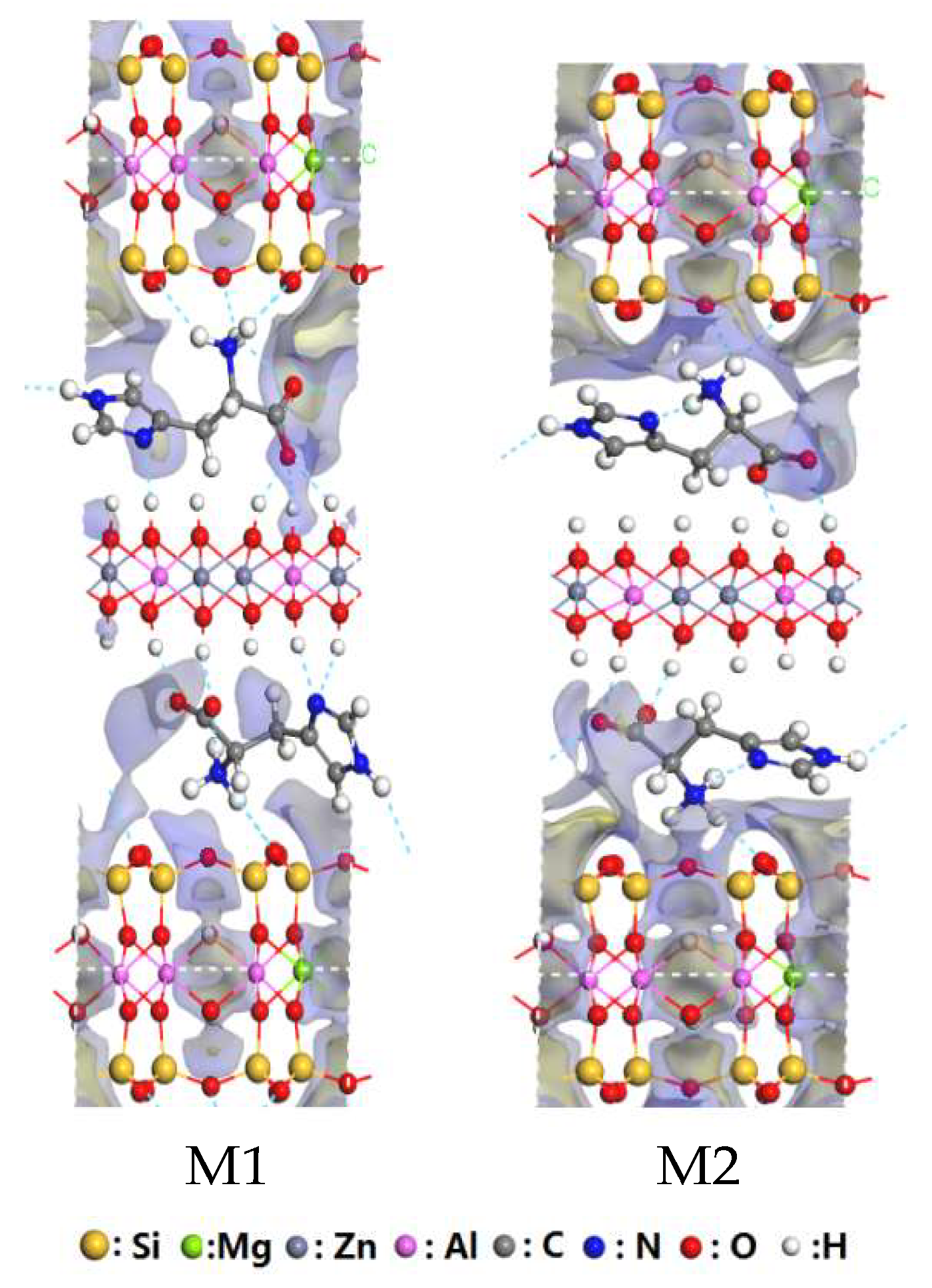
3.1. Structural Analysis of His–LDHs–MMT Complex
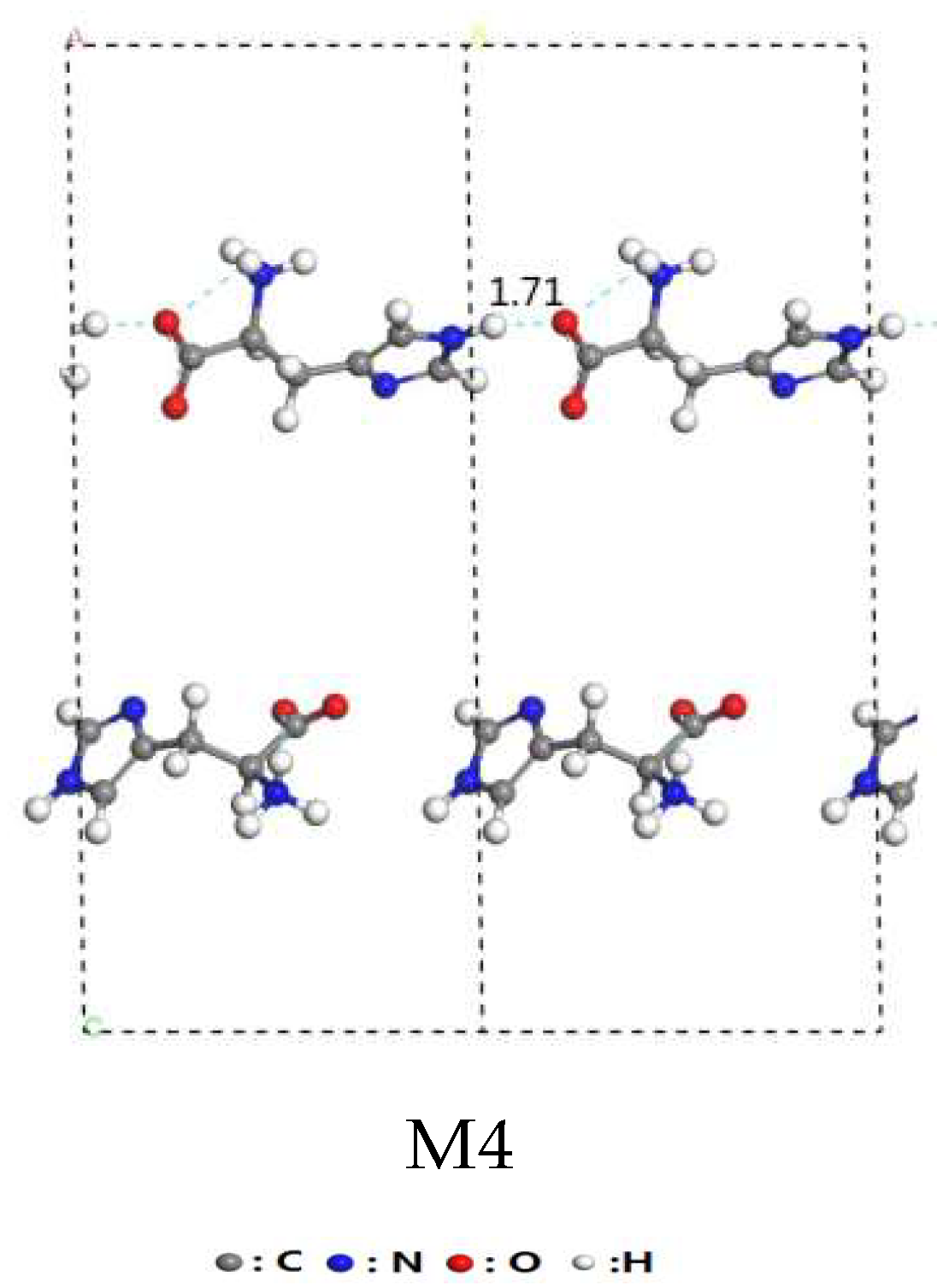
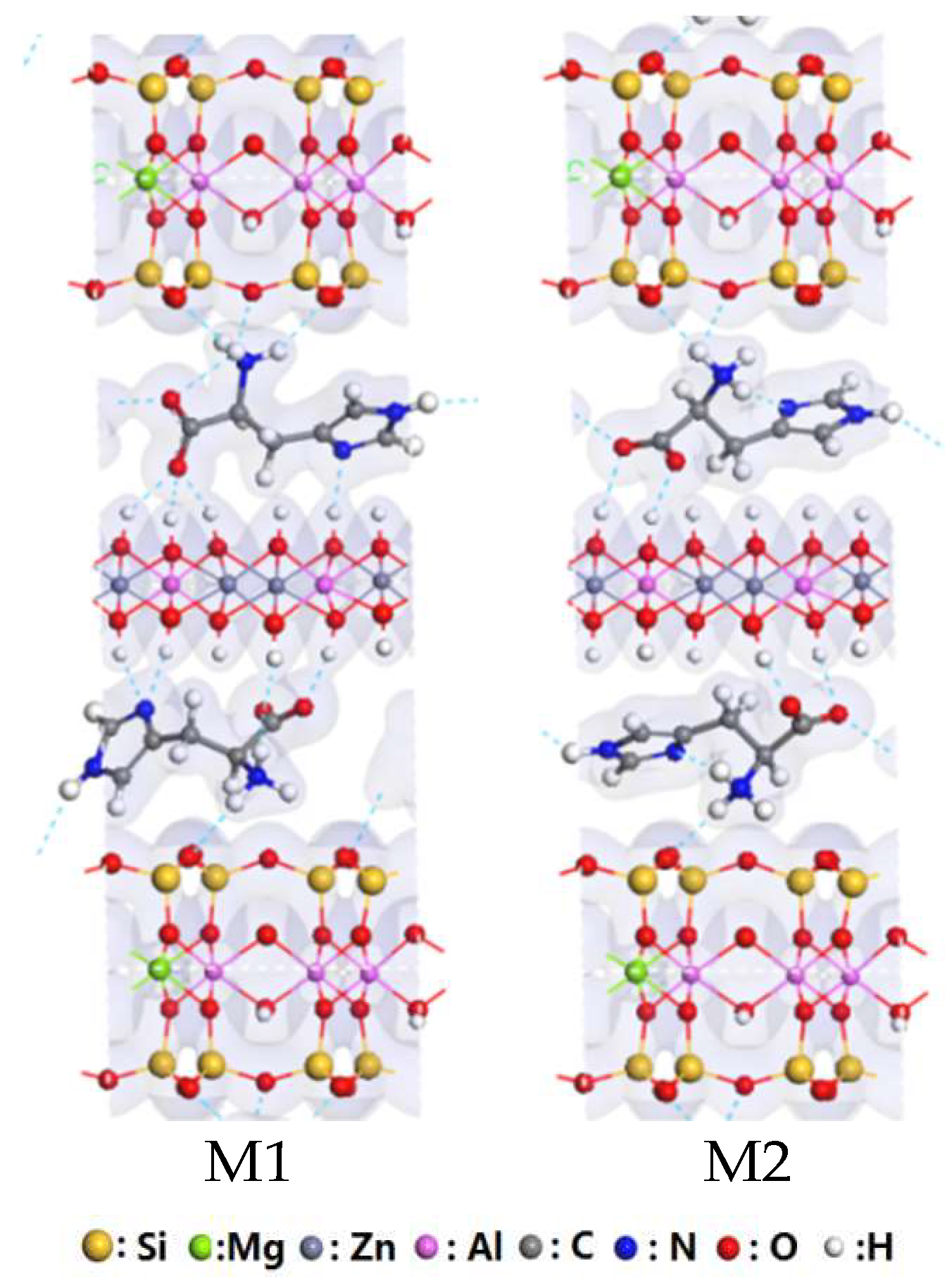
3.2. Interaction of Interlayer Guest of His–LDHs–MMT Complex
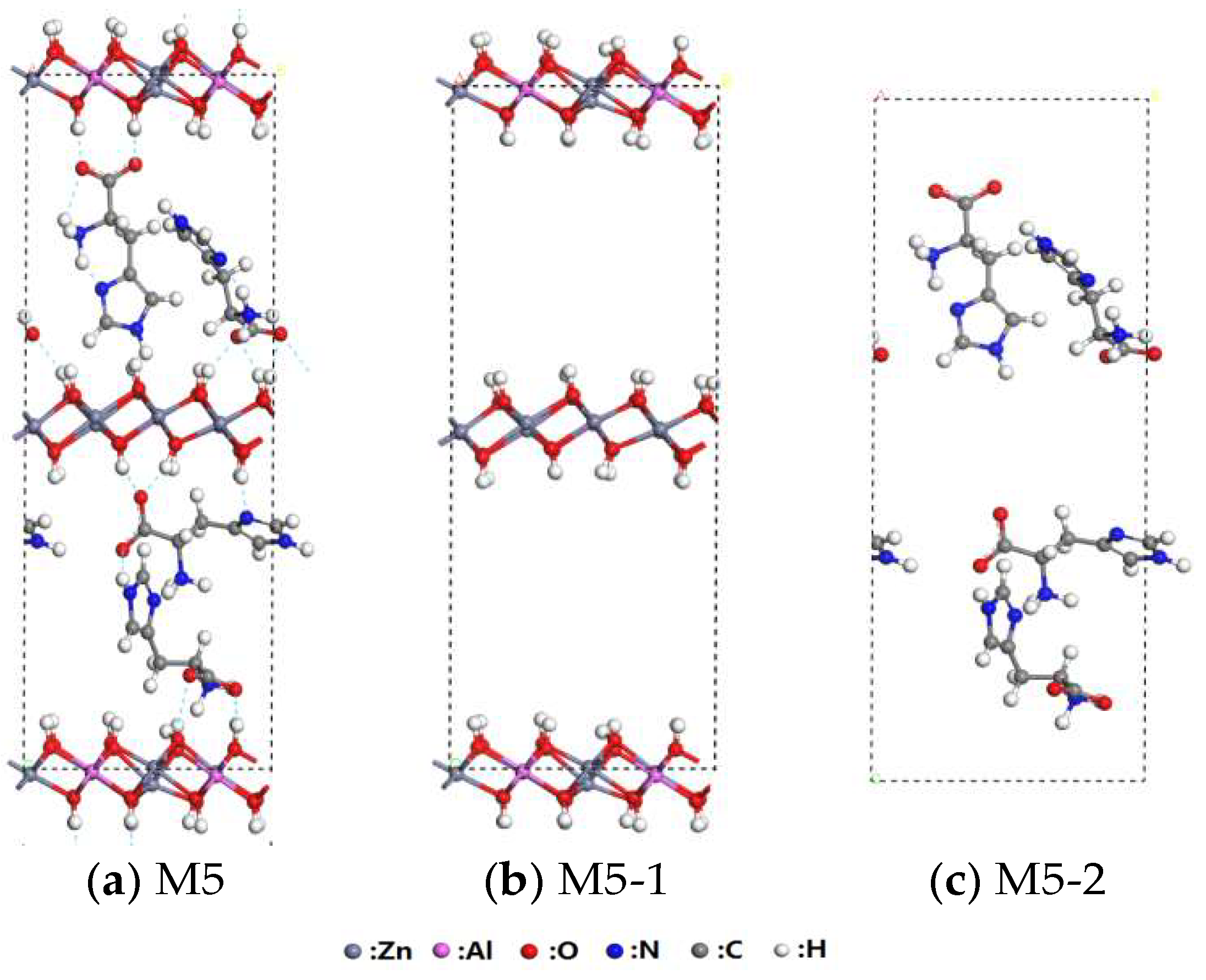
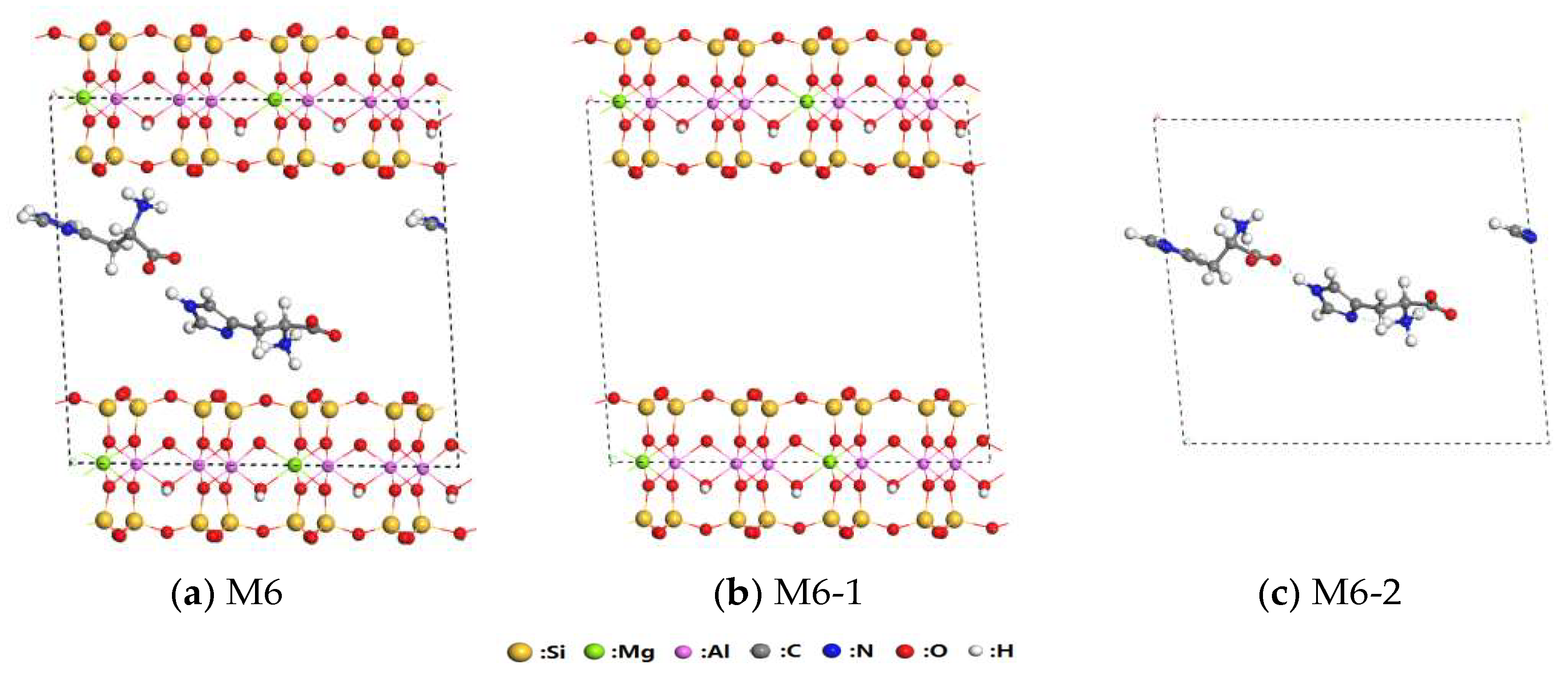
3.3. Interaction of His–LDHs and His–MMT
3.4. Interaction of His–LDHs–MMT Complex
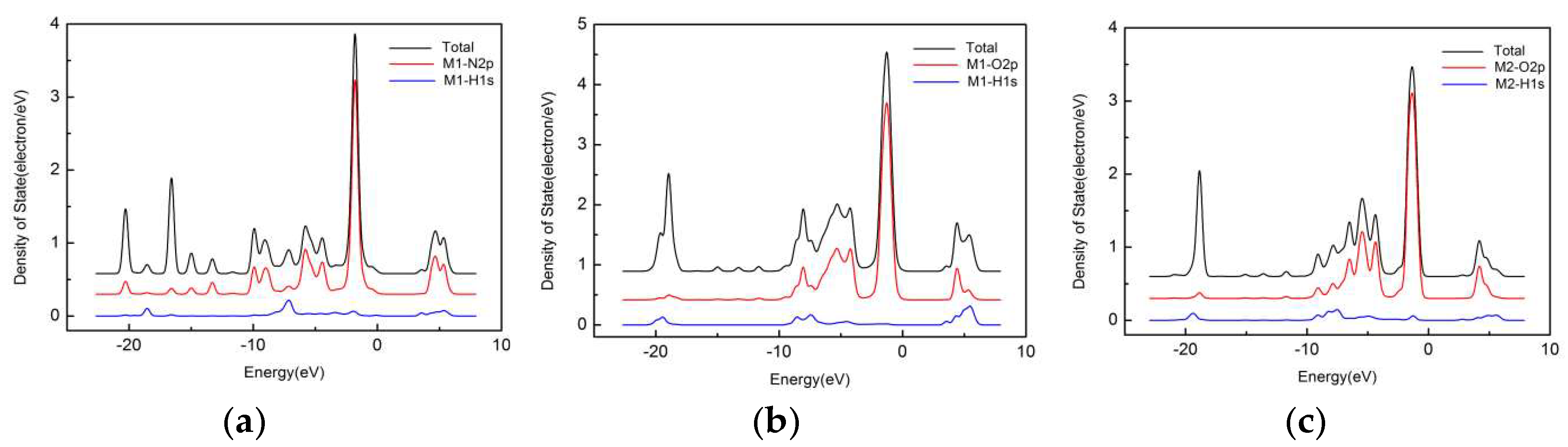
3.5. Electronic Structural Property of His–LDHs–MMT
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Ai, L.; Zhang, C.; Meng, L. Adsorption of methyl orange from aqueous solution on hydrothermal synthesized Mg-Al layered double hydroxide. J. Chem. Eng. Data 2011, 56, 4217–4225. [Google Scholar] [CrossRef]
- Lee, G.; Jeong, Y.; Takagaki, A.; Jung, J.C. Sonication assisted rehydration of hydrotalcite catalyst for isomerization of glucose to fructose. J. Mol. Catal. A Chem. 2014, 393, 289–295. [Google Scholar] [CrossRef]
- Serio, M.D.; Mallardo, S.; Carotenuto, G.; Tesser, R.; Santacesaria, E. Mg/Al hydrotalcite catalyst for biodiesel production in continuous packed bed reactors. Catal. Today 2012, 195, 54–58. [Google Scholar] [CrossRef]
- Wang, Y.B.; Jehng, J.M. Hydrotalcite-like compounds containing transition metals as solid base catalysts for transesterification. Chem. Eng. J. 2011, 175, 548–554. [Google Scholar] [CrossRef]
- Dadwhal, M.; Sahimi, M.; Tsotsis, T.T. Adsorption isotherms of arsenic on conditioned layered double hydroxides in the presence of various competing ions. Ind. Eng. Chem. Res. 2011, 50, 2220–2226. [Google Scholar] [CrossRef]
- Rives, V.; Arco, M.; Martin, C. Layered double hydroxides as drug carriers and for controlled release of non-steroidal antiinflammatory drugs (NSAIDs): A review. J. Control. Release 2013, 169, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Vial, S.; Prevot, V.; Lerous, F.; Forano, C. Immobilization of urease in ZnAl layered double hydroxides by soft chemistry routes. Microporous Mesoporous Mater. 2008, 107, 190–201. [Google Scholar] [CrossRef]
- Tarutani, N.; Tokudome, Y.; Jobbagy, M.; Viva, F.A.; Soler-Illia, G.J.A.A.; Takahashi, M. Single-nanometer-sized low-valence metal hydroxide crystals: Synthesis via epoxide-mediated alkalinization and assembly toward functional mesoporous materials. Chem. Mater. 2016, 28, 5606–5610. [Google Scholar] [CrossRef]
- Dewoolkar, K.D.; Vaidya, P.D. Tailored Ce- and Zr-doped Ni/hydrotalcite materials for superior sorption-enhanced steam methane reforming Int. J. Hydrogen Energy 2017, 42, 21762–21774. [Google Scholar] [CrossRef]
- Stawinski, W.; Wegrzyn, A.; Freitas, O.; Chmielarz, L.; Figueiredo, S. Dual-function hydrotalcite-derived adsorbents with sulfur storage properties: Dyes and hydrotalcite fate in adsorption-regeneration cycles. Microporous Mesoporous Mater. 2017, 250, 72–87. [Google Scholar] [CrossRef]
- Anirudhan, T.S.; Suchithra, P.S. Synthesis and characterization of tannin-immobilized hydrotalcite as a potential adsorbent of heavy metal ions in effluent treatments. Appl. Clay Sci. 2008, 42, 214–223. [Google Scholar] [CrossRef]
- Park, Y.; Ayoko, G.A.; Frost, R.L. Application of organoclays for the adsorption of recalcitrant organic molecules from aqueous media. J. Colloid Interface Sci. 2011, 354, 292–305. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, Z. Cation exchange interaction between antibiotic ciprofloxacin and montmorillonite. J. Hazard. Mater. 2010, 183, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Gemeay, A.H.; El-Sherbiny, A.S.; Zaki, A.B. Adsorption and kinetic studies of the intercalation of some organic compounds onto Na+-montmorillonite. J. Colloid Interface Sci. 2002, 245, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, P.K.; Samal, R.F. Fire retardancy and biodegradability of poly(methyl methacrylate)/montmorillonite nanocomposite. Polym. Degrad. Stab. 2007, 92, 1700–1702. [Google Scholar] [CrossRef]
- Kan, T.; Jiang, X.; Zhou, L.; Yang, M.; Duan, M. Removal of methyl orange from aqueous solutions using a bentonite modified with a new gemini surfactant. Appl. Clay Sci. 2011, 54, 184–187. [Google Scholar] [CrossRef]
- Liu, K.; Liu, T.; Chen, S.; Liu, D. Drug release behavior of chitosan–montmorillonite nanocomposite hydrogels following electrostimulation. Acta Biomater. 2008, 4, 1038–1045. [Google Scholar] [CrossRef] [PubMed]
- Zaghouane-Boudiaf, H.; Boutahala, M. Adsorption of 2,4,5-trichlorophenol by organo-montmorillonites from aqueous solutions: Kinetics and equilibrium studies. Chem. Eng. J. 2011, 170, 120–126. [Google Scholar] [CrossRef]
- Park, J.H.; Shin, H.J.; Kim, M.H.; Kim, J.S.; Kang, N.; Lee, J.Y.; Kim, K.T.; Lee, J.I.; Kim, D.D. Application of montmorillonite in bentonite as a pharmaceutical excipient in drug delivery systems. J. Pharm. Investig. 2016, 46, 363–375. [Google Scholar] [CrossRef]
- Terry, P.A. Characterization of Cr ion exchange with hydrotalcite. Chemosphere 2004, 57, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Goh, K.; Lim, T.; Dong, Z. Application of layered double hydroxides for removal of oxyanions: A review. Water Res. 2008, 42, 1343–1368. [Google Scholar] [CrossRef] [PubMed]
- Barriga, C.; Gaitan, M.; Pavlovic, I. Hydrotalcites as sorbent for 2,4,6-trinitrophenol: Influence of the layer composition and interlayer anion. J. Mater. Chem. 2002, 12, 1027–1032. [Google Scholar] [CrossRef]
- Pu, M.; Su, Y.; Zhang, X.F.; He, S.H. An Amino Acid Compound Assembled Montmorillonite-Hydrotalcite Layered Material and Preparation Method. China ZL 2013 101421982, 22 April 2013. [Google Scholar]
- Scheffler, U.; Mahrwald, R. Histidine-catalyzed asymmetric aldol addition of enolizable aldehydes: Insights into its mechanism. J. Org. Chem. 2012, 77, 2310–2330. [Google Scholar] [CrossRef] [PubMed]
- Lam, Y.H.; Houk, K.N.; Scheffler, U.; Mahrwald, R. Stereoselectivities of histidine-catalyzed asymmetric aldol additions and contrasts with proline catalysis: A quantum mechanical analysis. J. Am. Chem. Soc. 2012, 134, 6286–6295. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.F.; Wei, Q.Y.; Jiang, H.; Xie, P.F. L-proline and L-histidine co-catalyzed Baylis–Hillman reaction. Res. Chem. Intermed. 2012, 38, 639–644. [Google Scholar] [CrossRef]
- Liu, X.X.; Melman, A. Templated alkylation of hexahistidine with Baylis–Hillman esters. Chem. Commun. 2013, 49, 9042–9044. [Google Scholar] [CrossRef] [PubMed]
- Mulder, D.T.; McPhee, J.B.; Yu, S.A.R.; Stogios, P.J.; Savchenko, A.; Coombes, B.K. Multiple histidines in the periplasmic domain of the Salmonella enterica sensor kinase SsrA enhance signaling in response to extracellular acidification. Mol. Microbiol. 2015, 95, 678–691. [Google Scholar] [CrossRef] [PubMed]
- Rashid, Z.; Naeimi, H.; Zarnani, A.H.; Mohammadi, F.; Ghahremanzadeh, R. Facile fabrication of nickel immobilized on magnetic nanoparticles as an efficient affinity adsorbent for purification of his-tagged protein. Mater. Sci. Eng. C 2017, 80, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.W.; Dong, S.S.; Liu, J.H.; Nie, C.M.; Wen, G.B. Peroxidase activity of a myoglobin mutant with three distal histidines forming a metal-binding site: Implications for the cross-reactivity of cytochrome c oxidase. J. Mol. Catal. B Enzym. 2013, 91, 25–31. [Google Scholar] [CrossRef]
- Dong, S.S.; Du, K.J.; You, Y.; Liu, F.; Wen, G.B.; Lin, Y.W. Peroxidase-like activity of L29H myoglobin with two cooperative distal histidines on electrode using O2 as an oxidant. J. Electroanal. Chem. 2013, 708, 1–6. [Google Scholar] [CrossRef]
- Mahindra, A.; Jain, R. Regiocontrolled palladium-catalyzed and copper-mediated C–H bond functionalization of protected L-histidine. Org. Biomol. Chem. 2014, 12, 3792–3796. [Google Scholar] [CrossRef] [PubMed]
- Mahindra, A.; Bagra, N.; Jain, R. Palladium-catalyzed regioselective C-5 arylation of protected L-histidine: Microwave-assisted C-H activation adjacent to donor arm Org. Chem. 2013, 78, 10954–10959. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhang, Z.; Cheetham, J.; Ren, D.; Zhou, Z.S. Discovery and characterization of a photo-oxidative histidine-histidine cross-link in IgG1 antibody utilizing 18O-Labeling and mass spectrometry. Anal. Chem. 2014, 86, 4940–4948. [Google Scholar] [CrossRef] [PubMed]
- Vu, A.P.; Mguyen, T.N.; Do, T.T.; Doan, T.H. Clinical screening of paraquat in plasma samples using capillary electrophoresis with contactless conductivity detection: Towards rapid diagnosis and therapeutic treatment of acute paraquat poisoning in Vietnam. J. Chromatogr. B 2017, 1060, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Moti, M.; Ishikawara, F.; Tomoda, T.; Yamada, S. Use of capillary electrophoresis with dual-opposite end injection for simultaneous analysis of small ions in saliva samples from wrestlers undergoing a weight training program. J. Chromatogr. B 2016, 1012–1013, 178–185. [Google Scholar]
- Aisawa, S.; Sasaki, S.; Takahashi, S.; Hirahara, H.; Nakayama, H.; Narita, E. Intercalation of amino acids and oligopeptides into Zn–Al layered double hydroxide by coprecipitation reaction. J. Phys. Chem. Solids 2006, 67, 920–925. [Google Scholar] [CrossRef]
- Nakayama, H.; Wada, N.; Tsuhako, M. Intercalation of amino acids and peptides into Mg–Al layered double hydroxide by reconstruction method. Int. J. Pharm. 2004, 269, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Whilton, N.T.; Vickers, P.J.; Mann, S. Bioinorganic clays: Synthesis and characterization of amino- and polyamino acid intercalated layered double hydroxides. J. Mater. Chem. 1997, 7, 1623–1629. [Google Scholar] [CrossRef]
- Fudala, A.; Pálinko, I.; Kiricsi, I. Preparation and characterization of hybrid organic-inorganic composite materials using the amphoteric property of amino acids: Amino acid intercalated layered double hydroxide and montmorillonite. Inorg. Chem. 1999, 38, 4653–4658. [Google Scholar] [CrossRef]
- Yuan, Q.; Wei, M.; Evans, D.G.; Duan, X. Preparation and investigation of thermolysis of L-aspartic acid-intercalated layered double hydroxide. J. Phys. Chem. B 2004, 108, 12381–12387. [Google Scholar] [CrossRef]
- Kitadai, N.; Yokoyama, T.; Nakashima, S. In situ ATR-IR investigation of L-lysine adsorption on montmorillonite. J. Colloid Interface Sci. 2009, 338, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Mallakpour, S.; Dinari, M. Insertion of novel optically active poly(amide-imide) chains containing pyromellitoyl-bis-L-phenylalanine linkages into the nanolayered silicates modified with L-tyrosine through solution intercalation. Polymer 2011, 51, 2514–2523. [Google Scholar] [CrossRef]
- Fraser, D.G.; Greenwell, C.; Skipper, N.; Smalley, M.V.; Wilkinson, M.A.; Deme, B.; Heenan, R.K. Chiral interactions of histidine in a hydrated vermiculite. Phys. Chem. Chem. Phys. 2011, 13, 825–830. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Songurtein, D.; Yalcinlaya, E.E.; Ag, D.; Seleci, M.; Demirkol, D.O. Histidine modified montmorillonite: Laccase immobilization and application to flow injection analysis of phenols. Appl. Clay Sci. 2013, 68, 64–69. [Google Scholar] [CrossRef]
- He, S.H.; Pu, M.; Zhang, X.F.; Wang, C.X.; Wang, H.X. Analysis and simulations on the structure of sulfanilic acid zwitterion intercalated hydrotalcite and montmorillonite. RSC Adv. 2016, 6, 83656–83662. [Google Scholar] [CrossRef]
- Delley, B. From molecules to solids with the DMol3 approach. J. Chem. Phys. 2000, 113, 7756–7763. [Google Scholar] [CrossRef]
- Delley, B. An all-electron numerical method for solving the local density functional for polyatomic molecules. J. Chem. Phys. 1990, 92, 508–517. [Google Scholar] [CrossRef]
| a (nm) | c (nm) | Energy/Ha | |
|---|---|---|---|
| M1 | 0.53 | 2.27 | −14,666.2078 |
| M2 | 0.53 | 2.26 | −14,666.1890 |
| M3 | 0.53 | 2.98 | −14,666.0605 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.-X.; Pu, M.; Zhang, P.-H.; Gao, Y.; Yang, Z.-Y.; Lei, M. Structure Simulation and Host–Guest Interaction of Histidine-Intercalated Hydrotalcite–Montmorillonite Complex. Minerals 2018, 8, 198. https://doi.org/10.3390/min8050198
Wang C-X, Pu M, Zhang P-H, Gao Y, Yang Z-Y, Lei M. Structure Simulation and Host–Guest Interaction of Histidine-Intercalated Hydrotalcite–Montmorillonite Complex. Minerals. 2018; 8(5):198. https://doi.org/10.3390/min8050198
Chicago/Turabian StyleWang, Chen-Xi, Min Pu, Pei-Huan Zhang, Yang Gao, Zuo-Yin Yang, and Ming Lei. 2018. "Structure Simulation and Host–Guest Interaction of Histidine-Intercalated Hydrotalcite–Montmorillonite Complex" Minerals 8, no. 5: 198. https://doi.org/10.3390/min8050198
APA StyleWang, C.-X., Pu, M., Zhang, P.-H., Gao, Y., Yang, Z.-Y., & Lei, M. (2018). Structure Simulation and Host–Guest Interaction of Histidine-Intercalated Hydrotalcite–Montmorillonite Complex. Minerals, 8(5), 198. https://doi.org/10.3390/min8050198






