Towards Zn-Dominant Tourmaline: A Case of Zn-Rich Fluor-Elbaite and Elbaite from the Julianna System at Piława Górna, Lower Silesia, SW Poland
Abstract
1. Introduction
- X—Na+, K+, Ca2+, Pb2+, ☐ (vacancy),
- Y—Fe2+, Mg2+, Mn2+, Al3+, Li+, Fe3+, Cr3+, V3+, Ti4+, Zn2+, Cu2+, Ni2+, …
- Z—Al3+, Fe3+, Cr3+, V3+, Mg2+, Fe2+, ...
- T—Si4+, Al3+, B3+,
- B—B3+;
- V—OH−, O2−,
- W—OH−, F−, O2−.
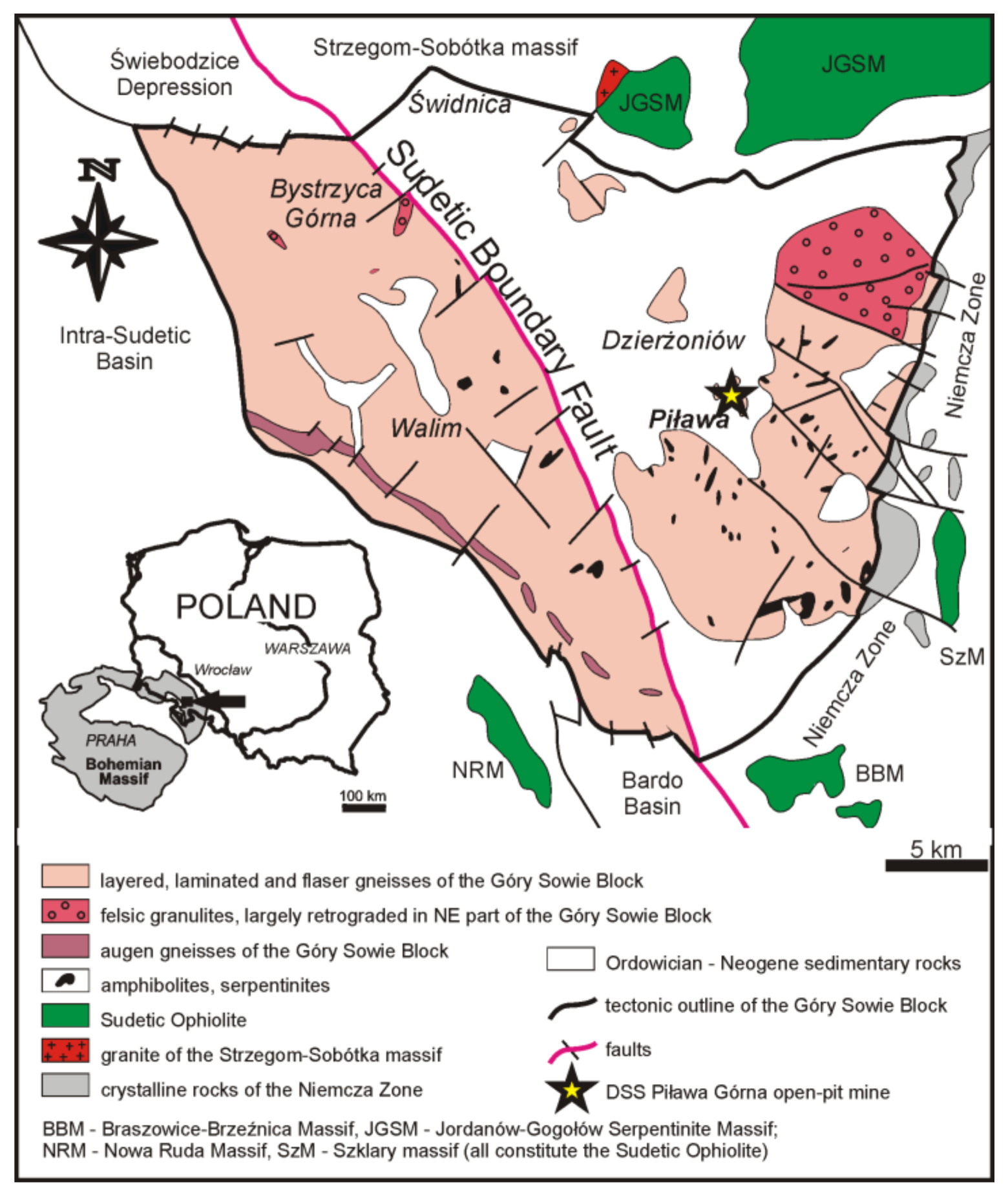
2. Geological Setting
3. Occurrence
4. Methods
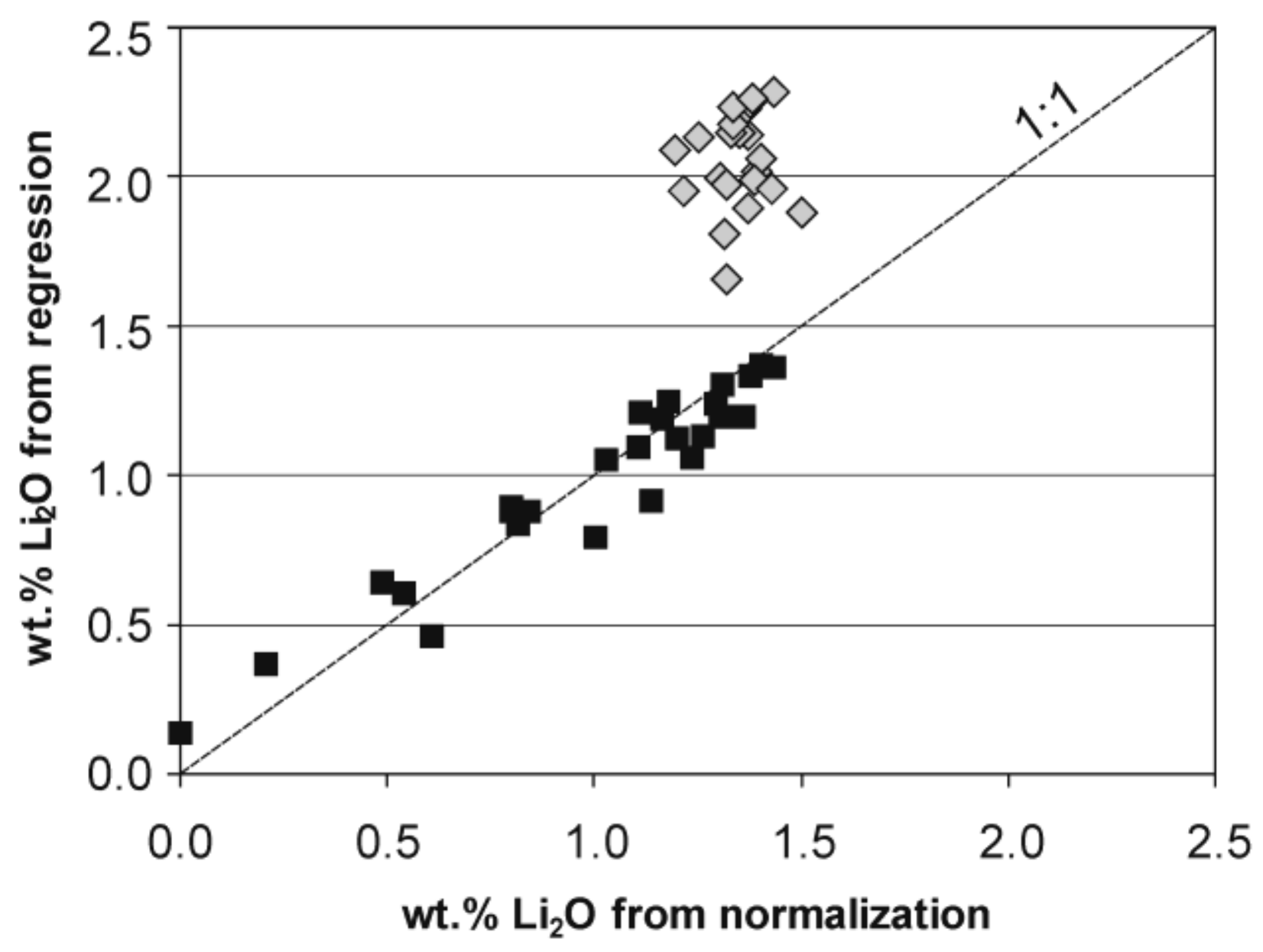
4.1. Electron Microprobe Analysis (EMPA)
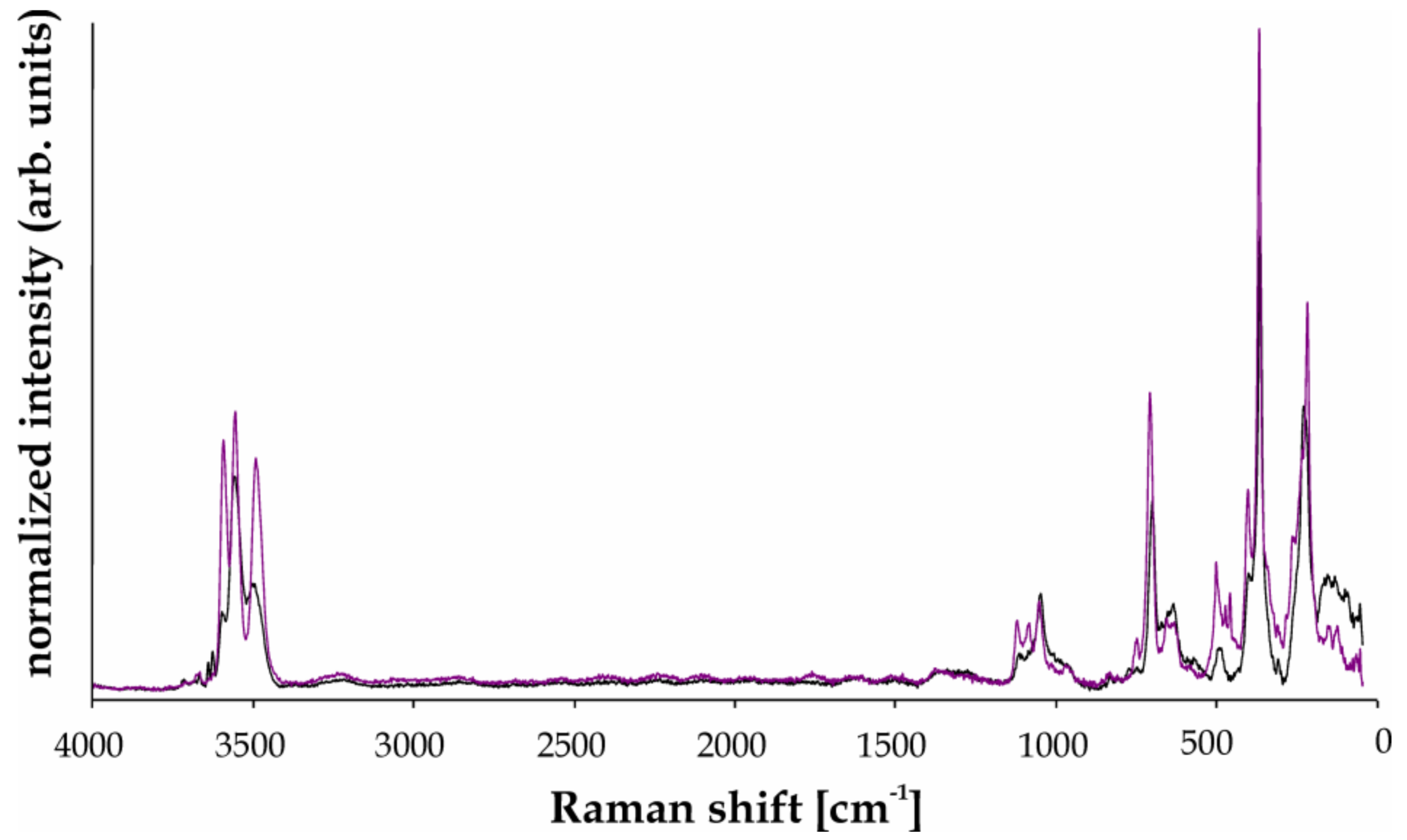
4.2. Raman Spectroscopy
5. Results
5.1. Primary Tourmalines
5.2. Secondary Dark Bluish Li-Bearing Tourmaline in Quartz
5.3. Secondary Dark Greenish Fluor-Elbaite in Muscovite Books
5.4. Zn-Rich Tourmaline from Gahnite Dissolution—Fluor-Elbaite + Elbaite Reprecipitation Zones
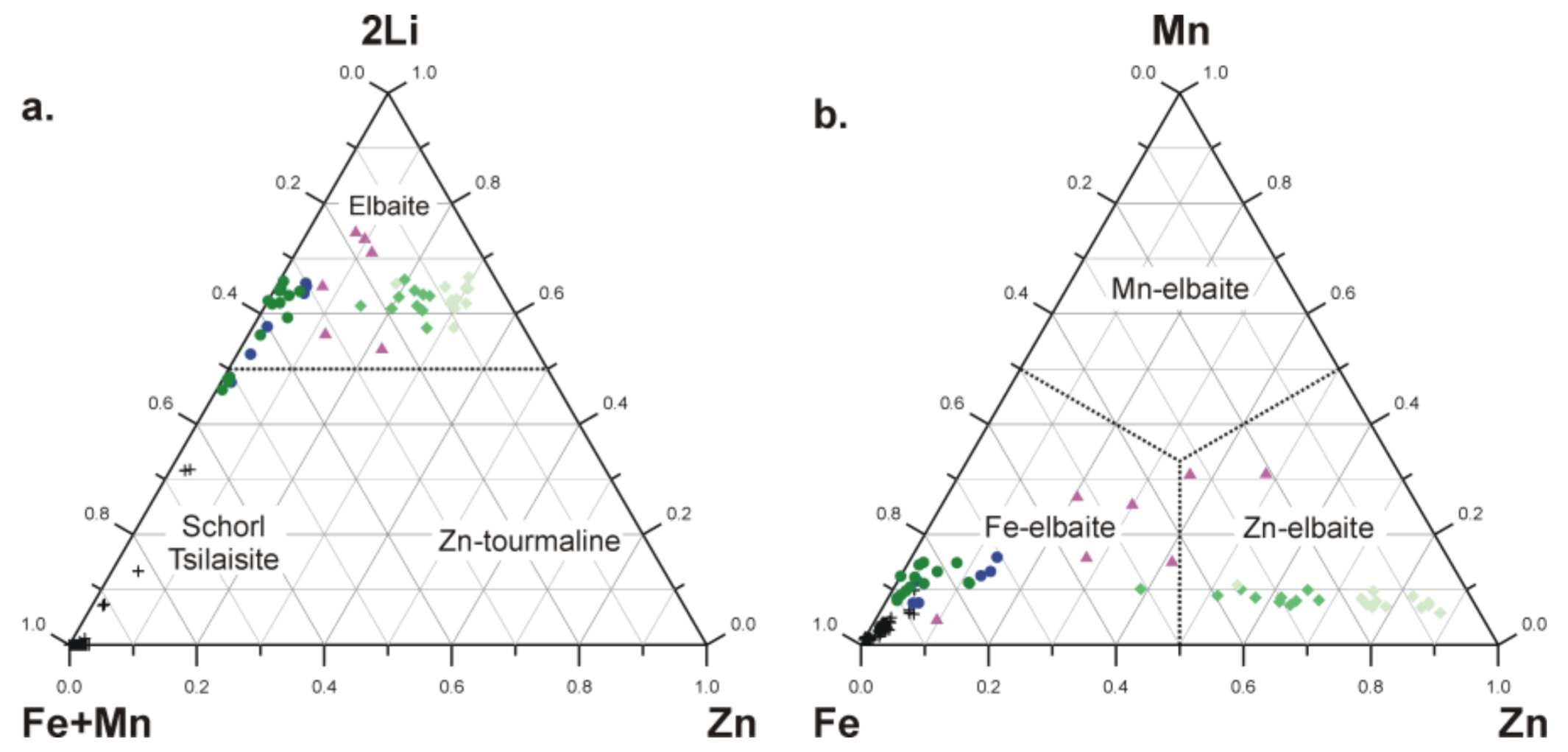
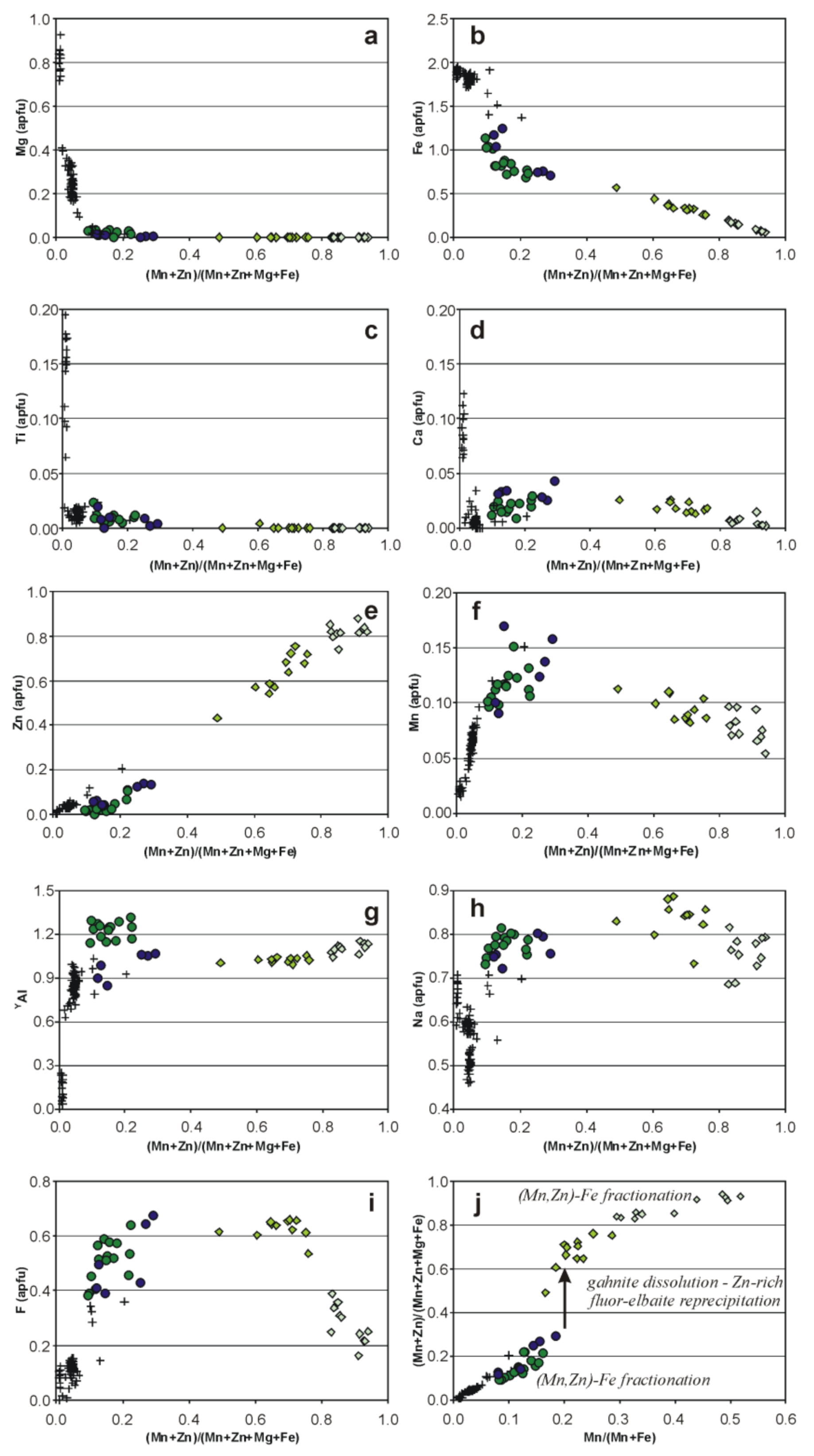
5.5. Compositional Relationships
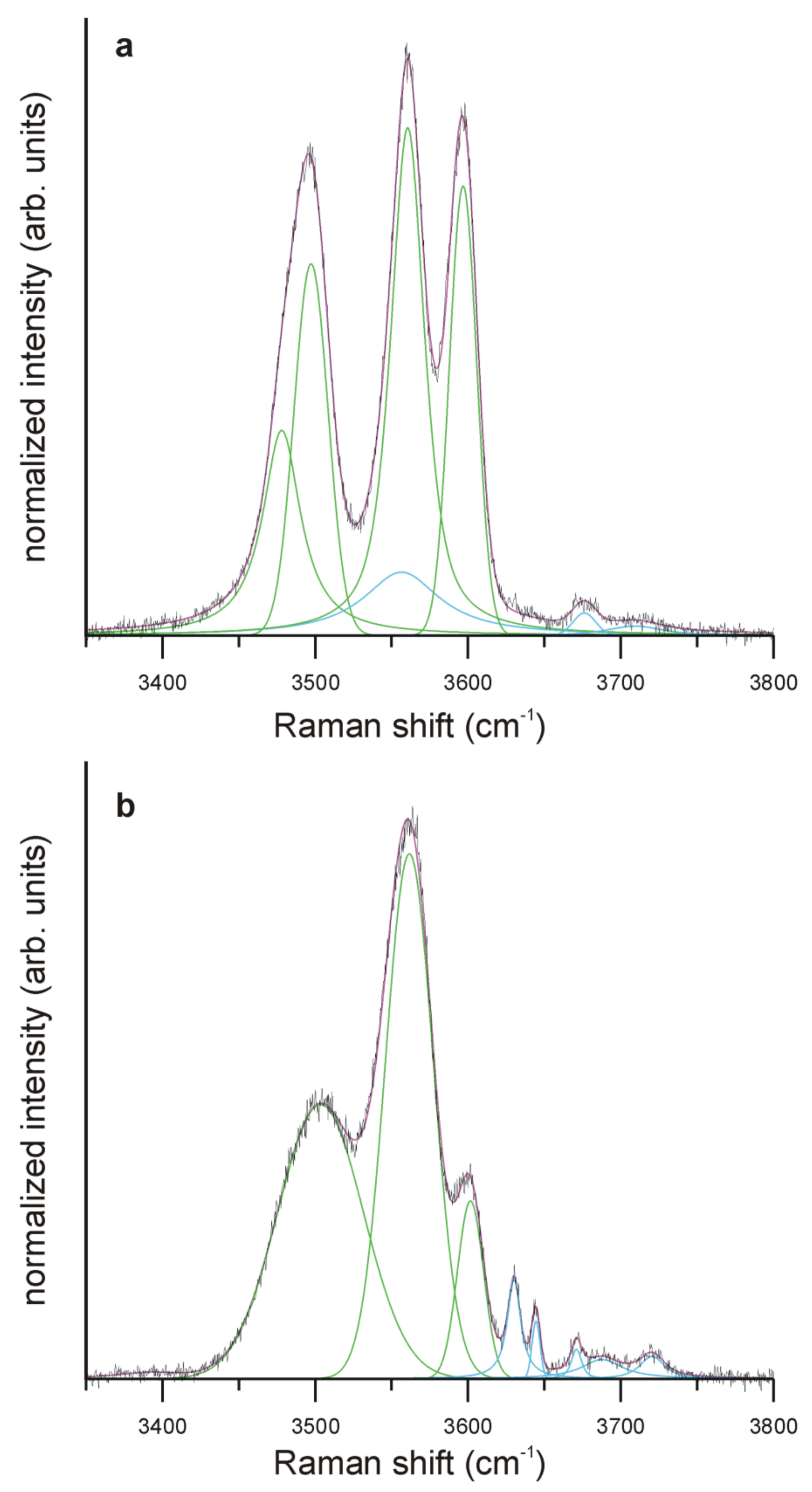
5.6. Raman Spectroscopy
6. Discussion: Is Natural Zn-Dominant Tourmaline Possible?
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Henry, D.J.; Novák, M.; Hawthorne, F.C.; Ertl, A.; Dutrow, B.L.; Uher, P.; Pezzotta, F. Nomenclature of the tourmaline-supergroup minerals. Am. Mineral. 2011, 96, 895–913. [Google Scholar] [CrossRef]
- Sokolov, M.; Martin, R.F. A Pb-dominant member of the tourmaline group, Minh Tien granitic pegmatite, Luc Yen District, Vietnam. Estudos Geol. 2009, 19, 352–353. [Google Scholar]
- Lottermoser, B.G.; Plimer, I.R. Chemical variation in tourmalines, Umberatana, South Australia. N. Jahrb. Mineral. Mon. 1987, 7, 314–326. [Google Scholar]
- Yu, J.M.; Jiang, S.Y. Chemical composition of tourmaline from the Yunlong tin deposit, Yunnan, China: Implications for ore genesis and mineral exploration. Miner. Petrol. 2003, 77, 67–84. [Google Scholar] [CrossRef]
- Johnson, M.L.; Wentzell, C.Y.; Elen, S. Multicolored bismuth-bearing tourmaline from Lundazi, Zambia. Gems Gemol. 1997, 33, 204–211. [Google Scholar] [CrossRef]
- Vereshchagin, O.S.; Rozhdestvenskaya, I.V.; Frank-Kamenetskaya, O.V.; Zolotarev, A.A.; Mashkovtsev, R.I. Crystal chemistry of Cu-bearing tourmalines. Am. Mineral. 2013, 98, 1610–1616. [Google Scholar] [CrossRef]
- Baksheev, I.A.; Kudryavtseva, O.E. Nickeloan tourmaline from the Berezovskoe gold deposit, Middle Urals, Russia. Can. Mineral. 2004, 42, 1065–1078. [Google Scholar] [CrossRef]
- Van Hinsberg, V.J.; Henry, D.J.; Dutrow, B.L. Tourmaline as a petrologic forensic mineral: A unique recorder of its geologic past. Elements 2011, 7, 327–332. [Google Scholar] [CrossRef]
- Grice, J.D.; Ercit, T.S. Ordering of Fe and Mg in the tourmaline crystal structure: The correct formula. N. Jahrb. Miner. Abh. 1993, 165, 245–266. [Google Scholar]
- Federico, M.; Andreozzi, G.B.; Lucchesi, S.; Graziani, G. Compositional variation of tourmaline in the granitic pegmatite dykes of the Cruzeiro Mine, Minas Gerais, Brazil. Can. Mineral. 1998, 36, 415–431. [Google Scholar]
- Hawkins, K.D.; MacKinnon, I.D.R.; Schneeberger, H. Influence of chemistry on the pyroelectric effect in tourmaline. Am. Mineral. 1995, 80, 491–501. [Google Scholar] [CrossRef]
- Adusumilli, M.S.; Castro, C.; Bhaskara Rao, A. Blue and green gem tourmalines from Gregório pegmatite, Rio Grande do Norte State, Brazil. In Proceedings of the 16th General Meeting, IMA, Pisa, Italy, 4–9 September 1994; pp. 1–13. [Google Scholar]
- Soares, D.R. Contribuição à Petrologia de Pegmatitos Mineralizados em Elementos Raros e em Elbeítas Gemológicas da Província Pegmatítica da Borborema, Nordeste do Brasil. Ph.D. Thesis, UFPE, Recife, Brazil, 2004. [Google Scholar]
- Ferreira, A.C.M.; Ferreira, V.P.; Soares, D.R.; Vilarroel-Leo, H.S. Chemical and mineralogical characterization of elbaite from the Alto Quixaba pegmatite, Seridó Province, NE Brazil. An. Acad. Bras. Ciênc. 2005, 77, 729–743. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jedwab, J. Tourmaline zincifere dans une pegmatite de Muika (Congo). Bull. Soc. Belge Géol. Paléontol. D’hydrol. 1962, 71, 132–135. [Google Scholar]
- Sokolov, P.B.; Gorskaya, M.G.; Kretser, Y.L. Zinc-bearing tourmalines from rare-metal pegmatites. Zap. Vses. Mineral. Obshchest. 1988, 117, 70–74. (In Russian) [Google Scholar]
- Szuszkiewicz, A.; Szełęg, E.; Pieczka, A.; Ilnicki, S.; Nejbert, K.; Turniak, K.; Banach, M.; Łodziński, M.; Różniak, R.; Michałowski, P. The Julianna pegmatite vein system at the Piława Górna mine, Góry Sowie Block, SW Poland–preliminary data on geology and descriptive mineralogy. Geol. Q. 2013, 57, 467–484. [Google Scholar] [CrossRef]
- Pieczka, A.; Łodziński, M.; Szełęg, E.; Ilnicki, S.; Nejbert, K.; Szuszkiewicz, A.; Turniak, K.; Banach, M.; Michałowski, P.; Różniak, R. The Sowie Mts. pegmatites (Lower Silesia, SW Poland): A current knowledge. Acta Mineral. Petrogr. Szeged 2012, 7, 105–106. [Google Scholar]
- Pieczka, A.; Szuszkiewicz, A.; Szełęg, E.; Nejbert, K.; Łodziński, M.; Ilnicki, S.; Turniak, K.; Banach, M.; Hołub, W.; Michałowski, P.; et al. (Fe,Mn)–(Ti,Sn)–(Nb,Ta) oxide assemblage in a little fractionated portion of a mixed (NYF + LCT) pegmatite from Piława Górna, the Sowie Mts. block, SW Poland. J. Geosci. 2013, 58, 91–112. [Google Scholar] [CrossRef]
- Pieczka, A.; Szuszkiewicz, A.; Szełęg, E.; Janeczek, J.; Nejbert, K. Granitic pegmatites of the Polish part of the Sudetes (NE Bohemian massif, SW Poland). In Fieldtrip Guidebook, Proceedings of the 7th International Symposium on Granitic Pegmatites, PEG 2015, Książ, Poland, 17–19 June 2015; Gadas, P., Novák, M., Szuszkiewicz, A., Cempírek, J., Eds.; Czech Republic: Brno, Czech Republic, 2015; pp. 73–103. [Google Scholar]
- Pieczka, A.; Szuszkiewicz, A.; Szełęg, E.; Ilnicki, S.; Nejbert, K.; Turniak, K. Samarskite-group minerals and alteration products: An example from the Julianna pegmatitic system, Piława Górna, SW Poland. Can. Mineral. 2014, 52, 303–319. [Google Scholar] [CrossRef]
- Pieczka, A.; Szełęg, E.; Szuszkiewicz, A.; Gołębiowska, B.; Zelek, S.; Ilnicki, S.; Nejbert, K.; Turniak, K. Cs-bearing beryl evolving to pezzottaite from the Julianna pegmatitic system, SW Poland. Can. Mineral. 2016, 54, 115–124. [Google Scholar] [CrossRef]
- Szuszkiewicz, A.; Pieczka, A.; Szełęg, E.; Turniak, K.; Ilnicki, S.; Nejbert, K. The euxenite-group minerals and products of their alteration in the hybrid Julianna granitic pegmatite, Piława Górna, Sudetes, southwestern Poland. Can. Mineral. 2016, 54, 879–898. [Google Scholar] [CrossRef]
- Pieczka, A.; Hawthorne, F.C.; Cooper, M.A.; Szełęg, E.; Szuszkiewicz, A.; Turniak, K.; Nejbert, K.; Ilnicki, S. Pilawite-(Y), Ca2(Y,Yb)2[Al4(SiO4)4O2(OH)2], a new mineral from the Piława Górna granitic pegmatite, southwestern Poland: Mineralogical data, crystal structure and association. Mineral. Mag. 2015, 79, 1143–1157. [Google Scholar] [CrossRef]
- Szełęg, E.; Zuzens, B.; Hawthorne, F.C.; Pieczka, A.; Szuszkiewicz, A.; Turniak, K.; Nejbert, K.; Ilnicki, S.S.; Friis, H.; Makovicky, E.; et al. Bohseite, ideally Ca4Be4Si9O24(OH)4, from the Piława Górna quarry, the Góry Sowie Block, SW Poland. Mineral. Mag. 2017, 81, 35–46. [Google Scholar] [CrossRef]
- Pieczka, A.; Hawthorne, F.C.; Ma, C.; Rossman, G.; Szełęg, E.; Szuszkiewicz, A.; Turniak, K.; Nejbert, K.; Ilnicki, S.S. Żabińskiite, ideally Ca(Al0.5Ta0.5)(SiO4)O, a new mineral of the titanite group from the Piława Górna pegmatite, the Góry Sowie Block, southwestern Poland. Mineral. Mag. 2017, 81, 591–610. [Google Scholar] [CrossRef]
- Turniak, K.; Pieczka, A.; Kennedy, A.K.; Szełęg, E.; Ilnicki, S.; Nejbert, K.; Szuszkiewicz, A. Crystallisation age of the Julianna pegmatite system (Góry Sowie Block, NE margin of the Bohemian massif): Evidence from U–Th–Pb SHRIMP monazite and CHIME uraninite studies. In Fieldtrip Guidebook, Proceedings of the 7th International Symposium on Granitic Pegmatites, PEG 201, Książ, Poland, 17–19 June 2015; Gadas, P., Novák, M., Szuszkiewicz, A., Cempírek, J., Eds.; Czech Republic: Brno, Czech Republic, 2015; pp. 111–112. [Google Scholar]
- Van Breemen, O.; Bowes, D.R.; Aftalion, M.; Żelaźniewicz, A. Devonian tectonothermal activity in the Sowie Góry gneissic block, Sudetes, southwestern Poland: Evidence from Rb–Sr and U–Pb isotopic studies. Ann. Geol. Soc. Pol. 1988, 58, 3–10. [Google Scholar]
- Timmermann, H.; Parrish, R.R.; Noble, S.R.; Kryza, R. New U–Pb monazite and zircon data from the Sudetes Mountains in SW Poland: Evidence for a single-cycle Variscan orogeny. J. Geol. Soc. 2000, 157, 265–268. [Google Scholar] [CrossRef]
- Kryza, R.; Fanning, C.M. Devonian deep-crustal metamorphism and exhumation in the Variscan Orogen: Evidence from SHRIMP zircon ages from the HT–HP granulites and migmatites of the Góry Sowie (Polish Sudetes). Geodin. Acta 2007, 20, 159–176. [Google Scholar] [CrossRef]
- Müller, A.; Szuszkiewicz, A.; Ilnicki, S.; Nejbert, K. Quartz chemistry of the Julianna pegmatites and their wall rocks, Piława Górna, Poland: Implications for the origin of pegmatite melts. In Abstracts, Proceedings of the 8th International Symposium on Granitic Pegmatites, PEG2017, Kristiansand, Norway, 13–15 June 2017; Müller, A., Rosing-Schow, N., Eds.; Norsk Geologisk Forening: Trondheim, Norway, 2017; pp. 84–88. [Google Scholar]
- Pouchou, I.L.; Pichoir, F. “PAP” (phi-rho-z) procedure for improved quantitative microanalysis. In Microbeam Analysis; Armstrong, I.T., Ed.; San Francisco Press: San Francisco, CA, USA, 1985; pp. 104–106. [Google Scholar]
- Pesquera, A.; Gil-Crespo, P.P.; Torres-Ruiz, F.; Torres-Ruiz, J.; Roda-Robles, E. A multiple regression method for estimating Li in tourmaline from electron microprobe analyses. Mineral. Mag. 2016, 80, 1129–1133. [Google Scholar] [CrossRef]
- SPECTRA-CALC V2.23. Galactic Industries Corp.: Main St., NH, USA.
- Whitney, D.L.; Evans, B.W. Abbreviations for names of rock-forming minerals. Am. Mineral. 2010, 95, 185–187. [Google Scholar] [CrossRef]
- Alvarez, M.A.; Coy-Yli, R. Raman spectra of tourmaline. Spectrochim. Acta A 1978, 34, 899–908. [Google Scholar] [CrossRef]
- Gasharova, B.; Mihailova, B.; Konstantinov, L. Raman spectra of various types of tourmalines. Eur. J. Mineral. 1997, 9, 935–940. [Google Scholar] [CrossRef]
- McKeown, D.A. Raman spectroscopy, vibrational analysis, and heating of buergerite tourmaline. Phys. Chem. Miner. 2008, 35, 259–270. [Google Scholar] [CrossRef]
- Hoang, L.H.; Hien, N.T.M.; Chen, X.B.; Minh, N.V.; Yang, I.-S. Raman spectroscopis study of various types of tourmalines. J. Raman Spectrosc. 2011, 42, 1443–1446. [Google Scholar] [CrossRef]
- Skoggby, H.; Bosi, F.; Lazor, P. Short-range order in tourmaline: A vibrational spectroscopic approach to elbaite. Phys. Chem. Miner. 2012, 39, 811–816. [Google Scholar] [CrossRef]
- Zhao, C.; Liao, L.; Xia, Z.; Sun, X. Temperature-dependent Raman and infrared spectroscopy study on iron-magnesium tourmalines with different Fe content. Vib. Spec. 2012, 62, 28–34. [Google Scholar] [CrossRef]
- Fantini, C.; Tavares, M.C.; Krambrock, K.; Moreira, R.L.; Righi, A. Raman and infrared study of hydroxyl sites in natural uvite, fluor-uvite, magnesio-foitite, dravite and elbaite tourmalines. Phys. Chem. Miner. 2014, 41, 247–254. [Google Scholar] [CrossRef]
- Berryman, E.J.; Wunder, B.; Ertl, A.; Koch-Müller, M.; Rhede, D.; Scheidl, K.; Giester, G.; Heinrich, W. Influence of the X-site composition tourmaline’s crystal structure: Investigation of synthetic K-dravite, dravite, oxy-uvite, and magnesio-foitite using SREF and Raman spectroscopy. Phys. Chem. Miner. 2016, 43, 83–102. [Google Scholar] [CrossRef]
- Watenphul, A.; Burgdorf, M.; Schlüter, J.; Horn, I.; Malcherek, T.; Mihailova, B. Exploring the potential of Raman spectroscopy for crystallochemical analyses of complex hydrous silicates: II. Tourmalines. Am. Mineral. 2016, 101, 970–985. [Google Scholar] [CrossRef]
- Mercurio, M.; Rossi, M.; Izzo, F.; Cappelletti, P.; Germinario, C.; Grifa, C.; Petrelli, M.; Vergara, A.; Langella, A. The characterization of natural gemstones using non-invasive FT-IR spectroscopy: New data on tourmalines. Talanta 2018, 178, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.B.; Hawthorne, F.C. Characterization of tourmaline crystals by Rietveld and single-crystal structure refinement: A comparative study. Geosci. J. 2002, 6, 237–243. [Google Scholar] [CrossRef]
- Hawthorne, F.C. Structural mechanism for light-element variations in tourmaline. Can. Mineral. 1996, 34, 123–132. [Google Scholar]
- Černý, P.; Meintzer, R.E.; Anderson, A.J. Extreme fractionation in rare-element granitic pegmatites: Selected examples of data and mechanism. Can. Mineral. 1985, 23, 381–421. [Google Scholar]



| apfu | Tur IA Sample 224 | Tur IB Sample 226 | |
|---|---|---|---|
| n = 13 | n = 13 | n = 54 | |
| SiO2 | 35.21(25) | 35.11(27) | 34.84(28) |
| TiO2 | 1.11(30) | 0.09(3) | 0.10(3) |
| B2O3(calc.) | 10.25(6) | 10.26(4) | 10.19(6) |
| Al2O3 | 30.86(38) | 34.28(48) | 34.38(32) |
| FeO | 13.27(30) | 13.04(31) | 12.66(29) |
| MnO | 0.14(3) | 0.39(18) | 0.45(6) |
| MgO | 3.19(23) | 1.03(43) | 0.89(18) |
| ZnO | 0.07(06) | 0.30(10) | 0.27(4) |
| CaO | 0.48(10) | 0.06(6) | 0.03(2) |
| Na2O | 2.01(10) | 1.77(10) | 1.64(14) |
| K2O | 0.04(2) | 0.03(1) | 0.04(1) |
| H2O | 3.25(6) | 3.16(11) | 3.07(5) |
| F2 | 0.14(7) | 0.14(8) | 0.23(3) |
| −O=F2 | −0.06(3) | −0.06(4) | −0.10(1) |
| Total | 99.96(56) | 99.60(44) | 98.68(59) |
| apfu | |||
| XNa+ | 0.660(33) | 0.582(33) | 0.544(48) |
| XK+ | 0.009(4) | 0.006(2) | 0.008(1) |
| XCa2+ | 0.087(18) | 0.010(11) | 0.006(3) |
| X☐ | 0.244(42) | 0.402(40) | 0.443(49) |
| ΣX | 1.000 | 1.000 | 1.000 |
| Mg2+ | 0.806(57) | 0.261(107) | 0.227(46) |
| Fe2+ | 1.882(43) | 1.847(41) | 1.807(43) |
| Mn2+ | 0.020(4) | 0.056(26) | 0.064(8) |
| Zn2+ | 0.009(8) | 0.038(12) | 0.034(5) |
| Al3+ | 6.142(63) | 6.787(108) | 6.912(37) |
| Ti4+ | 0.142(39) | 0.011(4) | 0.013(4) |
| Σ(Y + Z) | 9.000 | 9.000 | 9.000 |
| BB3+ | 3.000 | 3.000 | 3.000 |
| TSi4+ | 5.973(22) | 5.945(36) | 5.943(34) |
| TAl3+ | 0.027(22) | 0.055(36) | 0.057(34) |
| ΣT | 6.000 | 6.000 | 6.000 |
| O | 27.000 | 27.000 | 27.000 |
| VOH− | 3.000 | 3.000 | 3.000 |
| WO2− | 0.241(53) | 0.363(85) | 0.386(51) |
| WOH− | 0.683(62) | 0.565(117) | 0.491(51) |
| WF− | 0.076(35) | 0.073(45) | 0.123(16) |
| ΣW | 1.000 | 1.000 | 1.000 |
| Mn# | 0.010(2) | 0.029(14) | 0.034(4) |
| Zn# | 0.005(4) | 0.020(6) | 0.019(3) |
| Zn1# | 0.011(2) | 0.043(17) | 0.046(4) |
| wt % | Tur IIA | Tur IIB | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 223/9 | 217/3 | 217/4 | 217/6 | 217/5 | 112/28 | 112/23 | 112/16 | 112/17 | 112/20 | |
| SiO2 | 36.36 | 35.68 | 37.00 | 36.77 | 36.73 | 36.25 | 36.64 | 37.03 | 37.13 | 37.45 |
| TiO2 | 0.03 | 0.15 | 0.07 | 0.07 | 0.02 | 0.19 | 0.07 | 0.08 | 0.08 | 0.08 |
| B2O3(calc.) | 10.53 | 10.34 | 10.72 | 10.65 | 10.64 | 10.50 | 10.61 | 10.73 | 10.76 | 10.85 |
| Al2O3 | 35.14 | 35.47 | 36.12 | 36.71 | 36.64 | 36.60 | 37.79 | 37.97 | 38.09 | 38.74 |
| FeO | 10.96 | 9.95 | 8.64 | 5.44 | 5.52 | 8.24 | 7.49 | 6.53 | 5.67 | 5.10 |
| MnO | 1.24 | 0.69 | 0.73 | 0.90 | 0.99 | 0.73 | 0.69 | 0.85 | 0.82 | 0.97 |
| MgO | 0.05 | 0.07 | 0.08 | 0.03 | 0.02 | 0.13 | 0.13 | 0.10 | 0.11 | 0.13 |
| ZnO | 0.49 | 0.56 | 0.49 | 1.03 | 1.17 | 0.17 | 0.13 | 0.38 | 0.93 | 0.57 |
| CaO | 0.03 | 0.12 | 0.18 | 0.17 | 0.15 | 0.60 | 0.07 | 0.10 | 0.12 | 0.15 |
| Li2O(calc.) | 0.61 | 0.54 | 1.14 | 1.43 | 1.38 | 0.82 | 0.80 | 1.03 | 1.11 | 1.18 |
| Na2O | 1.74 | 2.17 | 2.38 | 2.54 | 2.51 | 2.28 | 2.35 | 2.52 | 2.41 | 2.47 |
| K2O | 0.02 | 0.04 | 0.05 | 0.02 | 0.00 | 0.03 | 0.02 | 0.00 | 0.03 | 0.00 |
| H2O(calc.) | 3.49 | 2.87 | 3.32 | 3.28 | 3.08 | 2.74 | 2.80 | 2.82 | 2.89 | 2.95 |
| F2 | 0.27 | 0.61 | 0.80 | 0.83 | 1.25 | 0.73 | 0.75 | 1.03 | 1.05 | 0.90 |
| −O=F2 | −0.12 | −0.26 | −0.34 | −0.35 | −0.53 | −0.31 | −0.32 | −0.43 | −0.44 | −0.38 |
| Total | 100.85 | 99.00 | 101.37 | 99.53 | 99.58 | 99.69 | 100.04 | 100.73 | 100.74 | 101.15 |
| apfu | ||||||||||
| XNa+ | 0.558 | 0.708 | 0.750 | 0.802 | 0.794 | 0.731 | 0.746 | 0.793 | 0.753 | 0.767 |
| XK+ | 0.005 | 0.008 | 0.010 | 0.004 | 0.000 | 0.006 | 0.004 | 0.000 | 0.006 | 0.000 |
| XCa2+ | 0.006 | 0.021 | 0.031 | 0.029 | 0.026 | 0.106 | 0.012 | 0.018 | 0.020 | 0.025 |
| X⃞ | 0.431 | 0.264 | 0.209 | 0.165 | 0.180 | 0.157 | 0.239 | 0.189 | 0.220 | 0.207 |
| ΣX | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Li+ | 0.404 | 0.365 | 0.741 | 0.941 | 0.906 | 0.543 | 0.528 | 0.670 | 0.721 | 0.761 |
| Mg2+ | 0.013 | 0.018 | 0.018 | 0.000 | 0.005 | 0.031 | 0.031 | 0.024 | 0.026 | 0.031 |
| Fe2+ | 1.513 | 1.400 | 1.171 | 0.742 | 0.754 | 1.141 | 1.026 | 0.885 | 0.767 | 0.684 |
| Mn2+ | 0.173 | 0.098 | 0.101 | 0.124 | 0.137 | 0.102 | 0.096 | 0.116 | 0.112 | 0.131 |
| Zn2+ | 0.059 | 0.069 | 0.058 | 0.124 | 0.141 | 0.021 | 0.016 | 0.045 | 0.111 | 0.067 |
| Al3+ | 6.834 | 7.030 | 6.902 | 7.060 | 7.054 | 7.139 | 7.293 | 7.250 | 7.253 | 7.315 |
| Ti4+ | 0.003 | 0.019 | 0.008 | 0.009 | 0.003 | 0.024 | 0.009 | 0.010 | 0.010 | 0.010 |
| Σ(Y + Z) | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 |
| BB3+ | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 |
| TSi4+ | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 |
| O | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 |
| VOH− | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 |
| WO2− | 0.011 | 0.462 | 0.000 | 0.000 | 0.000 | 0.593 | 0.555 | 0.429 | 0.351 | 0.392 |
| WOH− | 0.846 | 0.214 | 0.589 | 0.570 | 0.355 | 0.024 | 0.054 | 0.044 | 0.113 | 0.153 |
| WF− | 0.143 | 0.324 | 0.411 | 0.430 | 0.645 | 0.382 | 0.391 | 0.527 | 0.536 | 0.455 |
| ΣW | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Mn# | 0.103 | 0.066 | 0.079 | 0.143 | 0.154 | 0.082 | 0.086 | 0.116 | 0.127 | 0.161 |
| Zn# | 0.038 | 0.047 | 0.047 | 0.143 | 0.158 | 0.018 | 0.016 | 0.048 | 0.127 | 0.089 |
| Zn1# | 0.132 | 0.106 | 0.118 | 0.251 | 0.268 | 0.095 | 0.096 | 0.151 | 0.220 | 0.217 |
| Sch | F-sch | Elb | Elb | F-elb | Dh | Dh | F-elb | F-elb | F-elb | |
| wt % | Tur IB | Tur IB1 | Tur IIIA | Tur IIIB | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 225/1 | 225/2 | 225/3 | 225/4 | 225/5 | 225/6 | 225/7 | 225/8 | 225/9 | 225/10 | |
| SiO2 | 34.96 | 35.83 | 36.73 | 37.08 | 37.03 | 37.05 | 36.61 | 37.25 | 36.79 | 37.11 |
| TiO2 | 0.18 | 0.06 | 0.03 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| B2O3(calc.) | 10.13 | 10.38 | 10.64 | 10.74 | 10.73 | 10.73 | 10.61 | 10.79 | 10.66 | 10.75 |
| Al2O3 | 33.57 | 35.08 | 36.48 | 36.98 | 36.83 | 37.17 | 36.86 | 37.67 | 36.99 | 37.05 |
| FeO | 13.27 | 9.76 | 3.21 | 1.92 | 2.40 | 1.26 | 1.17 | 0.62 | 0.51 | 0.71 |
| MnO | 0.82 | 1.06 | 0.72 | 0.76 | 0.68 | 0.52 | 0.60 | 0.48 | 0.54 | 0.69 |
| MgO | 0.18 | 0.06 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| ZnO | 0.93 | 1.66 | 4.73 | 5.68 | 6.32 | 6.67 | 6.72 | 6.87 | 6.97 | 7.37 |
| CaO | 0.03 | 0.06 | 0.10 | 0.10 | 0.08 | 0.02 | 0.04 | 0.02 | 0.02 | 0.08 |
| Li2O(calc.) | 0.00 | 0.49 | 1.32 | 1.39 | 1.22 | 1.33 | 1.25 | 1.37 | 1.38 | 1.34 |
| Na2O | 1.99 | 2.15 | 2.52 | 2.62 | 2.33 | 2.43 | 2.16 | 2.33 | 2.50 | 2.49 |
| K2O | 0.03 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.02 | 0.00 | 0.00 | 0.00 |
| H2O(calc.) | 2.80 | 2.97 | 3.12 | 3.14 | 3.09 | 3.39 | 3.33 | 3.50 | 3.48 | 3.56 |
| F2 | 0.52 | 0.68 | 1.17 | 1.19 | 1.28 | 0.65 | 0.69 | 0.47 | 0.42 | 0.32 |
| −O=F2 | −0.22 | −0.29 | −0.49 | −0.50 | −0.54 | −0.28 | −0.29 | −0.20 | −0.18 | −0.13 |
| Total | 99.19 | 99.95 | 100.26 | 101.10 | 101.45 | 100.96 | 99.77 | 101.16 | 100.08 | 101.32 |
| apfu | ||||||||||
| XNa+ | 0.664 | 0.698 | 0.799 | 0.822 | 0.733 | 0.764 | 0.688 | 0.727 | 0.791 | 0.779 |
| XK+ | 0.007 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.004 | 0.000 | 0.000 | 0.000 |
| XCa2+ | 0.005 | 0.011 | 0.017 | 0.016 | 0.013 | 0.004 | 0.006 | 0.003 | 0.003 | 0.015 |
| X⃞ | 0.324 | 0.292 | 0.184 | 0.162 | 0.254 | 0.232 | 0.302 | 0.269 | 0.206 | 0.206 |
| ΣX | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Li+ | 0.000 | 0.331 | 0.865 | 0.906 | 0.792 | 0.867 | 0.824 | 0.884 | 0.906 | 0.869 |
| Mg2+ | 0.047 | 0.015 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Fe2+ | 1.904 | 1.367 | 0.439 | 0.260 | 0.326 | 0.170 | 0.161 | 0.084 | 0.075 | 0.098 |
| Mn2+ | 0.120 | 0.151 | 0.099 | 0.104 | 0.094 | 0.071 | 0.083 | 0.065 | 0.070 | 0.094 |
| Zn2+ | 0.117 | 0.205 | 0.570 | 0.679 | 0.755 | 0.798 | 0.813 | 0.817 | 0.840 | 0.880 |
| Al3+ | 6.789 | 6.924 | 7.023 | 7.051 | 7.033 | 7.095 | 7.119 | 7.150 | 7.109 | 7.061 |
| Ti4+ | 0.023 | 0.007 | 0.004 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Σ(Y + Z) | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 | 9.000 |
| BB3+ | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 |
| TSi4+ | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 | 6.000 |
| O | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 | 27.000 |
| VOH− | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 | 3.000 |
| WO2− | 0.515 | 0.327 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| WOH− | 0.201 | 0.313 | 0.398 | 0.389 | 0.344 | 0.665 | 0.643 | 0.759 | 0.785 | 0.837 |
| WF− | 0.284 | 0.360 | 0.602 | 0.611 | 0.656 | 0.335 | 0.357 | 0.241 | 0.215 | 0.163 |
| ΣW | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Mn# | 0.059 | 0.099 | 0.184 | 0.286 | 0.223 | 0.293 | 0.340 | 0.439 | 0.517 | 0.495 |
| Zn# | 0.058 | 0.130 | 0.565 | 0.723 | 0.699 | 0.824 | 0.835 | 0.907 | 0.923 | 0.901 |
| Zn1# | 0.108 | 0.205 | 0.604 | 0.751 | 0.723 | 0.836 | 0.848 | 0.913 | 0.929 | 0.910 |
| O-sch | F-sch | F-elb | F-elb | F-elb | Elb | Elb | Elb | Elb | Elb | |
| Tur IB | Tur IIIA | ||||||
|---|---|---|---|---|---|---|---|
| Fe*2+:Zn << Fe + Mn | Fe*2+:Zn >> Fe + Mn | ||||||
| Band (cm−1) | Rel. int. (%) | Calc. int. (%) | Assignment | Band (cm−1) | Rel. int. (%) | Calc. int. (%) | Assignment |
| 3478 | 17 | 31 | VOH-YAlZAlZAl-X☐ | ||||
| 3503 | 41 | 28 | VOH-YAl+ZAlZAl | 3497 | 18 | VOH-YAlZAlZAl-XNa | |
| 3556 | 11 | (9) | WOH- ……. -XNa | ||||
| 3562 | 43 | 53 | VOH-YFe*2+ZAlZAl | 3561 | 34 | 31 | VOH-YFe*2+ZAlZAl |
| 3602 | 8 | 10 | VOH-YLiZAlZAl | 3597 | 18 | 27 | VOH-YLiZAlZAl |
| 3630 | 3 | 2 | WOH-YFe*2+YAlYAl-X☐ | - | - | - | |
| 3644 | 1 | 1 | WOH-YFe*2+YFe*2+YAl-X☐ | - | - | - | WOH-YFe*2+YFe*2+YAl-X☐ |
| 3671 | 1 | 1 | WOH-YLiYFe*2+YAl-X☐ | 3676 | 1 | 1 | WOH-YLiYFe*2+YAl-X☐ |
| 3688 | 2 | 2 | WOH-YFe*2+YFe*2+YAl-XNa | - | - | - | WOH-YFe*2+YFe*2+YAl-XNa |
| 3720 | 1 | 2 | WOH-YFe*2+YFe*2+YFe*2+-XNa | 3709 | 1 | 1 | WOH-YFe*2+YFe*2+YFe*2+-XNa |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pieczka, A.; Gołębiowska, B.; Jeleń, P.; Włodek, A.; Szełęg, E.; Szuszkiewicz, A. Towards Zn-Dominant Tourmaline: A Case of Zn-Rich Fluor-Elbaite and Elbaite from the Julianna System at Piława Górna, Lower Silesia, SW Poland. Minerals 2018, 8, 126. https://doi.org/10.3390/min8040126
Pieczka A, Gołębiowska B, Jeleń P, Włodek A, Szełęg E, Szuszkiewicz A. Towards Zn-Dominant Tourmaline: A Case of Zn-Rich Fluor-Elbaite and Elbaite from the Julianna System at Piława Górna, Lower Silesia, SW Poland. Minerals. 2018; 8(4):126. https://doi.org/10.3390/min8040126
Chicago/Turabian StylePieczka, Adam, Bożena Gołębiowska, Piotr Jeleń, Adam Włodek, Eligiusz Szełęg, and Adam Szuszkiewicz. 2018. "Towards Zn-Dominant Tourmaline: A Case of Zn-Rich Fluor-Elbaite and Elbaite from the Julianna System at Piława Górna, Lower Silesia, SW Poland" Minerals 8, no. 4: 126. https://doi.org/10.3390/min8040126
APA StylePieczka, A., Gołębiowska, B., Jeleń, P., Włodek, A., Szełęg, E., & Szuszkiewicz, A. (2018). Towards Zn-Dominant Tourmaline: A Case of Zn-Rich Fluor-Elbaite and Elbaite from the Julianna System at Piława Górna, Lower Silesia, SW Poland. Minerals, 8(4), 126. https://doi.org/10.3390/min8040126





