Nöggerathite-(Ce), (Ce,Ca)2Zr2(Nb,Ti)(Ti,Nb)2Fe2+O14, a New Zirconolite-Related Mineral from the Eifel Volcanic Region, Germany
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. General Appearance and Mechanical Properties
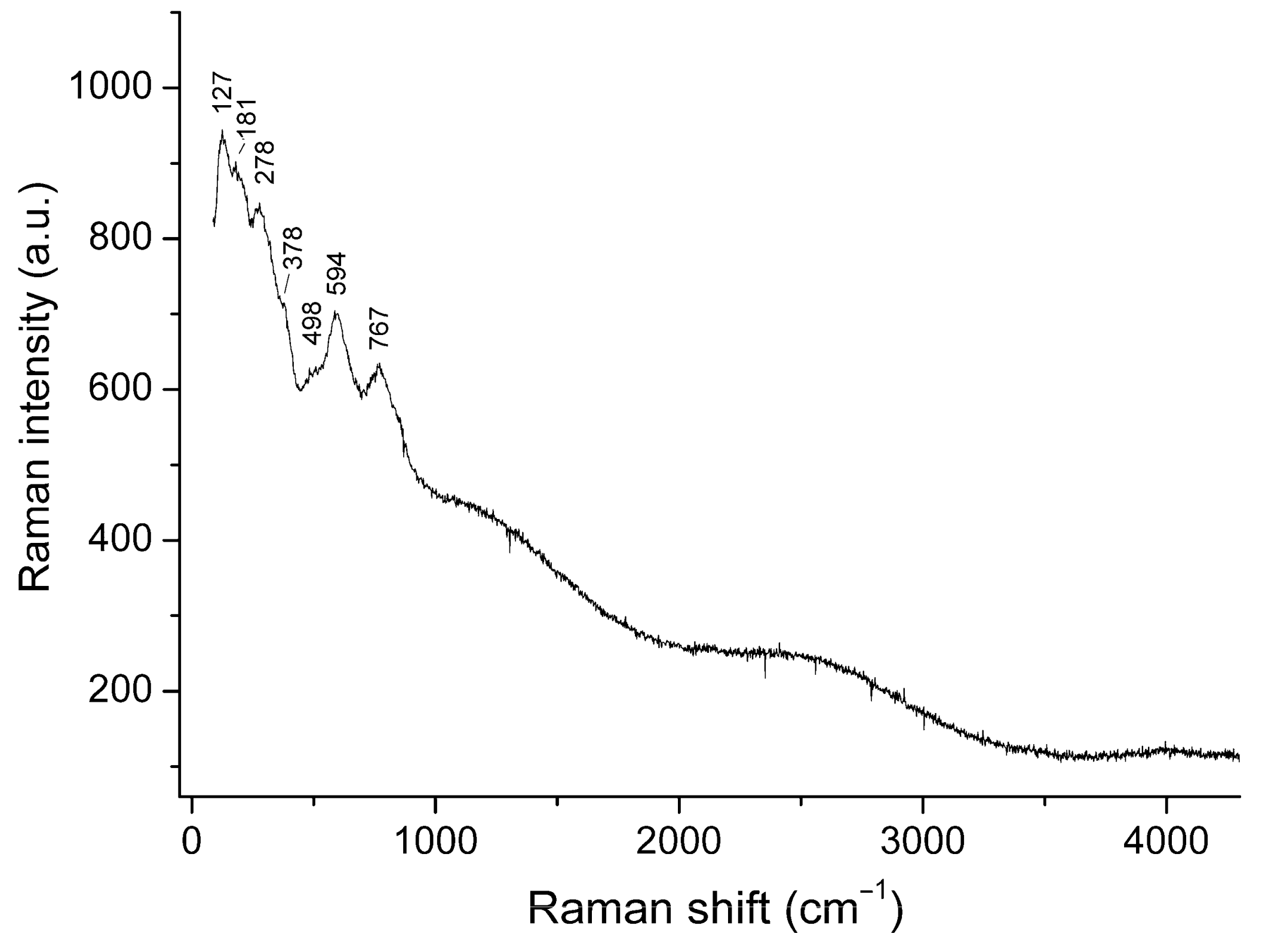
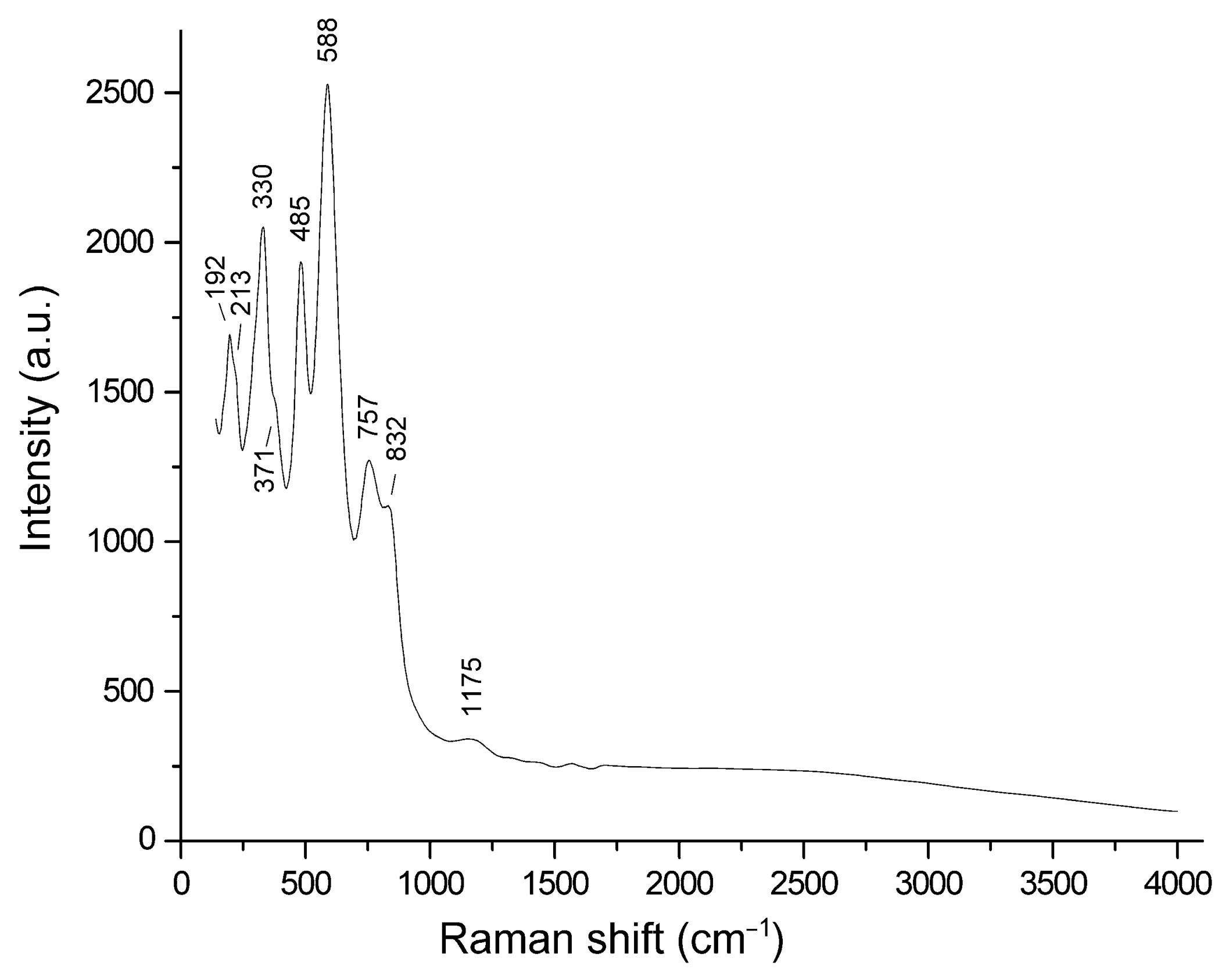
3.2. Raman Spectroscopy
3.3. Optical Properties
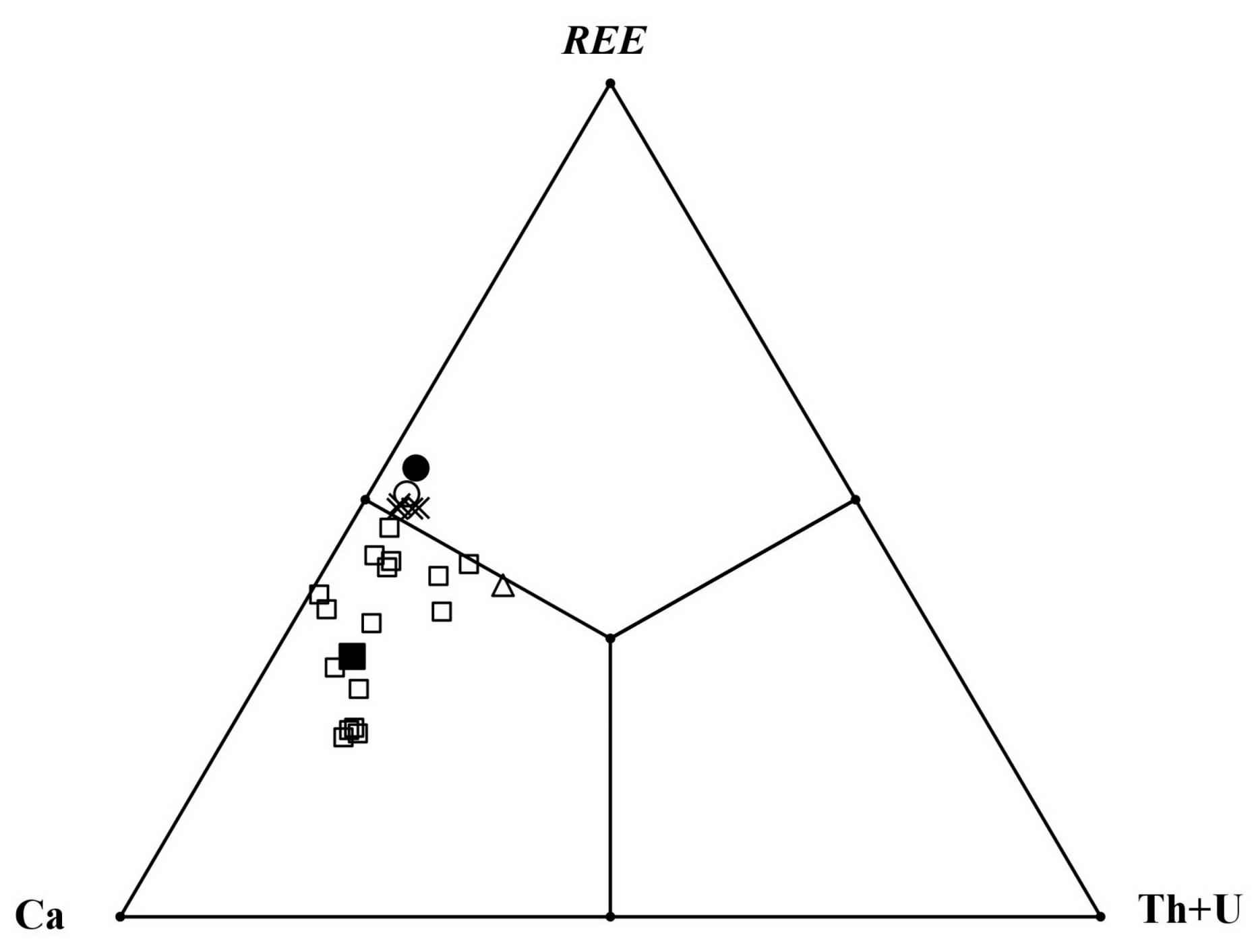
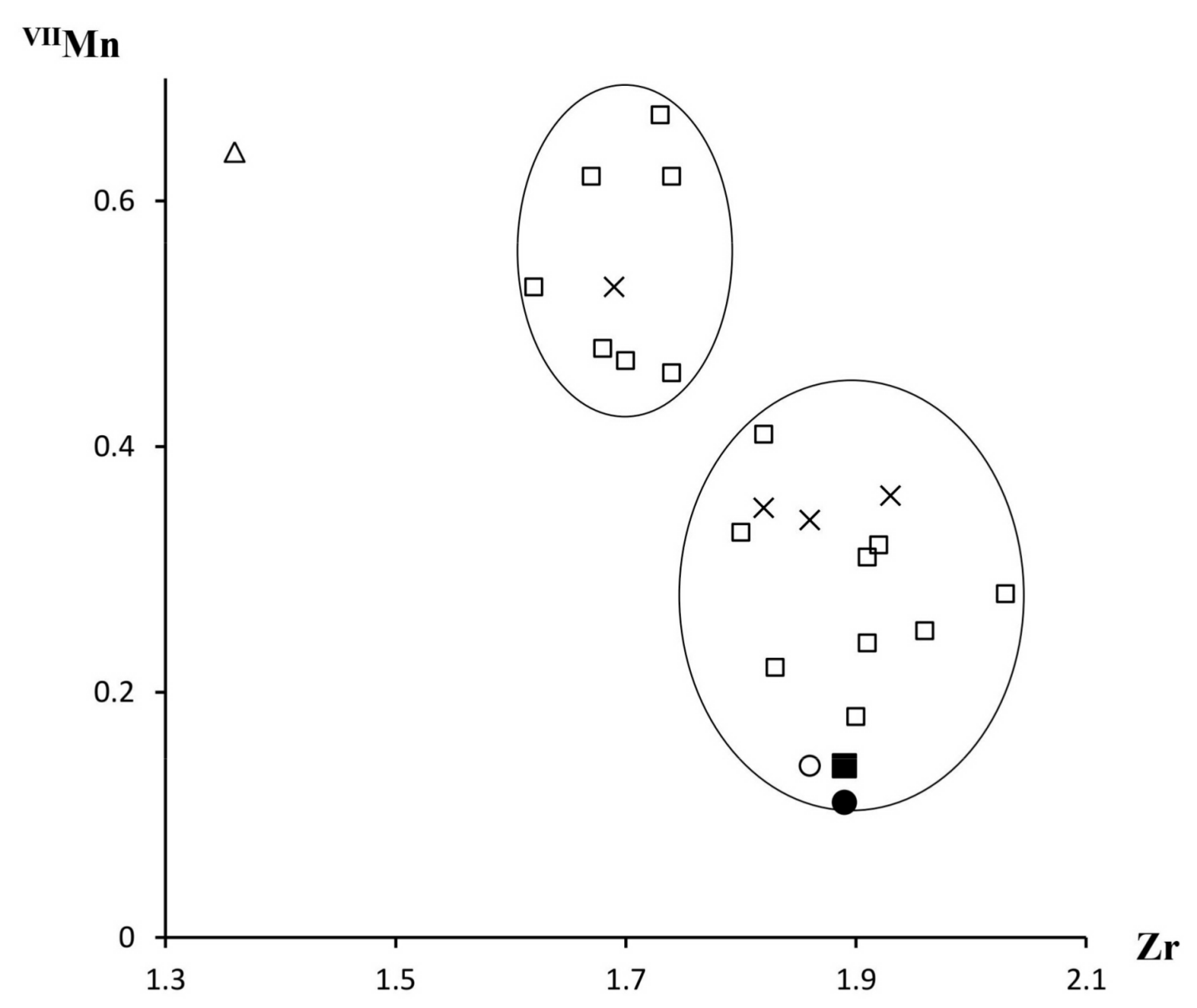
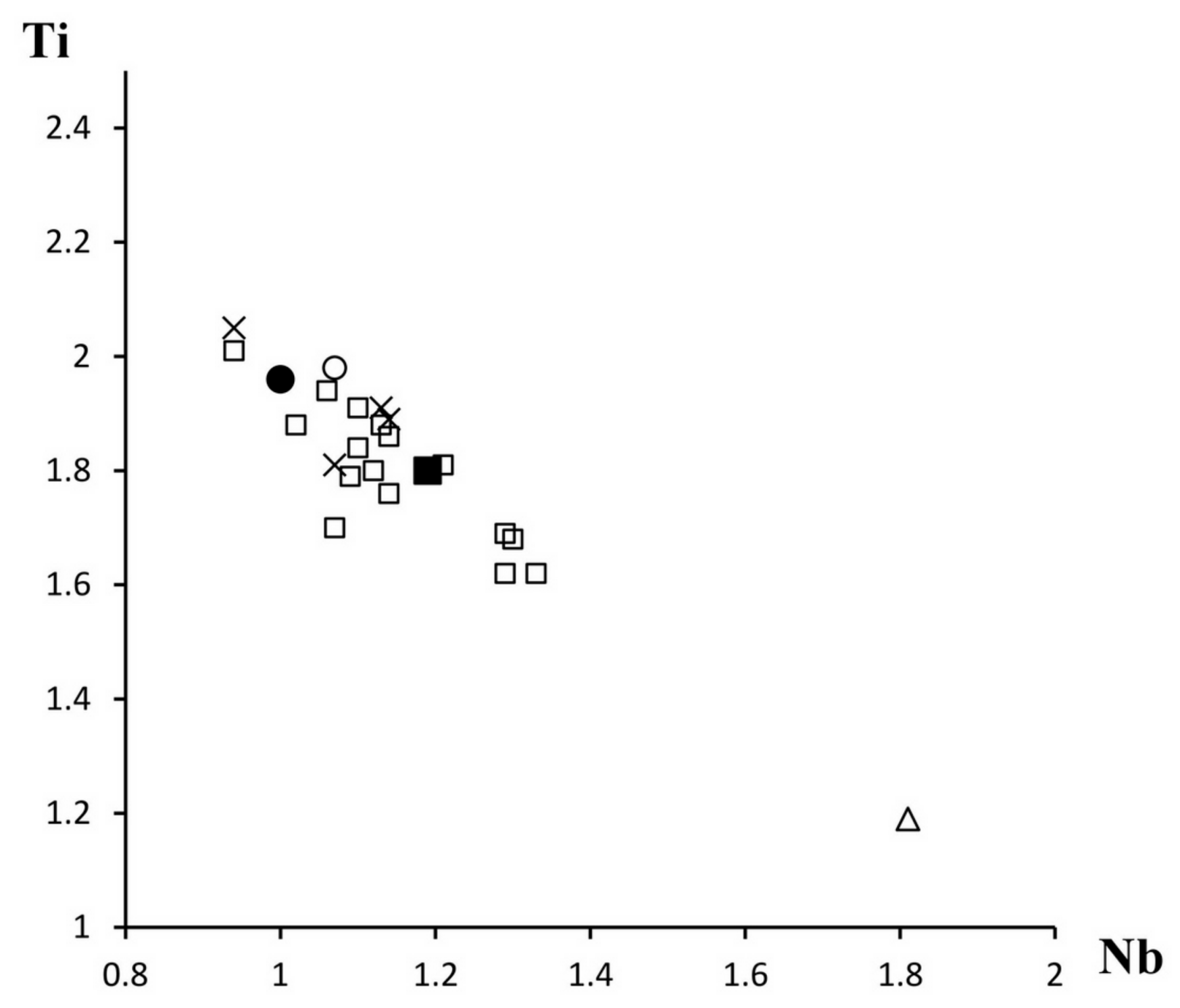
3.4. Chemical Composition
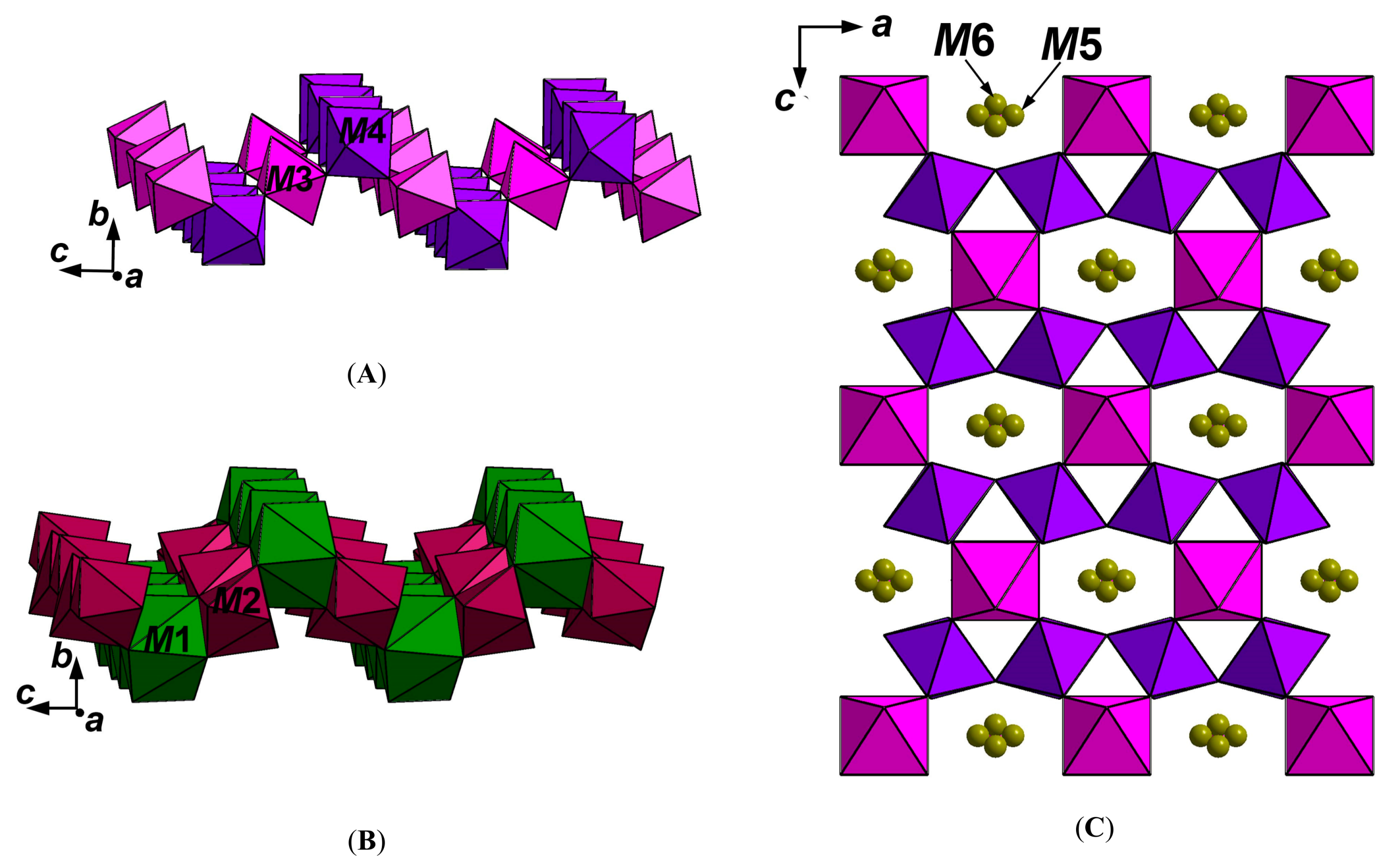
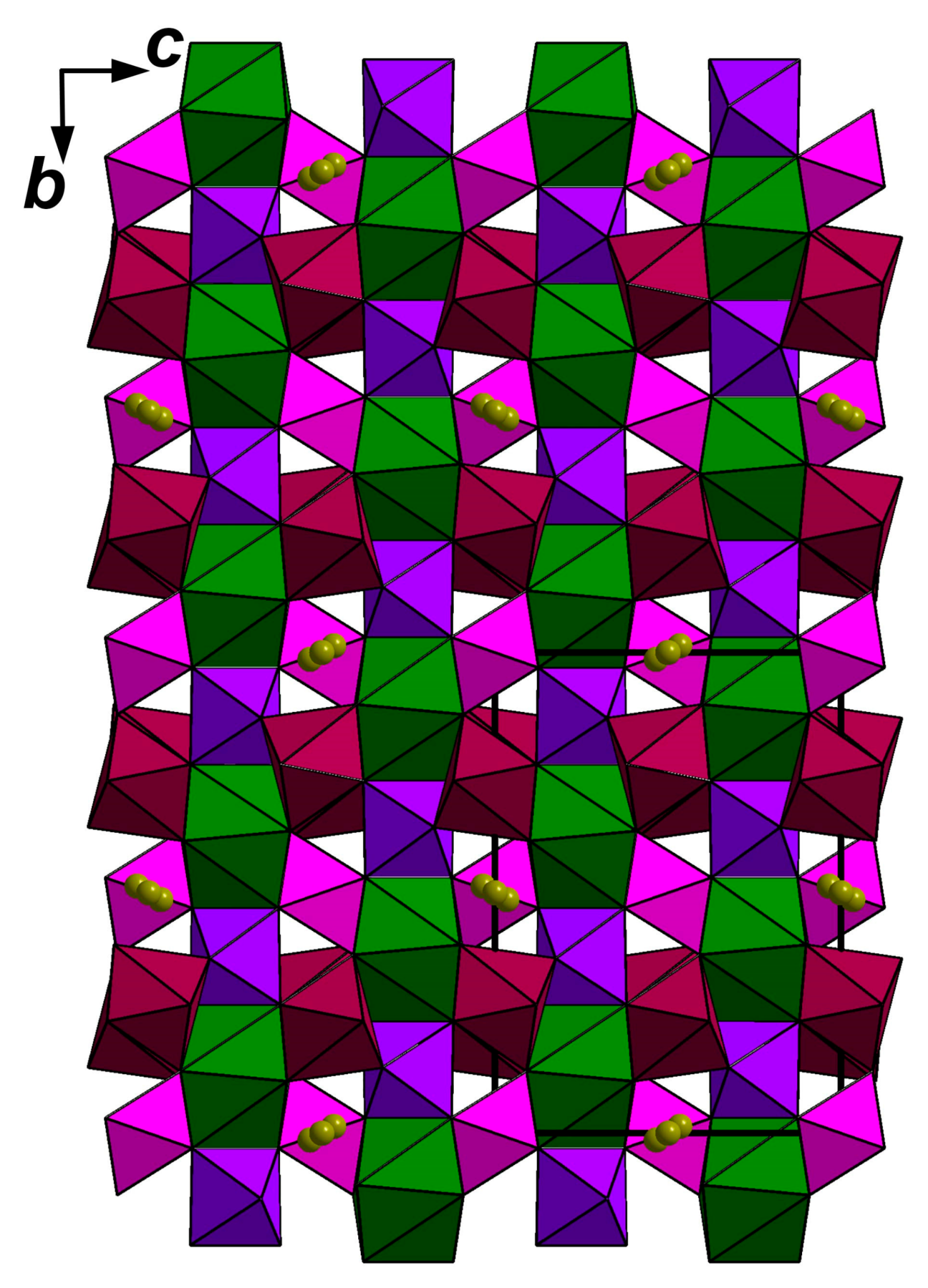
3.5. X-ray Diffraction Data and Crystal Structure
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ringwood, A.E.; Kelly, P.M. Immobilization of high-level waste in ceramic waste forms. Philos. Trans. R. Soc. A 1986, 319, 63–82. [Google Scholar] [CrossRef]
- Donald, I.W.; Metcalfe, B.L.; Taylor, R.N.J. The immobilization of high level radioactive wastes using ceramics and glasses. J. Mater. Sci. 1997, 32, 5851–5887. [Google Scholar] [CrossRef]
- Laverov, N.P.; Yudintsev, S.V.; Stefanovsky, S.V.; Omel’yanenko, B.I.; Nikonov, B.S. Murataite as a universal matrix for immobilization of actinides. Geol. Ore Depos. 2006, 48, 335–356. [Google Scholar] [CrossRef]
- Barinova, T.V.; Borovinskaya, I.P.; Ratnikov, V.I.; Ignat’eva, T.I. Self-propagating high-temperature synthesis for immobilization of high-level waste in mineral-like ceramics: 1. Synthesis and study of titanate ceramics based on perovskite and zirconolite. Radiochemistry 2008, 50, 316–320. [Google Scholar] [CrossRef]
- Zhang, Y.; Gregg, D.J.; Kong, L.; Jovanovich, M.; Triani, G. Zirconolite glass-ceramics for plutonium immobilization: The effects of processing redox conditions on charge compensation and durability. J. Nucl. Mater. 2017, 490, 238–241. [Google Scholar] [CrossRef]
- Williams, C.T.; Gieré, R. Zirconolite: A review of localities worldwide, and a compilation of its chemical compositions. Bull. Nat. Hist. Mus. Lond. 1996, 52, 1–24. [Google Scholar]
- Della Ventura, G.; Bellatreccia, F.; Williams, C.T. Zirconolite with significant REEZrNb(Mn,Fe)O7 from a xenolith of the Laacher See eruptive center, Eifel volcanic region, Germany. Can. Mineral. 2000, 38, 57–65. [Google Scholar] [CrossRef]
- Sinclair, W.; Eggleton, R.A. Structure refinement of zirkelite (zirconolite) from Kaiserstuhl, Germany. Am. Mineral. 1982, 67, 615–620. [Google Scholar]
- Chukanov, N.V.; Krivovichev, S.V.; Pakhomova, A.S.; Pekov, I.V.; Schäfer, Ch.; Vigasina, M.F.; Van, K.V. Laachite, (Ca,Mn)2Zr2Nb2TiFeO14, a new zirconolite-related mineral from the Eifel volcanic region, Germany. Eur. J. Mineral. 2014, 26, 103–111. [Google Scholar] [CrossRef]
- Zubkova, N.V.; Chukanov, N.V.; Pekov, I.V.; Ternes, B.; Schüller, W.; Pushcharovsky, D.Y. The crystal structure of natural nonmetamict Nb-rich zirconolite-3T from the Eifel paleovolcanic region, Germany. Z. Kristallogr. 2018, 233. [Google Scholar] [CrossRef]
- Litt, T.; Brauer, A.; Goslar, T.; Merk, J.; Balaga, K.; Mueller, H.; Ralska-Jasiewiczowa, M.; Stebich, M.; Negendank, J.F.W. Correlation and synchronisation of Lateglacial continental sequences in northern Central Europe based on annually laminated lacustrine sediments. Quat. Sci. Rev. 2001, 20, 1233–1249. [Google Scholar] [CrossRef]
- Schmitt, A.K.; Wetzel, F.; Cooper, K.M.; Zou, H.; Wörner, G. Magmatic longevity of Laacher See volcano (Eifel, Germany) indicated by U–Th dating of intrusive carbonatites. J. Petrol. 2010, 51, 1053–1085. [Google Scholar] [CrossRef]
- Frechen, J. Vorgänge der Sanidinit-Bildung im Laacher Seegebiet. Fortschritte der Mineralogie 1947, 26, 147–166. (In German) [Google Scholar]
- Schmitt, A.K. Laacher See revisited: High-spatial-resolution zircon dating indicates rapid formation of a zoned magma chamber. Geology 2006, 34, 597–600. [Google Scholar] [CrossRef]
- Britvin, S.N.; Dolivo-Dobrovolsky, D.V.; Krzhizhanovskaya, M.G. Software for processing the X-ray powder diffraction data obtained from the curved image plate detector of Rigaku RAXIS Rapid II diffractometer. Zap. Ross. Mineral. Obsh. 2017, 146, 104–107. (In Russian) [Google Scholar]
- Mazzi, F.; Munno, R. Calciobetafite (new mineral of the pyrochlore group) and related minerals from Campi Flegrei, Italy; crystal structures of polymignite and zirkelite: Comparison with pyrochlore and zirconolite. Am. Mineral. 1983, 68, 262–276. [Google Scholar]
- Berzelius, J. Undersökning af några Mineralier. 2. Polymignit. Kongl. Svenska Vetensk.-Acad. Handl. 1984, 338–345. (In Swedish) [Google Scholar]
- Brøgger, W.C. Die Mineralien der Syenitpegmatitgänge der südnorwegischen Augit und Nephelinsyenite. Z. Kristallogr. Speziellen Teil 1890, 16, 1–663. (In German) [Google Scholar]
- Bayliss, P.; Mazzi, F.; Munno, R.; White, T.J. Mineral nomenclature: Zirconolite. Mineral. Mag. 1989, 53, 565–569. [Google Scholar] [CrossRef]
- Chukhrov, F.V.; Bonshtedt-Kupletskaya, E.M. Mineraly Vol. II(3); Nauka: Moscow, Russia, 1967; pp. 188–190. (In Russian) [Google Scholar]
- Pudovkina, Z.V.; Chernitzova, N.M.; Pyatenko, Y.A. Crystallographic study of polymignite. Zap. Vses. Mineral. Obshch. 1969, 98, 193–199. (In Russian) [Google Scholar]
- Borodin, L.S.; Nazarenko, I.I.; Richter, T.L. On a new mineral zirconolite–A complex oxide of AB3O7 type. Dokl. Akad. Nauk SSSR 1956, 110, 845–848. (In Russian) [Google Scholar]
- Borodin, L.S.; Bykova, A.V.; Kapitonova, T.A.; Pyatenko, Y.A. New data on zirconolite and its new niobian variety. Dokl. Akad. Nauk SSSR 1960, 134, 1188–1192. (In Russian) [Google Scholar]

| Characteristics | Data and Methods |
|---|---|
| Crystal sizes, mm | 0.01 × 0.01 × 0.10 |
| Temperature, K | 293(2) |
| Radiation and wavelength, Å | MoKα; 0.71073 |
| F000 | 1530 |
| θ range for data collection, ° | 2.88–26.98 |
| h, k, l ranges | –9 → 7, –18 → 18, –12 → 12 |
| Reflections collected | 4962 |
| Independent reflections | 617 (Rint = 0.0215) |
| Independent reflections with I > 2σ(I) | 574 |
| Data reduction | Bruker SAINT |
| Structure solution | Direct methods |
| Refinement method | Full-matrix least-squares on F2 |
| Weighting coefficients a, b | 0.0251, 7.1886 |
| Number of refined parameters | 71 |
| Final R indices (with I > 2σ(I)) | R1 = 0.0198, wR2 = 0.0518 |
| R indices (with all data) | R1 = 0.0224, wR2 = 0.0550 |
| GoF | 1.161 |
| Largest diffraction peak and hole, e/Å3 | 1.53 and –0.70 |
| Wavelength, nm | R1 | R2 |
|---|---|---|
| 400 | 17.3 | 16.8 |
| 420 | 16.8 | 16.4 |
| 440 | 16.4 | 16.0 |
| 460 | 16.0 | 15.5 |
| 470 | 15.8 | 15.3 |
| 480 | 15.6 | 15.2 |
| 500 | 15.3 | 15.0 |
| 520 | 15.3 | 14.8 |
| 540 | 15.0 | 14.7 |
| 546 | 15.0 | 14.7 |
| 560 | 15.0 | 14.6 |
| 580 | 14.9 | 14.6 |
| 589 | 14.9 | 14.5 |
| 600 | 14.8 | 14.5 |
| 620 | 14.8 | 14.5 |
| 640 | 14.8 | 14.4 |
| 650 | 14.8 | 14.4 |
| 660 | 14.8 | 14.4 |
| 680 | 14.7 | 14.4 |
| 700 | 14.7 | 14.3 |
| Constituent | wt % | Range | Standard Deviation | Standard Used | X-ray Line Measured |
|---|---|---|---|---|---|
| CaO | 5.45 5.29 | 5.27–5.55 5.12–5.39 | 0.10 0.34 | Wollastonite | K |
| MnO | 4.19 4.16 | 4.07–4.32 4.06–4.34 | 0.09 0.13 | MnTiO3 | K |
| FeO | 7.63 6.62 | 7.46–7.79 6.23–6.83 | 0.14 0.28 | Fe | K |
| Al2O3 | 0.27 0.59 | 0.18–0.38 0.48–0.78 | 0.07 0.14 | Albite | K |
| Y2O3 | 0.00 0.90 | – 0.61–0.99 | – 0.16 | YPO4 | L |
| La2O3 | 3.17 3.64 | 3.05–3.28 3.47–3.84 | 0.10 0.16 | LaPO4 | L |
| Ce2O3 | 11.48 11.22 | 11.27–11.73 10.95–11.69 | 0.19 0.33 | CePO4 | L |
| Pr2O3 | 1.04 0.92 | 0.89–1.24 0.90–0.97 | 0.12 0.03 | PrPO4 | L |
| Nd2O3 | 2.18 2.46 | 2.10–2.34 2.28–2.81 | 0.08 0.25 | NdPO4 | L |
| ThO2 | 2.32 1.98 | 2.11–2.50 1.79–2.17 | 0.15 0.16 | ThO2 | M |
| TiO2 | 17.78 18.69 | 17.45–18.12 18.49–18.90 | 0.27 0.16 | TiO2 | M |
| ZrO2 | 27.01 27.69 | 26.82–27.26 27.51–27.86 | 0.19 0.11 | ZrO2 | L |
| Nb2O5 | 17.04 15.77 | 16.72–17.37 15.53–15.99 | 0.28 0.19 | LiNbO3 | L |
| Total | 99.59 99.82 | - | - | - | - |
| Iobs, % | dobs, Å | Icalc, % | dcalc, Å | h | k | l | Iobs, % | dobs, Å | Icalc, % | dcalc, Å | h | k | l |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 4 | 7.068 | 2 | 7.074 | 0 | 2 | 0 | 2 | 1.743 | 1 | 1.743 | 3 | 1 | 4 |
| 2 | 5.806 | 1 | 5.805 | 0 | 2 | 1 | - | - | 1 | 1.742 | 0 | 8 | 1 |
| 3 | 5.463 | 1 | 5.466 | 1 | 1 | 1 | 1 | 1.735 | 1 | 1.734 | 3 | 5 | 2 |
| 5 | 5.085 | 1 | 5.081 | 0 | 0 | 2 | 1 | 1.721 | 1 | 1.722 | 2 | 2 | 5 |
| 1 | 4.131 | 1 | 4.126 | 0 | 2 | 2 | 1 | 1.710 | 1 | 1.709 | 2 | 6 | 3 |
| 2 | 4.001 | 2 | 3.999 | 1 | 1 | 2 | 4 | 1.690 | 3 | 1.688 | 1 | 7 | 3 |
| 10 | 3.689 | 8 | 3.690 | 1 | 3 | 1 | 1 | 1.667 | 1 | 1.668 | 4 | 2 | 2 |
| 2 | 3.536 | 1 | 3.537 | 0 | 4 | 0 | 2 | 1.646 | 1 | 1.647 | 0 | 2 | 6 |
| 3 | 3.343 | 2 | 3.340 | 0 | 4 | 1 | - | - | 1 | 1.646 | 3 | 3 | 4 |
| 2 | 3.238 | 1 | 3.242 | 2 | 2 | 0 | - | - | 1 | 1.639 | 1 | 1 | 6 |
| 91 | 2.963 | 100 | 2.963 | 2 | 0 | 2 | 1 | 1.600 | 1 | 1.601 | 4 | 4 | 1 |
| 100 | 2.903 | 93 | 2.903 | 0 | 4 | 2 | 2 | 1.571 | 1 | 1.572 | 2 | 8 | 1 |
| 2 | 2.731 | 1 | 2.733 | 2 | 2 | 2 | - | - | 1 | 1.568 | 0 | 8 | 3 |
| 2 | 2.575 | 1 | 2.574 | 1 | 3 | 3 | 1 | 1.561 | 1 | 1.562 | 2 | 6 | 4 |
| 39 | 2.540 | 14 | 2.540 | 0 | 0 | 4 | 20 | 1.543 | 20 | 1.544 | 4 | 4 | 2 |
| - | - | 27 | 2.539 | 2 | 4 | 0 | 10 | 1.532 | 2 | 1.536 | 3 | 7 | 1 |
| 1 | 2.393 | 1 | 2.391 | 0 | 2 | 4 | - | - | 10 | 1.536 | 2 | 0 | 6 |
| 1 | 2.367 | 1 | 2.365 | 1 | 1 | 4 | - | - | 11 | 1.527 | 0 | 4 | 6 |
| 5 | 2.343 | 5 | 2.342 | 2 | 2 | 3 | 16 | 1.519 | 17 | 1.519 | 2 | 8 | 2 |
| - | - | 1 | 2.341 | 1 | 5 | 2 | 1 | 1.500 | 1 | 1.501 | 2 | 2 | 6 |
| 3 | 2.298 | 3 | 2.297 | 0 | 6 | 1 | 1 | 1.494 | 1 | 1.492 | 3 | 5 | 4 |
| 1 | 2.270 | 1 | 2.271 | 2 | 4 | 2 | 6 | 1.482 | 6 | 1.482 | 4 | 0 | 4 |
| 3 | 2.166 | 2 | 2.168 | 3 | 1 | 2 | - | - | 1 | 1.481 | 3 | 3 | 5 |
| 1 | 2.139 | 1 | 2.138 | 1 | 3 | 4 | 6 | 1.451 | 6 | 1.451 | 0 | 8 | 4 |
| 1 | 2.113 | 1 | 2.114 | 3 | 3 | 1 | 1 | 1.441 | 1 | 1.440 | 2 | 8 | 3 |
| 2 | 2.082 | 1 | 2.081 | 1 | 5 | 3 | 1 | 1.427 | 1 | 1.428 | 4 | 6 | 1 |
| 1 | 2.062 | 1 | 2.063 | 0 | 4 | 4 | - | - | 1 | 1.425 | 1 | 5 | 6 |
| 1 | 2.033 | 1 | 2.032 | 2 | 4 | 3 | 3 | 1.413 | 2 | 1.413 | 3 | 7 | 3 |
| 1 | 2.001 | 1 | 2.000 | 2 | 2 | 4 | 2 | 1.407 | 1 | 1.406 | 1 | 7 | 5 |
| 3 | 1.956 | 2 | 1.957 | 3 | 1 | 3 | 1 | 1.400 | 1 | 1.399 | 1 | 9 | 3 |
| - | - | 1 | 1.953 | 0 | 2 | 5 | 1 | 1.381 | 1 | 1.381 | 5 | 3 | 1 |
| 3 | 1.940 | 1 | 1.944 | 2 | 6 | 1 | 1 | 1.365 | 1 | 1.367 | 4 | 4 | 4 |
| - | - | 1 | 1.935 | 0 | 6 | 3 | - | - | 1 | 1.363 | 1 | 3 | 7 |
| 6 | 1.914 | 3 | 1.913 | 1 | 7 | 1 | 1 | 1.334 | 1 | 1.334 | 5 | 1 | 3 |
| 1 | 1.847 | 1 | 1.845 | 2 | 6 | 2 | - | - | 1 | 1.334 | 0 | 8 | 5 |
| - | - | 1 | 1.830 | 1 | 5 | 4 | - | - | 1 | 1.333 | 4 | 2 | 5 |
| 15 | 1.823 | 15 | 1.824 | 4 | 0 | 0 | - | - | 1 | 1.333 | 3 | 3 | 6 |
| - | - | 1 | 1.822 | 3 | 3 | 3 | 1 | 1.307 | 1 | 1.309 | 3 | 9 | 1 |
| 51 | 1.796 | 56 | 1.796 | 2 | 4 | 4 | - | - | 1 | 1.305 | 0 | 10 | 3 |
| 10 | 1.769 | 12 | 1.768 | 0 | 8 | 0 | 1 | 1.288 | 1 | 1.289 | 5 | 3 | 3 |
| Site | x | y | z | Ueq | Site Population | Q |
|---|---|---|---|---|---|---|
| VIIIM(1) | 0.25 | 0.11753(3) | –0.2500 | 0.00954(18) | Ce0.44Ca0.40Mn0.12Th0.04 | 8 |
| VIIM(2) | 0.5 | 0.23411(4) | 0.01426(5) | 0.0120(2) | Zr0.937(9)Mn0.063(9) | 8 |
| VIM(3) | 0.0 | 0.0 | 0.0 | 0.0112(3) | Nb0.608(8)Ti0.392(8) | 4 |
| VIM(4) | 0.25 | 0.13297(5) | 0.25 | 0.0129(2) | Ti0.74Nb0.24Al0.02 | 8 |
| IVM(5) | 0.4211(8) | 0.0 | 0.0 | 0.0301(9) | Fe0.24Mn0.04 | 8 |
| VM(6) | 0.5 | 0.0140(5) | 0.0342(9) | 0.0393(19) | Fe0.20Mn0.02 | 8 |
| O(1) | 0.1948(4) | 0.03150(18) | 0.1256(2) | 0.0236(6) | O | 16 |
| O(2) | 0.2131(4) | 0.23287(17) | 0.1201(2) | 0.0218(6) | O | 16 |
| O(3) | 0.5 | 0.1081(2) | –0.1007(4) | 0.0182(8) | O | 8 |
| O(4) | 0.0 | 0.1288(2) | –0.0912(4) | 0.0184(8) | O | 8 |
| O(5) | 0.5 | 0.1366(3) | 0.1756(4) | 0.0250(9) | O | 8 |
| Cation | Ligand | Distance | Cation | Ligand | Distance |
|---|---|---|---|---|---|
| M(1) | O(3) | 2.376(2) × 2 | M(3) | O(4) | 2.044(3) × 2 |
| M(1) | O(4) | 2.441(3) × 2 | M(4) | O(2) | 1.952(2) × 2 |
| M(1) | O(1) | 2.491(3) × 2 | M(4) | O(1) | 1.955(3) × 2 |
| M(1) | O(2) | 2.508(3) × 2 | M(4) | O(5) | 1.9759(16) × 2 |
| M(2) | O(4) | 2.091(3) | M(5) | O(3) | 1.928(4) × 2 |
| M(2) | O(2) | 2.122(3) × 2 | M(5) | O(1) | 2.134(5) × 2 |
| M(2) | O(3) | 2.132(4) | M(6) | O(3) | 1.855(8) |
| M(2) | O(5) | 2.142(4) | M(6) | O(3) | 1.910(9) |
| M(2) | O(2) | 2.354(3) × 2 | M(6) | O(5) | 2.251(9) |
| M(3) | O(1) | 1.961(3) × 4 | M(6) | O(1) | 2.426(5) × 2 |
| Site | Zirconolite-3O | Laachite | Nöggerathite-(Ce) |
|---|---|---|---|
| VIIIM(1) | Ca0.53Ce0.41Na0.04Th0.02 | Ca0.28Mn0.26Ln0.26Th0.14Y0.06 Ca0.32Mn0.28Ln0.24Th0.14Y0.02 | Ln0.44Ca0.40Mn0.12Th0.04 |
| VIIM(2) | Zr | Zr0.78Mn0.22 | Zr0.937Mn0.063 |
| VIM(3) | Ti0.52Nb0.47Ta0.01 | Nb0.82Ti0.18 | Nb0.608Ti0.392 |
| VIM(4) | Ti0.88Nb0.12 | Ti0.72Nb0.28 Nb0.44Ti0.40Al0.16 | Ti0.74Nb0.24Al0.02 |
| IVM(5) | Fe0.46 | Fe0.34Mn0.10Y0.06 | Fe0.24Mn0.04 |
| VM(6) | Fe0.03 | Fe0.20Mn0.02 | |
| Reference | [16] | [9] | This work |
| Mineral | Nöggerathite-(Ce) | Laachite | Zirconolite-3O |
|---|---|---|---|
| Idealized formula | (Ce,Ca)2Zr2(Nb,Ti) (Ti,Nb)2Fe2+O14 | Ca2Zr2Nb2TiFeO14 | CaZrTi2O7 |
| Crystal system Space group | Orthorhombic Cmca | Monoclinic C2/c | Orthorhombic Cmca |
| a, Å b, Å c, Å β, ° Z | 7.2985 14.1454 10.1607 90 4 | 7.3119 14.1790 10.1700 90.072 4 | 7.278–7.284 14.147–14.18 10.145–10.148 90 8 |
| Strong lines of the X-ray powder diffraction pattern: d, Å (I, %) | 2.963 (91) 2.903 (100) 2.540 (39) 1.823 (15) 1.796 (51) 1.543 (20) 1.519 (16) | 4.298 (22) 2.967 (100) 2.901 (59) 2.551 (32) 1.800 (34) 1.541 (24) 1.535 (23) 1.529 (23) | 3.176 (30) 2.914 (100) 2.506 (40) 1.980 (90) 1.792 (90) 1.517 (10) |
| Refractive index | 2.267 (mean, calc.) | 2.26 (mean, calc.) | 2.215 (meas., metamict); 2.26–2.31 (calc.) |
| Density, g cm–3 | 5.332 (calc.) | 5.42 (calc.) | 4.7 (meas.) 4.9 (calc.) |
| Sources | This work | [9] | [7,8,16,19,20,21,22] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chukanov, N.V.; Zubkova, N.V.; Britvin, S.N.; Pekov, I.V.; Vigasina, M.F.; Schäfer, C.; Ternes, B.; Schüller, W.; Polekhovsky, Y.S.; Ermolaeva, V.N.; et al. Nöggerathite-(Ce), (Ce,Ca)2Zr2(Nb,Ti)(Ti,Nb)2Fe2+O14, a New Zirconolite-Related Mineral from the Eifel Volcanic Region, Germany. Minerals 2018, 8, 449. https://doi.org/10.3390/min8100449
Chukanov NV, Zubkova NV, Britvin SN, Pekov IV, Vigasina MF, Schäfer C, Ternes B, Schüller W, Polekhovsky YS, Ermolaeva VN, et al. Nöggerathite-(Ce), (Ce,Ca)2Zr2(Nb,Ti)(Ti,Nb)2Fe2+O14, a New Zirconolite-Related Mineral from the Eifel Volcanic Region, Germany. Minerals. 2018; 8(10):449. https://doi.org/10.3390/min8100449
Chicago/Turabian StyleChukanov, Nikita V., Natalia V. Zubkova, Sergey N. Britvin, Igor V. Pekov, Marina F. Vigasina, Christof Schäfer, Bernd Ternes, Willi Schüller, Yury S. Polekhovsky, Vera N. Ermolaeva, and et al. 2018. "Nöggerathite-(Ce), (Ce,Ca)2Zr2(Nb,Ti)(Ti,Nb)2Fe2+O14, a New Zirconolite-Related Mineral from the Eifel Volcanic Region, Germany" Minerals 8, no. 10: 449. https://doi.org/10.3390/min8100449
APA StyleChukanov, N. V., Zubkova, N. V., Britvin, S. N., Pekov, I. V., Vigasina, M. F., Schäfer, C., Ternes, B., Schüller, W., Polekhovsky, Y. S., Ermolaeva, V. N., & Pushcharovsky, D. Y. (2018). Nöggerathite-(Ce), (Ce,Ca)2Zr2(Nb,Ti)(Ti,Nb)2Fe2+O14, a New Zirconolite-Related Mineral from the Eifel Volcanic Region, Germany. Minerals, 8(10), 449. https://doi.org/10.3390/min8100449







