A Density Functional Theory Study on the Effect of Lattice Impurities on the Electronic Structures and Reactivity of Fluorite
Abstract
:1. Introduction
2. Computational Details
3. Results and Discussion
3.1. Lattice Parameter
3.2. Fermi Level
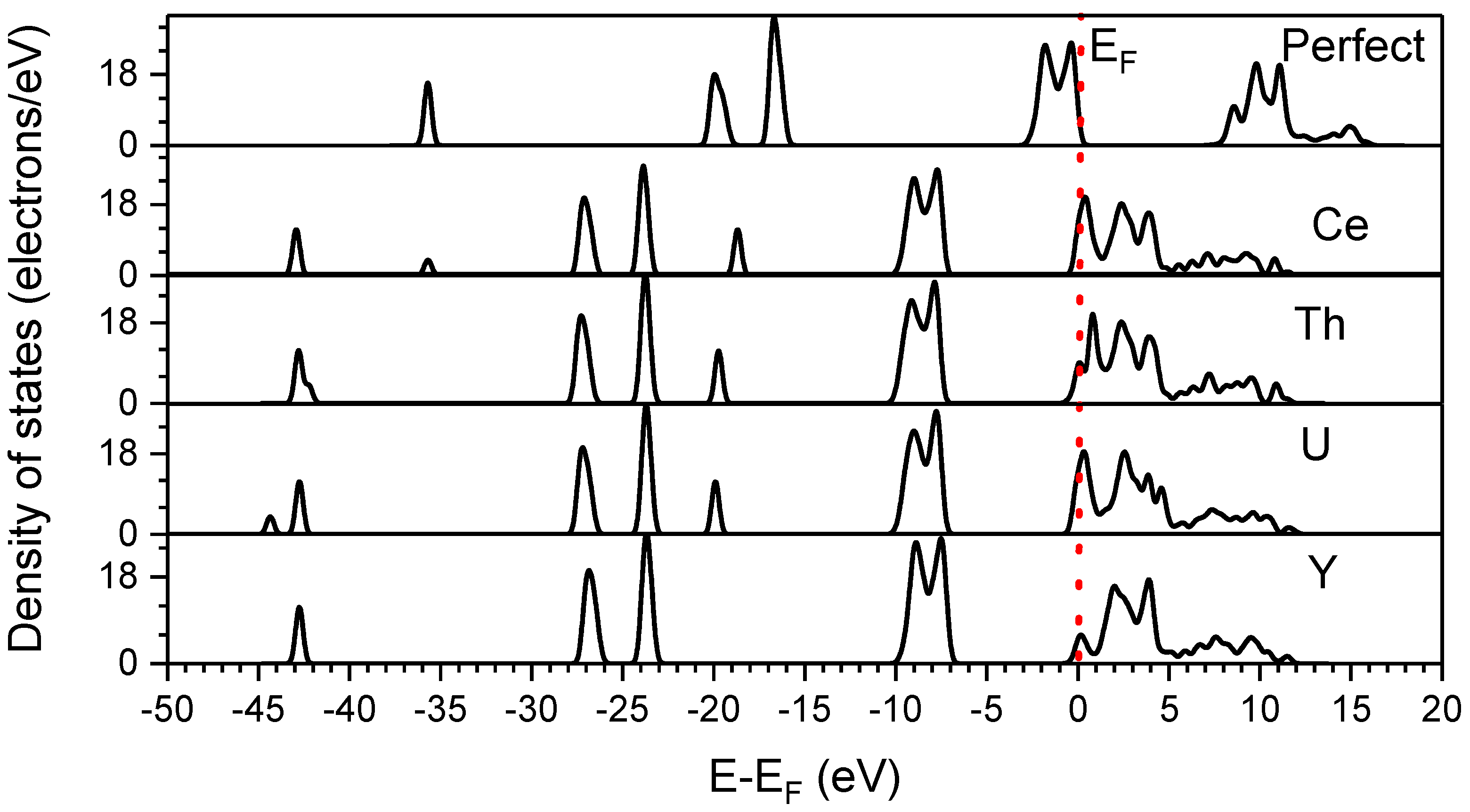
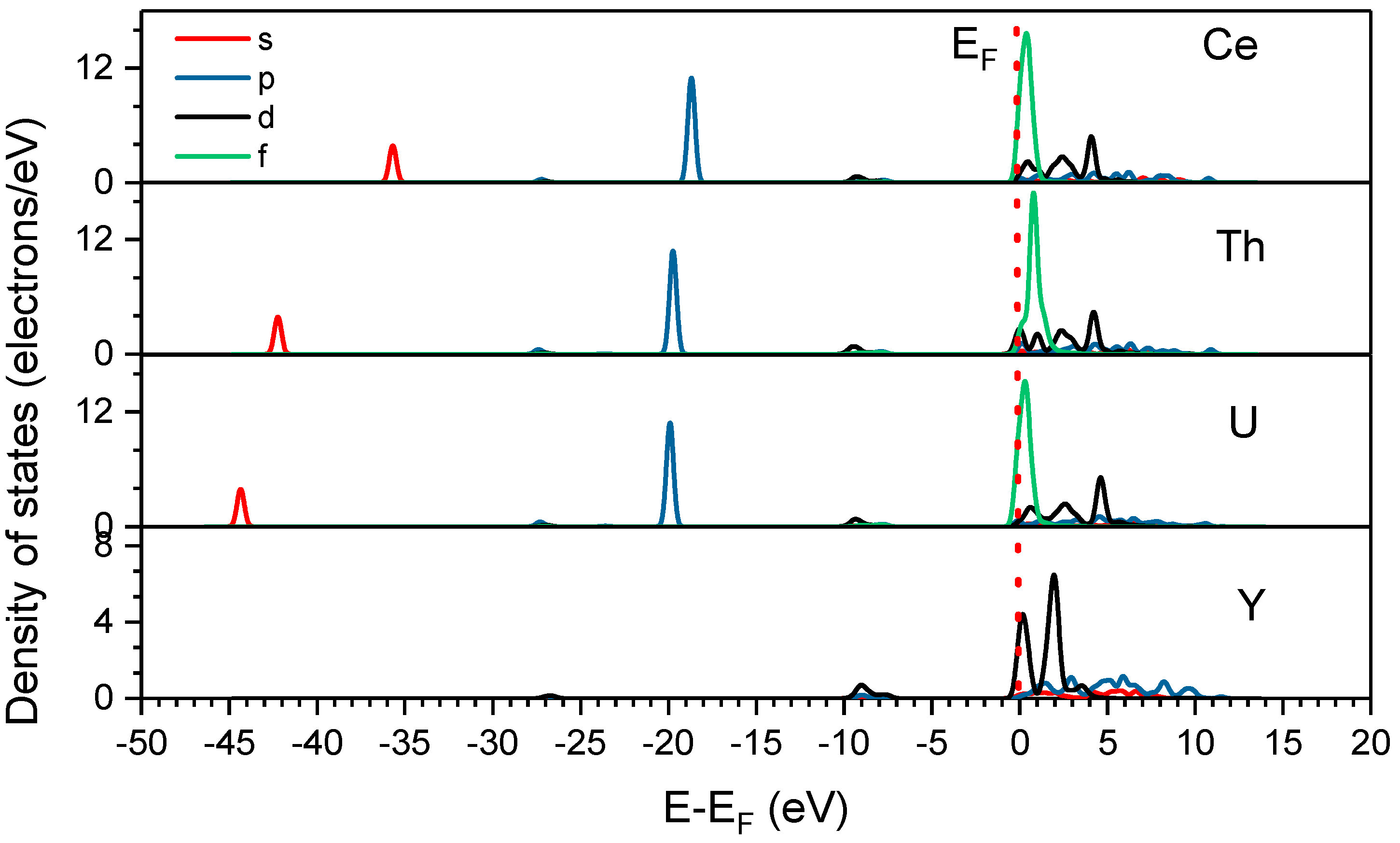
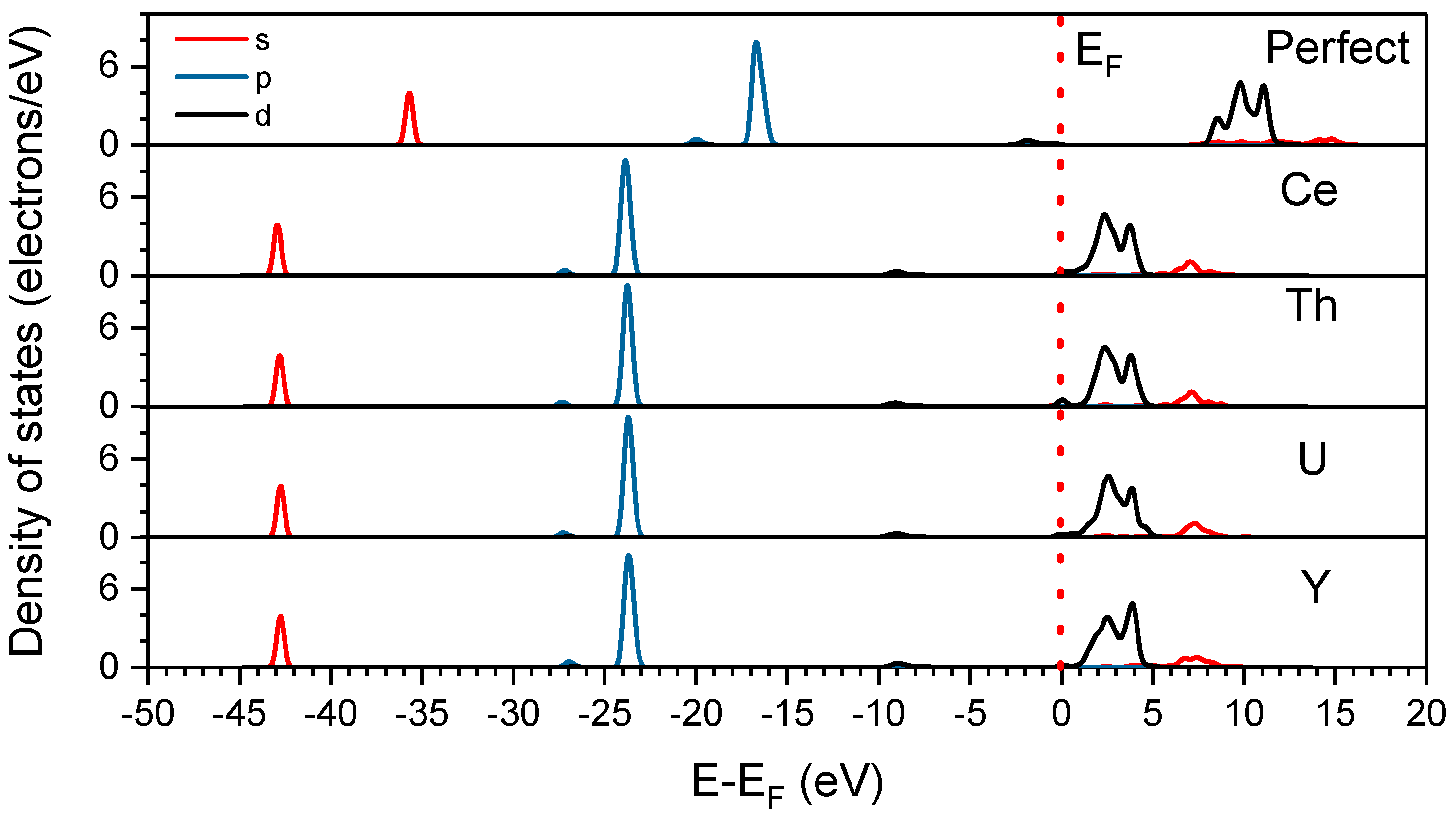
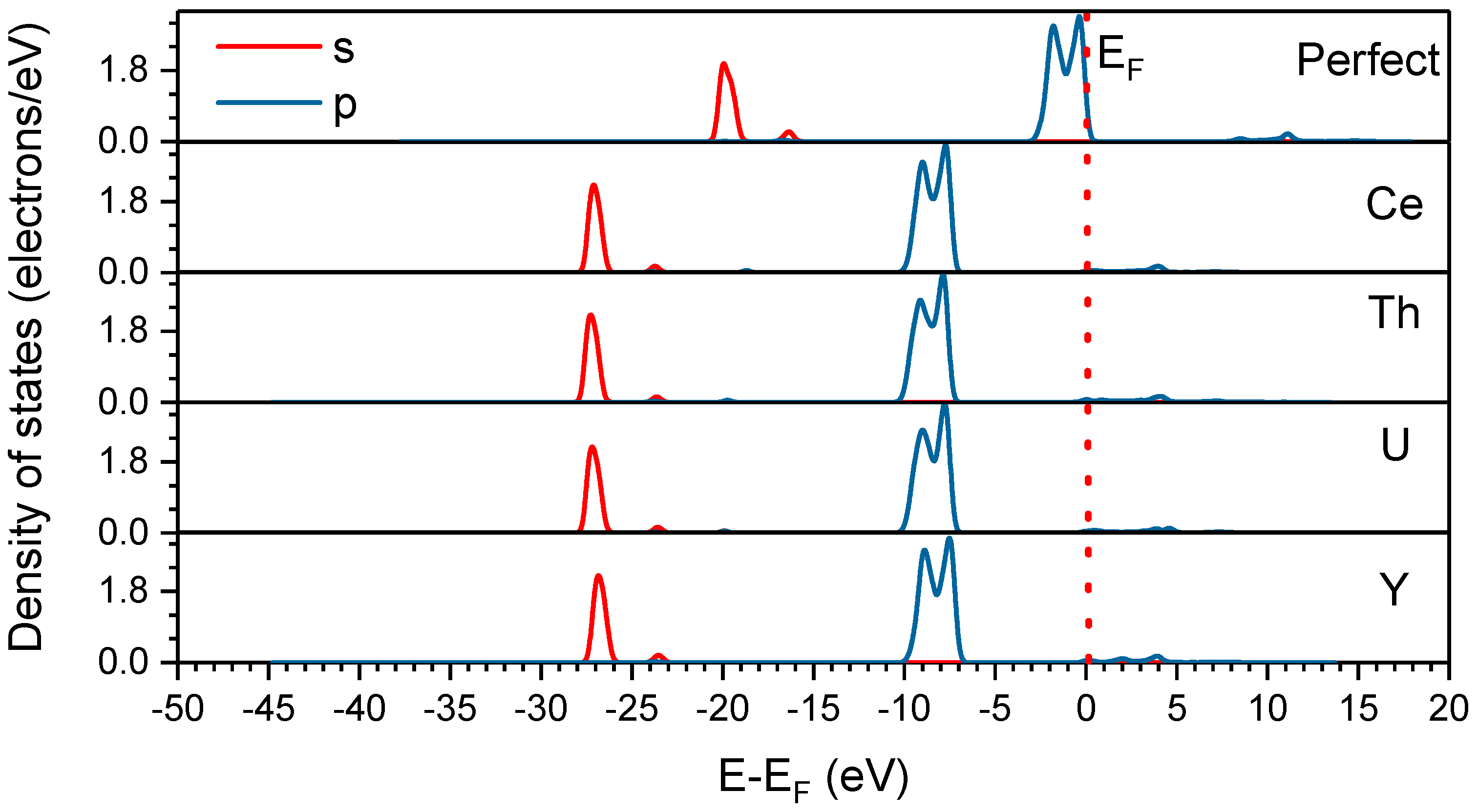
3.3. Density of States
3.4. Analysis of the Mulliken Population
3.5. Effect of Impurity on the Reactivity of Fluorite
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- El-Nadi, Y.A. Solvent extraction and recovery of Y(III) and Yb(III) from fluorspar mineral. Int. J. Miner. Metall. Mater. 2013, 20, 713–719. [Google Scholar] [CrossRef]
- Somasundaran, P.; Amankonah, J.O.; Ananthapadmabhan, K.P. Mineral–solution equilibria in sparingly soluble mineral systems. Colloids Surf. 1985, 15, 309–333. [Google Scholar] [CrossRef]
- Marinakis, K.I.; Shergold, H.L. The mechanism of fatty acid adsorption in the presence of fluorite, calcite and barite. Int. J. Miner. Process. 1985, 14, 161–176. [Google Scholar] [CrossRef]
- Pugh, R.; Stenius, P. Solution chemistry studies and flotation behaviour of apatite, calcite and fluorite minerals with sodium oleate collector. Int. J. Miner. Process. 1985, 15, 193–218. [Google Scholar] [CrossRef]
- Gao, Y.; Gao, Z.; Sun, W.; Hu, Y. Selective flotation of scheelite from calcite: A novel reagent scheme. Int. J. Miner. Process. 2016, 154, 10–15. [Google Scholar] [CrossRef]
- Gao, Z.; Bai, D.; Sun, W.; Cao, X.; Hu, Y. Selective flotation of scheelite from calcite and fluorite using a collector mixture. Miner. Eng. 2015, 72, 23–26. [Google Scholar] [CrossRef]
- Hu, Y.; Xu, Z. Interactions of amphoteric amino phosphoric acids with calcium-containing minerals and selective flotation. Int. J. Miner. Process. 2003, 72, 87–94. [Google Scholar] [CrossRef]
- Liu, T.; Zhao, Y.; Li, X. Preliminary Study of the Relationship between Irradiation Damage and Fluorite Colour. Acta Mineral. Sin. 1983, 4, 10. [Google Scholar]
- Peng, J.; Hu, R.; Qi, L.; Jiang, G. REE geochemistry of fluorite from the Qinglong antimony deposit and its geological implications. Sci. Geol. Sin. 2002, 37, 277–287. [Google Scholar]
- Gabelman, J.W. Migration of Uranium and Thorium: Exploration Significance; GSW Books: McLean, VA, USA, 1977. [Google Scholar]
- Catlow, C.R.A. Defect Structures in fluorite crystals. Acta Crystallogr. 1973, 32, 1944–1947. [Google Scholar]
- Hayes, W. Crystals with the Fluorite Structure: Electronic, Vibrational, and Defect Properties; Oxford University Press: Oxford, UK, 1974. [Google Scholar]
- Chen, Y.; Chen, J.; Lan, L.; Yang, M. The influence of the impurities on the flotation behaviors of synthetic ZnS. Miner. Eng. 2012, 27–28, 65–71. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, J.; Guo, J. A DFT study on the effect of lattice impurities on the electronic structures and floatability of sphalerite. Miner. Eng. 2010, 23, 1120–1130. [Google Scholar] [CrossRef]
- Yu-Qiong, L.I.; Chen, J.H.; Chen, Y.; Guo, J. Density functional theory study of influence of impurity on electronic properties and reactivity of pyrite. Trans. Nonferr. Met. Soc. China 2011, 21, 1887–1895. [Google Scholar]
- Chen, J.; Ke, B.; Lan, L.; Li, Y. DFT and experimental studies of oxygen adsorption on galena surface bearing Ag, Mn, Bi and Cu impurities. Miner. Eng. 2015, 71, 170–179. [Google Scholar]
- Speziale, S.; Duffy, T.S. Single-crystal elastic constants of fluorite (CaF2) to 9.3 GPa. Phys. Chem. Miner. 2002, 29, 465–472. [Google Scholar] [CrossRef]
- Payne, M.C.; Teter, M.P.; Allan, D.C.; Arias, T.A. Iterative minimization techniques for ab initio total-energy calculations: Molecular dynamics and conjugate gradients. Rev. Mod. Phys. 1992, 64, 1045. [Google Scholar] [CrossRef]
- Perdew, J.P.; Wang, Y. Accurate and simple analytic representation of the electron-gas correlation energy. Phys. Rev. B Condens. Matter 1992, 45, 13244. [Google Scholar] [CrossRef] [PubMed]
- Vanderbilt, D. Soft self-consistent pseudopotentials in a generalized eigenvalue formalism. Phys. Rev. B Condens. Matter 1990, 41, 7892. [Google Scholar] [CrossRef] [PubMed]
- Vilaplana, R.; Santamaríapérez, D.; Gomis, O.; Manjόn, F.J.; González, J.; Segura, A.; Muñoz, A.; Rodríguez-Hernández, P.; Pérez-González, E.; Marín-Borráa, V.; et al. Structural and vibrational study of Bi2Se3 under high pressure. Phys. Rev. B 2011, 84, 1160–1164. [Google Scholar]
- Bilal, M.; Ahmad, I.; Aliabad, H.A.R.; Asadabadi, S.J. Detailed DFT studies of the band profiles and optical properties of antiperovskites SbNCa 3, and BiNCa 3. Comput. Mater. Sci. 2014, 85, 310–315. [Google Scholar] [CrossRef]
- Chadi, D.J. Special points for Brillouin-zone integrations. Phys. Rev. B Condens. Matter 1977, 16, 5188–5192. [Google Scholar] [CrossRef]
- Macdonald, A.H. Comment on special points for Brillouin-zone integrations. Phys. Rev. B Condens. Matter 1978, 18, 5897–5899. [Google Scholar] [CrossRef]
- Evarestov, R.A.; Smirnov, V.P. Modification of the Monkhorst-Pack special points meshes in the Brillouin zone for density functional theory and Hartree-Fock calculations. Phys. Rev. B 2004, 70, 155–163. [Google Scholar] [CrossRef]
- Ramíarez, R.; Böhm, M.C. Simple geometric generation of special points in brillouin-zone integrations. Two-dimensional bravais lattices. Int. J. Quantum Chem. 1986, 30, 391–411. [Google Scholar] [CrossRef]
- Laasonen, K.; Pasquarello, A.; Car, R.; Lee, C.; Vanderbilt, D. Car-Parrinello molecular dynamics with Vanderbilt ultrasoft pseudopotentials. Phys. Rev. B Condens. Matter 1993, 47, 10142. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Kresse, G.; Furthmüller, J.; Hafner, J. Chemisorption of H on Pd(111): An ab initio approach with ultrasoft pseudopotentials. Phys. Rev. B Condens. Matter 1996, 54, 2157–2166. [Google Scholar] [CrossRef] [PubMed]
- Furthmüller, J.; Hafner, J.; Kresse, G. Ab initio calculation of the structural and electronic properties of carbon and boron nitride using ultrasoft pseudopotentials. Phys. Rev. B Condens. Matter 1994, 50, 15606–15622. [Google Scholar] [CrossRef] [PubMed]
- Wood, A.; Giersig, M.; Mulvaney, P. Fermi Level Equilibration in Quantum Dot-Metal Nanojunctions. J. Phys. Chem. B 2001, 105, 8810–8815. [Google Scholar] [CrossRef]
- Himpsel, F.J.; Hollinger, G.; Pollak, R.A. Determination of the Fermi-level pinning position at Si(111) surfaces. Phys. Rev. B Condens. Matter 1983, 28, 7014–7018. [Google Scholar] [CrossRef]
- Tengstedt, C.; Osikowicz, W.; Salaneck, W.R.; Parker, L.D.; Jsu, C.-H.; Fahlman, M. Fermi-level pinning at conjugated polymer interfaces. Appl. Phys. Lett. 2006, 88, 121. [Google Scholar] [CrossRef]
- Chen, J.H.; Wang, L.; Chen, Y.; Li, Y.Q.; Guo, J. Density functional theory of effects of vacancy defects on electronic structure and flotation of galena. Chin. J. Nonferr. Met. 2010, 20, 1815–1821. [Google Scholar]
- Imai, Y.; Mukaida, M.; Tsunoda, T. Calculation of electronic energy and density of state of iron-disilicides using a total-energy pseudopotential method, CASTEP. Thin Solid Films 2001, 381, 176–182. [Google Scholar] [CrossRef]
- Völkel, A.R.; Street, R.A.; Knipp, D. Carrier transport and density of state distributions in pentacene transistors. Phys. Rev. B 2002, 66, 248. [Google Scholar] [CrossRef]
- Roy, R.K.; Hirao, K.; Krishnamurty, S.; Pal, S. Mulliken population analysis based evaluation of condensed Fukui function indices using fractional molecular charge. J. Chem. Phys. 2001, 115, 2901–2907. [Google Scholar]
- Carbó-Dorca, R.; Bultinck, P. Quantum Mechanical Basis for Mulliken Population Analysis. J. Math. Chem. 2004, 36, 231–239. [Google Scholar] [CrossRef]
- Huzinaga, S.; Narita, S. Mulliken Population Analysis and Point Charge Model of Molecules. Isr. J. Chem. 2013, 19, 242–254. [Google Scholar] [CrossRef]
- Gao, Z.Y.; Li, C.W.; Sun, W.; Hu, Y.H. Anisotropic surface properties of calcite: A consideration of surface broken bonds. Colloid Surface A 2017, 520, 53–61. [Google Scholar] [CrossRef]
- Gao, Z.Y.; Sun, W.; Hu, Y.H. Mineral cleavage nature and surface energy: Anisotropic surface broken bonds consideration. Trans. Nonferr. Met. Soc. China 2014, 24, 2930–2937. [Google Scholar] [CrossRef]
- Gao, Z.Y.; Hu, Y.H.; Sun, W.; Drelich, J.W. Surface-charge anisotropy of scheelite crystals. Langmuir 2016, 32, 6282–6288. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Q.; Chen, J.H.; Chen, Y. Electronic Structures and Flotation Behavior of Pyrite Containing Vacancy Defects. Acta Phys. Chim. Sin. 2010, 26, 1435–1441. [Google Scholar]
- Free, M.L.; Miller, J.D. The significance of collector colloid adsorption phenomena in the fluorite/oleate flotation system as revealed by FTIR/IRS and solution chemistry analysis. Int. J. Miner. Process. 1996, 48, 197–216. [Google Scholar] [CrossRef]
- Jańczuk, B.; González-Martín, M.L.; Bruque, J.M.; del Pozo, J.M. The influence of oleate adsorption at the fluorite/water interface on fluorite surface free energy. Appl. Surf. Sci. 1993, 72, 201–207. [Google Scholar] [CrossRef]
- Gao, Z.Y.; Sun, W.; Hu, Y.H.; Liu, X.W. Anisotropic surface broken bond properties and wettability of calcite and fluorite crystals. Nonferr. Met. Soc. China 2012, 22, 1203–1208. [Google Scholar] [CrossRef]
- Yamabe, S.; Minato, T. Frontier Orbital Theory. Chem. Educ. 1992, 40, 450–454. [Google Scholar]
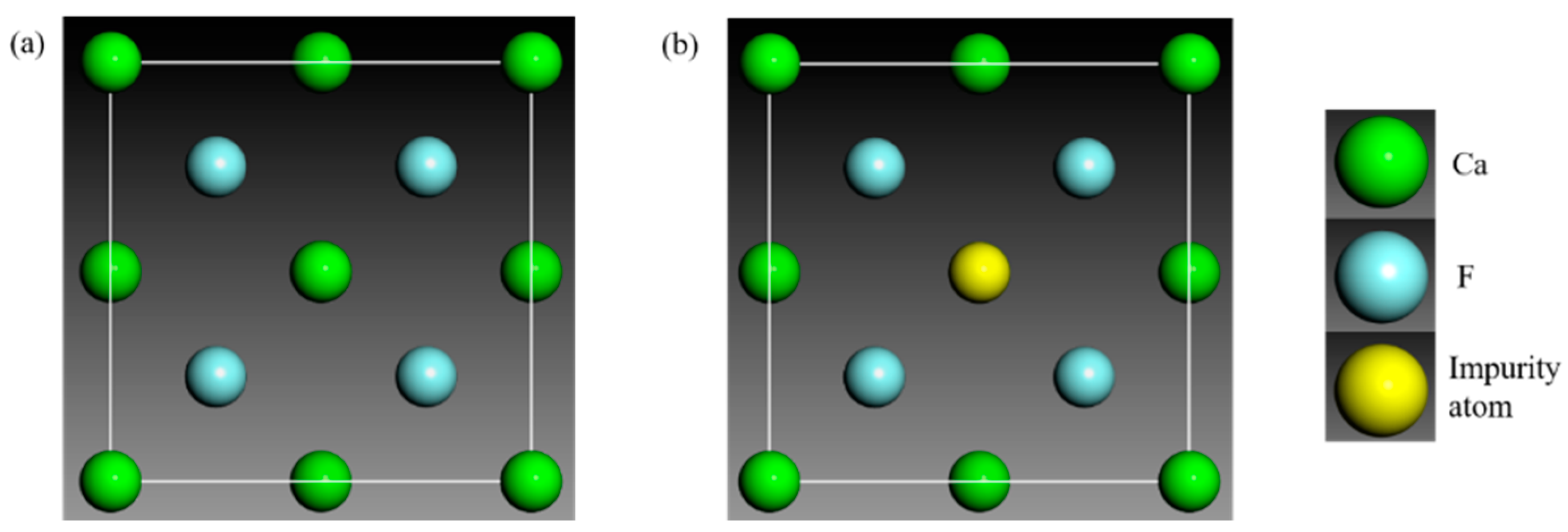
| Impurity Type | Lattice Parameter/Å | Deviation/% |
|---|---|---|
| No impurity | 5.463 | 0 |
| Ce | 5.561 | 1.79 |
| Th | 5.566 | 1.88 |
| U | 5.545 | 1.50 |
| Y | 5.542 | 1.44 |
| Species | Perfect | Ce | Th | U | Y |
|---|---|---|---|---|---|
| EF/eV | −5.698 | −2.24 | −2.54 | −2.55 | −2.509 |
| Defect Type | Species | Population | Total | Charge/e | |||
|---|---|---|---|---|---|---|---|
| s | p | d | f | ||||
| Perfect fluorite | F | 1.96 (0.04) | 5.71 (−0.71) | 0.00 | 0.00 | 7.67 | −0.67 |
| Ca | 2.15 (−0.15) | 6.00 (0) | 0.51 (1.49) | 0.00 | 8.66 | 1.34 | |
| Ce-bearing fluorite | F | 1.95 (0.05) | 5.66 (−0.66) | 0.00 | 0.00 | 7.61 | −0.61 |
| Ca | 2.19 (−0.19) | 6.00 (0) | 0.53 (1.47) | 0.00 | 8.72 | 1.28 | |
| Ce | 2.21 (−0.21) | 6.32 (−0.31) | 1.02 (0.98) | 1.40 (0.60) | 10.94 | 1.06 | |
| Th-bearing fluorite | F | 1.95 (0.05) | 5.64 (−0.64) | 0.00 | 0.00 | 7.59 | −0.59 |
| Ca | 2.18 (−0.18) | 6.00 (0) | 0.57 (1.43) | 0.00 | 8.75 | 1.25 | |
| Th | 2.28 (−0.28) | 6.31 (−0.31) | 1.88 (0.12) | 0.58 (1.42) | 11.05 | 0.95 | |
| U-bearing fluorite | F | 1.95 (0.05) | 5.64 (−0.64) | 0.00 | 0.00 | 7.59 | −0.59 |
| Ca | 2.18 (−0.18) | 6.00 (0) | 0.55 (1.45) | 0.00 | 8.73 | 1.27 | |
| U | 2.28 (−0.28) | 6.44 (−0.44) | 1.06 (0.94) | 3.31 (0.69) | 13.09 | 0.91 | |
| Y-bearing fluorite | F | 1.95 (0.05) | 5.67 (−0.67) | 0.00 | 0.00 | 7.62 | −0.62 |
| Ca | 2.20 (−0.20) | 6.00 (0) | 0.50 (1.5) | 0.00 | 8.70 | 1.30 | |
| Y | 2.30 (−0.30) | 6.32 (−0.32) | 1.32 (1.68) | 0.00 | 9.93 | 1.07 | |
| Defect Type | Bond | Population | Length (Å) |
|---|---|---|---|
| Perfect fluorite | F–Ca | 0.08 | 2.3590 |
| F–F | −0.04 | 2.7240 | |
| Ce-bearing fluorite | F–Ca | 0.07 | 2.3890 |
| F–F | −0.03 | 2.7126 | |
| F–Ce | 0.11 | 2.4668 | |
| Th-bearing fluorite | F–Ca | 0.06 | 2.3916 |
| F–F | −0.03 | 2.7164 | |
| F–Th | 0.10 | 2.4681 | |
| U-bearing fluorite | F–Ca | 0.07 | 2.3866 |
| F–F | −0.03 | 2.7216 | |
| F–U | 0.12 | 2.4447 | |
| Y-bearing fluorite | F–Ca | 0.07 | 2.3806 |
| F–F | −0.03 | 2.7026 | |
| F–Y | 0.13 | 2.4588 |
| Defect Type | Atomic Orbital Coefficient of LUMO |
|---|---|
| Perfect fluorite | 0.323Ca + 0.306F |
| Ce-bearing fluorite | 0.216Ca + 0.283F + 0.809Ce |
| Th-bearing fluorite | 0.006Ca + 0.036F + 0.993Th |
| U-bearing fluorite | 0.037Ca + 0.041F + 0.553U |
| Y-bearing fluorite | 0.064Ca + 0.111F + 0.974Y |
| Defect Type | EHOMO/eV | ELUMO/eV | /eV |
|---|---|---|---|
| Perfect fluorite | −9.175 | −2.197 | 3.424 |
| Ce-bearing fluorite | −3.05 | −2.894 | 2.727 |
| Th-bearing fluorite | −3.303 | −3.245 | 2.376 |
| U-bearing fluorite | −4.014 | −2.962 | 2.659 |
| Y-bearing fluorite | −9.395 | −3.107 | 2.514 |
| Oleic acid | −5.621 | −0.944 | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, W.; Gao, Z.; Sun, W.; Gao, J.; Hu, Y. A Density Functional Theory Study on the Effect of Lattice Impurities on the Electronic Structures and Reactivity of Fluorite. Minerals 2017, 7, 160. https://doi.org/10.3390/min7090160
Jiang W, Gao Z, Sun W, Gao J, Hu Y. A Density Functional Theory Study on the Effect of Lattice Impurities on the Electronic Structures and Reactivity of Fluorite. Minerals. 2017; 7(9):160. https://doi.org/10.3390/min7090160
Chicago/Turabian StyleJiang, Wei, Zhiyong Gao, Wei Sun, Jiande Gao, and Yuehua Hu. 2017. "A Density Functional Theory Study on the Effect of Lattice Impurities on the Electronic Structures and Reactivity of Fluorite" Minerals 7, no. 9: 160. https://doi.org/10.3390/min7090160
APA StyleJiang, W., Gao, Z., Sun, W., Gao, J., & Hu, Y. (2017). A Density Functional Theory Study on the Effect of Lattice Impurities on the Electronic Structures and Reactivity of Fluorite. Minerals, 7(9), 160. https://doi.org/10.3390/min7090160








