Synthesis of Ultramarine from Reservoir Silts
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Procedures
2.3. Characterization
3. Results and Discussion
3.1.Effects of Calcination Condition
3.1.1. Reaction Temperature
3.1.2. Reaction Duration
3.2.Effects of Raw Materials Ratio
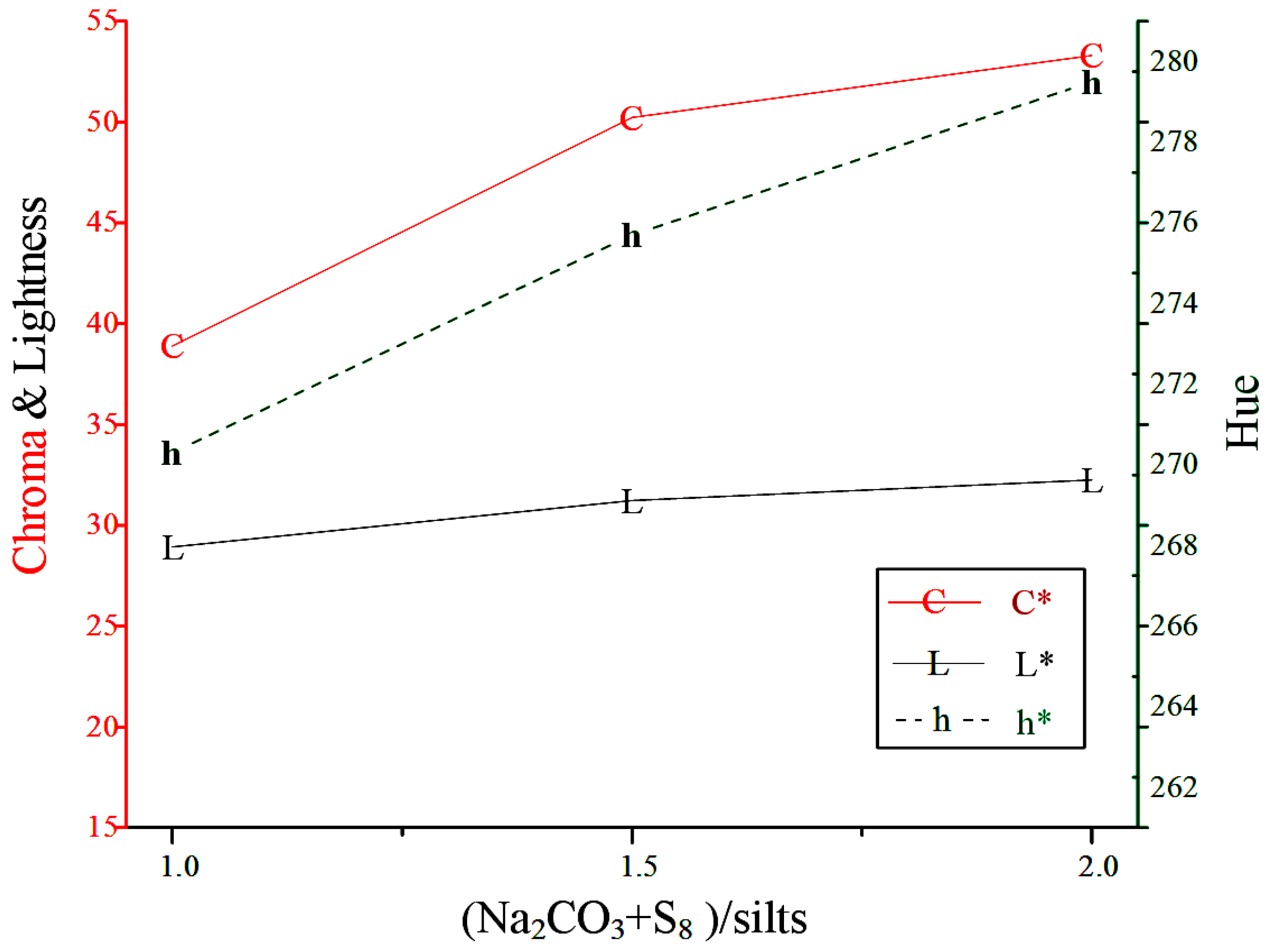
3.2.1. (Na2CO3+ S8)/Silts Ratio
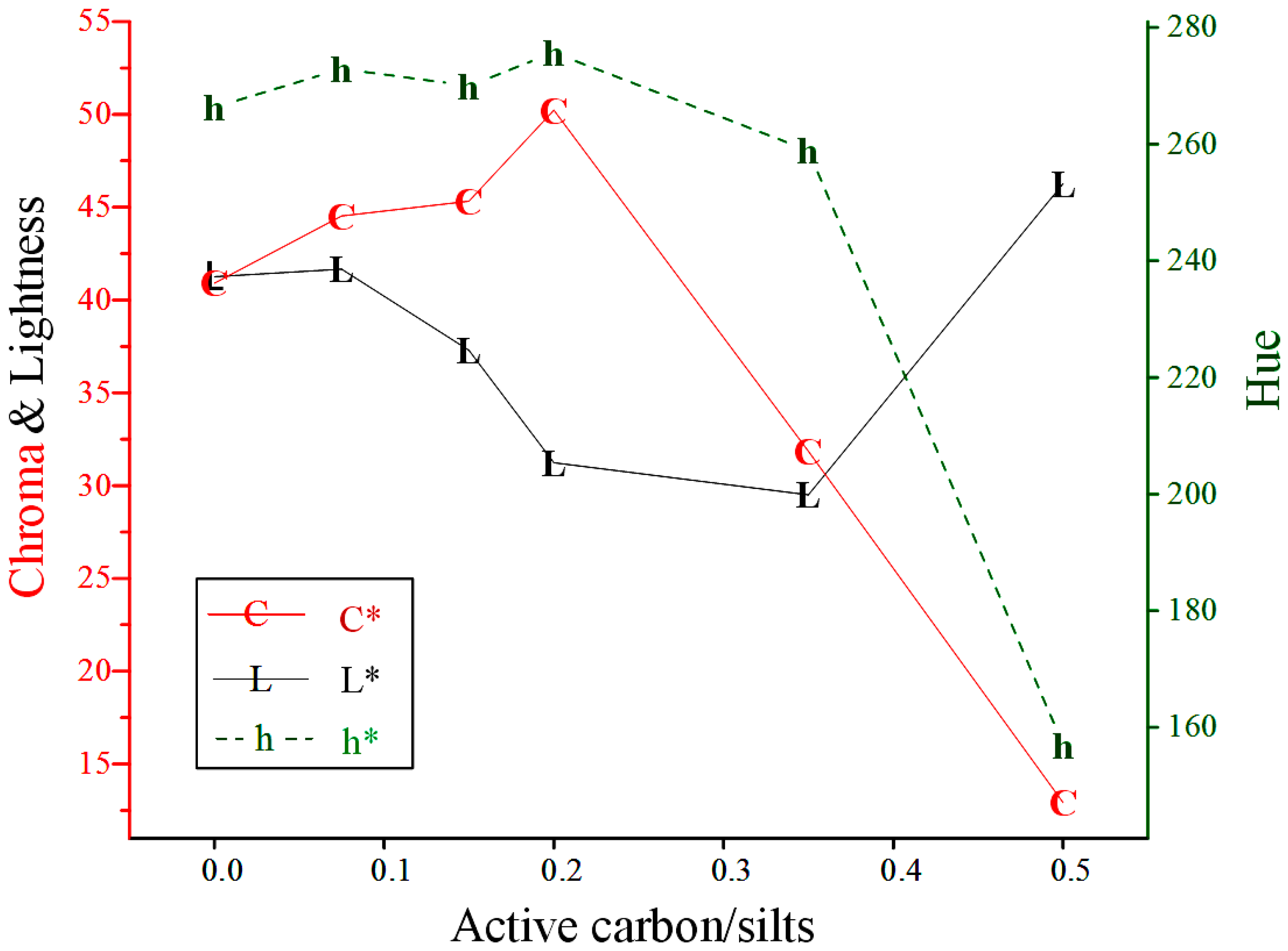
3.2.2. Active Carbon/Silts Ratio
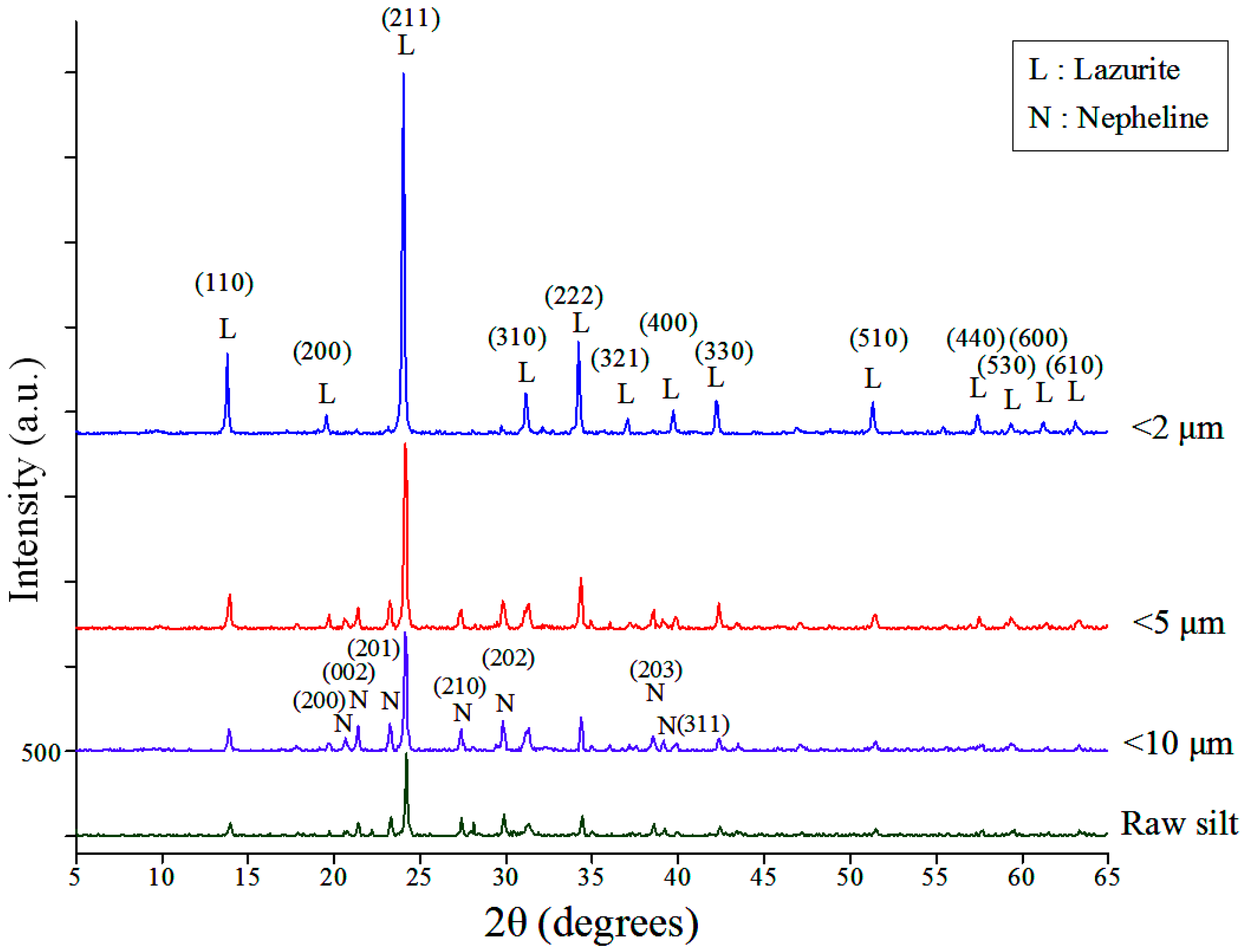
3.3. Effects of Mineral Composition of Starting Silts
4. Conclusions
Acknowledgment
Author Contributions
Conflicts of Interest
References
- Chaudhry, M.A.; Rehman, H.-U. Worldwide experience of sediment flushing through reservoirs. MURJET 2012, 31, 395–408. [Google Scholar]
- International Commission on Large Dams Introduction. Sedimentation and Sustainable Use of Reservoirs and River Systems; Stellenbosch University: Stellenbosch, South Africa, 2009; pp. 17–19. [Google Scholar]
- Sinotech Engineering Consultants LTD. The aquatic environment in Taiwan. In The Climate Change Knowledge Database and Data Integration Platform (4/4); Water Resources Agency, Ministry of Economic Affairs: Taipai, Taiwan, ROC, 2013; pp. 2–8. [Google Scholar]
- Wu, M.L. The Study on the Clay of Reservoir in Taiwan. Master’s Thesis, NCKU, Tainan, Taiwan, 2009. [Google Scholar]
- Kendrick, E.; Dann, S.E.; Hellgardt, K.; Weller, M.T. The effect of differents precursors on the synthesis of ultramarine blue using a modified test furnace. Stud. Surf. Sci. Catal. 2004, 154, 3059–3066. [Google Scholar]
- Hassan, I.; Peterson, R.C.; Grundy, H.D. The structure of lazurite, ideally Na6Ca2(A16Si6O24)S2, a member of the sodalite group. Acta Cryst. 1985, C41, 827–832. [Google Scholar]
- Climent-Pascual, E.; Sáez-Puche, R.; Gómez-Herrero, A.; Romero de Paz, J. Cluster ordering in synthetic ultramarine pigments. Microporous Mesoporous Mater. 2008, 116, 344–351. [Google Scholar] [CrossRef]
- Prener, J.S.; Ward, R. The preparation of ultramarines. J. Am. Chem. Soc. 1950, 72, 2780–2781. [Google Scholar] [CrossRef]
- Tarling, S.E.; Barnes, P. The structure and Si, Al distribution of the ultramarines. Acta Cryst. 1988, B44, 128–135. [Google Scholar] [CrossRef]
- Reinen, D.; Lindner, G.-G. The nature of the chalcogen colour centres in ultramarine-type solids. Chem. Soc. Rev. 1999, 28, 75–84. [Google Scholar] [CrossRef]
- Fabian, J.; Komiha, N.; Linguerri, R.; Rosmus, P. The absorption wavelengths of sulfur chromophors of ultramarines calculated by time-dependent density functional theory. J. Mol. Struc. Theochem 2006, 801, 63–69. [Google Scholar] [CrossRef]
- Clark, R.J.H.; Cobbold, D.G. Characterization of sulfur radical anions in solutions of alkali polysulfides in dimethylformamide and hexamethylphosphoramide and in the solid state in ultramarine blue, green, and red. Inorg. Chem. 1978, 17, 3169–3174. [Google Scholar] [CrossRef]
- Goslar, J.; Lijewski, S.; Hoffmann, S.K.; Jankowska, A.; Kowalak, S. Structure and dynamics of S3− radicals in ultramarine-type pigment based on zeolite A: Electron spin resonance and electron spin echo studies. J. Chem. Phys. 2009, 130, 204504. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, C.; Koch, W.; Lindner, G.-G.; Reinen, D. Electronic spectrum of S2−, the electron affinity of S2, and the binding energies of neutral and anionic S3 clusters. Phys. Rev. 1995, 52, 1024. [Google Scholar] [CrossRef]
- Cotton, F.A.; Harmon, J.B.; Hedges, R.M. Calculation of the ground state electronic structures and electronic spectra of di-and trisulfide radical anions by the scattered wave-SCF-Xα method. J. Am. Chem. Soc. 1976, 98, 1417–1424. [Google Scholar] [CrossRef]
- Orna, M.V. Colorant usage from antiquity to the Perkin Era. In The Chemical History of Color; Springer: Heidelberg/Berlin, Germany, 2013; pp. 47–78. [Google Scholar]
- Chivers, T. Ubiquitous trisulfur radical ion S3−. Nature 1974, 252, 32–33. [Google Scholar] [CrossRef]
- Chivers, T.; Elder, P.J.W. Ubiquitous trisulfur radical anion: Fundamentals and applications in materials science, electrochemistry, analytical chemistry and geochemistry. Chem. Soc. Rev. 2013, 42, 5996–6005. [Google Scholar] [CrossRef] [PubMed]
- Cork, W.B. Ultramarine pigments. In Pigments, Inorganic; Wiley-VCH: Weinheim, Germany, 1993; pp. 87–92. [Google Scholar]
- Landman, A.A.; De Waal, D. Fly ash as a potential starting reagent for the synthesis of ultramarine blue. Mater. Res. Bull. 2004, 39, 655–667. [Google Scholar] [CrossRef]
- Gobeltz, N.; Demortier, A.; Lelieur, J.P.; Duhayon, C. Encapsulation of the chromophores into the sodalite structure during the synthesis of the blue ultramarine pigment. J. Chem. Soc. Faraday Trans. 1998, 94, 2257–2260. [Google Scholar] [CrossRef]
- Arieli, D.; Vaughan, D.E.W.; Goldfarb, D. New Synthesis and insight into the structure of blue ultramarine pigments. J. Am. Chem. Soc. 2004, 126, 5776–5788. [Google Scholar] [CrossRef] [PubMed]
- Kumins, C.A.; Tuckahoe, N.Y. Process for Making Ultramarine. U.S. Patent 2544694, 13 March 1951. [Google Scholar]
- Lelieur, J.-P.V.; Duhayon, C.L.; Hautecoeur, N.S.; Demortier, A.M.; Lede, B.J.; Coopman, P.J.; Leghie, P. Ultramarine Pigment Synthesis Process. U.S. Patent 7632347 B2, 15 December 2009. [Google Scholar]
- Kowalak, S.; Pawłowska, M.; Miluśka, M.; Stróżyk, M.; Kania, J.; Przystajko, W. Synthesis of ultramarine from synthetic molecular sieves. Coll. Surf. A Physicochem. Eng. Asp. 1995, 101, 179–185. [Google Scholar] [CrossRef]
- Kowalak, S.; Jankowska, A.; Łączkowska, S. Preparation of various color ultramarine from zeolite A under environment-friendly conditions. Catal. Today 2004, 90, 167–172. [Google Scholar] [CrossRef]
- Kowalak, S.; Jankowska, A.; Zeidler, S. Ultramarine analogs synthesized from cancrinite. Microporous Mesoporous Mater. 2006, 93, 111–118. [Google Scholar] [CrossRef]
- Chen, K.T.; Ray, D.T. A study on the synthesis of zeolites and ultramarine pigment from reservoir sediments. Taiwan Min. Ind. 2013, 65, 19–27. [Google Scholar]
- De Menezes, R.A.; Da Paz, S.P.A.; Angélica, R.S.; De Freitas Neves, R.; Castella Pergher, S.B. Color and shade parameters of ultramarine zeolitic pigments synthesized from kaolin waste. Mater. Res. 2014, 17, 23–27. [Google Scholar] [CrossRef]
- Gobeltz, N.; Demortier, A.; Lelieur, J.P. Identification of the products of the reaction between sulfur and sodium carbonate. Inorg. Chem. 1998, 37, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Sancho, J.P.; Restrepo, O.J.; Garcia, P.; Ayala, J.; Fernandez, B.; Verdeja, L.F. Ultramarine blue from Asturian “hard” kaolins. Appl. Clay Sci. 2008, 41, 133–142. [Google Scholar] [CrossRef]
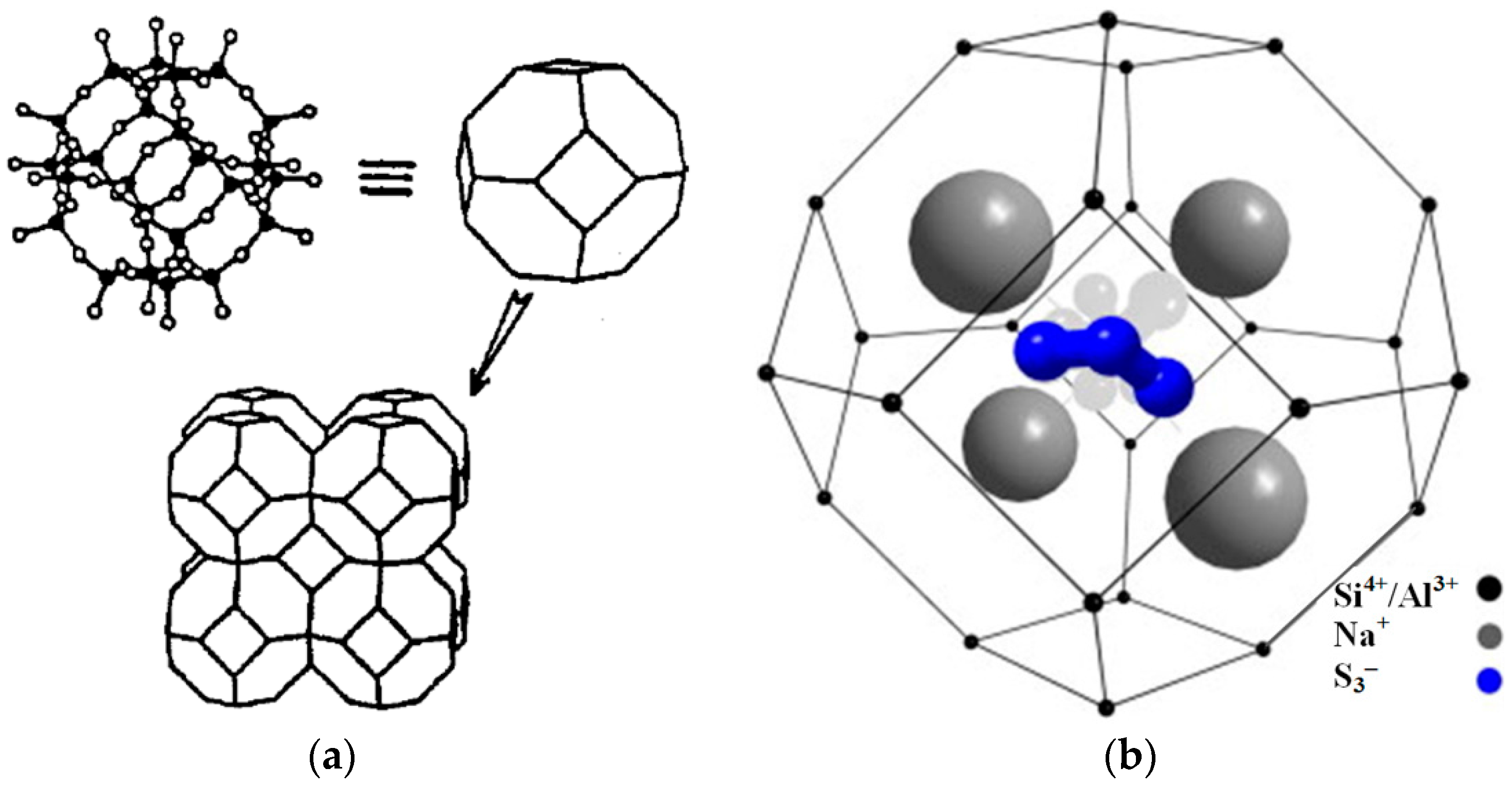
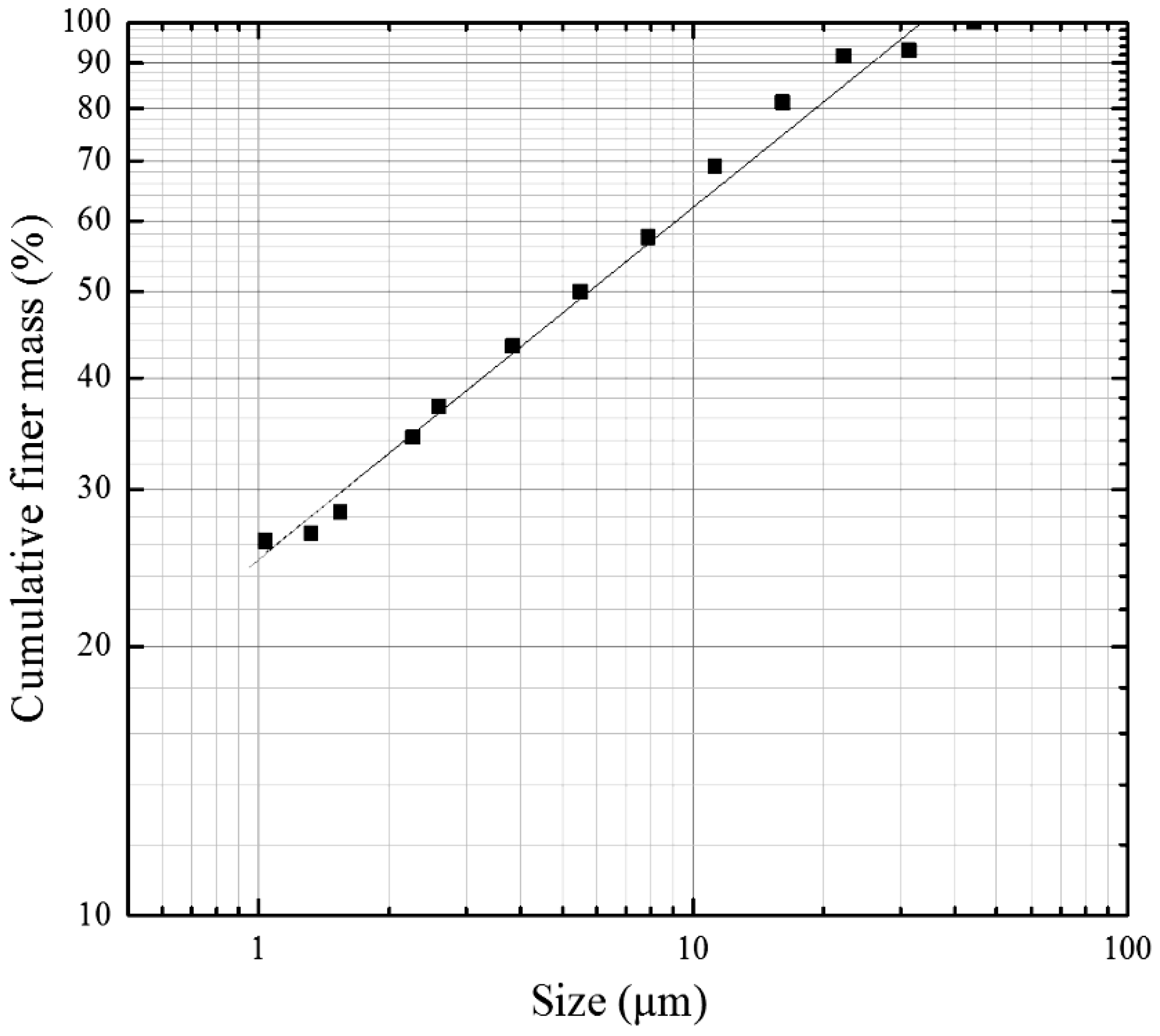


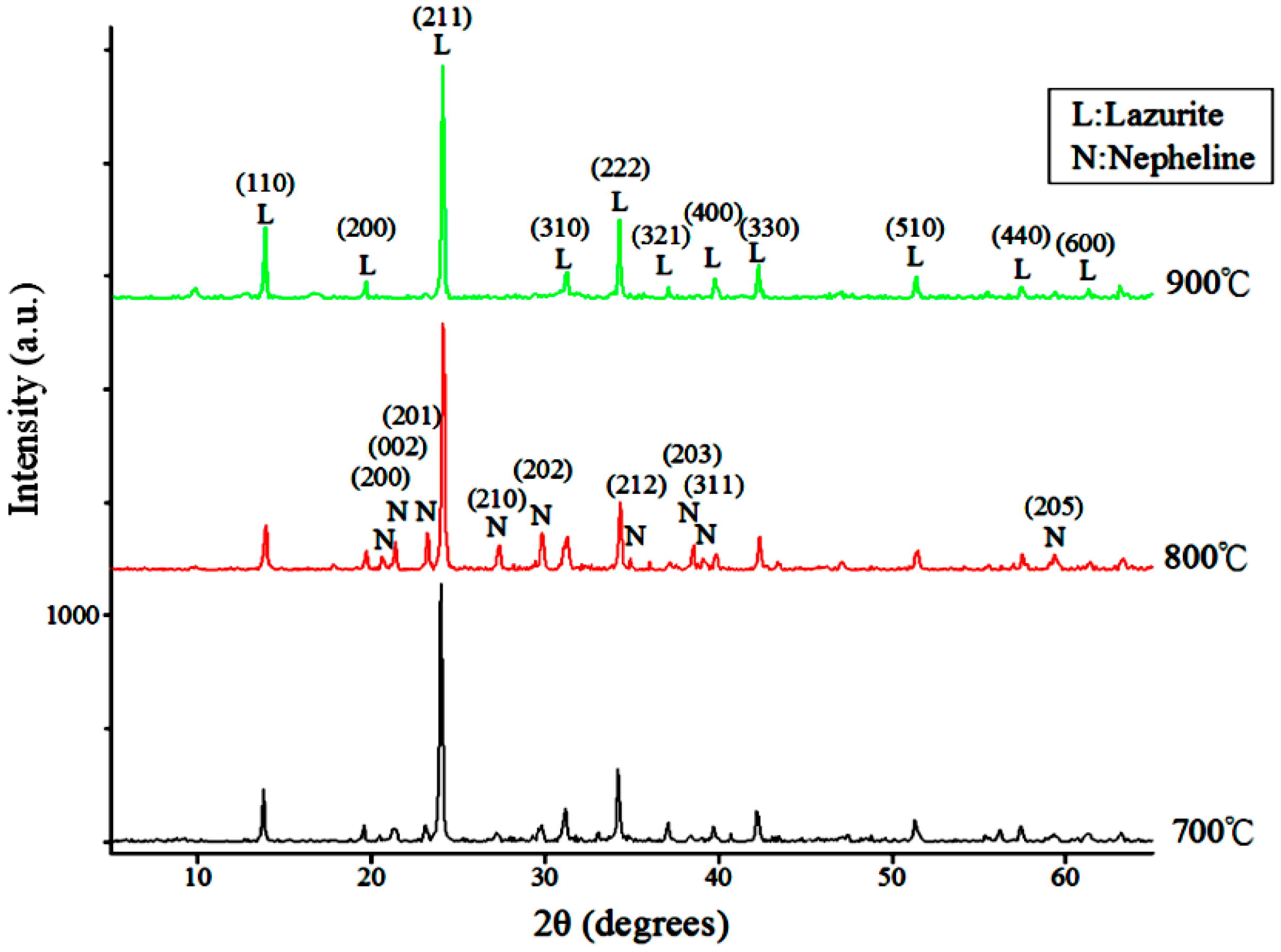
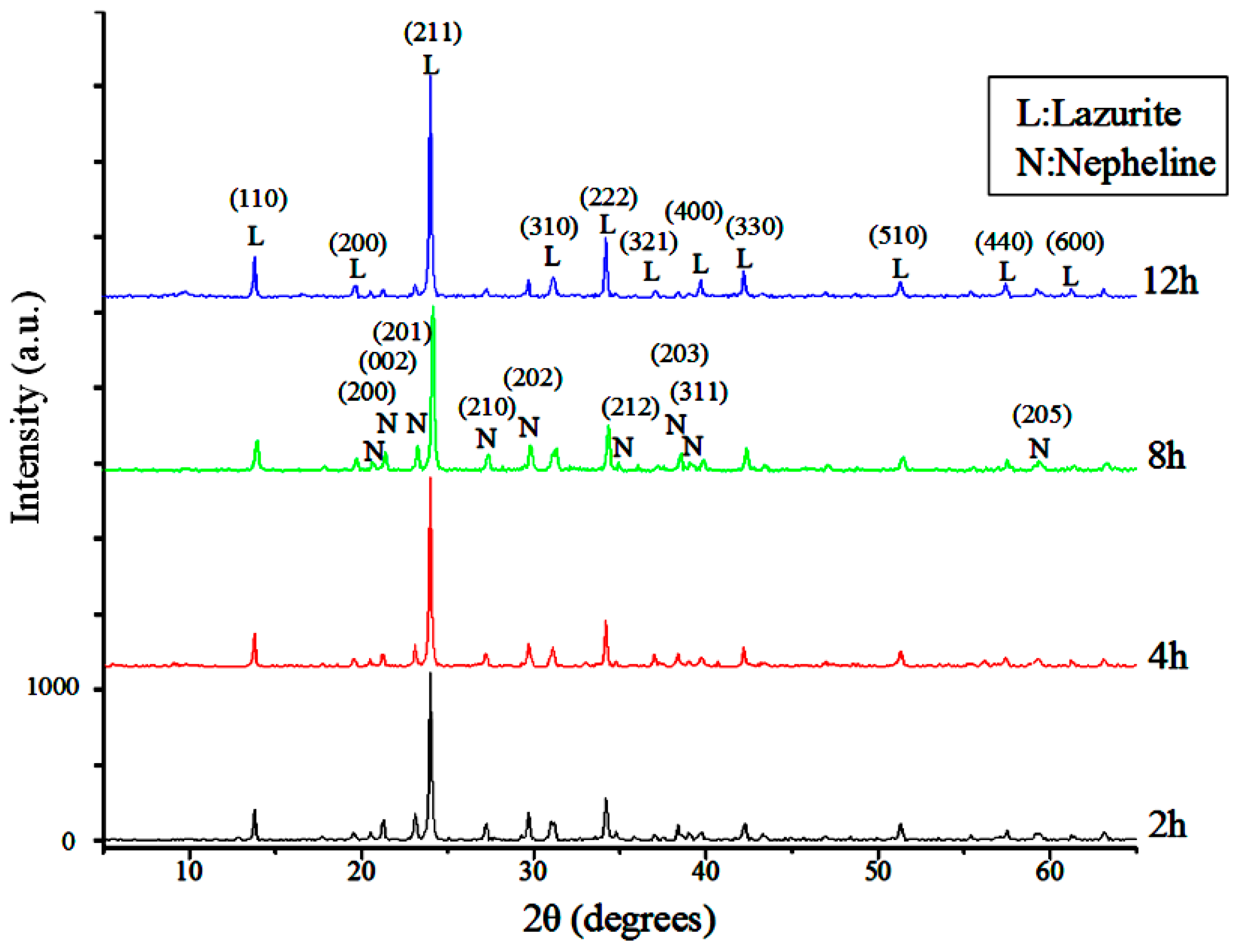

| Components | Raw | <10 μm | <5 μm | <2 μm |
|---|---|---|---|---|
| Na2O | a | a | 0.62 | 1.15 |
| MgO | 2.03 | 2.23 | 2.48 | 2.75 |
| K2O | 3.33 | 3.93 | 4.33 | 4.81 |
| CaO | 1.48 | 1.29 | 1.21 | 1.22 |
| Fe2O3 | 6.11 | 7.34 | 7.97 | 8.29 |
| Al2O3 | 19.61 | 22.25 | 24.74 | 27.74 |
| SiO2 | 63.97 | 60.86 | 56.9 | 51.48 |
| Si/Al | 2.77 | 2.32 | 1.95 | 1.58 |
| Calcination Temperature | L* | C* | h* | Pantone |
|---|---|---|---|---|
| 700 °C | 36.25 | 40.30 | 266.8 |  |
| 800 °C | 37.86 | 51.12 | 275.5 |  |
| 900 °C | 38.81 | 28.24 | 244.2 |  |
| Calcination Time | L* | C* | h* | Pantone |
|---|---|---|---|---|
| 2 h | 41.75 | 37.8 | 258.1 |  |
| 4 h | 42.27 | 36.15 | 256.6 |  |
| 8 h | 37.86 | 51.12 | 275.5 |  |
| 12 h | 39.78 | 33.08 | 250.8 |  |
| (Na2CO3 + S8)/Silts | C/Silts | L* | C* | h* | Pantone |
|---|---|---|---|---|---|
| 1 | 0.2 | 28.92 | 38.89 | 270.3 |  |
| 1.5 | 0.2 | 31.23 | 50.23 | 275.7 |  |
| 2 | 0.2 | 32.25 | 53.30 | 279.5 |  |
| (Na2CO3 + S8)/Silts | C/Silts | L* | C* | h* | Pantone |
|---|---|---|---|---|---|
| 1.5 | 0 | 41.26 | 40.90 | 266.3 |  |
| 1.5 | 0.075 | 41.66 | 44.53 | 272.9 |  |
| 1.5 | 0.15 | 37.29 | 45.33 | 270 |  |
| 1.5 | 0.2 | 37.86 | 51.12 | 275.5 |  |
| 1.5 | 0.35 | 29.50 | 31.86 | 258.9 |  |
| 1.5 | 0.5 | 46.27 | 12.92 | 157 |  |
| Particle Size | L* | C* | h* | Pantone |
|---|---|---|---|---|
| Raw | 30.19 | 22.40 | 253.3 |  |
| <10 μm | 34.68 | 35.94 | 266.4 |  |
| <5μm | 37.86 | 51.12 | 275.5 |  |
| <2μm | 38.48 | 52.38 | 271.2 |  |
| Sennelier l’Aquarelle 315 serie 2 | 24.04 | 61.52 | 299.6 |  |
| Lapis lazuli bead | 26 | 23.84 | 282.7 |  |
| Ultramarine synthesized from kaolin [31] | 33.7 | 53.7 | 290.1 |  |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsiao, Y.-H.; Shen, Y.-H.; Ray, D.-T. Synthesis of Ultramarine from Reservoir Silts. Minerals 2017, 7, 69. https://doi.org/10.3390/min7050069
Hsiao Y-H, Shen Y-H, Ray D-T. Synthesis of Ultramarine from Reservoir Silts. Minerals. 2017; 7(5):69. https://doi.org/10.3390/min7050069
Chicago/Turabian StyleHsiao, Yin-Hsiu, Yun-Hwei Shen, and Dah-Tong Ray. 2017. "Synthesis of Ultramarine from Reservoir Silts" Minerals 7, no. 5: 69. https://doi.org/10.3390/min7050069
APA StyleHsiao, Y.-H., Shen, Y.-H., & Ray, D.-T. (2017). Synthesis of Ultramarine from Reservoir Silts. Minerals, 7(5), 69. https://doi.org/10.3390/min7050069






