Strategies for Reduced Acid and Metalliferous Drainage by Pyrite Surface Passivation
Abstract
:1. Introduction
2. Materials and Methodology
2.1. KLC Tests Using Synthetic Mine Waste
2.2. KLC Tests Using Natural Iron Ore Wastes
2.3. Instrumental Analysis
3. Results and Discussion
3.1. KLC Tests Using Synthetic Waste
3.1.1. Leachate Chemistry
3.1.2. SEM Study
3.2. KLC Tests Using Natural Mine Wastes
3.2.1. Sample Characterisation
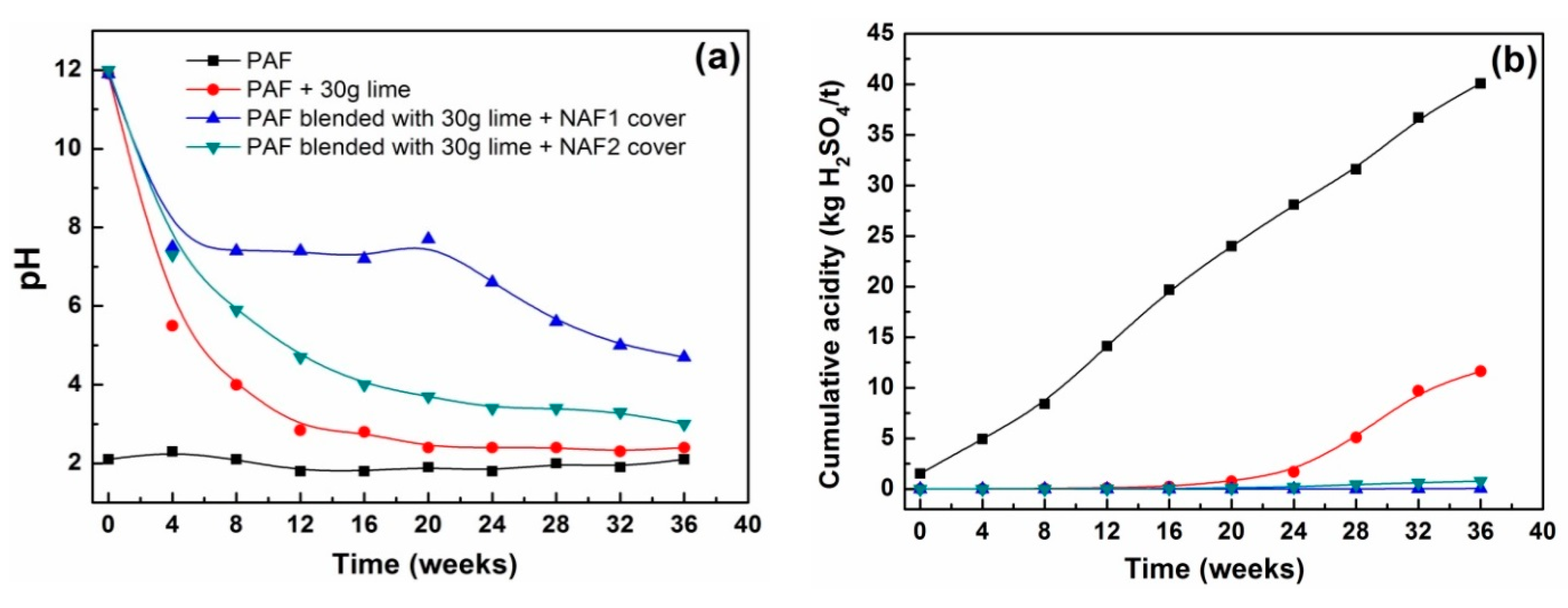
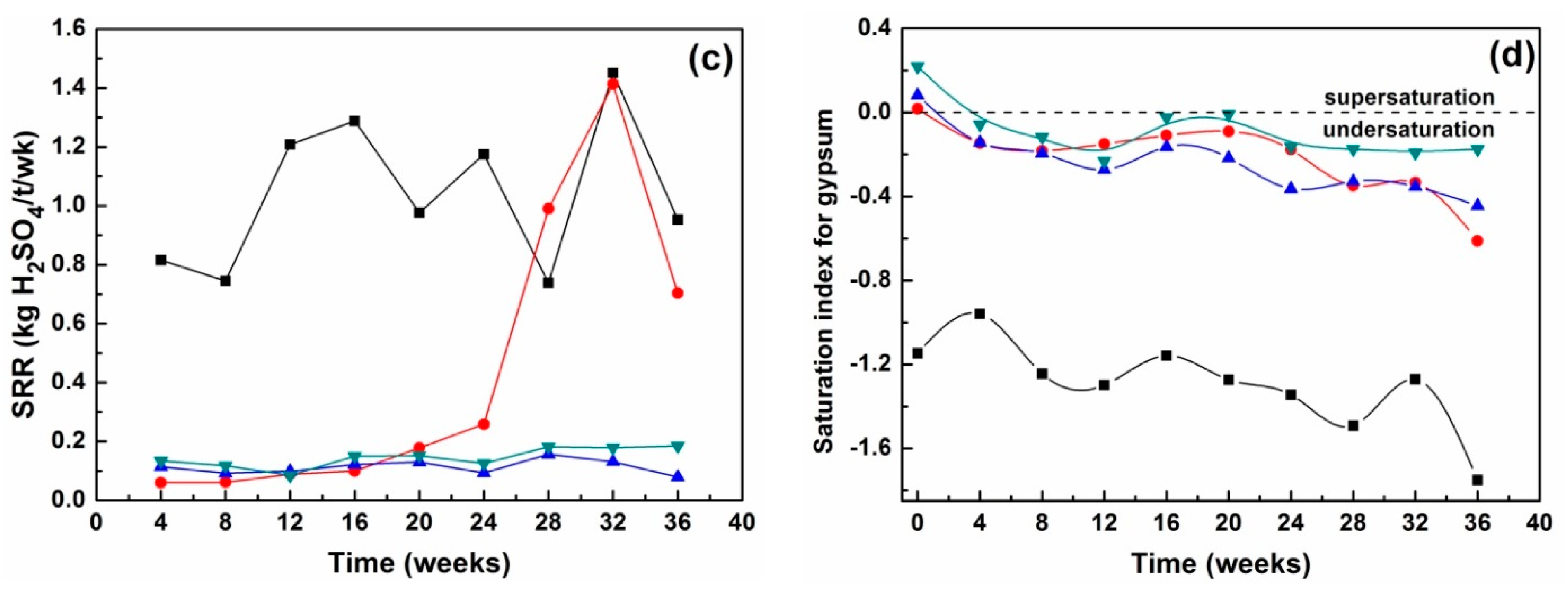
3.2.2. Leachate Chemistry
3.3. The Effectiveness of Passivation Strategies in the Treatment of Synthetic and Natural PAF Wastes
4. Conclusions and Implications
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cheng, H.; Hu, Y.; Luo, J.; Xu, B.; Zhao, J. Geochemical processes controlling fate and transport of arsenic in acid mine drainage (AMD) and natural systems. J. Hazard. Mater. 2009, 165, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Gerson, A.R.; Li, J.; Smart, R.S.C.; Saint, C.P.; Short, M.D.; Schumann, R.C. Responsible management of acid mine wastes: Geochemical and microbiological resources. In Responsible Mining: Sustainable Practices in the Mining Industry; Jarvie-Eggart, M., Muga, H.E., Eds.; SME Publications: Littleton, CO, USA, 2014; pp. 519–542. [Google Scholar]
- Schumann, R.; Miller, S.; Rusdinar, Y.; Smart, R.; Kawashima, N.; Weissman, D. Towards understanding the chemistry of limestone mitigation of acid and metalliferous drainage in waste rock dumps—From laboratory to field experiments. In Proceedings of the Sixth Australian Workshop on Acid and Metalliferous Drainage, Burnie, Austalia, 15–18 April 2005; ACMER, Sustainable Minerals Institute (Brisbane, Australia): Burnie, Austalia, 2005. [Google Scholar]
- Zeng, S.; Li, J.; Schumann, R.; Smart, R.S.C. Effect of ph and dissolved silicate on the formation of surface passivation layers for reducing pyrite oxidation. Comput. Water Energy Environ. Eng. 2013, 2, 50–55. [Google Scholar] [CrossRef]
- Smart, R.S.C.; Ciccarelli, J.; Zeng, S.; Fan, R.; Li, J.; Kawashima, N.; Gerson, A.; Schumann, R. Assessment of acid neutralization rate from site rock for amd control. In Proceedings of the 10th International Conference on Acid Rock Drainage & IWMA Annual Conference, Santiago, Chile, 21–24 April 2015.
- Gitari, W.M.; Petrik, L.F.; Etchebers, O.; Key, D.L.; Iwuoha, E.; Okujeni, C. Passive neutralisation of acid mine drainage by fly ash and its derivatives: A column leaching study. Fuel 2008, 87, 1637–1650. [Google Scholar] [CrossRef]
- Miller, S.D.; Stewart, W.S.; Rusdinar, Y.; Schumann, R.E.; Ciccarelli, J.M.; Li, J.; Smart, R.S.C. Methods for estimation of long-term non-carbonate neutralisation of acid rock drainage. Sci. Total Environ. 2010, 408, 2129–2135. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, R.; Nieto, J.M.; de Almodóvar, G.R. Utilization of fly ash to improve the quality of the acid mine drainage generated by oxidation of a sulphide-rich mining waste: Column experiments. Chemosphere 2007, 67, 1637–1646. [Google Scholar] [CrossRef] [PubMed]
- Smart, R.S.C.; Miller, S.D.; Stewart, W.S.; Rusdinar, Y.; Schumann, R.E.; Kawashima, N.; Li, J. In situ calcite formation in limestone-saturated water leaching of acid rock waste. Sci. Total Environ. 2010, 408, 3392–3402. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.; Andrina, J.; Richards, D. Overburden geochemistry and acid rock drainage scale-up investigations at the grasberg mine, Papua province, Indonesia. In Proceedings of the 6th ICARD, Cairns, Australia, 12–18 July 2003; pp. 111–121.
- Smart, R.S.C.; Skinner, B.; Levay, G.; Gerson, A.; Thomas, J.; Sobieraj, H.; Schumann, R.; Weisener, C.; Weber, P.; Miller, S. ARD Test Handbook; AMIRA International: Aurora, CO, USA, 2002; Available online: http://www.amira.com.au/documents/downloads/P387AProtocolBooklet.pdf (accessed on 13 March 2017).
- Davis, B.; Bourgeot, N.; Taylor, J. Using kinetic geochemical testwork to assist with mine planning, operations and post closure. In Proceedings of the Eighth Australian Workshop on Acid and Metalliferous Drainage, Adelaide, Australia, 29 April–2 May 2014; Miller, H., Preuss, L., Eds.; JKTechPty Ltd.: Adelaide, Australia, 2014; pp. 281–294. [Google Scholar]
- Rietveld, H. Line profiles of neutron powder-diffraction peaks for structure refinement. Acta Crystallogr. 1967, 22, 151–152. [Google Scholar] [CrossRef]
- Rietveld, H. A profile refinement method for nuclear and magnetic structures. J. Appl. Crystallogr. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Qian, G.; Li, Y.; Gerson, A.R. Applications of surface analytical techniques in earth sciences. Surf. Sci. Rep. 2015, 70, 86–133. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Appelo, C.A.J. Users’s Guide to Phreeqc (Version 2)—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; US Geological Society: Denver, CO, USA, 1999; pp. 1–326.
- Fan, R.; Short, M.D.; Zeng, S.; Qian, G.; Li, J.; Schumann, R.; Kawashima, N.; Smart, R.S.C.; Gerson, A.R. The formation of surface passivating layers on pyrite for reduced acid rock drainage. Environ. Sci. Technol. 2017. submitted. [Google Scholar]
- Schumann, R.; Kawakami, N.; Li, J.; Miller, S.; Smart, R.S.C.; Stewart, W. Passivating Surface Layer Formation on Pyrite in Neutral Rock Drainage. In Proceedings of the Securing the Future 8th ICARD, Skellefteå, Sweden, 23–26 June 2009; pp. 1–10. Available online: http://search.ror.unisa.edu.au/media/researcharchive/open/9915910957201831/53108999780001831 (accessed on 13 March 2017).
- Miller, S.; Schumann, R.C.; Smart, R.S.C.; Rusdinar, Y. ARD Control by Limestone Induced Armouring and Passivation of Pyrite Minerals Surfaces. In Proceedings of the Securing the Future and 8th ICARD, Skellefteå, Sweden, 23–26 June 2009; p. 10. Available online: http://www.inap.com.au/documents/Stuart_Miller_B6_T4_ARD-Control-by-Limestone-Induced-Armouring-and-Passivation-of-Pyrite-Min.pdf (accessed on 13 March 2017).
- Gerson, A.; Smart, R.S.C.; Li, J.; Kawashima, N.; Fan, R.; Zeng, S.; Schumann, R.; Levay, G.; Dielemans, P.; Mc Latchie, P.; et al. Mineralogy of mine site neutralising materials: A missing link in amd control planning. In Proceedings of the Eighth Australian Workshop on Acid and Metalliferous Drainage, Adelaide, Australia, 29 April–2 May 2014; Miller, H., Preuss, L., Eds.; JKTech Pty Ltd.: Adelaide, Australia, 2014; pp. 313–324. [Google Scholar]
- Sobek, A.A.; Schuller, W.A.; Freeman, J.R.; Smith, R.M. Field and Laboratory Methods Applicable to Overburdens and Minesoils; EPA-600/2-78-054; U.S. Environmental Protection Agency: Cincinnati, OH, USA, 1978; pp. 47–50.
- Schumann, R.; Stewart, W.; Miller, S.; Kawashima, N.; Li, J.; Smart, R. Acid-base accounting assessment of mine wastes using the chromium reducible sulfur method. Sci. Total Environ. 2012, 424, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Brocks, J.J.; Summons, R.E.; Buick, R.; Logan, G.A. Origin and significance of aromatic hydrocarbons in giant iron ore deposits of the late archean hamersley basin, Western Australia. Org. Geochem. 2003, 34, 1161–1175. [Google Scholar] [CrossRef]
- Hiroyoshi, N.; Miki, H.; Hirajima, T.; Tsunekawa, M. Enhancement of chalcopyrite leaching by ferrous ions in acidic ferric sulfate solutions. Hydrometallurgy 2001, 60, 185–197. [Google Scholar] [CrossRef]
- Li, Y.; Wei, Z.; Qian, G.; Li, J.; Gerson, A. Kinetics and mechanisms of chalcopyrite dissolution at controlled redox potential of 750 mv in sulfuric acid solution. Minerals 2016, 6, 83. [Google Scholar] [CrossRef]
- Moses, C.O.; Kirk Nordstrom, D.; Herman, J.S.; Mills, A.L. Aqueous pyrite oxidation by dissolved oxygen and by ferric iron. Geochim. Cosmochim. Acta 1987, 51, 1561–1571. [Google Scholar] [CrossRef]
- Bond, P.L.; Banfield, J.F. Design and performance of rrna targeted oligonucleotide probes for in situ detection and phylogenetic identification of microorganisms inhabiting acid mine drainage environments. Microb. Ecol. 2001, 41, 149–161. [Google Scholar] [PubMed]
- Brierley, C.L.; Le Roux, N.W. Bacterial leaching. CRC Crit. Rev. Microbiol. 1978, 6, 207–262. [Google Scholar] [CrossRef]
- Sánchez-Andrea, I.; Rodríguez, N.; Amils, R.; Sanz, J.L. Microbial diversity in anaerobic sediments at río tinto, a naturally acidic environment with a high heavy metal content. Appl. Environ. Microbiol. 2011, 77, 6085–6093. [Google Scholar] [CrossRef] [PubMed]
- Morales, T.A.; Dopson, M.; Athar, R.; Herbert, R.B. Analysis of bacterial diversity in acidic pond water and compost after treatment of artificial acid mine drainage for metal removal. Biotechnol. Bioeng. 2005, 90, 543–551. [Google Scholar] [CrossRef] [PubMed]
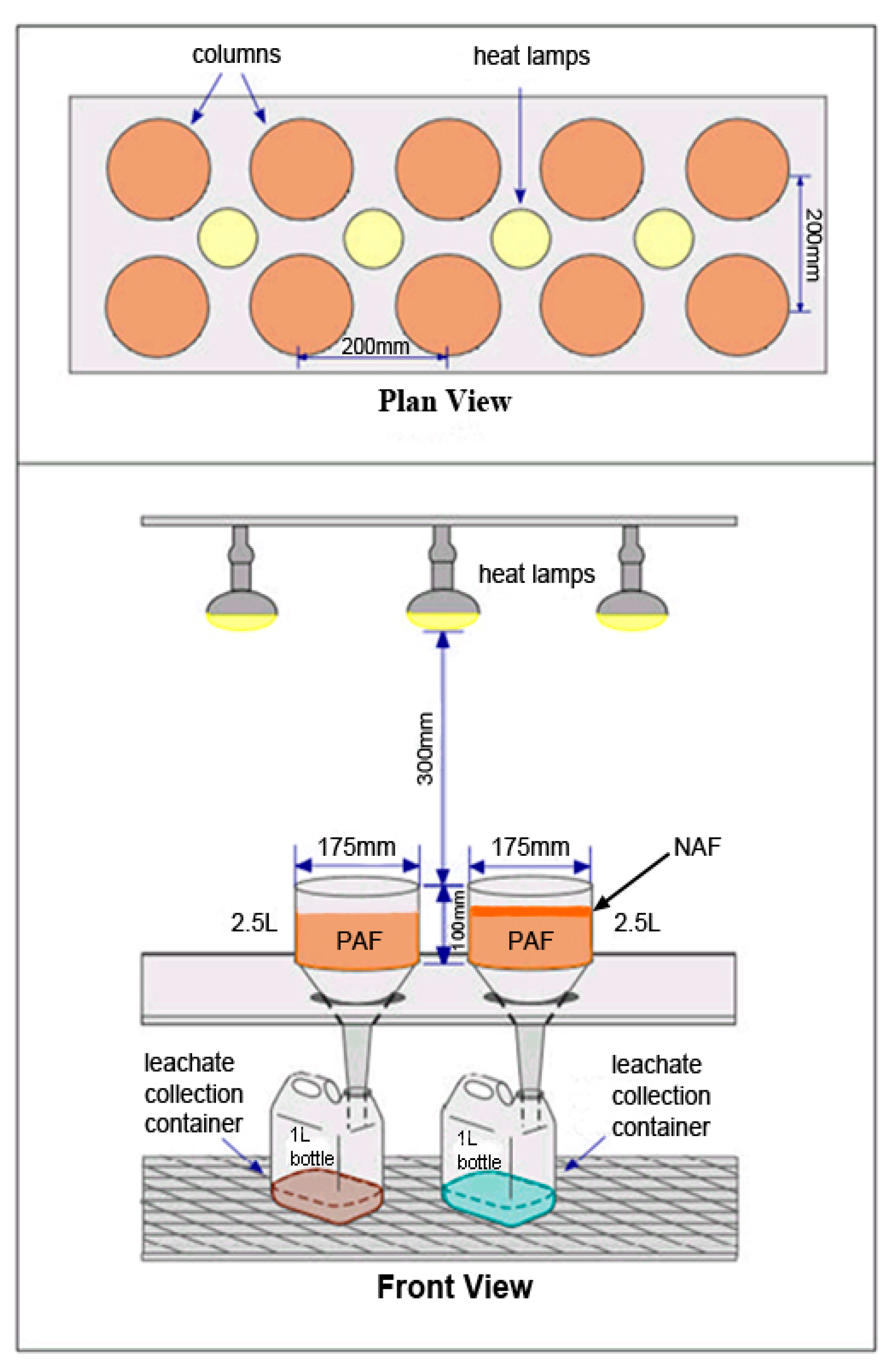
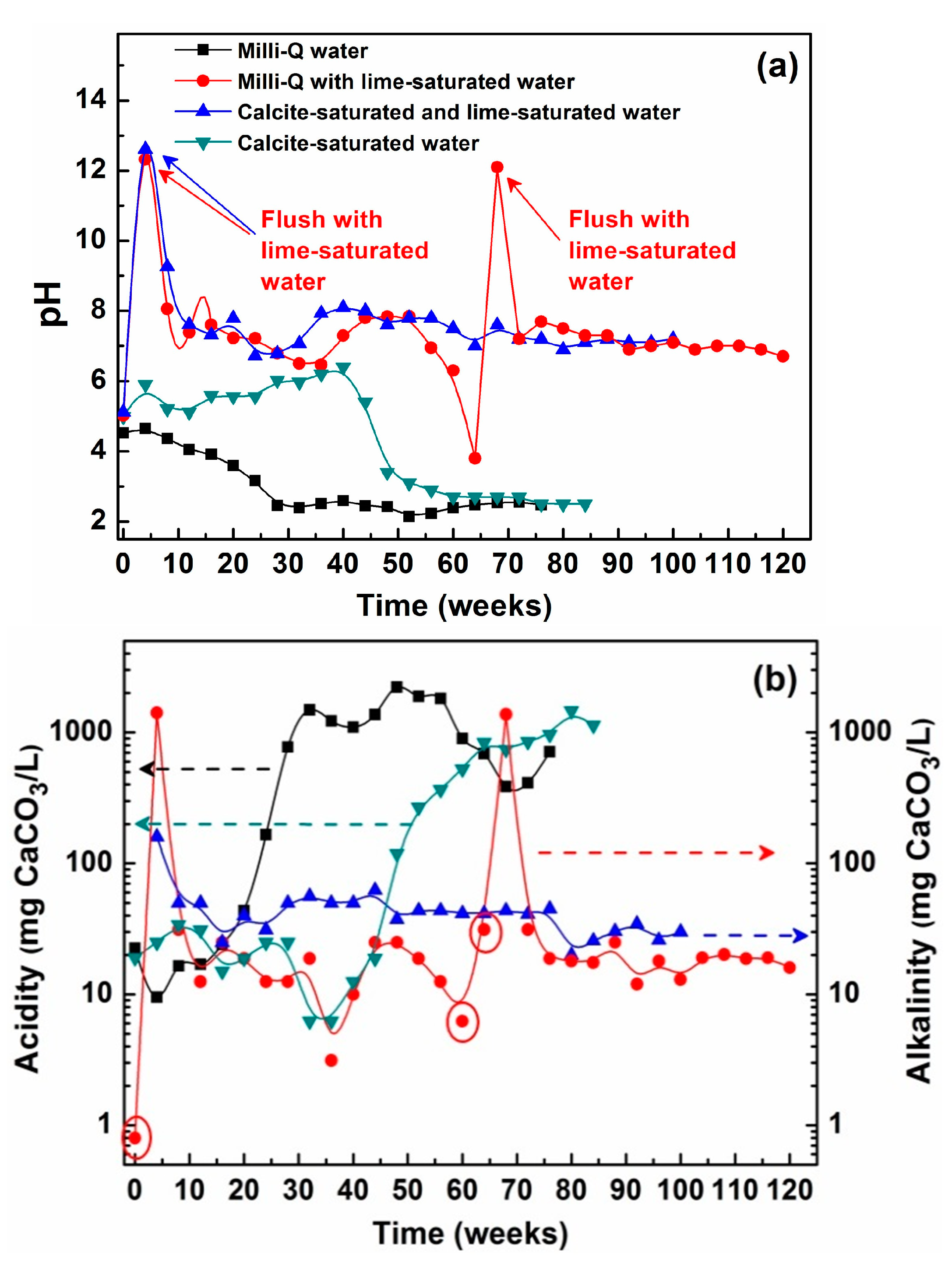

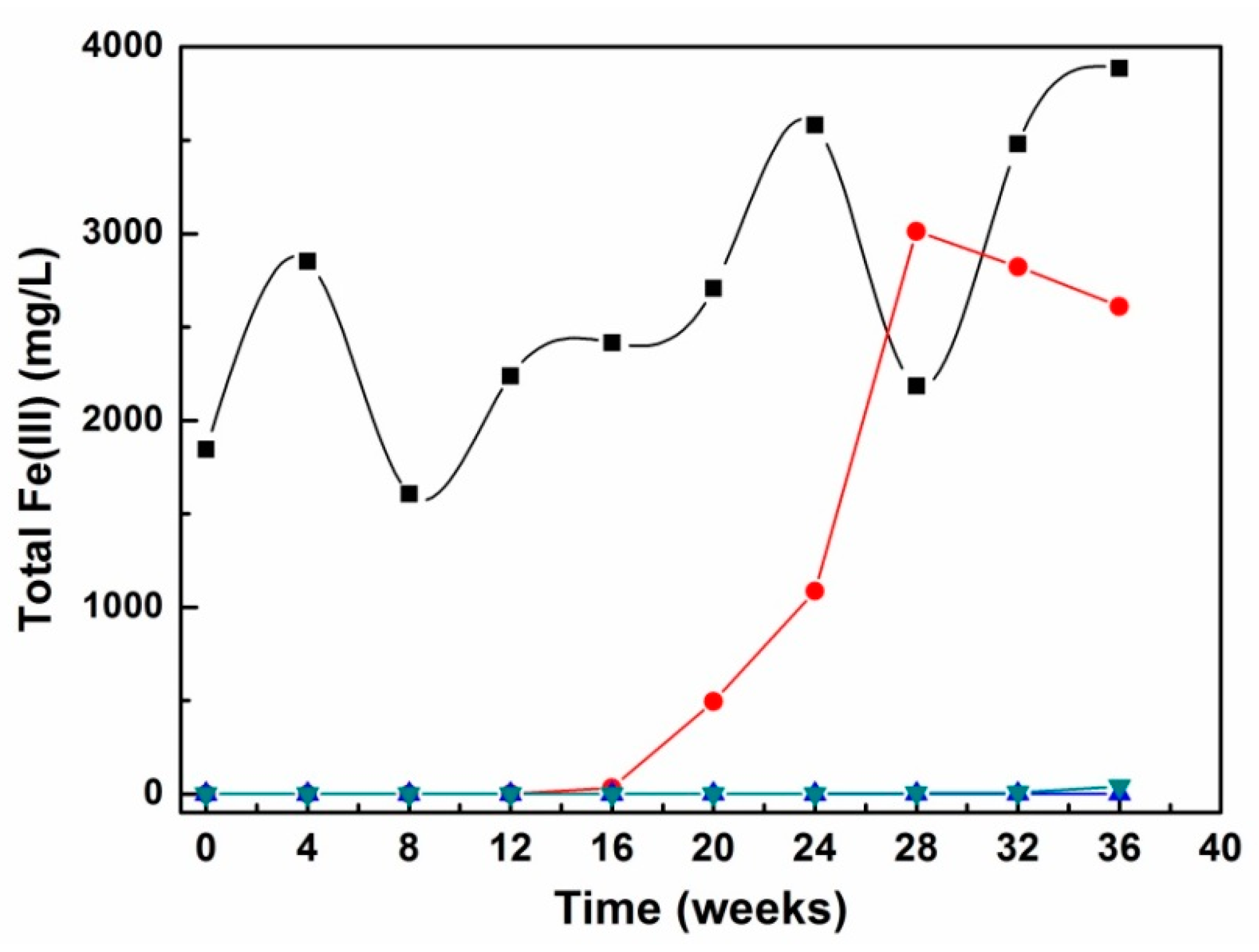
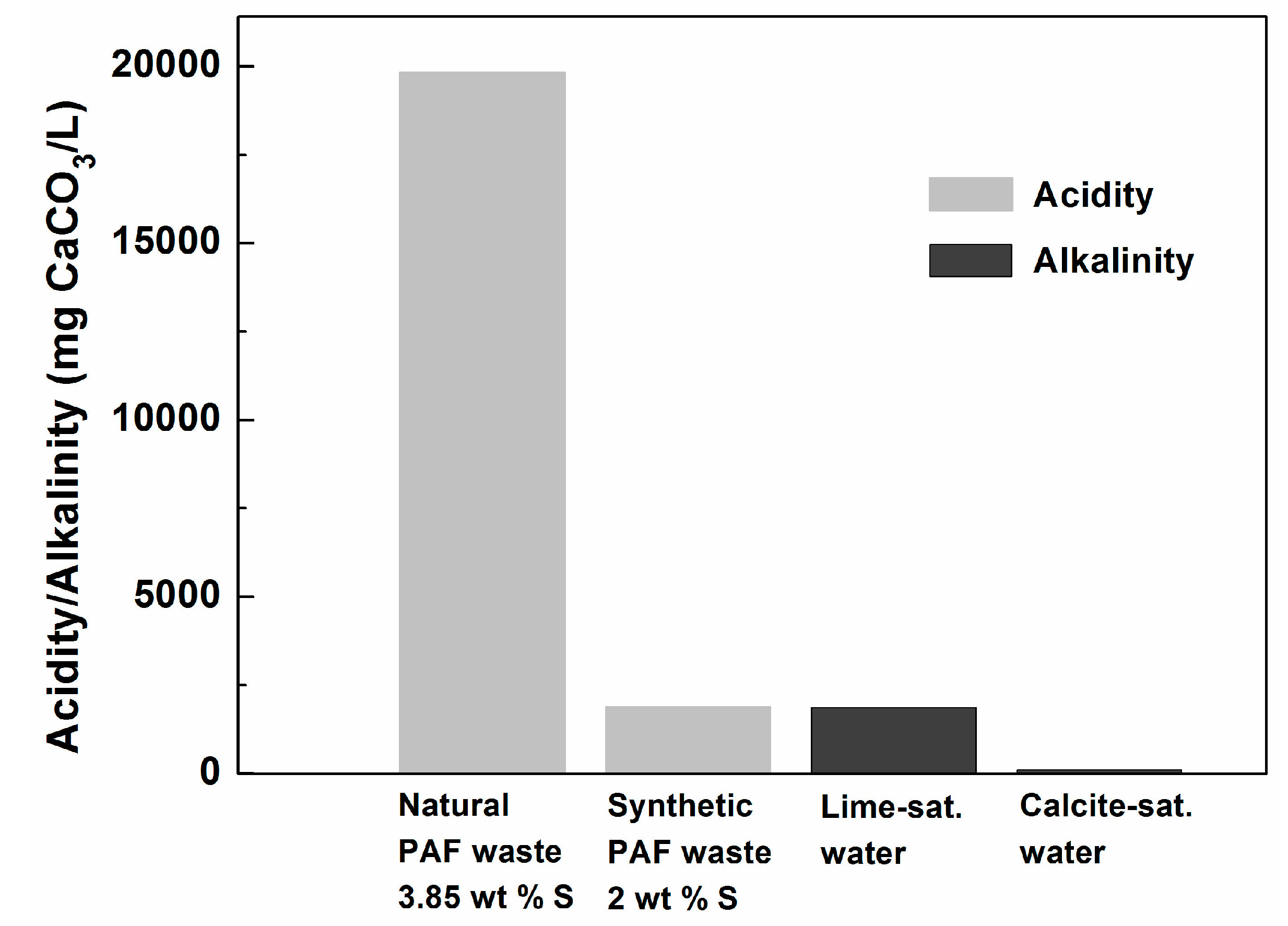
| KLC | Watering/Flushing Solution | Watering/Flushing Procedure |
|---|---|---|
| Control | Milli-Q | Milli-Q for all watering and flushing |
| Milli-Q with lime-saturated water | Milli-Q and lime-saturated water (alkalinity ≈1900 mg·CaCO3·L−1) | Milli-Q flush 0 (week 0); lime-saturated water (≈1900 mg·CaCO3·L−1) weeks 1–3 and flush 1 (week 4); Milli-Q watering thereafter until pH < 5.5, then lime-saturated watering/flushing resumed until pH > 5.5, after which Milli-Q watering/flushing until pH < 5.5 |
| Calcite-saturated and lime-saturated water | Calcite-saturated (alkalinity: ≈30 mg·CaCO3·L−1) and lime-saturated water | Same as for “Milli-Q with lime-saturated water” KLC except calcite-saturated water used for watering and flushing from flush 1 (week 4) |
| Calcite-saturated water | Calcite-saturated water | Milli-Q flush 0; calcite-saturated water thereafter |
| Sample (<4 mm) | Total Sample Weight Excl. Added Lime (kg) | NAF (kg) | KLC Configuration and Operation |
|---|---|---|---|
| PAF | 1.45 | 0 | Watering/flushing 1 as per standard method |
| PAF | 1.45 (blended with 30 g of Ca(OH)2) | 0 | Watering/flushing 1 as per standard method |
| PAF + NAF1 top cover | 1.75 (blended with 30 g of Ca(OH)2) | 0.3 | NAF added on top of PAF; watering/flushing 1 as per standard method |
| PAF + NAF2 top cover | 1.75 (blended with 30 g of Ca(OH)2) | 0.3 | NAF added on top of PAF; watering/flushing 1 as per standard method |
| Sample | ANC 1 | ANC 2 | NAG7 3 | MPA 5 | MPA* 6 | NAPP 7 | NAPP* 8 | Geochemical Classification |
|---|---|---|---|---|---|---|---|---|
| #1 (PAF) | 2.4 | 0 | 125 | 123 | 118 | 121 | 116 | PAF |
| #2 (NAF1) | 938 | 984 | / 4 | 1.5 | 1.2 | −936 | −937 | NAF |
| #3 (NAF2) | 391 | 379 | / | 1.5 | 1.2 | −389 | −390 | NAF |
| QEM-SCAN (wt %) | QXRD (wt %) | ||||||
|---|---|---|---|---|---|---|---|
| Phases | NAF1 | NAF2 | PAF | Phases | NAF1 | NAF2 | PAF |
| Fe disulphides 1 | 0.1 | 0.0 | 8.4 | Pyrite | − | − | 5 |
| Hem/Mag | 4.3 | 3.7 | 1.0 | Hematite | 9 | 4 | − |
| Geothite | 3.7 | 17.0 | 0.8 | Goethite | − | 18 | − |
| Mn minerals 2 | 0.0 | 11.0 | − | Magnetite | − | − | − |
| Dolomite | 77.2 | 37.6 | − | Dolomite | 77 | 48 | − |
| Ti minerals | 0.1 | 0.2 | 0.4 | Quartz | 2 | 1 | 42 |
| Quartz | 3.8 | 3.6 | 42.0 | Calcite | − | 5 | − |
| Feldspar | 0.8 | 0.5 | 17.4 | Nacrite | − | 7 | − |
| Mica | 2.0 | 0.8 | 24.1 | Chlorite | − | 5 | − |
| Clays | 1.6 | 3.1 | 1.4 | Muscovite | − | − | 22 |
| Iron silicates 3 | 4.1 | 19.0 | 3.5 | Microcline | − | − | 4 |
| Other silicates | 1.9 | 2.4 | 0.5 | Lizardite | − | − | 3 |
| Others | 0.4 | 1.1 | 0.3 | Amorphous 4 | 12 | 12 | 24 |
| Total | 100 | 100 | 100 | Total | 100 | 100 | 100 |
| Sample | Al | As | Au | B | Ba | Be | Bi | Ca | Cd | Co | Cr | Cu |
| PAF | 55,100 | 114 | <5 | 23 | 38 | 4 | <5 | 80 | 3 | 15 | 115 | 64 |
| NAF1 | 22,600 | <5 | <5 | 108 | 34 | <0.5 | <5 | 214,000 | <2.5 | <2.5 | 17 | <5 |
| NAF2 | 34,000 | 19 | <5 | 79 | 290 | 1 | <5 | 92,000 | <2.5 | 13 | 27 | 37 |
| Detection limit | 10 | 5 | 5 | 5 | 5 | 0.5 | 5 | 25 | 2.5 | 2.5 | 2.5 | 5.0 |
| Sample | Fe | K | La | Li | Mg | Mn | Mo | Na | Ni | P | Pb | Sb |
| PAF | 61,600 | 28,400 | 33 | 43 | 3900 | 40 | <5 | <50 | 49 | 256 | <5 | <5 |
| NAF1 | 29,700 | 1300 | 3.5 | 22 | 113,000 | 430 | <5 | 302 | <2.5 | 52 | <5 | <5 |
| NAF2 | 17,500 | 1700 | 27 | 93 | 45,300 | 27,800 | <5 | 824 | 45 | 264 | 5.5 | <5 |
| Detection limit | 5 | 50 | 2.5 | 2.5 | 25 | 5 | 5 | 50 | 2.5 | 25 | 5 | 5 |
| Sample | Se | Si | Sn | Sr | Ti | U | V | W | Zn | Zr | Cl | F |
| PAF | <5 | 269,000 | 12 | 45 | 1091 | 137 | 75 | <5 | 50 | 114 | <60 | 548 |
| NAF1 | 5.5 | 59,900 | 29 | 50 | 384 | 64 | 14 | <5 | 11 | 33 | <60 | 738 |
| NAF2 | <5 | 59,700 | 22 | 98 | 565 | 333 | 36 | <5 | 36 | 53 | <60 | 1627 |
| Detection limit | 5 | 25 | 5 | 2.5 | 5 | 50 | 5 | 5 | 5 | 5 | 60 | 100 |
| Sample | Total S | SCRS | Total C | Total organic C | Total inorganic C | |||||||
| PAF | 40,300 | 38,500 | 53,400 | 53,400 | 0 | |||||||
| NAF1 | 500 | 400 | 119,200 | 1100 | 118,100 | |||||||
| NAF2 | 500 | 400 | 46,500 | 1000 | 45,500 | |||||||
| Sample | Total Acid Generated 1 | Lime Added | Residual Calciumin Lime 2 | pH |
|---|---|---|---|---|
| Natural PAF | 38.6 | 0 | 0 | 2.1 |
| Natural PAF + lime | 15.7 | 27.4 | 25.5 | 2.4 |
| Natural PAF + lime+ NAF1 | 4.3 | 27.4 | 24.5 | 4.7 |
| Natural PAF + lime +NAF2 | 5.5 | 27.4 | 24.1 | 3.0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qian, G.; Schumann, R.C.; Li, J.; Short, M.D.; Fan, R.; Li, Y.; Kawashima, N.; Zhou, Y.; Smart, R.S.C.; Gerson, A.R. Strategies for Reduced Acid and Metalliferous Drainage by Pyrite Surface Passivation. Minerals 2017, 7, 42. https://doi.org/10.3390/min7030042
Qian G, Schumann RC, Li J, Short MD, Fan R, Li Y, Kawashima N, Zhou Y, Smart RSC, Gerson AR. Strategies for Reduced Acid and Metalliferous Drainage by Pyrite Surface Passivation. Minerals. 2017; 7(3):42. https://doi.org/10.3390/min7030042
Chicago/Turabian StyleQian, Gujie, Russell C. Schumann, Jun Li, Michael D. Short, Rong Fan, Yubiao Li, Nobuyuki Kawashima, Yan Zhou, Roger St. C. Smart, and Andrea R. Gerson. 2017. "Strategies for Reduced Acid and Metalliferous Drainage by Pyrite Surface Passivation" Minerals 7, no. 3: 42. https://doi.org/10.3390/min7030042
APA StyleQian, G., Schumann, R. C., Li, J., Short, M. D., Fan, R., Li, Y., Kawashima, N., Zhou, Y., Smart, R. S. C., & Gerson, A. R. (2017). Strategies for Reduced Acid and Metalliferous Drainage by Pyrite Surface Passivation. Minerals, 7(3), 42. https://doi.org/10.3390/min7030042








