Tellurium Enrichment in Jurassic Coal, Brora, Scotland
Abstract
:1. Introduction
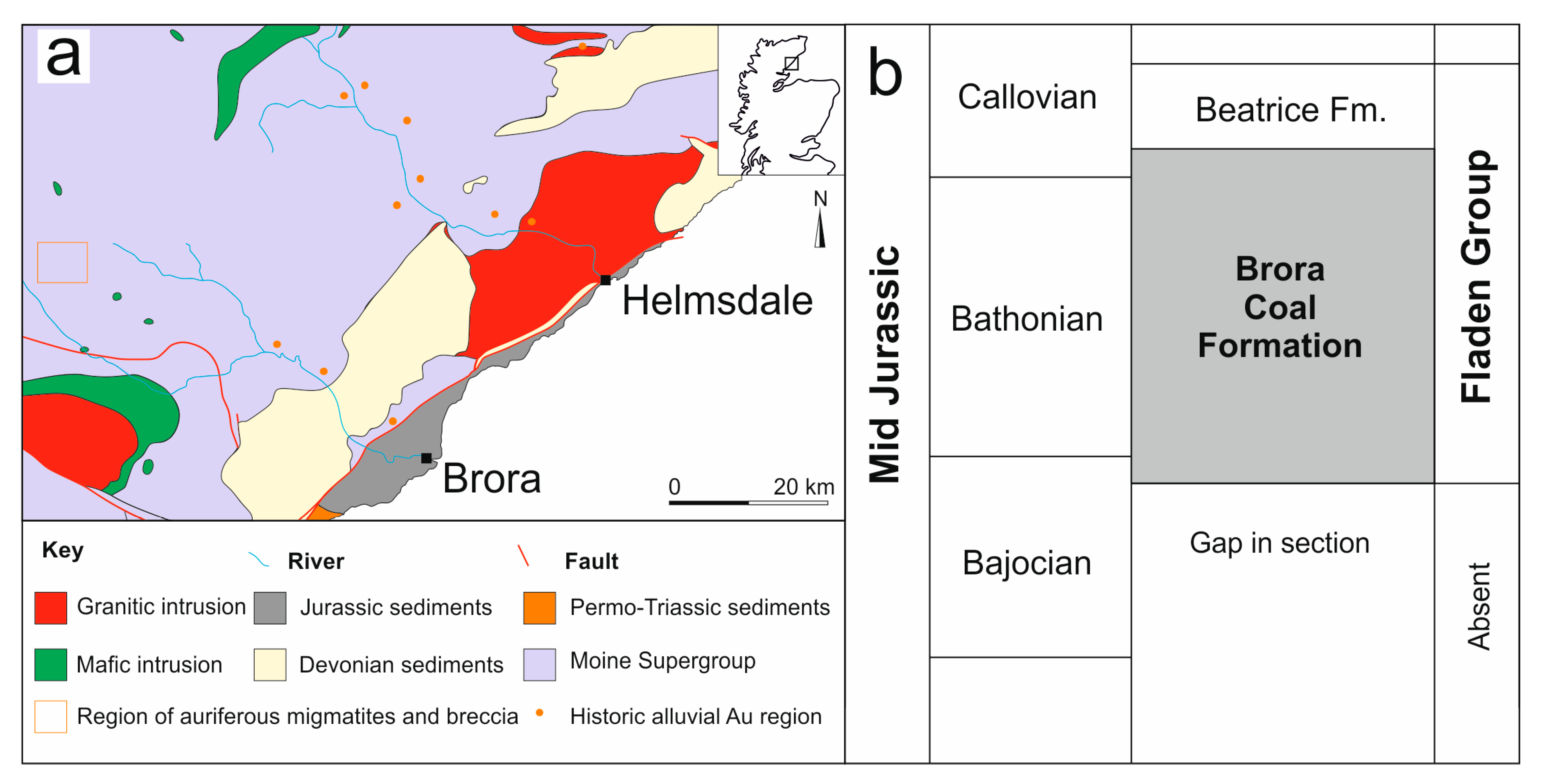
2. Geological Setting
3. Materials and Methods
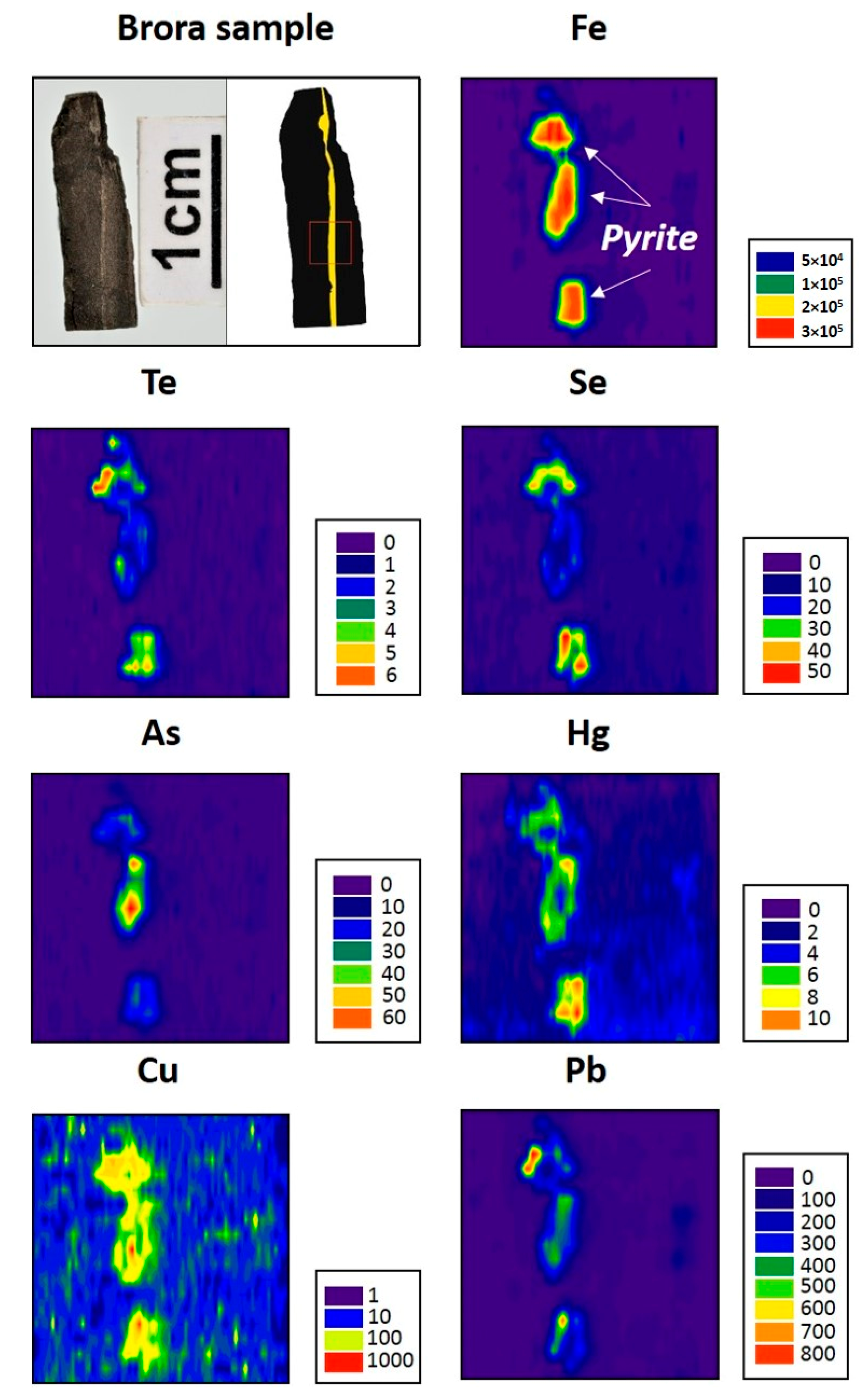
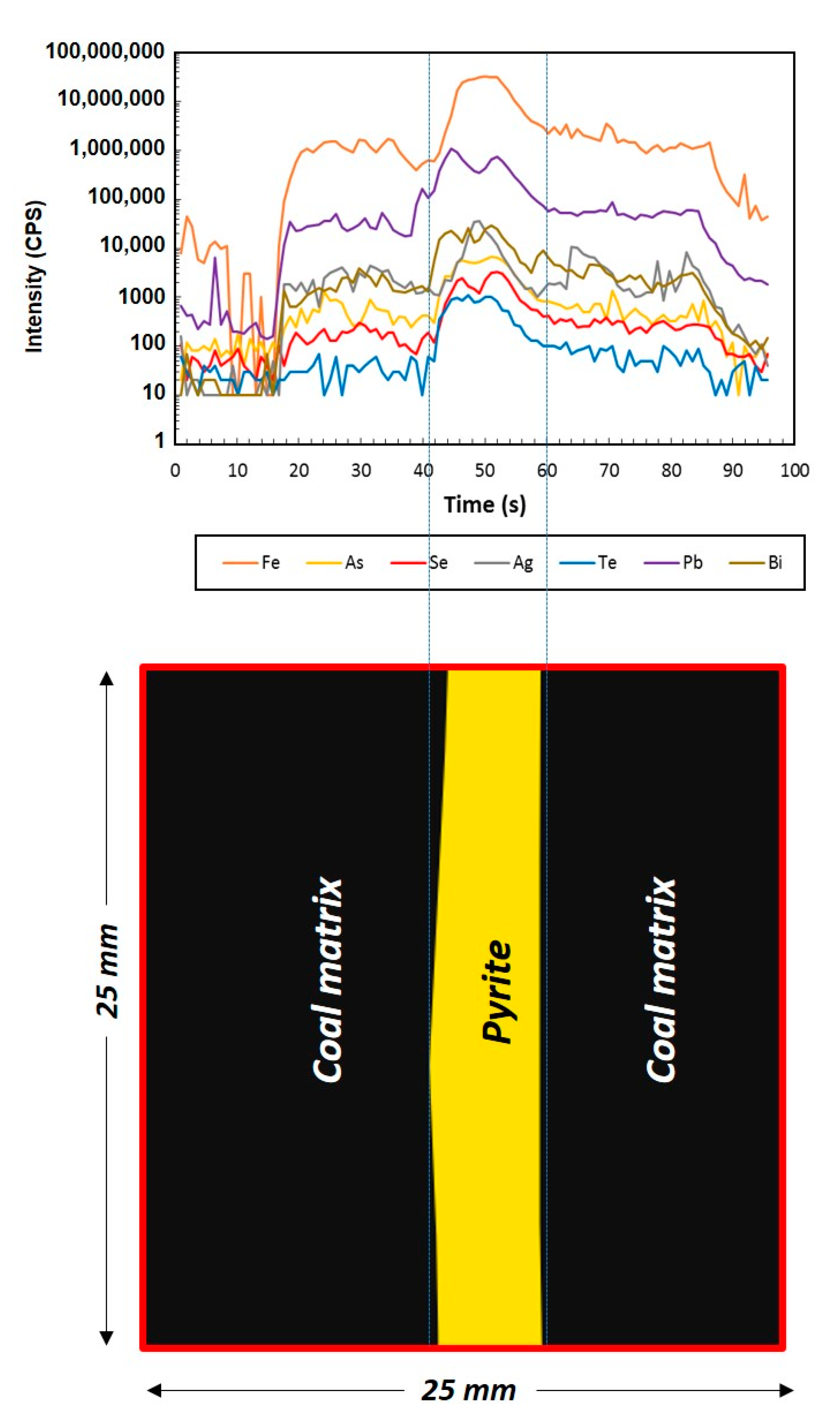
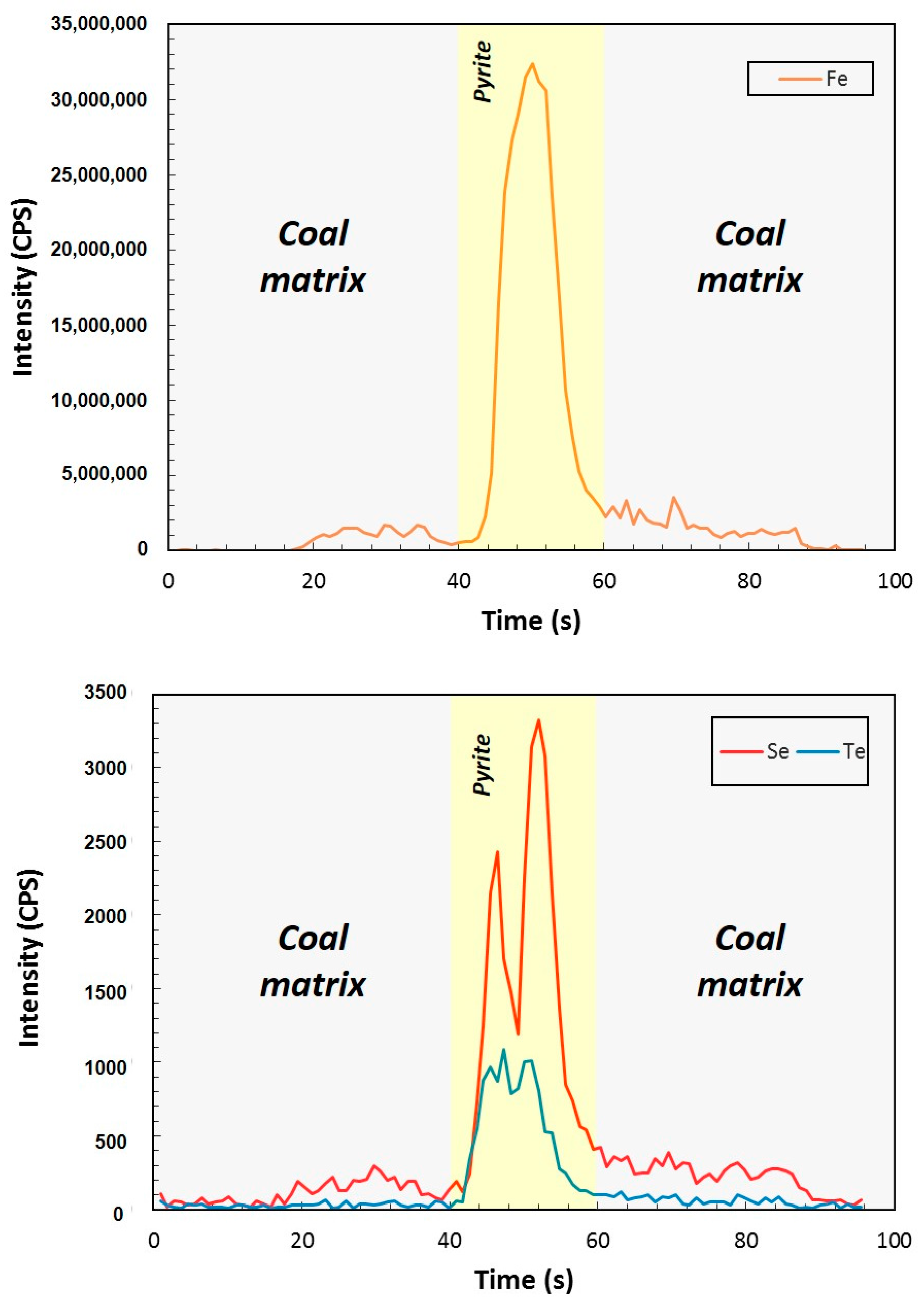
4. Results
5. Discussion
5.1. Tellurium in Brora Coal
5.2. Tellurium Source
5.3. Implications
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rudnick, R.L.; Gao, S. Composition of the continental crust. In The Crust; Rudnick, R.L., Ed.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 3, pp. 1–64. [Google Scholar]
- Hein, J.R.; Koschinsky, A.; Bau, M.; Roberts, L. Cobalt-Rich Ferromanganese Crusts in the Pacific. In Handbook of Marine Mineral Deposits; Cronan, D.S., Ed.; CRC Press: Boca Raton, FL, USA, 2000; pp. 239–279. [Google Scholar]
- Hein, J.R.; Koschinsky, A.; Halliday, A.N. Global occurrence of tellurium-rich ferromanganese crusts and a model for the enrichment of tellurium. Geochim. Cosmochim. Acta 2003, 67, 1117–1127. [Google Scholar] [CrossRef]
- Hein, J.R.; Conrad, T.A.; Staudigel, H. Seamount Mineral Deposits: A Source of Rare Metals for High-Technology Industries. Oceanography 2010, 23, 184–189. [Google Scholar] [CrossRef]
- Spinks, S.C.; Parnell, J.; Bellis, D.; Still, J. Remobilization and mineralization of selenium-tellurium in metamorphosed red beds: Evidence from the Munster Basin, Ireland. Ore Geol. Rev. 2016, 72, 114–127. [Google Scholar] [CrossRef]
- Parnell, J.; Spinks, S.; Bellis, D. Low-temperature concentration of tellurium and gold in continental red bed successions. Terra Nova 2016, 28, 221–227. [Google Scholar] [CrossRef]
- Castillo, J.R.; Lanaja, J.; Aznárez, J. Determination of germanium in coal ashes by hydride generation and flame atomic-absorption spectrophotometry. Analyst 1982, 107, 89–95. [Google Scholar] [CrossRef]
- Nadkarni, R.A. Applications of hydride generation—Atomic absorption spectrometry to coal analysis. Anal. Chim. Acta 1982, 135, 363–368. [Google Scholar] [CrossRef]
- Woo, I.H.; Watanabe, K.; Hashimoto, Y.; Lee, Y.K. Determination of Selenium and Tellurium in Coal by Graphite Furnace Atomic Absorption Spectrometry after Coprecipitation with Arsenic. Anal. Sci. 1987, 3, 49–51. [Google Scholar] [CrossRef]
- Scherer, V.; Linka, S.; Wirtz, S. Spectral radiation characteristics of coal ashes and slags. Int. J. Energy Clean Environ. 2005, 6, 326–341. [Google Scholar] [CrossRef]
- Davidson, D.F.; Lakin, H.W. Tellurium. In United States Mineral Resources; Brobst, D.A., Pratt, W.P., Eds.; US Govt. Print. Off.: Washington, DC, USA, 1973; Volume 820, pp. 627–630. [Google Scholar]
- Lottering, C.; Eksteent, J.J.; Steenekamp, N. Precipitation of rhodium from a copper sulphate leach solution in the selenium/tellurium removal section of a base metal refinery. J. S. Afr. Inst. Min. Metall. 2012, 112, 287–294. [Google Scholar]
- Pohl, W. Economic Geology: Principles and Practice; Wiley-Blackwell Publishing: Chichester, UK, 2011; p. 695. [Google Scholar]
- Schirmer, T.; Koschinsky, A.; Bau, M. The ratio of tellurium and selenium in geological material as a possible paleo-redox proxy. Chem. Geol. 2014, 376, 44–51. [Google Scholar] [CrossRef]
- Templeton, D.M.; Ariese, F.; Cornelis, R.; Danielsson, L.G.; Muntau, H.; van Leeuwen, H.P.; Lobinski, R. Guidelines for terms related to chemical speciation and fractionation of elements. Definitions, structural aspects, and methodological approaches (IUPAC Recommendations 2000). Pure Appl. Chem. 2000, 72, 1453–1470. [Google Scholar] [CrossRef]
- El-Shahawi, M.S.; Al-Saidi, H.M.; Al-Harbi, E.A.; Bashammakh, A.S.; Alsibbai, A.A. Speciation and determination of tellurium in water, soil, sediment and other environmental samples. In Speciation Studies in Soil, Sediment and Environmental Samples; Bakirdere, S., Ed.; CRC Press: Boca Raton, FL, USA, 2013; pp. 527–544. [Google Scholar] [CrossRef]
- Peiró, T.L.; Méndez, G.V.; Ayres, R.U. Rare and Critical Metals as By-Products and the Implications for Future Supply. In INSTEAD Facility and Research Working Paper; INSEAD: Fontainebleau, France, 2011. [Google Scholar]
- Ayres, R.U.; Peiró, T.L. Material efficiency: Rare and critical metals. Phil. Trans. R. Soc. B 2013, 371, 20110563. [Google Scholar] [CrossRef] [PubMed]
- Selenium-Tellurium Development Association. Available online: http://www.stda.org/se_te.html (accessed on 24 July 2017).
- Woodhouse, M.; Goodrich, A.; Margolis, R.; James, T.L.; Lokanc, M.; Eggert, R. Supply-Chain Dynamics of Tellurium, Indium, and Gallium within the Context of PV Manufacturing Costs. IEEE J. Photovolt. 2013, 3, 833–837. [Google Scholar] [CrossRef]
- Lusty, P.A.J.; Gunn, A.G. Challenges to global mineral resource security and options for future supply. In Ore Deposits in an Evolving Earth; Jenkin, G.R.T., Lusty, P.A.J., McDonald, I., Smith, M.P., Boyce, A.J., Wilkinson, J.J., Eds.; Geological Society London Special Publications: London, UK, 2014; Volume 393, pp. 265–276. [Google Scholar] [CrossRef]
- Chelvanathan, P.; Hossain, M.I.; Amin, N. Performance analysis of copper-indium-gallium-diselenide (CIGS) solar cells with various buffer layers by SCAPS. Curr. Appl. Phys. 2010, 10, 387–391. [Google Scholar] [CrossRef]
- Morales-Acevedo, A. Thin film CdS/CdTe solar cells: Research perspectives. Sol. Energy 2006, 80, 675–681. [Google Scholar] [CrossRef]
- Lam, K.; Porter, R. The distribution of palynomorphs in the Jurassic rocks of the Brora Outlier, N.E. Scotland. J. Geol. Soc. 1977, 134, 44–55. [Google Scholar] [CrossRef]
- Stephen, K.J.; Underhill, J.R.; Partington, M.A.; Hedley, R.J. The genetic sequence stratigraphy of the Hettangian to Oxfordian succession, Inner Moray Firth. In Proceedings of the 4th Conference Petroleum Geology of Northwest Europe, London, UK, 29 March–1 April 1992; Parker, J.R., Ed.; Geological Society: London, UK, 1993; pp. 485–505. [Google Scholar]
- MacLennan, A.M.; Trewin, N.H. Palaeoenvironments of the late Bathonian-mid-Callovian in the Innter Moray Firth. In Northwest European Micropalaeontology and Palynology; Keen, M.C., Batten, D.J., Eds.; British Micropalaeontological Society Series; Ellis Horwood: Chichester, UK, 1989; pp. 92–117. [Google Scholar]
- Richards, P.C.; Lott, G.K.; Johnson, H.; Knox, R.W.O’B.; Riding, J.B. 3. Jurassic of the Central and Northern North Sea. In Lithostratigraphic Nomenclature of the UK North Sea; Knox, R.W.O’B., Cordey, W.G., Eds.; British Geological Survey: Nottingham, UK, 1993; pp. 77–78. [Google Scholar]
- Hurst, A. Mid Jurassic stratigraphy and facies at Brora, Sutherland. Scott. J. Geol. 1981, 17, 169–177. [Google Scholar] [CrossRef]
- Linsley, P.N.; Potter, H.C.; McNab, G.; Racher, D. The Beatrice Oil Field, Inner Moray Firth, UK North Sea. In Grant Oil and Gas Fields of the Decade 1968–78; Halbouty, M.T., Ed.; The American Association of Petroleum Geologists: Tulsa, OK, USA, 1980; Volume 30, pp. 117–129. [Google Scholar]
- Curry, M.R.; Fisher, M.J. Coastal plain deposits in the Beatrice oilfield, U.K., North Sea (Abstract). In Proceedings of the 11th International Congress of the International Association of Sedimentologists, Hamilton, ON, Canada, 22–27 August 1982; p. 150. [Google Scholar]
- Andrews, I.J.; Brown, S. Stratigraphic evolution of the Jurassic, Moray Firth. In Petroleum Geology of North West Europe; Brooks, J., Glennie, K.W., Eds.; Graham & Trotman: London, UK, 1987; pp. 785–795. [Google Scholar]
- Crummy, J.; Hall, A.J.; Haszeldine, R.S.; Anderson, I.K. Potential for epithermal gold mineralization in east and central Sutherland, Scotland: Indications from River Brora headwaters. Metall. Mater. Trans. B 1997, 106, B9–B14. [Google Scholar]
- Plant, J.; Coleman, R.F. Application of neutron activation analysis to the evaluation of placer gold concentrations. In Proceedings of the 4th International Geochemical Exploration Symposium, London, UK, 17–20 April 1972; pp. 373–381. [Google Scholar]
- British Geological Survey Geology of Britain Viewer: Online Map Portal. Available online: http://www.bgs.ac.uk/discoveringGeology/geologyOfBritain/viewer.html (accessed on 24 July 2017).
- Turner, B.R.; Richardson, D. Geological controls on the sulphur content of coal seams in the Northumberland Coalfield, Northeast England. Int. J. Coal Geol. 2004, 60, 169–196. [Google Scholar] [CrossRef]
- Bragg, L.J.; Oman, J.K.; Tewalt, S.J.; Oman, C.L.; Rega, N.H.; Washington, P.M.; Finkelman, R.B. U.S. Geological Survey Coal Quality (COALQUAL) Database: Version 2.0; U.S. Geological Survey Open File Report 97-134; The Survey (USGS Distribution Branch): Washington, DC, USA, 1998.
- Yudovich, Y.E.; Ketris, M.P. Selenium in coal: A review. Int. J. Coal Geol. 2006, 67, 112–126. [Google Scholar] [CrossRef]
- Palmer, C.A.; Oman, C.L.; Park, A.J.; Luppens, J.A. The U.S. Geological Survey Coal Quality (COALQUAL) Database Version 3.0; U.S. Geological Survey Data Series; U.S. Geological Survey: Reston, VA, USA, 2015; Volume 975, p. 43. [CrossRef]
- Spears, D.A. The geochemistry and mineralogy of high-S coals, with examples mainly from the Yorkshire-Nottinghamshire coalfields, UK: An overview. Proc. Yorks. Geol. Soc. 2015, 60, 204–226. [Google Scholar] [CrossRef]
- Ketris, M.P.; Yudovich, Y.E. Estimations of Clarkes for Carbonaceous biolithes: World averages for trace element contents in black shales and coals. Int. J. Coal Geol. 2009, 78, 135–148. [Google Scholar] [CrossRef]
- Machel, H.G. Bacterial and thermochemical sulphate reduction in diagenetic settings—Old and new insights. Sediment. Geol. 2001, 140, 143–175. [Google Scholar] [CrossRef]
- Parnell, J.; Bellis, D.; Feldmann, J.; Bata, T. Selenium and tellurium enrichment in palaeo-oil reservoirs. J. Geochem. Explor. 2015, 148, 169–173. [Google Scholar] [CrossRef]
- Orr, W.L. Changes in sulfur content and isotopic ratios of sulfur during petroleum maturation—Study of Big Horn Basin Palaeozoic oils. Am. Assoc. Pet. Geol. 1974, 58, 2295–2318. [Google Scholar]
- Afifi, A.M.; Kelly, W.C.; Essene, E.J. Phase relations among tellurides, sulfides, and oxides: II. Applications to telluride-bearing ore deposits. Econ. Geol. 1988, 83, 395–404. [Google Scholar] [CrossRef]
- Jensen, E.P.; Barton, M.D. Gold deposits related to alkaline magmatism. Rev. Econ. Geol. 2000, 13, 279–314. [Google Scholar]
- Cook, N.J.; Ciobanu, C.L.; Spry, P.G.; Voudouris, P. The participants of IGCP-486. Understanding gold-(silver)-telluride-(selenide) deposits. Episodes 2009, 32, 249–263. [Google Scholar]
- Boyle, R.W. The geochemistry of gold and its deposits. Geol. Surv. Can. Bull. 1979, 280, 584. [Google Scholar]
- Ciobanu, C.L.; Cook, N.J.; Spry, P.G. Telluride and selenide minerals in gold deposits—How and why? Mineral. Petrol. 2006, 87, 163–169. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Watanbe, K.; Yumoto, K.; Otoshi, T. Tellurium as a Possible Tracer of Coal Combustion Effluent. J. Jpn. Soc. Air Pollut. 1989, 24, 45–51. [Google Scholar]
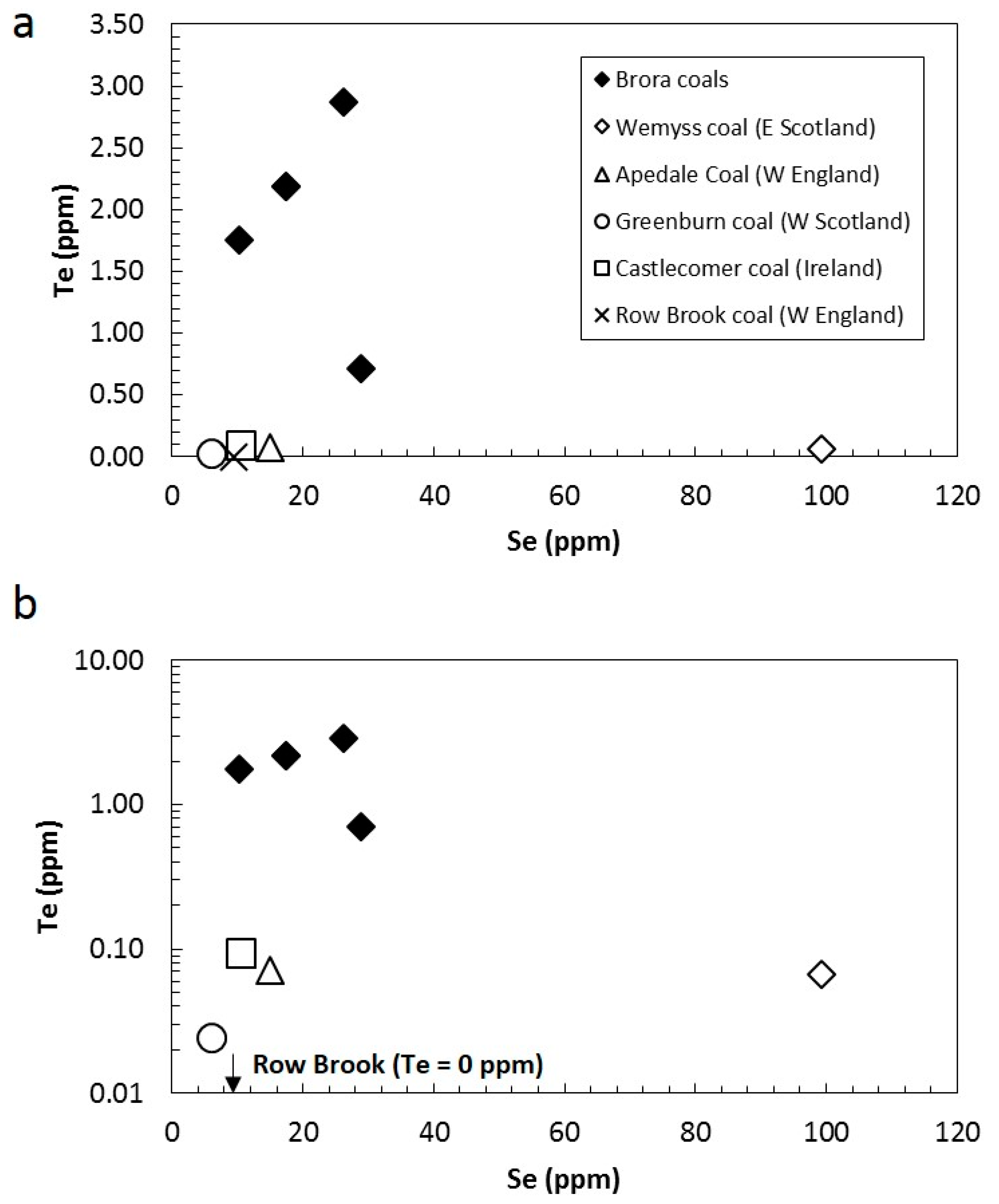
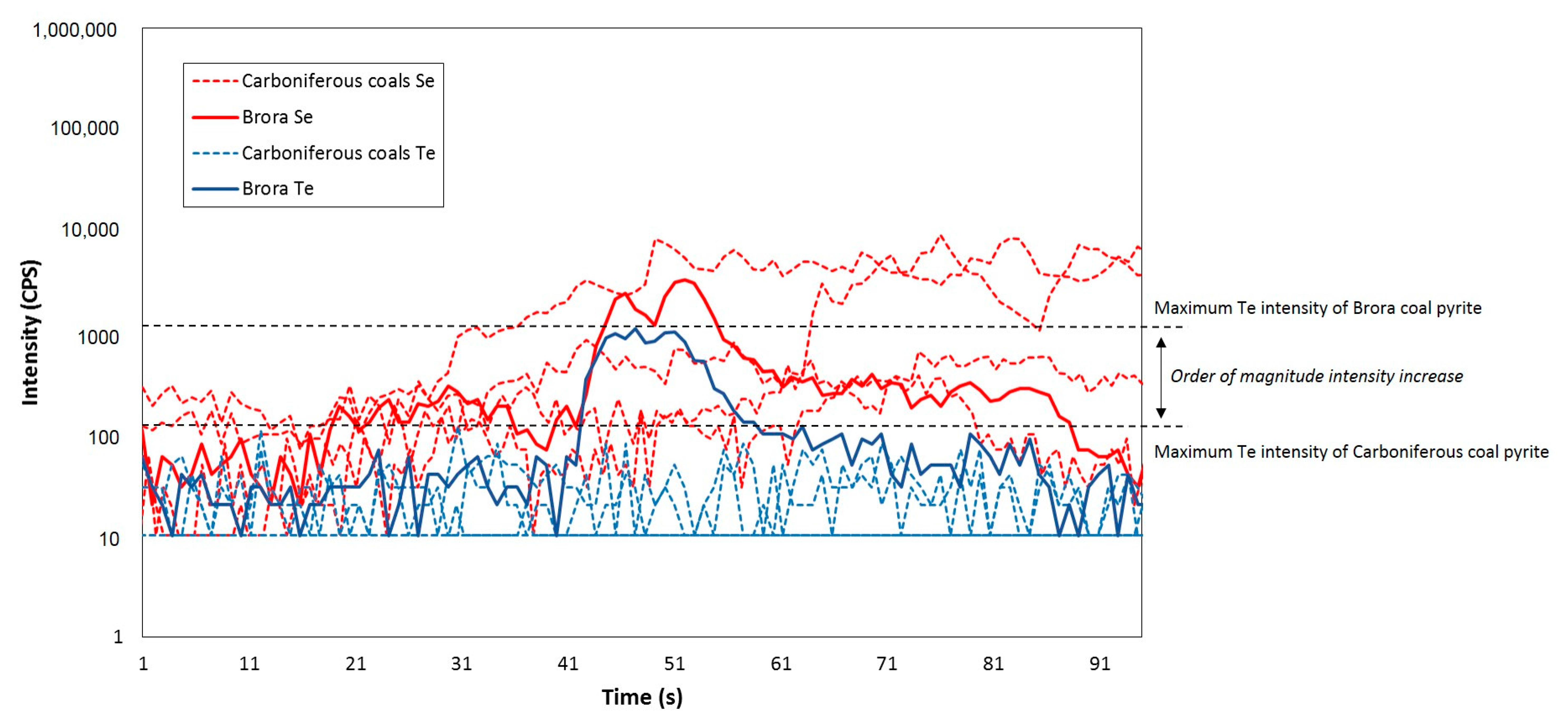
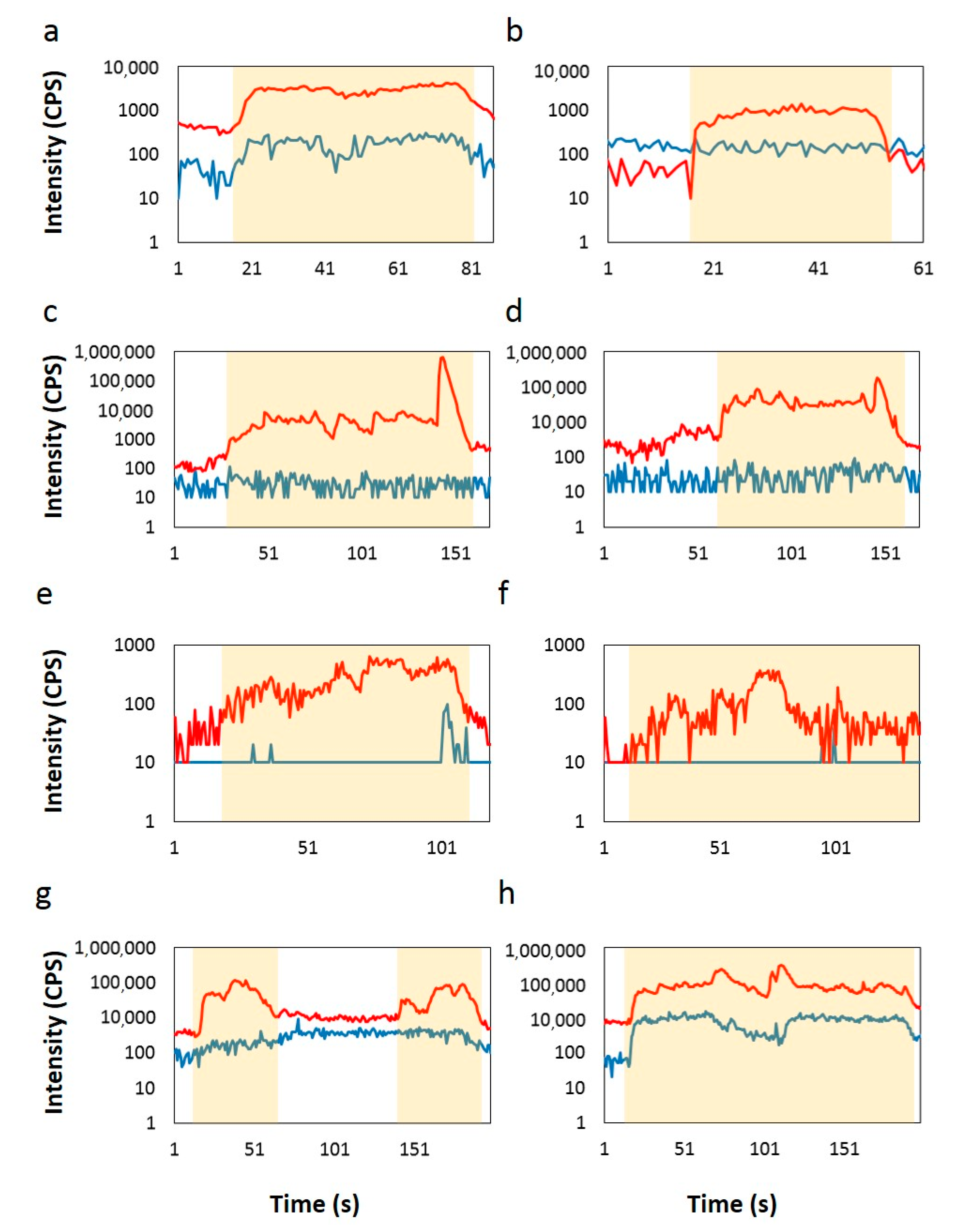
| Sample | Age | Se (ppm) | Te (ppm) | Se/Te |
|---|---|---|---|---|
| Brora 1 | Jurassic | 10.3 | 1.8 | 5.8 |
| Brora 2 | Jurassic | 26.2 | 2.9 | 9.1 |
| Brora 3 | Jurassic | 17.4 | 2.2 | 7.9 |
| Castlecomer | Carboniferous | 10.5 | 0.1 | 110.9 |
| Row Brook | Carboniferous | 9.4 | 0.0 | - |
| Wemyss | Carboniferous | 99.2 | 0.1 | 1487.4 |
| Apedale | Carboniferous | 15.1 | 0.1 | 213.3 |
| Greenburn | Carboniferous | 5.9 | 0.0 | 240.3 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bullock, L.; Parnell, J.; Perez, M.; Feldmann, J. Tellurium Enrichment in Jurassic Coal, Brora, Scotland. Minerals 2017, 7, 231. https://doi.org/10.3390/min7120231
Bullock L, Parnell J, Perez M, Feldmann J. Tellurium Enrichment in Jurassic Coal, Brora, Scotland. Minerals. 2017; 7(12):231. https://doi.org/10.3390/min7120231
Chicago/Turabian StyleBullock, Liam, John Parnell, Magali Perez, and Joerg Feldmann. 2017. "Tellurium Enrichment in Jurassic Coal, Brora, Scotland" Minerals 7, no. 12: 231. https://doi.org/10.3390/min7120231
APA StyleBullock, L., Parnell, J., Perez, M., & Feldmann, J. (2017). Tellurium Enrichment in Jurassic Coal, Brora, Scotland. Minerals, 7(12), 231. https://doi.org/10.3390/min7120231






