Further Investigations on Simultaneous Ultrasonic Coal Flotation
Abstract
:1. Introduction
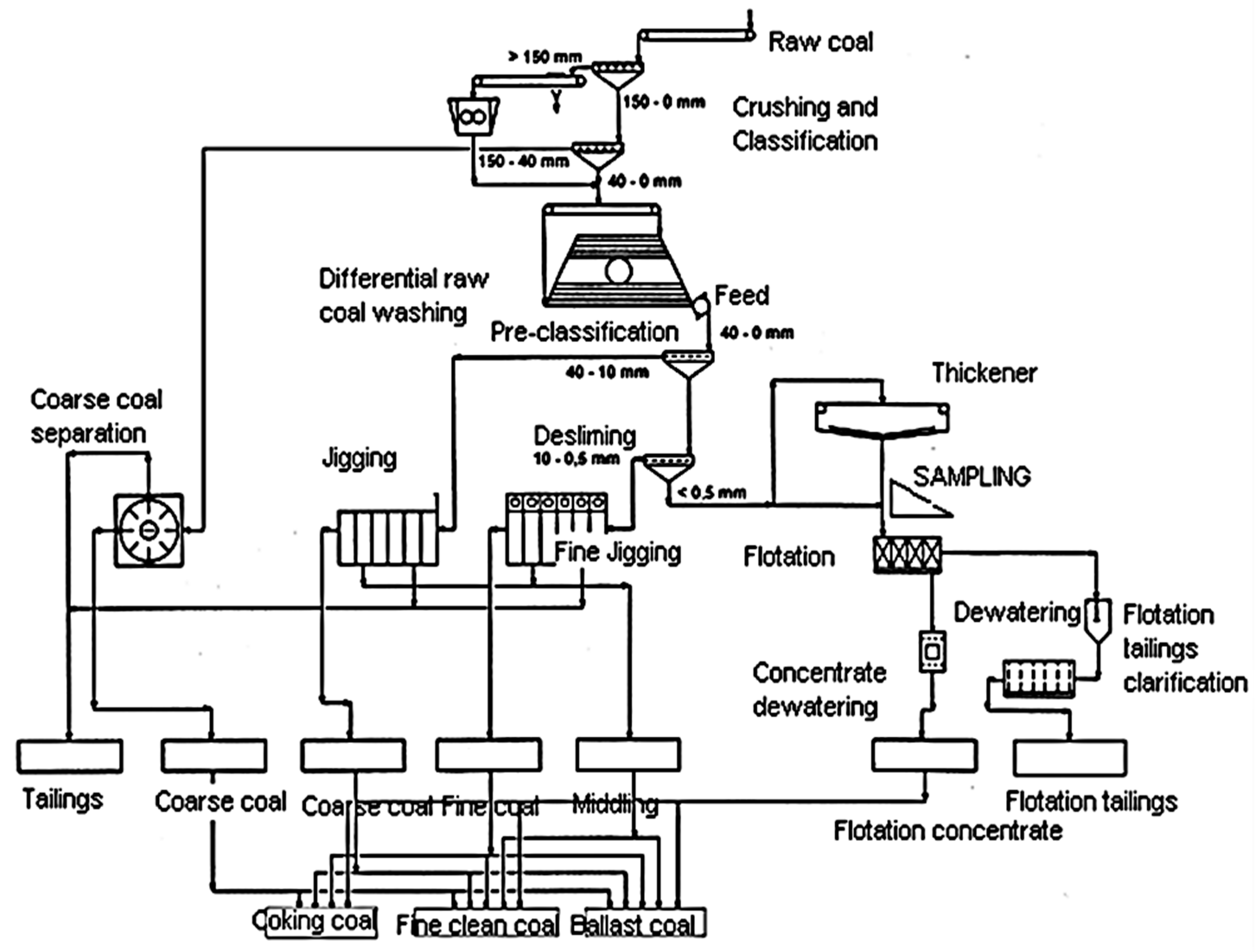
2. Materials and Methods
3. Experimental Results
- Combustible Recovery = [C × (100 − c)/F × (100 − f)] × 100
- Separation Efficiency = C × 100 × {[(100 − c) − (100 − f)]/[(100 − f) × f]}
- F: Weight of coal feed (%)
- C: Weight of clean coal (%)
- f: Ash of coal feed (%)
- c: Ash of clean coal (%)
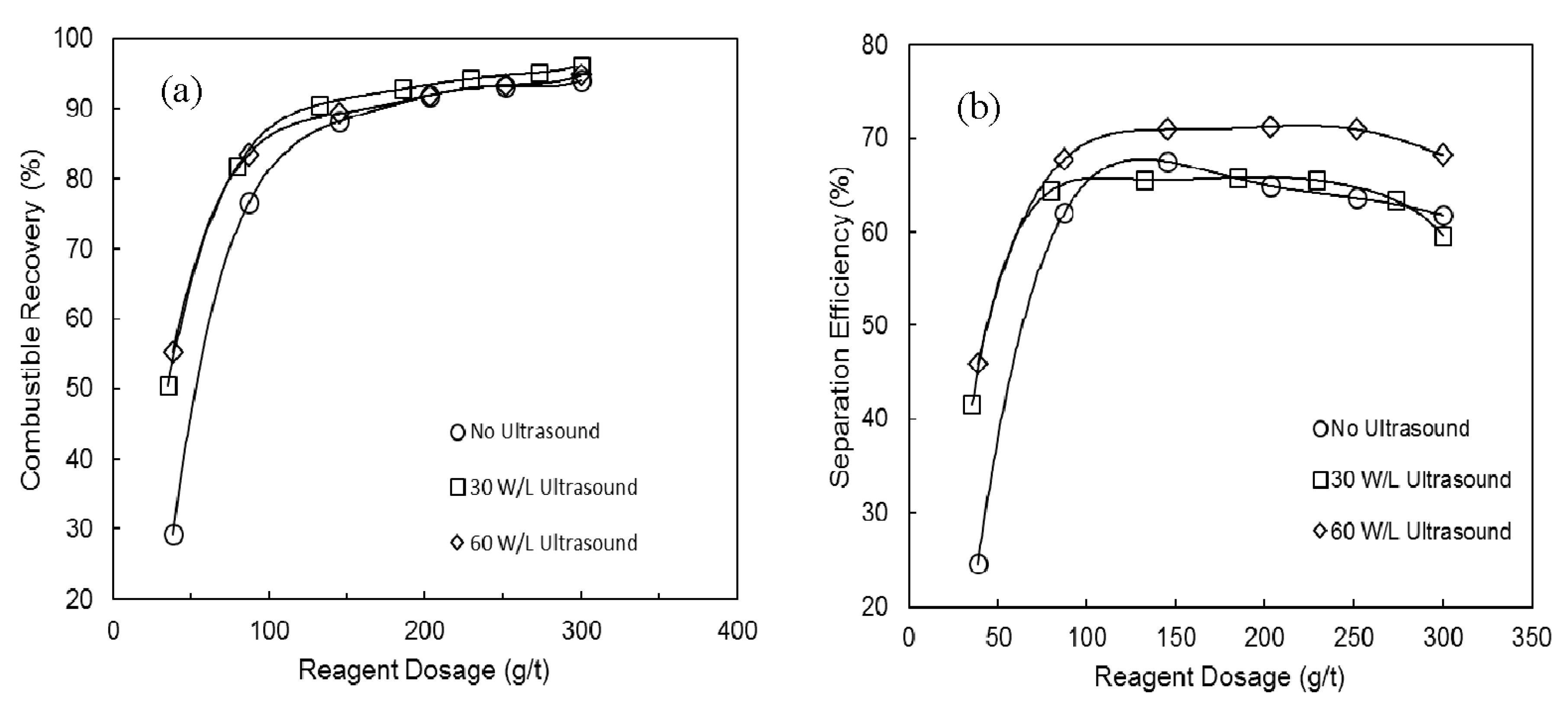
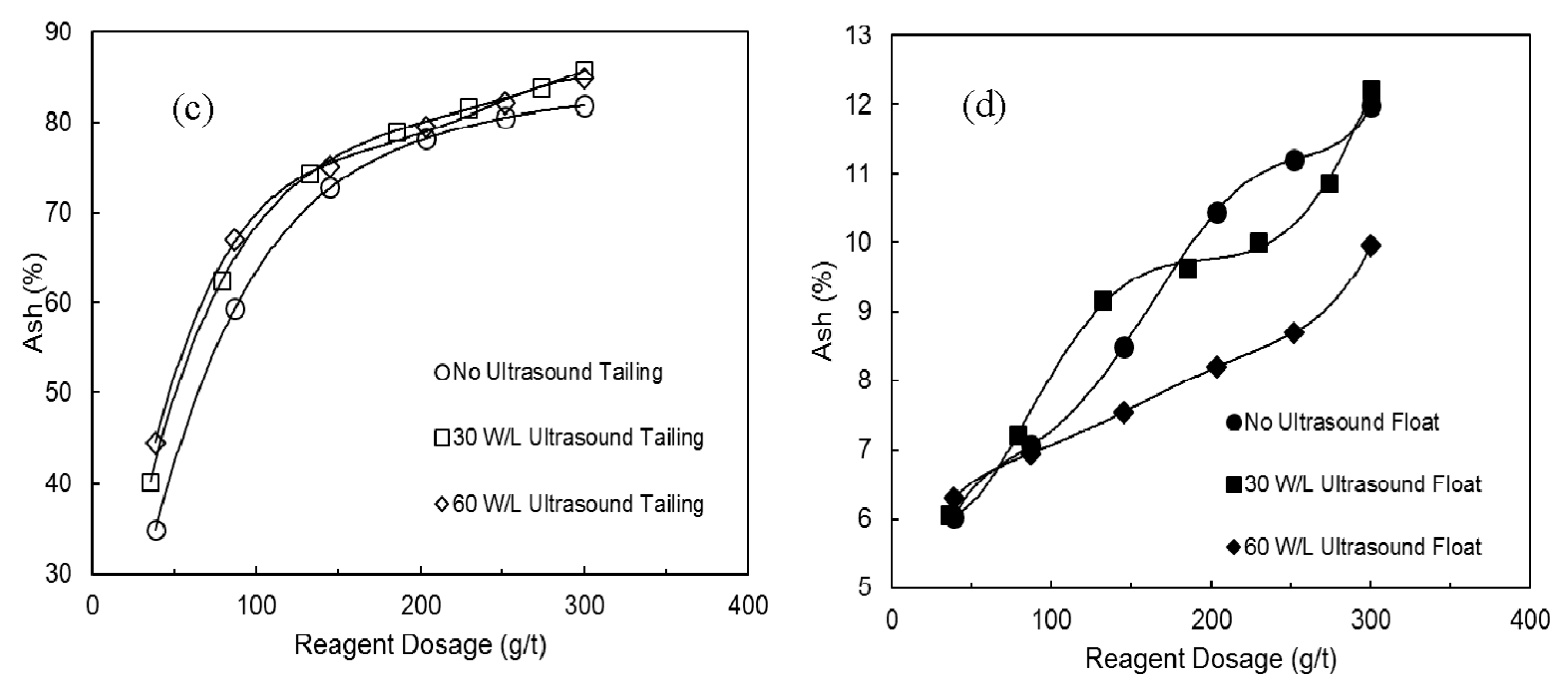
3.1. The Effects of Ultrasound on Combustible Recovery and Separation Efficiency
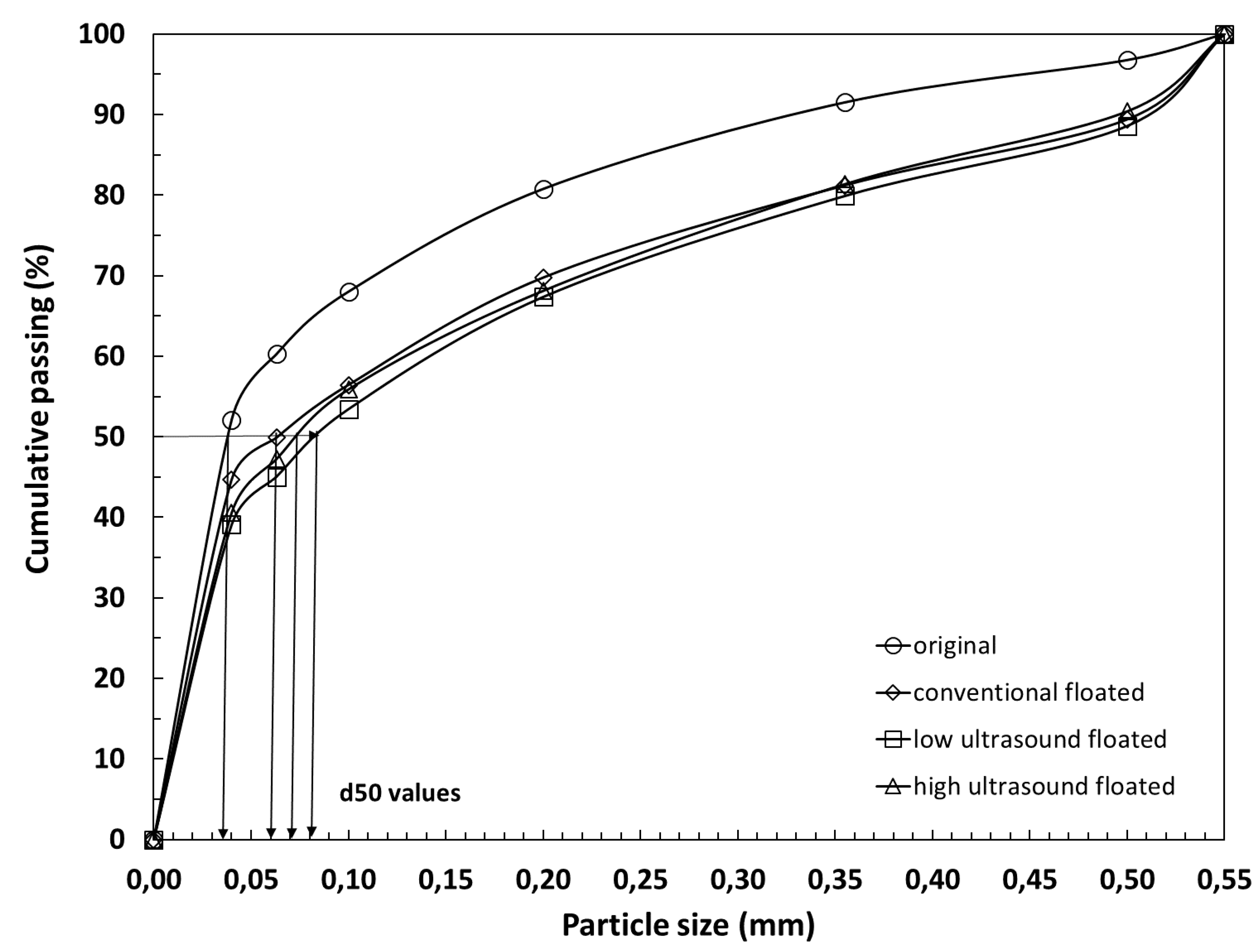
3.2. Effects of Ultrasound on Particle Size Distribution
4. Discussion and Conclusions
Acknowledgments
Conflicts of Interest
References
- Stoev, S.M.; Kuzev, L.; Metodiev, M.; Djendova, S. Vibroacoustic Improvements of Froth Flotation; Mavros, P., Matis, K.A., Eds.; Innovations in Flotation Technology. NATO ASI Series; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1992; Volume 208, pp. 383–407. [Google Scholar]
- Altun, N.E.; Hwang, J.Y.; Hicyilmaz, C. Enhancement of flotation performance of oil shale cleaning by ultrasonic treatment. Int. J. Miner. Process. 2009, 91, 1–13. [Google Scholar] [CrossRef]
- Ambedkar, B.; Nagarajan, R.; Jayanti, S. Investigation of high-frequency, high-intensity ultrasonics for size reduction and washing of coal in aqueous medium. Ind. Eng. Chem. Res. 2011, 50, 13210–13219. [Google Scholar] [CrossRef]
- Barry, B.; Klima, M.S.; Cannon, F.S. Effects of desliming and hydroacoustic cavitation treatment on the flotation of ultrafine bituminous coal. Int. J. Coal Prep. Util. 2017, 37, 213–224. [Google Scholar] [CrossRef]
- Cao, Q.; Cheng, J.; Feng, Q.; Wen, S.; Luo, B. Surface cleaning and oxidative effects of ultrasonication on the flotation of oxidized pyrite. Powder Technol. 2017, 311, 390–397. [Google Scholar] [CrossRef]
- Cilek, E.C.; Ozgen, S. Improvement of the flotation selectivity in a mechanical flotation cell by ultrasound. Sep. Sci. Technol. 2010, 45, 572–579. [Google Scholar] [CrossRef]
- Feng, D.; Aldrich, C. Effect of preconditioning on the flotation of coal. Chem. Eng. Commun. 2005, 192, 972–983. [Google Scholar] [CrossRef]
- Ghadyani, A.; Noaparast, M.; Tonkaboni, S.Z.S. A study on the effects of ultrasonic irradiation as pretreatment method on high-ash coal flotation and kinetics. Int. J. Coal Prep. Util. 2017, 1–18. [Google Scholar] [CrossRef]
- Gungoren, C.; Ozdemir, O.; Ozkan, S.G. Effects of temperature during ultrasonic conditioning in quartz-amine flotation. Physicochem. Probl. Miner. Process. 2017, 53, 687–698. [Google Scholar]
- Haghi, H.; Noaparast, M.; Tonkaboni, S.Z.S.; Mirmohammadi, M. A new experimental approach to improve the quality of low grade silica; the combination of indirect ultrasound irradiation with reverse flotation and magnetic separation. Minerals 2016, 6, 121. [Google Scholar] [CrossRef]
- Ozkan, S.G. Ultrasonic coal flotation. In Proceedings of the Abstracts of Limits of Knowledge International Scientific Conference Humboldt-Kolleg, Krakow, Poland, 22–25 June 2017; pp. 125–126. [Google Scholar]
- Ozkan, S.G.; Lotzien, R. Investigation of a newly designed ultrasonically assisted flotation cell. In Proceedings of the Abstracts of AIMS 2016—The 1st International Conference on Mining in Europe, Aachen, Germany, 18–19 May 2016; p. 61. [Google Scholar]
- Ozkan, S.G. Effects of simultaneous ultrasonic treatment on flotation of hard coal slimes. Fuel 2012, 93, 576–580. [Google Scholar] [CrossRef]
- Ozkan, S.G.; Gungoren, C. Enhancement of colemanite flotation by ultrasonic pre-treatment. Physicochem. Probl. Miner. Process. 2012, 48, 455–462. [Google Scholar]
- Ozkan, S.G.; Kuyumcu, H.Z. Design of a flotation cell equipped with ultrasound transducers to enhance coal flotation. Ultrason. Sonochem. 2007, 14, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Ozkan, S.G.; Kuyumcu, H.Z. Investigation of mechanism of ultrasound on coal flotation. Int. J. Miner. Process. 2006, 81, 201–203. [Google Scholar] [CrossRef]
- Ozkan, S.G.; Kuyumcu, H.Z. Zum einfluss von ultraschall auf die steinkohleflotation (The influence of ultrasonics on coal flotation). Aufbereit. Tech. 2006, 47, 23–33. (In German) [Google Scholar]
- Ozkan, S.G. Beneficiation of magnesite slimes with ultrasonic treatment. Miner. Eng. 2002, 15, 99–101. [Google Scholar] [CrossRef]
- Letmahe, C.; Benker, B.; Gunther, L. Intensivierung der schaumflotation durch einsatz von ultraschall. Aufbereit. Tech. 2002, 43, 32–40. (In German) [Google Scholar]
- Kang, W.Z.; Xun, H.X.; Kong, Z.; Li, M. Effects from changes in pulp nature after ultrasonic conditioning on high-sulfur coal flotation. Min. Sci. Technol. 2009, 19, 498–502. [Google Scholar] [CrossRef]
- Kang, W.Z.; Xun, H.X.; Hu, J. Study of the effect of ultrasonic treatment on the surface composition and the flotation performance of high-sulfur coal. Fuel Process. Technol. 2008, 89, 1337–1344. [Google Scholar] [CrossRef]
- Vargas-Hernández, Y.; Gaete-Garreton, L.; Magne Ortega, L.; Vergara-Belmar, R. High-power ultrasound as an alternative to high-intensity conditioning in flotation. In Proceedings of the World Congress on Ultrasonics, Paris, France, 7–10 September 2003; pp. 435–438. [Google Scholar]
- Riera-Franco de Sarabia, E.; Gallego-Juarex, J.A.; Rodrigez-Corral, G.; Elvira-Segura, L.; Gonzalez-Gomez, I. Application of high-power ultrasound to enhance fluid solid particle separation processes. Ultrasonics 2000, 38, 642–646. [Google Scholar] [CrossRef]
- Videla, A.R.; Morales, R.; Saint-Jean, T.; Gaete, L.; Vargas, Y.; Miller, J.D. Ultrasound treatment on tailings to enhance copper flotation recovery. Miner. Eng. 2016, 99, 89–95. [Google Scholar] [CrossRef]
- Zhang, H.X.; Bai, H.J.; Dong, X.S.; Wang, Z.Z. Enhanced desulfurizing flotation of different size fractions of high sulfur coal using sonoelectrochemical method. Fuel Process. Technol. 2012, 97, 9–14. [Google Scholar] [CrossRef]
- Zhang, H.X.; Ma, X.Y.; Dong, X.S.; Wang, Z.Z.; Bai, H.J. Enhanced desulfurizing flotation of high sulfur coal by sonoelectrochemical method. Fuel Process. Technol. 2012, 93, 13–17. [Google Scholar] [CrossRef]
- DIN-22017, Investigations of the raw material in hard-coal-mining - Froth-flotation-analysis.
- Harrison, C.D.; Raleigh, C.E., Jr.; Vujnovic, B.J. The use of ultrasound for cleaning coal. In Proceedings of the 19th Annual International Coal Preparation Exhibition and Conference, Lexington, KY, USA, 30 April–2 May 2002; pp. 61–67. [Google Scholar]
- Laskowski, J.S. Coal Flotation and Fine Coal Utilization, 1st ed.; Elsevier Science: Amsterdam, The Netherlands, 2001; Volume 14, p. 384. [Google Scholar]


| Particle Size Fractions (mm) | Weight (%) | Heat Value (kcal/kg) | Sulfur (%) | Ash (%) |
|---|---|---|---|---|
| +0.500 | 3.25 | 8153.24 | 0.84 | 3.93 |
| −0.500 + 0.355 | 5.25 | 7950.22 | 0.90 | 5.53 |
| −0.355 + 0.200 | 10.75 | 7239.66 | 0.86 | 7.83 |
| −0.200 + 0.100 | 12.75 | 6510.70 | 0.87 | 14.65 |
| −0.100 + 0.063 | 7.75 | 5662.56 | 0.93 | 15.96 |
| −0.063 + 0.040 | 8.13 | 5652.53 | 1.04 | 16.61 |
| −0.040 | 52.13 | 3038.86 | 1.19 | 46.21 |
| Total (Feed) | 100.00 | 4935.79 | 1.04 | 29.80 |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ozkan, S.G. Further Investigations on Simultaneous Ultrasonic Coal Flotation. Minerals 2017, 7, 177. https://doi.org/10.3390/min7100177
Ozkan SG. Further Investigations on Simultaneous Ultrasonic Coal Flotation. Minerals. 2017; 7(10):177. https://doi.org/10.3390/min7100177
Chicago/Turabian StyleOzkan, Safak Gokhan. 2017. "Further Investigations on Simultaneous Ultrasonic Coal Flotation" Minerals 7, no. 10: 177. https://doi.org/10.3390/min7100177
APA StyleOzkan, S. G. (2017). Further Investigations on Simultaneous Ultrasonic Coal Flotation. Minerals, 7(10), 177. https://doi.org/10.3390/min7100177




