Role of Fungi in the Biomineralization of Calcite
Abstract
:1. Introduction
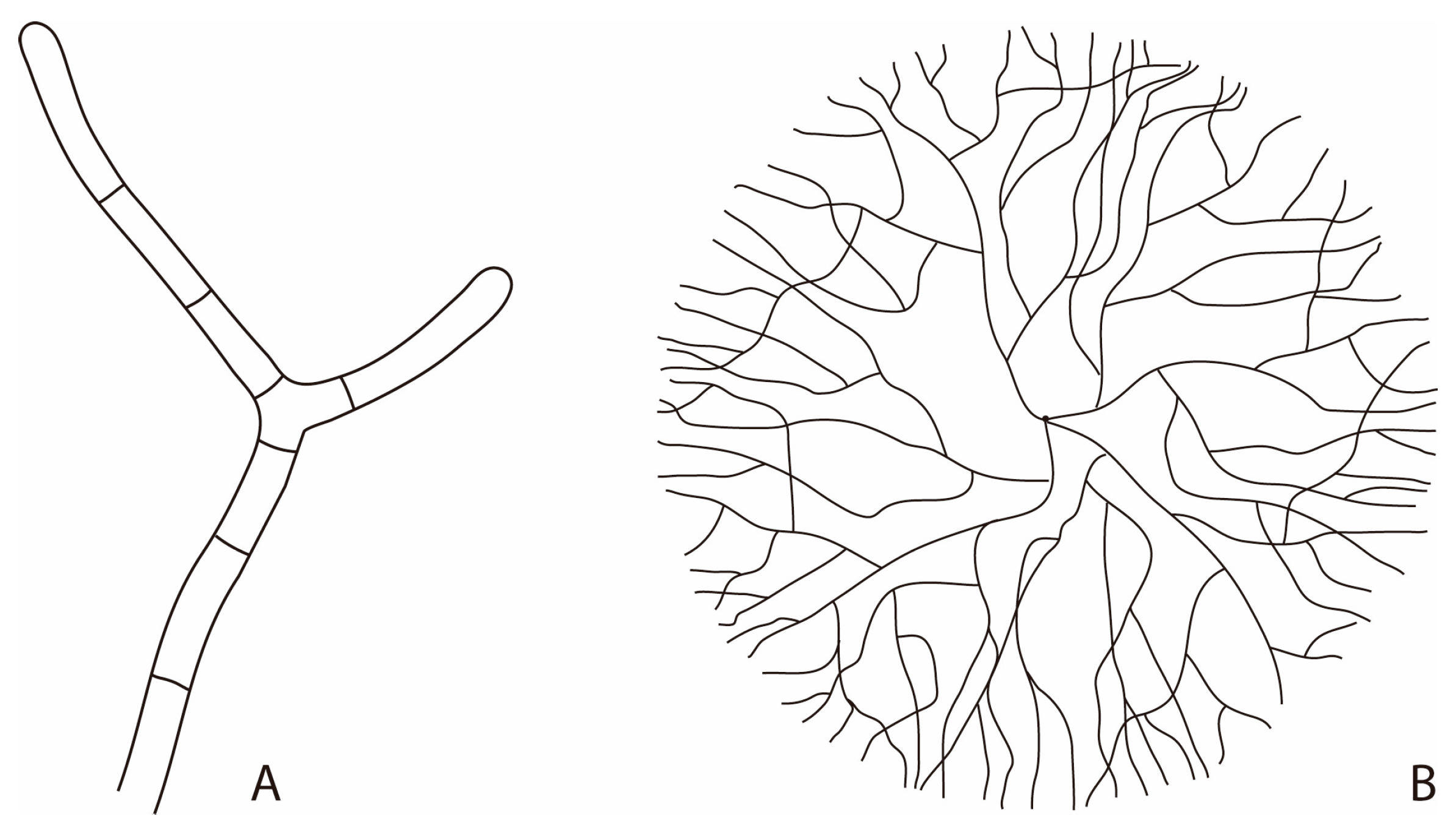
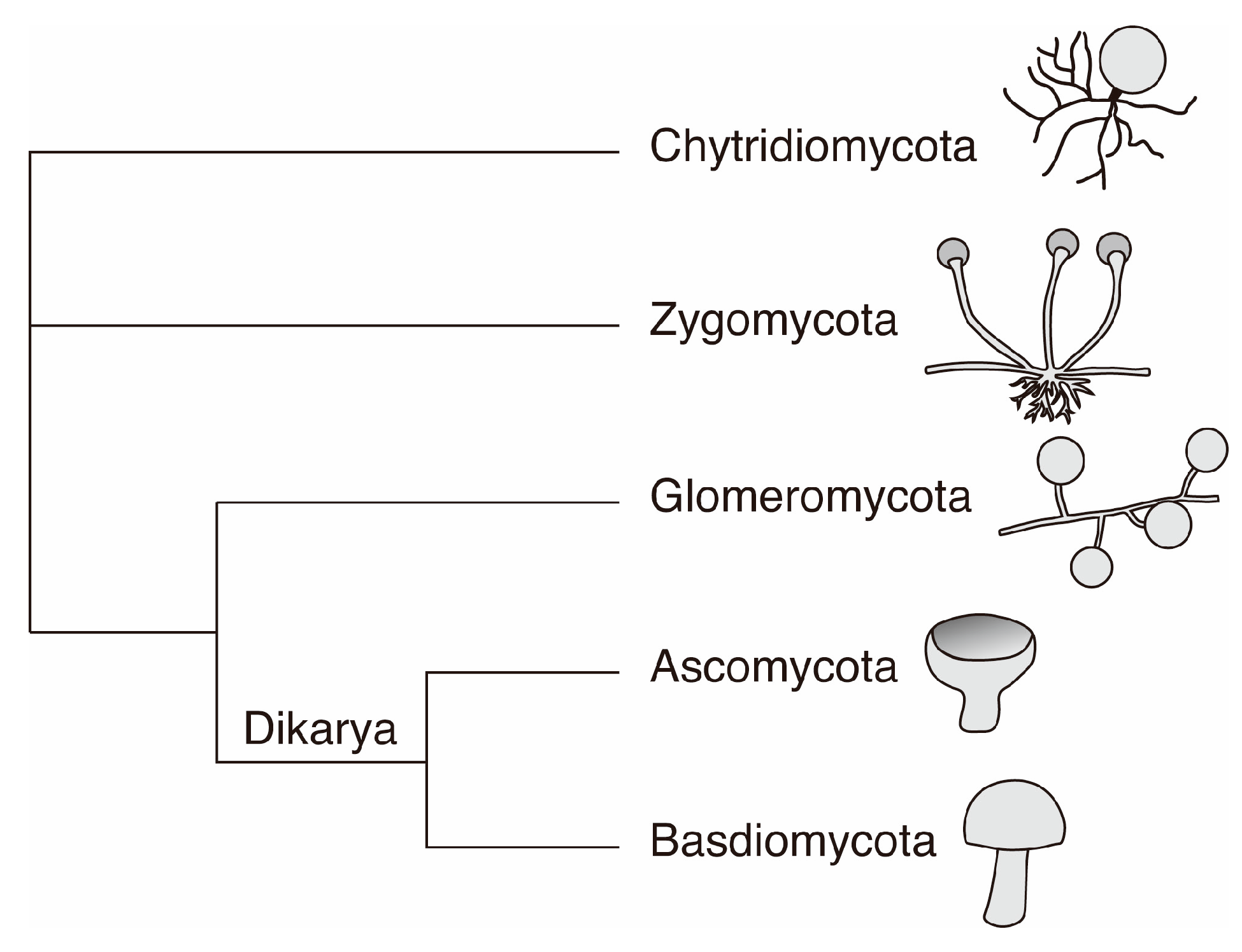
2. Fungi in the Geobiosphere
3. Importance of Fungi in the Biogeochemical Cycles of Elements
4. Fungal Biominerals
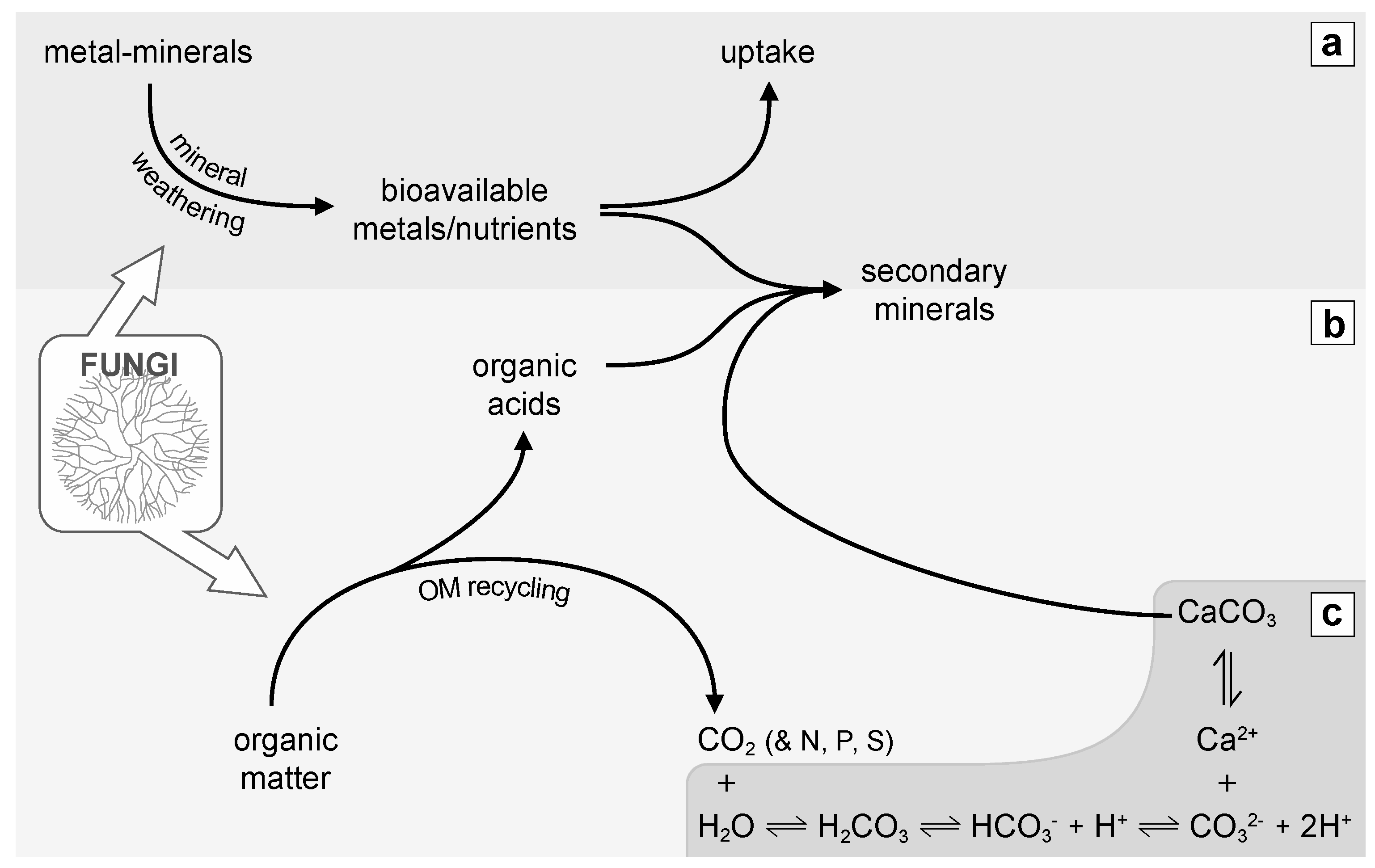
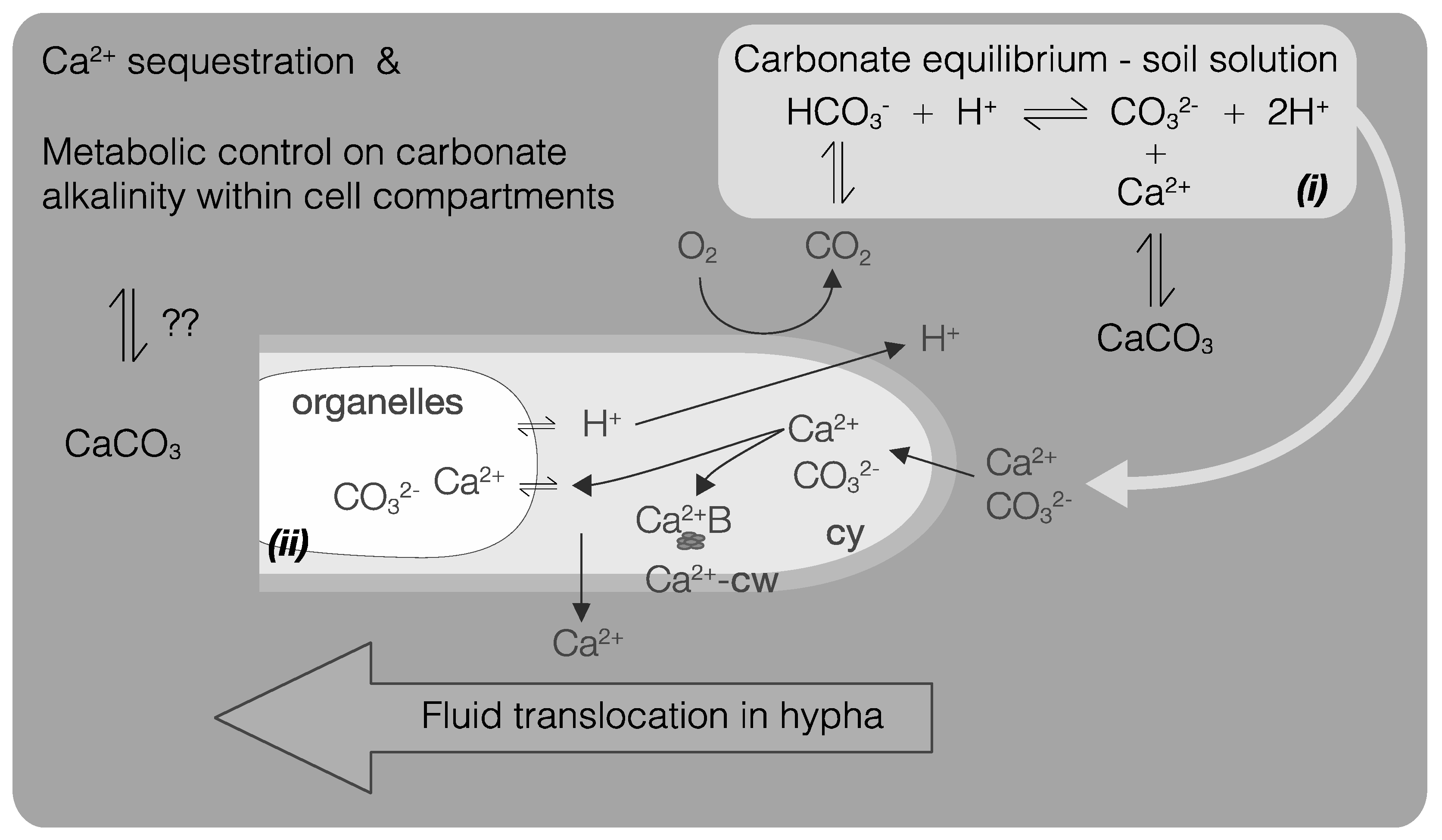
5. Fungi and CaCO3
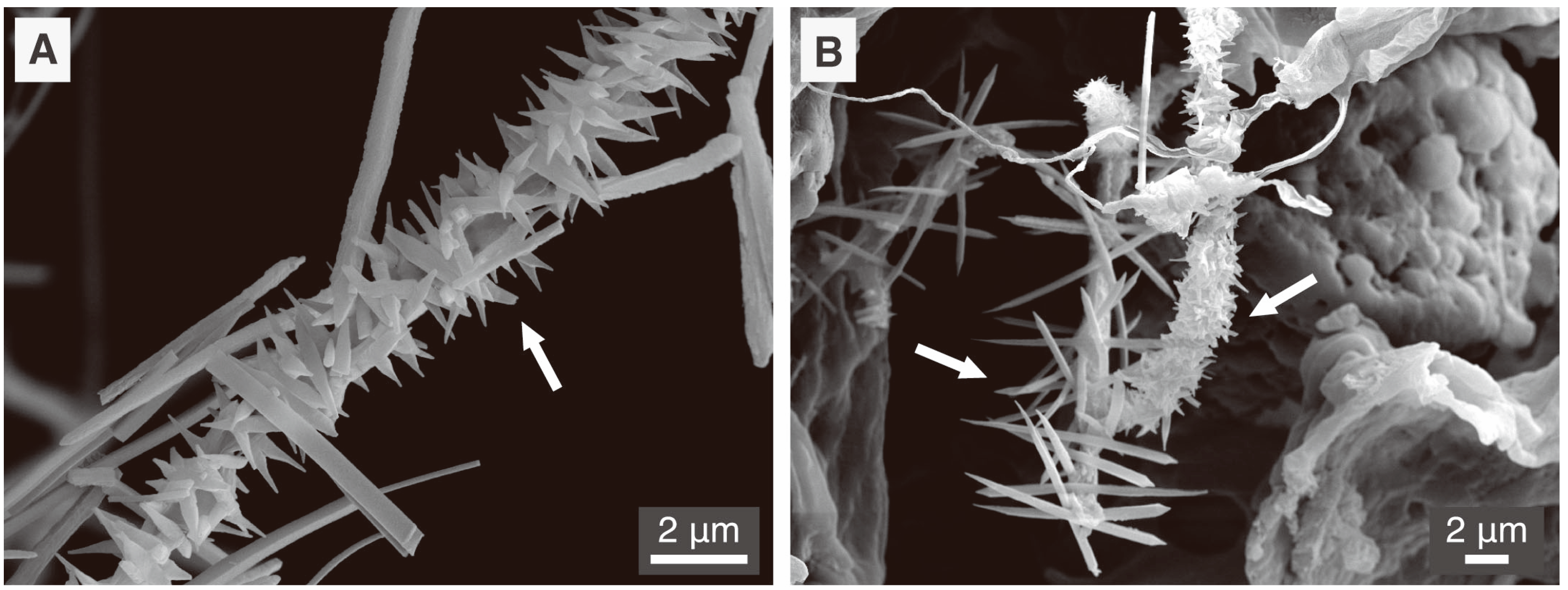
5.1. Fungal CaCO3 Produced in the Laboratory
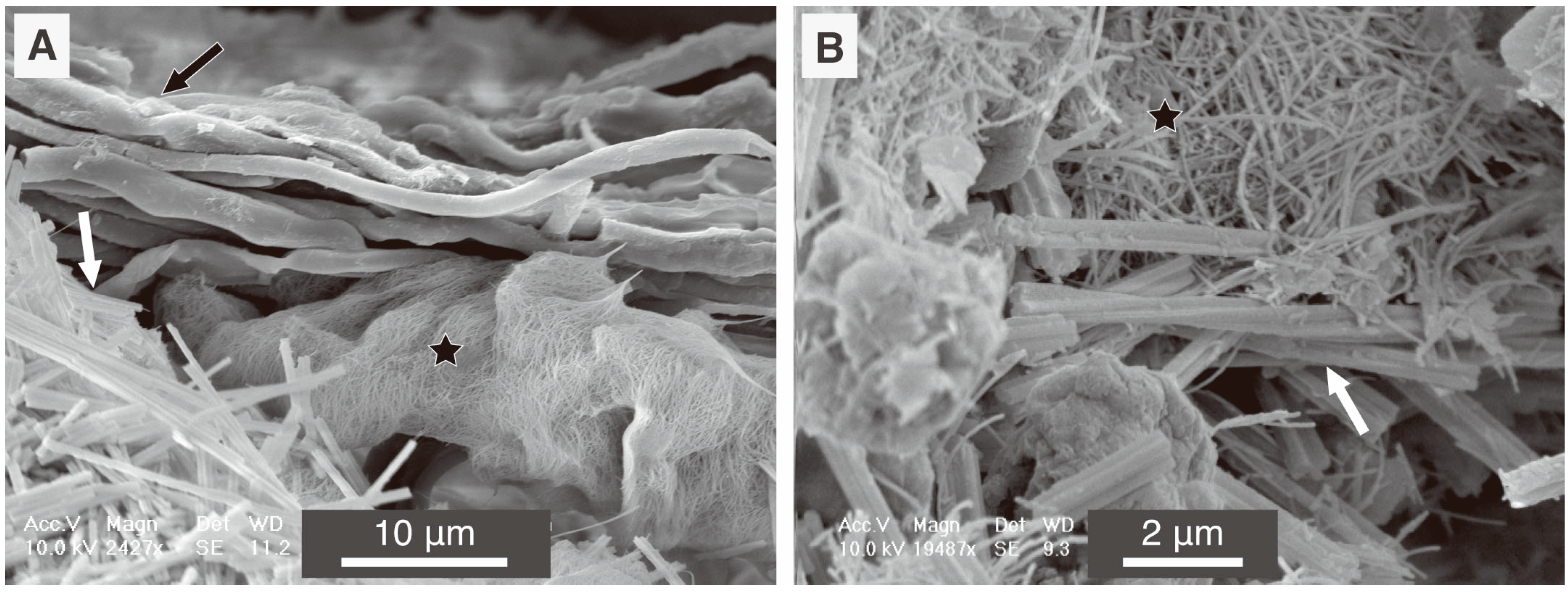
5.2. Fungal CaCO3 in the Natural Environment
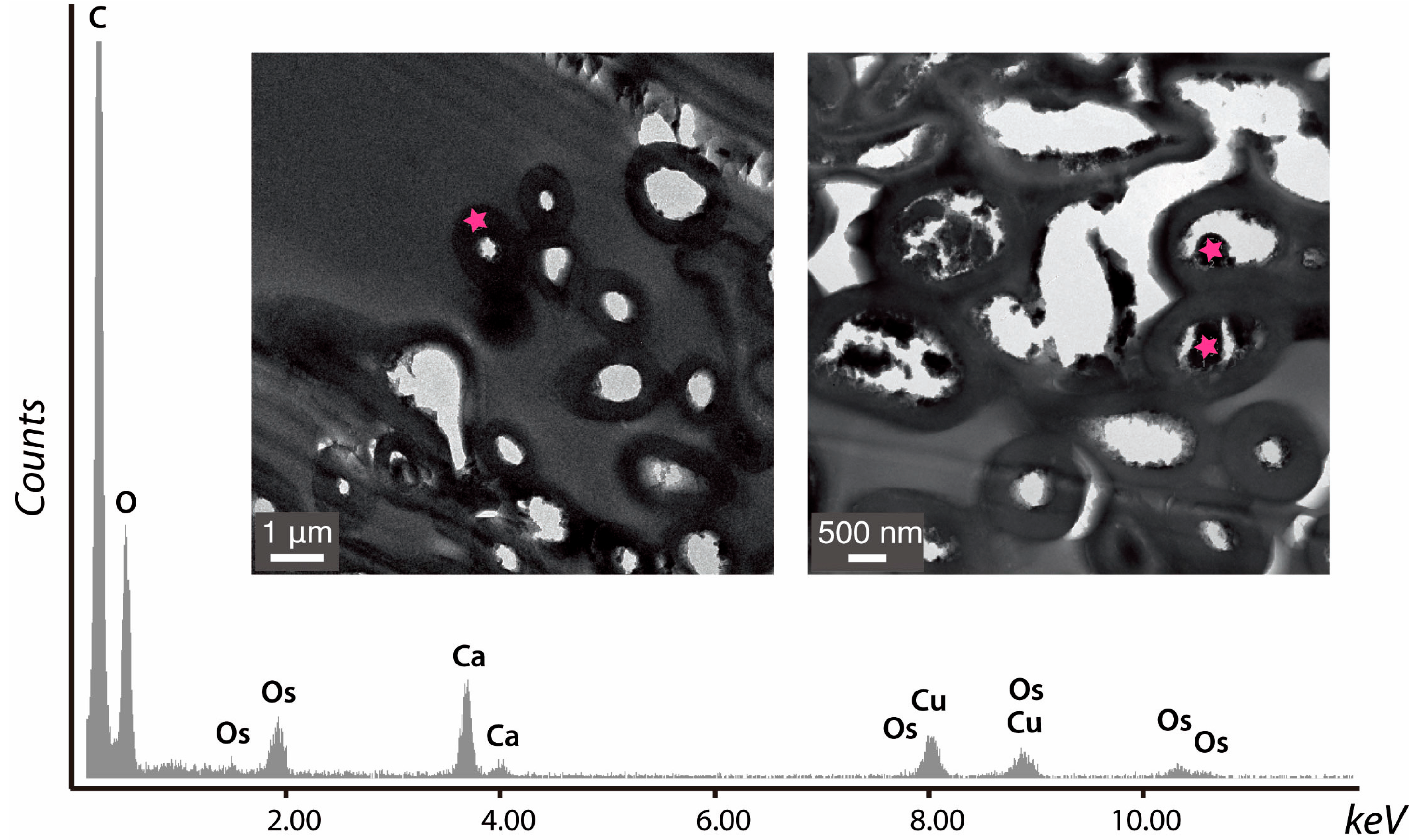
6. Methodological Issues in the Study of Fungal Biominerals from Natural Environments
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Weiner, S.; Dove, P.M. An overview of biomineralization processes and the problem of the vital effect. In Biomineralization; Dove, P.M., de Yoreo, J.J., Weiner, S., Eds.; Mineralogical Society of America: Chantilly, VA, USA, 2003; Volume 54, pp. 1–29. [Google Scholar]
- Dupraz, C.; Reid, R.P.; Braissant, O.; Decho, A.W.; Norman, R.S.; Visscher, P.T. Processes of carbonate precipitation in modern microbial mats. Earth Sci. Rev. 2009, 96, 141–162. [Google Scholar]
- Défarge, C. Organomineralization. In Encyclopedia of Geobiology; Reitner, J., Thiel, V., Eds.; Springer: Dodrecht, The Netherlands, 2011; pp. 697–701. [Google Scholar]
- Trichet, J.; Défarge, C. Non-biologically supported organomineralization. Bull. Inst. Océanogr. Monaco Numéro Spec. 1995, 14, 203–236. [Google Scholar]
- Madigan, M.T.; Martinko, J.M.; Bender, K.S.; Buckley, D.H.; Stahl, D.H. Brock Biology of Microorganisms, 14th ed.; Pearson Education, Inc.: Boston, MA, USA, 2015. [Google Scholar]
- Mann, S. Biomineralization: Principles and Concepts in Bioinorganic Materials Chemistry; Oxford University Press, Inc.: New York, NY, USA, 2001. [Google Scholar]
- Bazylinski, D.A.; Frankel, R.B. Biologically controlled mineralization in prokaryotes. Rev. Mineral. Geochem. 2003, 54, 217–247. [Google Scholar]
- Badger, M.R.; Price, G.D. The role of carbonic-anhydrase in photosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1994, 45, 369–392. [Google Scholar] [CrossRef]
- Castanier, S.; le Metayer-Levrel, G.; Perthuisot, J.-P. Bacterial roles in the precipitation of carbonate minerals. In Microbial Sediments; Riding, R.E., Awramik, S.M., Eds.; Springer-Verlag: Berlin, Germany; Heidelberg, Germany, 2000; pp. 32–39. [Google Scholar]
- Braissant, O.; Cailleau, G.; Aragno, M.; Verrecchia, E.P. Biologically induced mineralization in the tree Milicia excelsa (Moraceae): Its causes and consequences to the environment. Geobiology 2004, 2, 59–66. [Google Scholar]
- Visscher, P.T.; Stolz, J.F. Microbial mats as bioreactors: Populations, processes, and products. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2005, 219, 87–100. [Google Scholar]
- Dupraz, C.; Visscher, P.T. Microbial lithification in marine stromatolites and hypersaline mats. Trends Microbiol. 2005, 13, 429–438. [Google Scholar]
- Benzerara, K.; Morin, G.; Yoon, T.H.; Miot, J.; Tyliszczak, T.; Casiot, C.; Bruneel, O.; Farges, F.; Brown, G.E. Nanoscale study of As biomineralization in an acid mine drainage system. Geochim. Cosmochim. Acta 2008, 72, 3949–3963. [Google Scholar] [CrossRef]
- Bindschedler, S.; Cailleau, G.; Braissant, O.; Millière, L.; Job, D.; Verrecchia, E.P. Unravelling the enigmatic origin of calcitic nanofibres in soils and caves: Purely physicochemical or biogenic processes? Biogeosciences 2014, 11, 2809–2825. [Google Scholar] [CrossRef]
- Lowenstam, H.A.; Weiner, S. On Biomineralization; Oxford University Press, Inc.: New York, NY, USA, 1989. [Google Scholar]
- Ehrlich, H.I. Geomicrobiology, 4th ed.; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Skinner, H.C.W. Biominerals. Mineral. Mag. 2005, 69, 621–641. [Google Scholar] [CrossRef]
- Mann, S. The chemistry of form. Angew. Chem. Int. Ed. 2000, 39, 3393–3406. [Google Scholar] [CrossRef]
- Ritz, K.; Young, I.M. Interactions between soil structure and fungi. Mycologist 2004, 18, 52–59. [Google Scholar] [CrossRef]
- Sterflinger, K. Fungi as geologic agents. Geomicrobiol. J. 2000, 17, 97–124. [Google Scholar] [CrossRef]
- Gorbushina, A.A. Life on the rocks. Environ. Microbiol. 2007, 9, 1613–1631. [Google Scholar] [CrossRef] [PubMed]
- Vanderwolf, K.J.; Malloch, D.; McAlpine, D.F.; Forbes, G.J. A world review of fungi, yeasts, and slime molds in caves. Int. J. Speleol. 2013, 42, 77–96. [Google Scholar] [CrossRef]
- Shearer, C.A.; Descals, E.; Kohlmeyer, B.; Kohlmeyer, J.; Marvanovà, L.; Padgett, D.; Porter, D.; Raja, H.A.; Schmit, J.P.; Thorton, H.A. Fungal biodiversity in aquatic habitats. Biodivers. Conserv. 2007, 16, 49–67. [Google Scholar] [CrossRef]
- Nagahama, T.; Nagano, Y. Cultured and uncultured fungal diversity in deep-sea environments. In Biology of Marine Fungi; Springer-Verlag: Berlin, Germany; Heidelberg, Germany, 2012; pp. 173–187. [Google Scholar]
- Gadd, G.M. Geomycology: Biogeochemical transformations of rocks, minerals, metals and radionuclides by fungi, bioweathering and bioremediation. Mycol. Res. 2007, 111, 3–49. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.; Robson, G.D.; Trinci, A.P.J. 21st Century Guidebook to Fungi; Cambridge University Press: New York, NY, USA, 2011. [Google Scholar]
- Deacon, J.W. Fungal Biology; Blackwell Publishing Ltd: Malden, MA, USA, 2007. [Google Scholar]
- Klein, D.A.; Paschke, M.W. Filamentous fungi: The indeterminate lifestyle and microbial ecology. Microb. Ecol. 2004, 47, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Jongmans, A.G.; van Breemen, N.; Lundstrom, U.; van Hees, P.A.W.; Finlay, R.D.; Srinivasan, M.; Unestam, T.; Giesler, R.; Melkerud, P.A.; Olsson, M. Rock-eating fungi. Nature 1997, 389, 682–683. [Google Scholar] [CrossRef]
- Van Schöll, L.; Kuyper, T.W.; Smits, M.M.; Landeweert, R.; Hoffland, E.; van Breemen, N. Rock-eating mycorrhizas: Their role in plant nutrition and biogeochemical cycles. Plant Soil 2008, 303, 35–47. [Google Scholar] [CrossRef]
- Boswell, G.P.; Jacobs, H.; Ritz, K.; Gadd, G.M.; Davidson, F.A. The development of fungal networks in complex environments. Bull. Math. Biol. 2007, 69, 605–634. [Google Scholar] [CrossRef] [PubMed]
- Paul, E.A. Soil Microbiology, Ecology and Biochemistry; Academic Press: San Diego, CA, USA, 2007; p. 532. [Google Scholar]
- Jones, P.C.T.; Mollison, J.E.; Quenouille, M.H. A technique for the quantitative estimation of soil micro-organisms. Microbiology 1948, 2, 54–69. [Google Scholar]
- Ritz, K. Growth responses of some soil fungi to spatially heterogeneous nutrients. FEMS Microbiol. Ecol. 1995, 16, 269–279. [Google Scholar] [CrossRef]
- Wright, D.P.; Johansson, T.; Le Quéré, A.; Söderström, B.; Tunlid, A. Spatial patterns of gene expression in the extramatrical mycelium and mycorrhizal root tips formed by the ectomycorrhizal fungus Paxillus involutus in association with birch (Betula pendula) seedlings in soil microcosms. New Phytol. 2005, 167, 579–596. [Google Scholar] [CrossRef] [PubMed]
- Olsson, S. Mycelial density profiles of fungi on heterogeneous media and their interpretation in terms of nutrient reallocation patterns. Mycol. Res. 1995, 99, 143–153. [Google Scholar] [CrossRef]
- Harms, H.; Schlosser, D.; Wick, L.Y. Untapped potential: Exploiting fungi in bioremediation of hazardous chemicals. Nat. Rev. Microbiol. 2011, 9, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich-Nowoisky, J.; Burrows, S.; Xie, Z.; Engling, G.; Solomon, P.; Fraser, M.; Mayol-Bracero, O.; Artaxo, P.; Begerow, D.; Conrad, R. Biogeography in the air: Fungal diversity over land and oceans. Biogeosciences 2012, 9, 1125–1136. [Google Scholar] [CrossRef]
- Womack, A.M.; Artaxo, P.E.; Ishida, F.Y.; Mueller, R.C.; Saleska, S.R.; Wiedemann, K.T.; Bohannan, B.J.M.; Green, J.L. Characterization of active and total fungal communities in the atmosphere over the Amazon rainforest. Biogeosciences 2015, 12, 6337–6349. [Google Scholar] [CrossRef]
- Hibbett, D.S.; Binder, M.; Bischoff, J.F.; Blackwell, M.; Cannon, P.F.; Eriksson, O.E.; Huhndorf, S.; James, T.; Kirk, P.M.; Lücking, R. A higher-level phylogenetic classification of the Fungi. Mycol. Res. 2007, 111, 509–547. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis; Academic Press: New York, NY, USA, 2008. [Google Scholar]
- Stajich, J.E.; Berbee, M.L.; Blackwell, M.; Hibbett, D.S.; James, T.Y.; Spatafora, J.W.; Taylor, J.W. The fungi. Curr. Biol. 2009, 19, R840–R845. [Google Scholar] [CrossRef] [PubMed]
- Burford, E.P.; Fomina, M.; Gadd, G.M. Fungal involvement in bioweathering and biotransformation of rocks and minerals. Mineral. Mag. 2003, 67, 1127–1155. [Google Scholar] [CrossRef]
- Gadd, G.M.; Fomina, M.; Burford, E.P. Fungal roles and function in rock, mineral and soil transformations. In Microorganisms and Earth Systems—Advances in Geomicrobiology; Gadd, G.M., Semple, K.T., Lappin-Scott, H.M., Eds.; Cambridge University Press: New York, NY, USA, 2005; pp. 201–231. [Google Scholar]
- Parchert, K.J.; Spilde, M.N.; Porras-Alfaro, A.; Nyberg, A.M.; Northup, D.E. Fungal communities associated with rock varnish in black canyon, New Mexico: Casual inhabitants or essential partners? Geomicrobiol. J. 2012, 29, 752–766. [Google Scholar] [CrossRef]
- Gadd, G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiology 2010, 156, 609–643. [Google Scholar] [CrossRef] [PubMed]
- Fomina, M.A.; Alexander, I.J.; Colpaert, J.V.; Gadd, G.M. Solubilization of toxic metal minerals and metal tolerance of mycorrhizal fungi. Soil Biol. Biochem. 2005, 37, 851–866. [Google Scholar] [CrossRef]
- Kolo, K.; Keppens, E.; Preat, A.; Claeys, P. Experimental observations on fungal diagenesis of carbonate substrates. J. Geophys. Res. Biogeosci. 2007, 112. [Google Scholar] [CrossRef]
- Fomina, M.; Burford, E.P.; Hillier, S.; Kierans, M.; Gadd, G.M. Rock-building fungi. Geomicrobiol. J. 2010, 27, 624–629. [Google Scholar] [CrossRef]
- Gadd, G.M. Interactions of fungi with toxic metals. New Phytol. 1993, 124, 25–60. [Google Scholar] [CrossRef]
- Fomina, M.; Hillier, S.; Charnock, J.M.; Melville, K.; Alexander, I.J.; Gadd, G.M. Role of oxalic acid overexcretion in transformations of toxic metal minerals by Beauveria caledonica. Appl. Environ. Microbiol. 2005, 71, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Verrecchia, E.P. Fungi and sediments. In Microbial Sediments; Riding, R.E., Awramik, S.M., Eds.; Springer-Verlag: Berlin, Heidelberg, Germany, 2000. [Google Scholar]
- Hoffland, E.; Kuyper, T.W.; Wallander, H.; Plassard, C.; Gorbushina, A.A.; Haselwandter, K.; Holmstrom, S.; Landeweert, R.; Lundstrom, U.S.; Rosling, A.; et al. The role of fungi in weathering. Front. Ecol. Environ. 2004, 2, 258–264. [Google Scholar] [CrossRef]
- Cromack, K., Jr.; Sollins, P.; Todd, R.L.; Fogel, R.; Todd, A.W.; Fender, W.M.; Crossley, M.E.; Crossley, D.A., Jr. The role of oxalic acid and bicarbonate in calcium cycling by fungi and bacteria: Some possible implications for soil animals. In Proceedings of the VI International Soil Zoology Colloquium, Uppsala, Sweden, 21–25 June 1976.
- Gadd, G.M. Fungal production of citric and oxalic acid: Importance in metal speciation, physiology and biogeochemical processes. In Advances in Microbial Physiology; Academic Press: Cambridge, MA, USA, 1999; Volume 41, pp. 47–92. [Google Scholar]
- Adeyemi, A.O.; Gadd, G.M. Fungal degradation of calcium-, lead-and silicon-bearing minerals. Biometals 2005, 18, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Schilling, J.S.; Jellison, J. Oxalate regulation by two brown rot fungi decaying oxalate-amended and non-amended wood. Holzforschung 2005, 59, 681–688. [Google Scholar] [CrossRef]
- Tuason, M.M.S.; Arocena, J.M. Calcium oxalate biomineralization by Piloderma fallax in response to various levels of calcium and phosphorus. Appl. Environ. Microbiol. 2009, 75, 7079–7085. [Google Scholar] [CrossRef] [PubMed]
- Gadd, G.M. Fungi in Biogeochemical Cycles; Cambridge University Press: Cambridge, UK, 2006; Volume 24. [Google Scholar]
- Gadd, G.M. Bacterial and fungal geomicrobiology: A problem with communities? Geobiology 2008, 6, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Gadd, G.M.; Raven, J.A. Geomicrobiology of eukaryotic microorganisms. Geomicrobiol. J. 2010, 27, 491–519. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters; John Wiley & Sons Inc.: New York, NY, USA, 1996; p. 1022. [Google Scholar]
- Dutton, M.V.; Evans, C.S. Oxalate production by fungi: Its role in pathogenicity and ecology in the soil environment. Can. J. Microbiol. 1996, 42, 881–895. [Google Scholar] [CrossRef]
- Oggerin, M.; Rodriguez, N.; del Moral, C.; Amils, R. Fungal jarosite biomineralization in Rio Tinto. Res. Microbiol. 2014, 165, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Fomina, M.; Podgorsky, V.S.; Olishevska, S.V.; Kadoshnikov, V.M.; Pisanska, I.R.; Hillier, S.; Gadd, G.M. Fungal deterioration of barrier concrete used in nuclear waste disposal. Geomicrobiol. J. 2007, 24, 643–653. [Google Scholar] [CrossRef]
- Fomina, M.; Charnock, J.M.; Hillier, S.; Alvarez, R.; Livens, F.; Gadd, G.M. Role of fungi in the biogeochemical fate of depleted uranium. Curr. Biol. 2008, 18, R375–R377. [Google Scholar] [CrossRef] [PubMed]
- Rhee, Y.J.; Hillier, S.; Gadd, G.M. Lead transformation to pyromorphite by fungi. Curr. Biol. 2012, 22, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Crusberg, T.C. Biomineralization of copper by a fungus revealed by SEM. Microsc. Anal. 2004, 18, 11–13. [Google Scholar]
- Liang, X.; Hillier, S.; Pendlowski, H.; Gray, N.; Ceci, A.; Gadd, G.M. Uranium phosphate biomineralization by fungi. Environ. Microbiol. 2015, 17, 2064–2075. [Google Scholar] [CrossRef] [PubMed]
- Verrecchia, E.P.; Dumont, J.L. A biogeochemical model for chalk alteration by fungi in semiarid environments. Biogeochemistry 1996, 35, 447–470. [Google Scholar] [CrossRef]
- Burford, E.P.; Hillier, S.; Gadd, G.M. Biomineralization of fungal hyphae with calcite (CaCO3) and calcium oxalate mono- and dihydrate in Carboniferous limestone microcosms. Geomicrobiol. J. 2006, 23, 599–611. [Google Scholar] [CrossRef]
- Hou, W.; Dou, C.; Lian, B.; Dong, H. The interaction of fungus with calcite and the effects on aqueous geochemistry in karst systems. Carbonates Evaporites 2013, 28, 413–418. [Google Scholar] [CrossRef]
- Castanier, S.; le Métayer-Levrel, G.L.; Perthuisot, J.-P. Ca-carbonates precipitation and limestone genesis—The microbiogeologist point of view. Sediment. Geol. 1999, 126, 9–23. [Google Scholar] [CrossRef]
- Bowen, A.D.; Davidson, F.A.; Keatch, R.; Gadd, G.M. Induction of contour sensing in Aspergillus niger by stress and its relevance to fungal growth mechanics and hyphal tip structure. Fungal Genet. Biol. 2007, 44, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Verrecchia, E.P.; Braissant, O.; Cailleau, G. The oxalate-carbonate pathway in soil carbon storage: The role of fungi and oxalotrophic bacteria. In Fungi in Biogeochemical Cycles; Cambridge University Press: New York, NY, USA, 2006; pp. 289–310. [Google Scholar]
- Guggiari, M.; Bloque, R.; Aragno, M.; Verrecchia, E.; Job, D.; Junier, P. Experimental calcium-oxalate crystal production and dissolution by selected wood-rot fungi. Int. Biodeterior. Biodegrad. 2011, 65, 803–809. [Google Scholar] [CrossRef]
- Burbank, M.B.; Weaver, T.J.; Green, T.L.; Williams, B.C.; Crawford, R.L. Precipitation of calcite by indigenous microorganisms to strengthen liquefiable soils. Geomicrobiol. J. 2011, 28, 301–312. [Google Scholar] [CrossRef]
- Li, Q.; Csetenyi, L.; Gadd, G.M. Biomineralization of metal carbonates by Neurospora crassa. Environ. Sci. Technol. 2014, 48, 14409–14416. [Google Scholar] [CrossRef] [PubMed]
- Hou, W.; Lian, B.; Zhang, X. CO2 mineralization induced by fungal nitrate assimilation. Bioresour. Technol. 2011, 102, 1562–1566. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.L.; Heath, I.B. Roles of calcium-ions in hyphal tip growth. Microbiol. Rev. 1993, 57, 367–382. [Google Scholar] [PubMed]
- Pitt, D.; Ugalde, U.O. Calcium in fungi. Plant Cell Environ. 1984, 7, 467–475. [Google Scholar] [CrossRef]
- Manoli, F.; Koutsopoulos, S.; Dalas, E. Crystallization of calcite on chitin. J. Cryst. Growth 1997, 182, 116–124. [Google Scholar] [CrossRef]
- Whitney, K. Systems of biomineralization in the fungi. In Origin, Evolution and Modern Aspects of Biomineralization in Plants and Animals; Crick, R.E., Ed.; Springer-Verlag: Berlin, Heidelberg, Germany, 1989. [Google Scholar]
- Simkiss, K. The processes of biomineralization in lower plants and animals—An overview. Biominer. Lower Plants Anim. 1986, 30, 19–37. [Google Scholar]
- Schultze-Lam, S.; Fortin, D.; Davis, B.S.; Beveridge, T.J. Mineralization of bacterial surfaces. Chem. Geol. 1996, 132, 171–181. [Google Scholar] [CrossRef]
- Barton, H.A.; Northup, D.E. Geomicrobiology in cave environments: Past, current and future perspectives. J. Cave Karst Stud. 2007, 69, 163–178. [Google Scholar]
- Klappa, C. Calcified filaments in Quaternary calcretes: Organo-mineral interactions in the subaerial vadose environment. J. Sediment. Petrol. 1979, 49, 955–968. [Google Scholar] [CrossRef]
- Wright, V.P. The role of fungal biomineralization in the formation of Early Carboniferous soil fabrics. Sedimentology 1986, 33, 831–838. [Google Scholar] [CrossRef]
- Monger, H.C.; Daugherty, L.A.; Lindemann, W.C.; Liddell, C.M. Microbial precipitation of pedogenic calcite. Geology 1991, 19, 997–1000. [Google Scholar] [CrossRef]
- Cailleau, G.; Verrecchia, E.P.; Braissant, O.; Emmanuel, L. The biogenic origin of needle fibre calcite. Sedimentology 2009, 56, 1858–1875. [Google Scholar] [CrossRef]
- Bindschedler, S.; Millière, L.; Cailleau, G.; Job, D.; Verrecchia, E.P. Calcitic Nanofibres in Soils and Caves: A Putative Fungal Contribution to Carbonatogenesis; Geological Society, London, Special Publications: London, UK, 2010; Volume 336, pp. 225–238. [Google Scholar]
- Bindschedler, S.; Millière, L.; Cailleau, G.; Job, D.; Verrecchia, E.P. An ultrastructural approach to analogies between fungal structures and needle fiber calcite. Geomicrobiol. J. 2012, 29, 301–313. [Google Scholar] [CrossRef]
- Li, Q.; Csetenyi, L.; Paton, G.I.; Gadd, G.M. CaCO3 and SrCO3 bioprecipitation by fungi isolated from calcareous soil. Environ. Microbiol. 2015, 17, 3082–3097. [Google Scholar] [CrossRef] [PubMed]
- Addadi, L.; Raz, S.; Weiner, S. Taking advantage of disorder: Amorphous calcium carbonate and its roles in biomineralization. Adv. Mater. 2003, 15, 959–970. [Google Scholar] [CrossRef]
- Benzerara, K.; Menguy, N.; Guyot, F.; Dominici, C.; Gillet, P. Nanobacteria-like calcite single crystals at the surface of the tataouine meteorite. Proc. Natl. Acad. Sci. USA 2003, 100, 7438–7442. [Google Scholar] [CrossRef]
- Faatz, M.; Gröhn, F.; Wegner, G. Amorphous calcium carbonate: Synthesis and potential intermediate in biomineralization. Adv. Mater. 2004, 16, 996–1000. [Google Scholar] [CrossRef]
- Rautaray, D.; Ahmad, A.; Sastry, M. Biosynthesis of CaCO3 crystals of complex morphology using a fungus and an actinomycete. J. Am. Chem. Soc. 2003, 125, 14656–14657. [Google Scholar] [CrossRef] [PubMed]
- Rautaray, D.; Ahmad, A.; Sastry, M. Biological synthesis of metal carbonate minerals using fungi and actinomycetes. J. Mater. Chem. 2004, 14, 2333–2340. [Google Scholar] [CrossRef]
- Ahmad, A.; Rautaray, D.; Sastry, M. Biogenic calcium carbonate: Calcite crystals of variable morphology by the reaction of aqueous Ca2+ ions with fungi. Adv. Funct. Mater. 2004, 14, 1075–1080. [Google Scholar] [CrossRef]
- Sanyal, A.; Rautaray, D.; Bansal, V.; Ahmad, A.; Sastry, M. Heavy-metal remediation by a fungus as a means of production of lead and cadmium carbonate crystals. Langmuir 2005, 21, 7220–7224. [Google Scholar] [CrossRef] [PubMed]
- Masaphy, S.; Zabari, L.; Pastrana, J.; Dultz, S. Role of fungal mycelium in the formation of carbonate concretions in growing media—An investigation by SEM and synchrotron-based X-ray tomographic microscopy. Geomicrobiol. J. 2009, 26, 442–450. [Google Scholar] [CrossRef]
- Verrecchia, E.P.; Dumont, J.L.; Rolko, K.E. Do fungi building limestones exist in semi-arid regions? Naturwissenschaften 1990, 77, 584–586. [Google Scholar] [CrossRef]
- Verrecchia, E.P.; Verrecchia, K.E. Needle-fiber calcite—A critical-review and a proposed classification. J. Sediment. Res. Sect. Sediment. Petrol. Processes 1994, 64, 650–664. [Google Scholar]
- Cailleau, G.; Dadras, M.; Abolhassani-Dadras, S.; Braissant, O.; Verrecchia, E.P. Evidence for an organic origin of pedogenic calcitic nanofibres. J. Cryst. Growth 2009, 311, 2490–2495. [Google Scholar] [CrossRef]
- Millière, L.; Hasinger, O.; Bindschedler, S.; Cailleau, G.; Spangenberg, J.E.; Verrecchia, E.P. Stable carbon and oxygen isotope signatures of pedogenic needle fibre calcite. Geoderma 2011, 161, 74–87. [Google Scholar] [CrossRef]
- Kubiëna, W.L. Micropedology. Soil Sci. 1939, 47, 163. [Google Scholar] [CrossRef]
- Verges, V.; Madon, M.; Bruand, A.; Bocquier, G. Morphologie et cristallogenèse de microcristaux supergènes de calcite en aiguilles. Bull. Minéral. 1982, 105, 351–356. (In French) [Google Scholar]
- Jones, B.; Ng, K.C. The structure and diagenesis of rhizoliths from Cayman Brac, British West Indies. J. Sediment. Petrol. 1988, 58, 457–467. [Google Scholar]
- Callot, G.; Guyon, A.; Mousain, D. Relation between calcite needles and fungal hyphae in the soil. Agronomie 1985, 5, 209–216. [Google Scholar] [CrossRef]
- Phillips, S.E.; Self, P.G. Morphology, crystallography and origin of needle-fiber calcite in quaternary pedogenic calcretes of South Australia. Aust. J. Soil Res. 1987, 25, 429–444. [Google Scholar] [CrossRef]
- Ould Mohamed, S.; Bruand, A. Morphology and origin of secondary calcite in soils from Beauce, France. Dev. Soil Sci. 1993, 22, 27–36. [Google Scholar]
- Borsato, A.; Frisia, S.; Jones, B.; Van der Borg, K. Calcite moonmilk: Crystal morphology and environment of formation in caves in the Italian Alps. J. Sediment. Res. 2000, 70, 1171–1182. [Google Scholar] [CrossRef]
- Javaux, E.J. Biomarkers. In Encyclopedia of Astrobiology; Springer-Verlag: Berlin, Germany; Heidelberg, Germany, 2011; pp. 182–183. [Google Scholar]
- Simoneit, B.R.T. A review of current applications of mass spectrometry for biomarker/molecular tracer elucidations. Mass Spectrom. Rev. 2005, 24, 719–765. [Google Scholar] [CrossRef] [PubMed]
- Simkiss, K.; Wilbur, K.M. Biomineralization: Cell Biology and Mineralization; Academic Press: New York, NY, USA, 1989. [Google Scholar]
- Stumm, W. Chemistry of the Solid-Water Interface: Processes at the Mineral-Water and Particle-Water Interface in Natural Systems; John Wiley & Son Inc.: New York, NY, USA, 1992. [Google Scholar]
- Barton, H.A.; Spear, J.R.; Pace, N.R. Microbial life in the underworld: Biogenicity in secondary mineral formations. Geomicrobiol. J. 2001, 18, 359–368. [Google Scholar]
- Blyth, A.J.; Frisia, S. Molecular evidence for bacterial mediation of calcite formation in cold high-altitude caves. Geomicrobiol. J. 2008, 25, 101–111. [Google Scholar] [CrossRef]
- Benzerara, K.; Menguy, N. Looking for traces of life in minerals. Comptes Rendus Palevol 2009, 8, 617–628. [Google Scholar] [CrossRef]
- Yoon, T.H. Applications of soft X-ray spectromicroscopy in material and environmental sciences. Appl. Spectrosc. Rev. 2009, 44, 91–122. [Google Scholar] [CrossRef]
- Gilbert, P.; Abrecht, M.; Frazer, B.H. The organic-mineral interface in biominerals. Rev. Mineral. Geochem. 2005, 59, 157–185. [Google Scholar]
- Cavalazzi, B. Global to nano-scale relevance of Ca-carbonate biosignatures. Boll. Soc. Paleontol. Ital. 2013, 52, 139–140. [Google Scholar]
- Smits, M.M.; Herrmann, A.M.; Duane, M.; Duckworth, O.W.; Bonneville, S.; Benning, L.G.; Lundström, U. The fungal-mineral interface: Challenges and considerations of micro-analytical developments. Fungal Biol. Rev. 2009, 23, 122–131. [Google Scholar]
- Dykstra, M.J.; Reuss, L.E. Biological Electron Microscopy: Theory, Techniques, and Troubleshooting; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2011. [Google Scholar]
- Canaveras, J.C.; Hoyos, M.; Sanchez-Moral, S.; Sanz-Rubio, E.; Bedoya, J.; Soler, V.; Groth, I.; Schumann, P.; Laiz, L.; Gonzalez, I.; et al. Microbial communities associated with hydromagnesite and needle-fiber aragonite deposits in a karstic cave (Altamira, Northern Spain). Geomicrobiol. J. 1999, 16, 9–25. [Google Scholar]
- Benzerara, K.; Menguy, N.; Lopez-Garcia, P.; Yoon, T.H.; Kazmierczak, J.; Tyliszczak, T.; Guyot, F.; Brown, G.E. Nanoscale detection of organic signatures in carbonate microbialites. Proc. Natl. Acad. Sci. USA 2006, 103, 9440–9445. [Google Scholar]
- Benzerara, K.; Miller, V.M.; Barell, G.; Kumar, V.; Miot, J.; Brown, G.E.; Lieske, J.C. Search for microbial signatures within human and microbial calcifications using soft X-ray spectromicroscopy. J. Investig. Med. 2006, 54, 367–379. [Google Scholar] [CrossRef]
- Obst, M.; Wang, J.; Hitchcock, A.P. Soft X-ray spectro-tomography study of cyanobacterial biomineral nucleation. Geobiology 2009, 7, 577–591. [Google Scholar] [PubMed]
- Benzerara, K.; Yoon, T.H.; Tyliszczak, T.; Constantz, B.; Spormann, A.M.; Brown, G.E. Scanning transmission X-ray microscopy study of microbial calcification. Geobiology 2004, 2, 249–259. [Google Scholar] [CrossRef]
- Wagner, M. Single-cell ecophysiology of microbes as revealed by Raman microspectroscopy or secondary ion mass spectrometry imaging. Annu. Rev. Microbiol. 2009, 63, 411–429. [Google Scholar] [CrossRef] [PubMed]
- Kilburn, M.R.; Wacey, D. Nanoscale secondary ion mass spectrometry (NanoSIMS) as an analytical tool in the geosciences. In Principles and practive of analytical techniques in geosciences; Grice, K., Ed.; The Royal Society of Chemistry: Cambridge, UK, 2015; pp. 1–34. [Google Scholar]
- Anderson, I.C.; Cairney, J.W. Diversity and ecology of soil fungal communities: Increased understanding through the application of molecular techniques. Environ. Microbiol. 2004, 6, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Peay, K.G.; Kennedy, P.G.; Bruns, T.D. Fungal community ecology: A hybrid beast with a molecular master. Bioscience 2008, 58, 799–810. [Google Scholar] [CrossRef]
- Lindahl, B.D.; Nilsson, R.H.; Tedersoo, L.; Abarenkov, K.; Carlsen, T.; Kjoller, R.; Koljalg, U.; Pennanen, T.; Rosendahl, S.; Stenlid, J.; et al. Fungal community analysis by high-throughput sequencing of amplified markers : A user’s guide. New Phytol. 2013, 199, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, M.J.; Nelson, W.; Soderlund, C.; Maier, R.M.; Pryor, B.M. Assessing fungal community structure from mineral surfaces in Kartchner caverns using multiplexed 454 pyrosequencing. Microb. Ecol. 2015, 70, 175–187. [Google Scholar] [CrossRef] [PubMed]
- Jurado, V.; Fernandez-Cortes, A.; Cuezva, S.; Laiz, L.; Canaveras, J.C.; Sanchez-Moral, S.; Saiz-Jimenez, C. The fungal colonisation of rock-art caves: Experimental evidence. Naturwissenschaften 2009, 96, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Seifert, K.A. Progress towards DNA barcoding of fungi. Mol. Ecol. Resour. 2009, 9, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Porter, T.M.; Golding, G.B. Factors that affect large subunit ribosomal DNA amplicon sequencing studies of fungal communities: Classification method, primer choice, and error. PLoS ONE 2012, 7, e35749. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bindschedler, S.; Cailleau, G.; Verrecchia, E. Role of Fungi in the Biomineralization of Calcite. Minerals 2016, 6, 41. https://doi.org/10.3390/min6020041
Bindschedler S, Cailleau G, Verrecchia E. Role of Fungi in the Biomineralization of Calcite. Minerals. 2016; 6(2):41. https://doi.org/10.3390/min6020041
Chicago/Turabian StyleBindschedler, Saskia, Guillaume Cailleau, and Eric Verrecchia. 2016. "Role of Fungi in the Biomineralization of Calcite" Minerals 6, no. 2: 41. https://doi.org/10.3390/min6020041
APA StyleBindschedler, S., Cailleau, G., & Verrecchia, E. (2016). Role of Fungi in the Biomineralization of Calcite. Minerals, 6(2), 41. https://doi.org/10.3390/min6020041




