Use of Phosphate Solubilizing Bacteria to Leach Rare Earth Elements from Monazite-Bearing Ore
Abstract
:1. Introduction
2. Experimental Section
2.1. Bacterial Strains and Culture Conditions
2.2. Comparison of Halo Zone Formation, Organic Acid Production, and Phosphate Solubilization by Phosphate Solubilizing Bacteria
2.3. Ore Characterization
| Element | Ce | La | Nd | Pr | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | K2O | Sr | TiO2 | P2O5 |
| Content (wt%) | 3.50 | 2.09 | 0.75 | 0.21 | 12.77 | 0.73 | 39.80 | 7.52 | 6.26 | <0.001 | 0.47 | 0.02 | 5.33 |
2.4. Monazite-Bearing Ore Leaching by Phosphate Solubilizing Bacteria
3. Results and Discussion
3.1. Comparison of Phosphate Solubilization by Phosphate Solubilizing Bacteria
| Bacterial Species | Halo Size (mm) * | |
|---|---|---|
| Glucose | Sucrose | |
| P. rhizosphaerae DSM 16299T | 13.5 | 13.4 |
| P. putida DSM 291T | -** | 7.2 |
| P. fluorescens DSM 50090T | 5.6 | 7.8 |
| B. megaterium DSM 32T | 4.7 | 12.1 |
| P. polymyxa DSM 36T | - | - |
| E. meliloti DSM 30135T | - | - |
| A. brasilense DSM 1690T | - | - |
| A. lipoferum DSM 1842 | - | - |
| M. ciceri | 5.1 | 4.4 |
| A. aceti DSM 2002 | 41.0 | 12.58 |
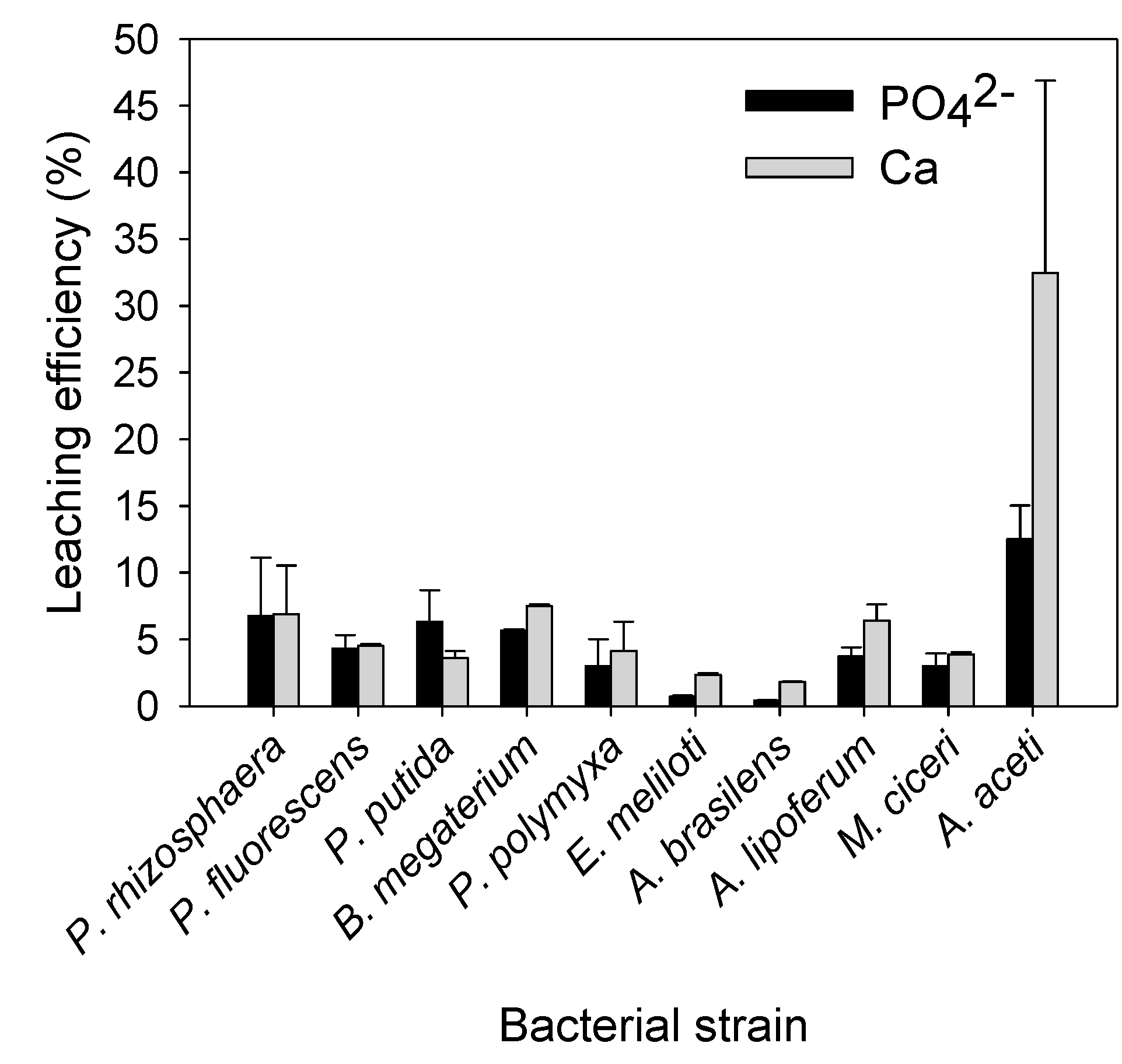
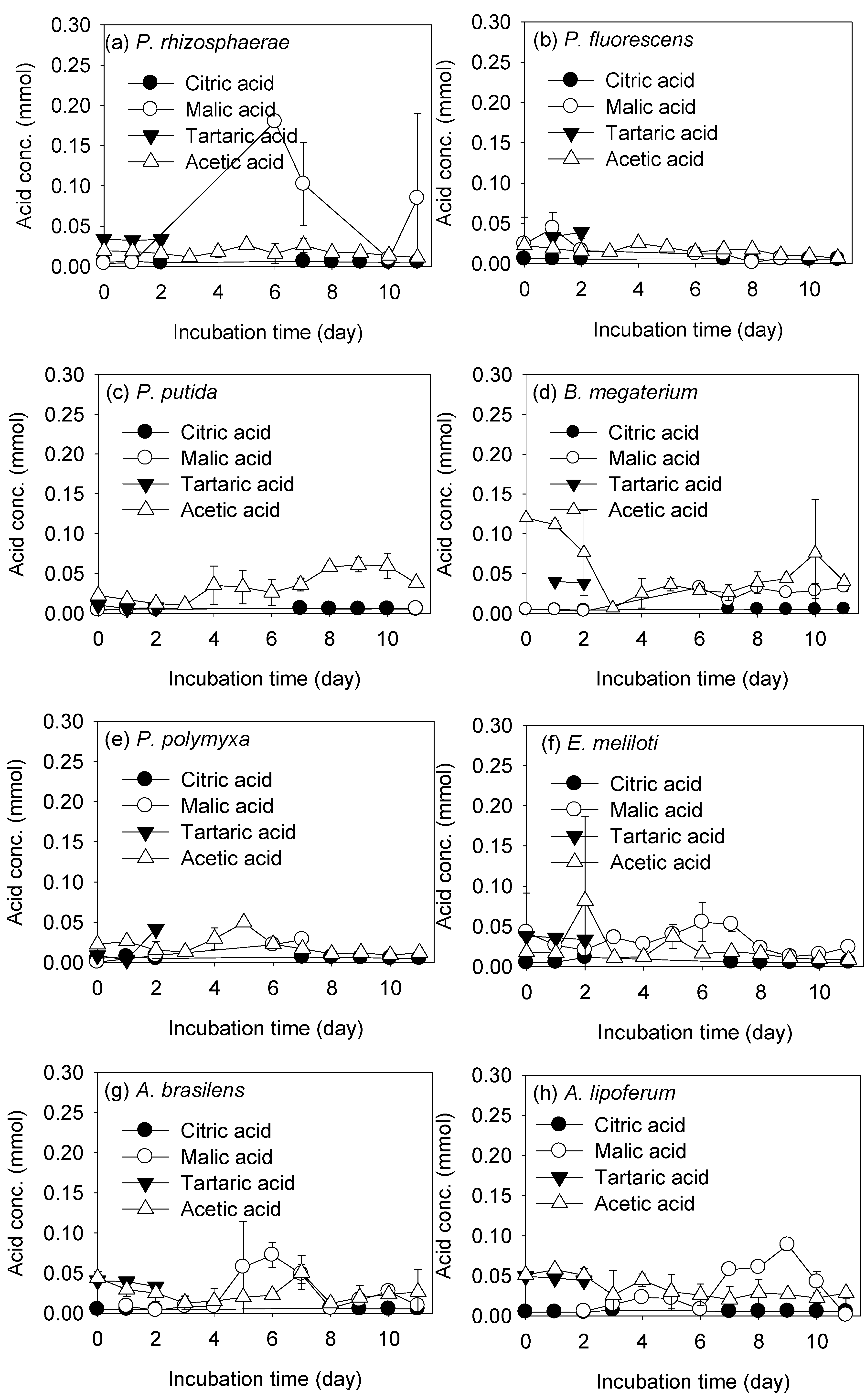
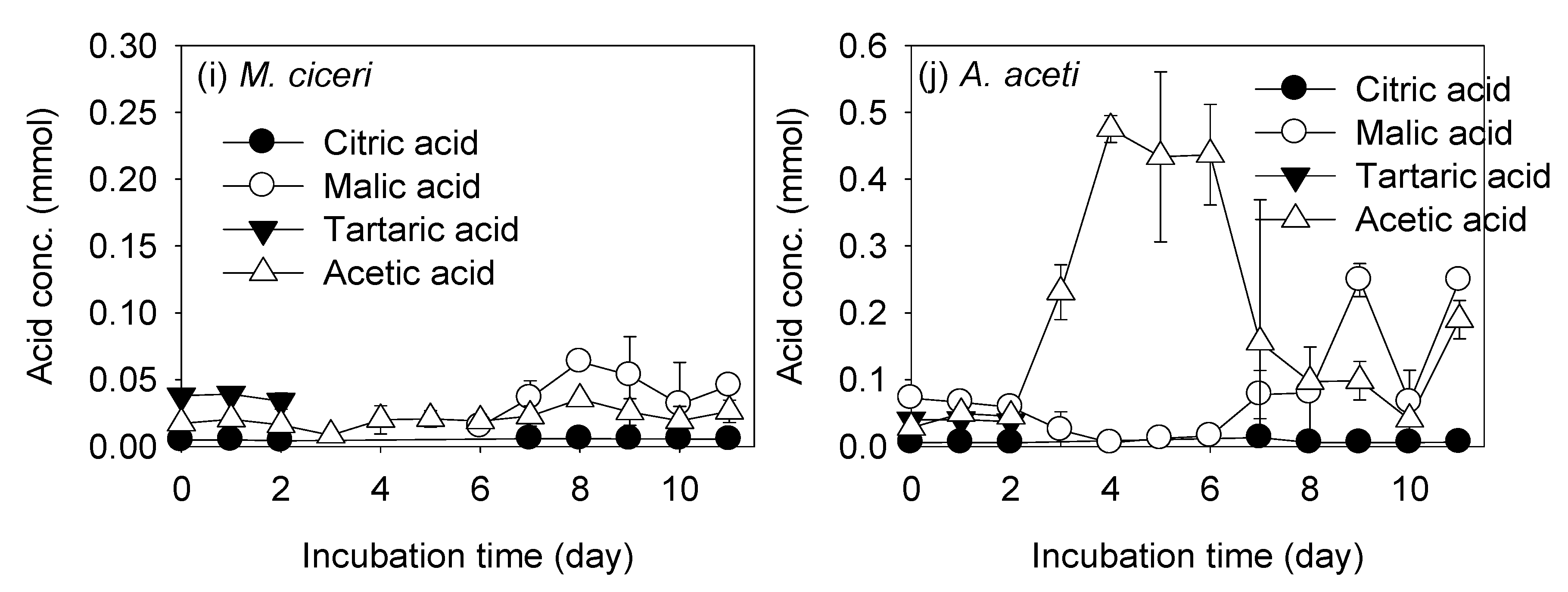
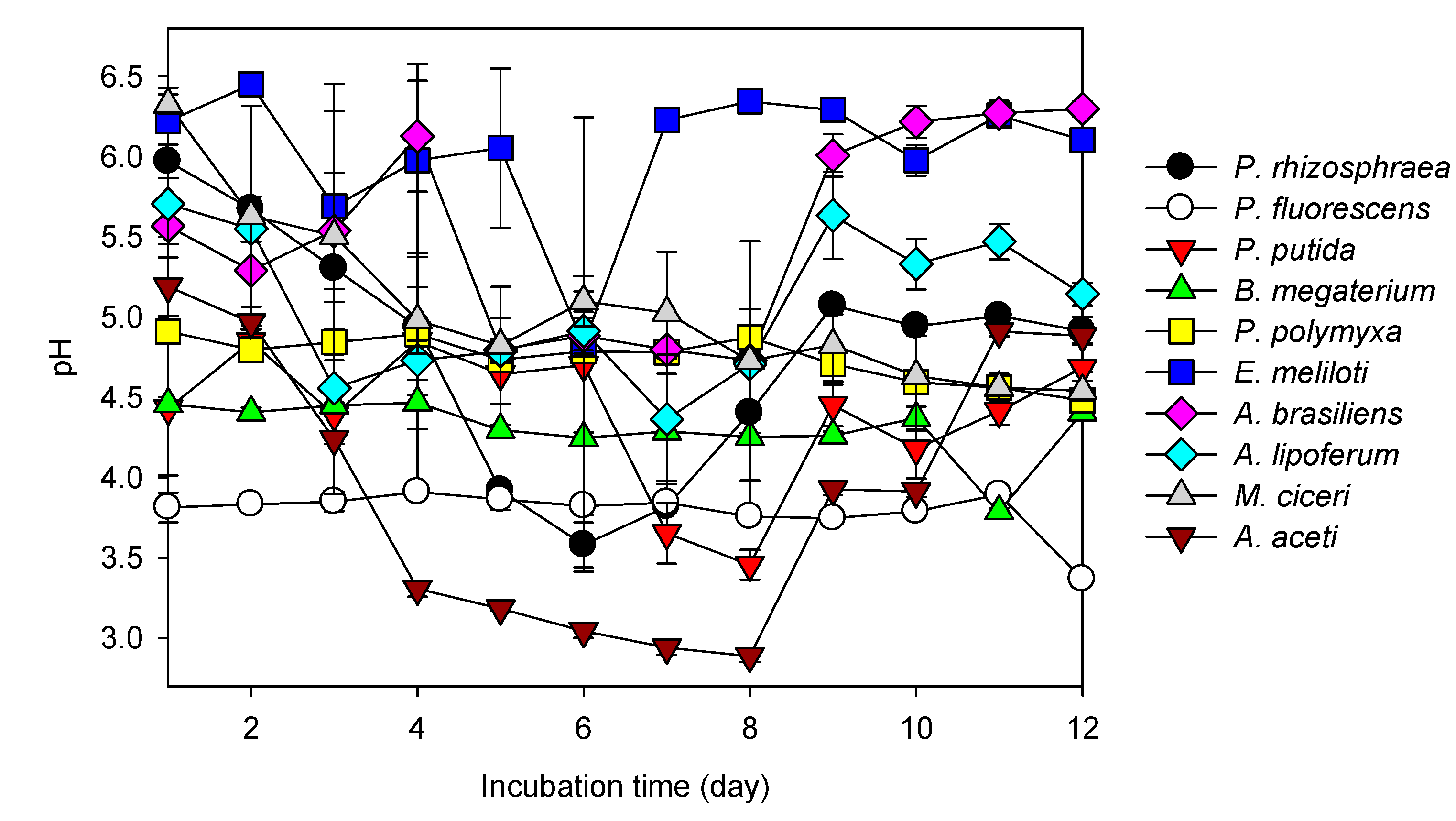
3.2. REE Leaching by the Test PSB from the Monazite-Bearing Ore and Comparison with the Screening Study Data
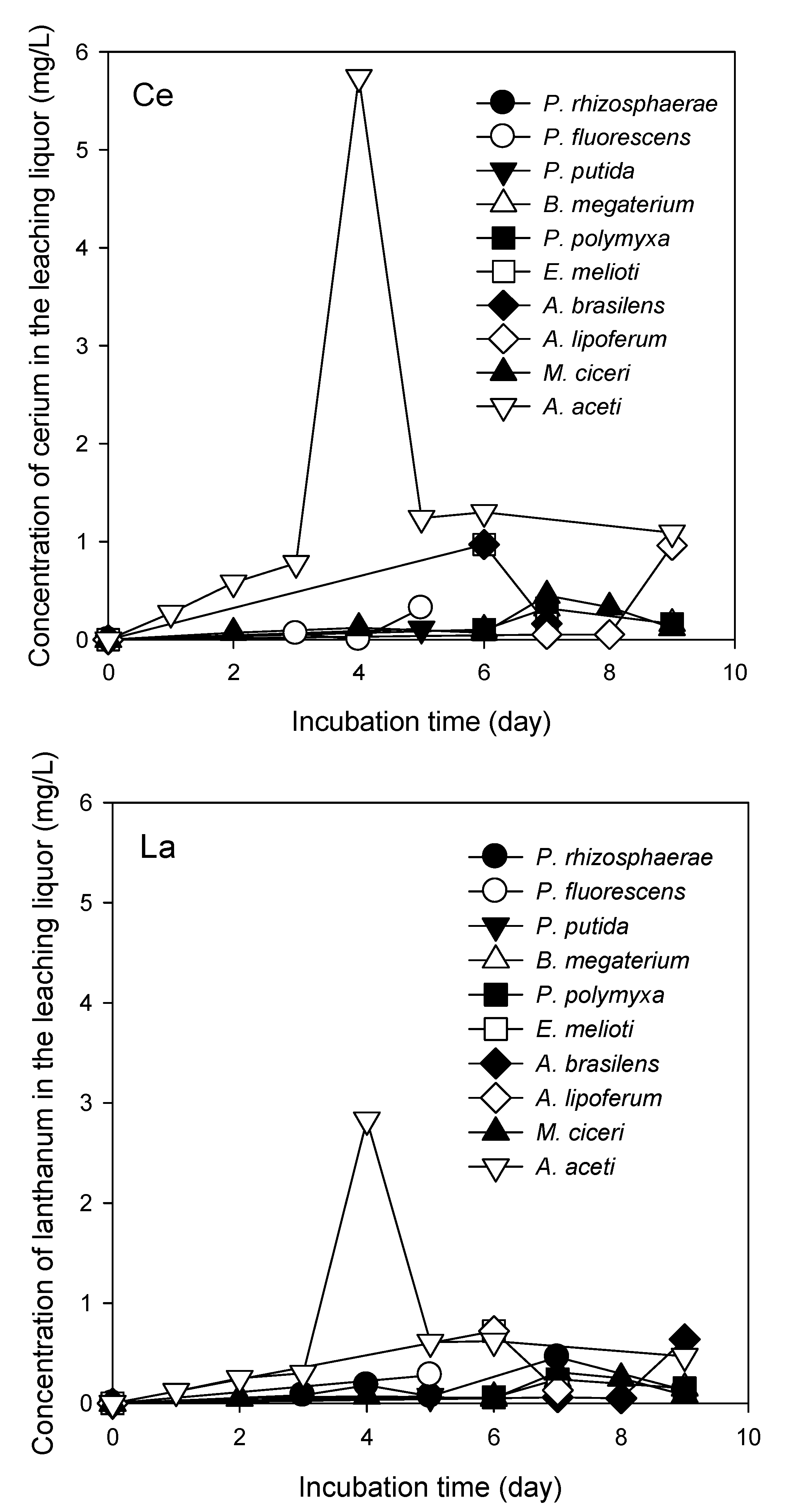
| Spot No. in Figure 5 | Mass % | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | O | Mg | Al | Si | P | S | Ca | Fe | La | Ce | Pr | |
| 1 | 82.1 | 4.88 | 1.67 | - | - | - | - | 7.06 | - | 0.18 | 4.1 | - |
| 2 | 46.0 | 22.7 | 1.28 | - | 5.4 | - | 8.2 | - | - | 3.36 | 12.5 | 0.62 |
| 3 | 61.3 | 16.4 | 1.05 | 0.24 | 8.77 | 2.15 | - | - | 9.26 | - | - | - |
| 4 | 70.6 | 15.8 | - | 4.33 | 9.3 | - | 0.56 | - | - | - | - | - |

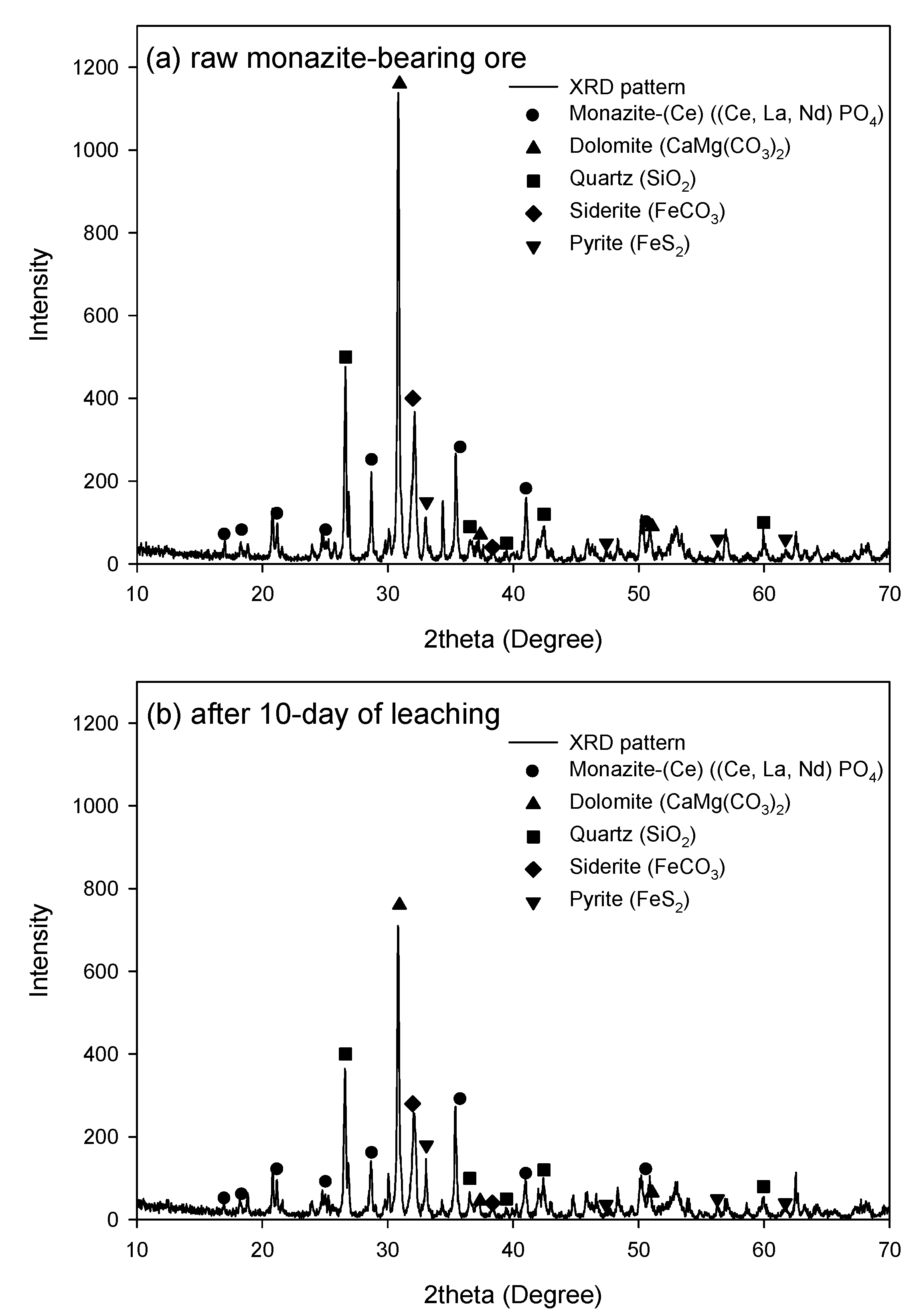
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- British Geological Survey. Rare Earth Elements. Available online: http://www.MineralsUK.com (accessed on 27 March 2015).
- Gupta, C.K.; Krishnamurthy, N. Extractive Metallurgy of Rare Earths; CRC Press: New York, NY, USA, 2005. [Google Scholar]
- Maestro, P.; Huguenin, D. Industrial applications of rare earths: Which way for the end of the century. J. Alloy. Compd. 1995, 225, 520–528. [Google Scholar] [CrossRef]
- Habashi, F. Handbook of Extractive Metallurgy; Wiley-VCH: Heidelberg, Germany, 1997. [Google Scholar]
- Zongsen, Y.; Minbo, C. Rare Earth Elements and Their Applications; Metallurgical Industry Press: Beijing, China, 1995. [Google Scholar]
- Park, H.-K.; Lee, J.-Y.; Cho, S.-W.; Kim, J.-S. Overview on the Technologies for Extraction of Rare Earth Metals. J. Korean Inst. Resour. Recycl. 2012, 21, 74–83. [Google Scholar] [CrossRef]
- Desouky, O.A.; El-Mougith, A.A.; Hassanien, W.A.; Awadalla, G.S.; Hussien, S.S. Extraction of some strategic elements from thorium–uranium concentrate using bioproducts of Aspergillus ficuum and Pseudomonas aeruginosa. Arabian J. Chem. 2011. [Google Scholar] [CrossRef]
- Hassanien, W.A.G.; Desouky, O.A.N.; HussienN, S.S.E. Bioleaching of some Rare Earth Elements from Egyptian Monazite using Aspergillus ficuum and Pseudomonas aeruginosa. Walailak J. Sci. Technol. 2014, 11, 809–823. [Google Scholar]
- Goyne, K.W.; Brantley, S.L.; Chorover, J. Rare earth element release from phosphate minerals in the presence of organic acids. Chem. Geol. 2010, 278, 1–14. [Google Scholar] [CrossRef]
- Chung, H.; Park, M.; Madhaiyan, M.; Seshadri, S.; Song, J.; Cho, H.; Sa, T. Isolation and characterization of phosphate solubilizing bacteria from the rhizosphere of crop plants of Korea. Soil. Biol. Biochem. 2005, 37, 1970–1974. [Google Scholar] [CrossRef]
- Gadagi, R.S.; Sa, T. New isolation method for microorganisms solulbilizing iron and aluminum phosphates using dyes. Soil Sci. Plant Nutr. 2002, 48, 615–618. [Google Scholar] [CrossRef]
- Gadd, G.M. Fungal Production of Citric and Oxalic Acid: Importance in Metal Speciation, Physiology and Biogeochemical Processes. In Advances in Microbial Physiology; Poole, R.K., Ed.; Academic Press: Waltham, MA, USA, 1999; Volume 41, pp. 47–92. [Google Scholar]
- Chen, Y.P.; Rekha, P.D.; Arun, A.B.; Shen, F.T.; Lai, W.A.; Young, C.C. Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl. Soil Ecol. 2006, 34, 33–41. [Google Scholar] [CrossRef]
- Rodriguez, H.; Fraga, R. Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol. Adv. 1999, 17, 319–339. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.; Moon, H.S.; Nam, K.; Kim, J.Y.; Kim, T.S. Application of phosphate-solubilizing bacteria for enhancing bioavailability and phytoextraction of cadmium (Cd) from polluted soil. Chemosphere 2012, 88, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Peix, A.; Rivas, R.; Mateos, P.F.; Martínez-Molina, E.; Rodríguez-Barrueco, C.; Velázquez, E. Pseudomonas rhizosphaerae sp. nov., a novel species that actively solubilizes phosphate in vitro. Int. J. Syst. Evol. Microbiol. 2003, 53, 2067–2072. [Google Scholar] [CrossRef] [PubMed]
- Gholami, A.; Shahsavani, S.; Nezarat, S. The Effect of Plant Growth Promoting Rhizobacteria (PGPR) on Germination, Seedling Growth and Yield of Maize. Int. J. Biol. Life Sci. 2009, 1, 35–40. [Google Scholar]
- Tilak, K.V.B.R.; Ranganayaki, N.; Pal, K.K.; De, R.; Saxena, A.K.; Nautiyal, C.S.; Mittal, S.; Tripathi, A.K.; Johri, B.N. Diversity of plant growth and soil health supporting bacteria. Curr. Sci. 2005, 89, 136–150. [Google Scholar]
- Halder, A.K.; Chakrabartty, P.K. Solubilization of inorganic phosphate by Rhizobium. Folia Microbiol. 1993, 38, 325–330. [Google Scholar] [CrossRef]
- Reyes, I.; Bernier, L.; Simard, R.R.; Tanguay, P.; Antoun, H. Characteristics of phosphate solubilization by an isolate of a tropical Penicillium rugulosum and two UV-induced mutants. FEMS Microbiol. Ecol. 1999, 28, 291–295. [Google Scholar] [CrossRef]
- Briggs, P.H.; Meier, A.L. The Determination of Forty Two Elements in Geological Materials by Inductively Coupled Plasma-Mass Spectrometry; US Department of the Interior, US Geological Survey: Denver, CO, USA, 1999.
- Katznelson, H.; Peterson, E.A.; Rouatt, J.W. Phosphate-dissolving microorganisms on seed and in the root zone of plants. Can. J. Bot. 1962, 40, 1181–1186. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Hwangbo, H.; Park, R.D.; Kim, Y.W.; Rim, Y.S.; Park, K.H.; Kim, T.H.; Suh, J.S.; Kim, K.Y. 2-Ketogluconic acid production and phosphate solubilization by Enterobacter intermedium. Curr. Microbiol. 2003, 47, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Švitel, J.; Šturdik, E. 2-Ketogluconic acid production by Acetobacter pasteurianus. Appl. Biochem. Biotechnol. 1995, 53, 53–63. [Google Scholar] [CrossRef]
- Dawson, R.M.C. Data for Biochemical Research; Clarendon Press: Oxford, UK, 1959. [Google Scholar]
- Qin, F.; Shan, X.-Q.; Wei, B. Effects of low-molecular-weight organic acids and residence time on desorption of Cu, Cd, and Pb from soils. Chemosphere 2004, 57, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Debela, F.; Arocena, J.M.; Thring, R.W.; Whitcombe, T. Organic acid-induced release of lead from pyromorphite and its relevance to reclamation of Pb-contaminated soils. Chemosphere 2010, 80, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Bolan, N.S.; Naidu, R.; Mahimairaja, S.; Baskaran, S. Influence of low-molecular-weight organic acids on the solubilization of phosphates. Biol. Fertil. Soils 1994, 18, 311–319. [Google Scholar] [CrossRef]
- Pine, L.; George, J.R.; Reeves, M.W.; Harrell, W.K. Development of a chemically defined liquid medium for growth of Legionella pneumophila. J. Clin. Microbiol. 1979, 9, 615–626. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, D.; Kim, J.; Kim, B.-s.; Jeong, J.; Lee, J.-c. Use of Phosphate Solubilizing Bacteria to Leach Rare Earth Elements from Monazite-Bearing Ore. Minerals 2015, 5, 189-202. https://doi.org/10.3390/min5020189
Shin D, Kim J, Kim B-s, Jeong J, Lee J-c. Use of Phosphate Solubilizing Bacteria to Leach Rare Earth Elements from Monazite-Bearing Ore. Minerals. 2015; 5(2):189-202. https://doi.org/10.3390/min5020189
Chicago/Turabian StyleShin, Doyun, Jiwoong Kim, Byung-su Kim, Jinki Jeong, and Jae-chun Lee. 2015. "Use of Phosphate Solubilizing Bacteria to Leach Rare Earth Elements from Monazite-Bearing Ore" Minerals 5, no. 2: 189-202. https://doi.org/10.3390/min5020189
APA StyleShin, D., Kim, J., Kim, B.-s., Jeong, J., & Lee, J.-c. (2015). Use of Phosphate Solubilizing Bacteria to Leach Rare Earth Elements from Monazite-Bearing Ore. Minerals, 5(2), 189-202. https://doi.org/10.3390/min5020189







