Characterization of Green Liquor Dregs, Potentially Useful for Prevention of the Formation of Acid Rock Drainage
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Mineralogical Characterization
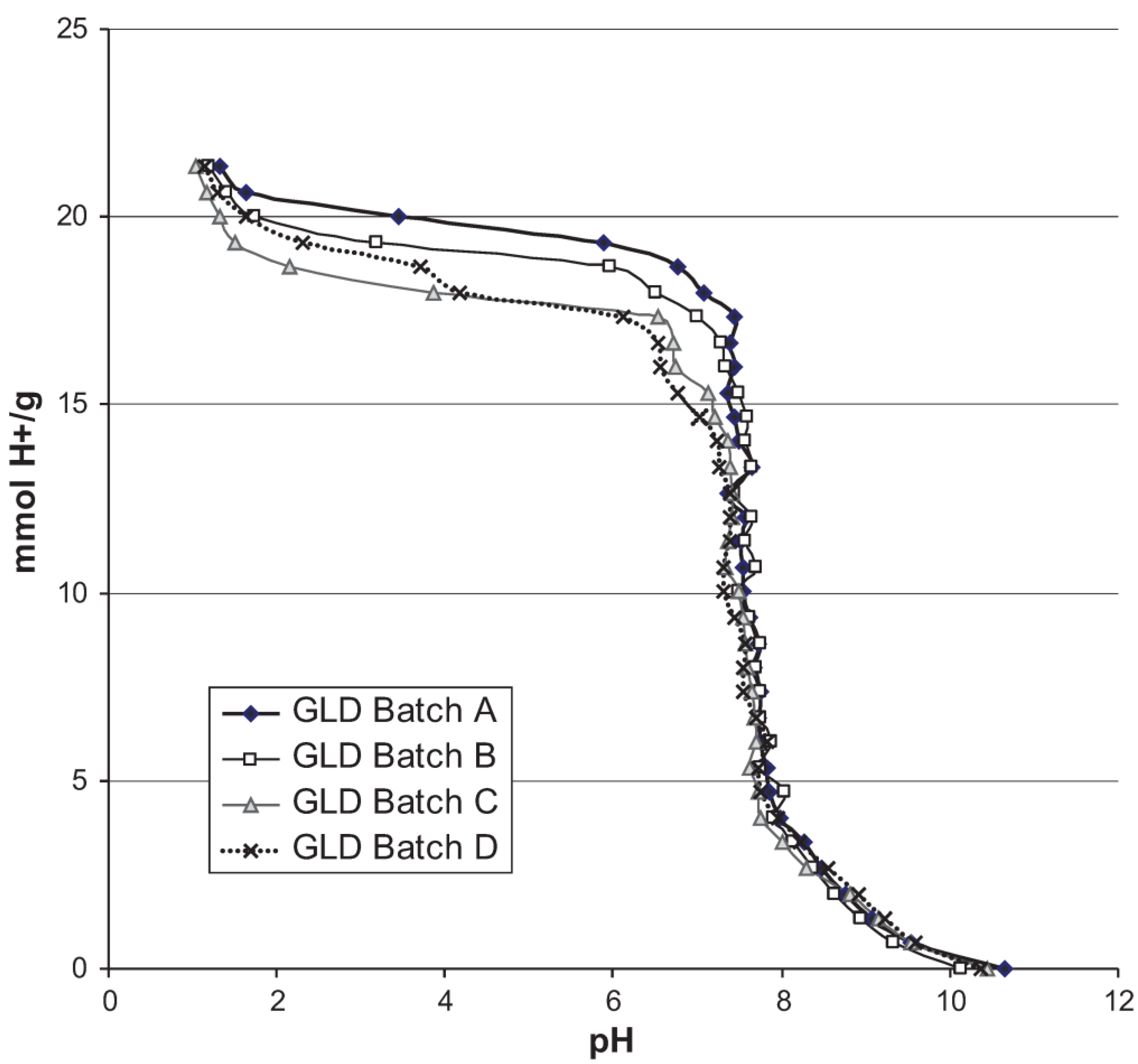
2.2.2. Chemical Characterization
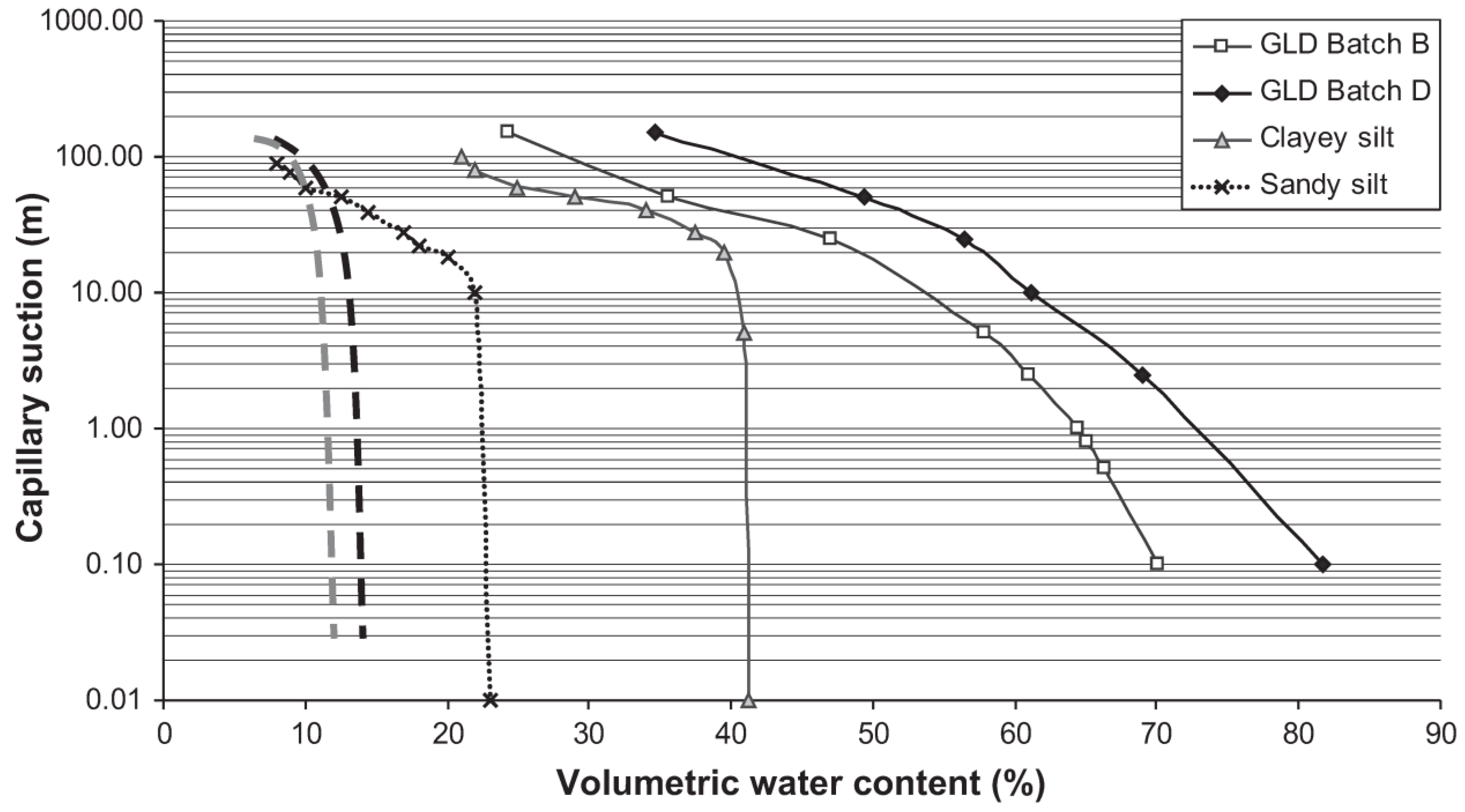
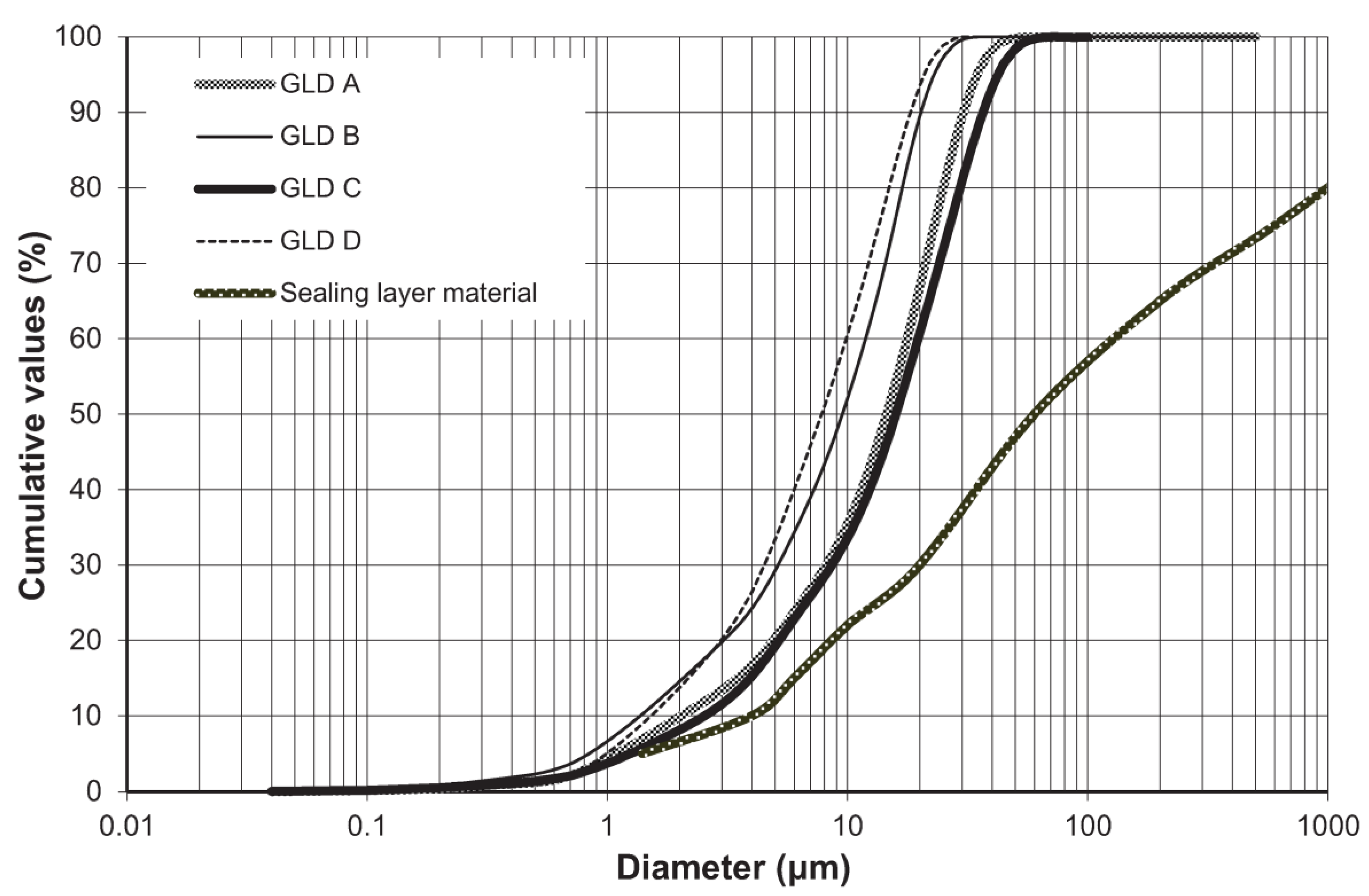
2.2.3. Physical Characterization
3. Results
| Element | Unit | Sample batch | |||||||
|---|---|---|---|---|---|---|---|---|---|
| A1 | A2 | B1 | B2 | C1 | C2 | D1 | D2 | ||
| Dry matter (DM) | % | 51.4 | 51.6 | 47.8 | 47.2 | 37.9 | 38.0 | 35.7 | 35.5 |
| Si | mg/kg | 4,960 | 5,020 | 6,630 | 6,850 | 9,720 | 10,500 | 12,300 | 12,400 |
| Al | mg/kg | 2,700 | 2,720 | 2,900 | 3,090 | 5,280 | 5,180 | 9,820 | 9,890 |
| Ca | mg/kg | 317,000 | 317,000 | 295,000 | 296,500 | 252,000 | 247,000 | 219,000 | 217,000 |
| Fe | mg/kg | 3,190 | 3,170 | 4,540 | 3,360 | 5,700 | 6,520 | 9,650 | 7,260 |
| K | mg/kg | 1,910 | 2,050 | 2,910 | 2,970 | 3,010 | 3,060 | 3,360 | 3,570 |
| Mg | mg/kg | 37,800 | 38,300 | 43,400 | 43,900 | 50,600 | 49,600 | 72,400 | 73,300 |
| Mn | mg/kg | 8,350 | 8,430 | 10,800 | 10,900 | 12,900 | 12,700 | 17,300 | 17,500 |
| P | mg/kg | 4,000 | 4,030 | 3,590 | 3,610 | 1,270 | 1,250 | 922 | 911 |
| LOI 1000 °C | % DM | 41.3 | 41.4 | 40.7 | 40.8 | 43.1 | 43.1 | 39.9 | 40.0 |
| As | mg/kg | 0.305 | 0.225 | 0.327 | 0.172 | 0.332 | 0.383 | 0.474 | 0.431 |
| Cd | mg/kg | 3.54 | 3.69 | 5.80 | 5.53 | 4.98 | 4.87 | 8.29 | 8.65 |
| Co | mg/kg | 3.34 | 3.04 | 3.96 | 3.67 | 4.83 | 4.72 | 5.63 | 5.77 |
| Cr | mg/kg | 71.9 | 80.2 | 93.2 | 94.3 | 92.0 | 97.7 | 126 | 127 |
| Cu | mg/kg | 101 | 109 | 112 | 108 | 179 | 159 | 224 | 202 |
| Hg | mg/kg | 0.04 | 0.04 | <0.04 | <0.04 | <0.04 | <0.04 | <0.04 | <0.04 |
| Ni | mg/kg | 29.3 | 31.6 | 53.6 | 46.7 | 51.7 | 40.7 | 60.8 | 49.5 |
| Pb | mg/kg | 2.27 | 2.56 | 2.86 | 2.96 | 3.28 | 3.26 | 5.88 | 6.13 |
| S | mg/kg | 7,880 | 8,780 | 10,500 | 9,150 | 13,500 | 13,200 | 18,600 | 18,700 |
| Zn | mg/kg | 701 | 764 | 1,120 | 1,060 | 1,230 | 1,200 | 1,840 | 1,800 |
| CaO | % DM | 44.3 | 44.3 | 41.0 | 41.0 | 35.3 | 34.6 | 30.6 | 30.4 |
| Element | GLD L/S 10 (mg/kg) | Leaching limit values at landfills for inert waste (mg/kg) | Leaching limit values at landfills for non-hazardous waste (mg/kg) | Relative mass released from GLD (%) |
|---|---|---|---|---|
| Si | 3.03 ± 0.00 | N/A | N/A | 0.01 |
| Al | 0.34 ± 0.19 | N/A | N/A | 0.01 |
| Ca | 29.4 ± 18.0 | N/A | N/A | 0.01 |
| Fe | 0.23 ± 0.00 | N/A | N/A | 0.01 |
| K | 1433 ± 12 | N/A | N/A | 97.0 |
| Mg | 8.3 ± 3.0 | N/A | N/A | 0.09 |
| Mn | 0.97 ± 0.76 | N/A | N/A | 0.01 |
| P | 3.07 ± 0.37 | N/A | N/A | 0.08 |
| As | 0.002 ± 0.002 | 0.5 | 2 | 0.32 |
| Cd | 0.0003 ± 0.0000 | 0.04 | 1 | 0.01 |
| Co | 0.0006± 0.0000 | N/A | N/A | 0.01 |
| Cr | 0.44 ± 0.07 | 0.5 | 10 | 0.7 |
| Cu | 0.045 ± 0.015 | 2 | 50 | 0.04 |
| Hg | 0.22 ± 0.00 | 0.01 | 0.2 | 0.08 |
| Ni | 7.01 ± 3.00 | 0.4 | 10 | 0.02 |
| Pb | 1.89 ± 0.00 | 0.5 | 10 | 0.06 |
| S | 7,990 ± 104 | Sulphate: 1,000 | Sulphate: 20,000 | 85.6 |
| Zn | 0.088 ± 0.051 | 4 | 10 | 0.01 |
| GLD batch | Surface area (m2/g) | pH | Hydraulic conductivity (m/s) | Bulk density (g/cm3) | Compact density (calculated) (g/cm3) | Compact density (by pycnometer) (g/cm3) | Porosity (%) |
|---|---|---|---|---|---|---|---|
| A | 12.1 ± 0.1 | 11.0 ± 0.1 | N/A | N/A | N/A | N/A | N/A |
| B | 16.8 ± 0.1 | 10.0 ± 0.0 | 1 × 10−8 ± 7 × 10−9 | 0.67 ± 0.00 | 2.60 ± 0.00 | 2.57 ± 0.00 | 73.2 ± 0.4 |
| C | 21.4 ± 0.0 | 10.6 ± 0.1 | N/A | N/A | N/A | N/A | N/A |
| D | 20.4 ± 0.2 | 10.1 ± 0.0 | 1 × 10−8 ± 7 × 10−9 | 0.44 ± 0.00 | 2.47 ± 0.00 | N/A | 82.2 ± 0.1 |
4. Discussion
4.1. Material Variation
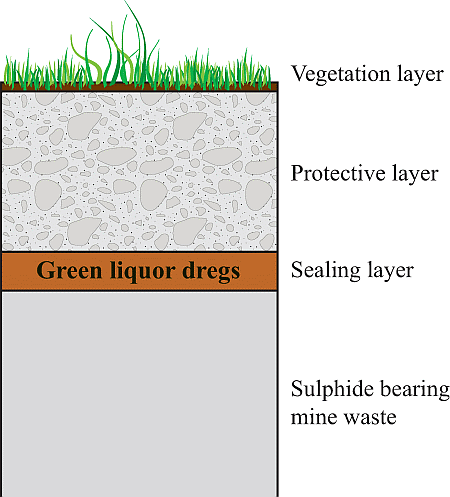
4.2. Use of GLD in Sealing Layers
4.3. Limitations
5. Conclusions
Acknowledgments
Authors Contributions
Conflicts of Interest
References and Notes
- Höglund, L.-O.; Herbert, R.; Lövgren, L.; Öhlander, B.; Neretniks, I.; Moreno, L.; Malmström, M.; Elander, P.; Lindvall, M.; Lindström, B. MiMi—Performance Assessment, Main Report; MiMi-Report 2003:3; MiMi Print: Stockholm, Sweden, 2004. [Google Scholar]
- International Network for Acid Drainage (INAP). The Global Acid Rock Drainage Guide (GARD Guide). Available online: http://www.gardguide.com (accessed on 12 December 2013).
- Carlsson, E. Sulphide-Rich Tailings Remediated by Soil Cover: Evaluation of Cover Efficiency and Tailings Geochemistry, Kristineberg, Northern Sweden. Ph.D. Thesis, Department of Environmental Engineering, Luleå University of Technology, 2002; p. 44. [Google Scholar]
- Hallberg, R.O.; Granhagen, A.; Liljemark, A. A fly ash/biosludge dry cover for the mitigation of AMD at the Falun mine. Chem. Erde Geochem. 2005, 65, 43–63. [Google Scholar] [CrossRef]
- Cabral, A.; Racine, I.; Burnotte, F.; Lefebvre, G. Diffusion of oxygen through a pulp and paper residue barrier. Can. Geotech. J. 2000, 201–207. [Google Scholar]
- Catalan, L.J.J.; Kumari, A. Efficency of lime mud residues from kraft mills to amend oxidized mine tailings before permanent flooding. J. Environ. Eng. Sci. 2005, 4, 241–256. [Google Scholar] [CrossRef]
- Bäckström, M.; Karlsson, S.; Sartz, L. Utvärdering och Demonstration av Efterbehandlingsalternativ för Historiskt Gruvavfall Med Aska och Alkaliska Restprodukter; Report 1099; Värmeforsk Service AB: Stockholm, Sweden, 2009. (In Swedish) [Google Scholar]
- Cousins, C.; Penner, G.H.; Liu, B.; Beckett, G.; Spiers, G. Organic matter degradation in paper sludge amendments over gold mine tailings. Appl. Geochem. 2009, 24, 2293–2300. [Google Scholar] [CrossRef]
- Doye, I.; Duchesne, J. Neutralisation of acid mine drainage with alkaline industrial residues: Laboratory investigation using batch-leaching tests. Appl. Geochem. 2003, 18, 1197–1213. [Google Scholar] [CrossRef]
- Bellaloui, A.; Chtaini, A.; Ballivy, G.; Narasiah, S. Laboratory investigation of the control of acid mine drainage using alkaline paper mill waste. Water Air Soil Pollut. 1999, 111, 57–73. [Google Scholar] [CrossRef]
- Chtaini, A.; Bellaloui, A.; Ballivy, G.; Narasiah, S. Field investigation of controlling acid mine drainage using alkaline paper mill waste. Water Air Soil Pollut. 2001, 125, 357–374. [Google Scholar] [CrossRef]
- Sartz, L.; Bäckström, M.; Karlsson, S.; Allard, B. Stabilization of Acid-Generating Waste Rock with Alkaline By-Products: Results from a Meso-Scale Experiment. In Proceedings of Securing the Future and 8th International Conference on Acid Rock Drainage (ICARD), Skellefteå, Sweden, 22–26 June 2009.
- Lundqvist, F.; Brelid, H.; Saltberg, A.; Gellerstedt, G.; Tomani, P. Removal of non-process elements from hardwood chips prior to kraft cooking. Appita 2006, 59, 493–499. [Google Scholar]
- SFS, 2001:1063, Swedish Ordinance of Waste; Sweden Ministry of Environment: Stockholm, Sweden, 2001. (In Swedish)
- Toikka, M. Minimization and Utilization of Green Liquor Dregs and Ashes in Pulp and Paper; Regional Environment Publication No. 82; Southeast Finland Regional Environment Centre, Ed.; Oy Edita Ab: Helsinki, Finland, 1998; pp. 1–82. [Google Scholar]
- Pousette, K.; Mácsik, J. Kartläggning av Restprodukter från Assi Domän AB: Research Report; Luleå University of Technology: Luleå, Sweden, 2000. (In Swedish) [Google Scholar]
- Hargelius, K. Pilotyta med Tätskikt på Ätrans Deponi, Fältförsök-Värö-FAVRAB-Hylte; Ramböll Sverige AB, Region Väst: Gothenburg, Sweden, 2008. (In Swedish) [Google Scholar]
- U.S. Environmental Protection Agency (EPA). Methods for the Determination of Metals in Environmental Samples; EPA/600/4-91/010; Office of Research and Development, U.S. Environmental Protection Agency: Washington, DC, USA, 1991.
- Swedish Standards Institute (SIS). Determination of Dry Matter and Ignition Residue in Water, Sludge and Sediment; SS 028113; SIS Förlag AB: Stockholm, Sweden, 1981. (In Swedish) [Google Scholar]
- Wyatt, P.H. A Technique for Determining the Acid Neutralizing Capacity of Till and Other Surficial Sediments; Paper No. 84-1A; Geological Survey of Canada: Ottawa, ON, Canada, 1984; pp. 597–600. [Google Scholar]
- Swedish Standards Institute (SIS). Characterisation of Waste, Leaching, Compliance Test for Leaching of Granular Waste Materials and Sludges; SS-EN 12457-4; SIS Förlag AB: Stockholm, Sweden, 2003. (In Swedish) [Google Scholar]
- Jia, Y.; Maurice, C.; Öhlander, B. Effect of the alkaline industrial residues fly ash, green liquor dregs, and lime mud on mine tailings oxidation when used as covering material. Environ. Earth Sci. 2013. [Google Scholar] [CrossRef]
- Weber, P.A.; Hughes, J.B.; Conner, L.B.; Lindsay, P.; Smart, R. Short-Term Acid Rock Drainage Characteristics Determined by Paste pH and Kinetic NAG Testing: Cypress, Prospect, New Zealand. In Proceedings of 7th International Conference on Acid Rock Drainage (ICARD), St. Louis, MO, USA, 26–30 March 2006.
- Swedish Standards Institute (SIS). Geotechnical Tests, Compression Properties, Oedometer Test, CRS-Test, Cohesive Soil; SS 02 71 26; SIS förlag AB: Stockholm, Sweden, 1991. (In Swedish) [Google Scholar]
- European Council. Council decision of 19 December 2002 establishing criteria and procedures for the acceptance of waste at landfills pursuant to Article 16 of and Annex II to Directive 1999/31/EC. Off. J. Eur. Commun. 2002, L11, 27–49. [Google Scholar]
- Swedish Geotechnical Institute (SIG). Deponiers Stabilitet, Vägledning för Beräkning; SGI Information 19; Swedish Geotechnical Institute: Linköping, Sweden, 2007. (In Swedish) [Google Scholar]
- Bussière, B.; Chapuis, R.P.; Aubertin, M. Unsaturated Flow Modeling for Exposed and Covered Tailings Dams. In Proceedings of the International Symposium on Major Challenges in Tailings Dams, Montreal, QC, Canada, 15 June 2003.
- Martins, F.M.; Martins, J.M.; Ferracin, L.C.; da Cunha, C.J. Mineral phases of green liquor dregs, slaker grits, lime mud and wood ash of a Kraft pulp and paper mill. J. Hazard. Mater. 2007, 147, 610–617. [Google Scholar] [CrossRef]
- Mbonimpa, M.; Aubertin, M.; Aachib, M.; Bussière, B. Diffusion and consumption of oxygen in unsaturated cover materials. Can. Geotech. J. 2003, 40, 916–932. [Google Scholar] [CrossRef]
- Erickson, J.; Tyler, E.J. Soil Oxygen Delivery to Wastewater Infiltration Surfaces. In Proceedings of 2000 National Onsite Wastewater Recycle Association (NOWRA), Laurel, MD, USA, 5 August 2000.
- Sivapullaiah, P.V.; Sridharan, A.; Stalin, V.K. Hydraulic conductivity of bentonite-sand mixtures. Can. Geotech. J. 2000, 37, 406–413. [Google Scholar] [CrossRef]
- Benson, C.H.; Trast, J.M. Hydraulic conductivity of thirteen compacted clays. Clays Clay Miner. 1995, 43, 669–681. [Google Scholar]
- Marion, D.; Nur, A.; Yin, H.; Han, D. Compressional velocity and porosity in sand-clay mixtures. Geophysics 1992, 57, 554–563. [Google Scholar] [CrossRef]
- Koltermann, C.E.; Gorelick, S.M. Fractional packing model for hydraulic conductivity derived from sediment mixtures. Water Resour. Res. 1995, 31, 3283–3297. [Google Scholar] [CrossRef]
- Jia, Y.; Luleå University of Technology, Luleå, Sweden. Unpublished Modeling Data. 2014.
- Kosmulski, M.; Dahlsten, P. High ionic strength electrokinetics of clay minerals. Colloids Surf. Physicochem. Eng. Aspects 2006, 291, 212–218. [Google Scholar] [CrossRef]
- Guichet, X.; Jouniaux, L.; Catel, N. Modification of streaming potential by precipitation of calcite in a sand-water system: Laboratory measurements in the pH range from 4 to 12. Geophys. J. Int. 2006, 166, 445–460. [Google Scholar] [CrossRef]
- Hunter, R.J. Zeta Potential in Colloid Science; Academic Press: London, UK, 1981. [Google Scholar]
- Maurice, C.; Villain, L.; Hargelius, K. Green Liquor Dregs for the Remediation of Abandoned Mine Tailings—Opportunities and Limitations. In Proceedings of Securing the Future and 8th International Conference on Acid Rock Drainage (ICARD), Skellefteå, Sweden, 22–26 June 2009.
- Naturvårdverket (Swedish Environmental Protection Agency). Avfall i Sverige 2010; Report 6520; CM Gruppen AB: Bromma, Sweden, 2012. (In Swedish)
- Jia, Y.; Stenman, D.; Mäkitalo, M.; Maurice, C.; Öhlander, B. Use of amended tailings as mine waste cover. Waste Biomass Valoriz. 2013, 4, 709–718. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mäkitalo, M.; Maurice, C.; Jia, Y.; Öhlander, B. Characterization of Green Liquor Dregs, Potentially Useful for Prevention of the Formation of Acid Rock Drainage. Minerals 2014, 4, 330-344. https://doi.org/10.3390/min4020330
Mäkitalo M, Maurice C, Jia Y, Öhlander B. Characterization of Green Liquor Dregs, Potentially Useful for Prevention of the Formation of Acid Rock Drainage. Minerals. 2014; 4(2):330-344. https://doi.org/10.3390/min4020330
Chicago/Turabian StyleMäkitalo, Maria, Christian Maurice, Yu Jia, and Björn Öhlander. 2014. "Characterization of Green Liquor Dregs, Potentially Useful for Prevention of the Formation of Acid Rock Drainage" Minerals 4, no. 2: 330-344. https://doi.org/10.3390/min4020330
APA StyleMäkitalo, M., Maurice, C., Jia, Y., & Öhlander, B. (2014). Characterization of Green Liquor Dregs, Potentially Useful for Prevention of the Formation of Acid Rock Drainage. Minerals, 4(2), 330-344. https://doi.org/10.3390/min4020330