Enhancing Fines Recovery by Hybrid Flotation Column and Mixed Collectors
Abstract
1. Introduction
2. Materials and Methods
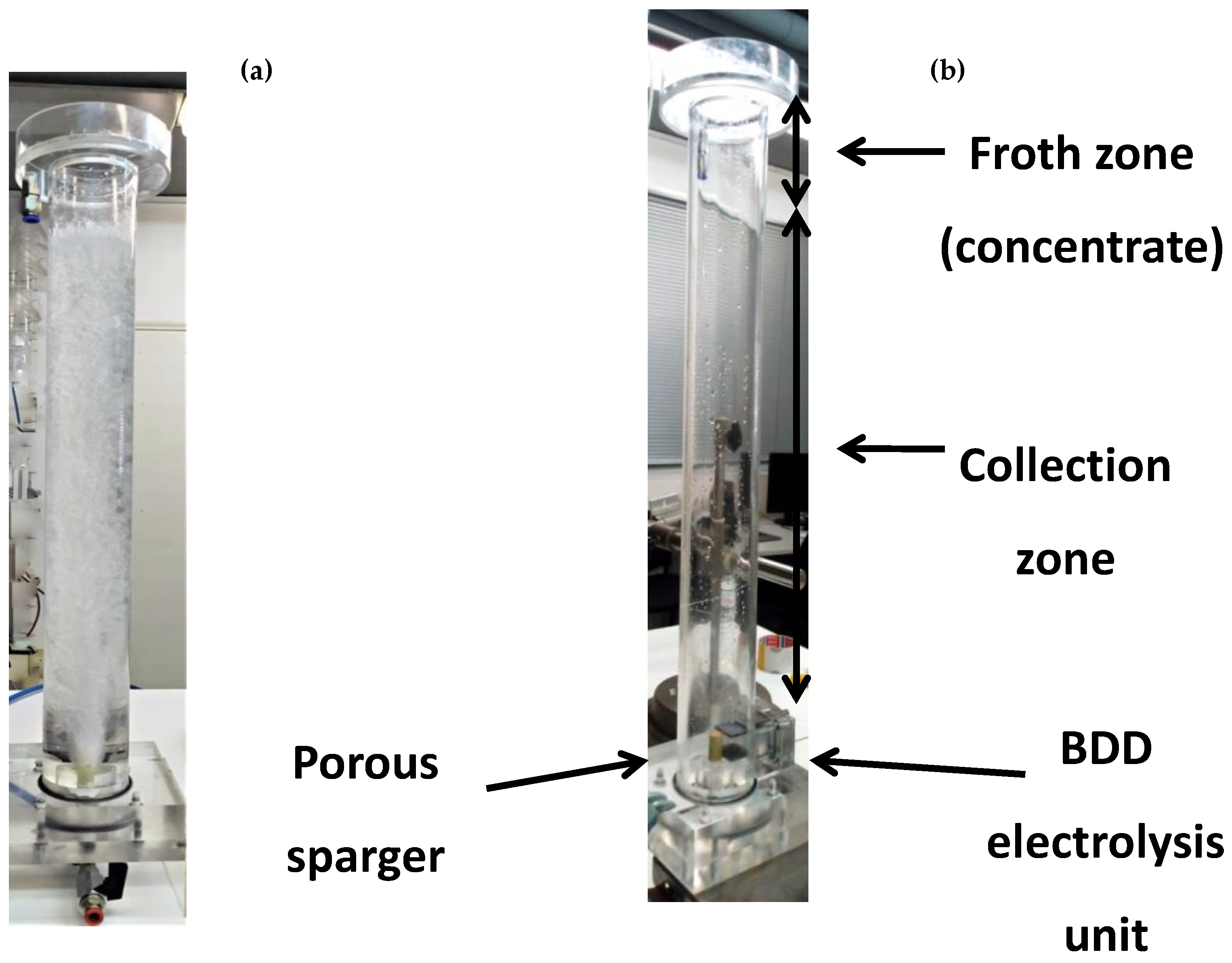
2.1. Experimental Set up-The Hybrid Flotation Column
2.2. Flotation Experiments
3. Results
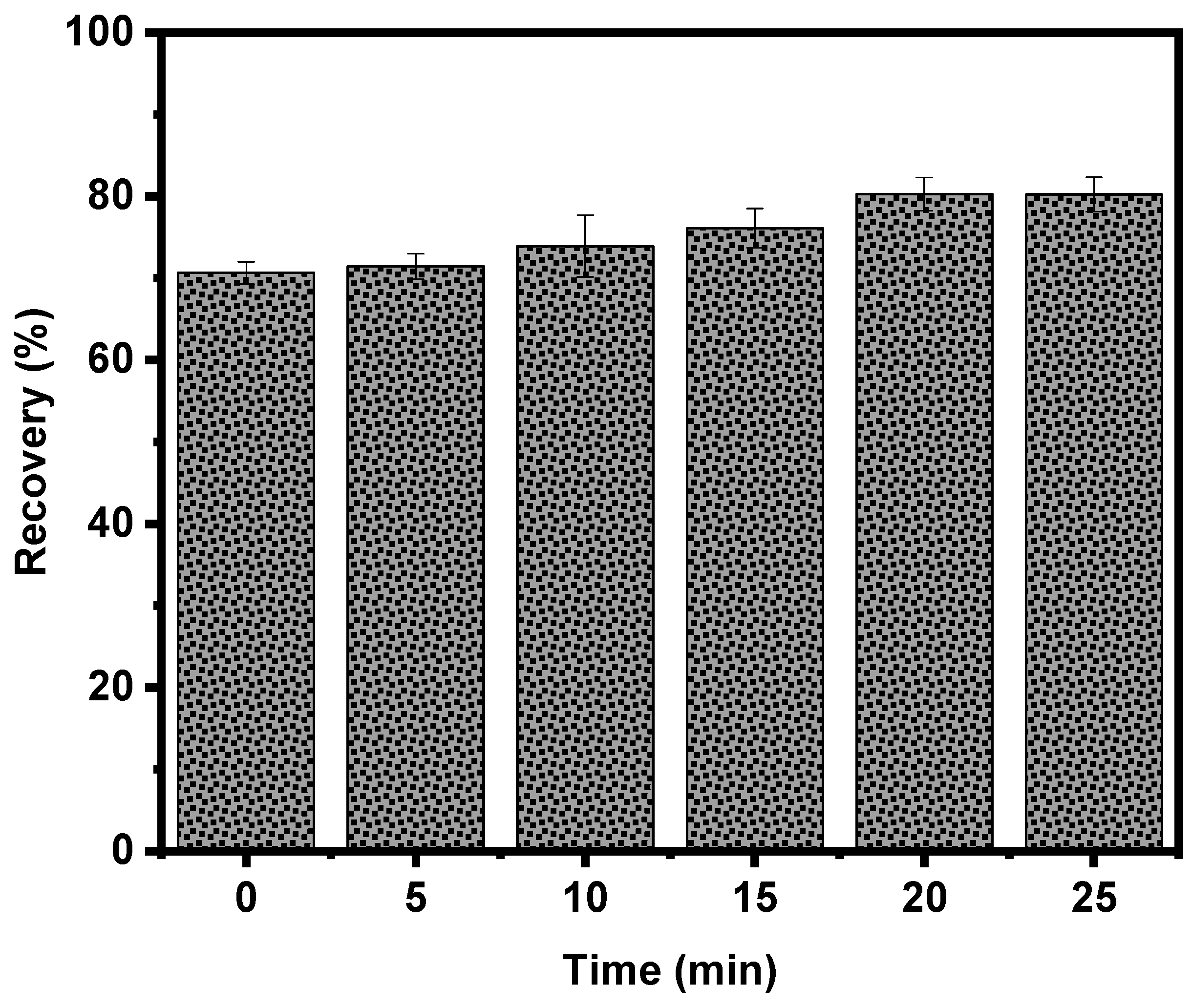
3.1. Effect of Conditioning Time with Electrolytic Bubbles
3.2. Effect of pH and Collector Concentration on Combined Magnesite Flotation
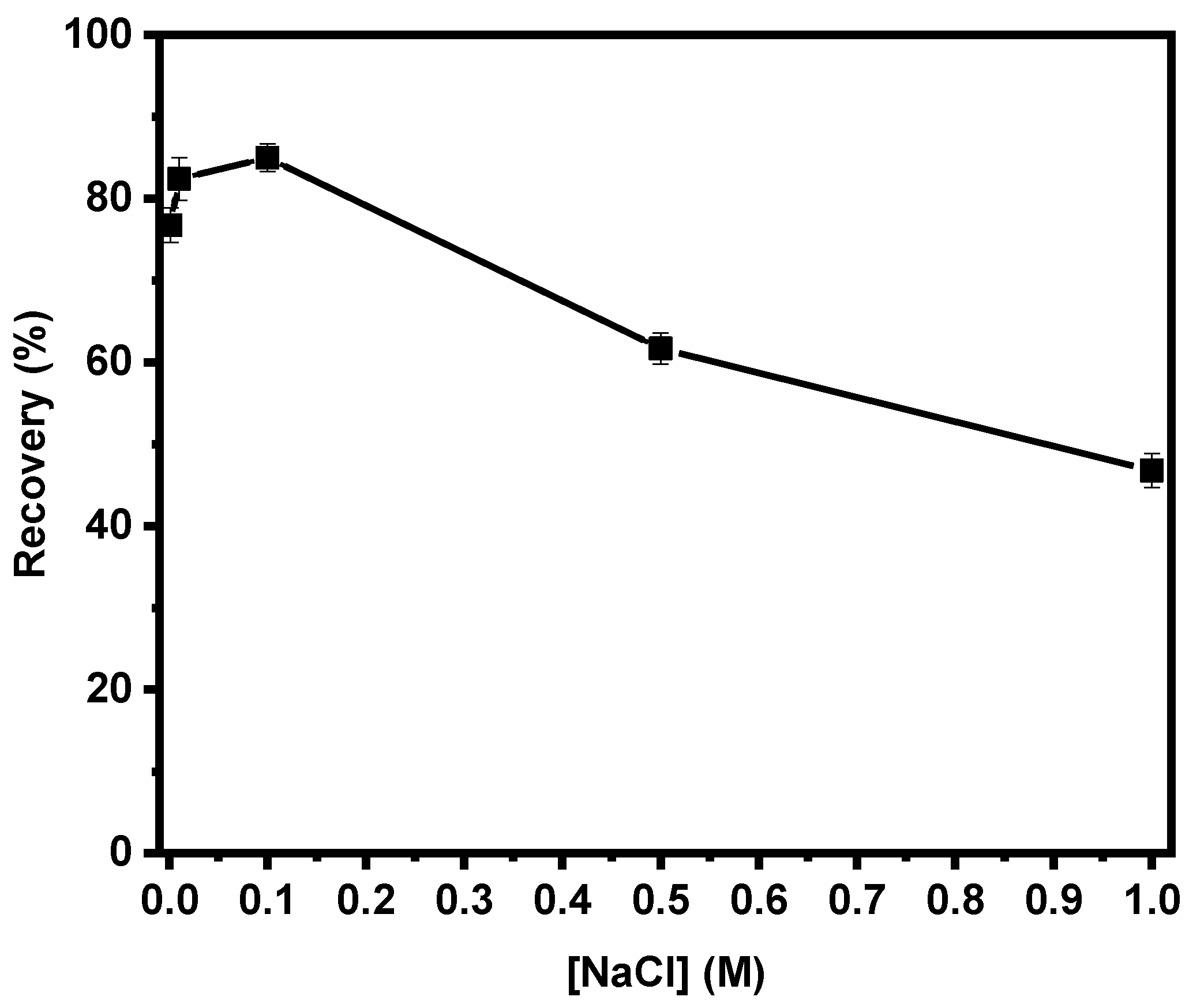
3.3. Effect of Electrolyte Concentration on Combined Flotation
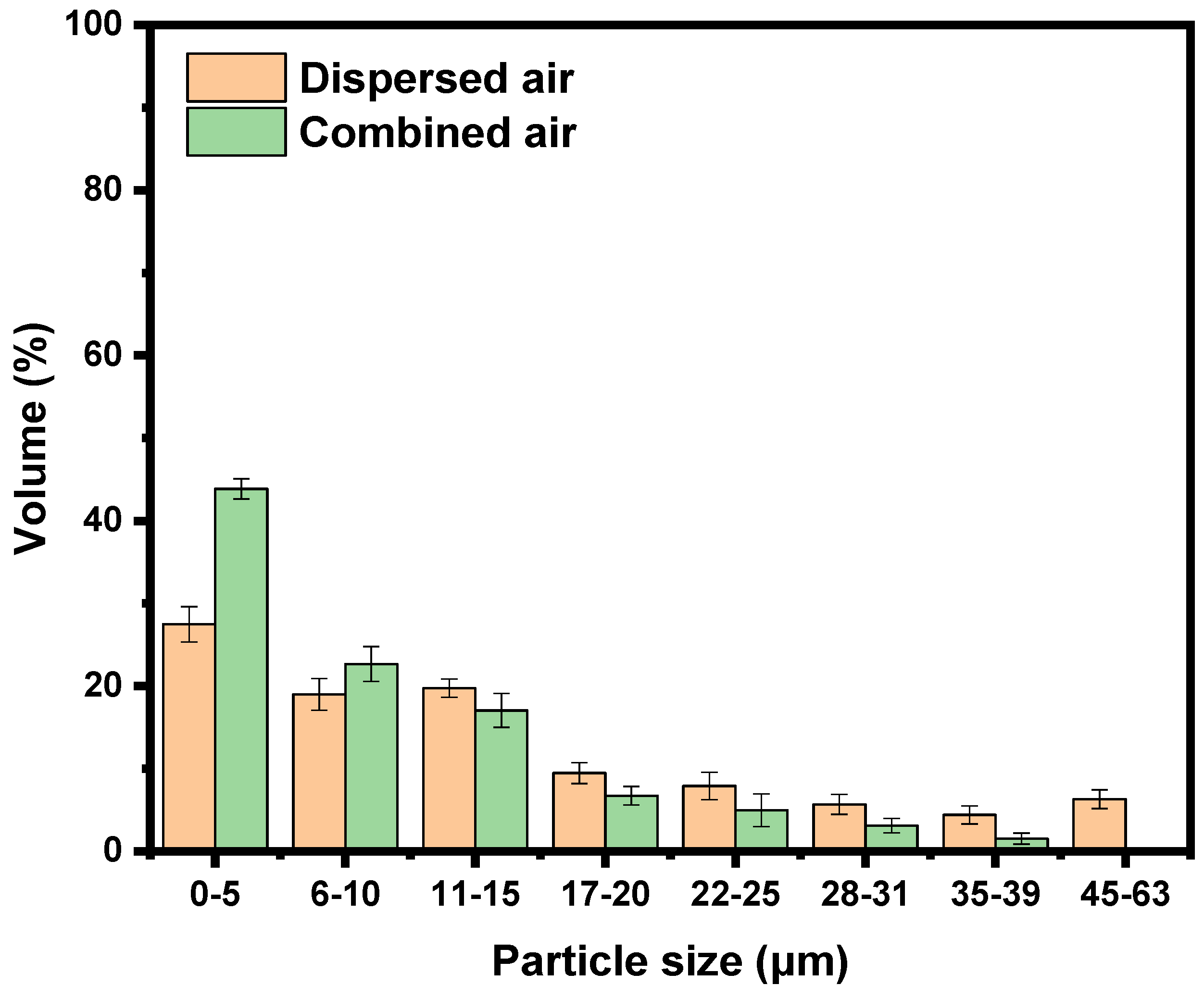
3.4. Particle Size Distribution of Froth Products
3.5. Flotation Mechanism
3.6. Flotation Kinetics Study
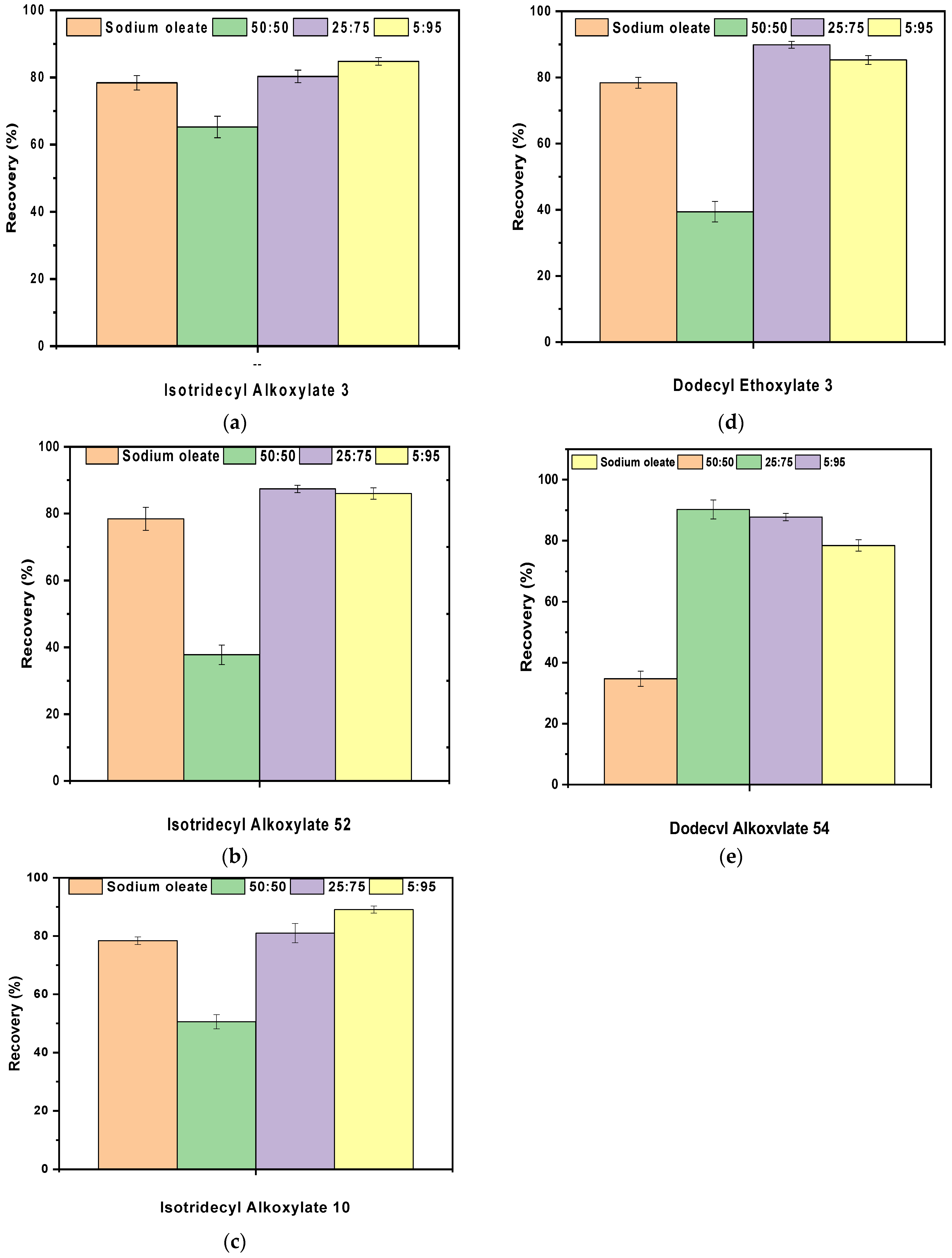
3.7. Synergistic Effect of Anionic/Non Ionic Collectors on Fine Magnesite Flotation
4. Conclusions
- The experimental results can be concluded as:
- The maximum treatment time of magnesite fine particles with electrolytic microbubbles with the optimal recovery was 20 min.
- Flotation experiments realized on the hybrid column with combined air showed an increase of about 8% in fines recovery. In addition, the particle size distribution of the recovered mineral showed an increase of about 37.4% in 0–5 μm particles compared to experiments conducted with dispersed air bubbles exclusively. To this end, it is distinct that the use of combined air favors the recovery of ultra-fine particles.
- The experimental data deriving from the kinetic study revealed that the experimental data follow the first-order model and furthermore that the particles of smaller particle size (−25 μm) are recovered faster than the other two fractions (−45 and −100 μm).The hybrid column kinetic study showed a 5% increase in the flotation rate of magnesite fines.
- The maximum ratios between sodium oleate and the non-ionic collectors were 5:95 and 25:75. In addition, maximum recovery was achieved when Dodecyl Alkoxylate 54 was utilized as the co-collector in a ratio of 25:75 increasing magnesite fines recovery by almost 12%.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Glembotskii, V.A.; Klassen, V.I.; Plaksin, I.N. Flotation; Primary Sources: New York, NY, USA, 1972; p. 633. [Google Scholar]
- Pyke, B.; Fornasiero, D.; Ralston, J. Bubble particle heterocoagulation under turbulent conditions. J. Colloid. Interface Sci. 2003, 265, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Beloborodov, V.; Fedotov, K. Ways to increase the efficiency of flotation process for complex ores. Oral Session. In Proceedings of the XXI International Mineral Processing Congress, July 23-27, Rome, Italy, 23–27 July 2000; pp. C8b–84–C8b–91. [Google Scholar] [CrossRef]
- Gaudin, A.M.; Schuhmann, J.R.; Schlechten, A.W. Flotation Kinetics. II. The Effect of Size on the Behavior of Galena Particles. J. Phys. Chem. 1942, 46, 902–910. [Google Scholar] [CrossRef]
- Li, H. The Roles of Non-Polar Oil in Froth Flotation of Fine Particles. Ph.D. Thesis, University of Alberta, Edmonton, AB, Canada, 2018. [Google Scholar] [CrossRef]
- Pease, J.D.; Curry, D.C.; Young, M.F. Designing Flotation Circuits for High Fines Recovery; Jameson, G.J., Ed.; Centenary of Flotation Symposium, The Australian Institute of Mining and Metallurgy: Carlton, Australian, 2005; pp. 905–912. [Google Scholar] [CrossRef]
- Evans, G.M.; Atkinson, B.W.; Jameson, G.J. The Jameson Cell. In Flotation Science and Engineering; Matis, Ed.; Marcel Dekker: New York, NY, USA, 1995. [Google Scholar]
- Mohanty, M.K.; Honaker, R.Q. Performance optimization of Jameson flotation technology for fine coal cleaning. Miner. Eng. 1999, 12, 367–381. [Google Scholar] [CrossRef]
- Cowburn, J.; Harbort, G.; Manlapig, E.; Pokrajcic, Z. Improving the recovery of coarse coal particles in a Jameson cell. Miner. Eng. 2006, 19, 609–618. [Google Scholar] [CrossRef]
- Jameson, G.J. New Directions in flotation machine design. Miner. Eng. 2010, 23, 835–841. [Google Scholar] [CrossRef]
- Trahar, W.J.; Warren, L.J. The flotability of very fine particles—a review. Int. J. Miner. Process. 1976, 3, 103–131. [Google Scholar] [CrossRef]
- Fuerstenau, D.W. Fine particle flotation. In Fine Particle Processing, Proceedings International Symposium, Las Vegas, Nevada, 24–28 February 1980; Somasundaran, P., Ed.; American Institute of Mining, Metallurgical, and Petroleum Engineers: Wilkes-Barre, PA, USA, 1980. [Google Scholar]
- Yoon, R.H.; Luttrell, G.H. The effect of bubble size on fine particle flotation. Miner. Process. Extr. Metall. 1989, 5, 101–122. [Google Scholar] [CrossRef]
- Dai, Z.; Fornasiero, D.; Ralston, J. Particle–bubble collision models—A review. Adv. Colloid Interface Sci. 2000, 85, 231–256. [Google Scholar] [CrossRef]
- Read, A.D.; Hollick, C.T. Selective flocculation techniques for recovery of fine particles. Min. Sci. Eng. 1976, 8, 202–213. [Google Scholar]
- Rubio, J.; Capponi, F.; Matiolo, E.; Nunes, G.N. Advances in flotation of mineral fines. Proceeding of the XXII International Mineral Processing Congress, Cape Town, South Africa, 29 September–3 October 2003. [Google Scholar]
- Somasundaran, P. Selective Flocculation of Fines. The Physical Chemistry of Mineral—Reagent Interactions in Sulfide Flotation; U.S. Bureau of Mines: Washington, DC, USA, 1978. [Google Scholar]
- Fornasiero, D.; Filippov, L. Innovations in the flotation of fine and coarse particles. J. Phys. Conf. Ser. 2017, 879, 012002. [Google Scholar] [CrossRef]
- Rulyov, N.N. Turbulent microflotation of fine disperse minerals (The general concept). In Proceedings of the Strategic Conference and Workshop: Flotation & Flocculation: From Fundamentals to Applications, Kailua-Kona, Hawai, 28 July–2 August 2003. [Google Scholar]
- Ahmed, N.; Jamesson, G.J. The effect of bubble size on the rate of flotation of fine particles. Int. J. Miner. Process. 1985, 14, 195–215. [Google Scholar] [CrossRef]
- Lu, S.; Pugh, R.; Forssberg, E. Surface Properties of Particles. In Interfacial Separation of Particles, 20th ed.; Elsevier Science: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Zhou, Z.A.; Li, H.; Chow, R.S.; Roberge, K. Role of carrier flotation in accelerating bitumen extraction recovery from mineable Athabasca oil sands. Can. J. Chem. Eng. 2013, 91, 1340–1348. [Google Scholar] [CrossRef]
- Yang, B.; Song, S. Hydrophobic agglomeration of mineral fines in aqueous suspensions and its application in flotation: A review. Surf. Rev. Lett. 2014, 21, 1430003. [Google Scholar] [CrossRef]
- Kohmuench, J.N.; Luttrell, G.; Mankosa, M. Coarse particle concentration using the HydroFloat Separator. Miner. Metall. Process 2000, 18, 61–67. [Google Scholar] [CrossRef]
- Mankosa, M.J.; Kohmuench, J.N.; Christodoulou, L.; Yan, E.S. Improving fine particle flotation using the Stackcell™ (raising the tail of the elephant curve). Miner. Eng. 2018, 121, 83–89. [Google Scholar] [CrossRef]
- Koh, P.T.L.; Schwarz, M.P. CFD modelling of bubble–particle collision rates and efficiencies in a flotation cell. Miner. Eng. 2003, 16, 1055–1059. [Google Scholar] [CrossRef]
- Hoseinian, F.S.; Rezai, B.; Kowsari, E.; Safari, M. Effect of impeller speed on the Ni(II) ion flotation. Geosyst. Eng. 2019, 22, 161–168. [Google Scholar] [CrossRef]
- Hoseinian, F.S.; Rezai, B.; Safari, M.; Deglon, D.A.; Kowsari, E. Effect of hydrodynamic parameters on nickel removal rate from wastewater by ion flotation. J. Environ. Manag. 2019, 244, 408–414. [Google Scholar] [CrossRef]
- Filippov, L.O.; Matinin, A.S.; Samiguin, V.D.; Filippova, I.V. Effect of ultrasound on flotation kinetics in the reactor-separator. J. Phys. Conf. Ser. 2013, 416, 012016. [Google Scholar] [CrossRef]
- Tsave, P.K.; Kostoglou, M.; Karapantsios, T.D.; Lazaridis, N.K. A Hybrid device for enhancing flotation of fine particles by combining micro-Bubbles with conventional bubbles. Minerals 2021, 11, 561. [Google Scholar] [CrossRef]
- Filippova, I.V.; Filippov, L.O.; Duverger, A.; Severov, V.V. Synergetic effect of a mixture of anionic and nonionic reagents: Ca mineral contrast separation by flotation at neutral pH. Miner. Eng. 2014, 66–68, 135–144. [Google Scholar] [CrossRef]
- Subrahmanyam, T.V.; Forssberg, K.S.E. Fine particles processing: Shear-flocculation and carrier flotation—A review. Int. J. Miner. Process. 1990, 30, 265–286. [Google Scholar] [CrossRef]
- Cho, Y.S.; Laskowski, J.S. Bubble coalescence and its effect on dynamic foam stability. Can. J. Chem. Eng. 2002, 80, 299–305. [Google Scholar] [CrossRef]
- Rulyov, N.N. Combined microflotation of fine minerals: Theory and experiment. mineral processing and extractive metallurgy. Trans. Inst. Min. Metall. C 2016, 125, 81–85. [Google Scholar] [CrossRef]
- Rulyov, N.N.; Tussupbayev, N.K.; Kravtchenco, O.V. Combined microflotation of fine quartz. Mineral processing and extractive metallurgy. Trans. Inst. Min. Metall. C 2015, 124, 217–233. [Google Scholar] [CrossRef]
- Rulyov, N.N.; Filippov, L.O.; Kravchenko, O.V. Combined microflotation of glass beads. Colloids Surf. A Physicochem. Eng. 2020, 598, 124810. [Google Scholar] [CrossRef]
- Rulyov, N.N.; Tussupbayev, N.K.; Turusbekov, D.K.; Semushkina, L.V.; Kaldybaeva, Z.A. Effect of Micro-bubbles as Flotation Carriers on Fine Sulphide Ore Beneficiation. Trans. Inst. Min. Metall. C 2018, 127, 133–139. [Google Scholar] [CrossRef]
- Sutherland, K.L. Physical Chemistry of Flotation. XI. Kinetics of the Flotation PROCESS. J. Chem. Phys. 1948, 52, 394–425. [Google Scholar] [CrossRef] [PubMed]
- Crawford, R.; Ralston, J. The influence of particle size and contact angle in mineral flotation. Int. J. Miner. Process. 1988, 23, 1–24. [Google Scholar] [CrossRef]
- Ralston, J.; Dukhin, S.S. The interaction between particles and bubbles. Colloids Surf. A Physicochem. Eng. Asp. 1999, 151, 3–14. [Google Scholar] [CrossRef]
- Duan, J.; Fornasiero, D.; Ralston, J. Calculation of the flotation rate constant of chalcopyrite particles in an ore. Int. J. Miner. Process. 2003, 72, 227–237. [Google Scholar] [CrossRef]
- Matis, K.A.; Gallios, G.P. Anionic flotation of magnesium carbonates by modifiers. Int. J. Miner. Process. 1989, 25, 261–274. [Google Scholar] [CrossRef]
- Brandão, P.R.G.; Poling, G.W. Anionic Flotation of Magnesite. Can. Metall. Q. 1982, 21, 211–220. [Google Scholar] [CrossRef]
- Fan, M.; Tao, D.; Honaker, R.; Luo, Z. Nanobubble generation and its application in froth flotation (Part III): Specially designed laboratory scale column flotation of phosphate. Min. Sci. Technol. 2010, 20, 317–338. [Google Scholar] [CrossRef]
- Liu, H.; Guofan, Z.; Luo, Y. Effect of depressants in the selective flotation of smithsonite and calcite using cationic collector. Physicochem. Probl. Miner. Process. 2020, 56, 1–10. [Google Scholar] [CrossRef]
- Yin, W.; Sun, H.; Tang, Y.; Hong, J.; Yang, B.; Fu, Y.; Han, H. Effect of pulp temperature on separation of magnesite from dolomite in sodium oleate flotation system. Physicochem. Probl. Miner. Process. 2019, 55, 1049–1058. [Google Scholar] [CrossRef]
- Bedekovic, G. A study of the effect of operating parameters in column flotation using experimental design. Physicochem. Probl. Miner. Process. 2016, 52, 523–535. [Google Scholar] [CrossRef]
- Dishon, M.; Zohar, O.; Sivan, U. From repulsion to attraction and back to repulsion: The effect of NaCl, KCl, and CsCl on the force between silica surfaces in aqueous solution. Langmuir 2009, 25, 2831–2836. [Google Scholar] [CrossRef]
- Marrucci, G.; Nicodemo, L. Coalescence of gas bubbles in aqueous solutions of inorganic electrolytes. Chem. Eng. Sci. 1967, 22, 1257–1265. [Google Scholar] [CrossRef]
- Uchida, T.; Liu, S.; Enari, M.; Oshita, S.; Yamazaki, K.; Gohara, K. Effect of NaCl on the lifetime of micro-and nanobubbles. Nanomaterials 2016, 6, 31. [Google Scholar] [CrossRef]
- Zieminski, S.A.; Whittemore, R.C. Behavior of gas bubbles in aqueous electrolyte solutions. Chem. Eng. Sci. 1971, 26, 509–520. [Google Scholar] [CrossRef]
- Montes-Atenas, G.; Garcia-Garcia, F.J.; Mermillod-Blondin, R.; Montes, S. Effect of suspension chemistry onto voltage drop: Application of electro-flotation. Powder Technol. 2010, 204, 1–10. [Google Scholar] [CrossRef]
- Huang, C.H.; Shen, S.Y.; Chen, C.W.; Dong, C.D.; Kumar, M.; Dakshinamoorthy, B.; Chang, J.H. Effect of chloride ions on electro-coagulation to treat industrial wastewater containing Cu and Ni. Sustainability 2020, 12, 7693. [Google Scholar] [CrossRef]
- Vinnett, L.; Waters, K.E. Representation of Kinetics Models in Batch Flotation as Distributed First-Order Reactions. Minerals 2020, 10, 913. [Google Scholar] [CrossRef]
- Wang, D.; Liu, Q. Hydrodynamics of froth flotation and its effects on fine and ultrafine mineral particle flotation: A literature review. Miner. Eng. 2021, 173, 107220. [Google Scholar] [CrossRef]
- Rosen, M.J.; Zhu, Z.H. Synergism in binary-mixtures of surfactants. 7. synergism in foaming and its relation to other types of synergism. J. Am. Oil Chem. Soc. 1988, 65, 663–668. [Google Scholar] [CrossRef]
- Xiong, Y. Bubble Size Effects in Coal Flotation and Phosphate Reverse Flotation Using a Pico-Nano Bubble Generator. Ph.D. Thesis, West Virginia University, Morgantown, VA, USA, 2014. [Google Scholar]
- Sobhy, A.; Tao, D. Nanobubble column flotation of fine coal particles and associated fundamentals. Int. J. Miner. Process. 2013, 124, 109–116. [Google Scholar] [CrossRef]
- Fan, M.; Tao, D.; Zhao, Y.; Honaker, R. Effect of nanobubbles on the flotation of different sizes of coal particle. Min. Metall. Explor. 2013, 30, 157–161. [Google Scholar] [CrossRef]
- Ma, F.; Tao, D.; Tao, Y. Effects of nanobubbles in column flotation of Chinese sub-bituminous coal. Int. J. Coal Prep. Util. 2019, 1–17, 1142–1146. [Google Scholar] [CrossRef]
- Svetovoy, V.B. Spontaneous chemical reactions between hydrogen and oxygen in nanobubbles. Curr. Opin. Colloid Interface Sci. 2021, 52, 101423. [Google Scholar] [CrossRef]
- Zhou, W.; Liu, K.; Wang, L.; Zhou, B.; Niu, J.; Ou, L. The role of bulk micro-nanobubbles in reagent desorption and potential implication in flotation separation of highly hydrophobized minerals. Ultrason. Sonochem. 2020, 64, 104996. [Google Scholar] [CrossRef]
- Nazari, S.; Shafaei, S.Z.; Hassanzadeh, A.; Azizi, A.; Gharabaghi, M.; Ahmadi, R.; Shahbazi, B. Study of effective parameters on generating submicron (nano)-bubbles using the hydrodynamic cavitation. Physicochem. Probl. Miner. Process. 2020, 56, 884–904. [Google Scholar] [CrossRef]
- Filippov, L.O.; Duverger, A.; Filippova, I.V.; Kasaini, H.; Thiry, J. Selective flotation of silicates and Ca-bearing minerals: The role of non-ionic reagent on cationic flotation. Miner. Eng. 2012, 36–38, 314–323. [Google Scholar] [CrossRef]
- Guimaraes, R.C.; Araujo, A.C.; Peres, A.E.C. Reagents in igneous phosphate ores flotation. Miner. Eng. 2005, 18, 199–204. [Google Scholar] [CrossRef]
- Lovell, V.M. Froth Characteristics in Phosphate Flotation in Flotation; Fuerstenau, M.C., Ed.; Mintek: New York, NY, USA, 1976. [Google Scholar]
- Rao, K.H.; Dwari, R.K.; Lu, S.; Vilinska, A.; Somasundaran, P. Mixed anionic/non-ionic collectors in phosphate gangue flotation from magnetite fines. TOMPJ 2010, 4, 14–24. [Google Scholar] [CrossRef]
- Sis, H.; Chander, S. Reagents used in the flotation of phosphate ores: A critical review. Miner. Eng. 2003, 16, 577–585. [Google Scholar] [CrossRef]
- Sis, H.; Chander, S. Adsorption and contact angle of single and binary mixtures of surfactants on apatite. Miner. Eng. 2003, 16, 839–848. [Google Scholar] [CrossRef]
- Chen, C.; Zhu, H.; Sun, W.; Hu, Y.; Qi, W.; Liu, R. Synergetic effect of the mixed anionic/non-ionic collectors in low temperature flotation of scheelite. Minerals 2017, 7, 87. [Google Scholar] [CrossRef]





| Kinetic Model | Magnesite Fraction | k (min−1) | Rmax (%) | R2 |
|---|---|---|---|---|
| First order | −100 μm | 0.61 ± 0.004 | 78.95 ± 1.83 | 0.990 |
| −45 μm | 0.89 ± 0.11 | 78.27 ± 2.80 | 0.980 | |
| −25 μm | 2.01 ± 0.19 | 87.17 ± 1.50 | 0.990 |
| k (min−1) | Rmax (%) | kmb/kb | R2 | |
|---|---|---|---|---|
| In presence of microbubbles (mb) | kmb = 2.2 ± 0.2 | 85 ± 1.7 | 1.05 ± 0.4 | 0.983 |
| Absence of microbubbles (b) | kb = 2.1 ± 0.2 | 80 ± 1.3 | 0.990 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsave, P.K.; Kostoglou, M.; Karapantsios, T.D.; Lazaridis, N.K. Enhancing Fines Recovery by Hybrid Flotation Column and Mixed Collectors. Minerals 2023, 13, 849. https://doi.org/10.3390/min13070849
Tsave PK, Kostoglou M, Karapantsios TD, Lazaridis NK. Enhancing Fines Recovery by Hybrid Flotation Column and Mixed Collectors. Minerals. 2023; 13(7):849. https://doi.org/10.3390/min13070849
Chicago/Turabian StyleTsave, Polyxeni K., Margaritis Kostoglou, Thodoris D. Karapantsios, and Nikolaos K. Lazaridis. 2023. "Enhancing Fines Recovery by Hybrid Flotation Column and Mixed Collectors" Minerals 13, no. 7: 849. https://doi.org/10.3390/min13070849
APA StyleTsave, P. K., Kostoglou, M., Karapantsios, T. D., & Lazaridis, N. K. (2023). Enhancing Fines Recovery by Hybrid Flotation Column and Mixed Collectors. Minerals, 13(7), 849. https://doi.org/10.3390/min13070849








