Size-Fractionated Weathering of Olivine, Its CO2-Sequestration Rate, and Ecotoxicological Risk Assessment of Nickel Release
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. OWCS Model
3.2. PNEC-Pro Model
3.3. Coupling OWCS and PNEC-Pro
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IPCC. Sixth Assessment Report. Impacts, Adaptation and Vulnerability. Intergovernmental Panel on Climate Change, WHO, UNEP. 2021. Available online: https://www.ipcc.ch/report/ar6/wg2/downloads/ (accessed on 25 September 2022).
- Dunsmore, H.E. A geological perspective on global warming and the possibility of carbon dioxide removal as calcium carbonate mineral. Energy Convers. Mgmt. 1992, 33, 565–572. [Google Scholar] [CrossRef]
- Bearat, H.; McKelvy, M.J.; Chizmeshya, A.V.G.; Gormley, D.; Nunez, R.; Carpenter, R.W.; Squires, L.; Wolf, G.H. Carbon sequestration via aqueous olivine mineral carbonation: Role of passivating layer formation. Environ. Sci. Technol. 2006, 40, 4802–4808. [Google Scholar] [CrossRef]
- Schuiling, R.D.; De Boer, P.L. Coastal spreading of olivine to control atmospheric CO2 concentrations; A critical analysis of viability. Comment: Nature and laboratory experiments are different. Short Comm. Int. J. Greenh. Gas Control 2010, 4, 855–856. [Google Scholar] [CrossRef]
- Baumeister, J.L. Chemical Weathering of the Mafic Minerals Serpentine and Olivine in Natural Environments. UNLV Thesis, Dept. Geoscience, University of Nevada, Reno, NV, USA, 2012. [Google Scholar]
- Hartmann, J.; West, A.J.; Renforth, P.; Koehler, P.; De La Rocha, C.L.; Wolf-Gladrow, D.A.; Duerr, H.H.; Scheffran, J. Enhanced chemical weathering as a geoengineering strategy to reduce atmospheric carbon dioxide, supply nutrients, and mitigate ocean acidification. Rev. Geophys. 2013, 51, 113–149. [Google Scholar] [CrossRef]
- Malik, A. Kinetics of Olivine Dissolution in Column Experiments. Ph.D. Thesis, University Hamburg, Hamburg, Germany, 2017. [Google Scholar]
- Beerling, D.J.; Leake, J.R.; Long, S.P.; Scholes, J.D.; Ton, J.; Nelson, P.N.; Bird, M.; Kantzas, E.; Taylor, L.L.; Sarkar, B.; et al. Farming with crops and rocks to address global climate, food and soil security. Nat. Plants 2018, 4, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.; Possinger, A. Atmospheric CO2 removed by rock weathering. Nature 2020, 583, 204–205. [Google Scholar] [CrossRef] [PubMed]
- Te Pas, E. Is the CO2 Hunter a Green, Black or White Mineral? Exploring the Enhanced Weathering Potential of Olivine, Basalt, Wollastonite, Anorthite, and Albite to Improve Agricultural Production and Mitigate Climate Change. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 2020. [Google Scholar]
- Schuiling, R.D.; Tickiell, O. Enhanced weathering of olivine to capture CO2. J. Appl. Geochem. 2010, 4, 510–519. [Google Scholar]
- Bakker, D.J.; Beumer, V.; Hartog, N.; Snijders, W.; Sule, M.; Vink, J.P.M. Applications of Olivine in RWS Constructions; Inventory of Possibilities for Pilots; Deltares Technical Report BGS-1203661: Utrecht, The Netherlands, 2011. (In Dutch) [Google Scholar]
- Vink, J.P.M.; Den Hamer, D. Olivine Captures CO2 in the City of Rotterdam; Possibilities for Practical Applications and Climate Targets; Deltares Technical Report 1206650: Utrecht, The Netherlands, 2012. (In Dutch) [Google Scholar]
- World Bank. What You Need to Know About the Measurement, Reporting, and Verification (MRV) of Carbon Credits. Available online: https://www.worldbank.org/en/news/feature/2022/07/27/what-you-need-to-know-about-the-measurement-reporting-and-verification-mrv-of-carbon-credits (accessed on 11 December 2022).
- Christoph, B.; Keel, G.; Leifeld, J. The Role of Atmospheric Carbon Dioxide Removal in Swiss Climate Policy-Fundamentals and Recommended Actions; Report by Risk Dialogue Foundation; Commissioned by the Federal Office for the Environment: Bern, Germany, 2019. [Google Scholar]
- Wood, B.J.; Kleppa, O.J. Thermochemistry of forsterite-fayalite olivine solutions. Geochim. Cosmochim. Acta 1981, 45, 529–534. [Google Scholar] [CrossRef]
- Donaldson, C.H. The rates of dissolution of olivine, plagioclase, and quartz in a basalt metl. Mineral. Mag. 1985, 49, 683–693. [Google Scholar] [CrossRef]
- Torres, M.A.; West, A.J.; Li, G. Sulphide oxidation and carbonate dissolution as a source of CO2 over geological timescales. Nature 2014, 507, 346–349. [Google Scholar] [CrossRef]
- Gbor, P.K.; Jia, C.Q. Critical evaluation of coupling particle size distribution with the shrinking core model. Chem. Eng. Sci. 2004, 59, 1979–1987. [Google Scholar] [CrossRef]
- Graff, C.P.; Wittrup, K.D. Theoretical analysis of antibody targeting of tumor spheroids: Importance of dosage for penetration and affinity for retention. Cancer Res. 2003, 63, 1288–1296. [Google Scholar] [PubMed]
- Wanta, K.C.; Perdana, I.; Petrus, H.T.B.M. Evaluation of shrinking core model in leaching process of Pomalaa nickel laterite using citric acid as leachant at atmospheric conditions. Mater. Sci. Eng. 2017, 162, 012018. [Google Scholar] [CrossRef]
- Wu, J.; Ahn, J.; Lee, J. Kinetic and mechanism studies using shrinking core model for copper leaching from chalcopyrite in methanesulfonic acid with hydrogen perioxide. Miner. Process. Metall. Rev. 2020, 42, 2021. [Google Scholar]
- Safari, V.; Arzpeyma, G.; Rashchi, F.; Mostoufi, N. A shrinking particle—shrinking core model for leaching of a zinc ore containing silica. Intl. J. Miner. Process. 2009, 93, 79–83. [Google Scholar] [CrossRef]
- Hangx, S.; Spiers, C.J. Coastal spreading of olivine to control atmospheric CO2 concentrations: A critical analysis of viability. Intl. J. Greenh. Gas Control 2009, 3, 757–767. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Schott, J. Kinetics and mechanism of forsterite dissolution at 25C and pH from 1 to 12. Geochim. Cosmochim. Acta 2000, 64, 3313–3325. [Google Scholar] [CrossRef]
- White, A.F.; Blum, A.E.; Bullen, T.D.; Vivit, D.V.; Schulz, M.; Fitzpatrick, J. The effect of temperature on experimental and natural chemical weathering rates of granitoid rocks. Geochim. Cosmochim. Acta 1999, 63, 3277–3291. [Google Scholar] [CrossRef]
- Nagy, K.L.; Blum, A.E.; Lasaga, A.C. Dissolution and precipitation kinetics of kaolinite at 80 degrees C and pH 3; the dependence on solution saturation state. Am. J. Sci. 1991, 291, 649–686. [Google Scholar] [CrossRef]
- Holdren, G.R.; Speyer, P.M. Reaction rate-surface area relationships during the early stages of weathering 1. Geochim. Cosmochim. Acta 1985, 49, 675–681. [Google Scholar] [CrossRef]
- Palandri, J.L.; Kharaka, Y.K. A compilation of Rate Parameters of Water-Mineral Interaction Kinetics for Application to Geochemical Modelling; Technical Report by the US Geological Survey: Reston, VA, USA, 2004. [Google Scholar]
- Olsen, A.A. Forsterite Dissolution Kinetics: Applications and Implications for Chemical Weathering. Ph.D. Thesis, Faculty Virginia Polytechnic Institute State University, Blacksburg, VA, USA, 2007. [Google Scholar]
- Vink, J.P.M.; Giesen, D.; Ahlrichs, E. Olivine weathering in field trials. In Effect of Natural Environmental Conditions on Mineral Dissolution and the Potential Toxicity of Nickel; Deltares Technical Report 11204378: Utrecht, The Netherlands, 2022. [Google Scholar]
- Vink, J.P.M.; Meeussen, J.C.L. BIOCHEM-ORCHESTRA: A scenario-DSS for heavy metal speciation and ecotoxicological impacts in river systems. Environ. Poll. 2007, 148, 833–841. [Google Scholar] [CrossRef] [PubMed]
- SCHER. Opinion on the Chemicals and the Water Framework Directive: Technical Guidance for Deriving Environmental Quality Standards; Scientific Committee on Health and Environmental Risks, European Union: Brussels, Belgium, 2010. [Google Scholar]
- Schlekat, C.E.; Van Genderen, E.; DeSchamphelaere, K.A.C.; Antunes, P.M.C.; Rogevich, E.C.; Stubblefield, W.A. Cross-species extrapolation of chronic nickel biotic ligand models. Sci. Total Environ. 2010, 408, 6148–6157. [Google Scholar] [CrossRef] [PubMed]
- Verschoor, A.; Vink, J.P.M.; Vijver, M.G.; De Snoo, G. Geographical and temporal variation in Cu, Zn, Ni bioavailability and species sensitivity. Environ. Sci. Technol. 2011, 45, 6049–6056. [Google Scholar] [CrossRef]
- Rüdel, H.; Díaz Muñiz, C.; Garelick, H.; Kandile, N.; Miller, B.; Pantoja Munoz, L.; Peijnenburg, W.G.M.; Purchase, D.; Shevah, Y.; Van Sprang, P.; et al. Consideration of the bioavailability of metal/metalloid species in freshwaters: Experiences regarding the implementation of biotic ligand model-based approaches in risk assessment frameworks. Environ. Sci. Pollut. Res. 2015, 22, 7405–7421. [Google Scholar] [CrossRef]
- Pagenkopf, G.K. Gill surface interaction model for trace-metal toxicity to fishes: Role of complexation, pH and water hardness. Environ. Sci. Technol. 1983, 17, 342. [Google Scholar] [CrossRef]
- Playle, R.C.; Dixon, D.G. Copper and Cadmium binding to fish gills: Estimates of Metal-Gill Stability constants and modelling of metal accumulation. Can. J. Fish. Aquat. Sci. 1993, 50, 2678. [Google Scholar] [CrossRef]
- Di Toro, D.M.; Allen, H.E.; Bergman, H.L.; Meyer, J.S.; Paquin, P.R.; Santore, R.C. Biotic ligand model of the acute toxicity of metals. 1. Technical basis. Environ. Toxicol. Chem. 2001, 20, 2383. [Google Scholar] [CrossRef]
- De Schamphelaere, K.A.C.; Lofts, S.; Janssen, C.R. Bioavailability models for predicting acute and chronic toxicity of zinc to algae, daphnids, and fish in natural surface water. Environ. Toxicol. Chem. 2005, 24, 1190. [Google Scholar] [CrossRef]
- European Commission. Common Implementation Strategy for the Water Framework Directive (2000/60/EC) Guidance Document No. 27 Technical Guidance for Deriving Environmental Quality Standards; European Communities: Brussels, Belgium, 2011. [Google Scholar]
- European Commission. Science for Environment Policy. Advances in Fresh Water Risk Assessment: Experiences with Biotic Ligand Models; European Communities: Brussels, Belgium, 2016; Issue 441. [Google Scholar]
- European Union. Guidance Document No. 38 Technical Guidance for Implementing Environmental Quality Standards (EQS) for Metals; Consideration of metal bioavailability and natural background concentrations in assessing compliance. 2000/60/EC; European Union: Brussels, Belgium, 2019. [Google Scholar]
- European Union. European Union Risk Assessment Report. Nickel and Nickel Compounds; Prepared by The Danish Environmental Protection Agency, on behalf of the European Union. Health and Consumer Protection DC, B-1049; European Union: Brussels, Belgium, 2008. [Google Scholar]
- Verschoor, A.; Vijver, M.G.; Vink, J.P.M. Refinement and cross-validation of nickel bioavailability in PNEC-pro, a regulatory tool for site-specific risk assessment of metals in surface waters. Environ. Toxicol. Chem. 2017, 36, 2367–2376. [Google Scholar] [CrossRef]
- Vink, J.P.M.; Verschoor, A.; Vijver, M.G. PNEC-pro. Software Release Version 6. Available online: http://www.pnec-pro.com (accessed on 25 September 2016).
- Kremer, D.; Etzold, J.S.; Boldt, P.; Blaum, K.M.; Hahn, H.; Wotruba, R. Telle Geological mapping and characterization of possible primary input materials for the mineral sequestration of carbon dioxide in Europe. Minerals 2019, 9, 485. [Google Scholar] [CrossRef]
- NIST/SEMATECH e-Handbook of Statistical Methods. 2021. Available online: https://www.itl.nist.gov/div898/handbook/ (accessed on 23 April 2021). [CrossRef]
- Renforth, P.; Von Strandmann, P.A.E.; Henderson, G.M. The dissolution of olivine added to soil: Implications for enhanced weathering. Appl. Geochem. 2015, 61, 109–118. [Google Scholar] [CrossRef]
- Köhler, P.; Abrams, J.F.; Völker, C.; Hauck, J.; Wolf-Gladrow, D.A. Geoengineering impact of open ocean dissolution of olivine on atmospheric CO2 surface ocean pH and marine biology. Environ. Res. Lett. 2013, 8, 014009. [Google Scholar] [CrossRef]
- Montserrat, F.; Renforth, P.; Hartmann, J.; Leermakers, M.; Knops, P.; Meysman, P.J.R. Olivine dissolution in seawater; implications for CO2 sequestration through enhanced weathering in coastal environments. Environ. Sci. Technol. 2017, 51, 3960–3972. [Google Scholar] [CrossRef]
- Amann, T.; Hartmann, J.; Struyf, E.; De Oliveira Garcia, W.; Fisher, E.K.; Janssens, I.; Meire, P.; Schoelynck, J. Constraints on enhanced weathering and related carbon sequestration: A cropland mesocosm approach. Biogeosciences 2020, 17, 103–119. [Google Scholar] [CrossRef]
- Dietzen, C.; Harrisona, R.; Michelsen-Correac, S. Effectiveness of enhanced mineral weathering as a carbon sequestration tool and alternative to agricultural lime: An incubation experiment. Int. J. Greenh. Gas Control 2018, 74, 251–258. [Google Scholar] [CrossRef]
- Bach, L.T.; Gill, S.J.; Rickaby, R.E.M.; Gore, S.; Renforth, P. CO2 removal with enhanced weathering and ocean alkalinity enhancement: Potential risks and co-benefits for marine pelagic ecosystems. Front. Clim. 2019, 1, 1–21. [Google Scholar] [CrossRef]
- Kersbergen, G. The possibilities of Olivine Enhanced Weathering in Paved Road Construction. A Literature Review; Technical report nr. 4233670; Utrecht University: Utrecht, The Netherlands, 2020. [Google Scholar]
- Haque, F.; Chiang, Y.W.; Santos, R.M. Risk assessment of Ni, Cr, and Si release from alkaline minerals during enhanced weathering. Open Agric. 2020, 5, 166–175. [Google Scholar] [CrossRef]
- Campbell, D.J. Interactions between trace metals and aquatic organisms: A critique of the free ion activity model. In Metal Speciation and Bioavailability in Aquatic Systems; Tessier, A., Turner, D., Eds.; John Wiley: New York, NY, USA, 1995; pp. 45–102. [Google Scholar]
- Vink, J.P.M. Measurement of heavy metal speciation over redox gradients in natural water-sediment interfaces and implications for uptake by benthic organisms. Environ. Sci. Technol. 2002, 36, 5130–5138. [Google Scholar] [CrossRef] [PubMed]
- Vink, J.P.M. The origin of speciation: Trace metal kinetics and bioaccumulation by Oligochaetes and Chironomids in undisturbed water-sediment interfaces. Environ. Pollut. 2009, 157, 519–527. [Google Scholar] [CrossRef]
- Erickson, R.J. The biotic ligand model approach for addressing effects of exposure water chemistry on aquatic toxicity of metals: Genesis and challenges. Environ. Toxicol. Chem. 2013, 32, 1212. [Google Scholar] [CrossRef]
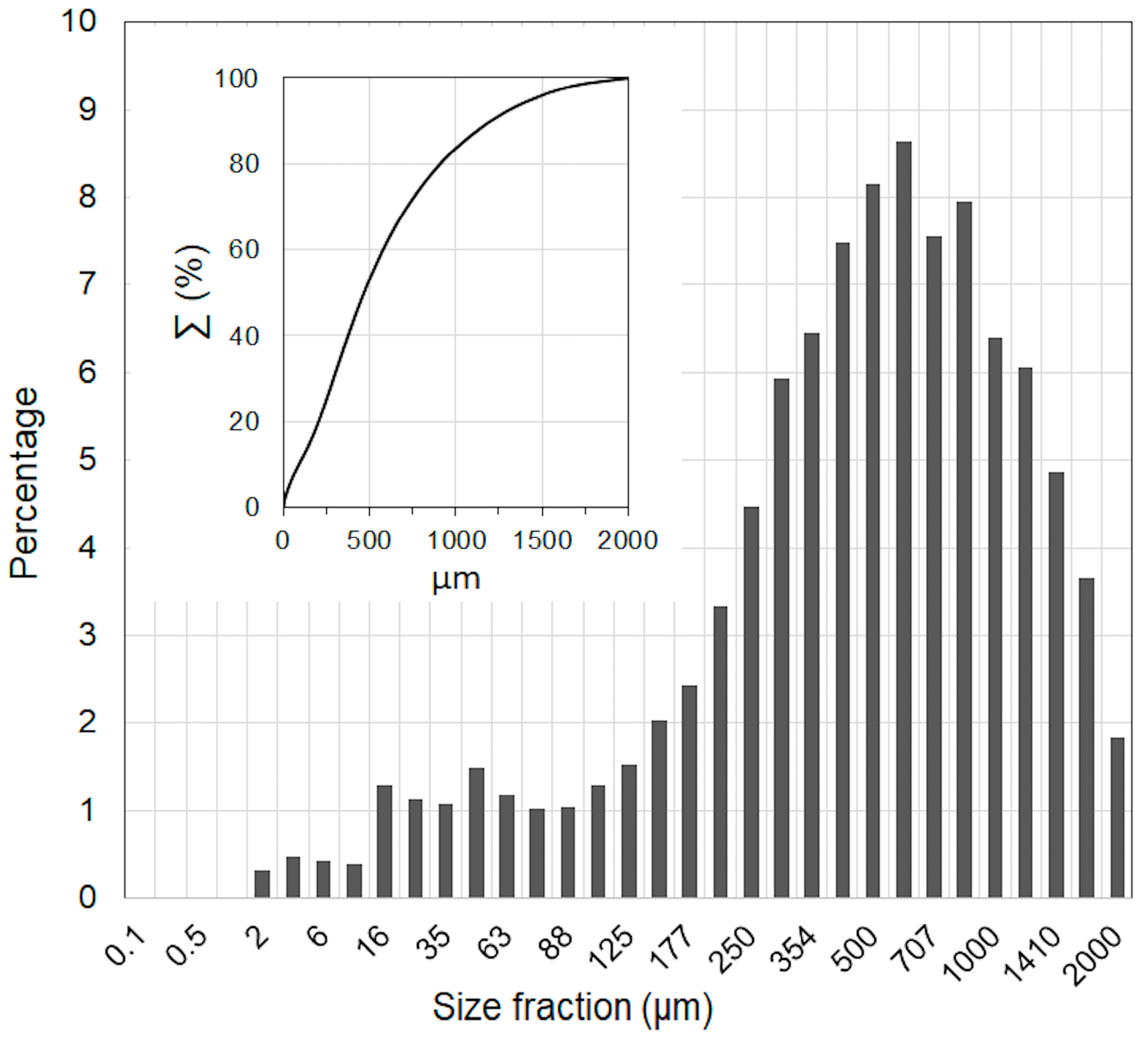

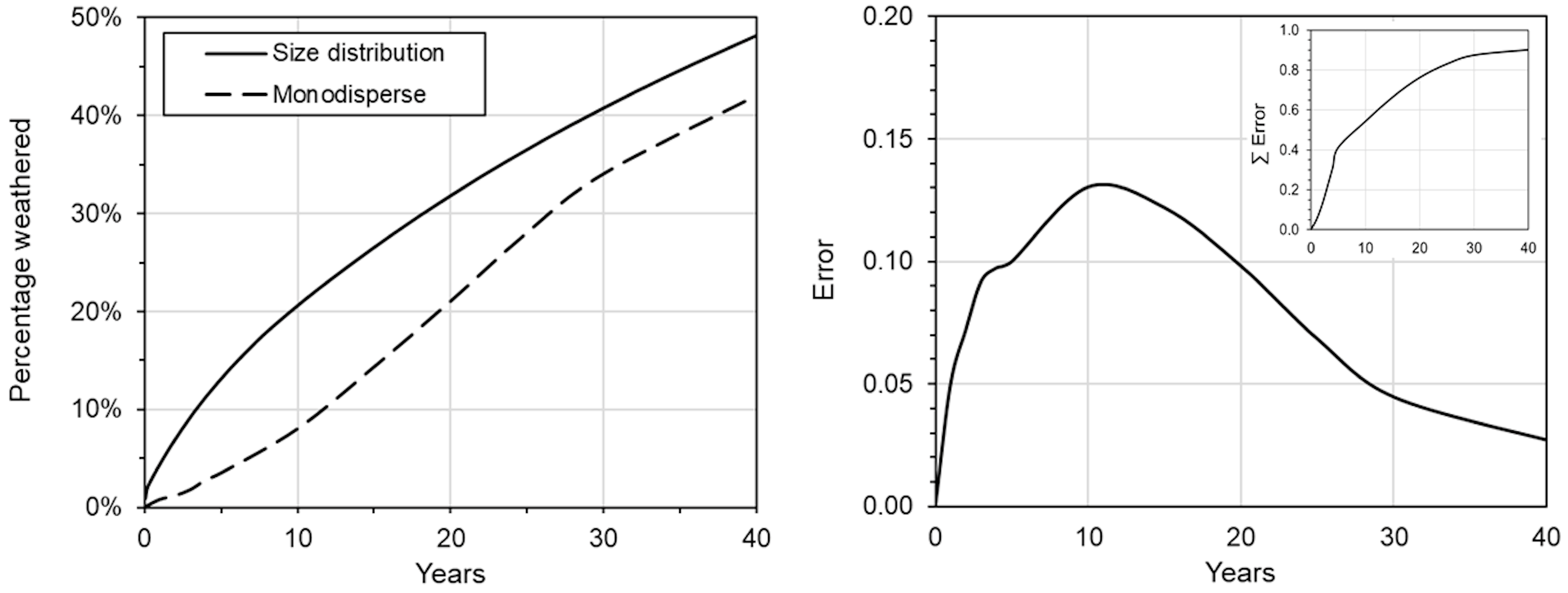
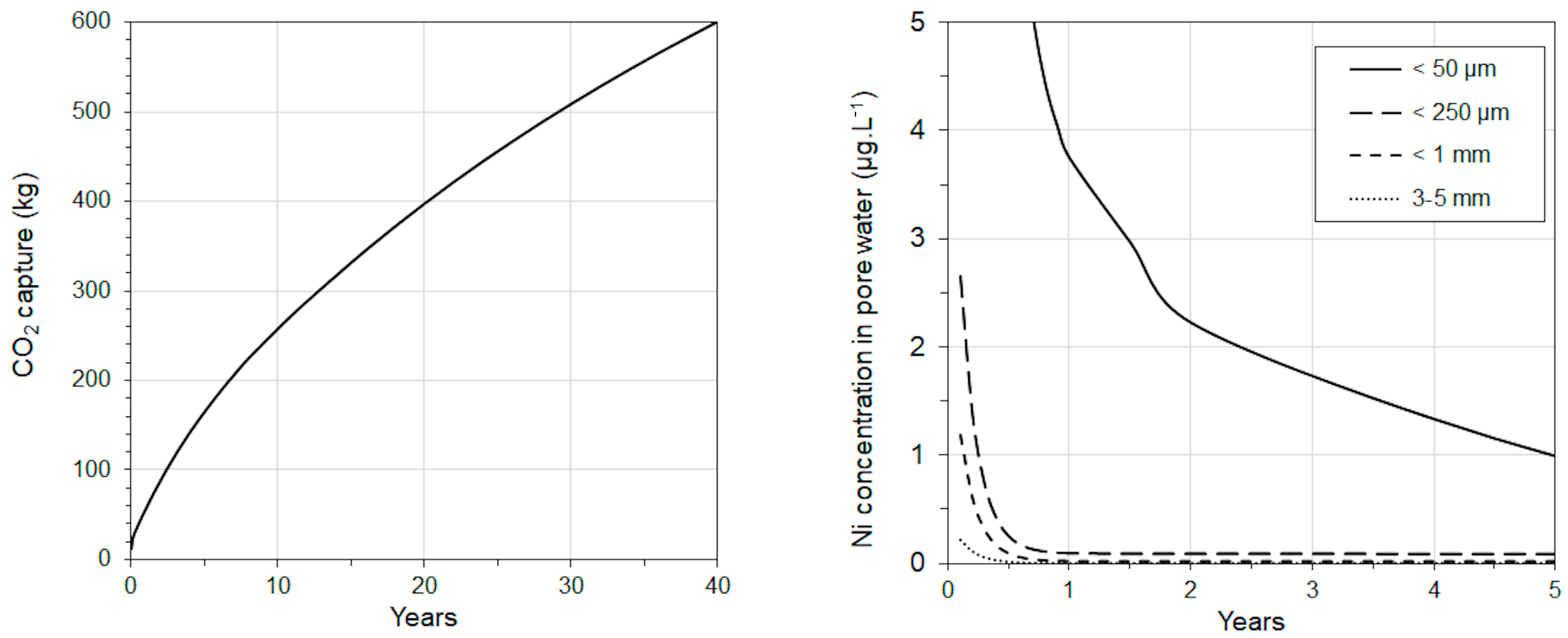
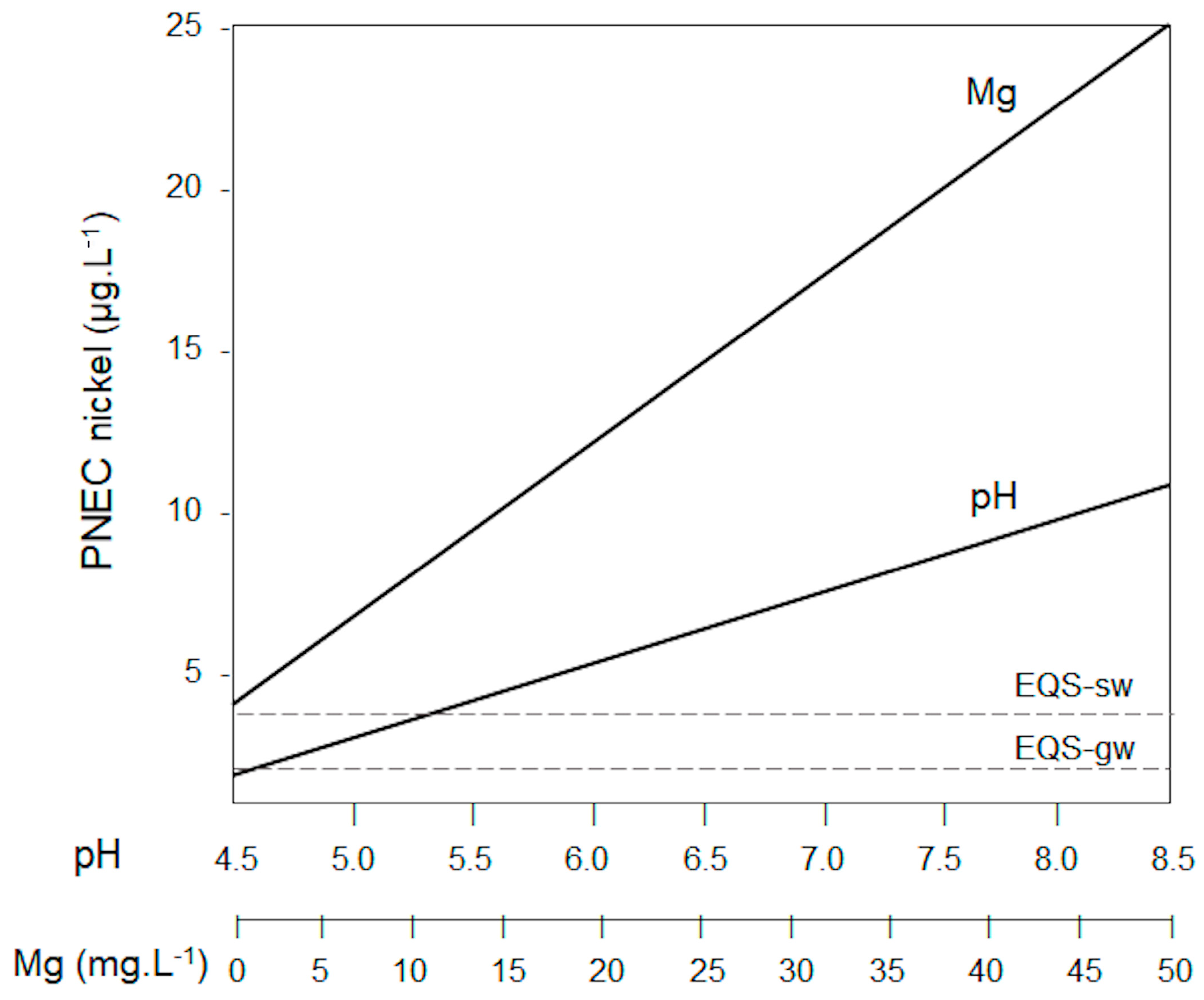
| Compound | Mass % | St. Dev. |
|---|---|---|
| SiO2 | 40.86 | ±0.01 |
| MgO | 35.11 | ±0.04 |
| Fe2O3 | 7.89 | ±0.01 |
| Al2O3 | 2.82 | ±0.01 |
| MnO | 0.13 | ±0.00 |
| Na2O | 0.06 | ±0.01 |
| TiO2 | 0.05 | ±0.00 |
| CaO | 2.15 | ±0.05 |
| K2O | 0.08 | ±0.01 |
| P2O5 | 0.01 | ±0.00 |
| Total | 89.16 | ±0.01 |
| Elements | mg∙kg−1 | |
| Cr | 2281 | ±62.4 |
| Ni | 1301 | ±0.6 |
| Sr | 32.2 | ±0.64 |
| Zr | 158.8 | ±1.3 |
| Ba | 35.8 | ±13.5 |
| Years of Weathering | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 3 | 5 | 10 | 15 | 30 | 40 | Original Mass (kg) | Remaining Mass (kg) | Dissolved (%) | ||
| Diameter | 2000 | 2000 | 1993 | 1989 | 1982 | 1971 | 1946 | 1928 | 55 | 48 | 13% | |
| 1410 | 1410 | 1407 | 1403 | 1398 | 1394 | 1377 | 1361 | 109 | 90 | 17% | ||
| 1000 | 999 | 993 | 989 | 982 | 971 | 946 | 928 | 144 | 110 | 24% | ||
| 707 | 706 | 700 | 696 | 689 | 678 | 653 | 635 | 243 | 156 | 36% | ||
| 420 | 419 | 413 | 409 | 402 | 391 | 366 | 348 | 199 | 90 | 55% | ||
| 250 | 249 | 243 | 239 | 232 | 221 | 196 | 178 | 102 | 23 | 77% | ||
| 150 | 149 | 143 | 139 | 132 | 121 | 96 | 78 | 49 | 2 | 97% | ||
| 88 | 87 | 81 | 77 | 70 | 59 | 34 | 16 | 33 | 0.0 | 100% | ||
| 50 | 49 | 43 | 39 | 32 | 21 | 0 | 0 | 37 | 0.0 | 100% | ||
| 16 | 15 | 9 | 5 | 0 | 0 | 0 | 0 | 13 | 0.0 | 100% | ||
| 8 | 7 | 1 | 0 | 0 | 0 | 0 | 0 | 16 | 0.0 | 100% | ||
| ∑ | 1000 | 519 | 48% | |||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vink, J.P.M.; Knops, P. Size-Fractionated Weathering of Olivine, Its CO2-Sequestration Rate, and Ecotoxicological Risk Assessment of Nickel Release. Minerals 2023, 13, 235. https://doi.org/10.3390/min13020235
Vink JPM, Knops P. Size-Fractionated Weathering of Olivine, Its CO2-Sequestration Rate, and Ecotoxicological Risk Assessment of Nickel Release. Minerals. 2023; 13(2):235. https://doi.org/10.3390/min13020235
Chicago/Turabian StyleVink, Jos P. M., and Pol Knops. 2023. "Size-Fractionated Weathering of Olivine, Its CO2-Sequestration Rate, and Ecotoxicological Risk Assessment of Nickel Release" Minerals 13, no. 2: 235. https://doi.org/10.3390/min13020235
APA StyleVink, J. P. M., & Knops, P. (2023). Size-Fractionated Weathering of Olivine, Its CO2-Sequestration Rate, and Ecotoxicological Risk Assessment of Nickel Release. Minerals, 13(2), 235. https://doi.org/10.3390/min13020235








