Abstract
Arsenic is a poisonous element and its super mobility can pose a major threat to the environment and human beings. Disposed arsenic-bearing waste or minerals over time may release arsenic into the groundwater, soil and then the food chain. Consequently, safe landfill deposition should be carried out to minimize arsenic bleeding. Cement-based stabilization/solidification and glass vitrification are two important methods for arsenic immobilization. This work compares the stability and intrinsic leaching properties of sequestered arsenic by cement encapsulation and glass vitrification of smelter high-arsenic flue dust (60% As2O3) and confirms if they meet or exceed the requirement of landfill disposition over a range of environmentally relevant conditions. The toxicity characterization leaching procedure (TCLP, 1311), synthetic precipitation leaching procedure (SPLP, 1312) and Australian standard (Aus. 4439.3) in short-term (18 h) and mass transfer from monolithic material using a semi-dynamic leaching tank (1315) in longer-term (165 days) were employed to assess arsenic immobility characteristic in three arsenic-cement (2%, 8.4% and 14.4%) and arsenic-glass (11.7%) samples. Moreover, calcium release from different matrices has been taken into consideration as a contributor to arsenic bleeding. Based on the USEPA guidelines, samples can be acceptable for landfilling only if As release is <5 mg/L. Results obtained from short-term leaching were almost similar for both cement and glass materials. However, high calcium release was observed from the cement-encapsulated materials. The pH of leachates after the test was highly alkaline for encapsulated materials; however, in glass material it was near neutral or slightly acidic. Method 1315 tests made a huge difference between the two materials and confirmed that cement encapsulation is not the best method for landfilling arsenic waste due to the high arsenic and calcium release over time with alkaline pH. However, glass material has shown promising results, i.e., the insignificant release of arsenic over time with an acceptable change in pH value. Overall, arsenic sequestration in glass is a better option compared with the cement-based solidification process.
1. Introduction
Arsenic is considered an element of high concern due to its toxic nature, mobilization, easy evaporation, bioaccumulation in the surrounding area, and it being a human carcinogen [1,2,3]. Naturally occurring arsenic (As) mainly exists in 60% arsenates, 20% sulfides, 10% oxides and sulfosalts, and the remaining 30% is divided between arsenites, arsenides, native elements, and metal alloys [4]. Arsenic minerals often coexist in most nonferrous metal ores such as copper, lead, zinc, nickel, gold, and silver minerals in different compositions such as sulfides and arsenides [4]. Well-known arsenic minerals are arsenian (As-rich) pyrite (Fe(S,As)2), and arsenopyrite (FeAsS) [4]. During metal extraction from minerals, especially sulfide minerals such as pyrite (FeS2), pyrrhotite (Fe1-xS, 0 < x < 0.2), and arsenopyrite, part of the waste is discharged in tailings, which leads to acid mine drainage (AMD) that contains a significant concentration of arsenic and is a major threat to the environment [5,6]. Moreover, it was reported that in Mozambique, the area near a coal-mining operation was contaminated as a result of heavy metal releases such as As (ca. 0.3–10.9 μg/L), selenium (Se), and boron (B) into the soil, groundwater, river sediments, and water and AMD generated from sulfide minerals (such as pyrite and arsenopyrite) tailing [7,8]. Moreover, in civil projects such as underground and tunnel construction, sedimentary rocks (5–10 mg/kg As) close to the marine area release acidic tailing, which contains heavy metals and poisonous metalloids such as As [9,10]. In the pyrometallurgy and hydrometallurgy processing of nonferrous metals, arsenic treatment is challenging and can be liberated as a mobile form; for instance, in copper production by smelters, copper waste concentrates contain >0.2% As [11].
The different secondary streams, e.g., tailings, tailing drainages, flue dust, etc., generated from ore processing contain a significant amount of arsenic [12,13,14,15]. Arsenic-bearing waste disposal could cause adverse impacts on the environment and human beings by entering into the groundwater, soil, and food chain. Therefore, there is a need for technologies to treat the arsenic-bearing wastes properly and make them suitable for reuse or safe landfill deposition. Important factors for choosing a proper As remediation method include As concentration in waste, the physical form of waste (solid or liquid), and minerals associated with the waste.
For liquid waste treatment containing arsenic (such as AMD), adsorption onto solid media has been used widely, especially for smaller facilities [16]. For instance, arsenic coprecipitation with alumina or iron-oxyhydroxide compounds such as schwertmannite, granular ferrihydrite (GFH), Bayoxide E-33 (E-33), iron-amended activated alumina (AAFS), and iron-impregnated sand has been performed to remove arsenic from wastewater (As > 1g/L), resulting from pyrometallurgical methods such as roasting and hydrometallurgical methods such as leaching [16,17]. However, for producing immobilized As-bearing iron-oxyhydroxide compounds, the Fe/As ratio should be higher than 4 and massive sludge formation should occur [17]. Scorodite (FeAsO4.2H2O) is an alternative for iron-oxyhydroxide compounds, which is stable under oxidizing conditions in the pH range of 2–5, and produces less sludge compared with iron-oxyhydroxide precipitation. high temperature (>150 °C) is required for scorodite crystallization [17], and at pH < 2 and pH > 6, ca. 20 mg/L and 5.38 mg/L As was released, respectively [18]. For lower As content (~10 mg/L) in the waste, in a proposed method by Park et al. [18] schwertmannite was utilized to concentrate As, and then Ca–Fe–AsO4 formed with low As dissolution [18].
Arsenic-bearing solid waste has been treated using extraction and stabilization/solidification (S/S) processes [19,20,21,22,23]. The stabilization/solidification method is one of the widely adopted ways to stabilize solid or semisolid arsenic and other hazardous waste by transforming them into less toxic phases using different components such as cement, lime, geopolymer, and pozzolanic materials as binders [24]. In the S/S method, metalloids in the soil are encapsulated by cement hydration, which minimizes As mobilization as a result of lower soil surface area and permeability [19,20,21,22,23]. However, this method is generally not considered viable for sulfidic waste due to the long-term instability of arsenic after landfill deposition of solidified hazardous waste. As a result of acidic liquor generation from sulfide ores, fast decay of cement occurs and could be a secondary source of pollution by generating AMD [18,25]. Geopolymerization is another way to stabilize As in form of construction materials by alkali activation of mine waste. Nevertheless, in this method, strong alkaline NaOH and sodium silicate (Na2SiO3) solutions are required [26]. The main disadvantage of the above-mentioned techniques is that the modified As waste is not suitable for regeneration and reusing in industry and should be disposed of in landfills [16].
Depending on the minerals associated with the As waste, the stabilization method will be selected. Tailings from gold-mining industries mainly consist of quartz and minute coal clay, which is considered granular silica sand [16]. In addition to S/S, vitrification process came into prominence as an alternative technology for As remediation, in which the fusion of heavy-metal-bearing waste at high temperature (1100–1600 °C) occurs and then during cooling it transforms into a stable (silicate) glass material, minimizing the threat to the environment [15,23,27,28,29,30,31]. The glass produced from waste has different applications in civil equipment (if their mechanical properties are qualified, they can be used in bricks or ceramics production), glass production, and hydraulic fracturing of oil wells [16]. Recently, GlassLock ProcessTM (Canada) as an environmentally-friendly method was developed by a Canadian company named Dundee Sustainable Technologies (DST), which showed promising immobilization of contaminants in acidic waste liquor, drainage, mineral concentrates (arsenopyrite, cobaltite, enargite, tennantite), and metallurgical flue dust, via its fusion into glass material using the vitrification process [32,33,34]. For As remediation of flue dust, silica, reused glass, and hematite have been mixed and vitrified the metallurgical waste into a 20% As-content glass [32,33,34]. Different leaching methods for the stability assessment of waste into solids have been explored by various research groups [12,35,36,37,38,39,40,41,42,43,44,45,46,47].
Environmental regulations made the United States Environmental Protection Agency (USEPA) define different leaching tests (static and semidynamic) for evaluating contaminant movement in metallurgical wastes. Regarding static tests, the toxicity characteristic leaching procedure (TCLP, USEPA method 1311), synthetic precipitation leaching procedure (SPLP, USEPA method 1312), and Australian standard (AUS. 4439.3) were defined to analyze the mobility of both organic and inorganic contaminants in liquid, solid, and multiphasic wastes. In the case of the semidynamic leaching test, USEPA method 1315 was introduced to measure the monolithic mass-transfer rates during the tank leaching test in the long term. These standard leaching tests are traditional single-point tests to simulate contaminant release under specific conditions and to confirm if the encapsulated waste meets or exceeds the requirement of safe landfill disposition over a range of environmentally relevant conditions [45,46,47,48].
From the environmental point of view, it is important to compare the immobilization properties of As after encapsulation by S/S and glass vitrification. For the first time, we compared the stability of arsenic-bearing flue dust in two different materials, i.e., cement and glass. TCLP (1311), SPLP (1312), Australian standard (AUS. 4439.3), and semi-dynamic tank leaching (1315) were performed to assess the stability of arsenic under specific conditions [45,46,47,48]. Moreover, calcium liberation from different matrices was also investigated as a result of high calcium release from cement-encapsulated samples. Available arsenic content, equilibrium pH screening, liquid–solid partitioning, and mass transport were taken into consideration during the evaluation of arsenic stability. The work was performed to evaluate the suitability of glass over cementitious material for arsenic remediation under different experimental scenarios followed by EPA and Australian standard guidelines.
2. Methodology
2.1. Materials
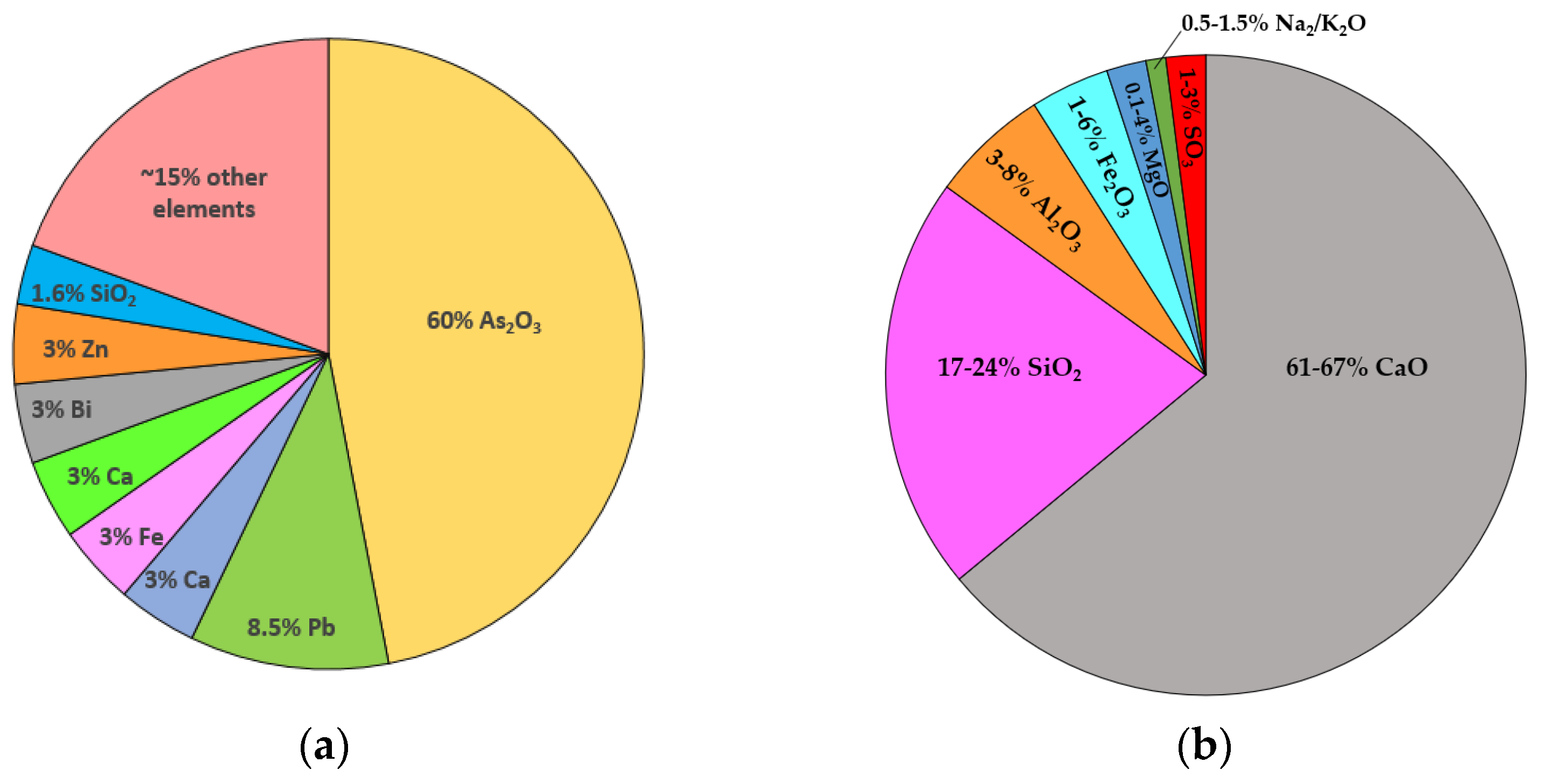
The arsenic-containing flue dust and flue-dust-encapsulated glass sample were procured from a local mining industry. The chemical composition of the smelter high-arsenic flue dust that was provided by the company is shown in Figure 1a. Glass samples provided by the company comprised arsenic-bearing flue dust in a mixture of silica, recycled glass, and hematite that was vitrified into an insoluble glass containing up to 20% arsenic. The Portland cement and lime (PL) method was used to solidify the flue dust in the PL matrix. Type 10 ordinary Portland cement (OPC) was used as cementitious binders to prepare samples, and its chemical composition is shown in Figure 1b. The materials, i.e., HNO3, H2SO4, CH3COOH, NaOH, and lime were of analytical reagent grade and procured from Fisher Scientific™ Canada. Each experiment was repeated thrice, and average values were reported. Ultrapure water was used to prepare the solutions throughout the study. The extraction bottles made of polypropylene were used to execute the experiments. A solution of 5% HNO3 was used to clean the vessels followed by rinsing with ultrapure water several times [49].
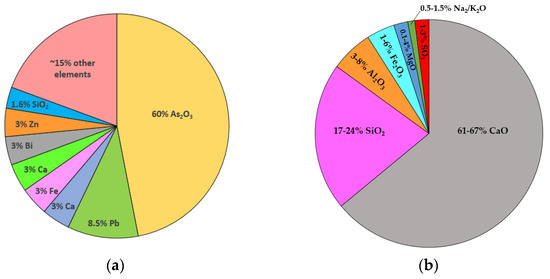
Figure 1.
Chemical composition of (a) smelter high-arsenic flue dust and (b) type 10 ordinary Portland cement (OPC).
2.2. Characterization Techniques
The flue dust and glass materials were digested using the sodium peroxide (Na2O2) fusion method followed by dilute nitric acid addition for a complete dissolution of minerals and determination of trace elements [50]. The filtered solutions after digestion were analyzed using the NexION ICP-MS (ELAN 9000, PerkinElmer, Woodbridge, ON, Canada) instrument for the concentration of different elements. The materials were also subjected to X’Pert Pro Philips powder diffractometer employing Cu-Kα radiation (λ = 1.54 Å) for further characterization. To observe surface material, a field-emission scanning electron microscope (FE-SEM Quanta 650, Thermo Fisher Scientific-FEI, Hillsboro, OR, USA) was employed.
2.3. Cement Encapsulation of Flue Dust
The flue dust sample containing a very high concentration of arsenic (45.35%) was dried at 60 °C for 24 h. Three cement-encapsulated materials were prepared with the addition of binders including lime (CaO), Portland cement, and water to the flue dust, as their compositions are shown in Table 1; considering the procedure described in Vandecasteele et al. [51] with slight modifications. Different recipes were defined by Vandecasteele et al. [51], in which depending on the sample characteristic the cement/waste mass ratio varied between 0.9–2.2, the lime/waste mass ratio was 0.8, and the water/cement ratio was between 0.68–3.33. In our procedure for cement encapsulation of flue dust, different amounts of flue dust were added to 120 g of slaked lime and 80 g of cement together with enough water to produce solidified material. The required time to harden the materials was 28 days. Moreover, three cylindrical molds were produced for 1315 tests. The dried molds were found to contain 2%, 8.4%, and 14.4% As for samples CF1, CF2, and CF3, respectively.

Table 1.
Composition of different solidified materials produced.
2.4. Methods
The United States Environmental Protection Agency (USEPA) and Australian Standard (AUS.) procedures were used to study the mobilization of arsenic from cement-encapsulated flue dust and glass material. All experiments were run in replicates and average data were reported with a standard deviation of <±3.2%.
2.4.1. Toxicity Characteristic Leaching Procedure (TCLP, USEPA1311)
In this procedure, simulated landfill leachate was used to test the arsenic bleeding from the material [46]. Since the arsenical materials involved in this research were all 100% solids, the first step of solid–liquid separation could be skipped. The encapsulated flue dust and glass materials were reduced to the required particle-size dimensions to increase the surface area per gram of material equal to or greater than 3.1 cm2 for different tests. The materials used for the TCLP test were provided in particle sizes of 4 mm < to <10 mm. The weight of the solid/extraction fluid ratio should be equal to 20. Therefore, at least 100 g of arsenic solid sample along with 2000 mL extraction fluid was added into a plastic container made of polypropylene (PP). Proper extraction fluid was chosen based on the preliminary evaluation, in which 5 g of the solid sample with an approximately 1 mm particle size should be mixed with 96.5 mL water in a flask and stirred heavily for 5 min on a magnetic stirrer. Then, pH should be recorded and based on the pH value proper extraction fluid will be chosen. In the current experiment, pH > 5, so glacial acetic acid (CH3CH2COOH) was adopted as the leaching agent as it mimics organic acids present in typical municipal landfill leachates. For the extraction-fluid preparation, 5.7 mL glacial acetic acid solution should be diluted with 1L water to reach approximate pH of 2.88 ± 0.05, as detailed in TCLP 1311 section 7.1.4 as Solution # 2 [46]. It is important to check the extraction fluid frequently for impurity avoidance, and pH should be monitored before the experiment. The leaching container was secured in a rotary reactor device, which rotated at 30 ± 2 rpm speed for 18 h. After tests, leaching solutions were subjected to determine the concentrations of calcium and arsenic.
2.4.2. Synthetic Precipitation Leaching Procedure (SPLP, USEPA 1312)
This method [45] is an alternative leaching procedure designed to evaluate the bleeding of arsenic from materials under acidic rainfall conditions. The extracting fluid was a 6:4 mass ratio of sulphuric acid and nitric acid with a pH range of 4.2. For pH adjustment, the diluted extraction fluid with deionized water can be used. The other operational procedures and experimental apparatus of SPLP follow the same standards as TCLP.
2.4.3. Australian Standard (AUS. 4439.3)
The Australian standard method follows the same experimental procedure as TCLP. The only difference is that the particle size of the material used should lie between 1.0 mm to 2.4 mm and the pH of leaching agent is 2.9 ± 0.05 buffered with an acetic acid solution [48].
2.4.4. Mass Transfer from Monolithic Material (USEPA 1315)
This procedure is used to determine mass-transfer-based release-rate information from monolithic cemented flue dust material in a sequential tank test [44]. The monolithic materials, i.e., cement-encapsulated flue dust and glass, were sequentially suspended in a series of tanks of leaching solution for predetermined leaching intervals. Three different monolithic cement-encapsulated flue dust samples and a glass sample with whole block geometries (surface area = 267.9 cm2, 248.6 cm2, 320.2 cm2, 165.8 cm2) were immersed in different leaching tanks filled with ultrapure water as extraction fluid to maintain liquid/surface area equal to 9 mL/cm2. The leaching tests were carried out for 165 days and samples were continuously subjected to fresh extraction fluid at the end of each leaching period. The elutes obtained from each tank at a specific cumulative leaching period were analyzed for arsenic content after filtration.
3. Results and Discussion
3.1. Characterization of Materials
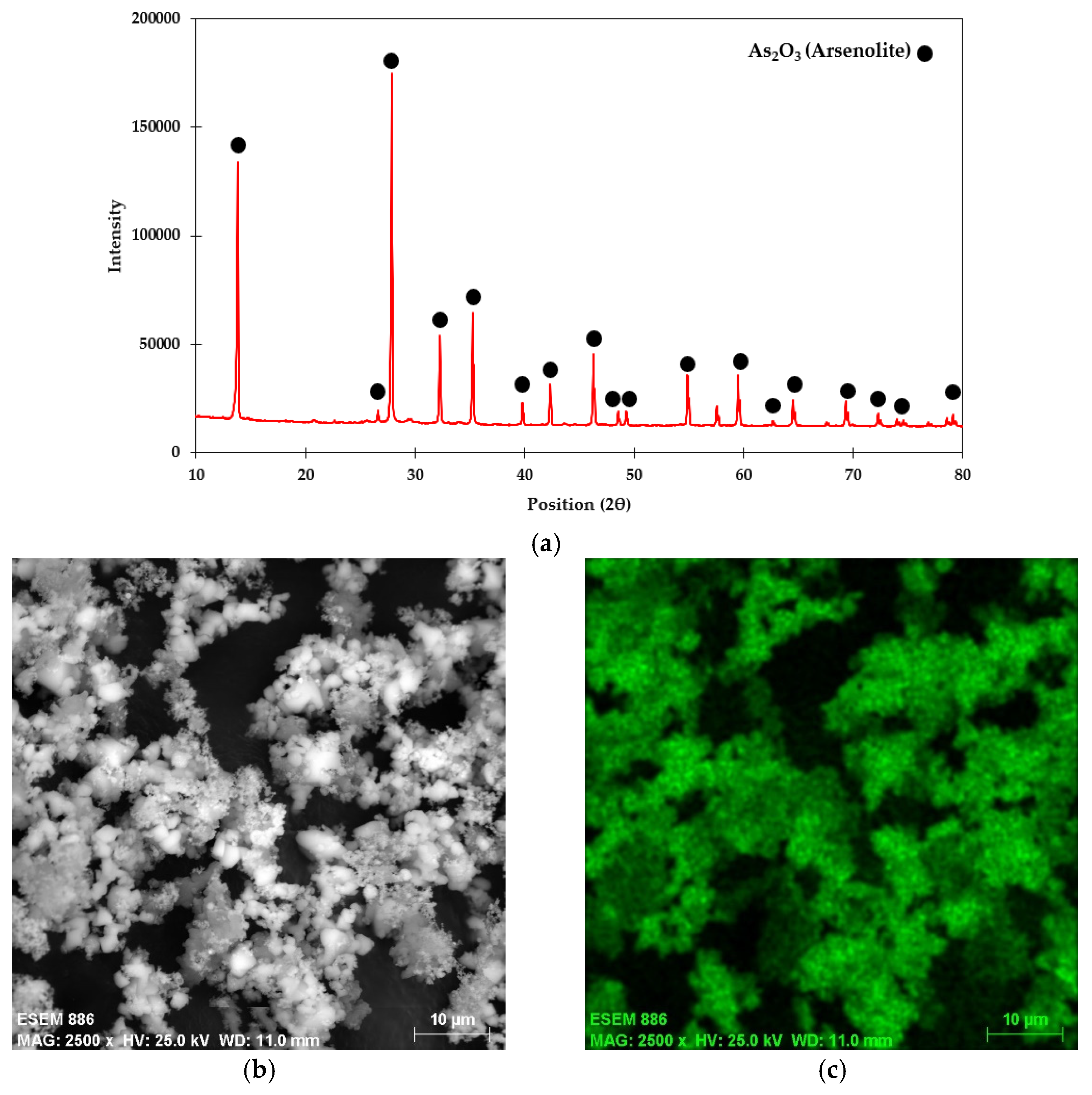
The arsenic concentration in digested liquor of flue dust was determined using the ICP-MS technique and was found to be 45.35%. The qualitative phase analysis of the flue dust using the XRD technique confirmed the presence of arsenolite (As2O3) crystalline phase (Ref. Code:00-036-1490) (Figure 2a). The SEM image of flue dust (Figure 2b) shows that the As2O3 crystals are cubical. The elemental analysis of arsenic on the surface (EDS) indicates that As appears mostly on the flue dust surface (Figure 2c), which is in good agreement with the XRD pattern.
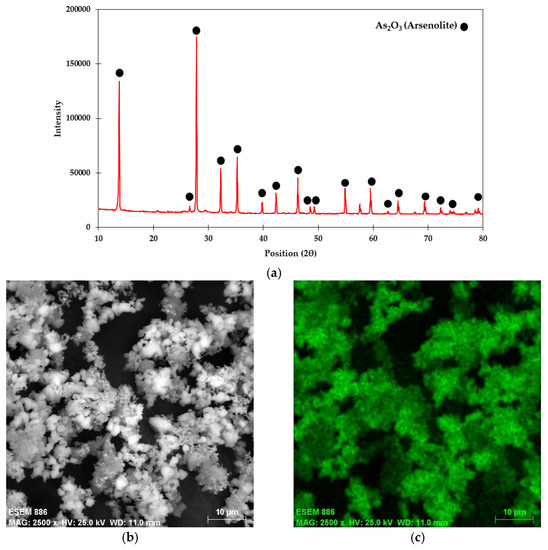
Figure 2.
(a) XRD pattern, (b) SEM image, and (c) arsenic EDS of smelter high-arsenic flue dust.
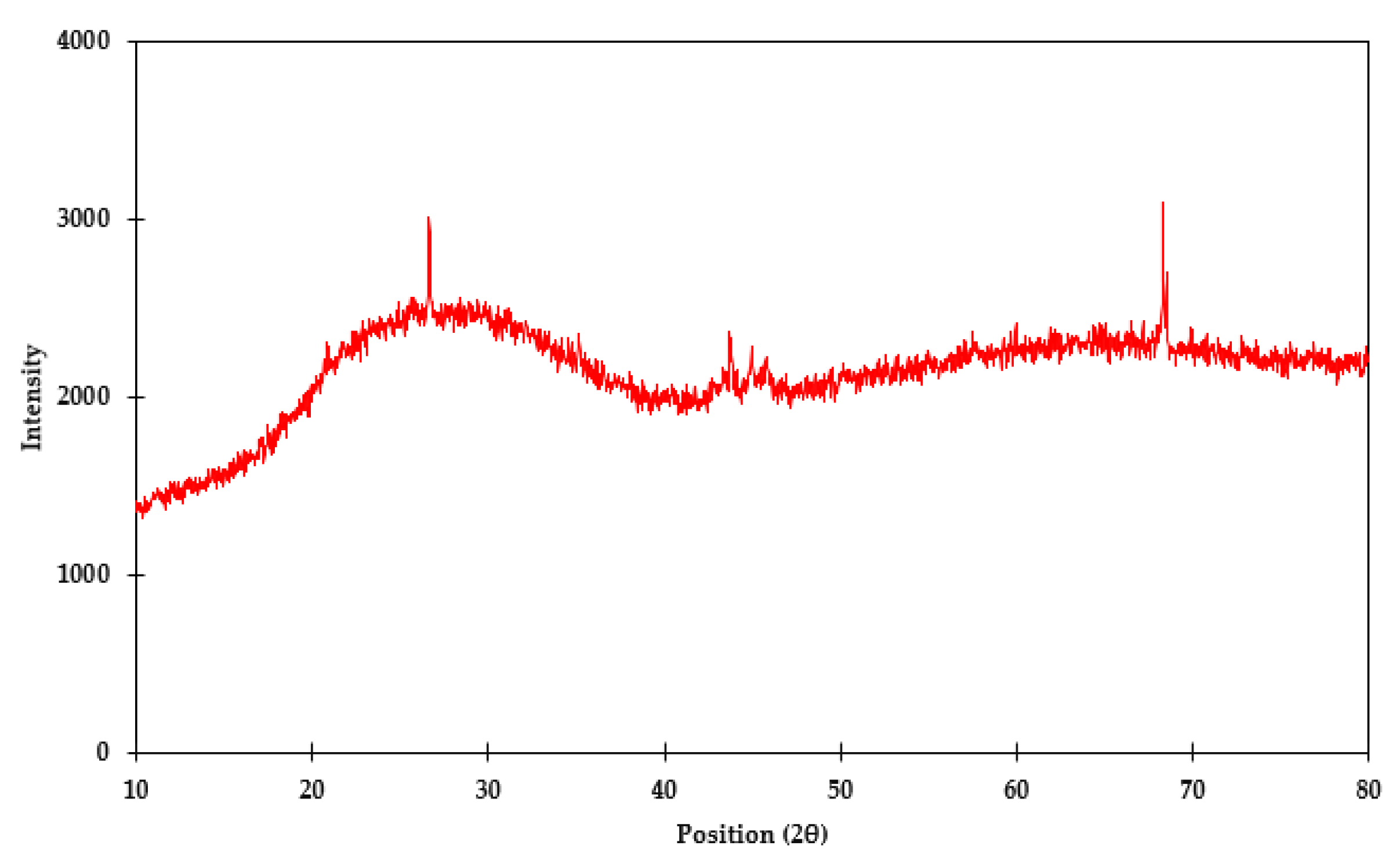
Regarding characterization of vitrified As in glass materials, 11.7% As was reported in the glass material by the material-supplying company. The major composition of digested liquor of the glass material is given in Table 2. The amorphous nature of the glass material was also confirmed with the XRD technique, as it shows a noisy pattern (Figure 3).

Table 2.
The major composition of digested liquor of glass material [32].
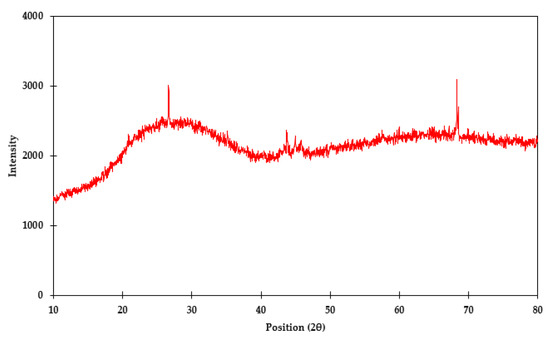
Figure 3.
XRD pattern of vitrified As in glass material.
3.1.1. TCLP and SPLP Tests
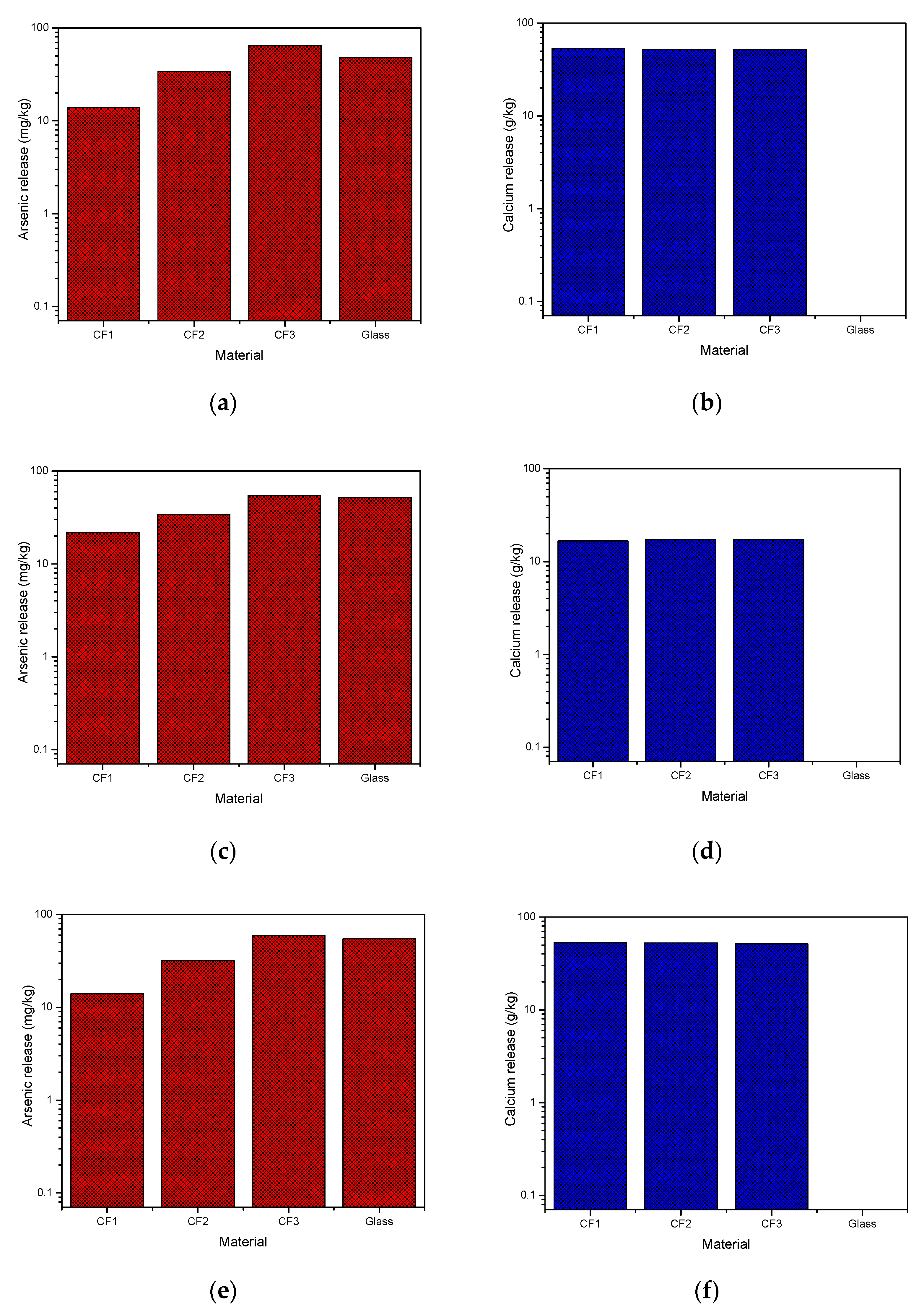
The TCLP method 1311, SPLP method 1312, and AUS. 4439.3 were applied to CF1, CF2, CF3, and glass samples for 18 h to measure arsenic stability in different structures and final pH and under different simulated conditions. In addition to arsenic, calcium release was also recorded as an effective parameter in arsenic leaching. Arsenic concentration in the leachate is correlated with the pH and the calcium concentration due to the formation of slightly soluble CaHAsO3. The calcium effect has been extensively discussed in further sections. Arsenic and calcium released in the solution from different samples are shown in Figure 4. Figure 4a,b showed that a maximum of 59 mg/kg arsenic and 53.4 g/kg calcium were released from samples CF3 and CF1, respectively, after the SPLP test. Based on the USEPA guidelines, samples were eligible for safe landfilling since arsenic release in the solution was less than 5 mg/L (Table 3). However, it should be mentioned that TCLP results, sometimes underestimate specific landfill leaching conditions and more standardized tests with different conditions such as SPLP are required to confirm whether a waste is acceptable for environmentally-friendly disposal or not [16]. Similarly, in the SPLP method, Figure 4c,d, a maximum of 55 mg/kg arsenic and 17.3 g/kg calcium were released from sample CF3. The mobilization characteristic of arsenic as an oxyanion increases in alkaline pH (>8.5) conditions [39], and since leachate pH in SPLP is high, the arsenic released in the solution is also high. The AUS. 4439.3 method resulted in a similar trend as observed in TCLP 1311 tests and a maximum of 60 mg/kg arsenic and 53 g/kg calcium were released from samples CF3 and CF1, respectively (Figure 4e,f). Comparing arsenic release in Figure 4a with Figure 4c,e indicates that TCLP is not as aggressive as the other two methods for glass materials; however, results obtained from all leaching experiments indicate that cement and glass samples are considered nonhazardous waste.
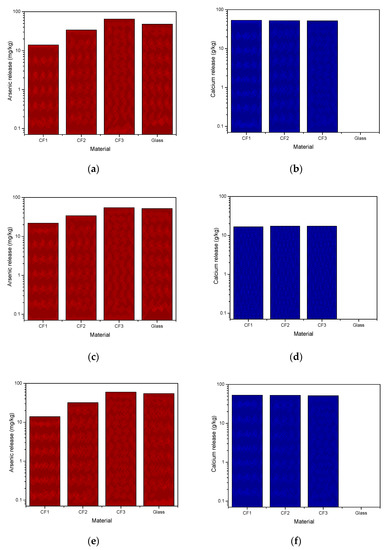
Figure 4.
Arsenic and calcium release from cement-encapsulated and glass materials (a,b) method 1311; (c,d) method 1312; (e,f) AUS. 4439.3.

Table 3.
Arsenic and calcium concentration in leachate.
Figure 4b,d,f show high calcium release in the case of cement-encapsulated samples. The glass sample showed minimum release of arsenic and calcium in comparison to the cement-encapsulated materials due to its compact nature and high porosity of cement-encapsulated materials. Final pH values of solutions after tests were in the range of 11.88–12.53 for cement-encapsulated samples; however, in the case of glass samples final pH values of solutions varied between 5.88–5.98, near neutral or slightly acidic, which minimizes arsenic mobility as an oxyanion. It should be added that microbial activity in the soil and near-neutral pH of an actual landfill result in reducing conditions for more arsenic leaching [16]. Consequently, considering As and Ca release and final pH, results revealed that glass material is a better option for flue dust sequestration and landfill deposition with low environmental risk.
3.1.2. Monolithic Mass-Transfer Test (USEPA 1315)
The bulk materials designed as CF1, CF2, CF3, and glass were subjected to semidynamic monolithic mass-transfer tests for 165 days. The samples were taken at definite time intervals and the medium was replaced with fresh deionized water each time. Samples were subjected to analysis of arsenic and calcium release with time.
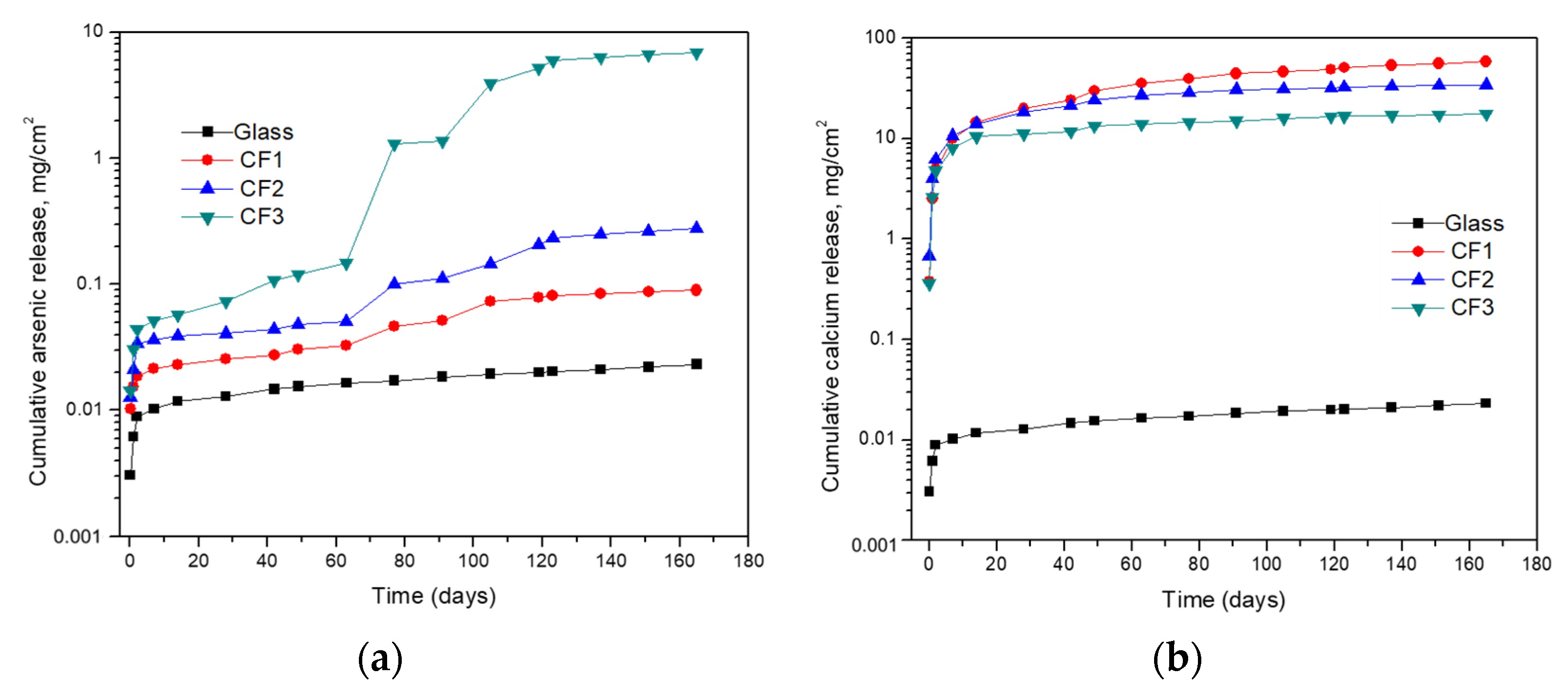
It is clear from Figure 5a,b that bleeding of arsenic and calcium from different samples showed a cumulative increase in their concentrations. However, the glass sample showed the minimum release of arsenic and calcium in comparison to the cement-encapsulated samples as a result of lower permeability and its significant intactness. The CF3 sample has the highest release of arsenic (6.96 mg/cm2) due to the presence of high amounts of flue dust, and so high arsenic content and CF1 showed the least release of arsenic (0.09 mg/cm2) due to the presence of less arsenic content in the sample and its bonding with calcium there to produce calcium arsenates (Ca3(AsO4)2). The release of calcium was related to the release of arsenic in cement-encapsulated samples due to the variation of arsenic content in the three samples. The driving force for the release of elements was much higher for the cement-encapsulated materials due to their high porosity. The slow release from glass material is due to its compact nature and weak interaction of the fluid with the material.
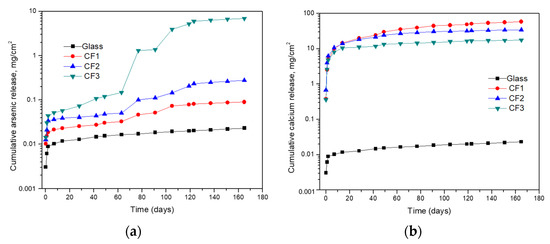
Figure 5.
(a) Arsenic and (b) calcium release from cement-encapsulated and glass materials in method 1315.
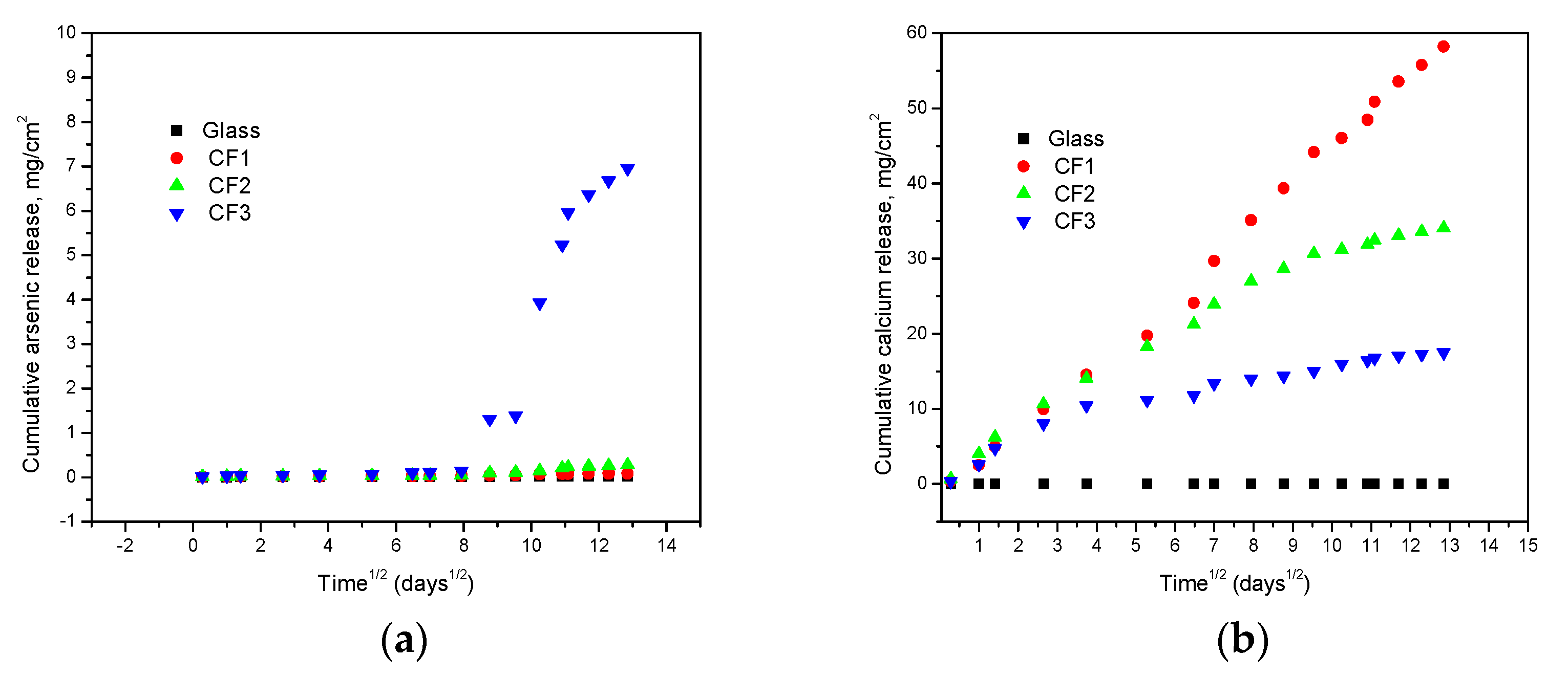
A plot between cumulative element release and (time)1/2 (Figure 6a,b) showed a linear relation followed by an ideal Fickian diffusion without other mass-transport limitations (USEPA, 2017e). The calcium release was much higher than arsenic in the case of cement-encapsulated samples due to the presence of high calcium content there. The sample with high arsenic content (CF3) showed a significant rise in arsenic concentration after a few days of the test probably due to retardation of content for the first few days.
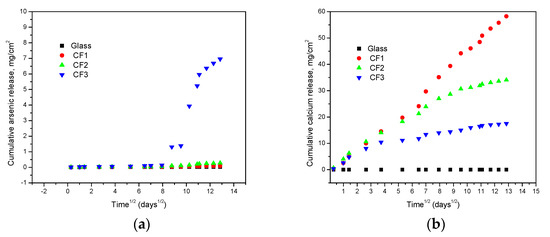
Figure 6.
Variation of cumulative (a) arsenic and (b) calcium with leaching time.
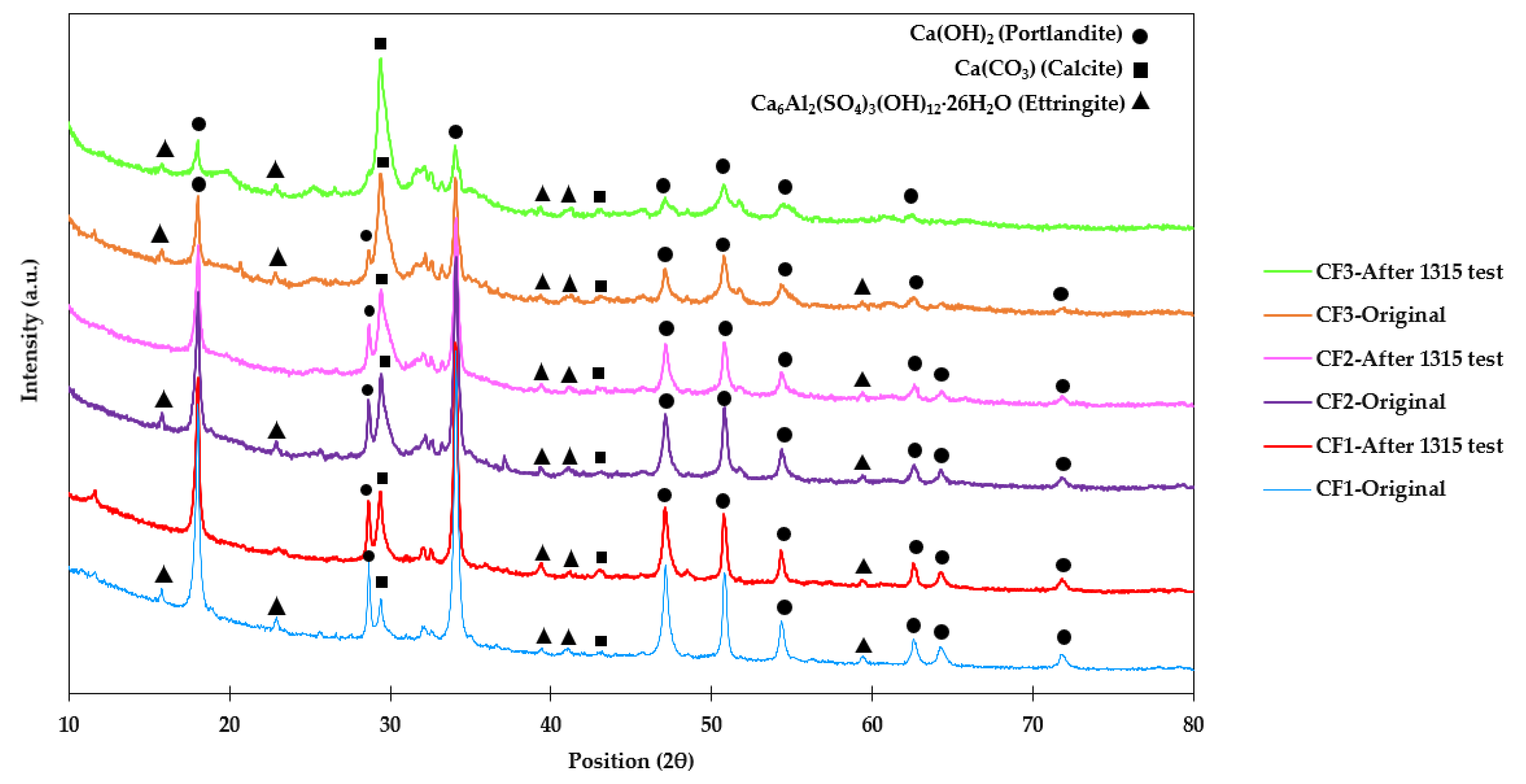
XRD patterns of CF1, CF2, and CF3 before and after method 1315 are shown in Figure 7. In this figure, two minerals were mostly assigned to characteristic peaks, including portlandite (Ca(OH)2) and calcite (carbonated portlandite) (Ca(CO3)). Portlandite appeared as sharp peaks with high intensity, compared to other peaks. At 28° and 34° (2θ), sharp peaks were found that show portlandite, and by increasing As content in the samples, the intensity of portlandite peaks decreased. Moreover, the peak at 29° was assigned to calcite, and from CF1 to CF3, the intensity of the peak increased and simultaneously its adjacent peak, at 28°, which indicates that portlandite decreased. In addition to these two minerals, ettringite (Ca6Al2(SO4)3(OH)12·26H2O) as the main cement hydration product was also detected as low-intensity peaks all over the spectra [52,53]. According to Li et al. [54], ettringite can boost the immobilization characteristic of arsenic by increasing the density, sorption, and compressive strength of cementitious material. However, a large amount of ettringite can have adverse effects on cement compressive strength and contribute to the dissociation of the internal structure [52,53,54].
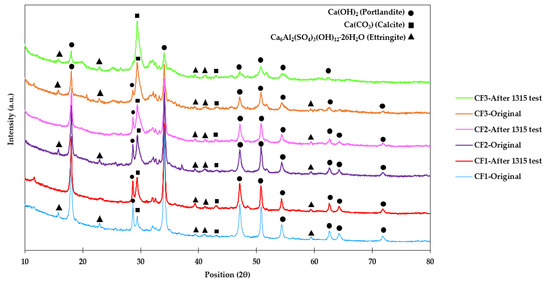
Figure 7.
XRD spectra of CF1, CF2, and CF3 before and after the 1315 test.
Figure 8 shows the SEM images of cement and glass material before and after the 1315 test. SEM images of the cement samples almost confirm the presence of ettringite crystals in the cement structure and the “Ostwald ripening” phenomena, in which small ettringite crystals under the desirable condition in large minerals pores grow and enhance the strength of the structure [52]. Thus, arsenic immobilization also improved. Yellow arrows in Figure 8 randomly show ettringite crystals. Generally, the morphologies of cement samples before and after leaching are quite similar; however, small ettringite crystals partially disappeared or lost their dense structures. The SEM image of the glass material before leaching shows a uniform surface, which almost remained unchanged after the 1315 test.


Figure 8.
SEM images of (a,b) CF1, (c,d) CF2, (e,f) CF3, and (g,h) glass material before and after test 1315, respectively.
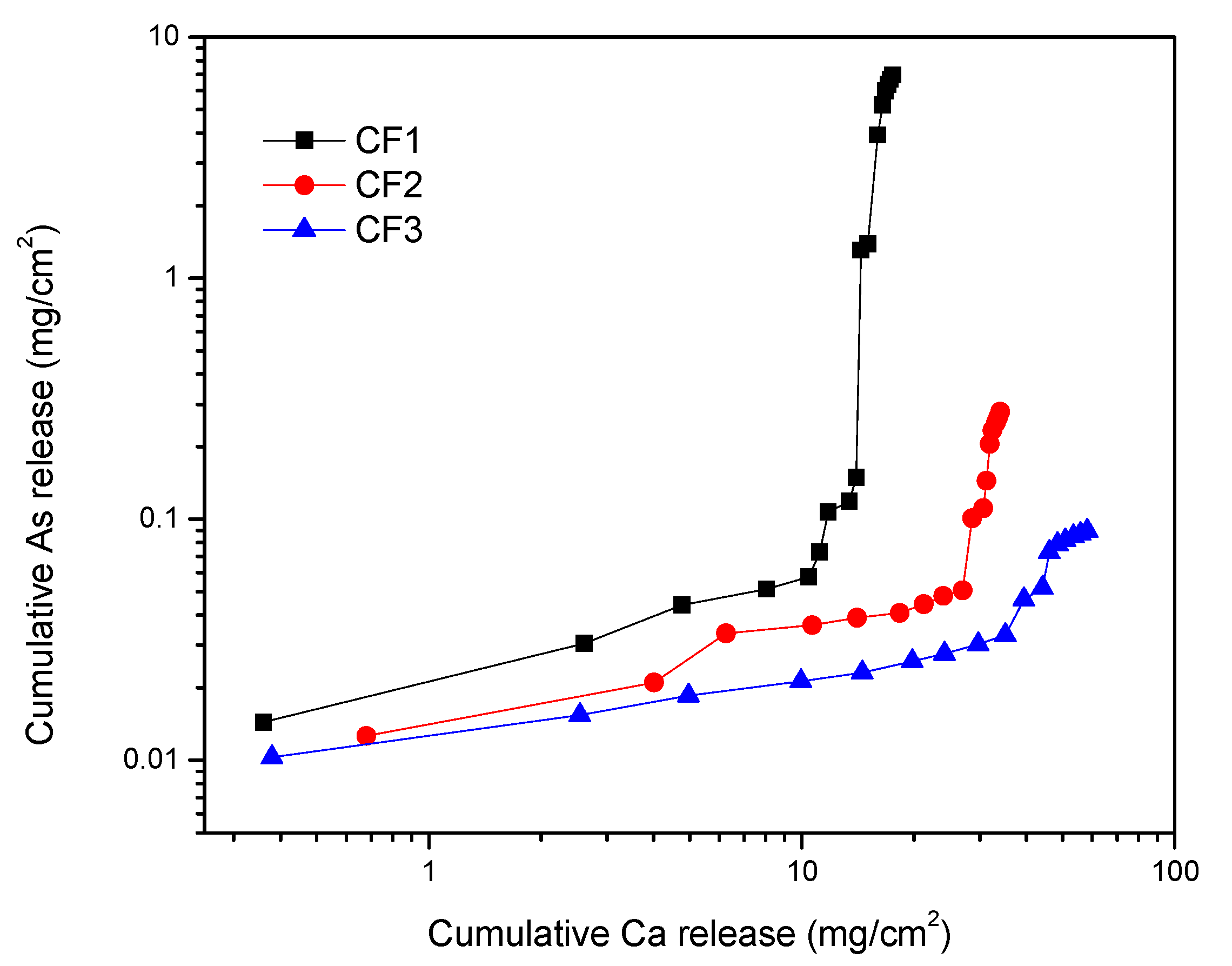
Figure 9 shows the effect of calcium bleeding on the arsenic leached from cement-encapsulated materials during the 1315 test. According to this figure, by increasing calcium release, arsenic release also increased. It is reported that arsenic concentration in the leachate is correlated with the pH and the calcium concentration due to the formation of slightly soluble CaHAsO3 [51]. It is reported that through arsenic removal from smelter flue dust by calcination, an excessive amount of CaO in a solution can increase the rate of As removal [54]. Moreover, during the cement hydration process, arsenic can be converted into calcium arsenate compounds, which are also considered responsible for arsenic immobilization through the cement-encapsulation process [54]. Moreover, for As fixation by gypsum precipitation, at high-pH calcium arsenate phase was precipitated [55,56]. Therefore, dissociation of calcium arsenate compounds under the 1315 leaching condition, in which pH is around neutral (6.95), may contribute to increasing As release.
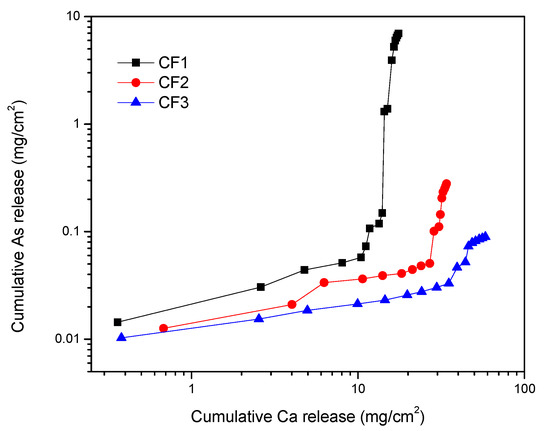
Figure 9.
Variation of cumulative leached calcium with cumulative leached arsenic from cement-encapsulated samples.
4. Conclusions
The TCLP, SPLP, and AUS. 4439.3 methods are traditional single-point tests to simulate release under specific conditions, and almost similar results were achieved for these tests from glass material and cement-encapsulated flue dust with high arsenic content (14.4%). However, high calcium release was observed from the cement-encapsulated materials. The pH of leachates after the test was highly alkaline for encapsulated materials; however, in glass material it was near neutral or slightly acidic. Method 1315 tests made a huge difference between the two materials and confirmed that cement encapsulation is not the best method for landfilling arsenic waste due to the high release of arsenic and calcium over time with alkaline pH. However, glass material showed insignificant release of arsenic over time with an acceptable change in pH value. This work revealed that cement encapsulation of flue dust can lower the arsenic bleeding in the leachate; however, it presents poor long-term leachability performances over the glass-vitrification method, which limits its applicability for the landfilling of hazardous waste such as smelter flue dust. On the other hand, sequestration of flue dust in glass material has been found to be a promising option due to satisfactory long-term leachability performances, which allow its applicability for the landfilling of hazardous waste.
The flue dust oxidation before encapsulation can lower the arsenic release in comparison to the arsenic release from encapsulated nonoxidized flue dust; however, it will increase the cost of the process. High release of calcium and high pH, which tend to increase the mobility of arsenic, are the other problems remaining relative to the GlassLock process. Other expected limitations such as the presence of halides, organics, sulfate, cyanide, calcium, or soluble salts of metals, i.e., Mn, Sn, Zn, Cu or Pb, may decrease the unconfined compressive strength or durability of the cement-encapsulated product or decrease the bond strength between the flue dust particles and the cement due to the presence of fine particulate matter. The above observations are in agreement with USEPA recommendation for vitrification as the best-developed available technologies for the following waste types: As-containing wastes including K031, K084, K101, K102, and D004 over cement solidification/stabilization.
Author Contributions
H.M.—Conceptualization, Investigation, Data curation, Resources, Writing—original draft A.A.—Data curation, Formal analysis, Writing—editing A.G.—Project administration, Supervision, Writing—review & editing, Funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
This research and APC were funded by NSERC Discovery Grant of Canada and Dundee Sustainable Technologies (DST), Canada (RGPIN-2015-05718).
Acknowledgments
The authors are thankful for the financial support during this work by NSERC Discovery Grant of Canada (RGPIN-2015-05718) and Dundee Sustainable Technologies (DST), Canada.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Nazari, A.M.; Radzinski, R.; Ghahreman, A. Review of arsenic metallurgy: Treatment of arsenical minerals and the immobilization of arsenic. Hydrometallurgy 2017, 174, 258–281. [Google Scholar] [CrossRef]
- Wu, C.; Mahandra, H.; Ghahreman, A. Novel Continuous Column Process for As(III) Oxidation from Concentrated Acidic Solutions with Activated Carbon Catalysis. Ind. Eng. Chem. Res. 2020, 59, 9882–9889. [Google Scholar] [CrossRef]
- Wu, C.; Mahandra, H.; Radzinski, R.; Ghahreman, A. Green catalytic process for in situ oxidation of Arsenic(III) in concentrated streams using activated carbon and oxygen gas. Chemosphere 2020, 261, 127688. [Google Scholar] [CrossRef]
- Tabelin, C.B.; Igarashi, T.; Villacorte-Tabelin, M.; Park, I.; Opiso, E.M.; Ito, M.; Hiroyoshi, N. Arsenic, selenium, boron, lead, cadmium, copper, and zinc in naturally contaminated rocks: A review of their sources, modes of enrichment, mechanisms of release, and mitigation strategies. Sci. Total Environ. 2018, 645, 1522–1553. [Google Scholar] [CrossRef] [PubMed]
- Park, I.; Baltazar Tabelin, C.; Jeon, S.; Li, X.; Seno, K.; Ito, M.; Hiroyoshi, N. A review of recent strategies for acid mine drainage prevention and mine tailings recycling. Chemosphere 2019, 219, 588–606. [Google Scholar] [CrossRef] [PubMed]
- Monjardin, C.E.F.; Senoro, D.B.; Magbanlac, J.J.M.; de Jesus, K.L.M.; Tabelin, C.B.; Natal, P.M. Geo-Accumulation Index ofManganese in Soils Due to Flooding in Boac andMogpog Rivers, Marinduque, Philippines with Mining Disaster Exposure. Appl. Sci. 2022, 12, 3527. [Google Scholar] [CrossRef]
- Marove, A.; Sotozono, R.; Tangviroon, P.; Tabelin, C.B.; Igarashi, T. Assessment of soil, sediment and water contaminations around open-pit coal mines in Moatize, Tete province, Mozambique. Environ. Adv. 2022, 8, 100215. [Google Scholar] [CrossRef]
- Marove, C.A.; Tangviroon, P.; Tabelin, C.B.; Igarashi, T. Leaching of hazardous elements from Mozambican coal and coal ash. J. Afr. Earth Sci. 2020, 168, 103861. [Google Scholar] [CrossRef]
- Arima, T.; Sasaki, R.; Yamamoto, T.; Tabelin, C.B.; Tamoto, S.; Igarashi, T. Effects of Environmental Factors on the Leaching and Immobilization Behavior of Arsenic from Mudstone by Laboratory and In Situ Column Experiments. Minerals 2021, 11, 1220. [Google Scholar] [CrossRef]
- Ho, G.D.; Tabelin, C.B.; Tangviroon, P.; Tamamura, S.; Igarashi, T. Effects of cement addition on arsenic leaching from soils excavated from projects employing shield-tunneling method. Geoderma 2021, 385, 114896. [Google Scholar] [CrossRef]
- Tabelin, C.B.; Park, I.; Phengsaart, T.; Jeon, S.; Villacorte-Tabelin, M.; Alonzo, D.; Yoo, K.; Ito, M.; Hiroyoshi, N. Copper and critical metals production from porphyry ores and E-wastes: A review of resource availability, processing/recycling challenges, socio-environmental aspects, and sustainability issues. Resour. Conserv. Recycl. 2021, 170, 105610. [Google Scholar] [CrossRef]
- Coussy, S.; Benzaazoua, M.; Blanc, D.; Moszkowicz, P.; Bussière, B. Arsenic stability in arsenopyrite-rich cemented paste backfills: A leaching test-based assessment. J. Hazard. Mater. 2011, 185, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Ni, W.; Zhang, Y.; Li, Y.; Shi, T.; Li, Z. Investigation into the semi-dynamic leaching characteristics of arsenic and antimony from solidified/stabilized tailings using metallurgical slag-based binders. J. Hazard. Mater. 2020, 381, 120992. [Google Scholar] [CrossRef] [PubMed]
- Jarošíková, A.; Ettler, V.; Mihaljevič, M.; Drahota, P.; Culka, A.; Racek, M. Characterization and pH-dependent environmental stability of arsenic trioxide-containing copper smelter flue dust. J. Environ. Manag. 2018, 209, 71–80. [Google Scholar] [CrossRef]
- Zhao, Z.; Wen, C.; Peng, B.; Liang, Y.; Jie-He, Y.; Yan, Z. Arsenic vitrification by copper slag based glass: Mechanism and stability studies. J. Non. Cryst. Solids 2017, 466–467, 21–28. [Google Scholar] [CrossRef]
- Shaw, J.K.; Fathordoobadi, S.; Zelinski, B.J.; Ela, W.P.; Saez, A.E. Stabilization of arsenic-bearing solid residuals in polymeric matrices. J. Hazard. Mater. 2008, 152, 1115–1121. [Google Scholar] [CrossRef] [PubMed]
- Tabelin, C.B.; Corpuz, R.D.; Igarashi, T.; Villacorte-Tabelin, M.; Ito, M.; Hiroyoshi, N. Hematite-catalysed scorodite formation as a novel arsenic immobilisation strategy under ambient conditions. Chemosphere 2019, 233, 946–953. [Google Scholar] [CrossRef] [PubMed]
- Park, I.; Ryota, T.; Yuto, T.; Tabelin, C.B.; Phengsaart, T.; Jeon, S.; Ito, M.; Hiroyoshi, N. A novel arsenic immobilization strategy via a two-step process: Arsenic concentration from dilute solution using schwertmannite and immobilization in Ca–Fe–AsO4 compounds. J. Environ. Manag. 2021, 295, 113052. [Google Scholar] [CrossRef]
- Li, Y.; Min, X.; Ke, Y.; Liu, D.; Tang, C. Preparation of red mud-based geopolymer materials from MSWI fly ash and red mud by mechanical activation. Waste Manag. 2019, 83, 202–208. [Google Scholar] [CrossRef]
- Li, Y.C.; Min, X.B.; Chai, L.Y.; Shi, M.Q.; Tang, C.J.; Wang, Q.W.; Liang, Y.; Lei, J.; Liyang, W.J. Co-treatment of gypsum sludge and Pb/Zn smelting slag for the solidification of sludge containing arsenic and heavy metals. J. Environ. Manag. 2016, 181, 756–761. [Google Scholar] [CrossRef]
- Liang, Y.; Min, X.; Chai, L.; Wang, M.; Liyang, W.; Pan, Q.; Okido, M. Stabilization of arsenic sludge with mechanochemically modified zero valent iron. Chemosphere 2017, 168, 1142–1151. [Google Scholar] [CrossRef] [PubMed]
- Peng, B.; Song, T.; Wang, T.; Chai, L.; Yang, W.; Li, X.; Li, C.; Wang, H. Facile synthesis of Fe3O4@Cu(OH)2 composites and their arsenic adsorption application. Chem. Eng. J. 2016, 299, 15–22. [Google Scholar] [CrossRef]
- Zhao, Z.; Song, Y.; Min, X.; Liang, Y.; Chai, L.; Shi, M. XPS and FTIR studies of sodium arsenate vitrification by cullet. J. Non. Cryst. Solids 2016, 452, 238–244. [Google Scholar] [CrossRef]
- Xu, H.; Min, X.; Wang, Y.; Ke, Y.; Yao, L.; Liu, D.; Chai, L. Stabilization of arsenic sulfide sludge by hydrothermal treatment. Hydrometallurgy 2020, 191, 105229. [Google Scholar] [CrossRef]
- Long, D.; Jia, J.; He, T.; Li, Y. Stabilization/solidification of arsenic sulfide residue and effect evaluation. Environ. Prot. Technol. 2014, 20, 7–11. [Google Scholar]
- Opiso, E.M.; Tabelin, C.B.; Maestre, C.V.; Aseniero, J.P.J.; Park, I.; Villacorte-Tabelin, M. Synthesis and characterization of coal fly ash and palm oil fuel ash modified artisanal and small-scale gold mine (ASGM) tailings based geopolymer using sugar mill lime sludge as Ca-based activator. Heliyon 2021, 7, e06654. [Google Scholar] [CrossRef]
- Barbieri, L.; Bonamartini, A.C.; Lancellotti, I. Alkaline and alkaline-earth silicate glasses and glass-ceramicsfrom municipal and industrial wastes. J. Eur. Ceram. Soc. 2000, 20, 2477–2483. [Google Scholar] [CrossRef]
- Basegio, T.; Beck, L.A.P.; Bernardes, A.M.; Bergmann, C.P. Vitrification: An alternative to minimize environmental impact caused byleather industry wastes. J. Hazard. Mater. 2009, 165, 604–611. [Google Scholar] [CrossRef]
- Colombo, P.; Brusatin, G.; Bernardo, E.; Scarinci, G. Inertization and reuse of waste materials by vitrification and fabrication of glass-based products. Curr. Opin. Solid State Mater. Sci. 2003, 7, 225–239. [Google Scholar] [CrossRef]
- Park, K.; Hyun, J.; Maken, S.; Jang, S.; Park, J.W. Vitrification of municipal solid waste incinerator fly ash using Brown’s gas. Energy Fuels 2005, 19, 258–262. [Google Scholar] [CrossRef]
- Pelino, M.; Karamanov, A.; Pisciella, P.; Crisucci, S.; Zonetti, D. Vitrification of Electric Arc Furnace Dusts. Waste Manag. 2002, 22, 945–949. [Google Scholar] [CrossRef]
- Mahandra, H.; Wu, C.; Ghahreman, A. Leaching characteristics and stability assessment of sequestered arsenic in flue dust based glass. Chemosphere 2021, 276, 130173. [Google Scholar] [CrossRef] [PubMed]
- Lalancette, J.M.; Lemieux, D.; Nasrallah, K.; Curiel, G.G.; Barbaroux, R. Method For Vitrification of Arsenic And Antimony. U.S. 9981295 B2, 29 May 2018. [Google Scholar]
- GlassLock, Process™. Dundee Sustainable Technologies (DST), Canada. 2020. [WWW Document]. Available online: http://dundeetechnologies.com/glasslock-process (accessed on 12 January 2022).
- Clavier, K.A.; Liu, Y.; Intrakamhaeng, V.; Townsend, T.G. Re-evaluating the TCLP’s role as the regulatory driver in the management of municipal solid waste incinerator Ash. Environ. Sci. Technol. 2019, 53, 7964–7973. [Google Scholar] [CrossRef] [PubMed]
- Clavier, K.A.; Watts, B.; Liu, Y.; Ferraro, C.C.; Townsend, T.G. Risk and performance assessment of cement made using municipal solid waste incinerator bottom ash as a cement kiln feed. Resour. Conserv. Recycl. 2019, 160, 270–279. [Google Scholar] [CrossRef]
- Coussy, S.; Benzaazoua, M.; Blanc, D.; Moszkowicz, P.; Bussière, B. Assessment of arsenic immobilization in synthetically prepared cemented paste backfill specimens. J. Environ. Manag. 2012, 93, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Kendall, D.S. Toxicity Characteristic Leaching Procedure and iron treatment of brass foundry waste. Environ. Sci. Technol. 2003, 37, 367–371. [Google Scholar] [CrossRef]
- Li, J.S.; Beiyuan, J.; Tsang, D.C.W.; Wang, L.; Poon, C.S.; Li, X.D.; Fendorf, S. Arsenic- containing soil from geogenic source in Hong Kong: Leaching characteristics and stabilization/solidification. Chemosphere 2017, 182, 31–39. [Google Scholar] [CrossRef]
- Lincoln, J.D.; Ogunseitan, O.A.; Shapiro, A.A.; Saphores, J.D.M. Leaching assessments of hazardous materials in cellular telephones. Environ. Sci. Technol. 2007, 41, 2572–2578. [Google Scholar] [CrossRef]
- Sanchez, F.; Gervais, C.; Garrabrants, A.C.; Barna, R.; Kosson, D.S. Leaching of inorganic contaminants from cement-based waste materials as a result of carbonation during intermittent wetting. Waste Manag. 2002, 22, 249–260. [Google Scholar] [CrossRef]
- Shakya, A.K.; Ghosh, P.K. Stability against arsenic leaching from biogenic arsenosulphides generated under reduced environment. J. Clean. Prod. 2019, 208, 1557–1562. [Google Scholar] [CrossRef]
- USEPA. The Arsenic Rule: Water Treatment Plant Residuals, Issues in Management and Disposal. 2020; [WWW Document]. Available online: https://www.epa.gov/sites/default/files/2015-09/documents/train6-residuals.pdf (accessed on 17 December 2021).
- USEPA. The Arsenic Rule—Waste Disposal Options. 2020; [WWW Document]. Available online: https://www.epa.gov/sites/production/files/2015-09/documents/disposal_options_janet_cherry.pdf (accessed on 17 December 2021).
- USEPA. SW-846 Test Method 1315: Mass Transfer Rates of Constituents in Monolithic or Compacted Granular Materials Using a Semi-Dynamic Tank Leaching Procedure. 2017; [WWW Document]. Available online: https://www.epa.gov/hw-sw846/sw-846-test-method-1315-mass-transfer-rates-constituents-monolithic-or-compacted-granular (accessed on 15 September 2021).
- USEPA. SW-846 Test Method 1312: Synthetic Precipitation Leaching Procedure. 1994; [WWW Document]. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/1312.pdf (accessed on 15 September 2021).
- USEPA. SW-846 Test Method 1311: Toxicity Characteristic Leaching Procedure. 1992; [WWW Document]. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/1311.pdf (accessed on 15 September 2021).
- Aus 4439.3. Wastes, Sediments and Contaminated Soils—Part 3. 2019. Available online: https://www.standards.org.au/standards-catalogue/sa-snz/manufacturing/ch-035/as--4439-dot-3-colon-2019 (accessed on 15 September 2021).
- Zhang, S.; Dai, S.; Finkelman, R.B.; Graham, I.T.; French, D.; Hower, J.C.; Li, X. Leaching characteristics of alkaline coal combustion by-products: A case study from a coal-fired power plant, Hebei Province, China. Fuel 2019, 255, 115710. [Google Scholar] [CrossRef]
- Galindo, C.; Mougin, L.; Nourreddine, A. An improved radiochemical separation of uranium and thorium in environmental samples involving peroxide fusion. Appl. Radiat. Isot. 2007, 65, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Vandecasteele, C.; Dutre, V.; Geysen, D.; Wauters, G. Solidification/stabilization of arsenic bearing fly ash from the metallurgical industry. Immobilisation mechanism of arsenic. Waste Manag. 2002, 22, 143–146. [Google Scholar] [CrossRef]
- Toit, G.; van der Merwe, E.M.; Kruger, R.A.; McDonald, J.M.; Kearsley, E.P. Characterisation of the Hydration Products of a Chemically and Mechanically Activated High Coal Fly Ash Hybrid Cement. Minerals 2022, 12, 57. [Google Scholar] [CrossRef]
- Hanum, F.F.; Desfitri, E.R.; Hayakawa, Y.; Kambara, S. Preliminary Study on Additives for Controlling As, Se, B, and F Leaching from Coal Fly Ash. Minerals 2018, 8, 493. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Min, X.; Ke, Y.; Fei, J.; Liu, D.; Tang, C. Immobilization potential and immobilization mechanism of arsenic in cemented paste backfill. Miner. Eng. 2019, 138, 101–107. [Google Scholar] [CrossRef]
- Liu, W.; Li, Z.; Han, J.; Li, W.; Wand, X.; Wang, N.; Qin, W. Selective Separation of Arsenic from Lead Smelter Flue Dust by Alkaline Pressure Oxidative Leaching. Minerals 2019, 9, 308. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Yuan, Z.; Wang, S.; Jia, Y.; Demopoulos, G.P. Incorporation of arsenic into gypsum: Relevant to arsenic removal and immobilization process in hydrometallurgical industry. J. Hazard. Mater. 2015, 300, 272–280. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).