Recent Ostracod Fauna of the Western Ross Sea (Antarctica): A Poorly Known Ingredient of Polar Carbonate Factories
Abstract
1. Introduction
- Characterize the ostracod assemblages from the WRSS, taking into account the level of undiscovered commonness, richness and abundance;
- Explore possible differences among ostracod assemblages in respect to bathymetric zones within the Ross Sea (i.e., inner and outer shelf, shelf break);
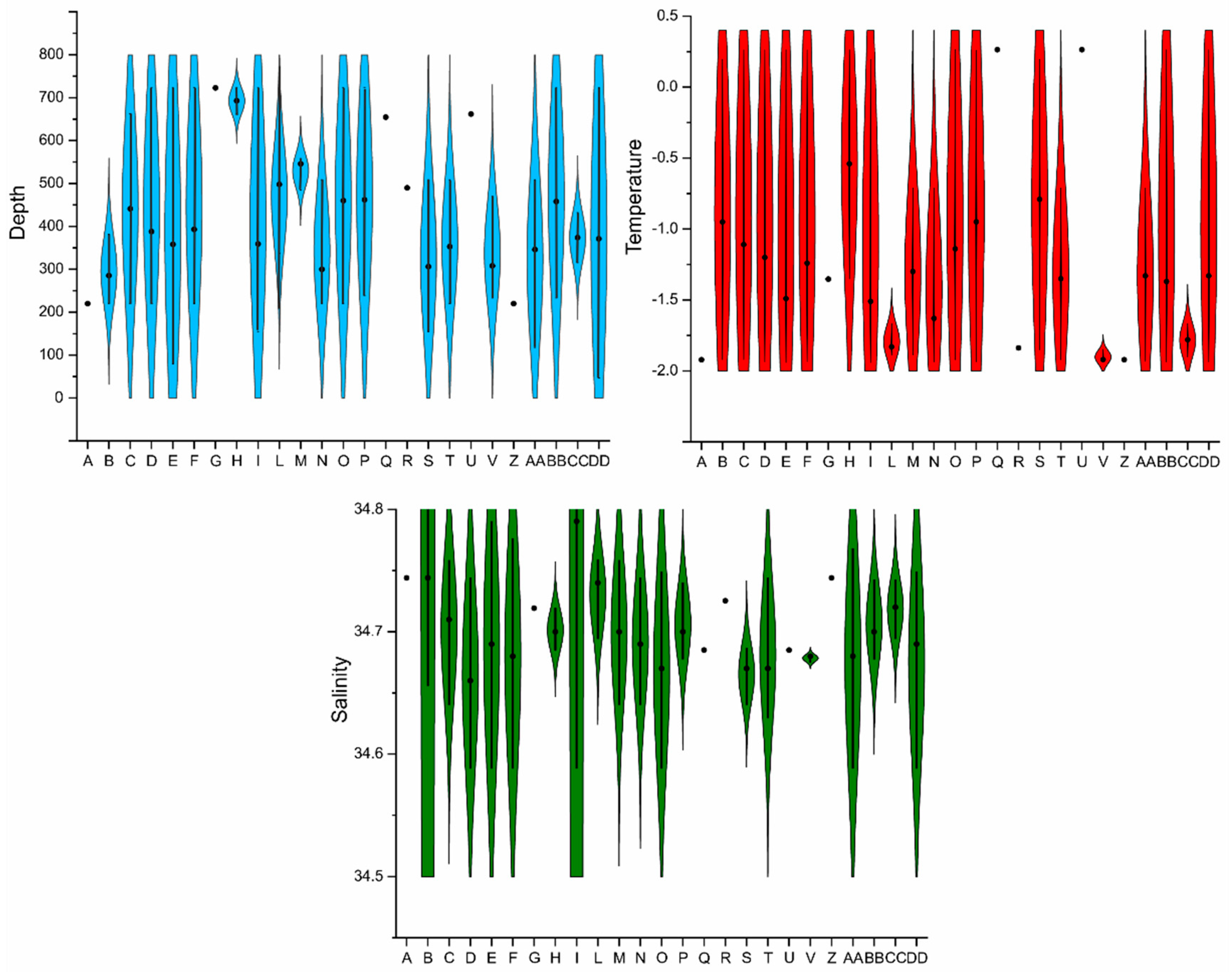
- Investigate which environmental parameters (depth, salinity and temperature) most affect ostracod assemblages to increase our understanding of the relationships between benthic ecosystems and oceanographic regimes in the Antarctic region.
2. Materials and Methods
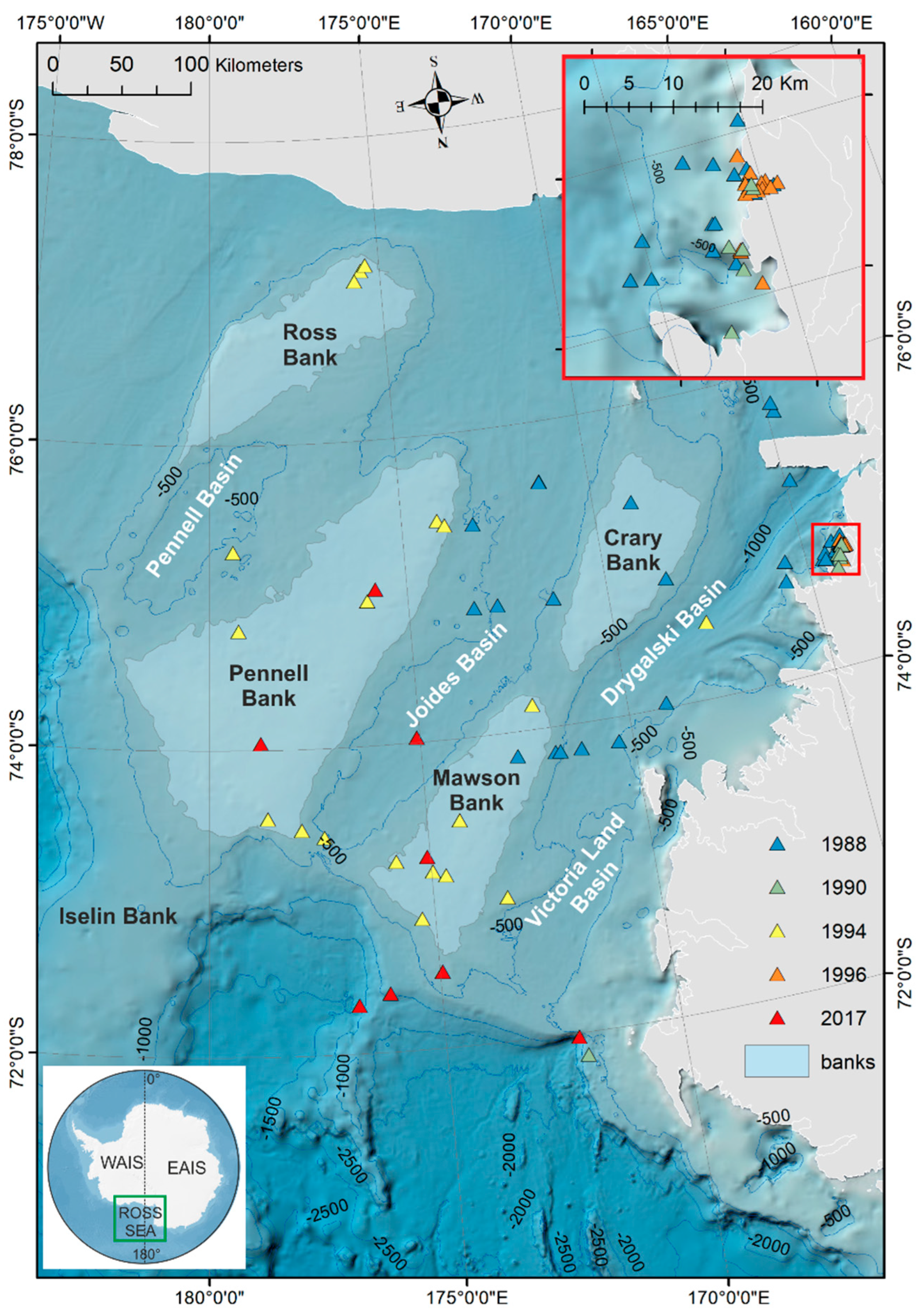
2.1. Study Area—Regional and Oceanographic Setting
2.2. Sediment of the Western Ross Sea Shelf
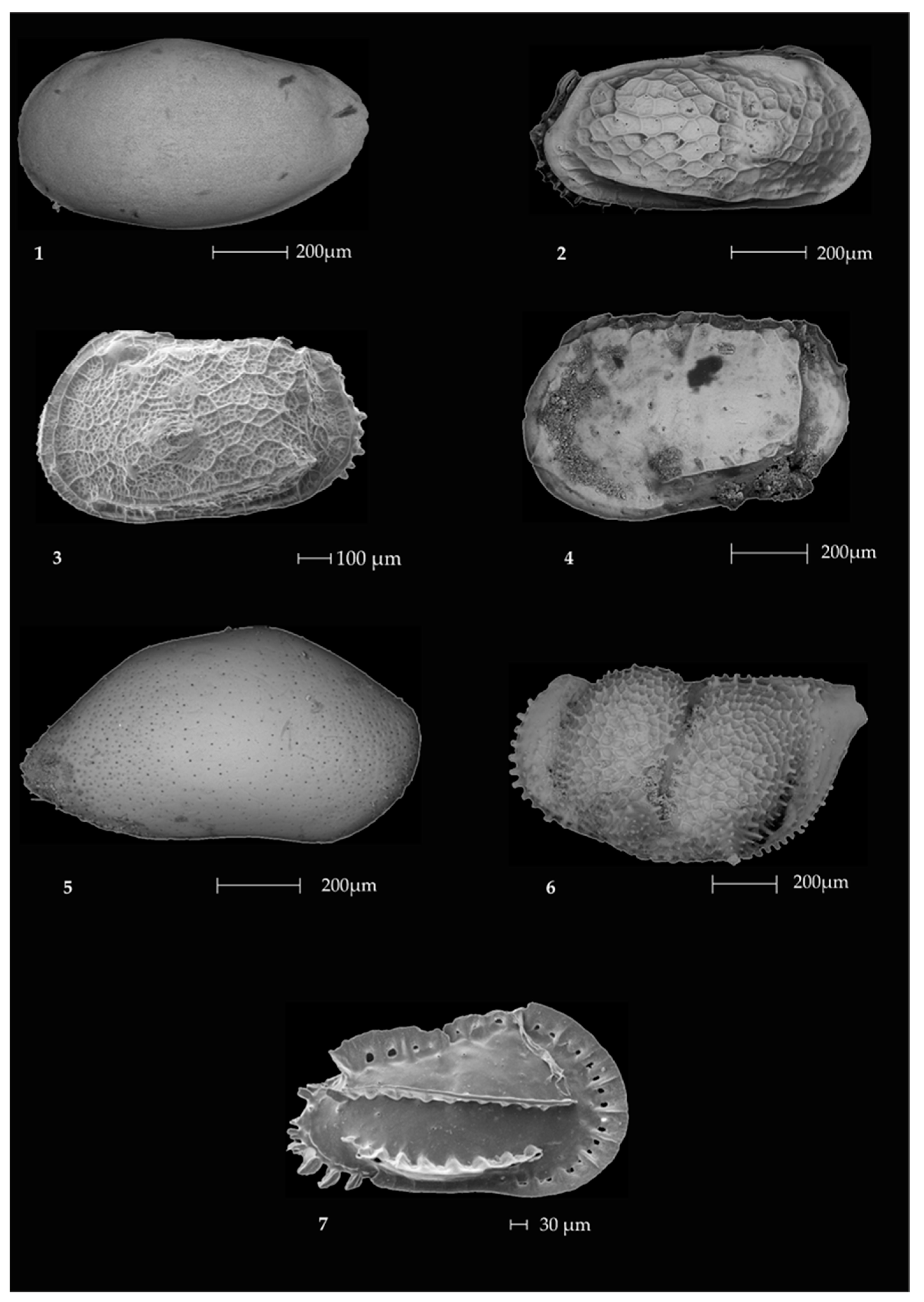
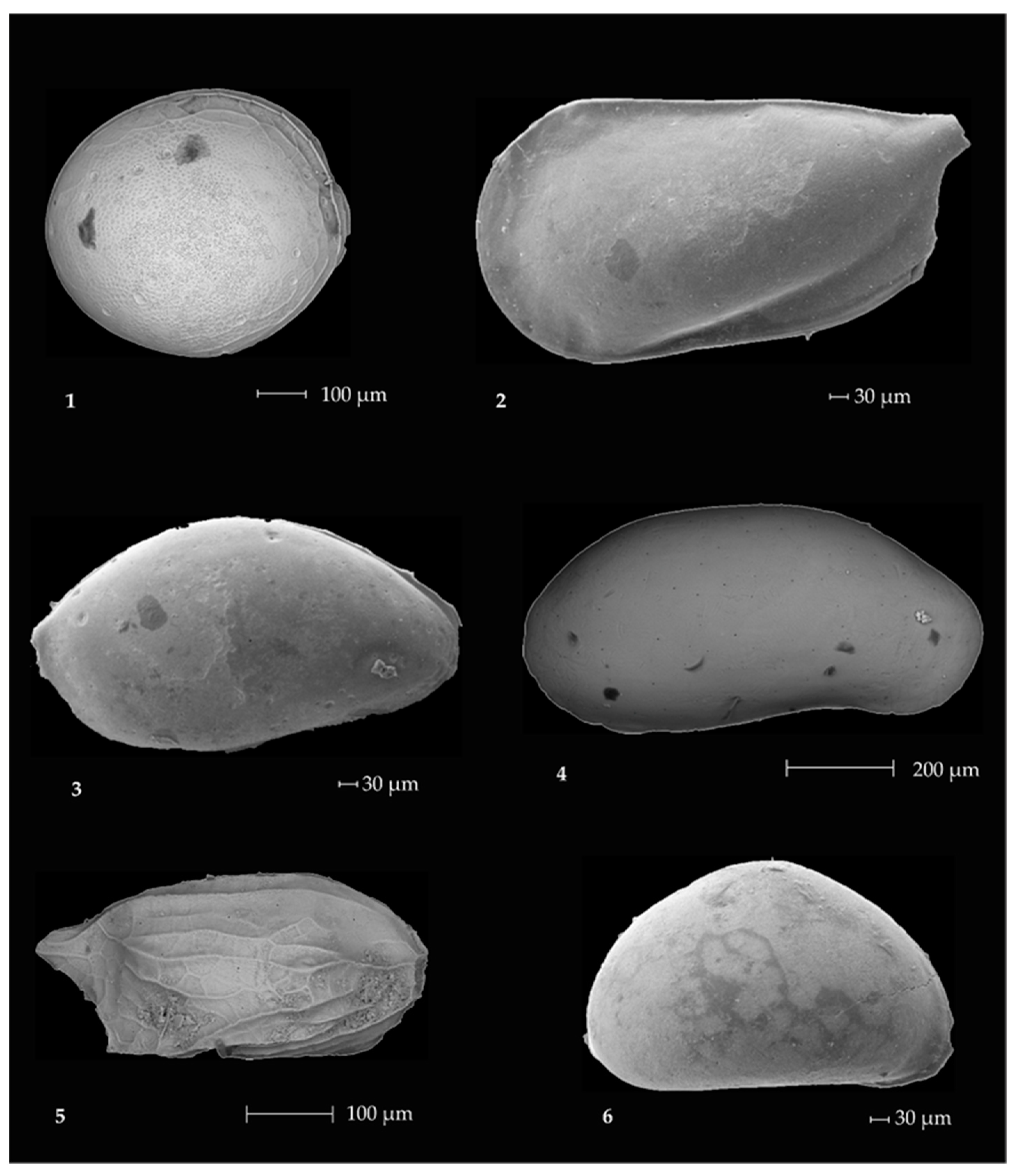
2.3. Ostracods
2.4. Statistical Analysis
2.5. GIS Analysis
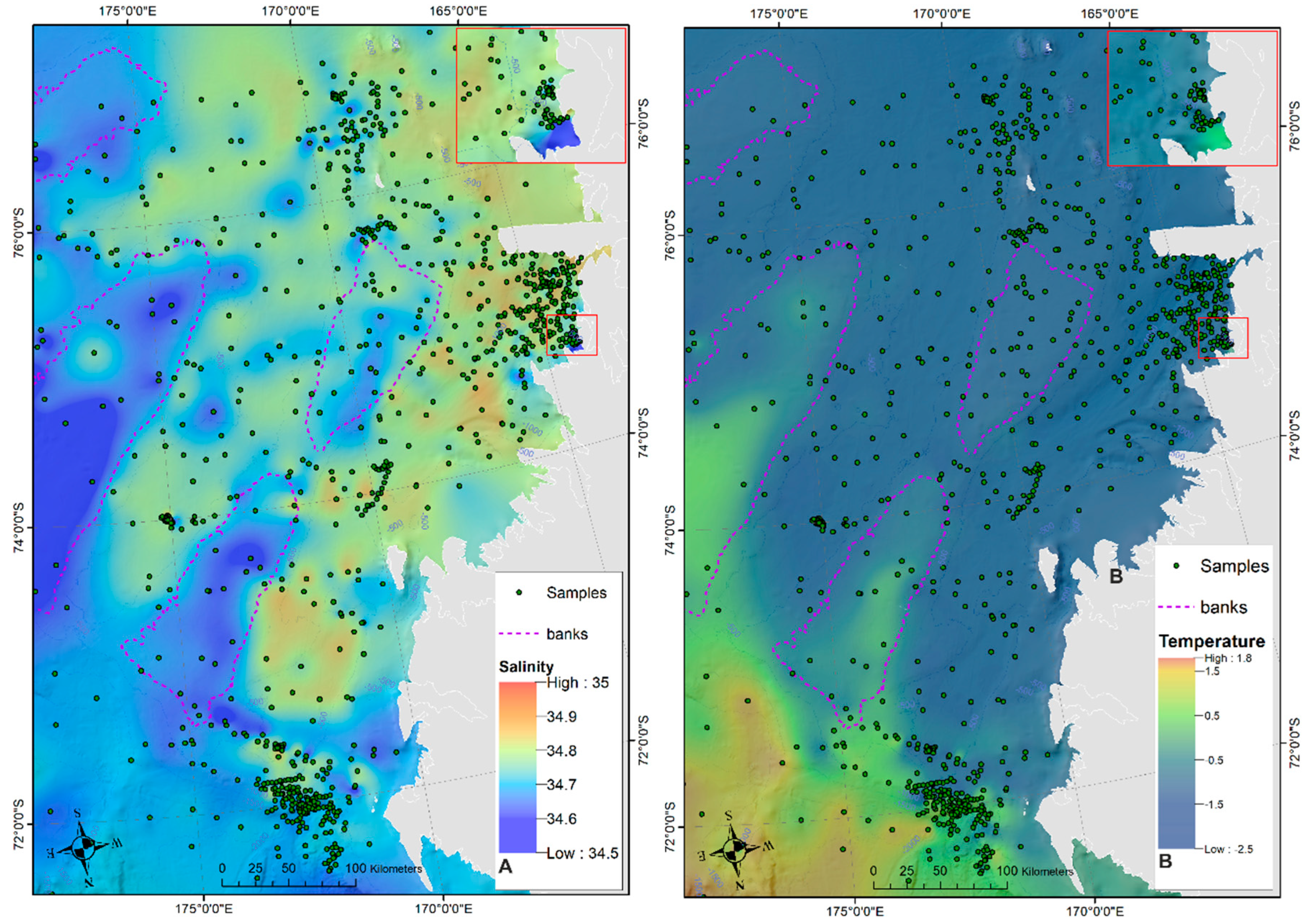
2.6. Oceanographic Data
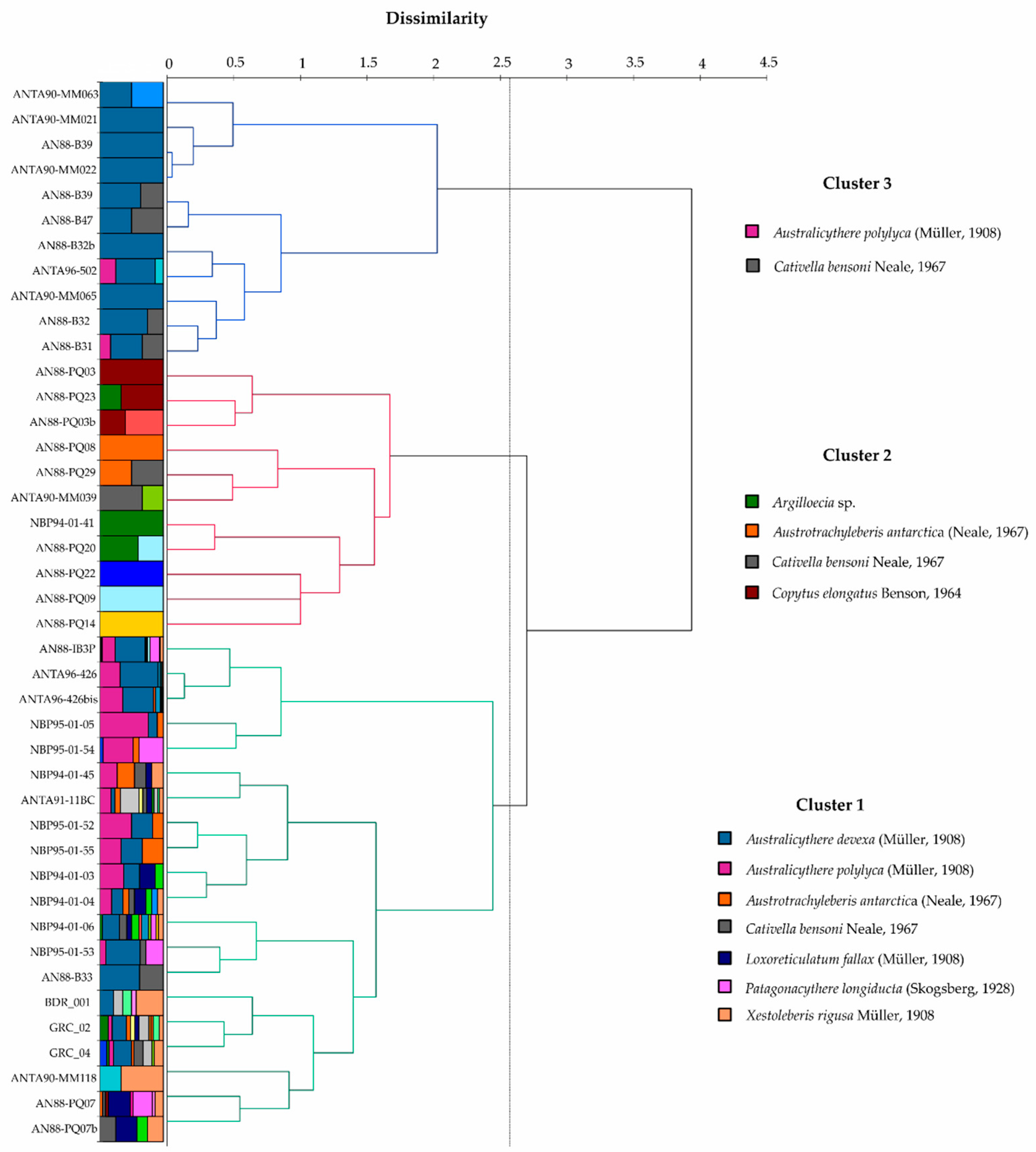
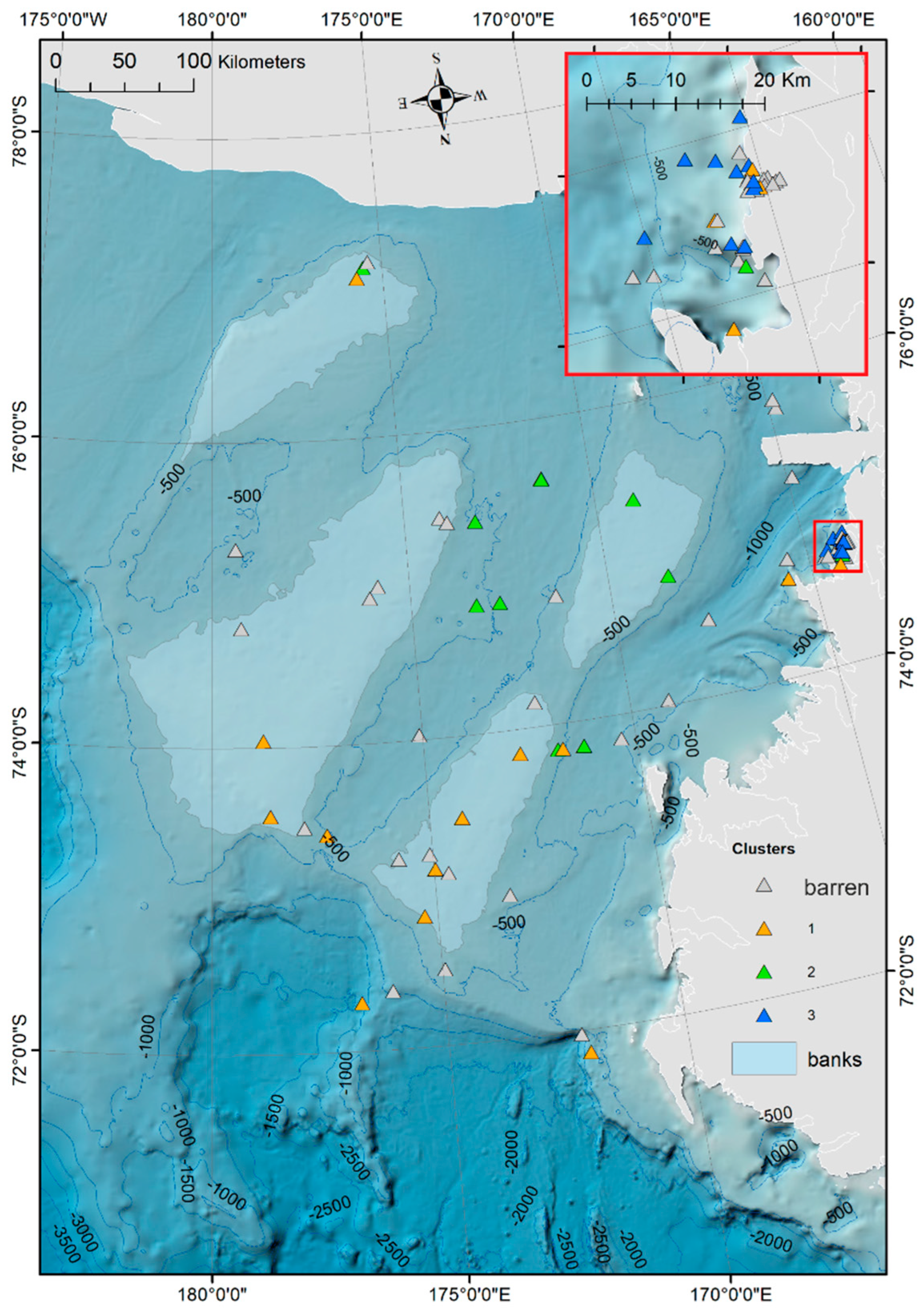
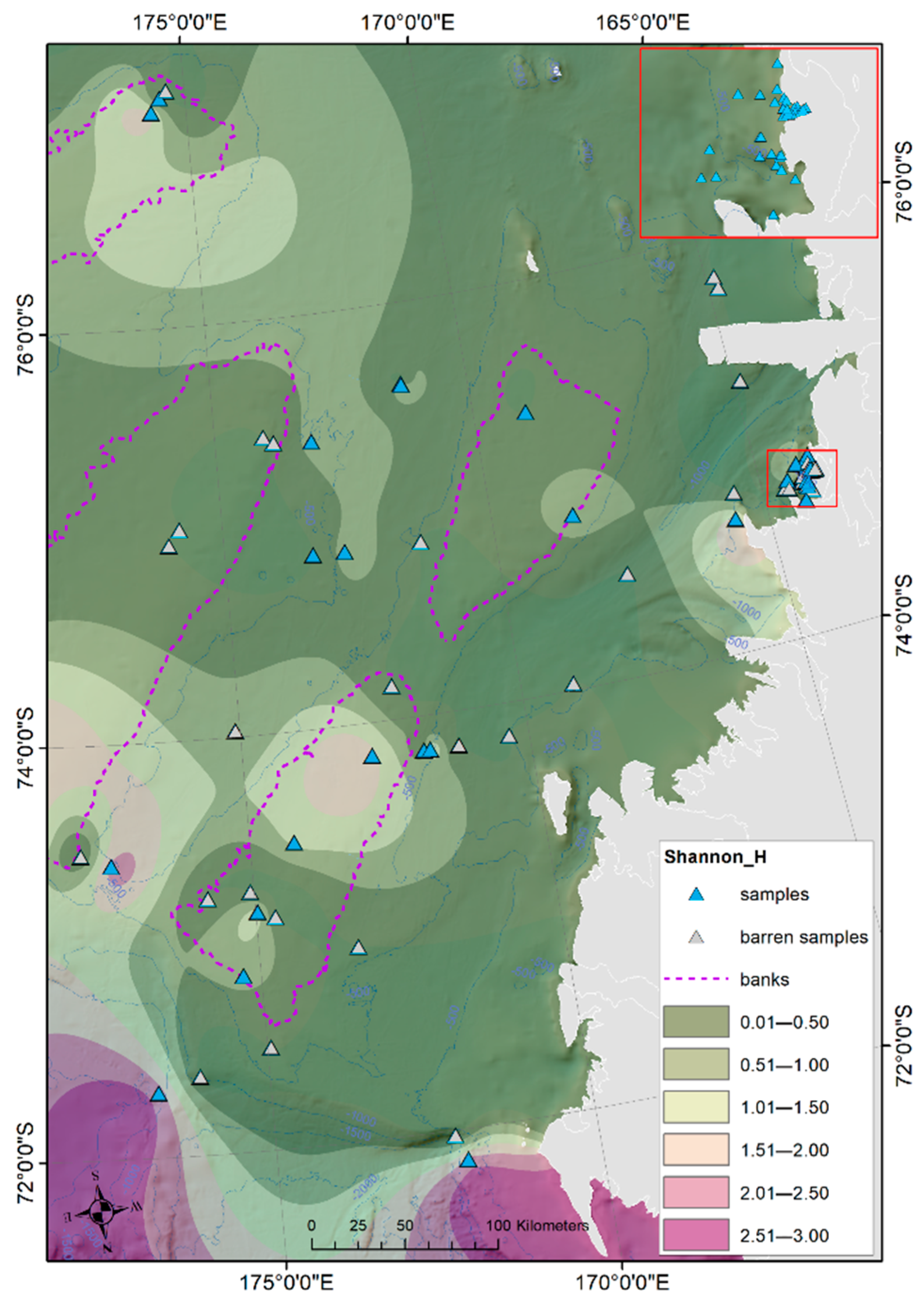
3. Results
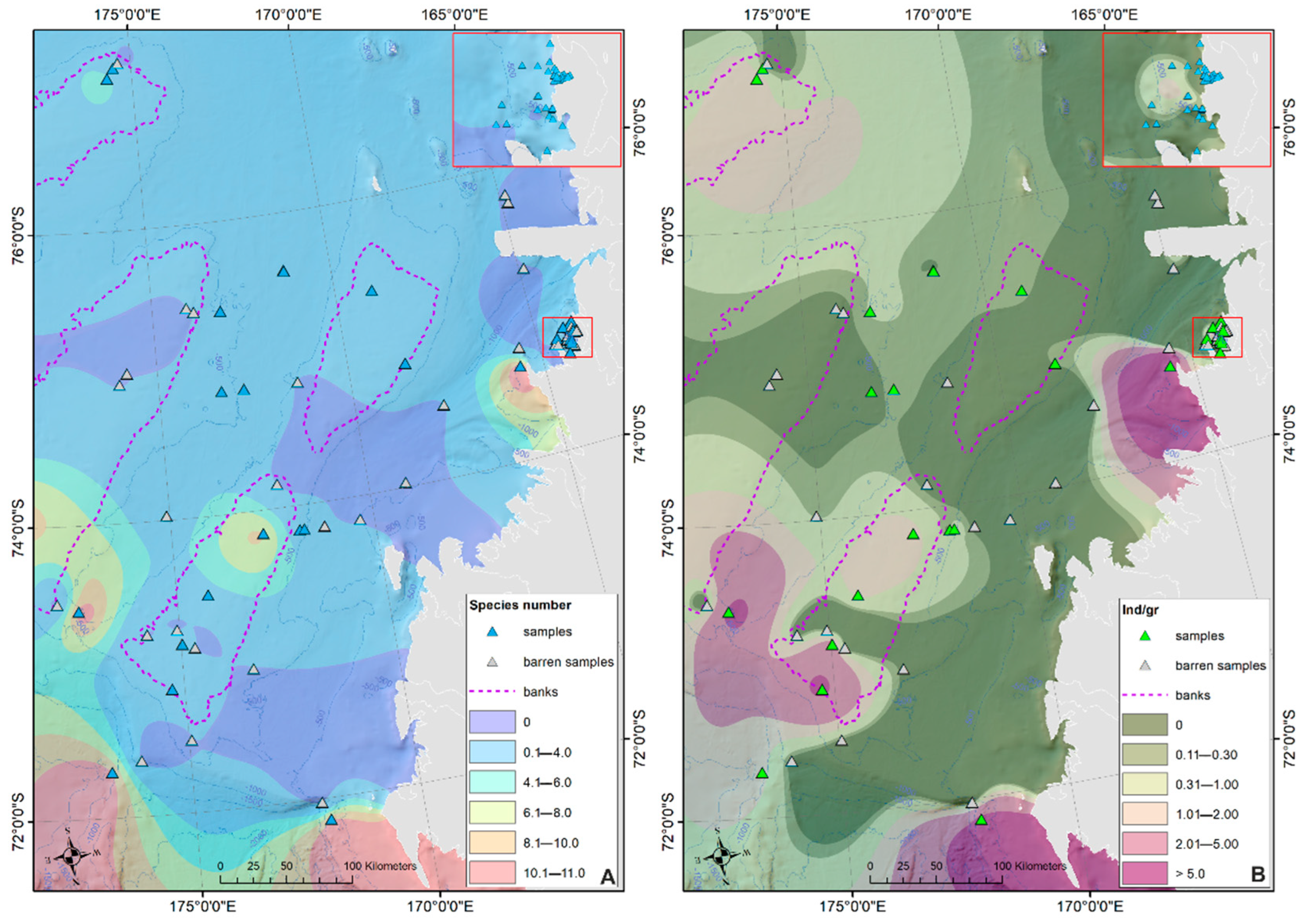
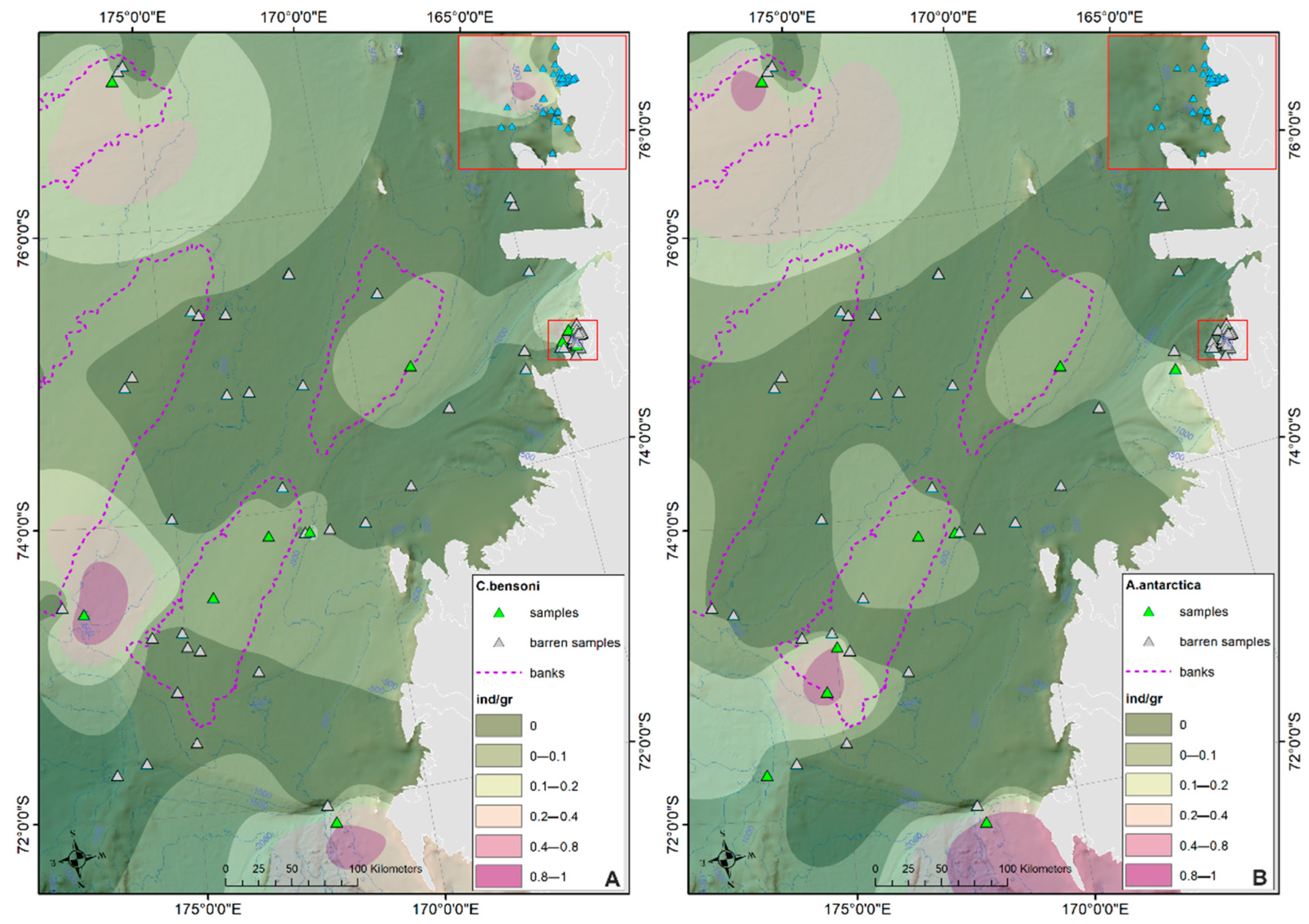
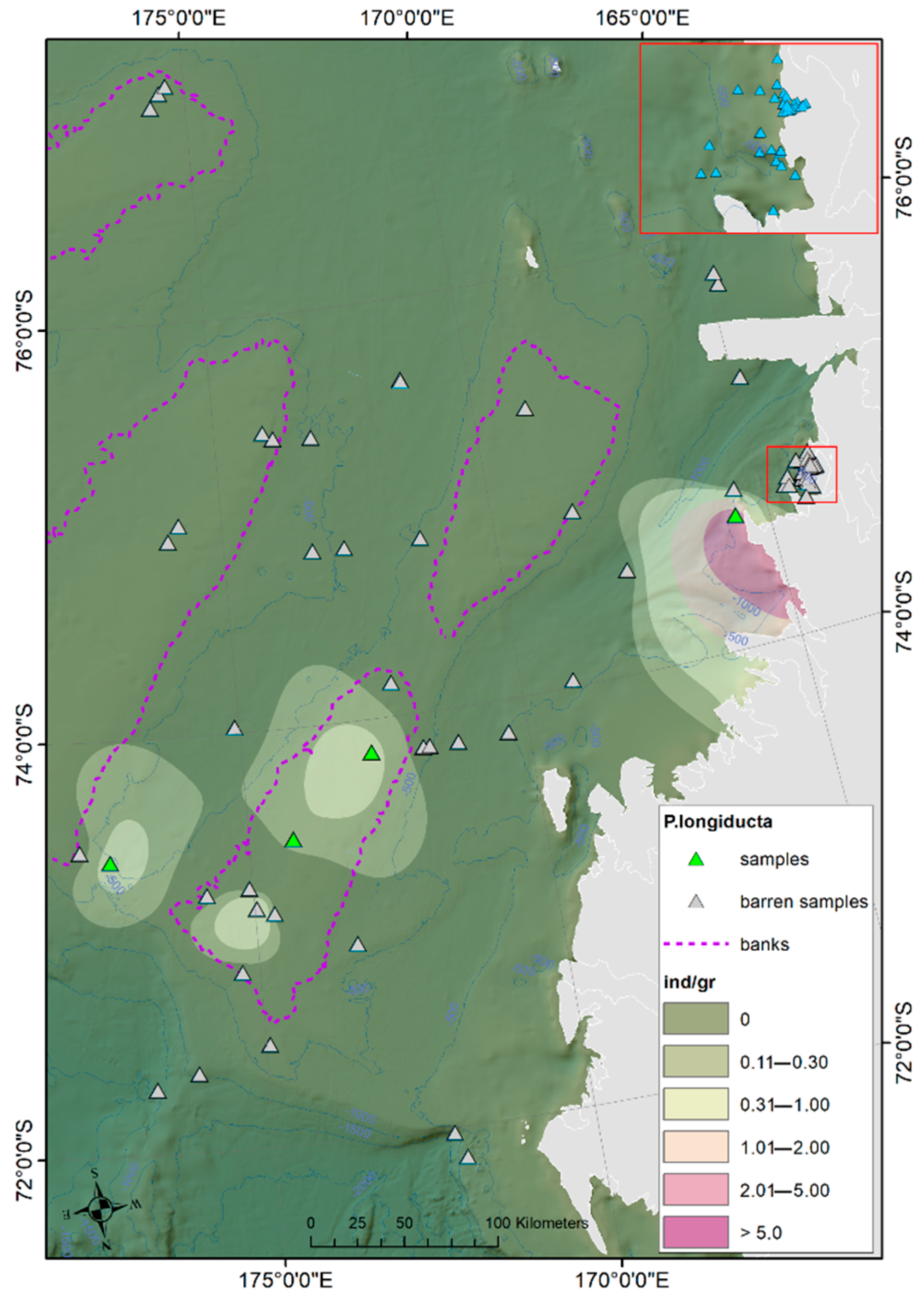
Ostracod Assemblages in the Western Ross Sea Shelf
4. Discussion
4.1. Analysis of the Ostracod Assemblages in Respect to WRSS Bathymetric Zones
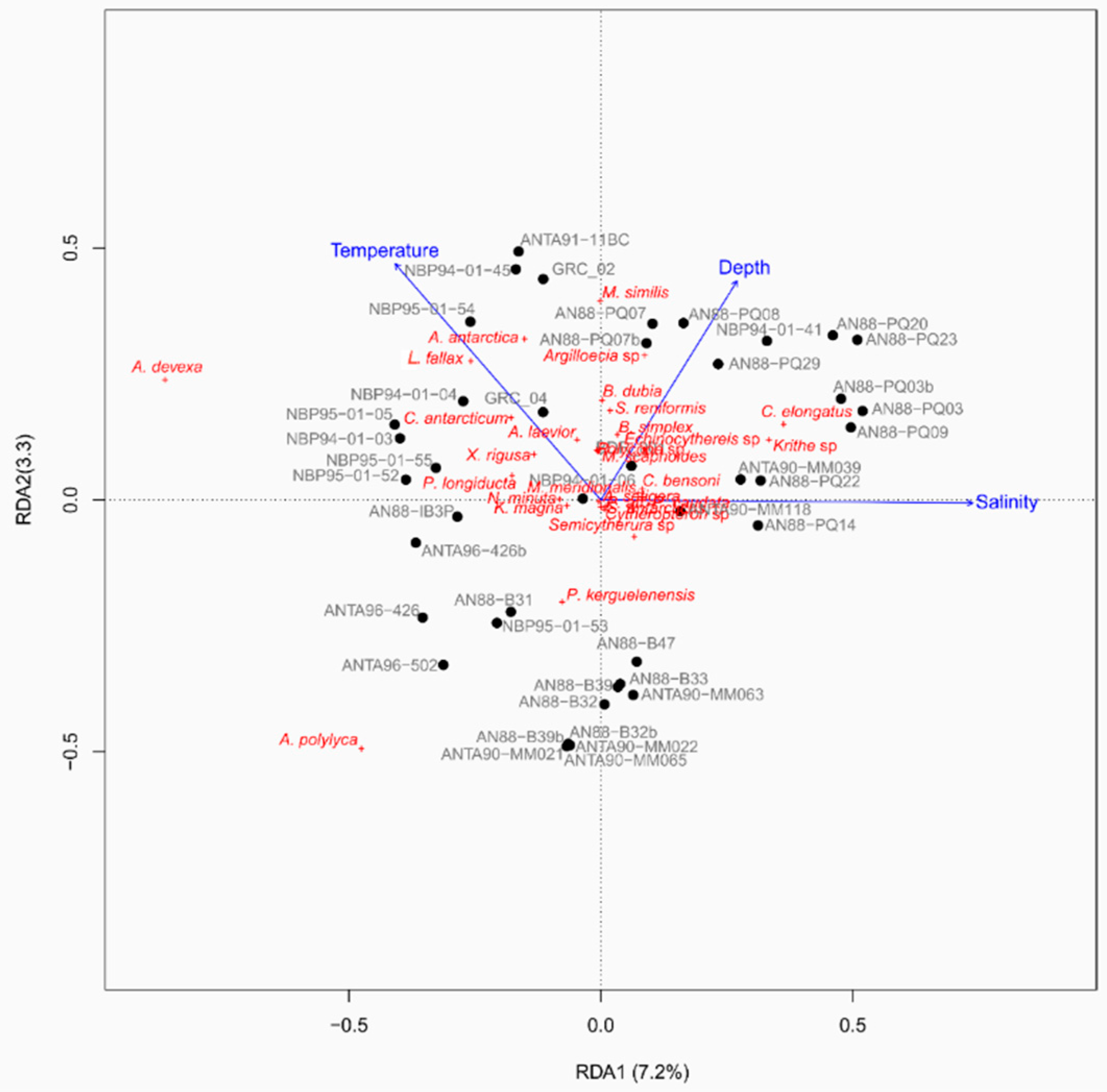
4.2. Distribution of Autochthonous Ostracod Assemblages in Response to the Oceanographic Settings
5. Conclusions
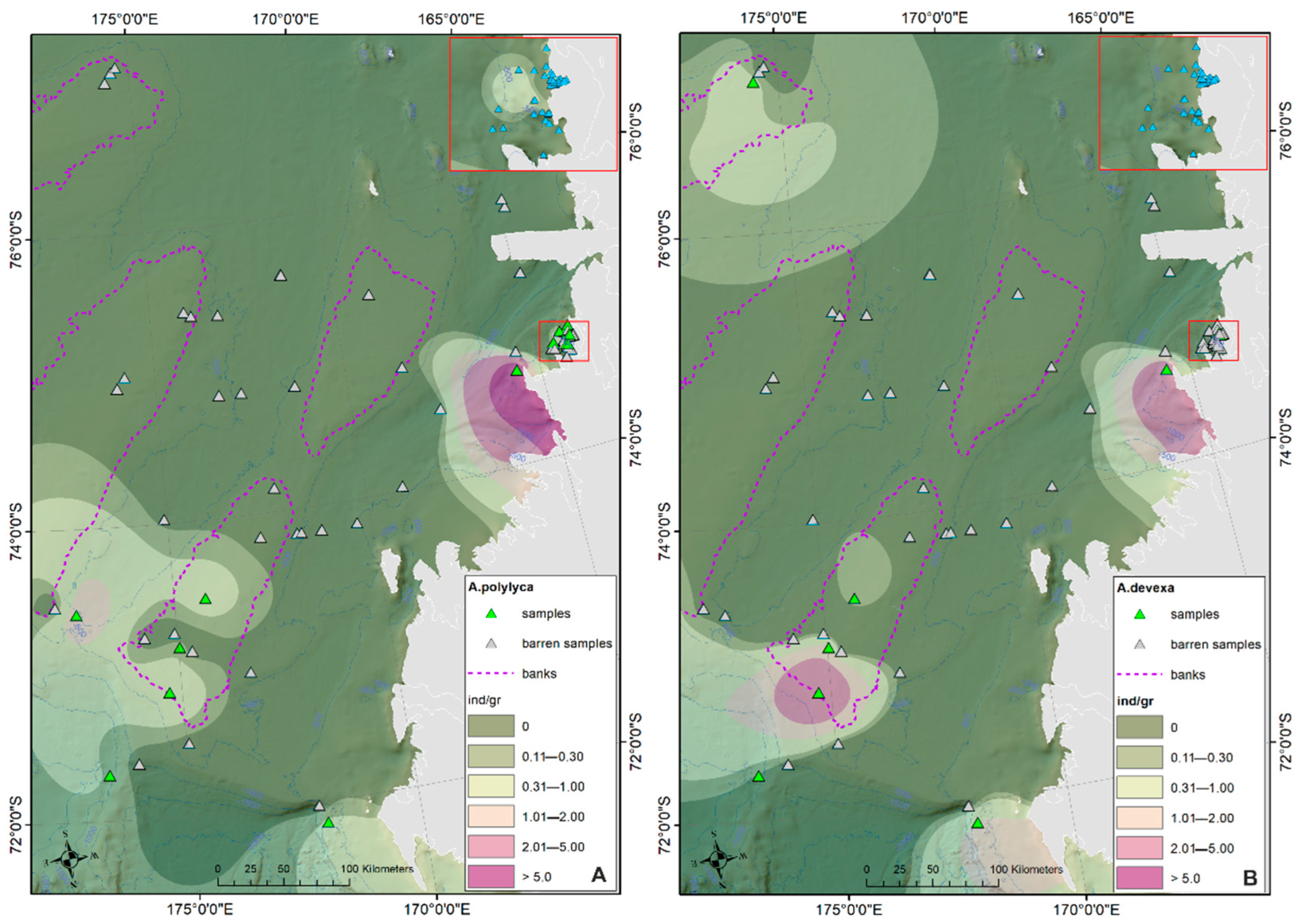
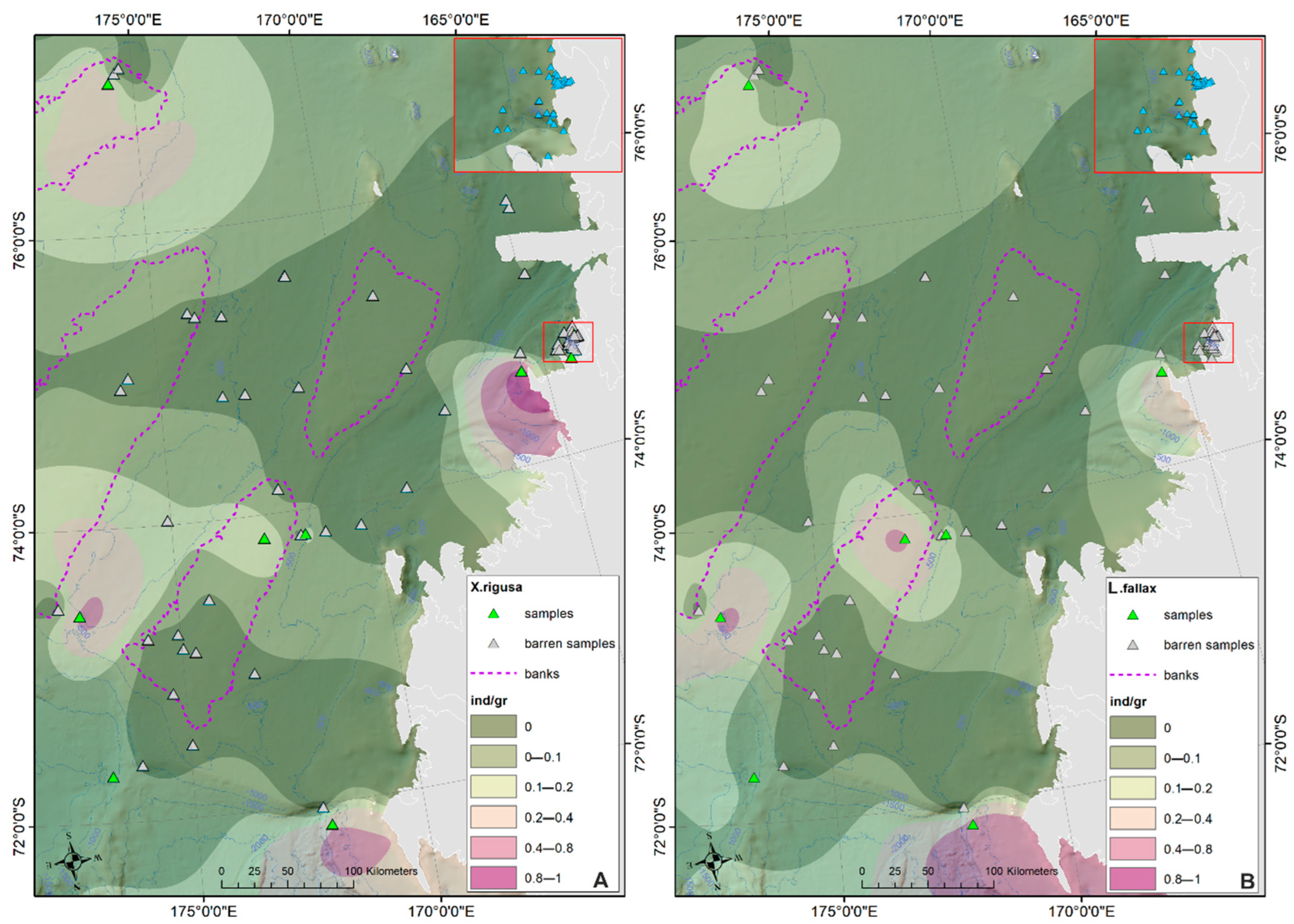
- The prevalent ostracod assemblages in WRSS are represented by Australicythere polylyca, Australicythere devexa, Xestoleberis rigusa, Loxoreticulatum fallax, Cativella bensoni, Austrotrachyleberis antarctica and Patagonacythere longiducta; all these species are able to colonize different shelf environments along an extensive range of water depths. These characteristics may have served as a potential source for shelf recolonization after the last glacial phase.
- Salinity and temperature are the oceanographic variables that better explain the main variance in the examined samples; in particular, the dominant ostracod species are positively correlated with temperature, outlining a possible close link between diffusion of the ostracod assemblages and CDW (a relatively warm water mass that flows onto the Ross Sea shelf at intermediate and surface levels), as a major factor controlling the nutrient intakes within the Ross Sea.
- Rising values of the ostracod assemblages were also recorded in and particularly north of the Terra Nova Bay, where nutrient enrichment is derived from warm water of circumpolar origin (Circumpolar Deep water—CDW) flowing from the open ocean southwards onto the continental shelf. Recent analyses have shown an increase in primary production supported by biogenic iron in the same area.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

References
- Yamaguchi, T. Valve calcification in the evolutionary history of marine ostracodes (Ostracoda). J. Crustacean Biol. 2018, 39, 253–260. [Google Scholar] [CrossRef]
- Benson, R.H. Ostracods and Palaeoceanography. Ostracoda in the Earth Sciences. In Proceedings of the 11th International Symposium on Ostracoda, Warrnambool, Australia, 8–12 July 1991; De Deckker, P., Colin, J.P., Peypouquet, J.P., Eds.; A.A. Balkema: Rotterdam, The Netherlands, 1988; pp. 1–26. [Google Scholar]
- Cronin, T.M.; Holtz, T.R.; Whatley, R.C. Quaternary paleoceanography of the deep Arctic Ocean based on quantitative analysis of Ostracoda. Mar. Geol 1994, 119, 305–332. [Google Scholar] [CrossRef]
- Cronin, T.M.; De Martino, D.M.; Dwyer, G.S.; Rodriguez-Lazaro, J. Deep-sea ostracode species diversity: Response to late Quaternary climate change. Mar. Micropaleontol. 1999, 37, 231–249. [Google Scholar] [CrossRef]
- Yasuhara, M.; Cronin, T.M.; Hunt, G.; Hodell, D.A. Deep–sea ostracods from the South Atlantic sector of the Southern Ocean during the Last 370,000 years. J. Paleontol. 2009, 83, 914–930. [Google Scholar] [CrossRef]
- Rodriguez-Lazaro, J.; Ruiz-Munõz, F. A General Introduction to Ostracods: Morphology, Distribution, Fossil Record and Applications. Dev. Quat. Sci. 2012, 17, 1–14. [Google Scholar] [CrossRef]
- Cronin, T.M.; Raymo, M.E. Orbital forcing of deep-sea benthic species diversity. Nature 1997, 385, 624–627. [Google Scholar] [CrossRef]
- Cronin, T.; Dwyer, G.; Baker, P.A.; Rodriguez Lazaro, J.; Demartino, D.M. Orbital and suborbital variability in North Atlantic bottom water temperature obtained from deep-sea ostracode Mg/Ca ratios. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2000, 162, 45–57. [Google Scholar] [CrossRef]
- Cronin, T.M.; Dwyer, G.S.; Farmer, J.; Bauch, H.A.; Spielhagen, R.F.; Jakobsson, M.; Nilsson, J.; Briggs, W.M., Jr.; Stepanova, A. Deep Arctic Ocean warming during the last glacial cycle. Nat. Geosci. 2012, 5, 631–634. [Google Scholar] [CrossRef]
- Rodriguez Lazaro, J.; Cronin, T. Quaternary glacial and deglacial Krithe (Ostracoda) in the thermocline of the Little Bahama Bank (NW Atlantic): Palaeoceanographic implications. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1999, 152, 339–364. [Google Scholar] [CrossRef]
- Didié, C.; Bauch, H.A.; Helmke, J.P. Late Quaternary deep-sea ostracodes in the polar and subpolar North Atlantic: Paleoecological and paleoenvironmental implications. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2002, 184, 195–212. [Google Scholar] [CrossRef]
- Dwyer, G.S.; Cronin, T.M.; Baker, P.A.; Rodriguez Lazaro, J. Changes in North Atlantic deep-sea temperature during climatic fluctuations of the last 25,000 years based on ostracode Mg/Ca ratios. Geochem. Geophys. Geosyst. 2000, 1, 17. [Google Scholar] [CrossRef]
- Bodergat, A.M. Les Ostracodes, témoins de leur environnement: Approche chimique et écologique en milieu lagunaire et océanique. Doc. Lab. Géol. Fac. Sci. Lyon. 1983, 88, 1–246. [Google Scholar]
- De Deckker, P. Ostracod paleoecology. The ostracoda: Application in quaternary research. Am. Geophys. Union Wash. 2002, 131, 121–134. [Google Scholar]
- Decrouy, L. Biological and environmental controls on isotopes in ostracod shells. In Ostracoda as Proxies for Quaternary Climate Change. Developments in Quaternary Science; Horne, D.J., Holmes, J., Rodriguez-Lazaro, J., Viehberg, F.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2012; Volume 17, pp. 165–181. [Google Scholar] [CrossRef]
- Dingle, R.V. Insular endemism in Recent Southern Ocean benthic Ostracoda from Marion Island: Paleozoogeographical and evolutionary implications. Rev. Esp. Micropaleontol. 2002, 34, 215–233. [Google Scholar]
- Dingle, R.V. Recent Subantarctic benthic ostracod faunas from the Marion and Prince Edward Islands Archipelago, Southern Ocean. Rev. Esp. Micropaleontol. 2003, 35, 119–155. [Google Scholar]
- Kaiser, S.; Griffiths, H.J.; Barnes, D.K.A.; Brandão, S.N.; Brandt, A.; O’Brien, P.E. Is there a distinct continental slope fauna in the Antarctic? Deep Sea Res. Part II Top. Stud. Oceanogr. 2011, 58, 91–104. [Google Scholar] [CrossRef]
- Brandão, S.N.; Vital, H.; Brandt, A. Southern Polar Front macroecological and biogeographical insights gained from benthic Ostracoda. Deep Sea Res. Part II Top. Stud. Oceanogr. 2014, 108, 33–50. [Google Scholar] [CrossRef]
- Ayress, M.; Neil, H.; Passlow, V.; Swanson, K.M. Benthonic ostracods and deep watermasses: A qualitative comparison of southwest Pacific, southern and Atlantic Oceans. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1997, 131, 287–302. [Google Scholar] [CrossRef]
- Brandão, S.N.; Horne, D.V. The Platycopid Signal of oxygen depletion in the ocean: A critical evaluation of the evidence from modern ostracod biology, ecology and depth distribution. Paleogeogr. Paleoclimatol. Paleoecol. 2009, 283, 126–133. [Google Scholar] [CrossRef]
- Brandão, S.N.; Dingle, R.V.; De Broyer, C. Biogeographic Atlas of the Southern Ocean; Koubbi, P., Griffiths, H.J., Raymond, B., d’Udekem d’Acoz, C., Van de Putte, A.P., Danis, B., David, B., Grant., S., Gutt, J., Eds.; Scientific Committee on Antarctic Research: Cambridge, UK, 2014; pp. 142–148. [Google Scholar]
- Brandt, A.; Gooday, A.; Brandão, S.; Brix, S.; Brökeland, W.; Cedhagen, T.; Choudhury, M.; Cornelius, N.; Danis, B.; De Mesel, I.; et al. First insights into the biodiversity and biogeography of the Southern Ocean deep sea. Nature 2007, 447, 307–311. [Google Scholar] [CrossRef]
- Majewski, W.; Olempska, E. Recent ostracods from Admiralty Bay, King George Island, West Antarctica. Pol. Polar Res. 2005, 26, 13–36. [Google Scholar]
- Yasuhara, M.; Kato, M.; Ikeya, N.; Seto, K. Modern benthic ostracodes from Lützow-Holm Bay, East Antarctica: Paleoceanographic, paleobiogeographic, and evolutionary significance. Micropaleontology 2007, 53, 469–496. [Google Scholar] [CrossRef]
- Brandão, S.N.; Saeedi, H.; Brandt, A. Macroecology of Southern Ocean benthic Ostracoda (Crustacea) from the continental margin and abyss. Zool. J. Linn. Soc. 2022, 194, 226–255. [Google Scholar] [CrossRef]
- Brandão, S.N.; Sauer, J.; Schön, I. Circumantarctic distribution in Southern Ocean benthos? A genetic test using the genus Macroscapha (Crustacea, Ostracoda) as a model. Mol. Phylogenet. Evolut. 2010, 55, 1055–1069. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.B. Factors controlling CaCO3 dissolution in the Weddell Sea from foraminiferal distribution patterns. Mar. Geol. 1975, 19, 315–332. [Google Scholar] [CrossRef]
- Rao, C.P. Modern Carbonates: Tropical, Temperate, Polar. Ph.D. Thesis, University of Tasmania, Hobbart, Tasmania, 1996. [Google Scholar]
- Hartmann, G. Antarktische benthische Ostracodan III. Auswertung der Reise des FFS “Walther Herwig” 68/1. 3. Teil: Süd-Orkney-Inseln. Mitt. Aus. Dem. Hambg. Zool. Mus. Und. Institut. 1988, 85, 141–162. [Google Scholar]
- Hartmann, G. Antarktische benthische Ostracodan IV. Auswertung der wahrend der Reise von FFS “Walther Herwig” (68/1) bei Sud-Georgien gesammelten Ostracodan. Mitt. Aus. Dem. Hambg. Zool. Mus. Und. Institut. 1989, 86, 209–230. [Google Scholar]
- Hartmann, G. Antarktische benthische Ostracodan, V. Auswertung der Sudwinterreise von FS “Polarstern” (Ps 9/V-1) im Bereich Elephant Island und der Antarktischen Halbinsel. Mitt. Aus. Dem. Hambg. Zool. Mus. Und. Institut. 1989, 86, 231–288. [Google Scholar]
- Hartmann, G. Antarktische benthische Ostracodan VI. Auswertung der Reise der “Polarstern” (Ant. VI-2). (1 Teil, Meiofauna und Zehnerserien sowie Versuch einer vorlaufigen Auswertung aller bislang vorliegenden Daten). Mitt. Aus. Dem. Hambg. Zool. Mus. Und. Institut. 1990, 87, 191–245. [Google Scholar]
- Hartmann, G. Antarktische benthische Ostracodan. VIII. Auswertung der Reise der “Meteor” (Ant. 11/4) in die Gewasser um Elephant Island und der Antarktischen Halbinsel. Helgol. Meeresunters. 1992, 46, 405–424. [Google Scholar] [CrossRef]
- Hartmann, G. Antarktische benthische Ostracodan IX. Ostracodan von der Antarktischen Halbinsel und von der Isla de los Estados (Feuerland/Argentinien). Auswertung der “Polarstern”—Reise PS ANT/X/1b. Mitt. Aus. Dem. Hambg. Zool. Mus. Und Institut. 1993, 90, 227–237. [Google Scholar]
- Hartmann, G. Antarktische benthische Ostracoden X. Bemerkungen zur Gattung Krithe mit Beschreibung einer neuen Untergattung Austrokrithe. Mitt. Aus. Dem. Hambg. Zool. Mus. Und. Institut. 1994, 91, 77–79. [Google Scholar]
- Hartmann, G. Antarktische und Subantarktische Podocopa (Ostracoda). In Antarktische und Subantarktische Podocopa (Ostracoda); Koeltz Scientific Books: Koenigstein, Germany, 1997; Volume 7, pp. 1–355. [Google Scholar]
- Neale, J.W. An ostracod fauna from Halley Bay, Coats Land, British Antarctic Territory. Br. Antarct. Surv. Sci. Rep. 1967, 58, 1–50. [Google Scholar]
- Whatley, R.C.; Moguilevsky, A.; Ramos, M.I.F.; Coxill, D.J. Recent deep and shallow water Ostracoda from the Antarctic Peninsula and the Scotia Sea. Rev. Esp. Micropaleontol. 1998, 30, 111–135. [Google Scholar]
- Benson, R.H. Recent Cytheracean Ostracodes from McMurdo Sound and the Ross Sea, Antarctica. Arthropoda 1964, 6, 1–36. [Google Scholar]
- Chapmann, F. Ostracoda from up thrust mud above the Drygalski Glacier, southeast of Mount Larsen. British Antarctic Expedition 1907–1909 under the command of Sir E. H. Shackleton. Rep. Sci. Investig. Geol. 1916, 2, 37–40. [Google Scholar]
- Chapmann, F. Ostracoda from elevated deposits on the slopes of Mount Erebus, between Cape Royds and Cape Barne. British Antarctic Expedition 1907–1909 under the command of Sir E. H. Shackleton. Rep. Sci. Investig. Geol. 1916, 2, 49–52. [Google Scholar]
- Chapmann, F. Report on the Foraminifera and Ostracoda: Out of marine muds from soundings in the Ross Sea. British Antarctic Expedition 1907–1909 under the command of Sir E. H. Shackleton. Rep. Sci. Investig. Geol. 1916, 2, 53–80. [Google Scholar]
- Gazdzicki, A.; Pugaczewska, H. Biota of the “Pecten conglomerate” (Polonez Cove Formation, Pliocene) of King George Island (South Shetland Islands, Antarctica). Studia. Geol. Polonica 1984, 79, 59–120. [Google Scholar]
- Szczechura, J.; Blaszyk, J. Ostracods from the Pecten conglomerate (Pliocene) of Cockburn Island, Antarctic peninsula. Palaeontol. Polonica 1996, 68, 175–186. [Google Scholar]
- Taviani, M.; Claps, M. Biogenic Quaternary Carbonates in the CRP-l Drillhole, Victoria Land Basin, Antarctica. Terra Antart. 1998, 5, 411–418. [Google Scholar]
- Cape Roberts Science Team. Studies from Cape Roberts Project, Initial Report on CRP-2/2A, Ross Sea, Antarctica. Terra Antart. 1999, 6, 1–168. [Google Scholar]
- Dingle, R.V. Ostracoda from CRP–1 and CRP–2/2A, Victoria Land Basin, Antarctica. Terra Antart. 2000, 7, 479–492. [Google Scholar]
- Dingle, R.V.; Majoran, S. Palaeo—climatic and—biogeographical implications of Oligocene Ostracoda from CRP–2/2A and CRP–3 drillholes, Victoria Land Basin, Antarctica. Terra Antart. 2001, 8, 369–382. [Google Scholar]
- Scherer, R.; Hannah, M.; Maffioli, P.; Persico, D.; Sjunneskog, C.; Strong, C.P.; Taviani, M.; Winter, D. the ANDRILL-MIS Science Team 2007. [PDF] da unl.edu Palaeontologic Characterisation and Analysis of the AND-1B Core, ANDRILL McMurdo Ice Shelf Project, Antarctica. Terra Antart. 2007, 14, 223–254. [Google Scholar]
- Taviani, M.; Hannah, M.; Harwood, D.M.; Ishman, S.E.; Johnson, K.; Olney, M.; Riesselman, C.; Tuzzi, E.; Beu, A.G.; Blair, S.; et al. Palaeontological characterisation and analysis of the AND-2A Core, ANDRILL Southern McMurdo Sound Project, Antarctica. Terra Antart. 2009, 15, 113–146. [Google Scholar]
- Speden, I.G. Fossiliferous Quaternary marine deposits in the McMurdo Sound Region, Antarctica. New Zealand. J. Geol. Geophys. 1962, 5, 746–777. [Google Scholar] [CrossRef]
- Briggs, W.M. Ostracoda from the Pleistocene Taylor Formation, Ross Island, and the Recent of the Ross Sea and McMurdo Sound region, Antarctica. Antarct. J. U.S. 1978, 14, 27–29. [Google Scholar]
- Taviani, M.; Reid, D.E.; Anderson, J.B. Skeletal an isotopic composition and paleoclimatic significance of late pleistocene carbonates, Ross Sea Antarctica. J. Sediment. Petrol. 1993, 63, 84–90. [Google Scholar]
- Brambati, A.; Fanzutti, G.P.; Finocchiaro, F.; Melis, R.; Pugliese, N.; Salvi, G.; Faranda, C. Some paleoecological remarks on the Ross Sea Shelf, Antarctica. In Ross Sea Ecology: Italiantartide Expeditions (1987–1995); Faranda, F., Guglielmo, E., Ianora, A., Eds.; Springer: Berlin, Germany, 1999; pp. 51–61. [Google Scholar]
- Cai, H.M. Holocene Ostracoda and sedimentary environment implication in the core NG931-1 from the Great Wall Bay, Antarctica. Antarct. Res. 1996, 7, 141–149. [Google Scholar]
- Rathburn, A.E.; Pichon, J.J.; Ayress, M.A.; De Deckker, P. Microfossil and stable-isotope evidence for changes in Late Holocene palaeoproductivity and palaeoceanographic conditions in the Prydz Bay region of Antarctica. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1997, 131, 485–510. [Google Scholar] [CrossRef]
- Whatley, R.C.; Roberts, R. Late Quaternary Ostracoda from a Core in the Weddell Sea, Antartica. Pesqui. Em Geociênc. 1999, 26, 11–19. [Google Scholar] [CrossRef][Green Version]
- Melis, R.; Salvi, G. Foraminifer and Ostracod Occurrence in a Cool-Water Carbonate Factory of the Cape Adare (Ross Sea, Antarctica): A Key Lecture for the Climatic and Oceanographic Variations in the Last 30,000 Years. Geosciences 2020, 10, 413. [Google Scholar] [CrossRef]
- Capotondi, L.; Bonomo, S.; Budillon, G.; Giordano, P.; Langone, L. Living and dead benthic foraminiferal distribution in two areas of the Ross Sea (Antarctica). Rend. Lincei. Sci. Fis. E Naturali. 2020, 31, 1037–1053. [Google Scholar] [CrossRef]
- Stewart, A.; Klocker, A.; Menemenlis, D. Acceleration and Overturning of the Antarctic Slope Current by Winds, Eddies, and Tides. J. Phys. Oceanogr. 2019, 49, 126–133. [Google Scholar] [CrossRef]
- Mosola, A.B.; Anderson, J.B. Expansion and rapid retreat of the West Antarctic Ice Sheet in eastern Ross Sea: Possible consequence of over-extended ice streams? Quat. Sci. Rev. 2006, 25, 2177–2196. [Google Scholar] [CrossRef]
- Halberstadt, A.R.W.; Simkins, L.M.; Greenwood, S.L.; Anderson, J.B. Past ice-sheet behaviour: Retreat scenarios and changing controls in the Ross Sea, Antarctica. Cryosphere 2016, 10, 1003–1020. [Google Scholar] [CrossRef]
- D’Sa, E.J.; Kim, H.C.; Ha, S.Y.; Joshi, I. Ross Sea Dissolved Organic Matter Optical Properties During an Austral Summer: Biophysical Influences. Front. Mar. Sci. 2021, 8, 749096. [Google Scholar] [CrossRef]
- Bowen, M.; Fernandez, D.; Forcen-Vazquez, A.; Gordon, A.; Huber, B.; Castagno, P.; Falco, P. The role of tides in bottom water export from the western Ross Sea. Sci. Rep. 2021, 11, 2246. [Google Scholar] [CrossRef]
- Budillon, G.; Castagno, P.; Aliani, S.; Spezie, G.; Padman, L. Thermohaline variability and Antarctic bottom water formation at the Ross Sea shelf break. Deep Sea Res. Part I Oceanogr. Res. Pap. 2011, 58, 1002–1018. [Google Scholar] [CrossRef]
- Gordon, A.L.; Orsi, A.; Muench, R.; Visbeck, M. Western Ross Sea continental slope gravity currents. Deep Sea Res. Part II Top. Stud. Oceanogr. 2009, 56, 796–817. [Google Scholar] [CrossRef]
- Castagno, P.; Falco, P.; Dinniman, M.S.; Spezie, G.; Budillon, G. Temporal variability of the Circumpolar Deep Water inflow onto the Ross Sea continental shelf. J. Mar. Syst. 2017, 166, 37–49. [Google Scholar] [CrossRef]
- Dinniman, M.S.; Klinck, J.M.; Smith Jr, W.O. Cross-shelf exchange in a model of the Ross Sea circulation and biogeochemistry. Deep Sea Res. II 2003, 50, 3103–3120. [Google Scholar] [CrossRef]
- Dinniman, M.S.; Klinck, J.M.; Smith, W.O., Jr. A model study of Circumpolar Deep Water on the West Antarctic Peninsula and Ross Sea continental shelves. Deep Sea Res. II 2011, 58, 1508–1523. [Google Scholar] [CrossRef]
- Klinck, J.M.; Dinniman, M.S. Exchange across the shelf break at high southern latitudes. Ocean Sci. 2010, 6, 513–524. [Google Scholar] [CrossRef]
- Kohut, J.; Hunter, E.; Huber, B. Small-scale variability of the cross-shelfflow over the outer shelf of the Ross Sea. J. Geophys. Res. 2013, 188, 1863–1876. [Google Scholar] [CrossRef]
- Whitworth, T.; Orsi, A.H. Antarctic Bottom Water production and export by tides in the Ross Sea. Geophys. Res. Lett. 2006, 33, L12609. [Google Scholar] [CrossRef]
- Jacobs, S.S.; Giulivi, C.F. Large multidecadal salinity trends near the Pacific–Antarctic continental margin. J. Clim. 2010, 23, 4508–4524. [Google Scholar] [CrossRef]
- Purkey, S.G.; Johnson, G.C. Antarctic bottom water warming and freshening: Contributions to sea level rise, ocean freshwater budgets, and global heat gain. J. Clim. 2013, 26, 6105–6122. [Google Scholar] [CrossRef]
- Gales, J.; Rebesco, M.; De Santis, L.; Bergamasco, A.; Colleoni, F.; Kim, S.; Accettella, D.; Kovacevic, V.; Liu, Y.; Olivo, E.; et al. Role of dense shelf water in the development of Antarctic submarine canyon morphology. Geomorphology 2021, 372, 107453. [Google Scholar] [CrossRef]
- Castagno, P.; Capozzi, V.; Ditullio, G.R.; Falco, P.; Fusco, G.; Rintoul, S.R.; Budillon, G. Rebound of shelf water salinity in the Ross Sea. Nat. Commun. 2019, 10, 5441. [Google Scholar] [CrossRef] [PubMed]
- Silvano, A.; Foppert, A.; Rintoul, S.R.; Holland, P.R.; Takeshi, T.; Kimura, N.; Castagno, P.; Falco, P.; Budillon, G.; Haumann, F.G. Recent recovery of Antarctic Bottom Water formation in the Ross Sea driven by climate anomalies. Nat. Geosci. 2020, 13, 780–786. [Google Scholar] [CrossRef]
- Orsi, A.H.; Wiederwohl, C.L. A recount of Ross Sea waters. Deep Sea Res. Part II Top. Stud. Oceanogr. 2009, 56, 778–795. [Google Scholar] [CrossRef]
- Rivaro, P.; Ianni, C.; Raimondi, L.; Manno, C.; Sandrini, S.; Castagno, P. Analysis of physical and biogeochemical control mechanisms on summertime surface carbonate system variability in the western Ross Sea (Antarctica) using in situ and satellite data. Remote Sens. 2019, 11, 238. [Google Scholar] [CrossRef]
- Rivaro, P.; Ardini, F.; Vivado, D.; Cabella, R.; Castagno, P.; Mangoni, O.; Falco, P. Potential Sources of Particulate Iron in Surface and Deep Waters of the Terra Nova Bay. Water 2020, 12, 3517. [Google Scholar] [CrossRef]
- Anderson, J.B.; Brake, C.F.; Myers, N.C. Sedimentation on the Ross Sea continental shelf, Antarctica. Mar. Geol. 1984, 57, 295–333. [Google Scholar] [CrossRef]
- Brambati, A.; Fanzutti, G.P.; Finocchiaro, F.; Simeoni, U. Sediments and sedimentological processes in the Ross Sea continental shelf (Antarctica): Results and preliminary conclusions. Boll. Di Oceanol. Teor. Appl. 1989, 7, 159–188. [Google Scholar]
- Prothro, L.O.; Simkins, L.M.; Majewski, W.; Anderson, J.B. Glacial retreat patterns and processes determined from integrated sedimentology and geomorphology records. Mar. Geol. 2018, 395, 104–119. [Google Scholar] [CrossRef]
- Müller, G. Die Ostracodan der Deutschen Sudpolar-Expedition 1901–1903. Wissenschaftliche Ergebnisse der deutschen Sudpolar expedition. Zoologie 1908, 10, 51–181. [Google Scholar]
- Brenchley, P.J.; Harper, D.A.T. Palaeoecology: Ecosystems, Environments and Evolution; Chapman and Hall: London, UK, 1998. [Google Scholar]
- Boomer, I.; Horne, D.J.; Slipper, I.J. The Use of Ostracods in Palaeoenvironmental Studies, or What can you do with an Ostracod Shell? In Bridging the Gap: Trends in the Ostracode Biological and Geological Sciences; The Paleontological Society Papers: Cambridge, UK, 2003; Volume 9, pp. 153–180. [Google Scholar] [CrossRef]
- Brouwers, E.M. Palaeobathymetry on the continental shelf based on examples using ostracods from the Gulf of Alaska. In Ostracoda in the Earth Sciences; De Deckker, P., Colin, J.-P., Peypouquet, J.-P., Eds.; Elsevier: Amsterdam, The Netherlands, 1988; pp. 55–76. [Google Scholar]
- Brouwers, E.M. Sediment transport detected from the analysis of ostracod population structures: An example from the Alaskan Continental Shelf. In Ostracoda in the Earth Sciences; De Deckker, P., Colin, J.-P., Peypouquet, J.-P., Eds.; Elsevier: Amsterdam, The Netherlands, 1988; pp. 231–244. [Google Scholar]
- Arndt, J.E.; Schenke, H.W.; Jakobsson, M.; Nitsche, F.; Buys, G.; Goleby, B.; Rebesco, M.; Bohoyo, F.; Hong, J.K.; Black, J.; et al. The international bathymetric chart of the Southern Ocean (IBCSO) version 1.0—A new bathymetric compilation covering circum Antarctic waters. Geophys. Res. Lett. 2013, 40, 3111–3117. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology, 3rd ed.; Elsevier, B.V.: Amsterdam, The Netherlands, 2012; pp. 1–1006. [Google Scholar]
- Shannon, C.E.; Weaver, W. The Mathematical Theory of Communication; University of Illinois Press: Urbana, IL, USA, 1963; pp. 1–55. [Google Scholar]
- Ter Braak, C.J.F.; Smilauer, P. CANOCO Release 4. Reference Manual and Users Guide to CANOCO for Windows: Software for Canonical Community Ordination; Microcomputer Power: Ithaca, NY, USA, 1998. [Google Scholar]
- Ter Braak, C.J.F.; Šmilauer, P. CANOCO Reference Manual and User’s Guide: Software for Ordination; Version 5.0; Microcomputer Power: Ithaca, NY, USA, 2012. [Google Scholar]
- Borcard, D.P.; Legendre, P.; Drapeau, P. Partialling out the spatial component of ecological variation. Ecology 1992, 73, 1045–1055. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Terzopoulos, D. The computation of visible-surface representations. IEEE Trans. Pattern Anal. Mach. Intell. 1988, 10, 417–438. [Google Scholar] [CrossRef]
- Fofonoff, N.P.; Millard, R.C. Algorithms for computation of fundamental properties of seawater. UNESCO Tech. Pap. Mar. Sci. 1983, 44, 53. [Google Scholar]
- Lord, A.; Boomer, I.; Brouwers, E.; Whittaker, J. Ostracod Taxa as Palaeoclimate Indicators in the Quaternary. Dev. Quat. Sci. 2012, 17, 37–45. [Google Scholar] [CrossRef]
- Hartmann-Schroder, G.; Hartmann, G. Zur Kenntnis des Eulitorals der chilenischen Pazifikküste und der argentinischen Küste Südpatagoniens unter besonderer Berücksichtigung der Polychaeten und Ostracoden. Teil III Ostracodan des Eulitorals. Mitt. Aus. Dem. Hambg. Zool. Mus. Und. Institut. 1962, 60, 169–270. [Google Scholar]
- Whatley, R.C.; Staunton, M.; Kaesler, R.L.; Moguilevsky, A. The taxonomy of Recent Ostracoda from the southern part of the Strait of Magellan. Rev. Esp. Micropaleontol. 1996, 28, 51–76. [Google Scholar]
- Whatley, R.C.; Staunton, M.; Kaesler, R.L. The depth distribution of recent marine Ostracoda from the southern Strait of Magellan. J. Micropalaeontol. 1997, 16, 121–130. [Google Scholar] [CrossRef]
- Siciński, J.; Jażdżewski, K.; Broyer, C.D.; Presler, P.; Ligowski, R.; Nonato, E.F.; Corbisier, T.N.; Petti, M.A.V.; Brito, T.A.S.; Lavrado, H.P.; et al. Admiralty Bay Benthos Diversity—A census of a complex polar ecosystem. Deep Sea Res. Part II Top. Stud. Oceanogr. 2011, 58, 30–48. [Google Scholar] [CrossRef]
- Ayress, M.; De Deckker, P.; Coles, G.P. A taxonomic and distributional survey of marine benthonic Ostracoda off Kerguelen and Heard Islands, South Indian Ocean. J. Micropalaeontol. 2004, 23, 15–38. [Google Scholar] [CrossRef][Green Version]
- Griffiths, H.J.; Anker, P.; Linse, K.; Maxwell, J.; Post, A.L.; Stevens, C.; Tulaczyk, S.; Smith, J.A. Breaking All the Rules: The First Recorded Hard Substrate Sessile Benthic Community Far Beneath an Antarctic Ice Shelf. Front. Mar. Sci. 2021, 8, 642040. [Google Scholar] [CrossRef]
- Cummings, V.J.; Bowden, D.A.; Pinkerton, M.H.; Halliday, N.J.; Hewitt, J.E. Ross Sea Benthic Ecosystems: Macro and Mega-faunal Community Patterns From a Multi-environment Survey. Front. Mar. Sci. 2021, 8, 629787. [Google Scholar] [CrossRef]
- Arrigo, K.R.; Van Dijken, G.L.; Strong, A.L. Environmental controls of marine productivity hot spots around Antarctica. J. Geophys. Res. Oceans 2015, 120, 5545–5565. [Google Scholar] [CrossRef]
- Isla, E. Environmental controls on sediment composition and particle fluxes over the Antarctic continental shelf. In Source-To Sink Fluxes in Undisturbed Cold Environments; Beylich, A., Dixon, J., Zwolinski, Z., Eds.; Cambridge University Press: Cambridge, UK, 2016; pp. 199–212. [Google Scholar] [CrossRef]
- Smith, C.R.; Mincks, S.; DeMaster, D.J. A synthesis of benthopelagic coupling on the Antarctic shelf: Food banks, ecosystem inertia and global climate change. Deep Sea Res. II Top. Stud. Oceanogr. 2006, 53, 875–894. [Google Scholar] [CrossRef]
- Cummings, V.J.; Hewitt, J.E.; Thrush, S.F.; Marriott, P.M.; Halliday, N.J.; Norkko, A.M. Linking Ross Sea coastal benthic ecosystems to environmental conditions: Documenting baselines in a changing world. Front. Mar. Sci. 2018, 5, 232. [Google Scholar] [CrossRef]
- Pineda-Metz, S.E.A.; Isla, E.; Gerdes, D. Benthic communities of the Filchner Region (Weddell Sea, Antarctica). Mar. Ecol. Prog. Ser. 2019, 628, 37–54. [Google Scholar] [CrossRef]
- Gutt, J.; Arndt, J.; Kraan, C.; Dorschel, B.; Schröder, M.; Bracher, A. Benthic communities and their drivers: A spatial analysis off the Antarctic Peninsula. Limnol. Oceanogr. 2019, 62, 2341–2357. [Google Scholar] [CrossRef]
- Dunbar, R.; Anderson, J.B.; Domack, E.W.; Jacobs, S.S. Oceanographic influences on sedimentation along the Antarctic continental shelf: Oceanology of the Antarctic Continental Shelf. In Antarctic Research Series, American Geophysical Union; Jacobs: Washington, WA, USA, 1985; pp. 291–312. [Google Scholar] [CrossRef]
- Capotondi, L.; Bergami, C.; Giglio, F.; Langone, L.; Ravaioli, M. Benthic foraminifera distribution in the Ross Sea (Antarctica) and its relationship to oceanography. Boll. Della Soc. Paleontol. Ital. 2018, 57, 187–202. [Google Scholar] [CrossRef]
- Elverhoi, A.; Roaldset, E. Glaciomarine sediments and suspended particulate matter, Weddell Sea shelf, Antarctica. Polar Res. 1983, 1, 1–21. [Google Scholar] [CrossRef]
- Jansen, J.; Hill, N.; Dunstan, P.; McKinlay, J.; Sumner, M.; Post, A.; Eléaume, M.; Armand, L.; Warnock, J.; Galton-Fenzi, B.; et al. Abundance and richness of key Antarctic seafloor fauna correlates with modelled food availability. Nat. Ecol. Evolut. 2018, 2, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Fabiano, M.; Danovaro, R. Meiofauna distribution and mesoscale variability in two sites of the Ross Sea (Antarctica) with contrasting food supply. Polar Biol. 1999, 22, 115–123. [Google Scholar] [CrossRef]
- D’Onofrio, S.; Pugliese, N. Foraminiferal and ostracod fauna from the Ross Sea, Antarctica: Preliminary results. Boll. Oceanol. Teor. Appl. 1989, 7, 129–137. [Google Scholar]
- Asioli, A. Living (stained) benthic foraminiferal distribution in Western Ross Sea (Antarctica). Paleopelagos 1995, 5, 201–214. [Google Scholar]
- Bertoni, E.; Bertello, L.; Capotondi, L.; Bergami, C.; Giglio, F.; Ravaioli, M.; Rossi, C.; Ferretti, A. Benthic foraminifera as indicators of hydrologic and environmental conditions in the Ross Sea (Antarctica). Geophys. Res. Abstr. 2012, 14, 10288. [Google Scholar]
- Kawahata, H.; Fujita, K.; Iguchi, A.; Inoue, M.; Iwasaki, S.; Kuroyanagi, A.; Maeda, A.; Manaka, T.; Moriya, K.; Takagi, H.; et al. Perspective on the response of marine calcifiers to global warming and ocean acidification—behavior of corals and foraminifera in a high CO2 world “hot house”. Prog Earth Planet. Sci. 2019, 6, 1–37. [Google Scholar] [CrossRef]
- Brandão, S.; Hoppema, M.; Kamenev, G.; Karanovic, I.; Riehl, T.; Tanaka, H.; Vital, H.; Hyunsu, Y.; Brandt, A. Review of Ostracoda (Crustacea) living below the Carbonate Compensation Depth and the deepest record of a calcified ostracod. Prog Oceanogr. 2019, 178, 102144. [Google Scholar] [CrossRef]
| Samples ID | Taxa_S | Shannon_H | Samples ID | Taxa_S | Shannon_H |
|---|---|---|---|---|---|
| NBP94-01-03 | 4 | 1.32 | AN88-PQ09 | 1 | 0.00 |
| NBP94-01-04 | 8 | 2.02 | AN88-PQ14 | 1 | 0.00 |
| NBP94-01-06 | 11 | 2.19 | AN88-PQ20 | 2 | 0.67 |
| NBP94-01-41 | 1 | 0.00 | AN88-PQ22 | 1 | 0.00 |
| NBP94-01-45 | 5 | 1.55 | AN88-PQ23 | 2 | 0.65 |
| NBP95-01-05 | 3 | 0.71 | AN88-PQ29 | 2 | 0.69 |
| NBP95-01-52 | 3 | 1.01 | AN88-PQ03b | 2 | 0.68 |
| NBP95-01-53 | 4 | 1.12 | AN88-PQ07b | 4 | 1.36 |
| NBP95-01-54 | 4 | 1.09 | GRC_02 | 11 | 2.23 |
| NBP95-01-55 | 3 | 1.10 | GRC_04 | 9 | 1.98 |
| AN88-B32 | 2 | 0.56 | BDR_001 | 5 | 1.44 |
| AN88-B33 | 2 | 0.66 | ANTA91-11BC | 11 | 2.13 |
| AN88-B39 | 2 | 0.65 | ANTA96-426 | 6 | 0.98 |
| AN88-B47 | 2 | 0.69 | ANTA96-426b | 8 | 1.24 |
| AN88-B31 | 3 | 1.01 | ANTA96-502 | 3 | 0.91 |
| AN88-B32b | 1 | 0.00 | ANTA90-MM021 | 1 | 0.00 |
| AN88-B39b | 1 | 0.00 | ANTA90-MM022 | 1 | 0.00 |
| AN88-IB3P | 12 | 1.60 | ANTA90-MM039 | 2 | 0.62 |
| AN88-PQ03 | 1 | 0.00 | ANTA90-MM063 | 2 | 0.69 |
| AN88-PQ07 | 8 | 1.66 | ANTA90-MM065 | 1 | 0.00 |
| AN88-PQ08 | 1 | 0.00 | ANTA90-MM118 | 2 | 0.62 |
| Species | Av. Dissim | Contrib. % | Cumulative % | Cluster 1 | Cluster 2 | Cluster 3 |
|---|---|---|---|---|---|---|
| Australicythere polylyca (Müller, 1908) | 23.37 | 23.84 | 23.84 | 1.226 | 0.000 | 0.148 |
| Australicythere devexa (Müller, 1908) | 19.64 | 20.04 | 43.87 | 0.966 | 0.000 | 0.010 |
| Xestoleberis rigusa (Müller, 1908) | 8.46 | 8.63 | 52.50 | 0.164 | 0.000 | 0.000 |
| Loxoreticulatum fallax (Müller, 1908; Hartmann, 1986) | 6.49 | 6.62 | 59.12 | 0.136 | 0.000 | 0.000 |
| Cativella bensoni (Neale, 1967) | 6.43 | 6.56 | 65.68 | 0.130 | 0.014 | 0.051 |
| Austrotrachyleberis antarctica (Neale, 1967) | 6.29 | 6.42 | 72.10 | 0.164 | 0.010 | 0.000 |
| Patagonacythere longiducta (Skogsberg, 1928) | 5.57 | 5.69 | 77.79 | 0.272 | 0.000 | 0.000 |
| Argilloecia sp. | 3.59 | 3.66 | 81.45 | 0.034 | 0.048 | 0.000 |
| Cytheropteron antarcticum (Chapman, 1916) | 2.42 | 2.47 | 83.91 | 0.062 | 0.000 | 0.000 |
| Macropyxis similis (Brady, 1880) | 2.28 | 2.32 | 86.24 | 0.044 | 0.000 | 0.000 |
| Copytus elongatus (Benson, 1964) | 1.55 | 1.58 | 87.82 | 0.003 | 0.021 | 0.000 |
| Bairdoppilata simplex (Brady, 1880) | 1.44 | 1.47 | 89.29 | 0.090 | 0.000 | 0.000 |
| Sclerochilus (Praesclerochilus) reniformis (Müller, 1908; Schornikov, 1982) | 1.26 | 1.28 | 90.57 | 0.011 | 0.014 | 0.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salvi, G.; Anderson, J.B.; Bertoli, M.; Castagno, P.; Falco, P.; Fernetti, M.; Montagna, P.; Taviani, M. Recent Ostracod Fauna of the Western Ross Sea (Antarctica): A Poorly Known Ingredient of Polar Carbonate Factories. Minerals 2022, 12, 937. https://doi.org/10.3390/min12080937
Salvi G, Anderson JB, Bertoli M, Castagno P, Falco P, Fernetti M, Montagna P, Taviani M. Recent Ostracod Fauna of the Western Ross Sea (Antarctica): A Poorly Known Ingredient of Polar Carbonate Factories. Minerals. 2022; 12(8):937. https://doi.org/10.3390/min12080937
Chicago/Turabian StyleSalvi, Gianguido, John B. Anderson, Marco Bertoli, Pasquale Castagno, Pierpaolo Falco, Michele Fernetti, Paolo Montagna, and Marco Taviani. 2022. "Recent Ostracod Fauna of the Western Ross Sea (Antarctica): A Poorly Known Ingredient of Polar Carbonate Factories" Minerals 12, no. 8: 937. https://doi.org/10.3390/min12080937
APA StyleSalvi, G., Anderson, J. B., Bertoli, M., Castagno, P., Falco, P., Fernetti, M., Montagna, P., & Taviani, M. (2022). Recent Ostracod Fauna of the Western Ross Sea (Antarctica): A Poorly Known Ingredient of Polar Carbonate Factories. Minerals, 12(8), 937. https://doi.org/10.3390/min12080937









