1. Introduction
Mordenite is an aluminosilicate mineral belonging to the zeolite group. The origin of the name comes from Morden County, Nova Scotia, Canada, where this fibrous mineral in radial aggregate was observed for the first time by How [
1]. The ideal chemical formula of mordenite is (Na
2,K
2,Ca)
4[Al
8Si
40O
96]·28H
2O and cell parameters are:
a = 18.05–18.25 Å,
b = 20.35–20.53 Å,
c = 7.49–7.55 Å, Z = 1 [
2,
3].
Mordenite (MOR framework-type) [
4] is characterized by puckered sheets consisting of six-membered rings parallel to (100), linked by four-membered rings. This structural arrangement produces strongly compressed eight-member (8MRc: 5.7 × 2.5 Å) and ellipsoidal twelve-member channels (12MRc: 7.0 × 6.5 Å) extending along
c. Those channels intersect with less-compressed eight-member channels running along
b (8MRb: 4.8 × 3.4 Å), forming a peculiar “plumbing system” accessible for extra-framework cation and molecule diffusion [
5]. The framework density, FD, is 17.2 T/1000 Å
3 and the average R, Ra = Si/(Si+Al) is 0.83 [
3,
4,
6]. The structure of mordenite was solved by Meier [
7] in the
Cmcm space group. However, this symmetry leads to an unfavorable T–O–T angle of 180°. Subsequently, a symmetry lowering to the acentric
Cmc2
1 space group provided a more accurate description of the extra-framework cation and H
2O molecule population, concurrently preventing the occurrence of the energetically unfavorable T–O–T angle of 180° [
8]. It is worth noting that Alberti et al., [
8] and Simoncic and Armbruster [
5], constrained couples of pseudo-centrosymmetric framework atoms to freely move but maintaining the coordinates related by the inversion center, imposing,
de facto, a
Cmcm symmetry to the framework. In addition, framework defects have been consistently reported, as testified by the occurrence of diffuse scattering in electron diffraction photographs or reconstruction of reciprocal layers from area detector measurement from synchrotron radiation diffraction experiments (see, for example, Simoncic and Armbruster [
5]).
Mordenite has been described in vugs of basalts and other volcanic rocks and intrusive (granite) rocks, and as a diagenetic product of silicic tuff, pitchstone, and volcanic sediments [
3]. Red compact masses of mordenite with radial and fibrous habits were often observed in amygdaloidal samples, associated with quartz, hematite, and other zeolites such as analcime, chabazite, heulandite, dachiardite, stilbite, natrolite, scolecite, epistilbite, ferrierite, erionite, and thomsonite. In diagenetically altered volcanic sediments, mordenite generally occurs in association with clinoptilolite, chabazite, erionite, phillipsite, ferrierite, and analcime [
2,
3]. Occurrences of mordenite are common worldwide, but generally its content in diagenetic deposits is higher than in hydrothermal ones. However, in both cases, mordenite occurs with the fibrous habit [
3,
9].
In Italy, mordenite has been described in some localities of Sardinia [
2,
10,
11], in the Island of Elba [
2,
8], in the Island of Ponza [
12], and many localities from Northern Italy [
2,
13,
14]. Except for Ponza mordenite, which has a diagenetic origin, all the mordenite crystals from Italy were originated in basaltic vugs.
In Northern Italy, mordenite is common in Lessini Mounts (Vicenza) and Fassa Valley (Trento), and it usually occurs in compact masses of fibers with a radiated habit, associated with microcrystalline quartz, hematite, and calcite [
2,
14]. In the Lessini Mounts, tertiary magmatic products of the Veneto Volcanic Province range from poorly to highly vesiculated basalts and basanites [
15]. Basalt vesicles are often filled with secondary minerals of hydrothermal origin, especially zeolites (analcime, chabazite, phillipsite, harmotome, gmelinite, yugawaralite, and willhendersonite [
16,
17,
18,
19]). Moreover, other fibrous zeolites (erionite, offretite, and ferrierite) have been recently discovered in several localities of Lessini Mounts: these findings are of particular importance due to their toxicological implications [
20,
21,
22,
23,
24]. Furthermore, in the Dolomites area (where Fassa Valley is located), a great amount of volcanic material is present. Here, volcanic and plutonic products of the magmatic series of Middle Triassic rocks are widespread, occurring as pyroclastic layers, reworked volcaniclastics, pillow lavas, pillow breccias, and hyaloclastics [
25,
26]. The vugs of these volcanic rocks are widely zeolitized, and several species are present, in particular analcime, natrolite, thomsonite, heulandite, clinoptilolite, chabasite, mordenite, dachiardite, and laumontite [
14,
27].
Zeolites are widely used in many fields, such as agronomy and ecology, in many industrial processes such as the production of detergents and drying agents, and in medicine [
3,
28,
29,
30]. In particular, mordenite is a very used zeolite because of its high silica/alumina ratio, which allows it to resist exposure to high temperatures and acids, either gaseous or liquids [
31,
32]. Mordenite is widely used both in natural and synthetic form, especially in the petrochemical industry, as a catalyst in industrial processes, as a host matrix in semiconductors, chemical sensors, and as a selective membrane for adsorptive separation of gas or liquid mixtures [
31,
32,
33,
34,
35,
36].
Despite the number of industrial applications of mordenite and its physicochemical similarities to the carcinogen fibrous erionite, there are very few studies on the hazard of mordenite involving animals [
37,
38,
39,
40,
41] or cell lines [
42,
43]. Moreover, sometimes the tested mordenite was not fibrous, contained more than 50% of other mineral phases or the proportion of fibers in the material was not specified, the experiment duration was short, small numbers of animals were tested, and information on survival was lack, as highlighted by Guthrie and IARC [
44,
45]. For this reason, mordenite is classified as Group 3 by IARC but detailed research is needed, as suggested by the recent literature. In recent decades, some studies focused on the possibility that mordenite could be a potential inhalation health hazard [
46,
47]. In general, the available information indicates that the health risk associated with mordenite is low or null, especially if compared to erionite [
44]. Erionite and mordenite show different cytotoxic capabilities. Mordenite resulted not mutagenic and the presence of ferrous ion in the crystals did not alter mordenite’s mutagenic potential [
43]. Many other fibrous zeolites such as erionite [
48,
49,
50], offretite [
23,
51], ferrierite [
24,
52,
53], mesolite, and thomsonite [
54] resulted in different toxicity and carcinogenicity response to humans.
The most important factors related to toxicity and carcinogenicity of the fibers are the size parameters (length, diameter, aerodynamic diameter), chemical composition and related bio-persistence, and other features (e.g., microtopography, surface area, structure, net charge, zeta potential, interacting capability, impurities, and toxic elements) [
48,
55,
56,
57,
58,
59,
60,
61,
62,
63,
64,
65].
Due to the abundance of mordenite in some volcanic rocks of Northern Italy and its suspected toxicity to humans, an accurate characterization of these fibers is of paramount importance. In the present work, a detailed morphological, chemical, and structural characterization of different mordenite samples from Northern Italy is presented. Data from Scanning Electron Microscopy with Energy Dispersive X-ray (SEM-EDX), Electron MicroProbe Analysis (EMPA), X-ray Powder Diffraction (XRPD), Thermo Gravimetric Analysis-Differential Scanning Calorimetry (TGA-DSC), and Inductively Coupled Plasma Atomic Emission Spectroscopy (ICP-AES) were combined to characterize from the morphological, chemical, and structural point of view this zeolite. Particular attention was paid to the small fibers detected on the investigated samples.
3. Results
The sample FAS1 is composed of an intergrowth of elongated acicular crystals resulting in soft pink masses of centimetric size. Some large individuals (about 30 μm in width and 280 μm in length) are present from SEM observation (
Figure 1a). However, the crystals show a strong cleavage and fragmentation along the elongation axis, resulting in a chaotic pattern of very small fibers (
Figure 1b). The size variability of these fibers is significant, ranging from 0.1 μm to 6 μm in terms of width, and from 4 μm to 80 μm in terms of length. However, the great part of the measured fibers belongs to a small size range. In particular, almost all the fibers (about 96% of the total) have a diameter value lower than 3 μm. The remaining fraction has a width between 3 μm and 5 μm (about 3%) or is bigger than 5 μm (about 1%). Regarding the length values, about 57% of the total of the measured fibers is lower than 20 μm, while the remaining part (about 43%) lies in the range from 20 to 100 μm (
Table 1). For regulatory purposes, the most considered sizes of inhalable fibers are: length >5 μm, diameter <3 μm, diameter–length ratio >1:3 [
76]. Moreover, according to Stanton’s hypothesis [
77], fibers with length >8 μm and diameter <0.25 μm are the most able to induce lung disease. For these reasons, accurate measurement of mordenite fibers is of paramount importance.
The sample GC1 is formed by soft woolly masses of centimetric size, with variable colors ranging from pink to brown, but always with a soft appearance. In SEM images, the acicular-to-fibrous crystals show a very elongated habit (
Figure 1c). The bigger acicular individuals have a length of about 140 μm and about 20 μm in width, but also in this sample, high cleavage evidence is present (
Figure 1d). Similar to the previous samples, also the mordenite fibers from GC1 show a wide size variability, ranging from 0.1 μm to 7 μm in width and from 5 μm to 70 μm in length, but the fibers are distributed in a small range of dimensions. About 92% of the total measured fibers have a width thinner than 3 μm, while the remaining fraction has a bigger size: about 6% lies in the range 3 μm–5 μm, and about 2% is bigger than 5 μm. As regards the length, about 64% of the total is lower than 20 μm, while about 36% lies in the range of 20 μm–100 μm (
Table 1).
Similarly, in the sample SP1, very elongated crystals with acicular-to-fibrous habit, strictly intergrow, resulting in pink masses of centimetric size, sometimes with a soft aspect. From SEM image observation, in general, the bigger fragments (about 80 μm in width and 300 μm in length, (
Figure 1e) show a strong cleavage and fragmentation and are formed by a great number of smaller needles and fibers (
Figure 1f). The mechanical behavior of the fibers is always rigid. Sometimes, also the small fibers show a cleavage pattern parallel to the elongation axis. Accurate measurements of the detected fibers were performed on several SEM images. The results are summarized in
Table 1. In particular, the small fibers separated from bigger crystals show wide size variability, ranging from less than 0.1 μm to 8 μm in width and from 4 μm to 170 μm in length. Regarding diameter, 78% of the fibers have a width up to 3 μm, about 6% range between 3 and 5 μm, while for the remainder (about 15%) the diameter is bigger than 5 μm. As regards the length values, 46% of the total of the measured fibers are lower than 20 μm, while about 40% lie in the range from 20 μm to 100 μm. The remaining part (about 13%) of the fibers are longer than 100 μm, up to 170 μm.
The TGA analysis showed a significant weight loss at 450 °C, associated with H2O loss. The resulting water content of the studied mordenite samples is 14.70 wt% for FAS1, 13.00 wt% for GC1 sample, and 12.70 wt% for SP1 sample.
Microchemical data collected by SEM-EDX and EMPA provided similar oxides concentrations (
Table 2), and they scale reasonably well albeit, as expected, differences were observed caused by the onset of the different mechanisms discussed in Pacella et al. [
68]. In general, obtaining reliable EMPA data was difficult owing to the morphological and mechanical features of the fibers and only a minor fraction of the analytical points measured were considered positive. This is especially true for sample SP1, whose analyses generally returned high E% values and a total extra-framework site scattering (s.s.) significantly lower than expected from crystal–chemical reasoning and comparison with reference data. For this reason, we will discuss in the following the microchemical data collected by SEM-EDX.
The chemical formulae of the mordenite samples, obtained as an average of reliable SEM-EDX point analysis, are shown in
Table 3. The unit formulae were calculated based on 96 oxygens.
The three samples have a similar chemical composition and may be identified as Na-rich mordenites. The prevailing extra-framework cations are in the Na > Ca > K relationship. The M/(M+D) ratio (M = Na + K; D = Ca + Mg + Mn) is in the 0.617–0.638 range. K content becomes relevant in GC1 (0.36 apfu) and especially in SP1 (0.91 apfu). Mg content is very low and consistently <0.2 apfu.
ICP-AES, performed on non-purified samples, indicated the occurrence of low contents of Ba, in the range 578–7400 mg/kg, and Sr in the range 710–3620 mg/kg (
Table 4). The occurrence of minor Ba was confirmed by EMPA at some analytical points (max. 0.20 wt% BaO, corresponding to 0.05 apfu Ba in GC1). A significant amount of Fe (range 2070–7500 mg/kg) and Mn (601–27,200 mg/kg) were also detected by ICP-AES (
Table 4).
The value of D
ae has been calculated for all the mordenite samples using the equation of Gonda and Abd El Khalik [
66]. The 50th percentile of diameter and length were used as the mean value, as suggested by Di Giuseppe [
47]. The size parameters, the theoretical density of the mordenite samples and the related D
ae values obtained by Gonda’s equation [
66] are shown in
Table 5. For the sample FAS1 the resulting D
ae is 2.69 μm, for GC1 is 1.19 μm, while for the sample SP1 is 3.91 μm.
Although mordenite crystallizes in
Cmc2
1, similarly to several crystallographic studies involving the analysis of its structure (e.g., Passaglia et al., Elsen et al., Martucci et al., Lotti et al. [
12,
78,
79,
80]) we refined the various samples in the centrosymmetric
Cmcm space group. This is justified by the relevant pseudo-centrosymmetry of the mordenite structure and by the corresponding significant reduction in the number of refined parameters.
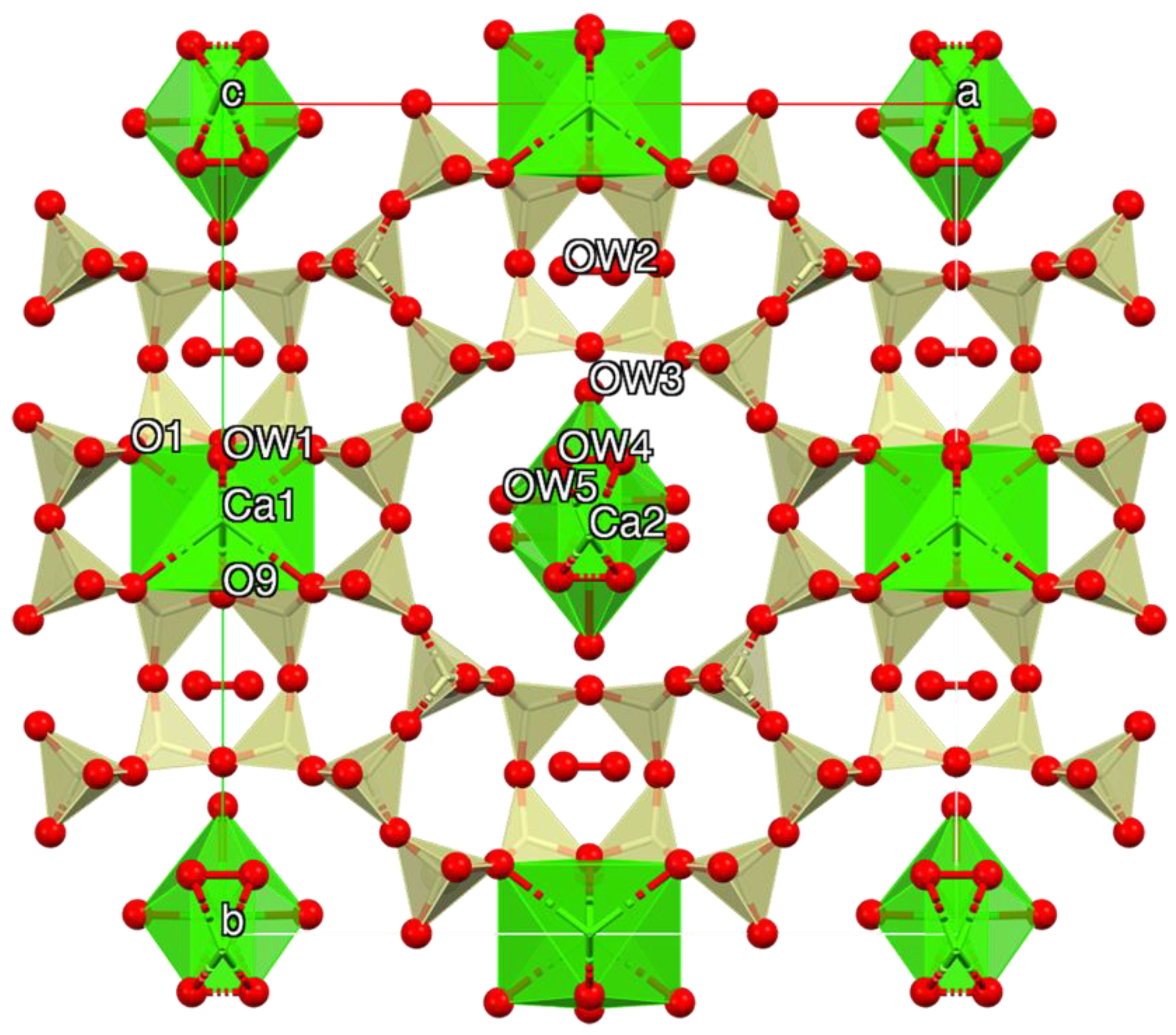
Starting structural parameters were taken from Mortier et al. [
81] for a rehydrated Ca-exchanged mordenite owing to chemical similarities. The model includes two partly occupied cation sites (Ca1 and Ca2) located, respectively, at the center of the strongly compressed eight-member channel (8MRc) and within the ellipsoidal twelve-membered ring (12MRc) channel. H
2O molecules populate five sites (OW1 within 8MRc; OW3, OW4 and OW5 within 12MRc; OW2 within the side-pocket parallel to (010)), in some cases partly occupied and possibly affected by H
2O/cation disorder.
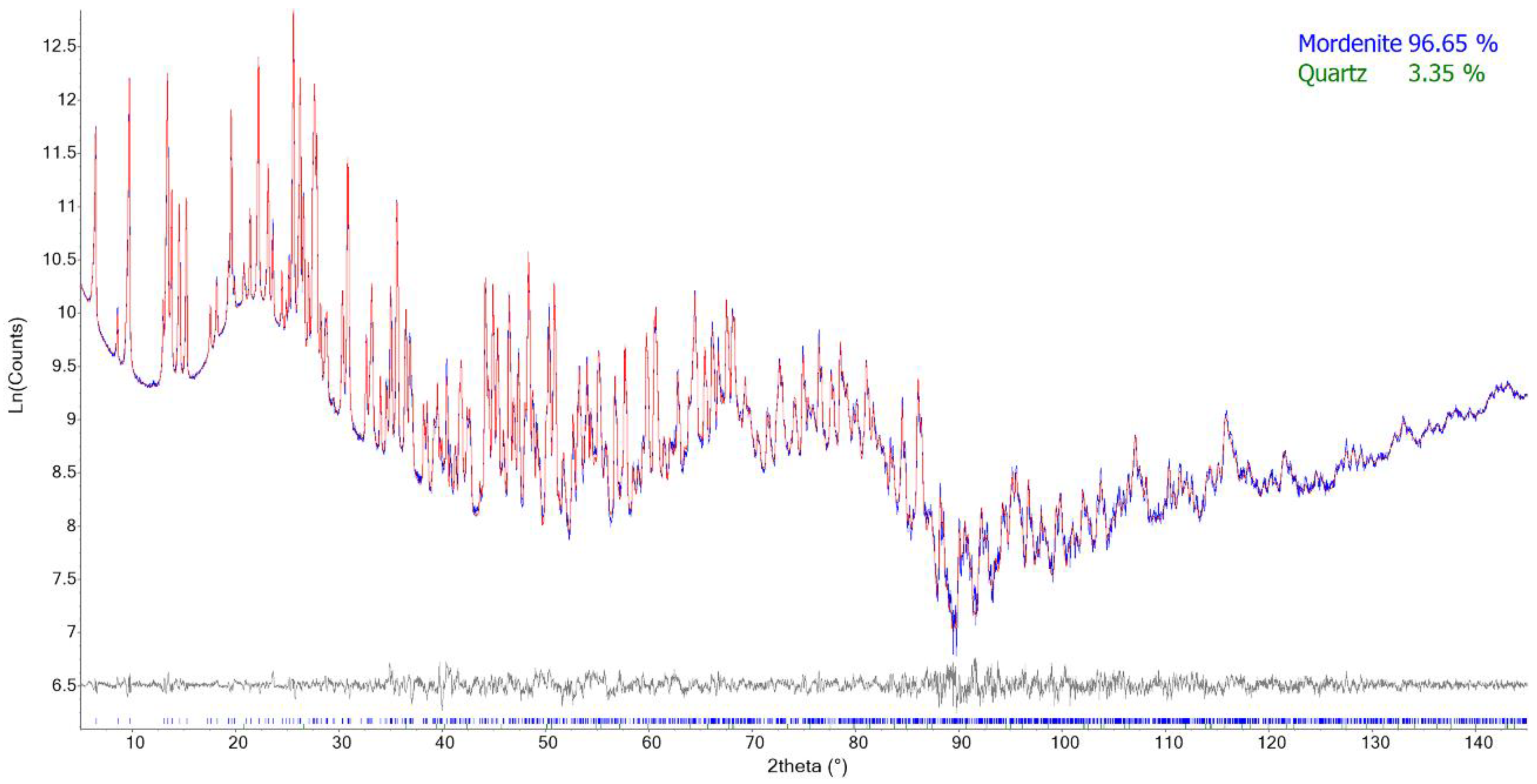
Preliminary analysis of the various patterns revealed the occurrence of quartz (FAS1: abundant; GC1 and SC1: minor) and a 15 Å phyllosilicate phase (FAS1: minor). In the initial stages of the refinements, isotropic displacement parameters of mordenite were refined for groups of atoms (all tetrahedral cations, all framework oxygens, all extra-framework cations), keeping fixed those of the oxygen atoms of H
2O to the average value calculated from the structural data of Simoncic and Armbruster [
5]. In the final cycle of refinement, individual isotropic displacement parameters of the framework atoms were refined. Moreover, s.s. at extra-framework cation and H
2O sites were refined. Neutral scattering curves of Si, O, and Ca were used for modeling the framework and the extra-framework cation sites. The population of Al vs. Si at the various tetrahedral sites T was iteratively adjusted to conform to the calculated population from the analysis of the corresponding <T–O> using the Jones’s equation [
82]. Fully ionized O-2 scattering curves were used for modelling the H
2O molecule sites to empirically compensate for the presence of the bonded hydrogen atoms. Using this approach, the total H
2O pfu was refined to a value close to that expected from crystal–chemical reasoning and TGA data. Starting structural data of quartz were those of Le Page and Donnay [
83]. The full structure refinement of quartz was performed in the case of sample FAS1, whereas, in the remaining samples, only the scale factor was optimized along with a single parameter for modeling the crystallite dependence of the peak broadening.
Miscellaneous data of the refinements are reported in
Table 6, and relevant structural data in
Table 7. Rietveld plots of sample SP1 are shown, as an example, in
Figure 2. CIFs were deposited as
Supplementary Materials and are available for download at the journal’s site.
4. Discussion
Mordenite crystals from the three investigated samples are characterized by similar structural features (
Figure 3). The <T–O> bond distances refined in the 1.602–1.647 Å range pointing out to some Si,Al ordering. Individual T–O bond distances are slightly spread as compared to refinements carried out in
Cmc2
1 [
5,
8]. This behavior is coherent with the absence of restraints on bond distances and is marginally more pronounced for FAS1 owing to the presence in mixture of a relevant amount of quartz (
Table 6). The unit cell volumes are in the following relationship: FAS1 > SP1 > GC1 (
Table 6), and this behavior correlates with both the decreasing Al content determined from SEM-EDX and that estimated from the Jones’s equation [
82] (
Table 2). It has been shown that this equation, as well as other more accurate calculation procedures [
86,
87], underestimates the Al content of mordenite samples [
5]. This underestimation has been attributed to the shortening of T–O bond distances caused by the apparent symmetry being higher than the true one [
87]. For example, the calculated R = Si/(Si+Al) of the sample of Ca-exchanged mordenite analyzed by Mortier et al. [
81] is 0.912 as compared to 0.846 from chemical data. Present data confirm such behavior as R was calculated in the 0.895–0.914 range, which is higher than 0.817–0.832 from SEM-EDX analyses. The latter range is coherent with the chemical analyses of natural mordenite samples (R = 0.80–0.86 [
3]). Al consistently exhibits the following site-specific occupation preference: T3 > T4 > T1 > T2 sequence (
Table 8) in close agreement with reference data [
5,
81,
87], confirming its prevailing allocation at T3 and, subordinately, at T4, i.e., the tetrahedra build the four-membered ring. The retrieved mutual extra-framework cations/H
2O molecule distribution is clearly affected by the simplification used to refine the structure in the
Cmcm space group. This approximation does not permit a detailed discussion of those structural details and for this reason, only a few features will be underlined. Both position and s.s. of the extra-framework cation and H
2O sites are close to those of Mortier et al. [
81], except for OW2, which was allowed to move freely from the center of the pocket in correspondence of the extra-framework site C of Mortier [
85], and as a result, it was found to be slightly off-axis. The total extra-framework cation s.s. of the three samples is rather constant, being in the 76.8(10)–79.5(14) e
− range and is smaller than that determined from SEM-EDX (82.1–92.2). The Ca1 site is eight-fold coordinated to six oxygen atoms of the framework (4 × O1; 2 × O9) and to two H
2O molecules lying at OW1, which is consistently fully occupied. Ca2 is at bonding distance exclusively with H
2O molecules (OW3, OW4, and OW5). OW3 and OW5 are almost fully occupied, whereas OW4 is approximately half-occupied in agreement with the mutually excluding short OW4–OW4 contacts. This leads to a seven-fold coordination for Ca2. The H
2O content is also constant, being in the 29.4(3)–31.7(4) molecule pfu range, in reasonable agreement with both present and reference chemical data [
3]. It is worth noting that OW2 is at bonding distances with other H
2O molecule sites allocated in the neighboring channels (OW1 in 8MRc and OW3 in 12MRc, respectively). TGA points to a higher H
2O content of FAS1 with respect to GC1 and SP1 coherently with the refined s.s. (
Table 7). The total number of H
2O molecules pfu derived from the structure refinements is regularly higher than that determined by TGA, suggesting the occurrence of some H
2O/cation disorder. This hypothesis is supported by the total extra-framework cation s.s. of the three samples from Rietveld refinement being smaller than that retrieved from SEM-EDX.
As previously described, the three samples show comparable chemical compositions, and the only differences regard extra-framework cations, water content, and the amount of impurities. Despite the crystal habit being always fibrous, significant differences in the fiber’s size were observed in the studied samples. The obtained D
ae values are 2.69 μm for the sample FAS1, 3.91 μm for SP1, and 1.19 μm for GC1. Considering that particles with D
ae value up to 10 μm can pass the larynx (thoracic fraction) and particles with D
ae < 4 μm are able to reach the alveolar region (respirable fraction) [
88], in all the studied samples a fraction of fibers are respirable and could enter into the alveolus. However, the variability of the D
ae value highlights the possibility that the hazard of mordenite fibers to humans could be significantly different, depending on the sample features, and needs to be evaluated case by case. Considering the similar structural and chemical features compared to other carcinogenic zeolites (e.g., erionite), fibrous mordenite should be considered potentially hazardous for humans.
In acid conditions (e.g., cellular environment—macrophages) the Si/Al framework ratio is fundamental to guide the dissolution rate. Zeolites with a high Si/Al ratio show a slower dissolution, resultantly being more biodurable [
51,
89]. In particular, zeolite dissolution can result in (I) stoichiometric framework degradation and silicate precipitation (Si/Al = 1); (II) partially dissolved silicate framework with selective removal of aluminum and then partially dissolved silicate particles (Si/Al = 2); (III) intact silicate framework (Si/Al = 3). In the last case, the aluminum is insufficient to weaken the structure and cause silicon dissolution. Thus, little or no precipitation is observed, and amorphous undissolvable silicate particles remain intact [
89]. For this reason, it is important to consider the higher Si/Al ratio of the studied mordenite samples (4.47 for FAS1, 4.94 for GC1, and 4.72 for SP1) compared to erionite (about 3.5) [
47].
Moreover, as observed by EMPA and ICP-AES, low but significant contents of Fe, Mn, Ba, and Sr were detected on natural samples. The role of some elements detected on the fibers or in attached impurities (including minor and trace elements), such as Fe, Mn, As, Pb, Cr, Ni, Po, and others, is very important for the toxicity of fibrous minerals as demonstrated by the wide literature [
51,
61,
65,
90]. Therefore, the oxidation state and the amount of iron or other cations on mordenite fibers could play an important role in its toxicity, as observed for asbestos and other zeolites. In fact, the presence of iron nanoparticles is considered as one of the key factors for explaining fiber toxicity. Iron exposed at the fiber surface, especially Fe
2+, is considered to play a primary role for Reactive Oxygen Species (ROS) production [
90]. Moreover, Mn resulted in being cytotoxic to human lung cells (in vitro) at concentration ranges of 0.2–200 μM [
91].
Considering the important amount of mordenite fiber and mass in the volcanic rocks of the sampling areas, and the resulting features of these fibers, attention could be focused on managing this mineral and hosting rocks, especially in case of active mining or quarrying activities and in the use of such material. However, further detailed studies are needed. As known, the evaluation of the toxicity and carcinogenicity of mineral fibers is a complex task. Still, at the same time, it is fundamental to plan activities and safety rules, especially regarding occupationally exposed subjects. Our work represents a further step through the knowledge of morphometric, physical, and chemical characteristics of mordenite. As a matter of fact, together with bio-durability, surface activity, and correlation with human epidemiological observations, these parameters play a crucial role in evaluation of toxicity and carcinogenicity of mineral fibers [
92,
93].
5. Conclusions
At present, fibrous mordenite is considered a non-carcinogenic mineral for humans and is classified as Group 3 by IARC [
44]. However, this classification is essentially due to the lack of detailed data on this zeolite. Despite the abundance of natural mordenite in many worldwide zeolitized volcanic rocks, and the number of industrial applications of this mineral, few studies have investigated the chemical, structural, physical, and toxicological character of mordenite fibers. In the present work, three different samples of fibrous mordenite from Northern Italy were investigated in detail, with the aim of defining their size parameters, the crystal structure, and the chemical composition of mordenite and detected impurities.
The calculated Dae values are 1.19 μm for the GC1 sample, 2.69 μm for FAS1, and 3.91 μm for SP1, meaning that all the studied samples could spread fibers which can be considered “respirable” for humans. From the chemical point of view, all the mordenite samples are Na-rich (Na > Ca > K). The presence of Ba, Sr, Fe, and Mn was also detected in non-purified samples, and these elements could play a crucial role in the toxicity of mordenite fibers. The structural features of the investigated samples are similar, and the unit cell volume follows the series FAS1 > SP1 > GC1, reflecting Al content decreasing. The higher Si/Al ratio of mordenite (about 4.7) compared to erionite (about 3.5) could also play an important part in its biodurability.
These results contribute to increasing the knowledge of this important fibrous zeolite, especially from the mineralogical (crystal structure and chemistry) and physical (morphology and size parameters) points of view, and are the basis for a further study on the toxicological implications of this zeolite. In any case, the data obtained in this work indicate that fibrous mordenite may represent a potential health hazard and should be tested for toxicity and carcinogenicity, as it could be a non-safe zeolite.