Sustainable Use of Copper Resources: Beneficiation of Low-Grade Copper Ores
Abstract
:1. Introduction
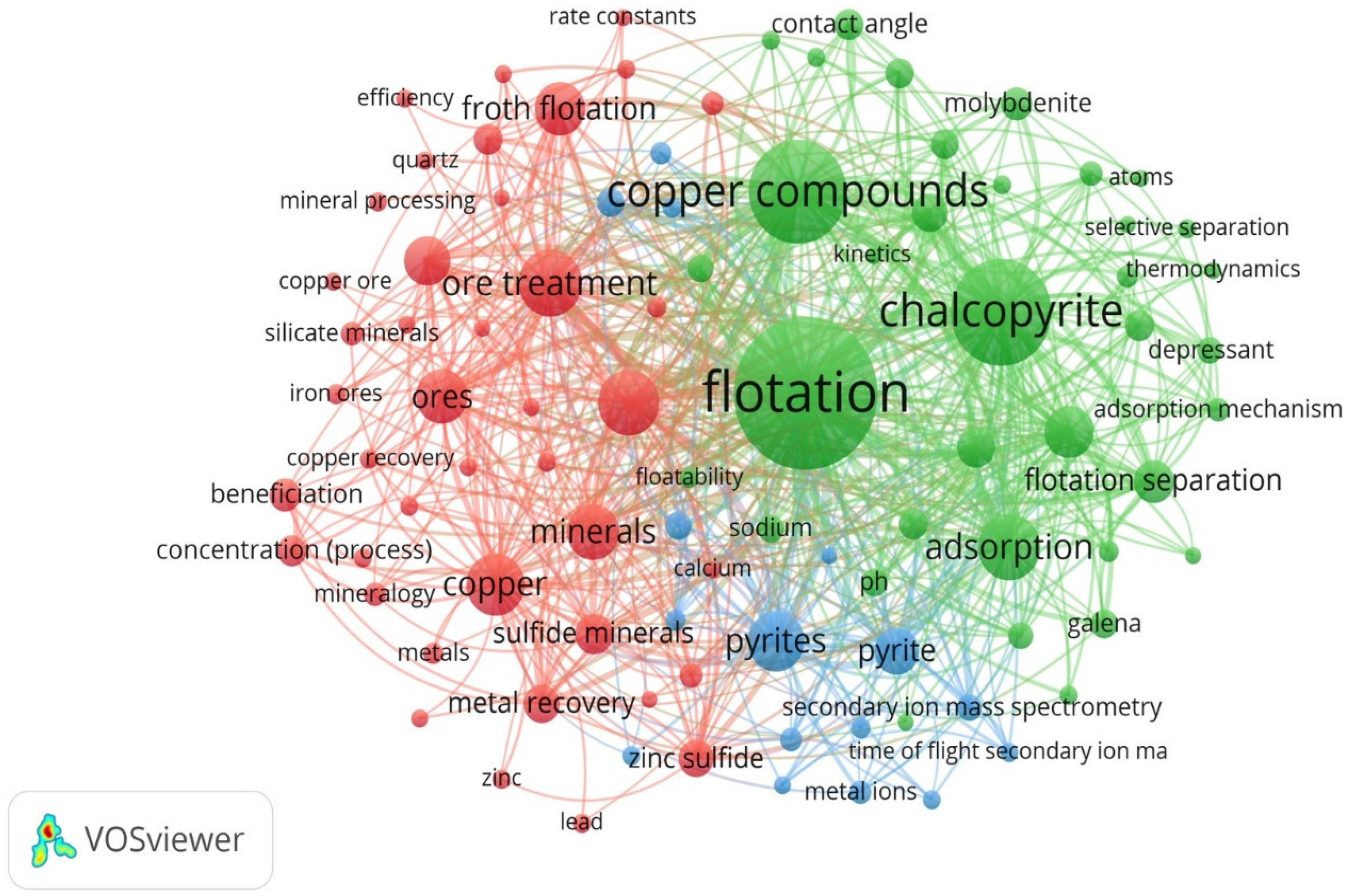
2. Visualization of Recent Research Trends of Copper Using Bibliographic Analysis
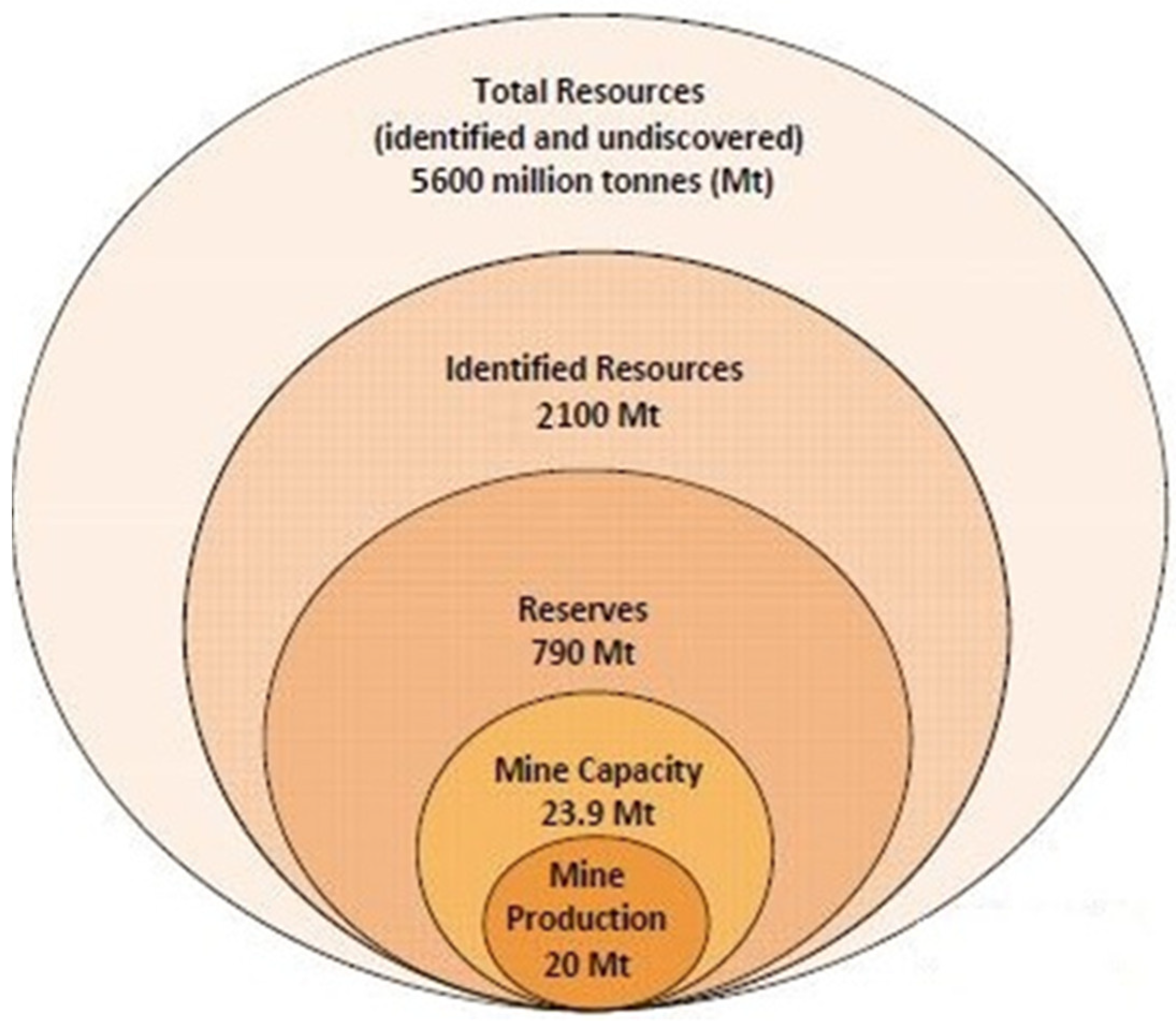
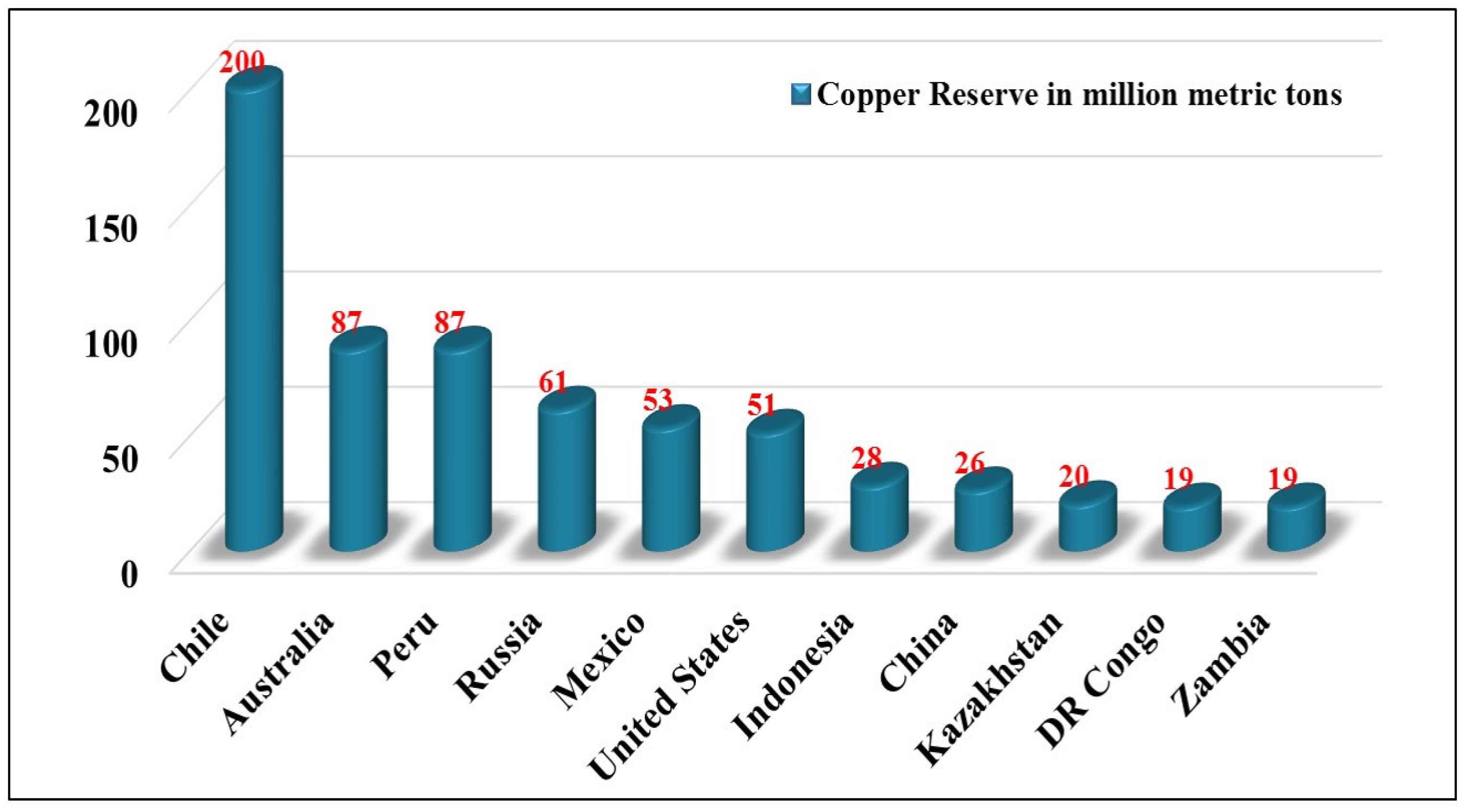
3. Global Copper Reserve, Demand and Production and Consumption
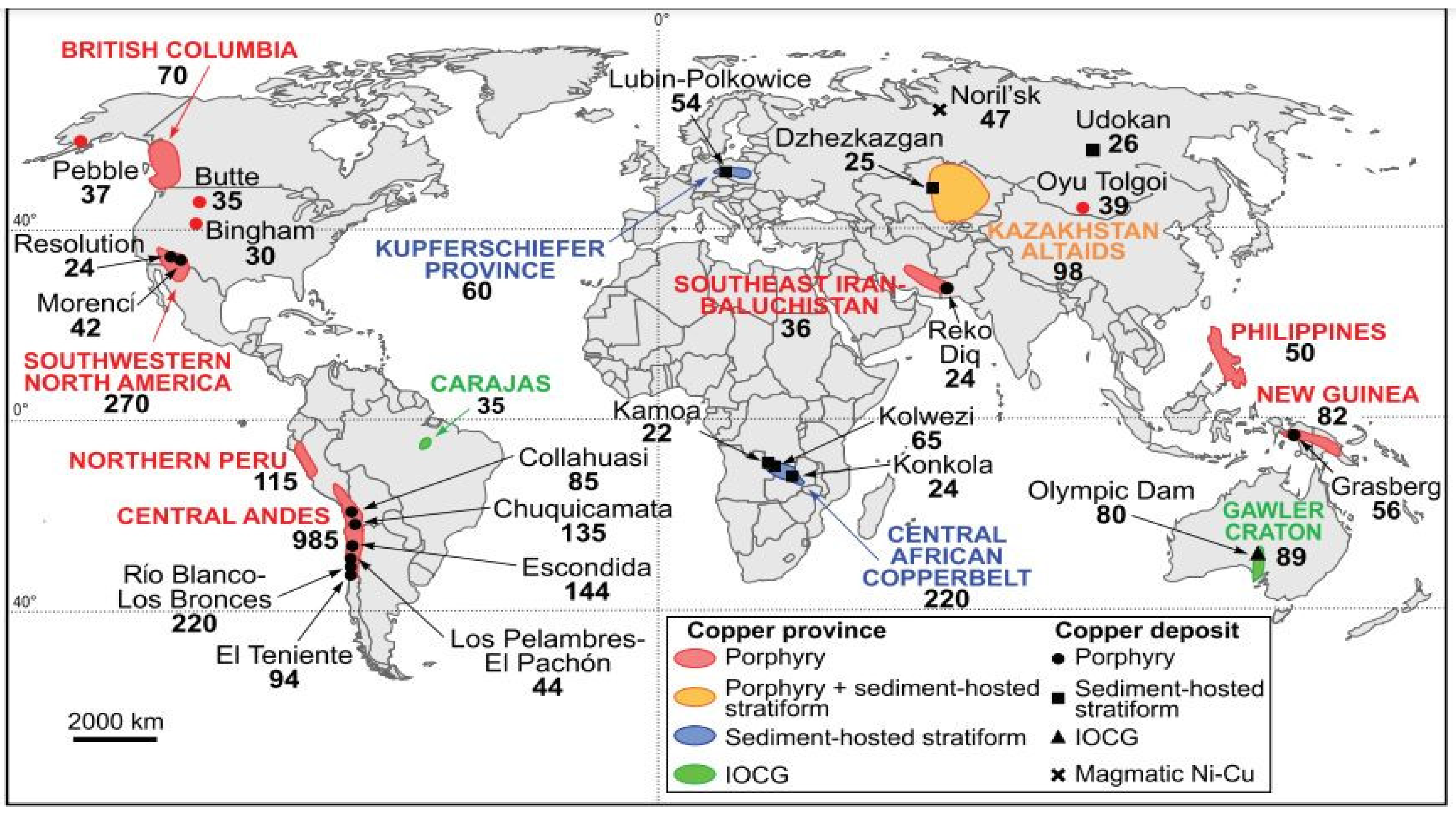
4. Copper Mineralogy
5. Processing of Copper Ore
5.1. Gravity Concentration or Separation
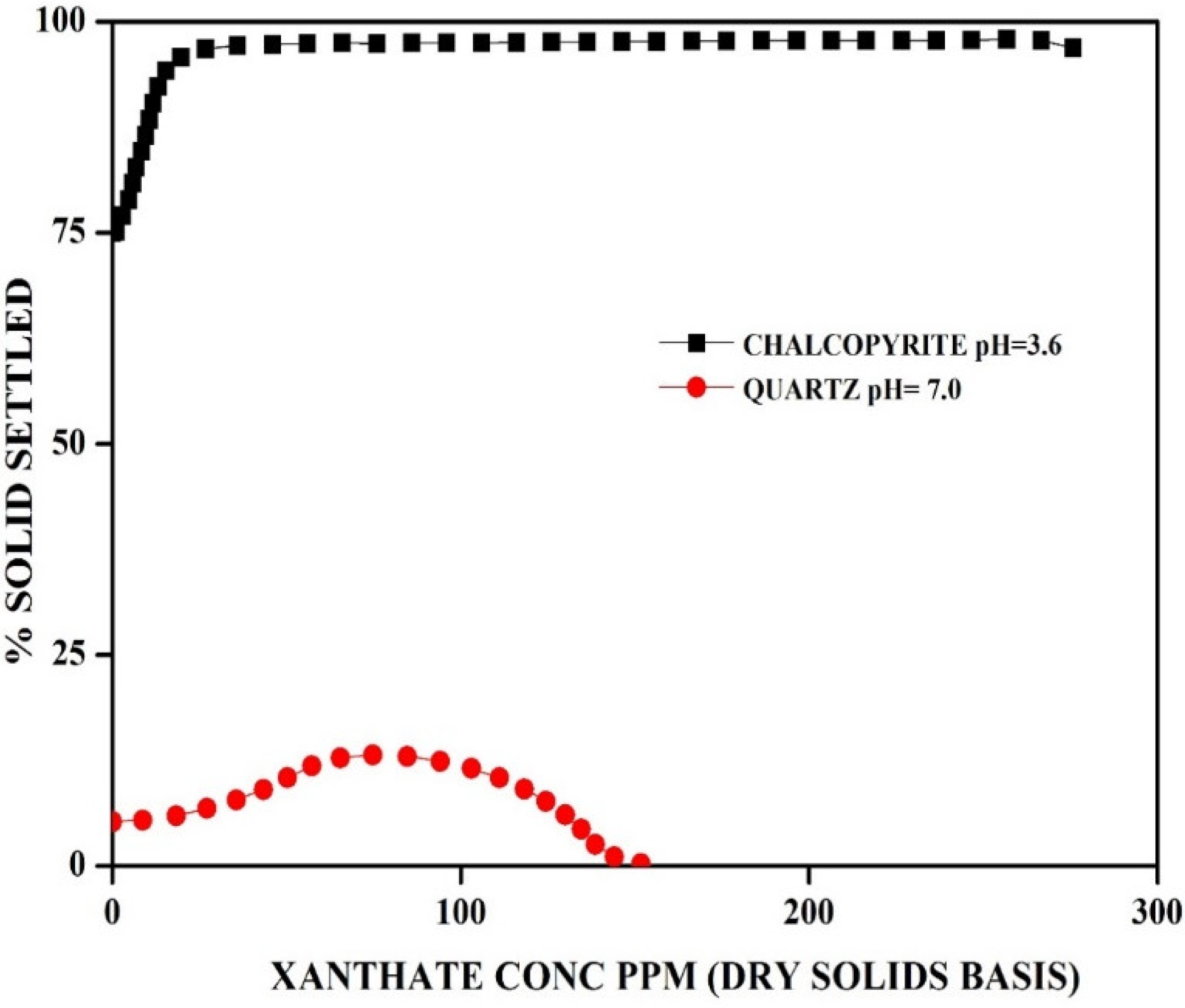
5.2. Selective Flocculation
6. Flotation of Copper Ore
6.1. Copper Sulphide Minerals
6.1.1. Chalcopyrite
6.1.2. Bornite
6.1.3. Chalcocite
6.2. Copper Oxide Minerals
- (i)
- The froth flotation method after the sulphidization stage or
- (ii)
- The mixture of flotation and hydrometallurgy or
- (iii)
- Flotation segregation, whereas the hydrometallurgical route generally processes oxide copper ores with high dissolution rates.
6.2.1. Malachite
| Hydroxamate | Abbreviation | Structure | Manufacturer |
|---|---|---|---|
| Benzo | BHA | 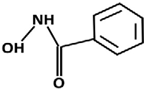 | Sigma Aldrich (St. Louis, MO, USA) |
| Salicyl | SHA |  | Alfa aesar (Haverhill, MA, USA) |
| Aceto | AHA |  | Alfa aesar (Haverhill, MA, USA) |
| K-Butyl | PBH | 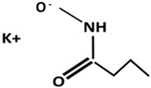 | Synthesized |
| Octyl | OHA | 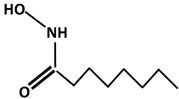 | Synthesized |
| K-Octyl | POH |  | Synthesized |
| Tetradecyl | THA |  | Synthesized |
6.2.2. Chrysocolla
6.2.3. Cuprite
| Ore Mineral; Gangue Mineral | Sample Source (Ore Deposit/Country) | Collector | Remarks | References |
|---|---|---|---|---|
| Malachite; Quartz | AMR Mineral and Metal Inc., Canada | Benzo Hydroxamate[Best] Salicyl Hydroxamate Aceto Hydroxamate K-butyl hydroxamate Octyl Hydroxamate K-octyl Hydroxamate Tetradecyl Hydroxamate | Synthesized ore (mixture of malachite and quartz) | [89] |
| Chrysocolla; Quartz | Miami, Arizona | Hexyl Mercaptan, Dodecyl Mercaptan (Higher Xanthate) | Natural Ore | [99] |
| Miami, Arizona | Aminothiophenol | Natural Ore | [98] | |
| Cuprite; Quartz, calcite | Copper Company of Dishui Corporation, Xinjiang, China. | Sodium Iso amyl Xanthate (SIAX) | Natural Ore | [104] |
| Cu Sulphide (Chalcopyrite and bornite) and Cu Oxide (Malachite with small Azurite); Quartz | Sherwood Copper’s Minto mines, Canada | N-octyl hydroxamate (AM28) | Synthesized ore (Mixture of Cu Sulphide Cu Oxide::(70:30)) | [81] |
6.3. Mixed Copper Ore
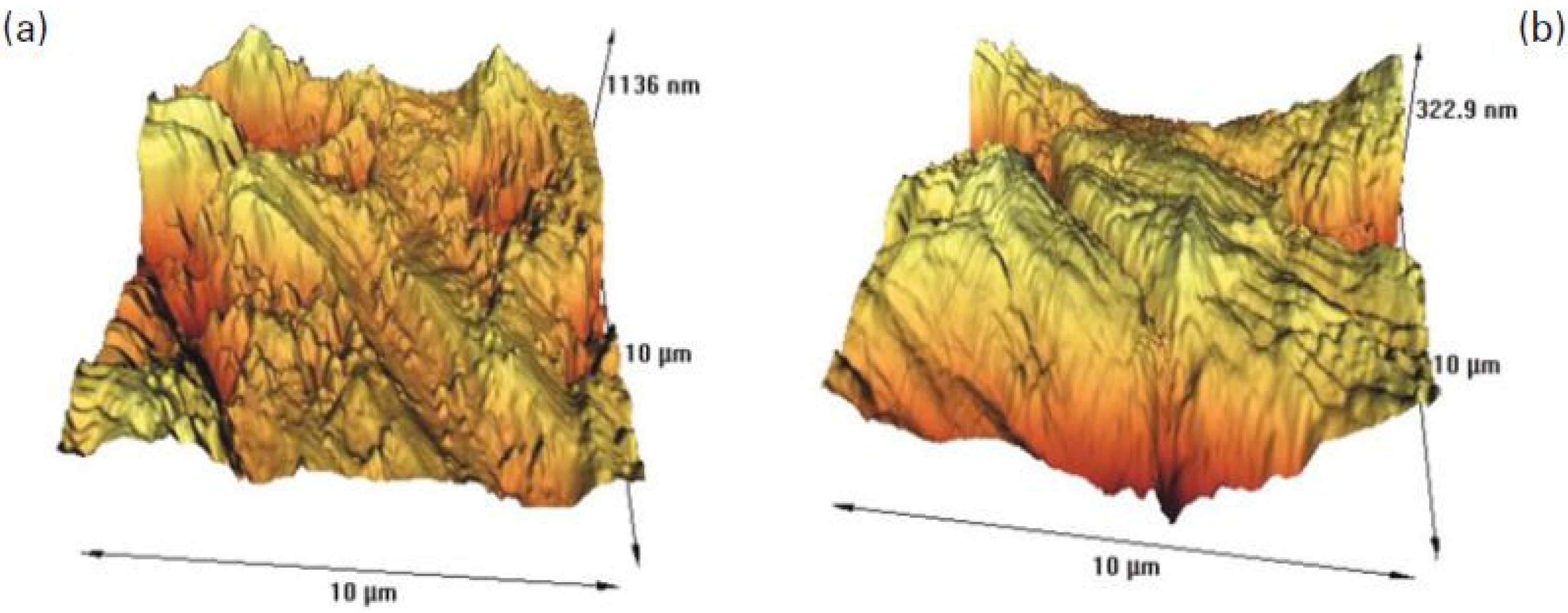
6.4. Effect of Mineralogy on Copper Mineral Zeta Potential
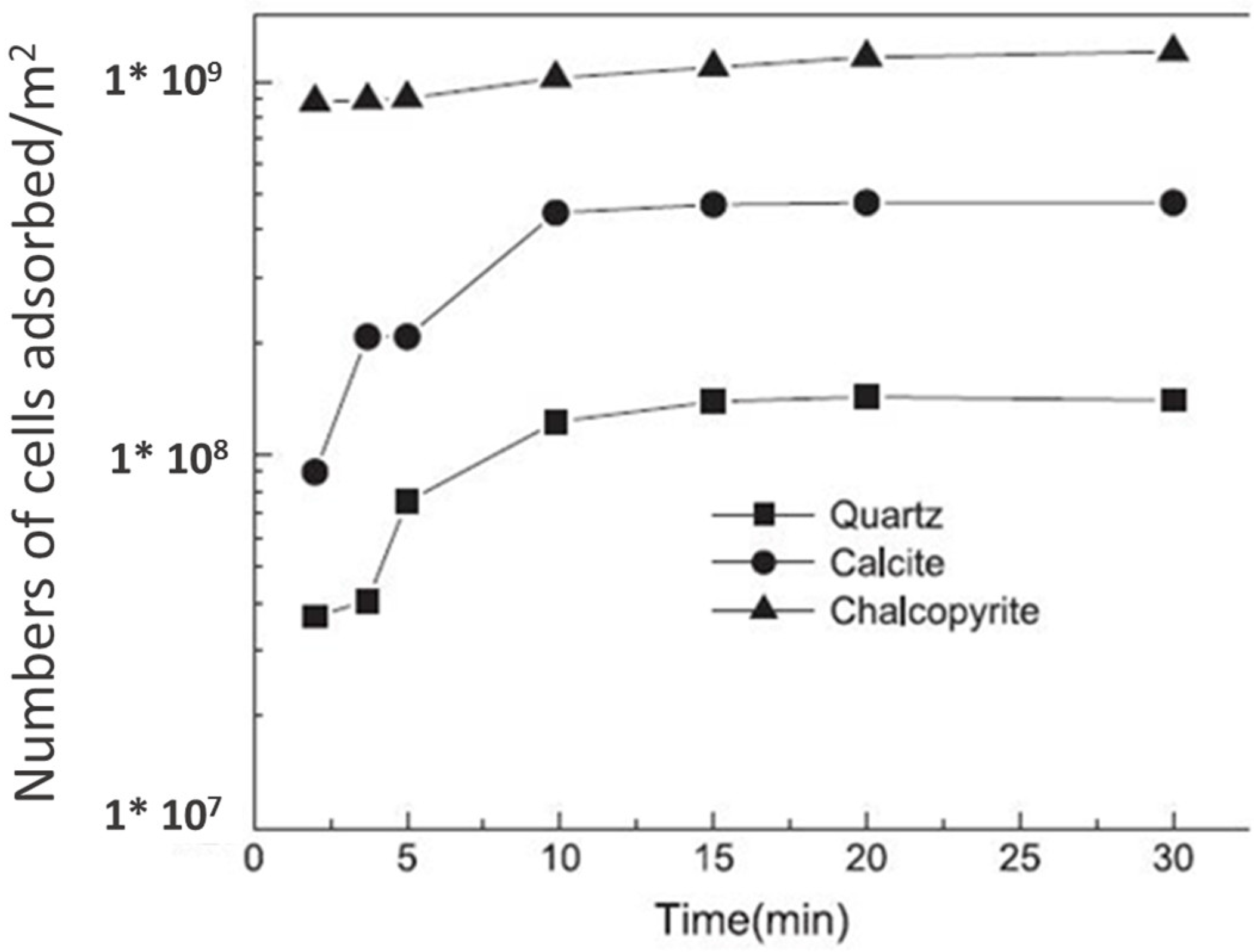
6.5. Bio-Collector for the Flotation of Copper
6.6. Effect of Water Quality on Copper Flotation
7. Problems Associated with Copper Tailings
8. Different Aspects of Copper Plant Flowsheet/Practice
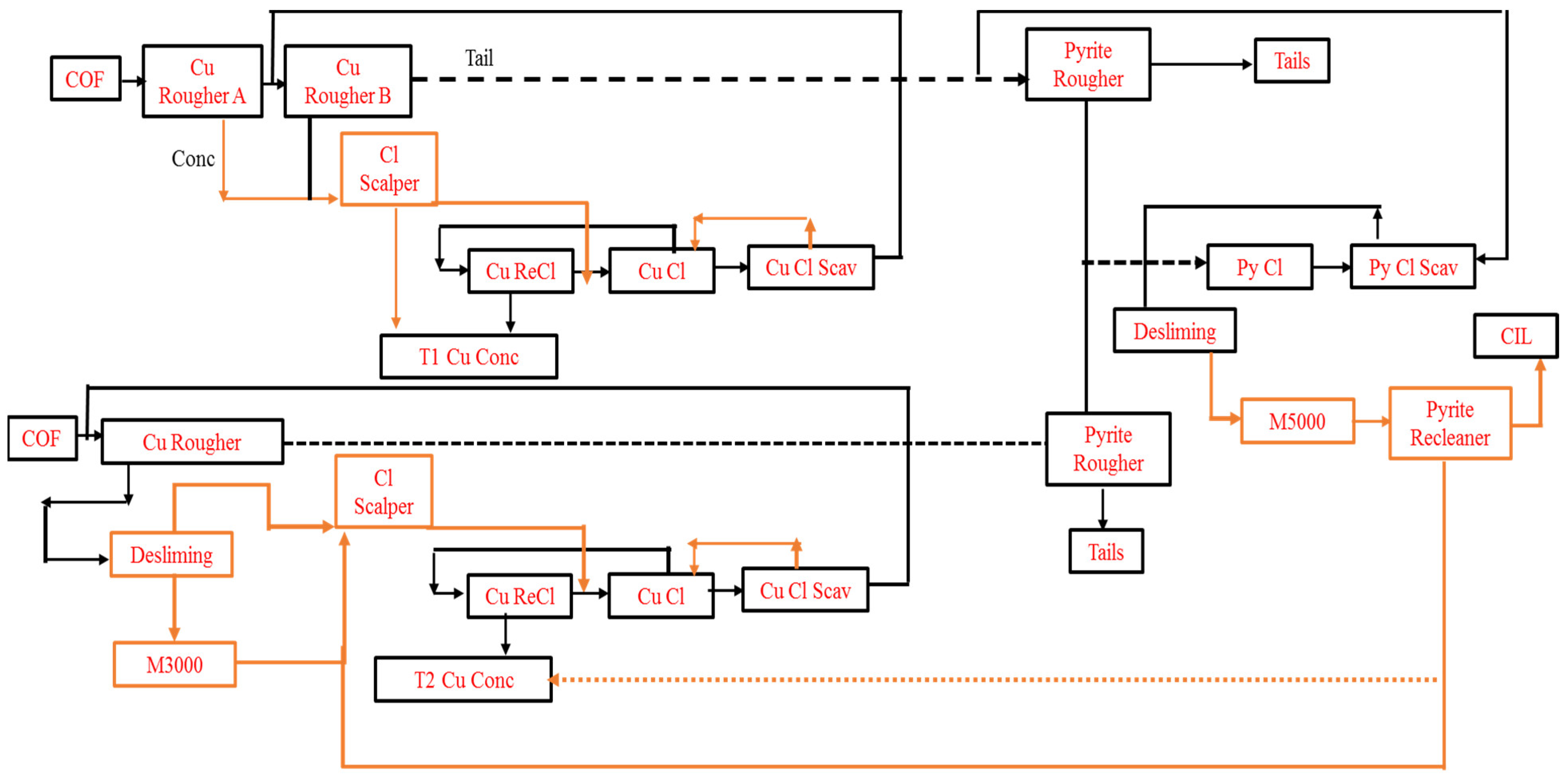
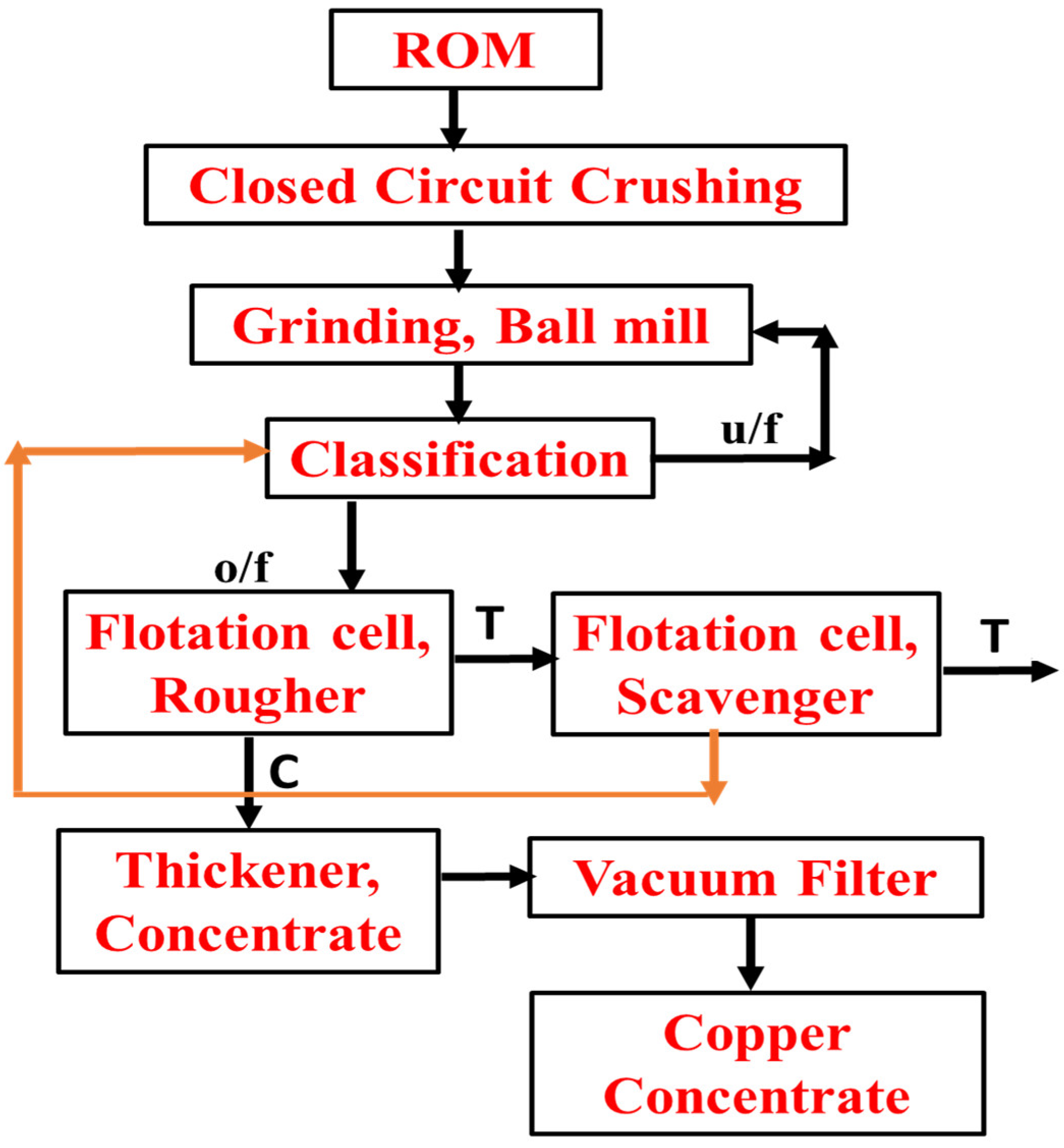
8.1. Dexing Copper Plant, China
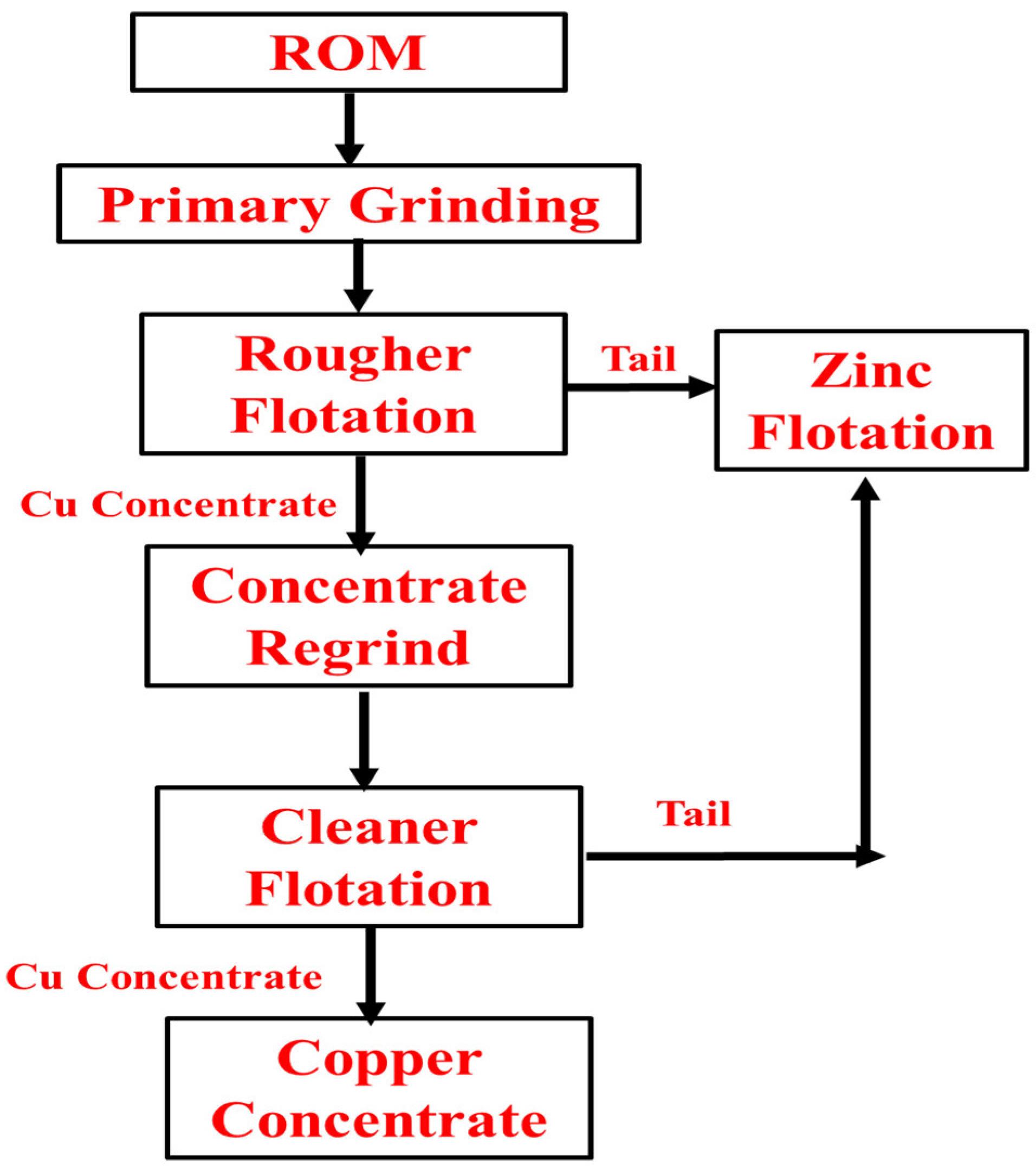
8.2. Sungun Copper Plant, Iran
8.3. Telfer Copper Processing Plant, Western Australia
8.4. Mosabani and Malanjkhand Copper Plant, India
8.5. Cerro de Maimón Copper Plant, Dominican Republic
9. Future Prospect of Pretreatment Methods for Copper Minerals
9.1. Microwave Pretreatment
9.2. High-Voltage Pulse Treatment
9.3. Ultrasonic Pretreatment
10. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- LePan, N. Visualizing Copper’s Role in the Transition to Clean Energy. Available online: https://www.visualcapitalist.com/visualizing-coppers-role-in-the-transition-to-clean-energy/ (accessed on 6 March 2022).
- Moreno-Leiva, S.; Haas, J.; Junne, T.; Valencia, F.; Godin, H.; Kracht, W.; Nowak, W.; Eltrop, L. Renewable energy in copper production: A review on systems design and methodological approaches. J. Clean. Prod. 2020, 246, 118978. [Google Scholar] [CrossRef]
- Hund, K.; La Porta, D.; Fabregas, T.; Laing, T.; Drexhage, J. Minerals for Climate Action: The Mineral Intensity of the Clean Energy Transition; World Bank Group; World Bank: Washington, DC, USA, 2020. [Google Scholar]
- International Copper Association and the Copper Alliance®—Copper Alliance. Available online: https://copperalliance.org/ (accessed on 3 September 2020).
- Copper Development Association Inc. Copper’s Bright Past and Brighter Future; Copper Development Association Inc.: New York, NY, USA, 2022. [Google Scholar]
- Bide, T. British Geological Survey Report—Copper; NERC Open Research Archive: Nottingham, UK, 2007. [Google Scholar]
- Schipper, B.W.; Lin, H.-C.; Meloni, M.A.; Wansleeben, K.; Heijungs, R.; van der Voet, E. Estimating global copper demand until 2100 with regression and stock dynamics. Resour. Conserv. Recycl. 2018, 132, 28–36. [Google Scholar] [CrossRef]
- Prasad, U. Economic Geology: Economic Mineral Deposits; CBS Pub.: New Delhi, India, 2006; ISBN 812-390-460-6. [Google Scholar]
- Kleijn, R.; van der Voet, E.; Kramer, G.J.; van Oers, L.; van der Giesen, C. Metal requirements of low-carbon power generation. Energy 2011, 36, 5640–5648. [Google Scholar] [CrossRef]
- Evans, A.M. Ore Geology and Industrial Minerals: An Introduction; John Wiley & Sons: Hoboken, NJ, USA, 2009; ISBN 144-431-405-X. [Google Scholar]
- Skinner, B.J. Chapter 10 A Second Iron Age Ahead? In Studies in Environmental Science; Elsevier: Amsterdam, The Netherlands, 1979; Volume 3, pp. 559–575. [Google Scholar] [CrossRef]
- Guy, P.; Trahar, W. The Effects of Oxidation and Mineral Interaction on Sulphide Flotation; Elsevier: Amsterdam, The Netherlands, 1985. [Google Scholar]
- Trahar, W. A rational interpretation of the role of particle size in flotation. Int. J. Miner. Process. 1981, 8, 289–327. [Google Scholar] [CrossRef]
- Zhang, X.; Han, Y.; Kawatra, S.K. Effects of Grinding Media on Grinding Products and Flotation Performance of Sulfide Ores. Miner. Process. Extr. Met. Rev. 2021, 42, 172–183. [Google Scholar] [CrossRef]
- Molaei, N.; Hoseinian, F.S.; Rezai, B. A Study on the Effect of Active Pyrite on Flotation of Porphyry Copper Ores. Physicochem. Probl. Miner. Process. 2018, 54, 922–933. [Google Scholar]
- Jena, S.; Pattanaik, A.; Venugopal, R. Statistical Analysis of Species Interaction and Optimization of Copper Flotation Process. J. Mines Met. Fuels 2019, 67, 326–331. [Google Scholar]
- Abramov, A.A.; Forssberg, K.S.E. Chemistry and Optimal Conditions for Copper Minerals Flotation: Theory and Practice. Miner. Process. Extr. Met. Rev. 2005, 26, 77–143. [Google Scholar] [CrossRef]
- Bakalarz, A.; Gloy, G.; Luszczkiewicz, A. Flotation of Sulfide Components of Copper Ore in the Presence of n-Dodecane. Miner. Process. Extr. Met. Rev. 2015, 36, 103–111. [Google Scholar] [CrossRef]
- Gül, A. The Role of Na2S2O5and Activated Carbon on the Selective Flotation of Chalcopyrite from a Copper Ore Using a Dithiophosphine-Type Collector. Miner. Process. Extr. Met. Rev. 2007, 28, 235–245. [Google Scholar] [CrossRef]
- Katwika, C.N.; Kime, M.-B.; Kalenga, P.N.M.; Mbuya, B.I.; Mwilen, T.R. Application of Knelson Concentrator for Beneficiation of Copper–Cobalt Ore Tailings. Miner. Process. Extr. Met. Rev. 2019, 40, 35–45. [Google Scholar] [CrossRef]
- Poulter, S.; Fitzmaurice, C.; Steward, G. The Knelson Concentrator: Application and Operation at Rosebery. In Proceedings of the 5th Mill Operators Conf, Roxby Downs, Australia, 16–20 October 1994. [Google Scholar]
- Ramakokovhu, M.M.; Kasaini, H.; Mbaya, R.K. Leaching Characteristics of Upgraded Copper Flotation Tailings. Int. J. Mater. Metall. Eng. 2012, 6, 859–863. [Google Scholar]
- Kime, M.B.; Kaniki, A.T. Determining milling parameters of a pear-shaped ball mill for grinding high-talc oxidized copper-cobalt ore. Econ. Anal. 2017, 8, 136306708. [Google Scholar] [CrossRef]
- Perek, K.T.; Arslan, F. Effect of Mechanical Activation on Pressure Leaching of Küre Massive Rich Copper Ore. Miner. Process. Extr. Met. Rev. 2010, 31, 191–200. [Google Scholar] [CrossRef]
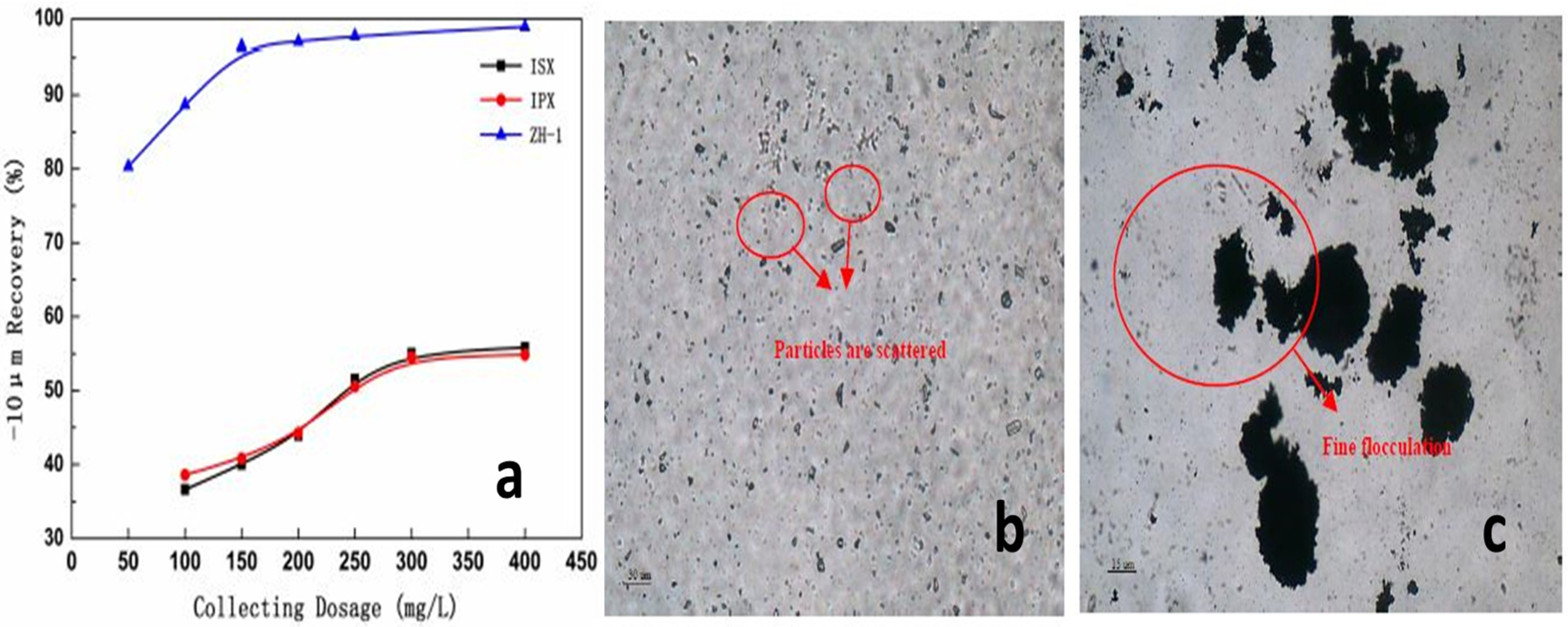
- Mweene, L.; Subramanian, S. Selective Dispersion-Flocculation and Flotation Studies on a Siliceous Copper Ore. Physicochem. Probl. Miner. Process. 2018, 54, 1282–1291. [Google Scholar]
- Patra, P.; Natarajan, K. Microbially induced flocculation and flotation for separation of chalcopyrite from quartz and calcite. Int. J. Miner. Process. 2004, 74, 143–155. [Google Scholar] [CrossRef]
- Acar, S.; Somasundaran, P. Flocculation of sulfides and the role of a complexing agent in it. Int. J. Miner. Process. 1989, 27, 111–123. [Google Scholar] [CrossRef]
- Copper Reserves by Country. 2020. Available online: https://www.statista.com/statistics/273637/copper-reserves-by-country/ (accessed on 7 March 2022).
- Dong, D.; van Oers, L.; Tukker, A.; van der Voet, E. Assessing the Future Environmental Impacts of Copper Production in China: Implications of the Energy Transition. J. Clean. Prod. 2020, 274, 122825. [Google Scholar] [CrossRef]
- International Copper Study Group. The World Copper Factbook; International Copper Study Group: Lisbon, Portugal, 2014. [Google Scholar]
- U.S. Geological Survey. Mineral Commodity Summaries 2020; U.S. Geological Survey: Reston, VA, USA, 2020; p. 204.
- Banger, K.M. Principles of Engineering Geology. Available online: https://firstwaybookshop.com/product/textbook-of-geology-general-and-engineering-by-k-m-bangar/ (accessed on 7 March 2022).
- Sillitoe, R. Copper Provinces. Society of Economic Geologists; Special Publication: London, UK, 2012; Volume 16, pp. 1–18. [Google Scholar]
- Tatsch, J.H. Copper Deposits: Origin, Evolution, and Present Characteristics: An Analysis of the Present Copper Deposits in Terms of the Geometrical, Mechanical, Thermal, and Chemical Aspects of the Earth’s Behavior during the Past 4.6 Billion Years; Tatsch Associates: Coos Bay, OR, USA, 1975; ISBN 0-912890-08-8. [Google Scholar]
- Bateman, A. Economic Mineral Deposits; John Wiley & Sons, Inc.: Carrollton, TX, USA, 1975. [Google Scholar]
- Wills, B.; Napier-Munn, T.; Julius Kruttschnitt Mineral Research Centre. Wills’ Mineral Processing Technology: An Introduction to the Practical Aspects of Ore Treatment and Mineral Recovery; Butterworth-Heinemann: Oxford, UK, 2006. [Google Scholar]
- Fuerstenau, M.; Jameson, G.; Yoon, R. Froth Flotation: A Century of Innovation; Society for Mining, Metallurgy, and Exploration Inc.: Littleton, CO, USA, 2007; 8975p. [Google Scholar]
- Ancia, P. Comparison of the Knelson and Falcon Centrifugal Separators, Proceedings of the Proc. Conf. on Inovation in Physical Separation Technologies, Richard Mozley Symp., Falmouth, UK, 4–5 June 1997; Mozeley, R., Ed.; University of Liège: Liège, Belgium, 1997; pp. 53–62. [Google Scholar]
- Jena, S.S.; Angadi, S.I.; Mandre, N.R.; Venugopal, R. An Investigation into Pre-concentration of Low-Grade Silica-Rich Malanjkhand Copper Ore by Wilfley Table. Trans. Indian Inst. Met. 2021, 74, 571–581. [Google Scholar] [CrossRef]
- Carta, M.; Ghiani, M.; Rossi, G. Beneficiation of a Complex Sulfide Ore by an Integrated Process of Flotation and Bioleaching. Complex Sulphide Ores 1980, 178–185. [Google Scholar]
- Chadwick, J. CAMEC—The Cobalt Champion, Reports on What Is Today Probably the Leading Cobalt Miner in the World. Int. Min. 2008, 8–16. [Google Scholar]
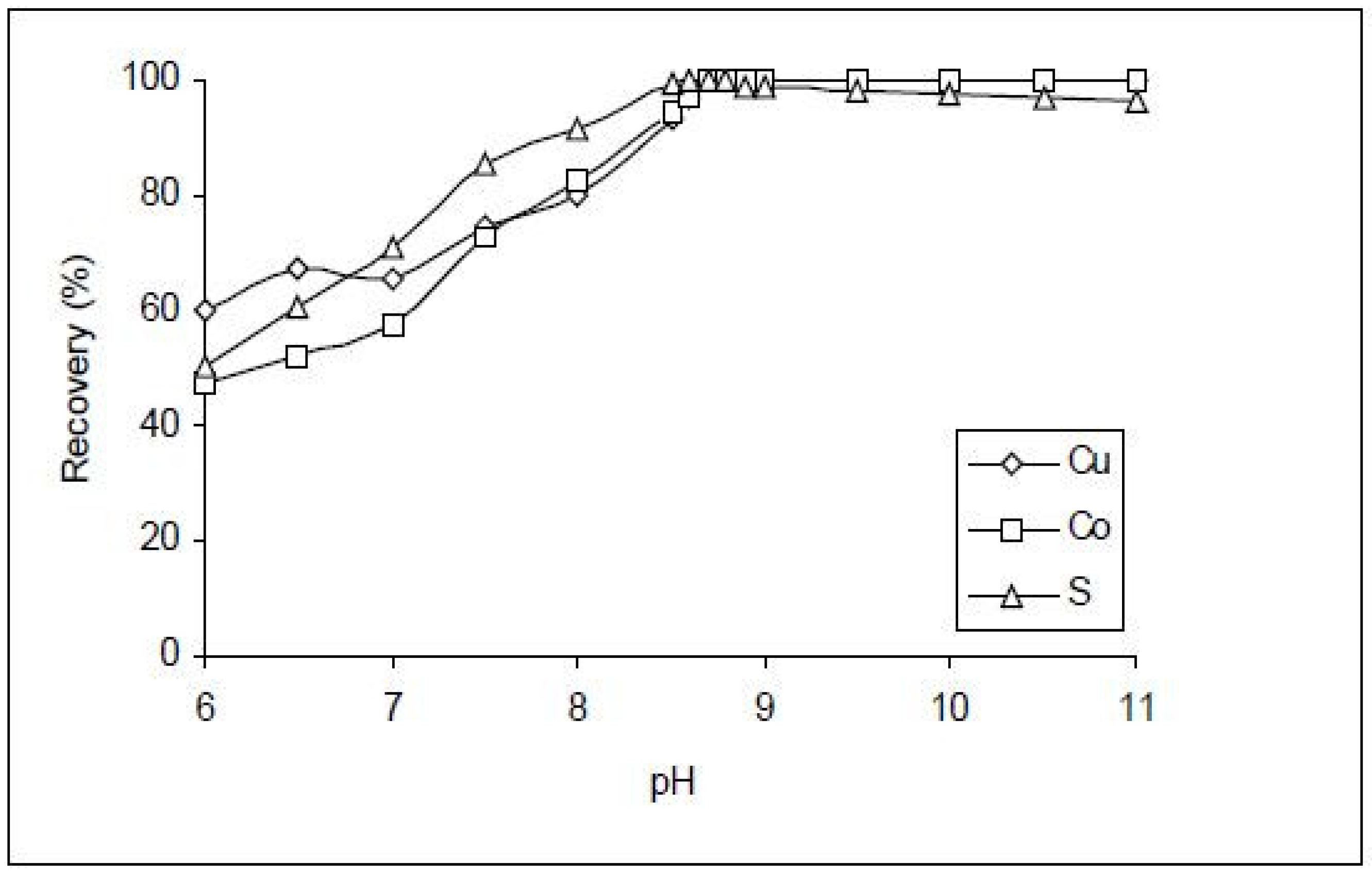
- Lutandula, M.S.; Maloba, B. Recovery of cobalt and copper through reprocessing of tailings from flotation of oxidised ores. J. Environ. Chem. Eng. 2013, 1, 1085–1090. [Google Scholar] [CrossRef]
- Jena, S.S.; Gharai, M.; Mandre, N.R.; Venugopal, R. Mineralogical Characterization and Gravity Separation of Lean Grade Mixed Copper Ore of Malanjkhand Deposit. Trans. Indian Inst. Met. 2019, 72, 245–255. [Google Scholar] [CrossRef]
- Yarar, B.; Kitchener, J. Selective Flocculation of Minerals. Pt. 1. Basic Principles. Pt. 2. Experimental Investigation of Quartz, Calcite, and Galena. Trans. Inst. Min. Met. 1970, 79, 760. [Google Scholar]
- Sresty, G.; Somasundaran, P. Selective flocculation of synthetic mineral mixtures using modified polymers. Int. J. Miner. Process. 1980, 6, 303–320. [Google Scholar] [CrossRef]
- Ackerman, P.; Harris, G.; Klimpel, R.; Aplan, F. Evaluation of flotation collectors for copper sulfides and pyrite, I. Common sulfhydryl collectors. Int. J. Miner. Process. 1987, 21, 105–127. [Google Scholar] [CrossRef]
- Attia, Y.; Kitchener, J. Development of Complexing Polymers for the Selective Flocculation of Copper Minerals. In Proceedings of the 11th International Mineral Processing Congress, Cagliara, Italy, 20–26 April 1975. [Google Scholar]
- Heyes, G.; Trahar, W. The Natural Flotability of Chalcopyrite. Int. J. Miner. Process. 1977, 4, 317–344. [Google Scholar] [CrossRef]
- Yoon, R. Collectorless flotation of chalcopyrite and sphalerite ores by using sodium sulfide. Int. J. Miner. Process. 1981, 8, 31–48. [Google Scholar] [CrossRef]
- Luttrell, G.H.; Yoon, R.-H. The collectorless flotation of chalcopyrite ores using sodium sulfide. Int. J. Miner. Process. 1984, 13, 271–283. [Google Scholar] [CrossRef]
- Mitchell, T.K.; Nguyen, A.; Evans, G.M. Heterocoagulation of chalcopyrite and pyrite minerals in flotation separation. Adv. Colloid Interface Sci. 2005, 114, 227–237. [Google Scholar] [CrossRef]
- Gardner, J.; Woods, R. An electrochemical investigation of the natural flotability of chalcopyrite. Int. J. Miner. Process. 1979, 6, 1–16. [Google Scholar] [CrossRef]
- Trahar, W. A laboratory study of the influence of sodium sulphide and oxygen on the collectorless flotation of chalcopyrite. Int. J. Miner. Process. 1983, 11, 57–74. [Google Scholar] [CrossRef]
- Allison, S.A.; Goold, L.A.; Nicol, M.; Granville, A. A determination of the products of reaction betweer various sulfide minerals and aqueous xanthate solution, and a correlation of the products with electrode rest potentials. Metall. Trans. 1972, 3, 2613–2618. [Google Scholar] [CrossRef]
- Leppinen, J.; Basilio, C.; Yoon, R. In-situ FTIR study of ethyl xanthate adsorption on sulfide minerals under conditions of controlled potential. Int. J. Miner. Process. 1989, 26, 259–274. [Google Scholar] [CrossRef]
- Venugopal, R.; Viswanatha, D.; Sanyal, P.; Rao, T.C. Flotation Characteristics of Chalcopyrite Ore. Inst. Eng. J.-MN 1993, 74, 12–16. [Google Scholar]
- Ram, A.; Venugopal, R.; Rao, T. Reagent-Particle Size Interaction in Copper Ore Flotation. Trans. Indian Inst. Met. 1990, 43, 356–362. [Google Scholar]
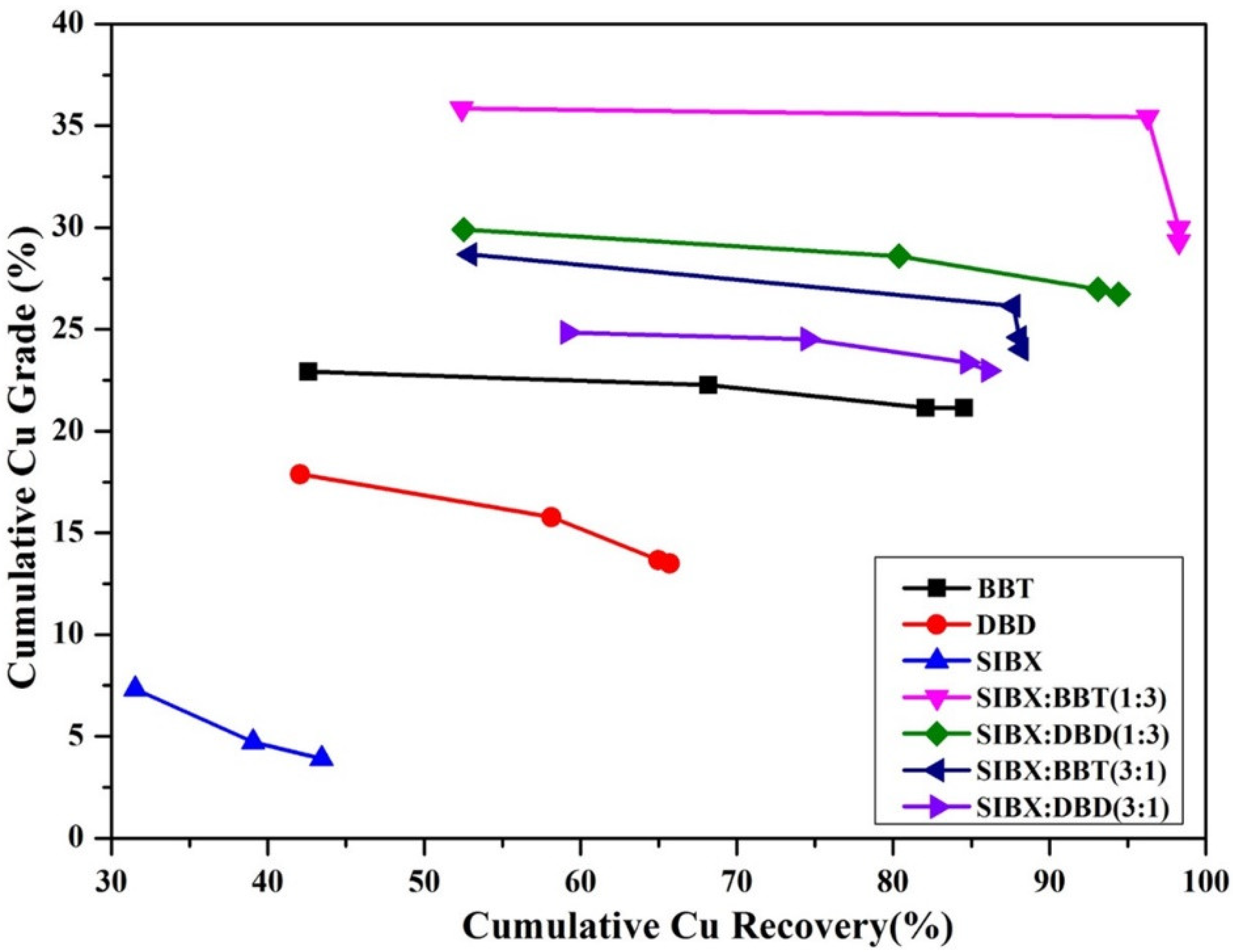
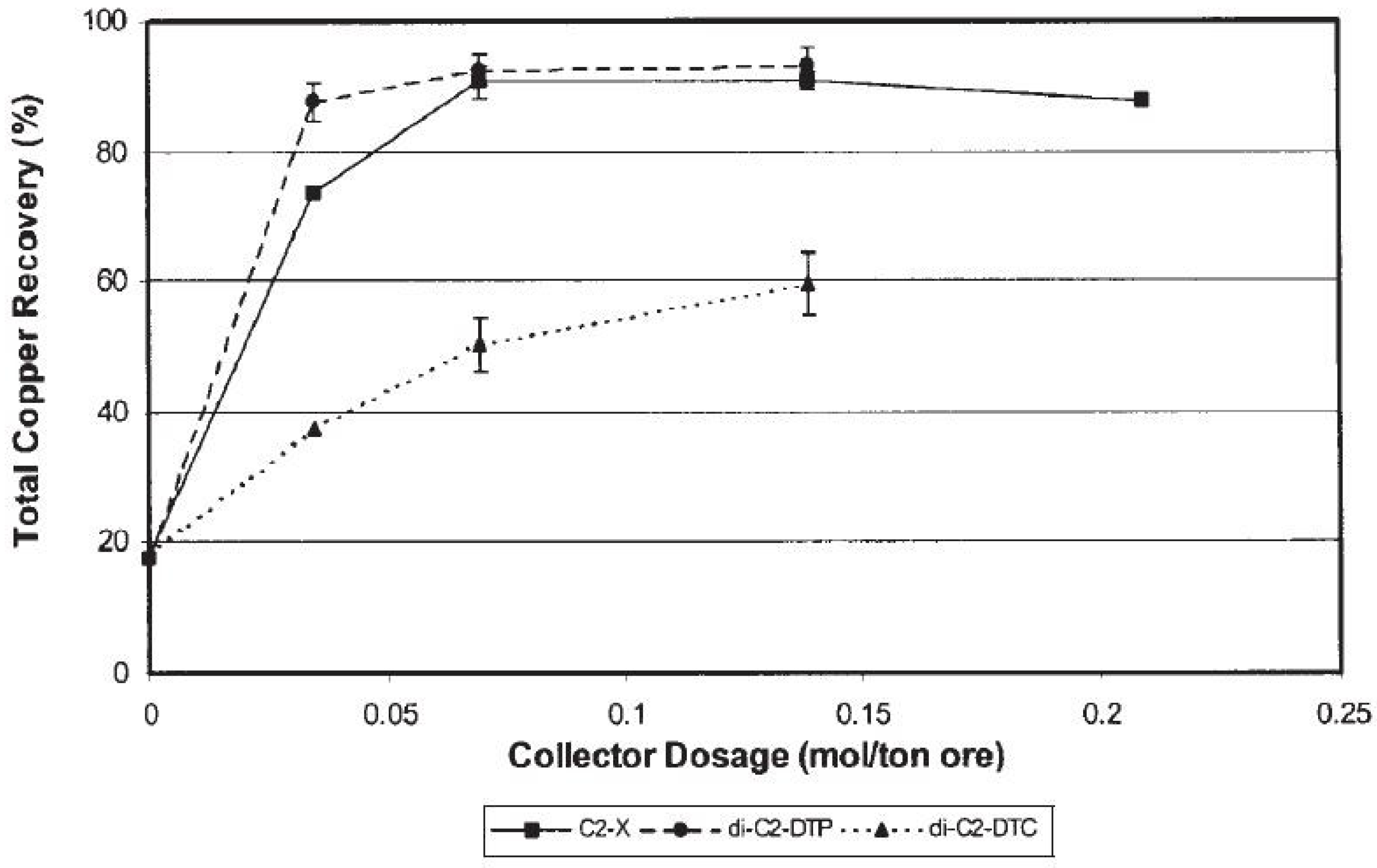
- Dhar, P.; Thornhill, M.; Kota, H.R. Comparison of single and mixed reagent systems for flotation of copper sulphides from Nussir ore. Miner. Eng. 2019, 142, 105930. [Google Scholar] [CrossRef]
- Dhar, P.; Thornhill, M.; Kota, H.R. Investigation of Copper Recovery from a New Copper Ore Deposit (Nussir) in Northern Norway: Dithiophosphates and Xanthate-Dithiophosphate Blend as Collectors. Minerals 2019, 9, 146. [Google Scholar] [CrossRef] [Green Version]
- Dhar, P.; Thornhill, M.; Kota, H.R. Investigation of Copper Recovery from a New Copper Deposit (Nussir) in Northern Norway. Miner. Process. Extr. Met. Rev. 2019, 40, 380–389. [Google Scholar] [CrossRef] [Green Version]
- Owusu, C.; Fornasiero, D.; Addai-Mensah, J.; Zanin, M. Influence of pulp aeration on the flotation of chalcopyrite with xanthate in chalcopyrite/pyrite mixtures. Int. J. Miner. Process. 2015, 134, 50–57. [Google Scholar] [CrossRef]
- Hangone, G.; Bradshaw, D.; Ekmekci, Z. Flotation of a Copper Sulphide Ore from Okiep Using Thiol Collectors and Their Mixtures. J. South. Afr. Inst. Min. Metall. 2005, 105, 199–206. [Google Scholar]
- Finkelstein, N.; Poling, G. The Role of Dithiolates in the Flotation of Sulphide Minerals; Mintek: Randburg, South Africa, 1977. [Google Scholar]
- Fuerstenau, M. Thiol Collector Adsorption Processes. US Bur. Mines Inf. Circ. 1980, 8818, 7–23. [Google Scholar]
- Harris, P.; Finkelstein, N. The Interaction of Chalcocite, Oxygen and Xanthates, National Institute for Metallurgy, Randburg, South Africa. Report No. 1895. Int. J. Mineral Process. 1977, 12, 33–37. [Google Scholar]
- Fuerstenau, M. Chemistry of Collectors in Solution. South African Institute of Mining and Metallurgy. In Principles of Flotation; SAIMM: Johannesburg, South Africa, 1982; pp. 1–16. [Google Scholar]
- Sheridan, M.; Nagaraj, D.; Fornasiero, D.; Ralston, J. The use of a factorial experimental design to study collector properties of N-allyl-O-alkyl thionocarbamate collector in the flotation of a copper ore. Miner. Eng. 2002, 15, 333–340. [Google Scholar] [CrossRef]
- Wang, D. Flotation Reagents: Applied Surface Chemistry on Minerals Flotation and Energy Resources Beneficiation; Springer: Berlin/Heidelberg, Germany, 2016; ISBN 981-102-028-0. [Google Scholar]
- Hanson, J.S.; Fuerstenau, D.W. The electrochemical and flotation behavior of chalcocite and mixed oxide/sulfide ores. Int. J. Miner. Process. 1991, 33, 33–47. [Google Scholar] [CrossRef]
- Twidle, T.R.; Engelbrecht, P.C. Developments in the Flotation of Copper at Black Mountain. J. South. Afr. Inst. Min. Metall. 1984, 84, 164–178. [Google Scholar]
- Liu, G.; Zhong, H.; Xia, L.; Wang, S.; Xu, Z. Improving copper flotation recovery from a refractory copper porphyry ore by using ethoxycarbonyl thiourea as a collector. Miner. Eng. 2011, 24, 817–824. [Google Scholar] [CrossRef]
- Xiong, K.; Zheng, G.S. Process Mineralogy and Flotation Kinetic of a Copper Oxide Ore during Sulfuration Flotation; Trans Tech Publication: Freienbach, Switzerland, 2013; Volume 634, pp. 3460–3465. [Google Scholar]
- Farrokhpay, S.; Ametov, I.; Grano, S. Improving the recovery of low grade coarse composite particles in porphyry copper ores. Adv. Powder Technol. 2011, 22, 464–470. [Google Scholar] [CrossRef]
- Farrokhpay, S. Effect of Feed Classification by Hydrocycloning on Copper Recovery in Flotation. Can. Metall. Quart. 2010, 49, 107–112. [Google Scholar] [CrossRef]
- Chander, S.; Fuerstenau, D. The Effect of Potassium Diethyldithiophosphate on the Electrochemical Properties of Platinum, Copper and Copper Sulfide in Aqueous Solutions. J. Electroanal. Chem. Interfacial Electrochem. 1974, 56, 217–247. [Google Scholar] [CrossRef]
- Schlesinger, M.E.; Sole, K.C.; Davenport, W.G.; Alvear, G.R. Extractive Metallurgy of Copper; Elsevier: Amsterdam, The Netherlands, 2021; ISBN 012-821-903-3. [Google Scholar]
- Ge, B.L.; Fu, Y.X.; Li, Q. A Copper Oxide Ore Treatment by Flotation; Trans Tech Publication: Freienbach, Switzerland, 2013; Volume 813, pp. 230–233. [Google Scholar]
- Bulatovic, S. Flotation of Oxide Copper and Copper Cobalt Ores. In Handbook of Flotation Reagents: Chemistry, Theory and Practice; Elsevier: Amsterdam, The Netherlands, 2010; Volume 2, pp. 47–65. [Google Scholar]
- Herrera-Urbina, R.; Laskowski, J.; Fuerstenau, D. A Process for the Flotation of Chrysocolla. In Proceedings of the In XXV International Mineral Processing Congress Proceeding, Brisbane, Australia, 6–10 September 2010; pp. 1959–1969. [Google Scholar]
- Lee, K.; Archibald, D.; McLean, J.; Reuter, M.A. Flotation of mixed copper oxide and sulphide minerals with xanthate and hydroxamate collectors. Miner. Eng. 2009, 22, 395–401. [Google Scholar] [CrossRef]
- Lenormand, J.; Salman, T.; Yoon, R. Hydroxamate Flotation of Malachite. Can. Metall. Q. 1979, 18, 125–129. [Google Scholar] [CrossRef]
- Ziyadanogullari, R.; Aydin, F. A New Application for Flotation of Oxidized Copper Ore. J. Miner. Mater. Charact. Eng. 2005, 4, 67–73. [Google Scholar] [CrossRef]
- Glembotskii, V.; Klassen, V.; Plaksin, I. Flotation; Primary Sources: New York, NY, USA, 1972. [Google Scholar]
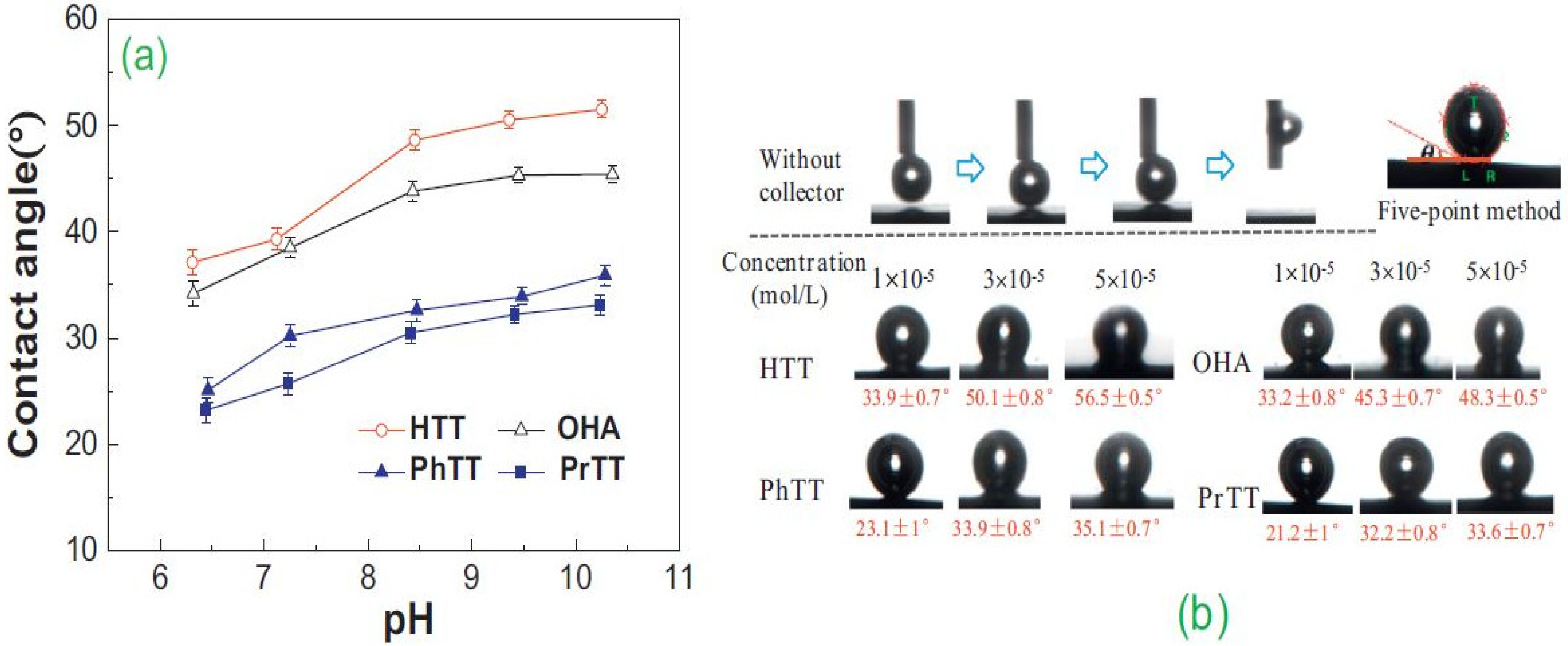
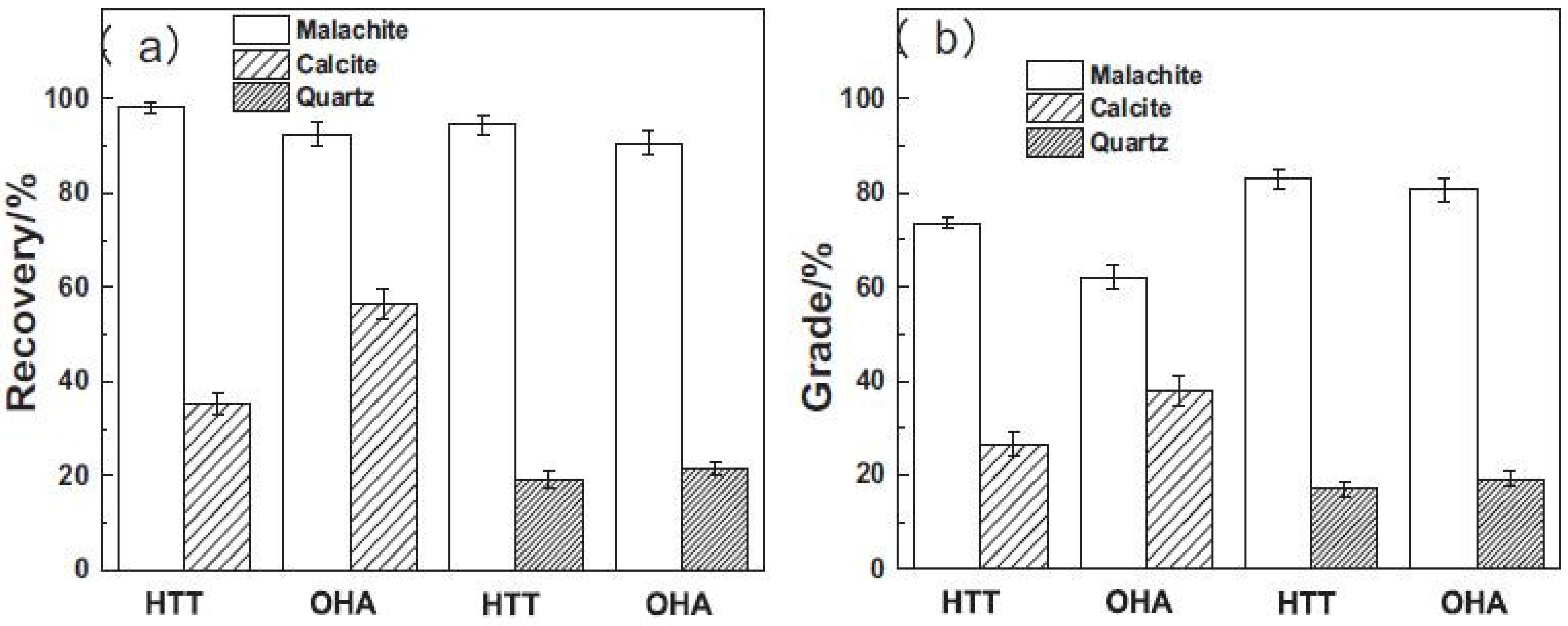
- Marion, C.; Jordens, A.; Li, R.; Rudolph, M.; Waters, K. An evaluation of hydroxamate collectors for malachite flotation. Sep. Purif. Technol. 2017, 183, 258–269. [Google Scholar] [CrossRef]
- Choi, J.; Choi, S.Q.; Park, K.; Han, Y.; Kim, H. Flotation behaviour of malachite in mono- and di-valent salt solutions using sodium oleate as a collector. Int. J. Miner. Process. 2016, 146, 38–45. [Google Scholar] [CrossRef]
- Li, F.; Zhong, H.; Xu, H.; Jia, H.; Liu, G. Flotation behavior and adsorption mechanism of α-hydroxyoctyl phosphinic acid to malachite. Miner. Eng. 2015, 71, 188–193. [Google Scholar] [CrossRef]
- Peterson, H.; Fuerstenau, M.; Rickard, R.; Miller, J. Chrysocolla Flotation by the Formation of Insoluble Surface Chelates. Trans. Am. Inst. Min. Eng. 1965, 232, 388–392. [Google Scholar]
- Yang, X.F.; Liu, Q.J.; Deng, R.D. Flotation of Copper Oxide Minerals Using Ethylene Phosphate as Activators; Trans Tech Publication: Freienbach, Switzerland, 2012; Volume 581, pp. 975–982. [Google Scholar]
- Lee, J.; Nagaraj, D.; Coe, J. Practical aspects of oxide copper recovery with alkyl hydroxamates. Miner. Eng. 1998, 11, 929–939. [Google Scholar] [CrossRef]
- Feng, Q.; Zhao, W.; Wen, S.; Cao, Q. Copper sulfide species formed on malachite surfaces in relation to flotation. J. Ind. Eng. Chem. 2017, 48, 125–132. [Google Scholar] [CrossRef]
- Zhu, R.; Gu, G.; Chen, Z.; Wang, Y.; Song, S. A New Collector for Effectively Increasing Recovery in Copper Oxide Ore-Staged Flotation. Minerals 2019, 9, 595. [Google Scholar] [CrossRef] [Green Version]
- Rao, S.; Finch, J. Base metal oxide flotation using long chain xanthates. Int. J. Miner. Process. 2002, 69, 251–258. [Google Scholar] [CrossRef]
- Li, Z.; Rao, F.; Lou, X.; Song, S.; López-Valdivieso, A. Floc-Flotation of Malachite Fines with an Octyl Hydroxamate and Kerosene Mixture. Minerals 2019, 9, 301. [Google Scholar] [CrossRef] [Green Version]
- Sun, Q.-Y.; Yin, W.-Z.; Li, D.; Fu, Y.-F.; Xue, J.-W.; Yao, J. Improving the sulfidation−flotation of fine cuprite by hydrophobic flocculation pretreatment. Int. J. Miner. Met. Mater. 2018, 25, 1256–1262. [Google Scholar] [CrossRef]
- Liu, J.; Hu, Z.; Liu, G.; Huang, Y.; Zhang, Z. Selective Flotation of Copper Oxide Minerals with A Novel Amino-Triazole-Thione Surfactant: A Comparison to Hydroxamic Acid Collector. Miner. Process. Extr. Met. Rev. 2019, 41, 96–106. [Google Scholar] [CrossRef]
- Fuerstenau, D.; Herrera-Urbina, R.; Laskowski, J. Surface Properties and Flotation Behaviour of Chrysocolla in the Presence of Potassium Octylhydroxamate. In Proceedings of the 2nd Latin-American Congr. Froth Flotation, Concepcion, Chile, 19–23 August 1985; pp. 19–23. [Google Scholar]
- Liu, J.; Liu, G.; Huang, Y.; Zhang, Z. Tetrazinan-thione collectors for copper oxide mineral: Synthesis and flotation mechanism. Appl. Surf. Sci. 2019, 491, 624–632. [Google Scholar] [CrossRef]
- Aplan, F.; Fuerstenau, D. The Flotation of Chrysocolla by Mercaptan. Int. J. Miner. Process. 1984, 13, 105–115. [Google Scholar] [CrossRef]
- Barbaro, M.; Urbina, R.H.; Cozza, C.; Fuerstenau, D.; Marabini, A. Flotation of oxidized minerals of copper using a new synthetic chelating reagent as collector. Int. J. Miner. Process. 1997, 50, 275–287. [Google Scholar] [CrossRef]
- Nagaraj, D.; Somasundaran, P. Commercial Chelating Extractants as Collectors: Flotation of Copper Minerals Using” LIX” Reagents. Trans. Am. Inst. Min. Engrs. 1979, 266, 1892–1897. [Google Scholar]
- Xiong, K.; Wen, S.M.; Zheng, G.S.; Bai, S.J.; Shen, H.Y. Flotation Research on Cuprite-Type Oxide Copper in XinJiang; Trans Tech Publication: Freienbach, Switzerland, 2012; Volume 524, pp. 987–992. [Google Scholar]
- Han, G.; Wen, S.; Wang, H.; Feng, Q. Surface sulfidization mechanism of cuprite and its response to xanthate adsorption and flotation performance. Miner. Eng. 2021, 169, 106982. [Google Scholar] [CrossRef]
- Han, G.; Wen, S.; Wang, H.; Feng, Q. Sulfidization regulation of cuprite by pre-oxidation using sodium hypochlorite as an oxidant. Int. J. Min. Sci. Technol. 2021, 31, 1117–1128. [Google Scholar] [CrossRef]
- Rule, W.T. Recovery of Copper from Copper Oxide Minerals. U.S. Patent 4324654A, 13 April 1982. [Google Scholar]
- Wang, Y.J.; Wen, S.M.; Liu, D.; Cao, Q.B.; Feng, Q.C.; Lv, C. Sulphidizing Flotation of Copper Oxide Ore; Trans Tech Publication: Freienbach, Switzerland, 2013; Volume 807, pp. 2279–2283. [Google Scholar]
- Zhao, Y.; Liu, S.; Liu, H.; Wang, D.; Li, T. Flotation Test of a High Silica Copper Oxide Ore in Yunnan; Atlantis Press: Paris, France, 2015. [Google Scholar]
- Saleh, A.-H.M.; Ramadan, A.M.; Moharam, M.R. Beneficiation of Egyptian Abu-Swayel Copper Ore by Flotation. Physicochem. Probl. Miner. Process. 2008, 42, 119–130. [Google Scholar]
- Singh, R.; Chandrashekhar, S.; Rath, R.; Nayak, B.; Bhattacharyya, K. Designing Process for Concentration of a Low Grade Copper Ore. In Proceedings of the International Seminar on Mineral Processing Technology, Trivandrum, India, 22–24 April 2008; pp. 1–6. [Google Scholar]
- Arakatsu, A.; Nakazawa, H.; Naruse, H. Rotating multitube biocontactor for treating sewage. U.S. Patent No. 4,022,689, 10 May 1977. [Google Scholar]
- Pang, W.H.; Liu, Q.J.; Ding, P. Experimental Research on Flotation of Copper Oxide Ore in Guizhou; Trans Tech Publication: Freienbach, Switzerland, 2013; Volume 813, pp. 238–241. [Google Scholar]
- Ou, L.M.; Yin, B.Y. A Flotation Technique for a Sulfide-Oxidized Cu-Co Mixed Ore; Trans Tech Publication: Freienbach, Switzerland, 2012; Volume 402, pp. 564–571. [Google Scholar]
- Xiong, F.; Li, Y.J.; Zhang, Z.H.; Lan, Y.Z. Flotation Responce of the Refractory Yangla Copper Oxide Ores with Hydroxamate Collectors; Trans Tech Publication: Freienbach, Switzerland, 2013; Volume 803, pp. 131–136. [Google Scholar]
- Xiong, F.; Li, Y.J.; Zhang, Z.; Du, G.F.; Lan, Y.Z. Application of Chelating Collectors in the Flotation of Copper Minerals; Trans Tech Publication: Freienbach, Switzerland, 2013; Volume 868, pp. 417–422. [Google Scholar]
- Cao, Z.-F.; Zhong, H.; Liu, G.-Y.; Zhao, S.-J. Techniques of copper recovery from Mexican copper oxide ore. Min. Sci. Technol. 2009, 19, 45–48. [Google Scholar] [CrossRef]
- Prabhakar, S.; Bhaskar Raju, G.; Misra, V.; Khangaonkar, P. Studies on the Flotation of Mixed Oxide-Sulphide Ores of Copper from Malanjkhand Deposit India. In Minerals, Materials and Industry, Proceedings of the 14th Congress of the Council of Mining and Metallurgy Institutions, Edinburgh, Scotland, 2–6 July 1990; The Institution of Mining and Metallurgy: London, UK, 1990. [Google Scholar]
- Phetla, T.P.; Muzenda, E. A Multistage Sulphidisation Flotation Procedure for a Low Grade Malachite Copper Ore. Int. J. Chem. Nucl. Mater. Metall. Eng. 2010, 4, 26–32. [Google Scholar]
- Subramanian, S.; Natarajan, K. Zeta Potential Studies on Some Iron Ore Minerals in the Presence of Starches. Trans. Indian Inst. Met. 1988, 41, 255–263. [Google Scholar]
- Quast, K. A review of hematite flotation using 12-carbon chain collectors. Miner. Eng. 2000, 13, 1361–1376. [Google Scholar] [CrossRef]
- Kallay, N.; Torbić, Z.; Barouch, E.; Jednačak-Bišćan, J. The determination of isoelectric point for metallic surfaces. J. Colloid Interface Sci. 1987, 118, 431–435. [Google Scholar] [CrossRef]
- Salopek, B.; Krasic, D.; Filipovic, S. Measurement and Application of Zeta-Potential. Rud.-Geol.-Naft. Zb. 1992, 4, 147. [Google Scholar]
- Ottewill, R.H. The Rideal Lecture. Colloidal Dispersions. Faraday Discuss. Chem. Soc. 1990, 90, 1–15. [Google Scholar] [CrossRef]
- Mokone, T.; van Hille, R.; Lewis, A. Effect of solution chemistry on particle characteristics during metal sulfide precipitation. J. Colloid Interface Sci. 2010, 351, 10–18. [Google Scholar] [CrossRef]
- Kosmulski, M. Surface Charging and Points of Zero Charge; CRC Press: Boca Raton, FL, USA, 2009; ISBN 042-909-339-X. [Google Scholar]
- Reyes-Bozo, L.; Escudey, M.; Vyhmeister, E.; Higueras, P.; Godoy-Faúndez, A.; Salazar, J.L.; Valdés-González, H.; Wolf-Sepúlveda, G.; Herrera-Urbina, R. Adsorption of biosolids and their main components on chalcopyrite, molybdenite and pyrite: Zeta potential and FTIR spectroscopy studies. Miner. Eng. 2015, 78, 128–135. [Google Scholar] [CrossRef]
- Rath, R.; Subramanian, S.; Sivanandam, V.; Pradeep, T. Studies on the Interaction of Guar Gum with Chalcopyrite. Can. Metall. Q. 2001, 40, 1–11. [Google Scholar] [CrossRef]
- Júnior, J.A.A.; Baldo, J.B. The Behavior of Zeta Potential of Silica Suspensions. New J. Glass Ceram. 2014, 4, 29–37. [Google Scholar] [CrossRef] [Green Version]
- Fullston, D.; Fornasiero, D.; Ralston, J. Zeta Potential Study of the Oxidation of Copper Sulfide Minerals. Colloids Surf. A Physicochem. Eng. Asp. 1999, 146, 113–121. [Google Scholar] [CrossRef]
- Fan, H.; Qin, J.; Liu, J.; Liu, G.; Yang, X. Investigation into the flotation of malachite, calcite and quartz with three phosphate surfactants. J. Mater. Res. Technol. 2019, 8, 5140–5148. [Google Scholar] [CrossRef]
- Liu, G.; Huang, Y.; Qu, X.; Xiao, J.; Yang, X.; Xu, Z. Understanding the Hydrophobic Mechanism of 3-Hexyl-4-Amino-1, 2, 4-Triazole-5-Thione to Malachite by ToF-SIMS, XPS, FTIR, Contact Angle, Zeta Potential and Micro-Flotation. Colloids Surf. A Physicochem. Eng. Asp. 2016, 503, 34–42. [Google Scholar] [CrossRef]
- Liu, G.; Xiao, J.; Liu, J.; Qu, X.; Liu, Q.; Zeng, H.; Yang, X.; Xie, L.; Zhong, H.; Liu, Q. In Situ Probing the Self-Assembly of 3-Hexyl-4-Amino-1, 2, 4-Triazole-5-Thione on Chalcopyrite Surfaces. Colloids Surf. A Physicochem. Eng. Asp. 2016, 511, 285–293. [Google Scholar] [CrossRef]
- Shen, P.; Liu, D.; Xu, X.; Jia, X.; Zhang, X.; Liu, D.; Liu, R. Effect of Ethylene Diamine Phosphate on the Sulfidization Flotation of Chrysocolla. Minerals 2018, 8, 216. [Google Scholar] [CrossRef] [Green Version]
- González, G.; Soto, H. The effect of thermal treatment on the flotation of chrysocolla. Int. J. Miner. Process. 1978, 5, 153–162. [Google Scholar] [CrossRef]
- Gonzalez, G.; Laskowski, J. The Point of Zero Charge of Oxidized Copper Minerals: Tenorite, Malachite and Chrysocolla. J. Electroanal. Chem. Interfacial Electrochem. 1974, 53, 452–456. [Google Scholar] [CrossRef]
- Li, Z.; Rao, F.; Song, S.; Uribe-Salas, A.; López-Valdivieso, A. Reexamining the Adsorption of Octyl Hydroxamate on Malachite Surface: Forms of Molecules and Anions. Miner. Process. Extr. Met. Rev. 2019, 41, 178–186. [Google Scholar] [CrossRef]
- Ellis, J. Analysis of accidents and incidents occurring during transport of packaged dangerous goods by sea. Saf. Sci. 2011, 49, 1231–1237. [Google Scholar] [CrossRef]
- Singh, K.; Ihlenfeld, C.; Oates, C.; Plant, J.; Voulvoulis, N. Developing a screening method for the evaluation of environmental and human health risks of synthetic chemicals in the mining industry. Int. J. Miner. Process. 2011, 101, 1–20. [Google Scholar] [CrossRef]
- Boening, D. Aquatic Toxicity and Environmental Fate of Xanthates. Min. Eng. 1998, 50, 65–68. [Google Scholar]
- Young, T.L.; Greene, M.G.; Bauer, K.; Reber, N.R.; Young, S.K. Flotation of Sulfide Mineral Species with Oils. U.S. Patent 6,827,220, 7 December 2004. [Google Scholar]
- Greene, M.G.; Walton, K.B.; Dimas, P.A.; Laney, D.G.; Young, S.K.; Young, T.L.; Reber, N.R. Collectors for Flotation of Molybdenum-Containing Ores. U.S. Patent 12/963,712, 14 June 2012. [Google Scholar]
- Owusu, C.; Quast, K.; Addai-Mensah, J. The use of canola oil as an environmentally friendly flotation collector in sulphide mineral processing. Miner. Eng. 2016, 98, 127–136. [Google Scholar] [CrossRef]
- Alvarez, J.; Castro, S. Flotation of Chalcocite and Chalcopyrite in Seawater and Salty Water. In Proceedings of the IV EncontroNacional de Tratamento de Minerios, Anais, São José Dos Campos, Brazil, 2–6 May 1976; Volume 1, pp. 39–44. [Google Scholar]
- Castro, S. Challenges in Flotation of Cu-Mo Sulfide Ores in Sea Water. Water Miner. Process. 2012, 29–40. [Google Scholar]
- Smith, L.; Heyes, G. The Effect of Water Quality on the Collectorless Flotation of Chalcopyrite and Bornite. In Proceedings of the Third International Congress on Water management in the Mining Industry, Santiago, Chile, 6–8 June 2012. [Google Scholar]
- Jeldres, R.I.; Forbes, L.; Cisternas, L.A. Effect of Seawater on Sulfide Ore Flotation: A Review. Miner. Process. Extr. Metall. Rev. 2016, 37, 369–384. [Google Scholar] [CrossRef]
- Bakalarz, A.; Duchnowska, M.; Luszczkiewicz, A. The Effect of Process Water Salinity on Flotation of Copper Ore from Lubin Mining Region (SW Poland); EDP Sciences: Les Ulis, France, 2017; Volume 18, p. 01007. [Google Scholar]
- Agboola, O.; Babatunde, D.E.; Fayomi, O.S.I.; Sadiku, E.R.; Popoola, P.; Moropeng, L.; Yahaya, A.; Mamudu, O.A. A review on the impact of mining operation: Monitoring, assessment and management. Results Eng. 2020, 8, 100181. [Google Scholar] [CrossRef]
- Falagán, C.; Grail, B.M.; Johnson, D.B. New approaches for extracting and recovering metals from mine tailings. Miner. Eng. 2017, 106, 71–78. [Google Scholar] [CrossRef]
- Gordon, R.B. Production residues in copper technological cycles. Resour. Conserv. Recycl. 2002, 36, 87–106. [Google Scholar] [CrossRef]
- Chen, T.; Lei, C.; Yan, B.; Xiao, X. Metal recovery from the copper sulfide tailing with leaching and fractional precipitation technology. Hydrometallurgy 2014, 147, 178–182. [Google Scholar] [CrossRef]
- Yin, Z.; Sun, W.; Hu, Y.; Zhang, C.; Guan, Q.; Wu, K. Evaluation of the possibility of copper recovery from tailings by flotation through bench-scale, commissioning, and industrial tests. J. Clean. Prod. 2018, 171, 1039–1048. [Google Scholar] [CrossRef]
- Zhu, X.-N.; Nie, C.-C.; Zhang, H.; Lyu, X.-J.; Tao, Y.-J.; Qiu, J.; Li, L.; Zhang, G.-W. Recovery of high-grade copper from waste printed circuit boards by mechanical-grinding assisted flotation. J. Clean. Prod. 2019, 232, 1251–1256. [Google Scholar] [CrossRef]
- Garmsiri, M.R.; Nosrati, A. Dewatering of Copper Flotation Tailings: Effect of Feed Dilution on the Thickener Performance. Miner. Process. Extr. Met. Rev. 2019, 40, 141–147. [Google Scholar] [CrossRef]
- Vazifeh, Y.; Jorjani, E.; Bagherian, A. Optimization of reagent dosages for copper flotation using statistical technique. Trans. Nonferrous Met. Soc. China 2010, 20, 2371–2378. [Google Scholar] [CrossRef]
- Seaman, D.; Burns, F.; Adamson, B.; Seaman, B.; Manton, P. Telfer Processing Plant Upgrade–the Implementation of Additional Cleaning Capacity and the Regrinding of Copper and Pyrite Concentrates. In Proceedings of the 11th Mill Operators Conference, Hobart, Australia, 29–31 October 2012; pp. 373–381. [Google Scholar]
- Van Eck, N.J.; Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 2010, 84, 523–538. [Google Scholar] [CrossRef] [Green Version]
- Shastri, S.; Raju, K.; Rao, K.; Rao, G. Mineral Resources and Beneficiation Plant Practices in India. Miner. Eng. 2000, 14, 1665. [Google Scholar]
- Ruiz, J.; Lizardo, J.; Umipig, C.; Ccuno, F. The Implementation Processing Plant Modifications at Cormidom, Cerro De Maimon, Perilya. In Proceedings of the IMPC 2016: XXVIII International Mineral Processing Congress Proceedings, Quebec City, QC, Canada, 11 September 2016; Volume 4, pp. 240–250. [Google Scholar]
- Kaya, E. Comminution Behaviour of Microwave Heated Two Sulphide Copper Ores. Indian J. Chem. Technol. 2010, 17, 455–461. [Google Scholar]
- Azghdi, S.; Barani, K. Effect of microwave treatment on the surface properties of chalcopyrite. Miner. Met. Process. 2018, 35, 141–147. [Google Scholar] [CrossRef]
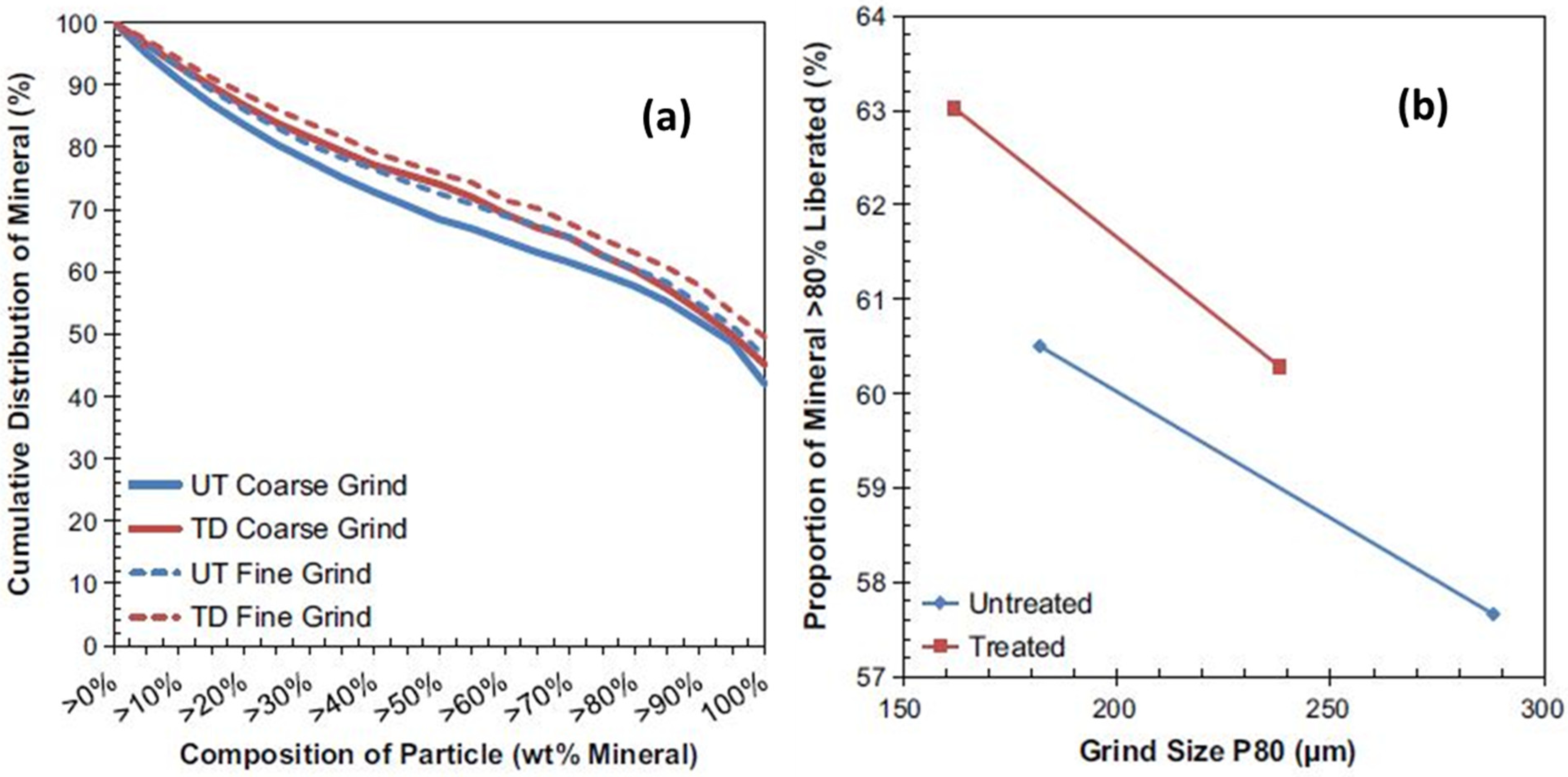
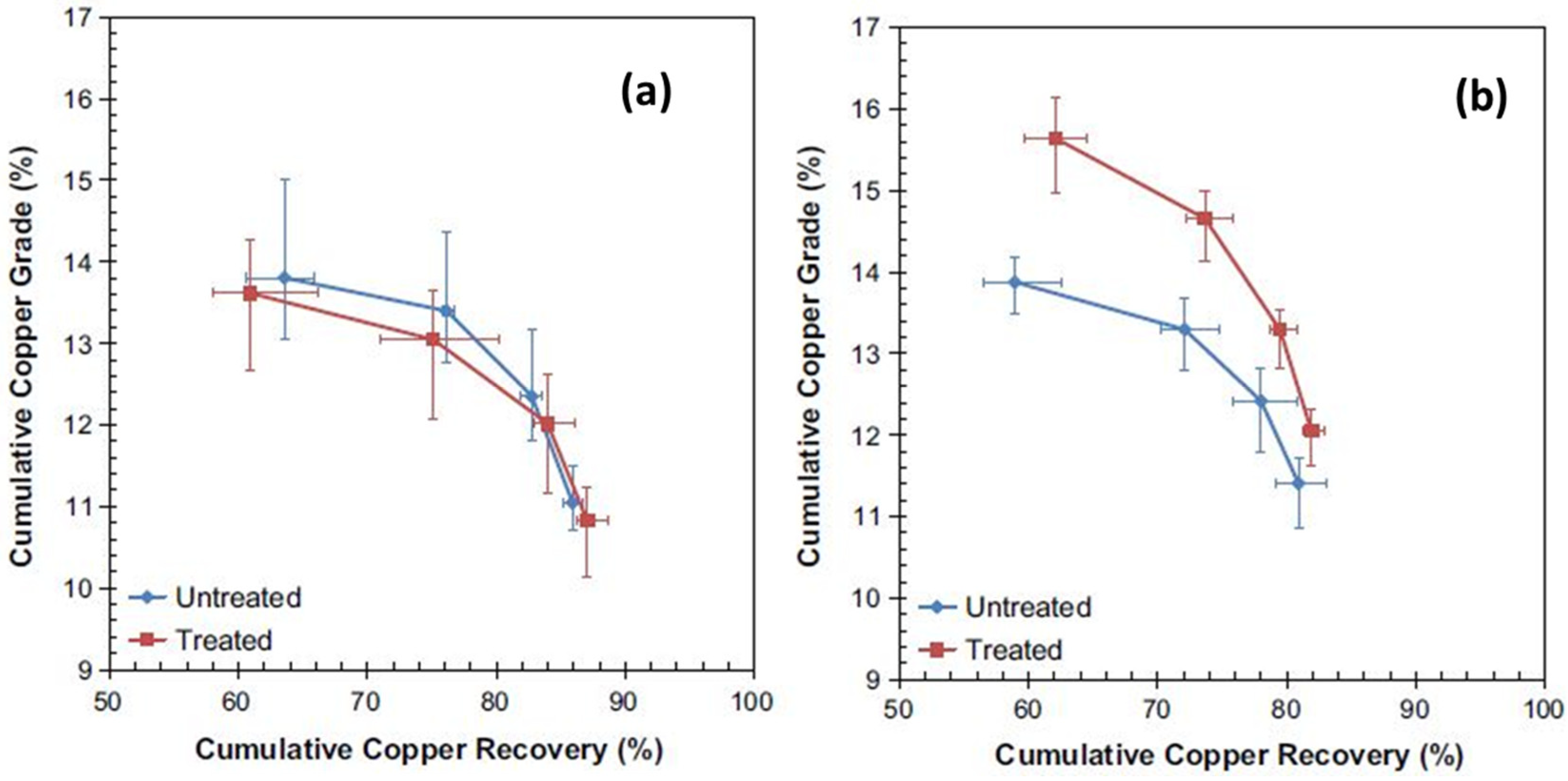
- Batchelor, A.; Jones, D.; Plint, S.; Kingman, S. Increasing the grind size for effective liberation and flotation of a porphyry copper ore by microwave treatment. Miner. Eng. 2016, 94, 61–75. [Google Scholar] [CrossRef]
- Shi, F.; Zuo, W.; Manlapig, E. Pre-concentration of copper ores by high voltage pulses. Part 2: Opportunities and challenges. Miner. Eng. 2015, 79, 315–323. [Google Scholar] [CrossRef]
- Zuo, W.; Shi, F.; Manlapig, E. Pre-concentration of copper ores by high voltage pulses. Part 1: Principle and major findings. Miner. Eng. 2015, 79, 306–314. [Google Scholar] [CrossRef]
- Taheri, B.; Lotfalian, M. Effect of Ultrasonic Pre-Treatment and Aeration on Flotation Separation of Chalcopyrite from Pyrite. Iran. J. Chem. Chem. Eng. (IJCCE) 2018, 37, 199–207. [Google Scholar] [CrossRef]

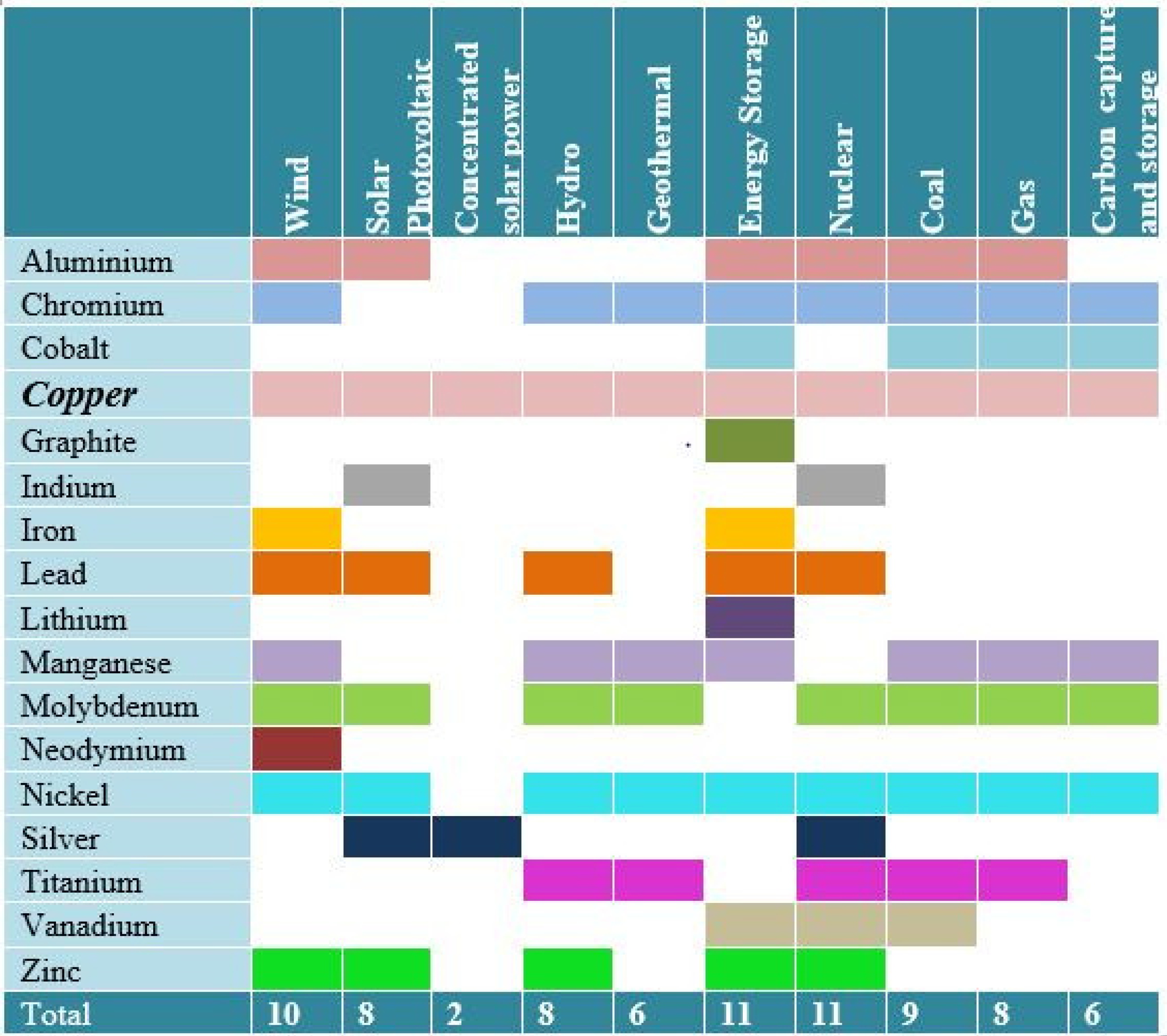


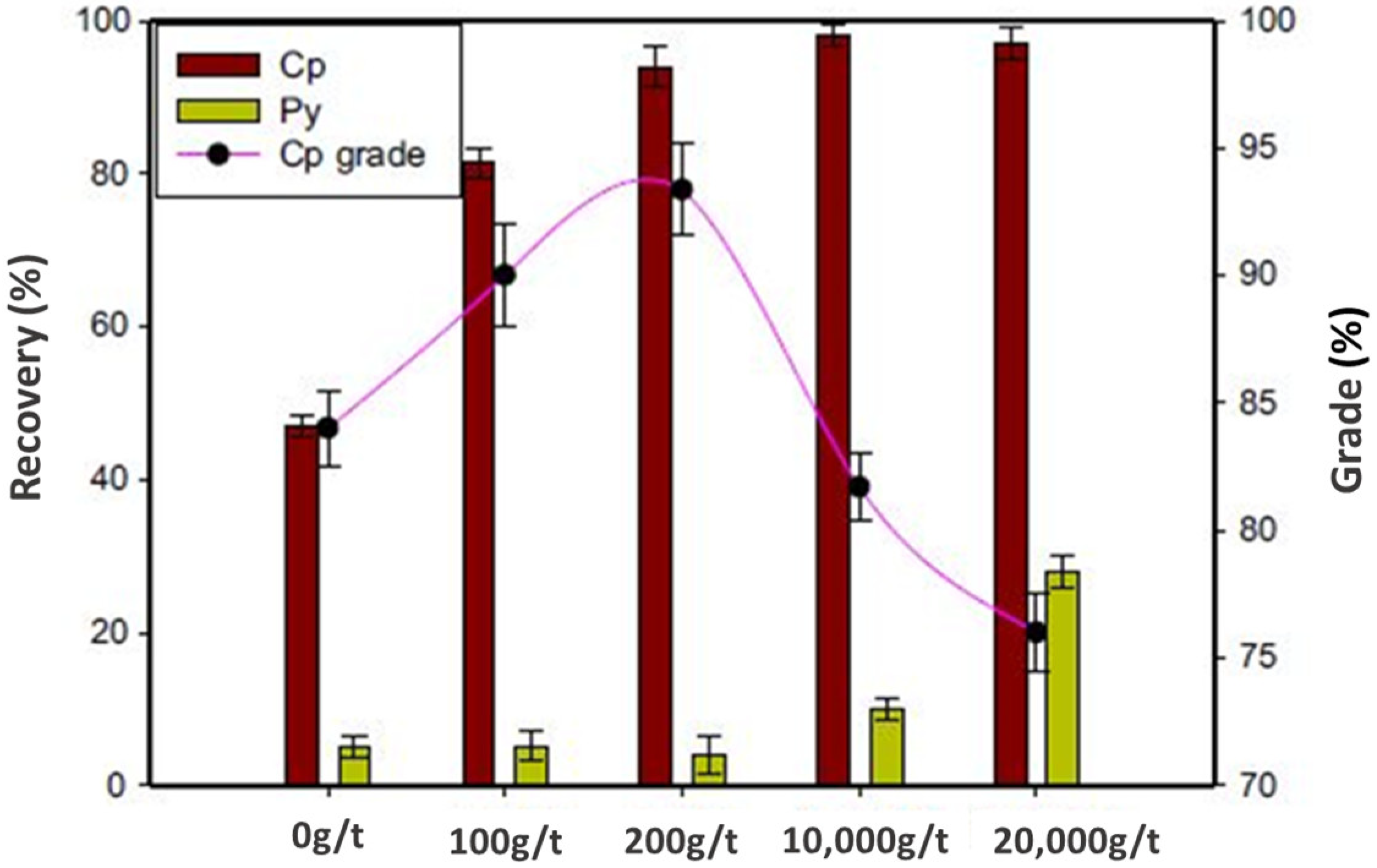
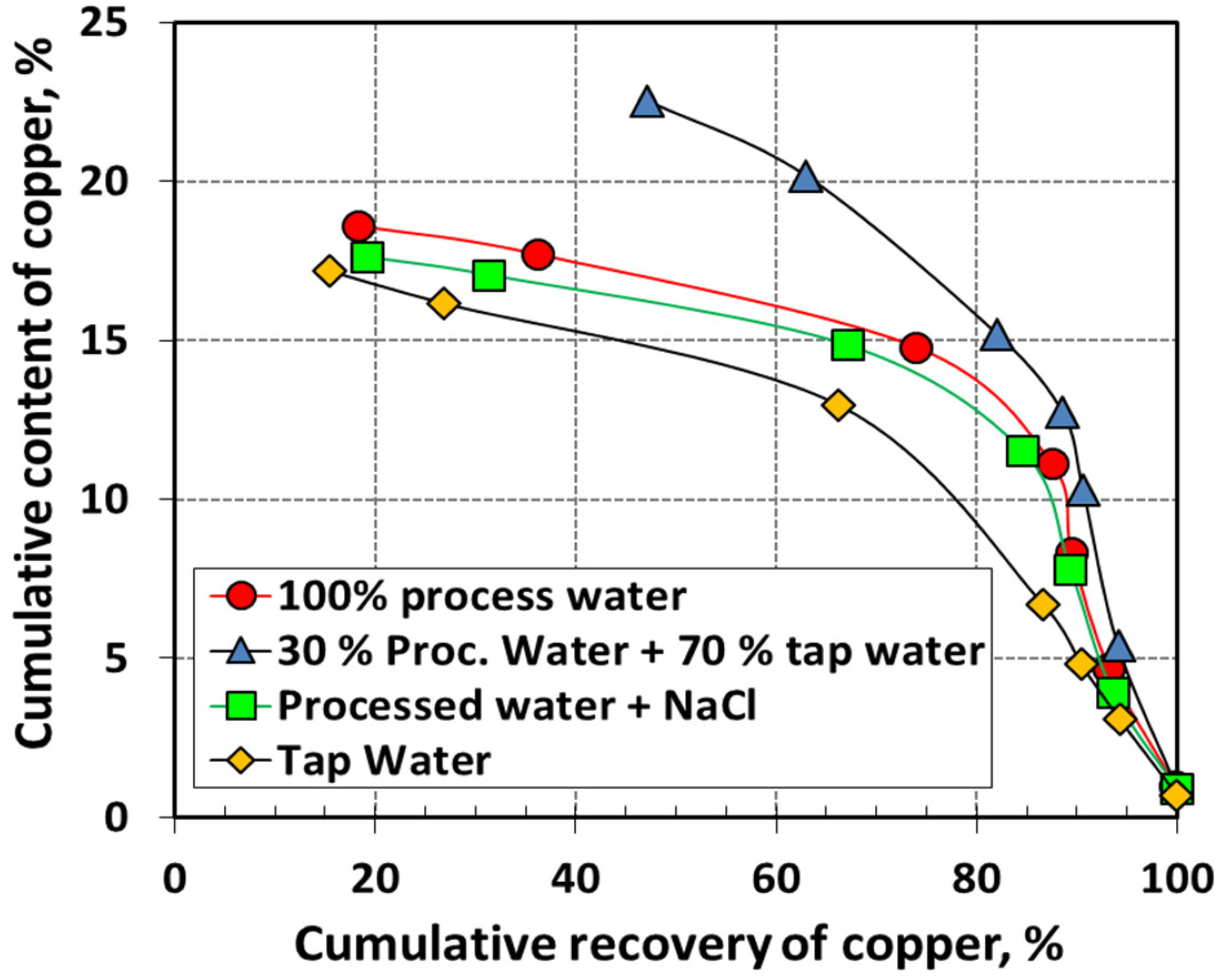

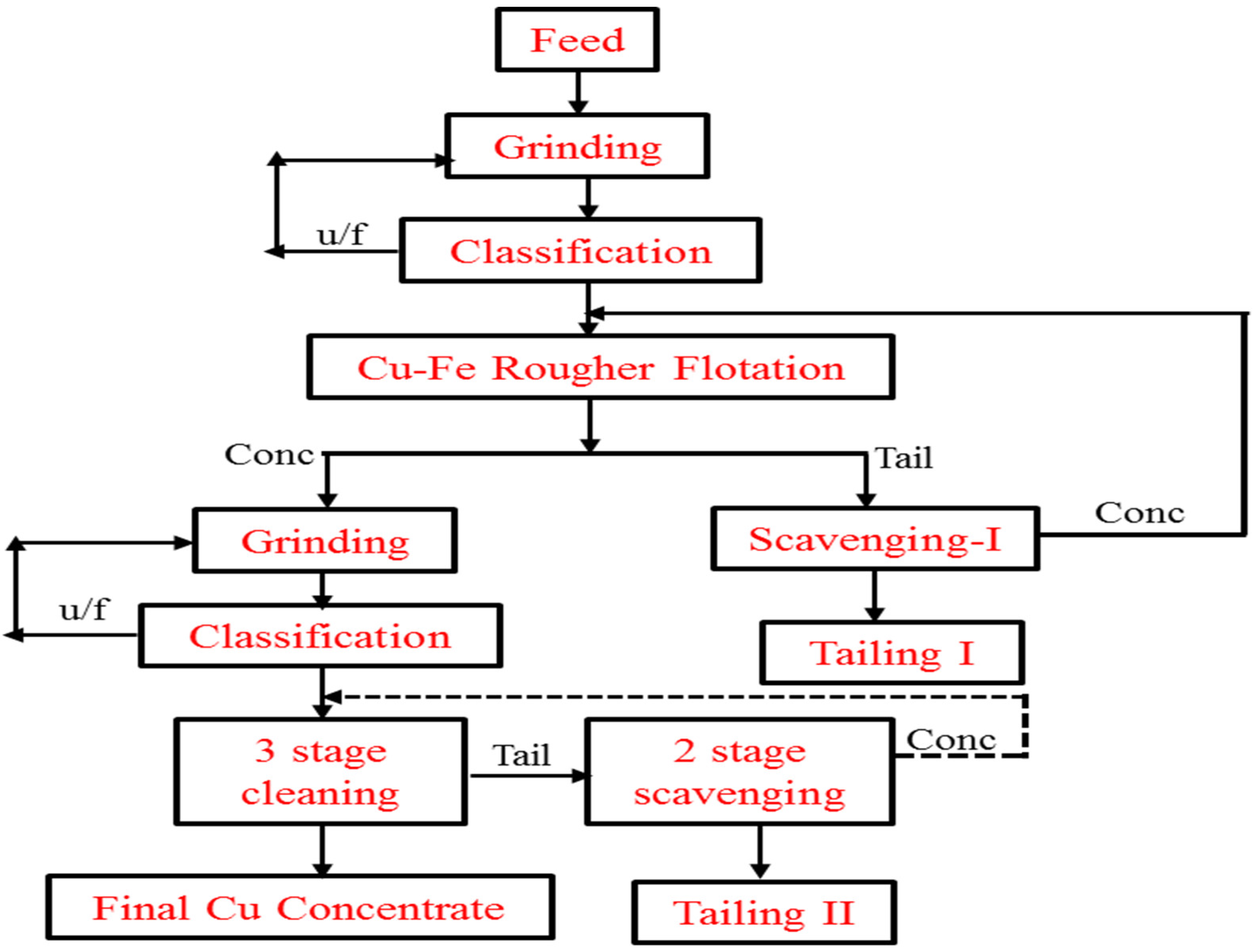
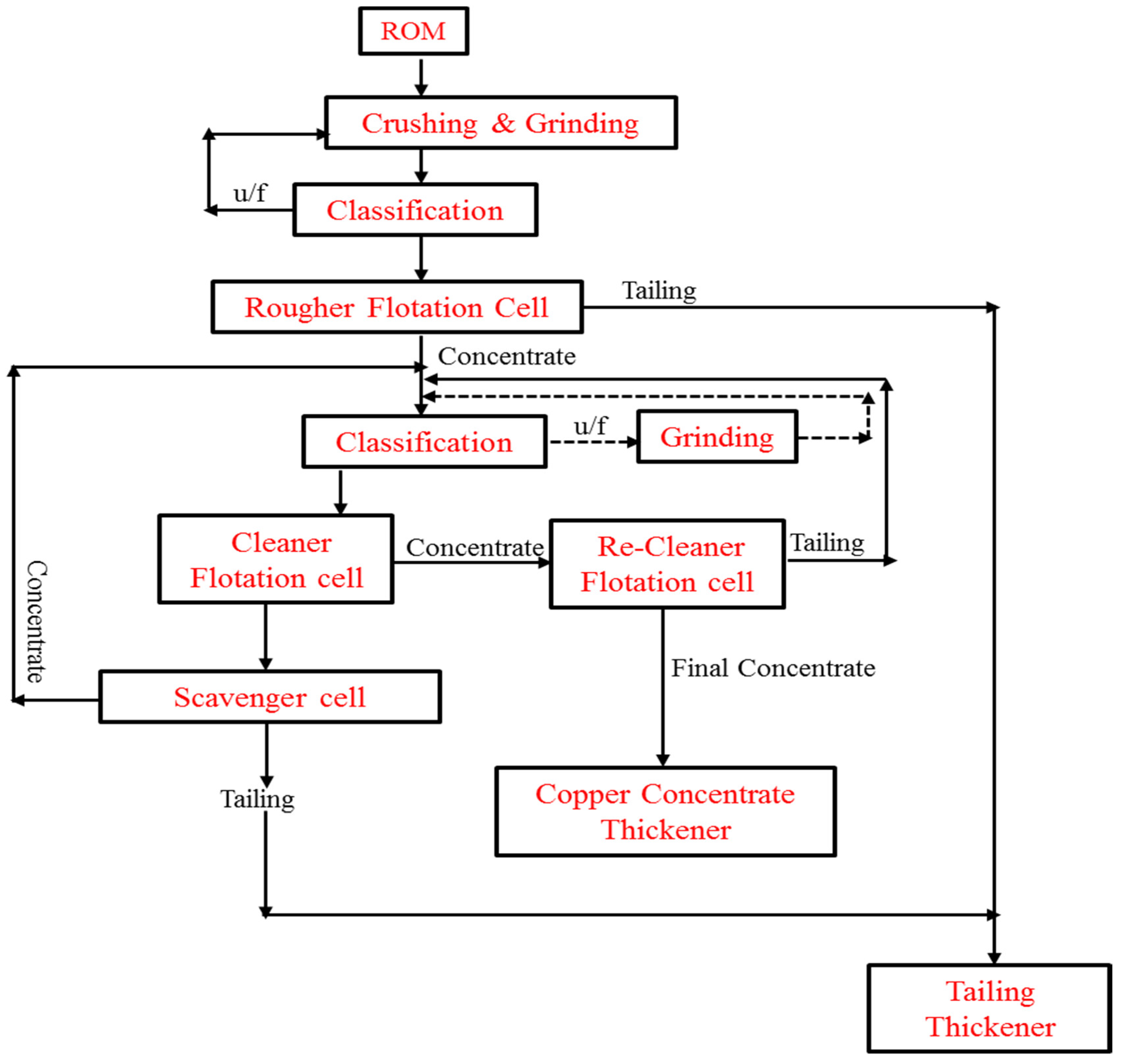




| Country Name | Mine Production (kt) | |
|---|---|---|
| 2018 | 2019 | |
| United States | 1220 | 1300 |
| Australia | 920 | 960 |
| Chile | 5830 | 5600 |
| China | 1590 | 1600 |
| Congo [Kinshasa] | 1230 | 1300 |
| Indonesia | 651 | 340 |
| Kazakhstan | 603 | 700 |
| Mexico | 751 | 770 |
| Peru | 2440 | 2400 |
| Russia | 751 | 750 |
| Zambia | 854 | 790 |
| Other Countries | 3540 | 3800 |
| World Total (Rounded) | 20,400 | 20,000 |
| Deposit Type | Description | Major Minerals | Examples |
|---|---|---|---|
| Porphyry | Large, low-grade stock work disseminated deposits | Primary-Chalcopyrite, bornite. Secondary-Chalcocite, covellite, malachite, azurite, cuprite, molybdenum. By-product material: Rhenium, gold, silver, and tin. | Chuquicamata and La Escondida, Chile; Bingham Canyon, USA; Grasberg, Indonesia; Malanjkhand deposit; India |
| Sediment-hosted | Strata bound disseminations of copper minerals occurring in a range of sedimentary rock. | Chalcocite, bornite with lesser chalcopyrite, galena, pyrite and hematite. By product material: zinc, silver, cobalt | Nchanga, Zambia; Lubin, Poland; White Pine, USA; Khetri deposit, India |
| Red-bed | Copper mineralization occurring in oxidized zones in sedimentary and volcanic rocks | Copper sulphide (1–4% Cu) with silver and rhenium. | Dzhezkazgan, Kazakhstan; Mantos Blancos, Chile |
| Volcanic Massive Sulphide [VMS] | Mineralization hosted by submarine volcano-sedimentary sequences | Chalcopyrite, sphalerite, pyrite, lead, gold, silver. Cobalt, tin, manganese, cadmium, bismuth | Bathurst and Kidd Creek, Canada; Rio Tinto, Spain |
| Magmatic Sulphide | Sulphide concentrations associated with a variety of mafic and ultramafic magmatic rocks | Primarily Chalcopyrite with Pyrrhotite, Pentlandite. Minor amount of PGE, galena, sphalerite, gold, silver, magnetite. | Sudbury and Voisey’s Bay, Canada; Noril’sk-Talanakh, Russia; Kambalda, Australia |
| Deposit type | Description | Major mineral found | Examples |
| Epithermal | Vein stock works and breccias associated with high-level or near surface volcanic-related low temperature hydrothermal systems | Chalcocite, covellite, bornite, native gold, chalcopyrite | El Indio, Chile; Lepanto, Philippines; Monywa, Burma |
| Copper Skarns | Mineralization by chemical alteration associated with intrusions into carbonate rocks | Copper (Chalcopyrite, bornite and tennanite) | Ok Tedi, Papua New Guinea; Copper Canyon, USA; Rosita, Nicaragua |
| Vein Style | Mineralized structures often developed along fractures varying from centimeters up to tens of meters wide from a variety of sources | Chalcopyrite, bornite, pyrite, quartz, silver, gold, occasionally uranium, antimony and phosphorous. | El Indio, Chile; Copper Hills, Australia; Butte, USA; Singhbhum, India |
| Supergene | In-situ natural secondary enrichment of primary mineralization | Chalcocite, covellite, bornite | Erdenet, Mongolia; La Escondida, Chile; Sarcheshmeh, Iran |
| Sedimentary Exhalative | Bodies of polymetallic sulphides deposited from hydrothermal fluids vented into sedimentary basins with no obvious volcanic source | Chalcopyrite, pyrite, sphalerite, galena, pyrite, pyrrhotite, barite, silver | Broken Hill and McArthur River, Australia; Red Dog, Alaska |
| Mineral Name | Chemical Formula | Cu% | Specific Gravity | Hardness | Property |
|---|---|---|---|---|---|
| Native Copper (Cu) | Cu | 100 | 8.9 | Non-magnetic | |
| Sulphides | |||||
| Chalcopyrite (Cpy) | CuFeS2 | 34.6 | 4.1–4.3 | 3.5–4 | Non-magnetic |
| Chalcocite (Cc) | Cu2S | 79.9 | 5.5–5.8 | 2.5–3 | |
| Digenite (Dg) | Cu9S5 | 78.1 | 5.5–5.7 | ||
| Covellite (Cv) | CuS | 66.4 | 4.6–4.8 | 1.5–2 | |
| Enargite (Eng) | Cu3AsS4 | 49.0 | 4.4 | 3 | |
| Bornite (Bn) | Cu5FeS4 | 63.3 | 4.9–5.3 | Magnetic after heating | |
| Oxides | |||||
| Cuprite (Cpr) | Cu2O | 88.8 | 6.1 | 3.5–4 | Non-magnetic |
| Tenorite (Tnr) | CuO | 79.7 | 6.5 | ||
| Malachite (Mal) | CuCO3.Cu(OH)2 | 57.3 | 3.9 | ||
| Azurite (Azu) | 2CuCO3.Cu(OH)2 | 55.1 | 3.7 | ||
| Silicate | |||||
| Chrysocolla (Ccl) | CuSiO3.2H2O | 37.9 | 2.2 | 2–4 | Non-magnetic |
| Specific Gravity | Concentration Criterion (CC) | |
|---|---|---|
| Valuable Heavy Minerals | Gangue/Lighter Mineral | |
| Chalcopyrite = 4.1 | (Primarily Quartz) Quartz = 2.65 | 1.87 |
| Malachite = 3.9 | 1.76 | |
| Covellite = 4.6 | 2.18 | |
| Chalcocite = 5.5 | 2.73 | |
| Bornite = 4.9 | 2.36 | |
| Digenite = 5.5 | 2.73 | |
| CC > 2.5, Gravity separation is easy; CC < 1.25, Gravity separation is not feasible | ||
| Mineral | Collector | Species Responsible for Hydrophobicity |
|---|---|---|
| Bornite (Cu5FeS4) | C2-X | CuX |
| Chalcopyrite (CuFeS2) | X2 | |
| Chalcocite (Cu2S) | CuX, Cu(X)2 | |
| Covellite (CuS) | X2, CuX | |
| Bornite (Cu5FeS4) | di-C2-DTP | Cu(DTP)2 |
| Chalcopyrite (CuFeS2) | Cu(DTP)2 | |
| Chalcocite (Cu2S) | Cu(DTP)2, Cu(DTP) | |
| Covellite (CuS) | Cu(DTP)2 | |
| Bornite (Cu5FeS4) | di-C2-DTC | Cu(DTC)2 |
| Chalcopyrite (CuFeS2) | Cu(DTC), Cu(DTC)2 | |
| Chalcocite (Cu2S) | Cu(DTP) | |
| Covellite (CuS) | Cu(DTC)2 |
| Ore | Collector | Frother | pH | Reference |
|---|---|---|---|---|
| Chalcopyrite | Sodiumisopropyl Xanthate (SIPX) | Polypropylene Glycol (Dowfroth 250; AR grade) | 10.5 | [62] |
| Isopropyl ethyl thiocarbamate | Methyl Isobutyl Carbinol (MIBC) | 6.3–5.1 | [71] | |
| Sodium butyl xanthate (SBX) | Mixture of C5–C8 alcohol | 8.0–8.5 | [72] | |
| N-propyl-N-ethoxycarbonyl thiourea (PECTU) | Mixture of C5–C8 alcohol | 8.5–9.0 | ||
| Sodium ethyl xanthate (NaEX) | DowFroth 250 | [73] | ||
| Potassium amyl xanthate (PAX) | Methyl Isobutyl Carbinol (MIBC) | 10.0–10.5 | [74] | |
| Bornite |
| - | 9 | [65] |
| Chalcocite |
| - | 9.3 | [75] |
| MIBC Polypropylene Dowfroth 250 | 9.4 | [68] |
| Ore | Deposit /Country | Collector | Frother | Modifier | Remark | Reference |
|---|---|---|---|---|---|---|
| Atacamite + paracamite + chalcopyrite | Hanna Mining Company, Ohio. | Potassium octyl hydroxamate (POH): Potassium Amyl Xathate (PAX): 1:2 | Pine Oil: MIBC: 1:1 | - | Patentable work for the effective flotation of mixed cu ore using hydroxamate collector | [104] |
| Chalcopyrite ([major) + Malachite, Azurite (traces) | Indian copper mine | Sodium Iso Propyl Xanthate (SIPX) Potassium Octyl hydroxamate (POH) | MIBC | NaOH (Ph), Na2S (Sulphidizer) and Na2SiO3 (Depressant) | Single Rougher step followed by three stage cleaning yield final concentrate of 31.77% cu from 0.95% Cu feed | [109] |
| Sulphide (Bornite + Chalcopyrite) and Oxide (Malachite + Azurite) | Sherwood Copper’s Minto mines, Canada | Potassium Amyl Xanthate (PAX) + Potassium n-octyl hydroxamate (AM28) | MIBC | Sulphidizer (NaHS) | AM28 recovered copper oxide mineral without affecting recovery of sulphide minerals | [116] |
| (Cu Oxide (88%) + Cu Sulphide (12%)) | Central Africa | Potassium amyl Xanthate (PAX)-Sulphide collector N-Octyl hydroxamate and Oleofloat 6540-Oxide collector | Dowfroth 200 | Sulphidizer (NaHS) | Mixed natural ore | [116] |
| Copper Mixed ore (Chalcopyrite + Malachite) | Malanjkhand copper mine, India | Sodium Iso-propyl Xanthate (SIPX) Sodium Diethyldithiocarbamate (SDTC) | Pine Oil | Sodium Silicate | Only copper sulphide recovery is focused even after its mixed type | [115] |
| Mineral | Ore Deposit | IEP (at pH) | Remark | Reference |
|---|---|---|---|---|
| Chalcopyrite | Malanjkhand Copper Mine, India | 2.9–3.3 | Lower IEP is due to the presence of abundant silicate gangue | [43,124,125] |
| Malachite | Guangxi Province, China | 8.4–8.7 | High purity mineral | [128,129,130] |
| Lupe Mine, Mexico | 9.1 | Presence of Pseudo malachite (5%) | [134] | |
| Chrysocolla | Dongchuan, Yunnan Province, China | Not found | Pure Mineral with 40% SiO2 | [131] |
| Exotica Mine, Chile | Not found | Purest Lump with 43% SiO2 | [132] |
| Stream | Element (wt%) | ||||
|---|---|---|---|---|---|
| Cu | Fe | Al | Mg | O | |
| Raw tailing feed | 0.61 | 2.44 | 9.17 | 3.42 | 50.16 |
| Upgraded feed | 1.02 | 1.64 | 6.54 | 1.52 | 54.97 |
| Sl. No. | Mineralogy | Place and Country | CuFG (%) | CCG; CuCR (%) | CuTG (%) | Beneficiation Units | Reagents | References |
|---|---|---|---|---|---|---|---|---|
| 1 | Cpy, cc, dg, bor, mal, cup, cv, qtz | Dexing copper Mine, China | 0.443 | 23.16; 75.6 | 0.11 | Classification and Flotation (3-stage) | SBX: PECTU as collector, Lime pH modifier | [71] |
| 2 | Cpy, cc, cv, py | Sungun Copper, Iran | 0.61 | 3.78; 87.03 | 0.09 | Classification and Flotation | SIPX and AP407 as collector; Ether Polyglycol and MIBC as frother. | [153] |
| 3 | Cpy, py, po, asp, qtz | Mosabani copper mines, Singhbhum, India | 1.5 | 25; 96.86 | 0.05 | Classification and Flotation (2-stage) | SIPX as collector, Pine oil as frother, soda ash as pH modifier | [156] |
| 4 | Cpy, cv, dg, py, mal, azu, qtz, kfs | Malanjkhand copper mines, Balaghat, India | 1.2 | 25; 83.6 | 0.08 | Classification and Flotation (3-stage) | SIPX as collector, Pine oil as frother, Lime as pH modifier | [156] |
| 5 | Cpy, sph, cv, bor | Cerro de Maimón copper mine, Dominican Republic | 2.7 | 28; 83.3 | 0.49 | Grinding, rougher flotation, regrinding, and cleaner flotation | Aero 9810 as collector, MIBC as frother, Lime as pH modifier | [157] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jena, S.S.; Tripathy, S.K.; Mandre, N.R.; Venugopal, R.; Farrokhpay, S. Sustainable Use of Copper Resources: Beneficiation of Low-Grade Copper Ores. Minerals 2022, 12, 545. https://doi.org/10.3390/min12050545
Jena SS, Tripathy SK, Mandre NR, Venugopal R, Farrokhpay S. Sustainable Use of Copper Resources: Beneficiation of Low-Grade Copper Ores. Minerals. 2022; 12(5):545. https://doi.org/10.3390/min12050545
Chicago/Turabian StyleJena, Silpa Sweta, Sunil Kumar Tripathy, N. R. Mandre, R. Venugopal, and Saeed Farrokhpay. 2022. "Sustainable Use of Copper Resources: Beneficiation of Low-Grade Copper Ores" Minerals 12, no. 5: 545. https://doi.org/10.3390/min12050545
APA StyleJena, S. S., Tripathy, S. K., Mandre, N. R., Venugopal, R., & Farrokhpay, S. (2022). Sustainable Use of Copper Resources: Beneficiation of Low-Grade Copper Ores. Minerals, 12(5), 545. https://doi.org/10.3390/min12050545









