Recovery of Catapleiite and Eudialyte from Non-Magnetic Fraction of Eudialyte ore Processing of Norra Kärr Deposit
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Characterization
2.3. Magnetic Separation Stage
2.4. Flotation Experiments
2.5. Reagents
3. Results and Discussion
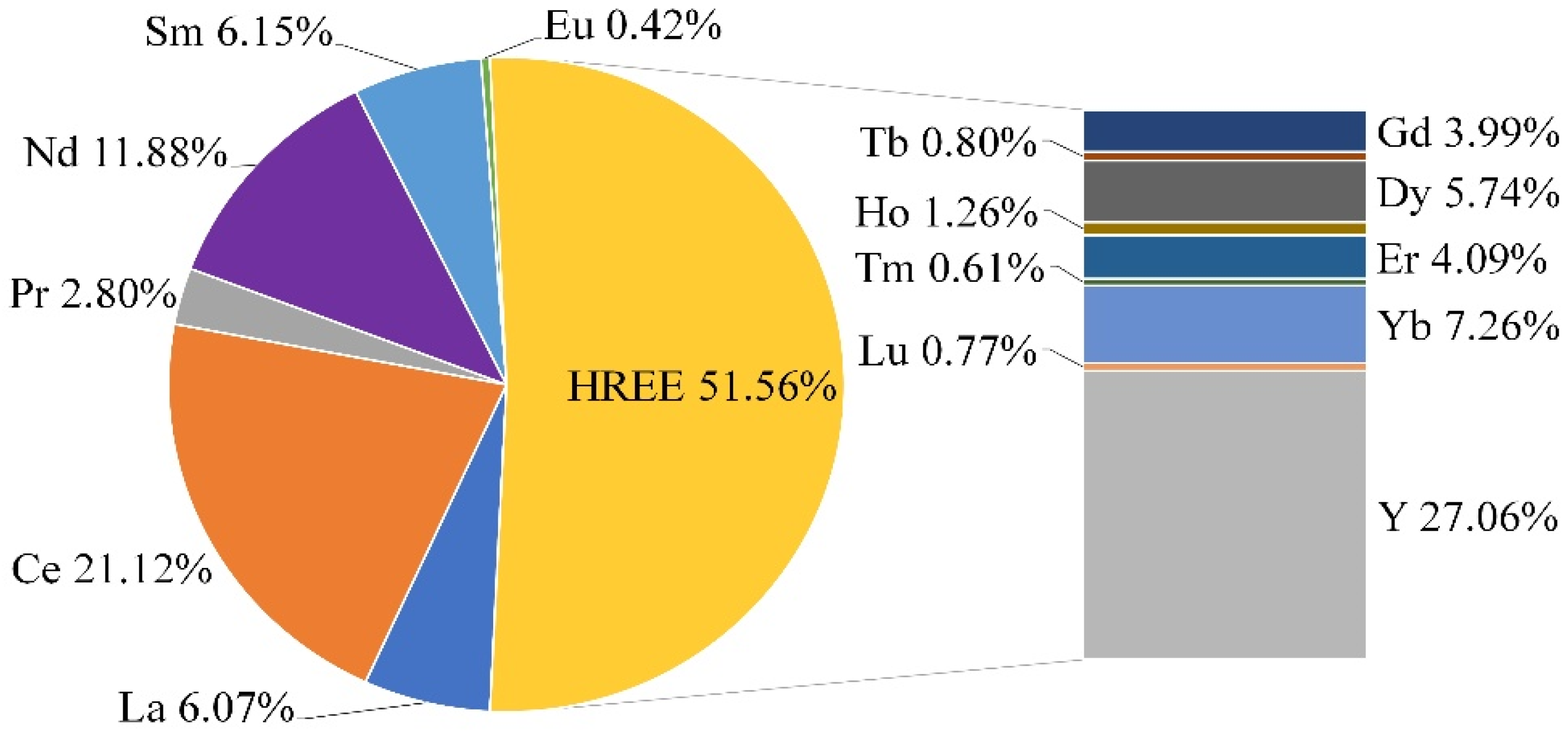
3.1. Characterization of the Non-Magnetic Fraction (Flotation Feed)
3.2. Flotation of Zirconosilicate Minerals from Non-Magnetic Fraction
3.2.1. Selection of Collectors for the Flotation of Zirconosilicate Minerals
3.2.2. Effect of Collector Dosage and pH Value
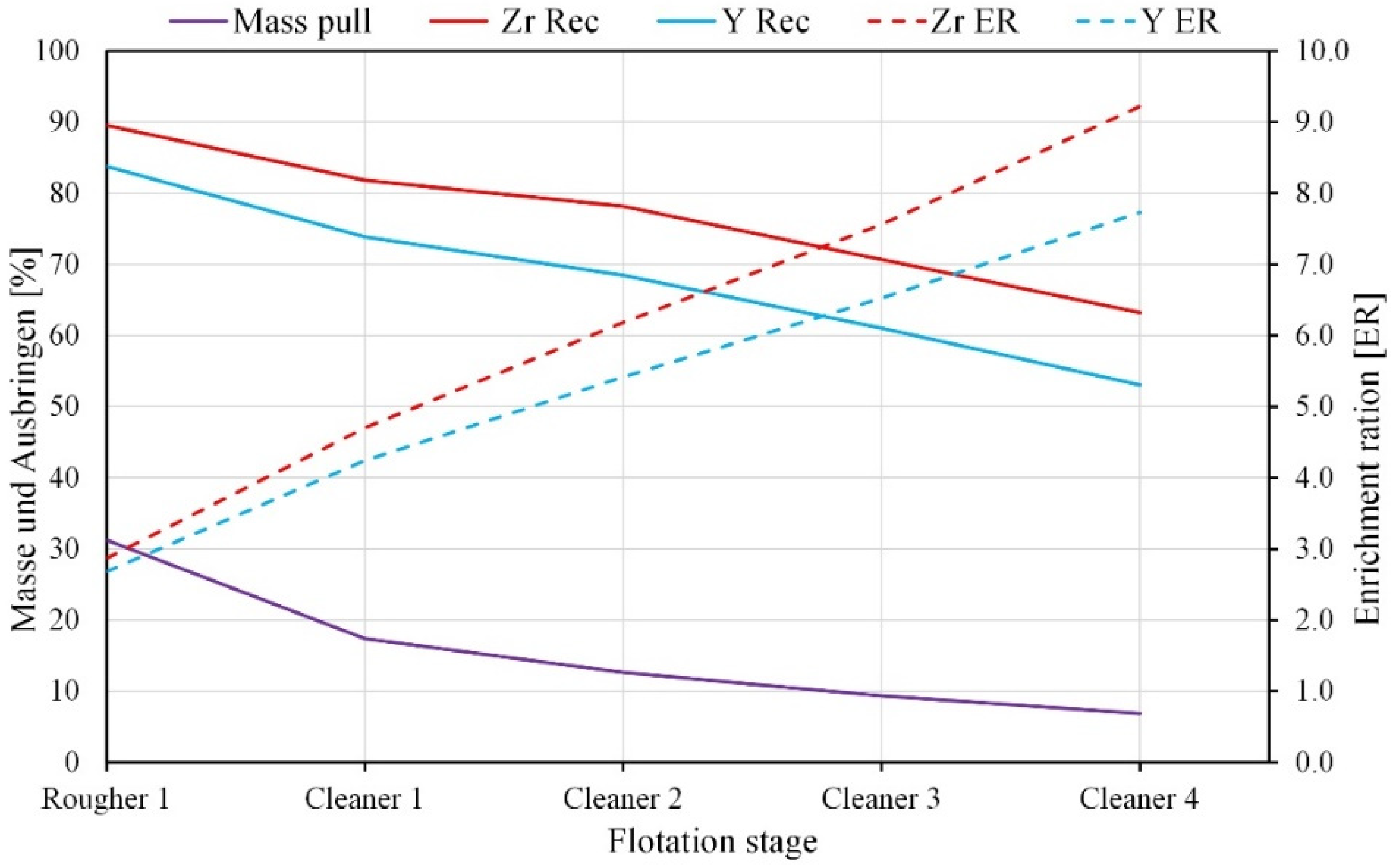
3.2.3. Characterization of Flotation Products
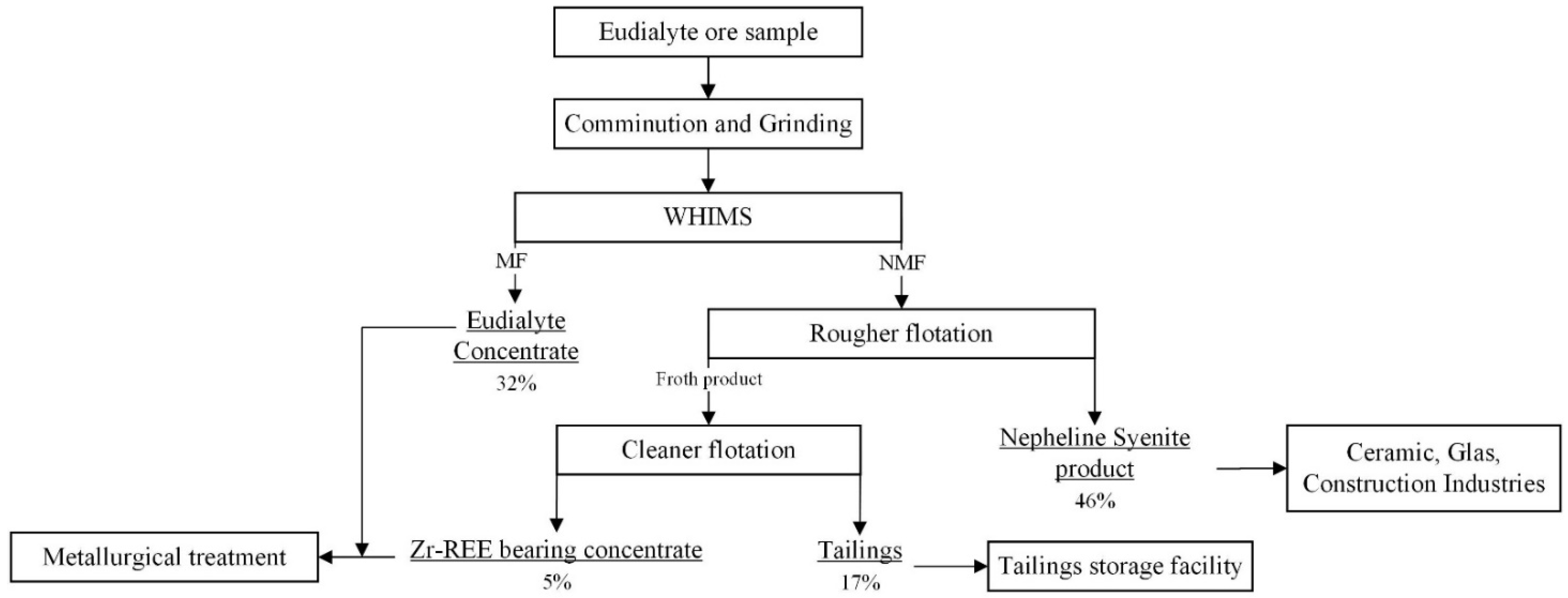
3.3. Development of an Alternative Beneficiation Flowsheet for Norra Kärr Deposit
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sovacool, B.K.; Ali, S.H.; Bazilian, M.; Radley, B.; Nemery, B.; Okatz, J.; Mulvaney, D. Sustainable minerals and metals for a low-carbon future. Science 2020, 367, 30–33. [Google Scholar] [CrossRef]
- Tkaczyk, A.H.; Bartl, A.; Amato, A.; Lapkovskis, V.; Petranikova, M. Sustainability evaluation of essential critical raw materials: Cobalt, niobium, tungsten and rare earth elements. J. Phys. D Appl. Phys. 2018, 51, 203001. [Google Scholar] [CrossRef] [Green Version]
- Silin, I.; Hahn, K.; Gürsel, D.; Kremer, D.; Gronen, L.; Stopić, S.; Friedrich, B.; Wotruba, H. Mineral processing and metallurgical treatment of lead vanadate ores. Minerals 2020, 10, 197. [Google Scholar] [CrossRef] [Green Version]
- Jaroni, M.S.; Friedrich, B.; Letmathe, P. Economical feasibility of rare earth mining outside China. Minerals 2019, 9, 576. [Google Scholar] [CrossRef] [Green Version]
- Dostal, J. Rare Earth Element Deposits of Alkaline Igneous Rocks. Resources 2017, 6, 34. [Google Scholar] [CrossRef]
- Marks, M.A.; Markl, G. A global review on agpaitic rocks. Earth Sci. Rev. 2017, 173, 229–258. [Google Scholar] [CrossRef]
- Sjöqvist, A.; Cornell, D.; Andersen, T.; Erambert, M.; Ek, M.; Leijd, M. Three Compositional Varieties of Rare-Earth Element Ore: Eudialyte-Group Minerals from the Norra Kärr Alkaline Complex, Southern Sweden. Minerals 2013, 3, 94–120. [Google Scholar] [CrossRef]
- Atanasova, P.; Marks, M.A.W.; Heinig, T.; Krause, J.; Gutzmer, J.; Markl, G. Distinguishing Magmatic and Metamorphic Processes in Peralkaline Rocks of the Norra Kärr Complex (Southern Sweden) Using Textural and Compositional Variations of Clinopyroxene and Eudialyte-group Minerals. J. Petrol. 2017, 58, 361–384. [Google Scholar] [CrossRef] [Green Version]
- Vaccarezza, V.; Anderson, C. An Overview of Beneficiation and Hydrometallurgical Techniques on Eudialyte Group Minerals. Min. Metall. Explor. 2020, 37, 39–50. [Google Scholar] [CrossRef]
- Stark, T.; Silin, I.; Wotruba, H. Mineral Processing of Eudialyte Ore from Norra Kärr. J. Sustain. Metall. 2017, 3, 32–38. [Google Scholar] [CrossRef]
- Forrester, K.; Leijd, M.; Oczlon, M.; Holmström, H.; Saxon, M. Beneficiation of rare earth element enriched eudialyte from the Norra Kärr peralkaline intrusion with wet high intensity magnetic separation. In Proceedings of the Conference of Metallurgists, Vancouver, BC, Canada, 28 September–1 October 2014. [Google Scholar]
- Silin, I.; Stark, T.; Wolfrum, S.; Wotruba, H. Method and Device for Producing an Eudialyte Concentrate by Direct Flotation. European Patent Number 3150283A1, 30 September 2015. [Google Scholar]
- Voßenkaul, D.; Birich, A.; Müller, N.; Stoltz, N.; Friedrich, B. Hydrometallurgical Processing of Eudialyte Bearing Concentrates to Recover Rare Earth Elements Via Low-Temperature Dry Digestion to Prevent the Silica Gel Formation. J. Sustain. Metall. 2017, 3, 79–89. [Google Scholar] [CrossRef]
- Ma, Y.; Stopic, S.; Friedrich, B. Hydrometallurgical Treatment of an Eudialyte Concentrate for Preparation of Rare Earth Carbonate. Johns. Matthey Technol. Rev. 2019, 63, 2–13. [Google Scholar] [CrossRef]
- Ma, Y.; Stopic, S.; Gronen, L.; Friedrich, B. Recovery of Zr, Hf, Nb from eudialyte residue by sulfuric acid dry digestion and water leaching with H2O2 as a promoter. Hydrometallurgy 2018, 181, 206–214. [Google Scholar] [CrossRef]
- Balinski, A.; Wiche, O.; Kelly, N.; Reuter, M.A.; Scharf, C. Separation of rare earth elements from contaminants and valuable components by in-situ precipitation during the hydrometallurgical processing of eudialyte concentrate. Hydrometallurgy 2020, 194, 105345. [Google Scholar] [CrossRef]
- Balinski, A.; Atanasova, P.; Wiche, O.; Kelly, N.; Reuter, M.A.; Scharf, C. Selective Leaching of Rare Earth Elements (REEs) from Eudialyte Concentrate after Sulfation and Thermal Decomposition of Non-REE Sulfates. Minerals 2019, 9, 522. [Google Scholar] [CrossRef] [Green Version]
- Cheremisina, O.V.; Volkova, O.; Litvinova, T.E. Influence of anion nature on acid leaching of silicate minerals and solvent extraction of rare and rare-earth elements. Geochemistry 2020, 80, 125507. [Google Scholar] [CrossRef]
- Chanturiya, V.A.; Bunin, I.Z.; Ryazantseva, M.V.; Chanturiya, E.L.; Samusev, A.L.; Anashkina, N.E. Mechanism of the Change in the Structural, Chemical, and Technological Properties of Eudialyte upon Combined Energy Effects. Bull. Russ. Acad. Sci. Phys. 2019, 83, 716–720. [Google Scholar] [CrossRef]
- Davidson, T. Amended and Restated Prefeasibility Study. NI 43-101. Technical report for the Norra Kärr Rare Earth Element Deposit.; GBM: Gränna, Sweden, 2015. [Google Scholar]
- Zubkova, N.V.; Ksenofontov, D.A.; Chukanov, N.V.; Pekov, I.V.; Artamonova, A.A.; Koshlyakova, N.N.; Bychkov, A.Y.; Pushcharovsky, D.Y. Crystal Chemistry of the Microporous Zirconosilicate Na6Zr3[Si9O27], a Product of High-Temperature Transformation of Catapleiite, and Its Ag-Exchanged Form. Minerals 2020, 10, 243. [Google Scholar] [CrossRef] [Green Version]
- Ksenofontov, D.A.; Grebenev, V.V.; Zubkova, N.V.; Pekov, I.V.; Kabalov, Y.K.; Chukanov, N.V.; Pushcharovsky, D.Y.; Artamonova, A.A. Behavior of Catapleiite under Heating and Crystal Structure of its High-Temperature Transformation Product, a New Phase Na6Zr3[Si9O27] with Nine-membered Rings of SiO4 Tetrahedra. Geol. Ore Depos. 2019, 61, 696–705. [Google Scholar] [CrossRef]
- Borst, A.M.; Friis, H.; Andersen, T.; Nielsen, T.F.D.; Waight, T.E.; Smit, M.A. Zirconosilicates in the kakortokites of the Ilímaussaq complex, South Greenland: Implications for fluid evolution and high-field-strength and rare-earth element mineralization in agpaitic systems. Mineral. Mag. 2016, 80, 5–30. [Google Scholar] [CrossRef] [Green Version]
- van de Ven, M.; Borst, A.; Davies, G.; Hunt, E.; Finch, A. Hydrothermal Alteration of Eudialyte-Hosted Critical Metal Deposits: Fluid Source and Implications for Deposit Grade. Minerals 2019, 9, 422. [Google Scholar] [CrossRef] [Green Version]
- Bulatovic, S.M. Handbook of Flotation Reagents: Chemistry, Theory and Practice; Flotation of Industrial Minerals; Elsevier: Kidlington, UK, 2015; Volume 3, ISBN 9780444530837. [Google Scholar]
- Bjelopavlic, M.; Ralston, J.; Reynolds, G. Adsorption of Monoalkyl Phosphates at the Zircon-Aqueous Solution Interface. J. Colloid Interface Sci. 1998, 208, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Marion, C.; Li, R.; Williams, H.; Hundt, D.; Waters, K.E. A physico-chemical investigation into the separation of zircon and quartz by flotation. In Proceedings of the 29th International Mineral Processing Congress (IMPC 2018), Moscow, Russia, 17–21 September 2018; pp. 4062–4072. [Google Scholar]
- Sobieraj, S.; Ralston, J.; Smart, R.S.C. Flotation of Zircon from Mineral Sands. Flotacja cyrkonu z piasków mineralnych (in Polish). Physicochem. Probl. Miner. Process. 1991, 24, 233–243. [Google Scholar]
- Jeswiet, J.; Szekeres, A. Energy Consumption in Mining Comminution. Procedia CIRP 2016, 48, 140–145. [Google Scholar] [CrossRef] [Green Version]
- Silin, I.; Rubinstein, Y.; Volobayev, I.; Liono, A.; Gürsel, D.; Wotruba, H. Valorisation of Abandoned Low Grade Tailings in Albania for Recovery of Metal Concentrates and Mineral Products. Russ. J. Non Ferr. Met. 2021, 62, 483–494. [Google Scholar] [CrossRef]
- Spooren, J.; Binnemans, K.; Björkmalm, J.; Breemersch, K.; Dams, Y.; Folens, K.; González-Moya, M.; Horckmans, L.; Komnitsas, K.; Kurylak, W.; et al. Near-zero-waste processing of low-grade, complex primary ores and secondary raw materials in Europe: Technology development trends. Resour. Conserv. Recycl. 2020, 160, 104919. [Google Scholar] [CrossRef]
- Sánchez-Soto, P.J.; Garzón, E.; Pérez-Villarejo, L.; Angelopoulos, G.N.; Eliche-Quesada, D. Mining Wastes of an Albite Deposit as Raw Materials for Vitrified Mullite Ceramics. Minerals 2021, 11, 232. [Google Scholar] [CrossRef]
- Lorenz, W.; Gwosdz, W. Quarzrohstoffe: Mit 69 Tabellen; Schweizerbart: Stuttgart, Germany, 1999; ISBN 978-3-510-95839-9. [Google Scholar]
- Bagani, M.; Balomenos, E.; Panias, D. Nepheline syenite as an alternative source for aluminum production. Minerals 2021, 11, 734. [Google Scholar] [CrossRef]
- Samantray, J.; Anand, A.; Dash, B.; Ghosh, M.K.; Behera, A.K. Nepheline Syenite—An Alternative Source for Potassium and Aluminium. In Rare Metal Technology 2019; Azimi, G., Kim, H., Alam, S., Ouchi, T., Neelameggham, N.R., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 145–159. ISBN 978-3-030-05739-8. [Google Scholar]
- Everard, L.J. An Assessment of the Resource Potential for Nepheline Syenite at Cygnet and Elsewhere in Tasmania: Record 1996/13; Tasmanian Geological Survey: Tasmania, Australia, 1998.
- Burat, F.; Kangal, O.; Onal, G. An alternative mineral in the glass and ceramic industry: Nepheline syenite. Miner. Eng. 2006, 19, 370–371. [Google Scholar] [CrossRef]
- Çinar, M.; Durgut, E. Mineral beneficiation of Kırşehir nepheline syenite with combination of dry magnetic separation and flotation methods. Physicochem. Probl. Miner. Process. 2019, 55, 1227–1238. [Google Scholar] [CrossRef]
- Kangal, M.O.; Bulut, G.; Yeşilyurt, Z.; Basturkcu, H.; Burat, F. Characterization and production of Turkish nepheline syenites for industrial applications. Physicochem. Probl. Miner. Process. 2019, 55, 605–616. [Google Scholar] [CrossRef]
- Ahmed, H.A.M. Dry versus wet upgrading of nepheline syenite ores. Physicochem. Probl. Miner. Process. 2011, 46, 107–118. [Google Scholar]
- Wills, B.A. Wills’ Mineral. Processing Technology: An Introduction to the Practical Aspects of Ore Treatment and Mineral. Recovery, 8th ed.; Elsevier Science: Amsterdam, The Netherlands, 2015; ISBN 9780080970530. [Google Scholar]
- Ivanova, V.; Mitrofanova, G. Flotation of eudialyte: Correlation of experimental data with the results of quantum-chemical calculations. In Proceedings of the XVI Balkan Mineral Processing Congress, Belgrade, Serbia, 15–17 June 2015; pp. 347–351. [Google Scholar]
- Qiu, H.; Kersebaum, J.; Wollmann, A.; Feuge, N.; Haas, A.; Goldmann, D.; Wilhelm, R. Improvement of the froth flotation of LiAlO2 and melilite solid solution via pre-functionalization. Sci. Rep. 2021, 11, 20443. [Google Scholar] [CrossRef] [PubMed]
- Baudet, G.; Morteani, G.; Strub, M.P. Application of Phosphoric Esters to Flotation of Finely Divided Oxidized Ores: Final Report, C.E.C. Research Contract MA1M-0059-C; Department Mineralurgie: Orleans, France, 1992. [Google Scholar]
- O’Lenick, A.J. Surfactants: Strategic Personal Care Ingredients; Allured Publ. Corp.: Carol Stream, IL, USA, 2005; ISBN 978-1932633085. [Google Scholar]
- Espiritu, E.R.L.; Naseri, S.; Waters, K.E. Surface chemistry and flotation behavior of dolomite, monazite and bastnäsite in the presence of benzohydroxamate, sodium oleate and phosphoric acid ester collectors. Colloids Surf. A Physicochem. Eng. Asp. 2018, 546, 254–265. [Google Scholar] [CrossRef]
- Ney, P. Zeta-Potentiale und Flotierbarkeit von Mineralen; Springer: Vienna, Austria; New York, NY, USA, 1973; ISBN 978-3-211-81104-7. [Google Scholar]
- Nayfonov, T.; Belborodov, V.; Zakharova, I. Flotation technology for beneficiation of eudialyte ore. In Proceedings of the XVII International Mineral Processing Congress, Dresden, Germany, 23–28 September 1991; Volume 4, pp. 131–138. [Google Scholar]
- Silin, I.; Huben, J.; Wotruba, H.; Ognyanova, A. Study on the Characterisation and Processing of Iron Ore after Grinding by HPGR. In Proceedings of the 29th International Mineral Processing Congress (IMPC 2018), Moscow, Russia, 17–21 September 2018; pp. 2388–2397. [Google Scholar]
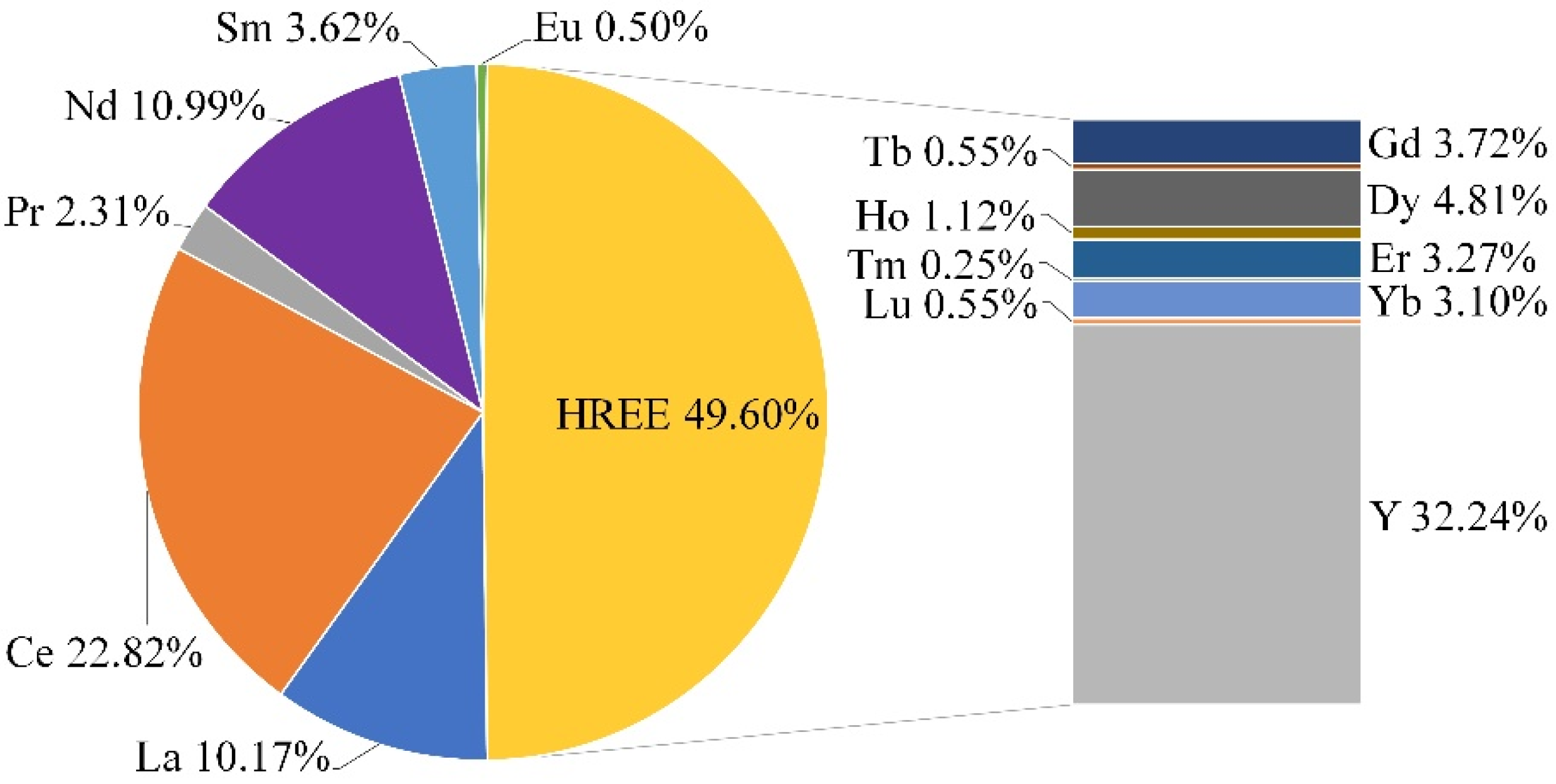
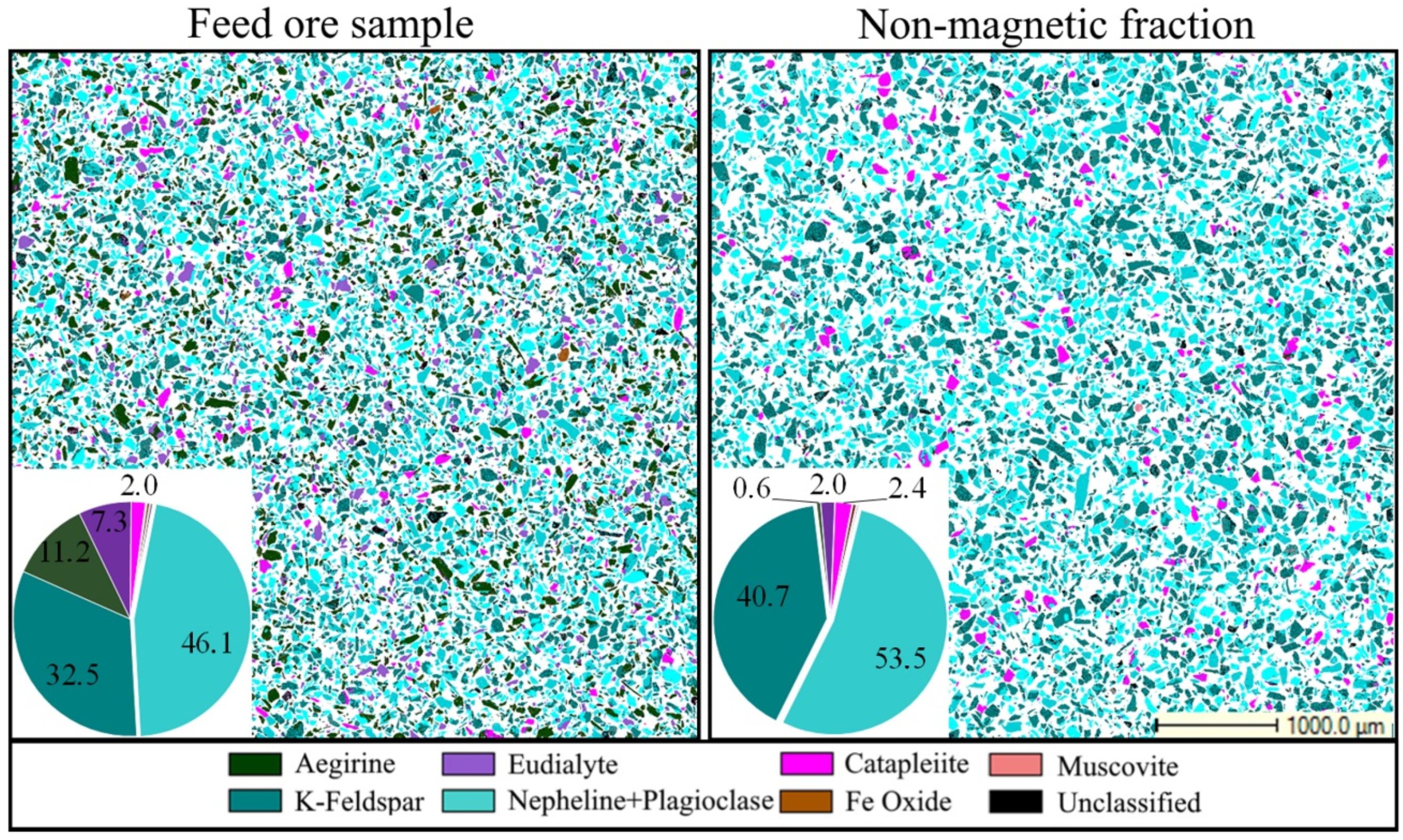
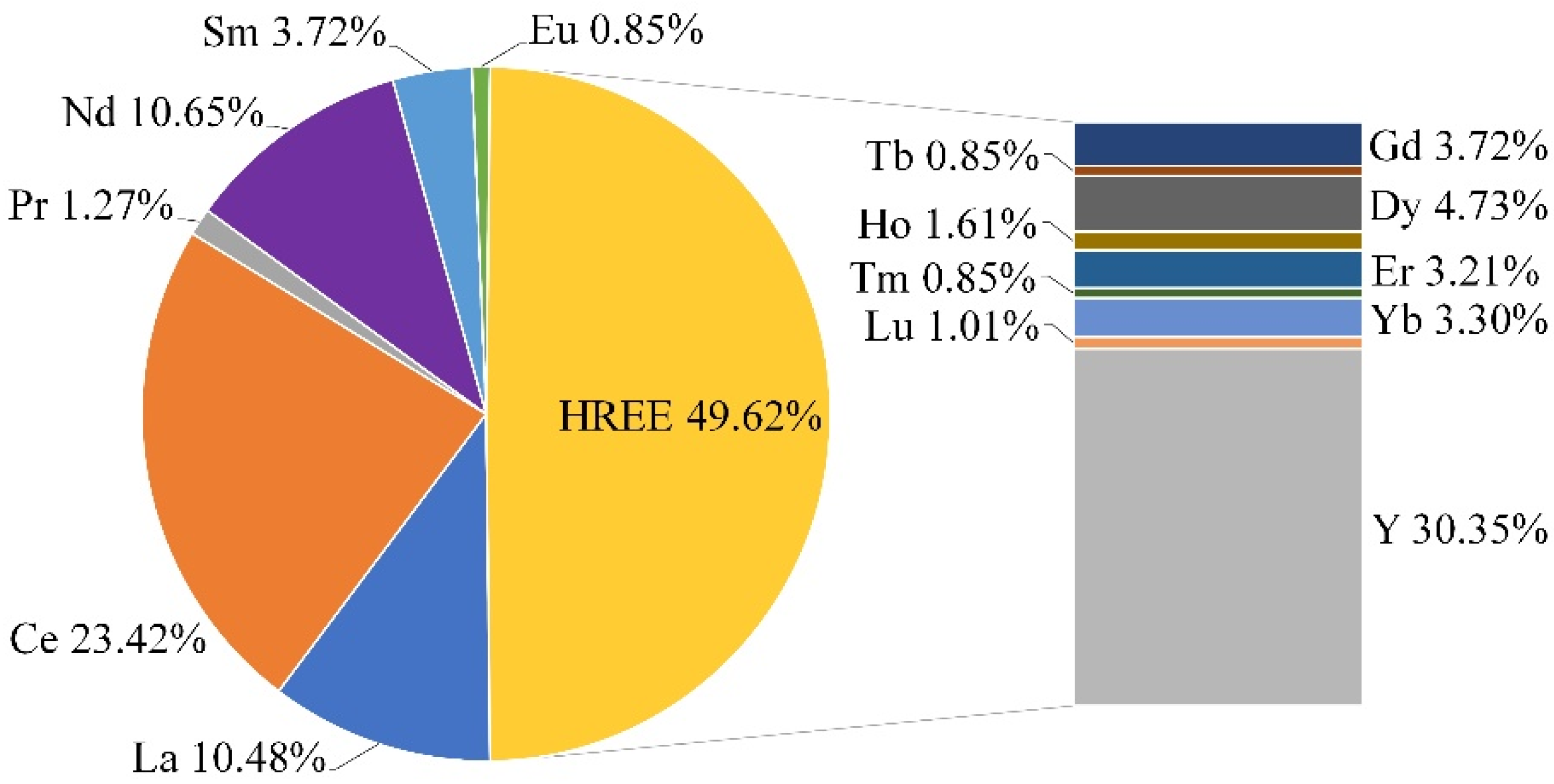
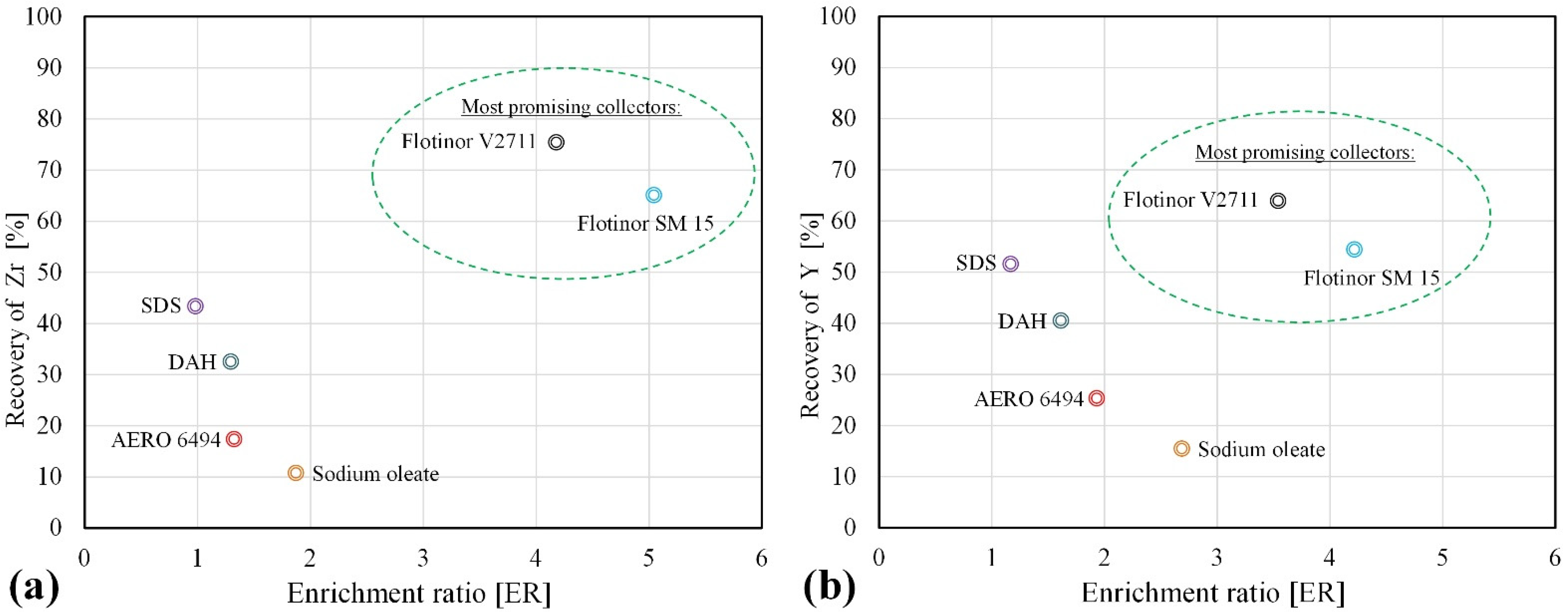
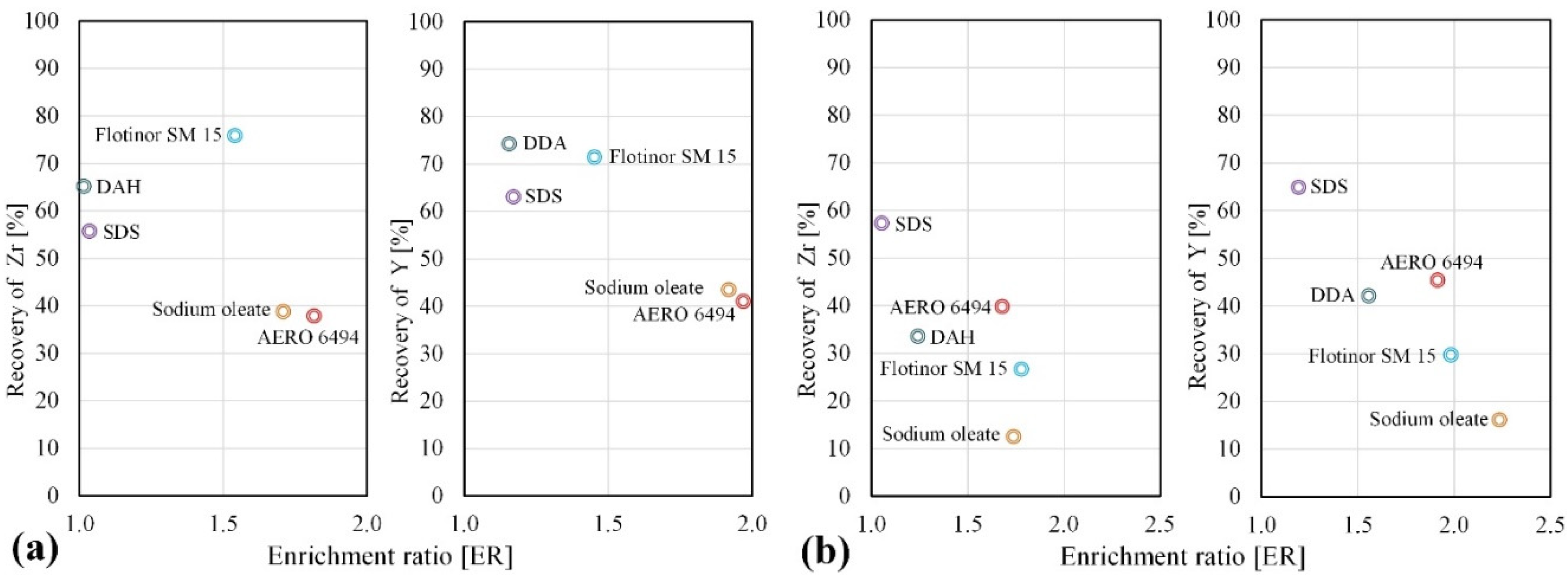
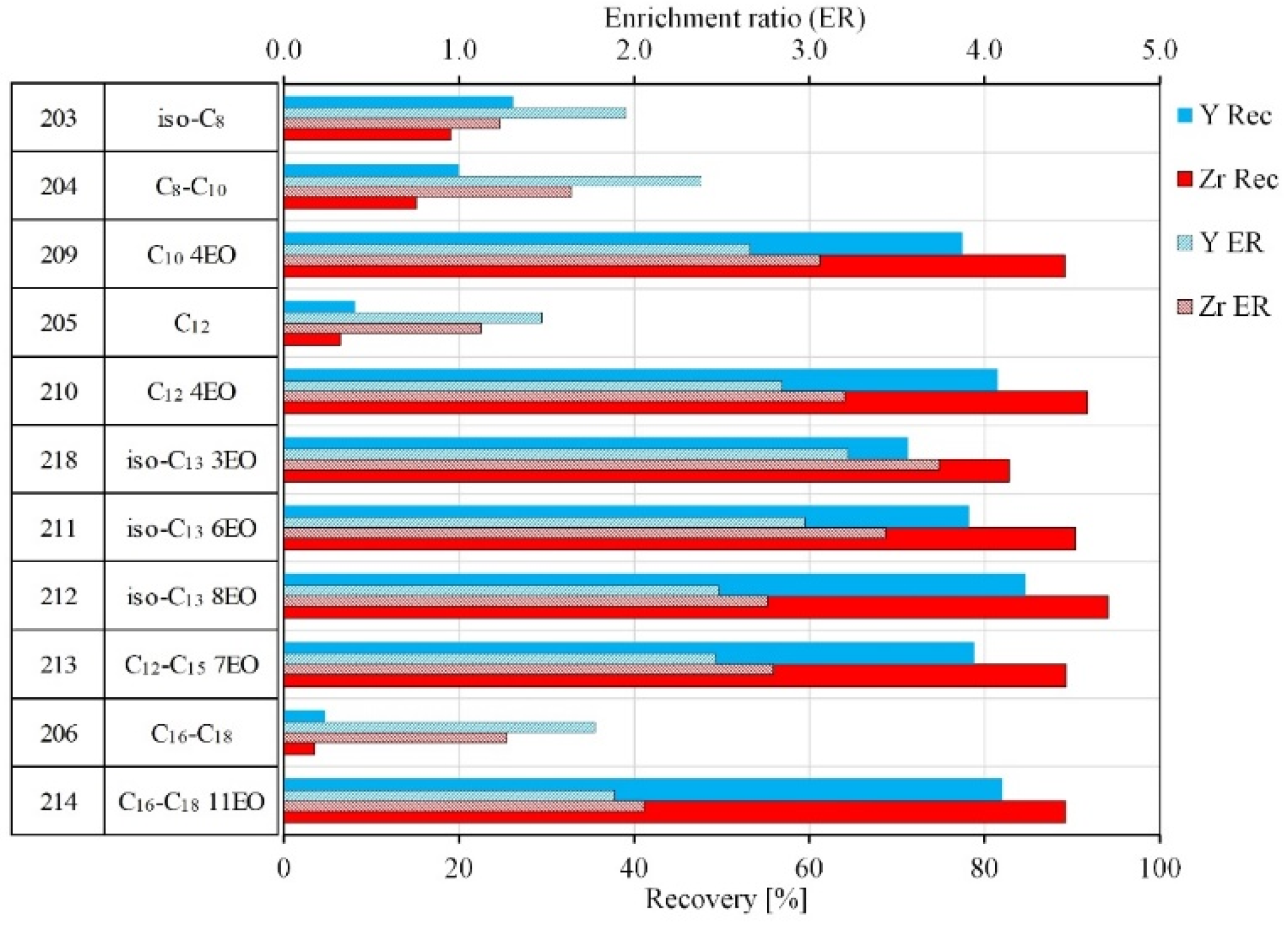
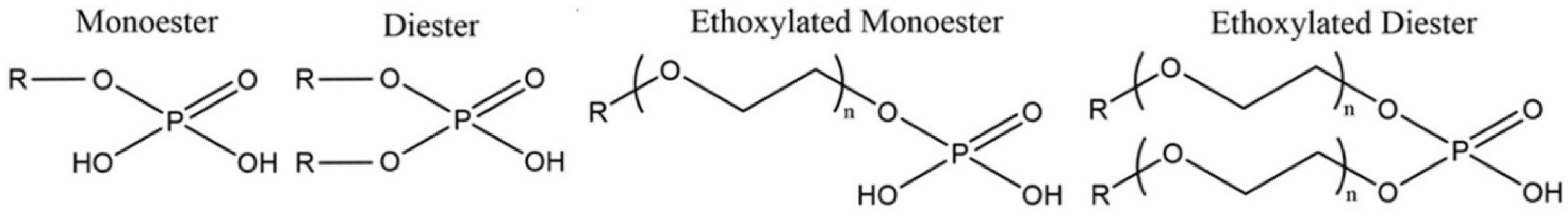
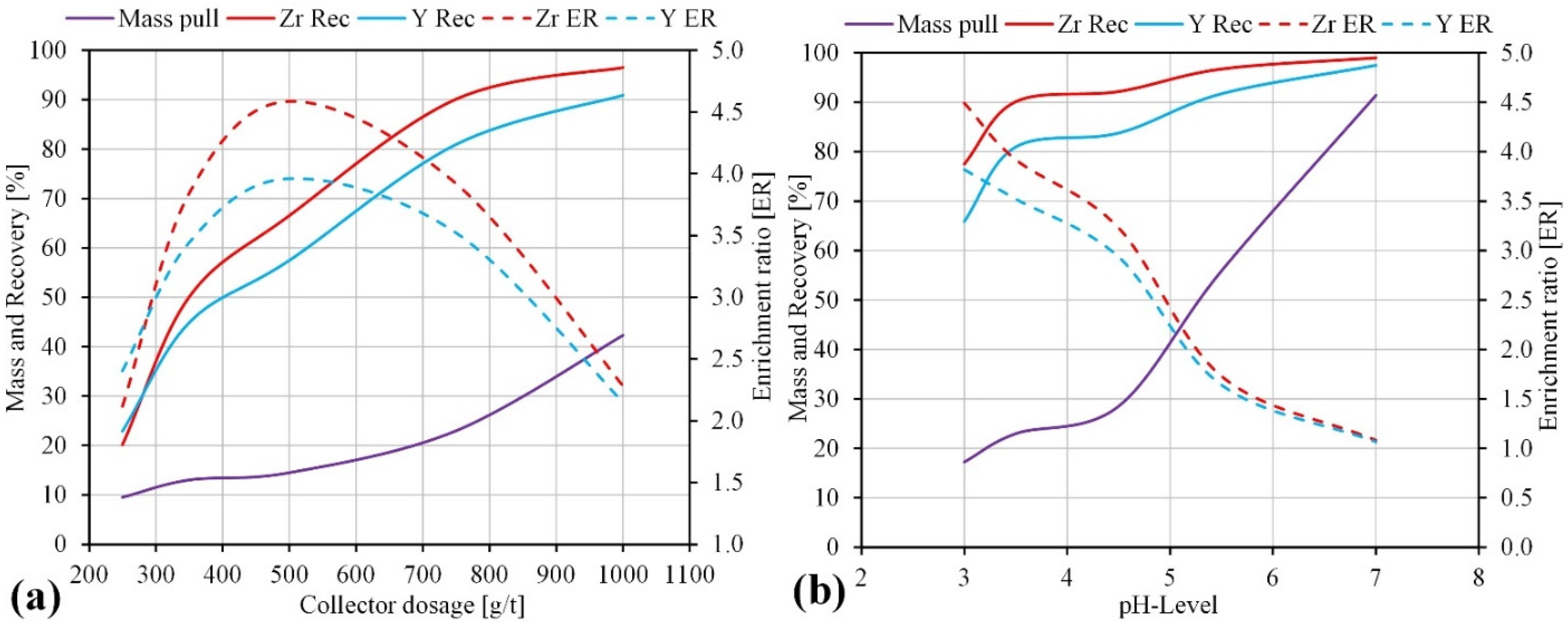

| Si | Na | Al | K | Zr | Fe | Ca | Hf | Nb | Th | U | REE | HREE | LREE |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 24.60 | 7.60 | 9.71 | 3.27 | 1.23 | 2.44 | 0.89 | 0.025 | 0.033 | 0.0013 | 0.001 | 0.403 | 0.203 | 0.200 |
| Trade Name | Chemical Composition | Supplier |
|---|---|---|
| Sodium dodecyl sulfate (SDS) | Sodium lauryl sulphate | Carl Roth GmbH |
| Sodium oleate | Oleic acid sodium salt | |
| Dodecylamine hydrochloride (DAH) | Dodecylamine hydrochloride | TCI Chemical Industry |
| AERO 6494 Promoter | Alkyl hydroxamate | Solvay |
| Flotinor SM 15; Flotinor V2711 | Mixture of mono- and di-phosphoric acid esters | Clariant International Ltd. |
| ILCO PHOS Series | ILCO Chemikalien GmbH |
| Products | Mass, % | Zr | REE | ||
|---|---|---|---|---|---|
| Content, % | Recovery, % | Content, % | Recovery, % | ||
| Magnetic fraction | 32.2 | 1.83 | 47.83 | 0.936 | 79.06 |
| Non-magnetic fraction | 67.8 | 0.95 | 52.17 | 0.118 | 20.94 |
| Feed | 100.0 | 1.23 | 100.00 | 0.382 | 100.00 |
| Si | Na | Al | K | Zr | Fe | Ca | Hf | Nb | Th | U | REE | HREE | LREE |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 25.2 | 7.22 | 11.20 | 3.87 | 0.95 | 0.22 | 0.66 | 0.019 | 0.010 | 0.0012 | 0.001 | 0.118 | 0.060 | 0.059 |
| Zr | Hf | Ti | Fe | Mn | Nb | Ta | Ca | Na | Th | U | REE | HREE | LREE |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 9.12 | 0.2 | 0.028 | 0.81 | 0.26 | 0.0746 | 0.0047 | 1.11 | 4.82 | 0.0005 | 0.0142 | 1.5154 | 0.734 | 0.7814 |
| SiO2 | Al2O3 | Na2O | K2O | CaO | Fe2O3(T) | TiO2 | MnO | ZrO2 | P2O5 | MgO | BaO | Other | LOI |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 56.575 | 22.9 | 10.28 | 5.175 | 0.425 | 0.17 | 0.0017 | 0.02 | 0.095 | 0.0055 | 0.1 | 0.01 | 1.253 | 2.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silin, I.; Gürsel, D.; Büchter, C.; Weitkämper, L.; Wotruba, H. Recovery of Catapleiite and Eudialyte from Non-Magnetic Fraction of Eudialyte ore Processing of Norra Kärr Deposit. Minerals 2022, 12, 19. https://doi.org/10.3390/min12010019
Silin I, Gürsel D, Büchter C, Weitkämper L, Wotruba H. Recovery of Catapleiite and Eudialyte from Non-Magnetic Fraction of Eudialyte ore Processing of Norra Kärr Deposit. Minerals. 2022; 12(1):19. https://doi.org/10.3390/min12010019
Chicago/Turabian StyleSilin, Ivan, Devrim Gürsel, Christian Büchter, Lars Weitkämper, and Hermann Wotruba. 2022. "Recovery of Catapleiite and Eudialyte from Non-Magnetic Fraction of Eudialyte ore Processing of Norra Kärr Deposit" Minerals 12, no. 1: 19. https://doi.org/10.3390/min12010019
APA StyleSilin, I., Gürsel, D., Büchter, C., Weitkämper, L., & Wotruba, H. (2022). Recovery of Catapleiite and Eudialyte from Non-Magnetic Fraction of Eudialyte ore Processing of Norra Kärr Deposit. Minerals, 12(1), 19. https://doi.org/10.3390/min12010019






